Abstract
The objective of this study was to investigate the aroma profile and major flavor compounds in Xiaoqu liquor by a combination of sensory profiling, GC-O/GC-MS analysis, and SBSE-GC-MS. A total of 57 important volatile compounds (FD ≥ 2) were screened based on the retention indices, aromatic characteristics, standard comparison, and aroma extract dilution analysis (AEDA). By calculating the odor activity values (OAVs), 32 aroma active compounds were identified in Xiaoqu liquor. In the aroma recombination experiments, these active compounds were dissolved in 46% ethanol water at their natural concentrations, resulting in successful simulations of the typical aromas of Xiaoqu liquor, including fruity aroma, sweet aroma, and grain aroma. Omission experiments showed that the critical compounds that contribute to the characteristic flavor of Xiaoqu liquor include ethyl octanoate, acetaldehyde, 1,1-diethoxyethane, isoamyl acetate, ethyl acetate, valeric acid, 1-octen-3-ol, and ethyl isovalerate. The results provide some guidance for upgrading the fermentation and microbial strain in the production of Xiaoqu liquor.
1. Introduction
Based on the aroma, Chinese liquor can be classified into fen-flavor, strong-flavor, Maotai-flavor, rice-flavor and other flavor types, which have highly different stylistic features in their aromas [1]. Fen-flavor liquor is the earliest liquor in China and is honored as the origin of all liquors. It is pure, sweet, mild, and natural with a fresh aftertaste [2]. According to the distiller’s yeast used in the production process, fen-flavor liquor can be further classified into Daqu, Fuqu, and Xiaoqu types. Xiaoqu fen-flavor liquor is mainly produced in Hubei, Chongqing, and Yunnan, China. Owing to its sweet, fresh, mild, natural, and pure tastes, it is very popular among consumers in the South Central China. The unique flavor characteristics are attributed to the production techniques of saccharification before fermentation, steaming and distillation, as well as management of vinasse and raw materials [3,4].
In recent years, gas chromatography olfactometry (GC-O), solid-phase microextraction (SPME), and gas chromatography/mass spectrometry (GC/MS) have been widely applied to the research on the aroma compounds in liquor [5], and Chinese researchers have conducted a number of in-depth studies of the strong-flavor, Daqu fen-flavor, and Maotai-flavor liquor. For example, Wang et al. have investigated the major components in Maotai-flavor liquor by liquid-liquid extraction and GC/MS and carried out quantification analysis of 48 aroma compounds; finally, 29 compounds were identified as the skeleton components, and the results were validated by aroma recombination and omission experiments [6]. Fan et al. conducted a quantitative analysis of 66 flavor compounds in soft-style liquor with Head Space Solid Phase Microextraction (HS SPME) and identified the key flavor compounds such as ethyl hexanoate and 2-methyl ethyl butyrate by calculating the odor activity values (OAVs) [7]. These studies have greatly expanded the understanding of the chemical composition of Chinese liquor. Up to date, about 700 micro components have been detected in Daqu fen-flavor liquor; however, there have been few reports about the flavor compounds in Xiaoqu liquor. The flavor of Xiaoqu liquor not only greatly determines the taste and quality of the liquor itself but also plays a feedback regulatory role in the production techniques and microbial system. Therefore, it is highly necessary to further dissect the flavor of Xiaoqu liquor and the related compounds.
This study aims to identify the volatile compounds by GC-O, stir bar sorptive extraction (SBSE), and GC-MS in Xiaoqu liquor based on the sensory quantitative description. Besides, the key aroma compounds in Xiaoqu liquor were identified by aroma extract dilution analysis (AEDA) and OAV determination. Finally, the results were validated by aroma recombination and omission experiments, and the flavor compounds in Xiaoqu liquor were fully dissected, which may provide important reference and support for improving the production techniques of Xiaoqu liquor.
2. Materials and Methods
2.1. Materials and Reagents
The Xiaoqu liquor was produced by the Jing Brand Co. Ltd. (Daye, Hubei, China), with sorghum as the raw material and green-covering Guanyin Tuqu as the distiller’s yeast. The production process involved fermentation at 33 °C for 15 d in the stainless-steel groove, brewing under pressure, and condensation distillation, with the volume fraction of ethanol in the distillation products being controlled at nearly 55%. The sample was designated as JPXQL.
1,1-Diethyloxyethane, n-propanol, isobutanol, n-butanol, isopentanol, pentanol, acetic acid, propionic acid, butyric acid, valeric acid, caproic acid, heptanic acid, octoic acid, isovaleric acid, ethyl butyrate, ethyl isovalerate, isoamyl acetate, ethyl valerate, ethyl caproate, ethyl heptanoate, ethyl octanoate, ethyl nonanoate, ethyl decanoate, ethyl phenylacetate, ethyl dodecanoate, ethyl phenylpropionate, hexanol, furfural, benzaldehyde, phenylacetaldehyde, and phenylethanol were purchased from SAFC99 (chromatographic purity > 98%). Ethyl 2-hydroxyhexanoate, 5-methylfurfural, naphthalene, 4-methylguaiacol, 4-Ethylguaiacol, phenol, 2-Nonanone, 1-octene-3-ol, 1-nonanol, diethyl succinate, hexylacetate, ethyl undecanoate, γ-nononolactone, tetramethylpyrazine, 2-ethylhexanol, tert-pentanol, amyl acetate, and n-alkanes (C7–C30) were purchased from Sigma-Aldrich (chromatographic purity > 97%, Shanghai, China). Diethyl azelate, ethyl palmitate, isoamyl butyrate, styreneT, and phenethyl acetate were obtained from CNW (purity > 98%). Ethyl formate was purchased from Dr. Ehrenstorfer Gmbh (chromatographic purity > 90%). Ethyl acetate and ethyl lactate were purchased from the Chinese Medicine Wokai (chromatographic purity > 99%, Shanghai, China). Methanol (purity > 40%) and diethyl pimelate (purity > 97%) were purchased from XiYa Reagent (Shandong, China). Ethyl benzoate was purchased from Toronto Research Chemicals (TRC), Canada (Toronto, ON, Canada). Diethyl succinate was purchased from Aladdin Bio-Chem Technology Co., LTD. (purity > 98%, Shanghai, China). Ethyl oleate and ethyl linoleate were purchased from ANPEL (purity > 95%, Shanghai, China).
All the standards used in the study were chromatographically pure. Sodium hydroxide, sulphuric acid, sodium bicarbonate, and sodium chloride were of analytical purity. Chromatographically pure anhydrous ethanol, ether, pentane, and acetonitrile were obtained from the Beijing Mreda Technology Co., Ltd. (Beijing, China). Phosphoric acid and potassium dihydrogen phosphate of analytical purity were purchased from China Pharmaceutical (Shanghai Chemical Reagent Co. Ltd., Shanghai, China). The water used was ultrapure water.
2.2. Instruments
The instruments employed included electronic AB135-S analytical balance (METTLER-TOLEDO, Shanghai, China), Agilent 7890B-5977C Gas Chromatograph-mass Spectrometer (Agilent Technologies Inc., Palo Alto, CA, USA), DB-FFAP Capillary-column Chromatography (60 m × 0.25 mm, 0.25 μm), ODP2 Gas Chromatography Olfactometry, MPS2 Three-in-one Automatic Sample, Twister for SBSE (0.5 mm in film thickness, 10 mm in length; PDMS) (German GERSTEL, Demilheim, Germany); and DVB/CAR/PDMS Three-phase Solid Phase microextraction (1 cm 50/30 µm, American SUPELCO, Bellefonte, PA, USA).
2.3. Methods
2.3.1. Sensory Evaluation of Xiaoqu Liquor
The sensory evaluation of Xiaoqu liquor was performed according to previous studies [8,9] and EN ISO 8589 “Sensory Analysis General Guidance for the Design of Test Rooms”. The sensory evaluation panel was composed by 10 professional liquor tasters (among which five are national level liquor tasters) selected from Jing Brand Co. Ltd. A series of standard solutions of different aromas at different intensities were prepared. The panelists were then invited to examine the aroma one by one. The intensity (quality or time of duration) was evaluated by the panel or individual panelists with a five-grade marking system, and the description methods are presented in Table 1. Subsequently, the descriptors of the sensory characteristics of Xiaoqu liquor were sorted out, including eight descriptors: fruity, floral, grain, sweet, fermented grain, chaff, sour, and green. The aroma of the liquor samples was described by the panelists after 90 s of olfactory evaluation in a professional liquor tasting room, and the interval between two rounds was 30 min. The panelists would make evaluation of the aroma intensity based on the screened aroma indices, and the average values were taken to plot the aroma profiles.

Table 1.
Sensory quantitative description of Xiaoqu liquor.
2.3.2. SBSE Conditions
The stir bar of SBSE was allowed to age for 60 min at the injection port before use. Two milliliters of sample was transferred into a 20-mL headspace bottle. Then, 8 mL ultrapure water was added, and the stir bar was placed into the bottle, followed by heating and stirring for 1 h (1100 rpm, 55 °C). After that, the stir bar was taken out and designated as stir bar 1. Then, 1.5 g NaCl was added into the sample, and another stir bar was placed in the bottle. The above process was repeated to obtain stir bar 2. The two stir bars were then transferred to a thermal desorption device for desorption (250 °C, 15 min) and further GC-MS analysis.
2.3.3. GC-MS Analysis
The gas chromatography was conducted with the following conditions: injection port temperature, 250 °C; carrier gas, helium (purity ≥ 99.999%); constant flow at the rate of 1.42 mL/min; column temperature programming: initial temperature 40 °C, held for 5 min, increased to 250 °C at a rate of 3.5 °C/min, and held for 10 min. The mass spectral analysis was performed with the following conditions: ion source, electron bombardment ion source (EI); energy, 70 eV; temperature, 230 °C; MS Quad temperature, 150 °C; MS transfer line temperature, 280 °C; solvent delay, 0 min; full ion scanning in the range of 20–500 amu.
2.3.4. GC-MS-O Analysis
The gas chromatography and mass spectral analysis were conducted under the same conditions as the above section. The parameters for the olfactometric device were set as: air flow rate, 50 mL/min; sniff port temperature 200 °C; the effluents flowed into the olfactometric device at the capillary column with a split ratio of 1:1. The aroma compounds in the Xiaoqu liquor were analyzed by the aroma extract dilution analysis (AEDA) method. The samples were serially diluted with diethyl ether at the ratio of 1:2; the GC-O aroma analysis was conducted by three trained panelists; and the aroma characteristics were recorded. Flavor dilution factor (FD factor) was used to indicate the aroma intensity [10]. Usually, the compounds with FD ≥ 2 were considered as aroma active compounds.
2.3.5. Aroma Recombination and Omission Experiments
The aroma recombination and omission experiments were performed according to the previously reported methods [11]. The recombination medium was prepared by dissolving 2 g/L lactic acid in 46% ethanol by volume, and the compounds with OAVs ≥ 1 were all dissolved in the medium at their natural concentrations, which were then equilibrated for 10 min under ambient temperature and used as the aroma recombination solution. For the omission experiments, all the compounds with OAVs between 1 and 10 were dissolved in the medium at their natural concentrations, and those with OAVs ≥ 10 were omitted. The solutions were equilibrated under ambient temperature for 10 min to obtain the solution for omission experiments. Then, 20 mL of recombination solution and omission solution was respectively taken and placed into glasses specialized for tasting that were covered with aluminum foil. At the same time, a small glass of original Xiaoqu liquor was prepared. All the samples were randomly numbered for sensory evaluation. The descriptive evaluation was performed in the same way as in Section 2.3.2.
2.4. Data Processing
2.4.1. Qualitative Method
The mass spectra of each volatile compound were compared with those in the NIST11.L Database (Agilent Technologies Inc.), and the compounds with positive and negative matching scores higher than 800 were screened. The retention indices of each compound were calculated, and a comprehensive qualitative analysis was carried out based on the retention indices reported in previous literature. Finally, an accurate qualitative analysis was performed by comparison with the standards.
2.4.2. Quantitative Method
Standard solutions were prepared, and 2-ethyl hexanol was used as the internal standard. GC-MS-SBSE was used, and quantification ion was chosen to plot the standard curve by the five-spot method.
2.4.3. Odor Active Value (OAV) Calculation
OAV was defined as the ratio of the concentration (C) of the odor compound to the threshold value (T), namely OAV = C/T. An OAV < 1 indicates that the compound has little contribution to the overall aroma of the sample, while an OAV ≥ 1 indicates that the compound has significant contribution to the overall aroma, which was identified as a critical aroma compound [12]. The higher the OVAs were, the more significant the contribution of the compound would be.
3. Results and Discussion
3.1. Qualitative Analysis of Xiaoqu Liquor
Aroma compounds are very important characteristics of Chinese liquor or Xiaoqu liquor. A total of 80 aroma compounds were identified from Xiaoqu liquor by GC-MS-O (Figure 1). AEDA proposed by Matsui et al. [13] is an effective approach to screen the critical aroma compounds. The aroma compounds extracted from the sample were diluted sequentially with dichloromethane at a 1:1 ratio, and each dilution was analyzed once by each panelist until the aroma could not be detected. The fold of dilution was then regarded as the flavor dilution (FD) factor of the compound. Generally, a high value of FD indicates that the compound has a greater contribution to the overall flavor of the sample [14]. As shown in Table 2, a total of 13 compounds with FD ≥ 8 were detected in Xiaoqu liquor, including ethyl octanoate, acetaldehyde, 1,1-diethoxyethane, isoamyl acetate, ethyl hexanoate, dimethyl trisulfide, 1-octen-3-ol, valeric acid, ethyl acetate, ethyl isovalerate, isobutanol, ethyl caprate, and ethyl laurate. Among these compounds, ethyl octanoate had the highest FD value of 256 and correspondingly the greatest contribution to the aroma, which is consistent with its contribution to the aroma of Daqu liquor [11]. Besides, compared with in Daqu liquor, acetaldehyde, esoamyl acetate, and ethyl laurate have great contributions to the aroma of Xiaoqu liquor, among which acetaldehyde contributes to the fruity and grass aroma (FD = 32), isoamyl acetate mainly contributes to the banana aroma (FD = 32), and ethyl laurate contributes to the sweet and fruity aroma (FD = 8). The identification of the 13 aroma compounds such as ethyl octanoate provides a basis for quantifying the contributions of the key aroma compounds in Xiaoqu liquor.
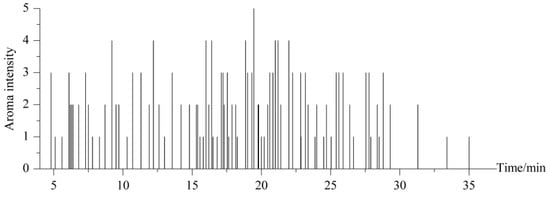
Figure 1.
Aroma intensity of JPXQL by GC-MS-O.

Table 2.
Volatile compounds in JPXQL determined by GC-O.
3.2. GC-FID and SBSE-GC-MS Analysis
By GC-O analysis, a total of 57 compounds were identified to contribute to the aroma of Xiaoqu liquor, and their contributions were quantified by the FD values. However, the extraction and condensation process might have caused some loss of the aroma compounds, which might affect the evaluation results of the aroma [15]. Hence, it is necessary to conduct quantitative analysis of the 57 compounds and calculate their OAVs, so as to more accurately demonstrate the contribution of each compound to the overall aroma of Xiaoqu liquor. To this end, GC-FID combined with SBSE-GC-MS was used for the quantitative analysis, which has been validated to be an effective method in previous studies. The standard curve and the contents of compounds are shown in Table 3. In Xiaoqu liquor, the content of ethyl octanoate (5502.82 μg/L) was significantly higher than that in Daqu liquor (2163.91 μg/L), but the contents of ethyl lactate (442,370 μg/L) and ethyl acetate (537,920 μg/L) were lower [11]. Besides, higher contents of acetaldehyde (151,980 μg/L), isoamyl acetate (7099.8 μg/L), and ethyl laurate (2827.21 μg/L) were found in Xiaoqu liquor. It can be speculated that different contents of such important aroma compounds contribute to the unique flavor of Xiaoqu liquor.

Table 3.
Quantitative results of volatile compounds in JPXQL.
3.3. Analysis of Aroma Active Compounds in Xiaoqu Liquor
Both AEDA and OAV can be used to effectively evaluate the aroma of flavor substances in a single matrix. However, neither of the two methods considers the possible interactions among different flavor substances in the same matrix, such as the enhancement and inhibition effects that have been confirmed by previous studies. The compounds with higher OAVs are usually considered to have more significant contributions to the overall aroma of the sample, and those with OAVs above 1 are generally regarded as aroma active compounds. As shown in Table 4, by calculating and comparing the OVAs of the 57 compounds, a total of 32 compounds were found to have OAVs above 1 in Xiaoqu liquor, and the number is smaller than that in Daqu liquor (50 compounds) [11]. The top 10 compounds in OVAs were ethyl octanoate, acetaldehyde, isoamyl acetate, 1,1-diethoxyethane, ethyl hexanoate, valeric acid, dimethyl trisulfide, 1-octen-3-ol, ethyl acetate, and isobutanol. All together, there were 13 ester compounds, which is in agreement with the fact that ester aroma is dominant in the overall aroma of Chinese liquor [16]. It has been reported that ester compounds have important influence on the sensory characteristics such as the dominant aroma and flavor of Chinese liquor and contribute to the fruity and floral aroma of fen-flavor liquor [17]. In Xiaoqu liquor, the contents of ethyl acetate and ethyl lactate were the highest, and some other ester compounds in lower contents had great contributions to the overall flavor due to their lower odor threshold values and higher OAVs, such as ethyl octanoate (sweet aroma like brandy), isoamyl acetate (representative of sweet aroma in fruit taste), ethyl hexanoate (sweet aroma, fruity aroma, and cellar aroma), ethyl valerate, ethyl isovalerate, and ethyl butyrate [18,19].

Table 4.
Odor activity values (OAVs) of the key flavor compounds in JPXQL.
The aldehyde compounds with great contributions to the aroma of Xiaoqu liquor were acetaldehyde and acetal. Acetaldehyde has a spicy odor and can contribute to fruity, coffee, wine, and grass aromas in a concentration dependent manner. Acetal has a fruity aroma and can promote the soft sensory feeling and aged aroma of the liquor [20]. Among the alcohol compounds, besides ethanol, other compounds with relatively higher OAVs included isobutanol, n-propanol, and 1-octen-3-ol, among which isobutanol and n-propanol have a slight wine aroma, while 1-octen-3-ol is the representative of mushroom aroma. Alcohol compounds can help to make the liquor mellow and thick and are the precursors for the production of ester compounds as well [21].
Acetic acid and lactic acid were the acids with the highest content in Xiaoqu liquor; however, they had little contribution to the flavor due to their relatively higher threshold values. The acid compounds that have certain influence on the aroma of Xiaoqu liquor included valeric acid and butyric acid, which have water odor and putrefactive odor and thus exert some negative effects on the flavor. The compounds that contribute to the aroma of Xiaoqu liquor mainly include acetaldehyde, ethyl phenylpropionate, and phenethyl acetate, which confer the liquor with elegant floral aroma and sweet aroma.
3.4. Aroma Recombination and Omission Experiments
Aroma recombination and omission experiments are general methods to validate the critical aroma compounds [11]. On a quantitative basis, various aroma compounds at different concentrations are added into certain medium to produce an overall aroma [22,23]. In addition, sensory analysis has also been used to study the relationship between flavor substances in the liquor and the liquor flavor. The 32 compounds with OAVs ≥ 1 were recombined, and the aroma was compared with that of the base liquor. Figure 2 shows that the recombination solution had rich aroma, particularly fruity aroma, grain aroma, and distiller’s grain aroma. A comparison of the aroma properties in the original liquor and recombination sample showed that they had highly similar aroma profiles with only very slight differences. These slight differences could be due to that the recombination sample did not undergo the aging process of the original liquor, and the reactions among compounds did not fully occur because of the short time of establishment, or the medium for the recombination did not contain some non-volatile compounds that interact with the aroma compounds. Although there were slight differences in aroma properties between the two samples, the statistical results showed that there were no significant differences in the eight properties analyzed in the aroma profiling, indicating that the simulation system of Xiaoqu liquor was successfully established. These results also imply the high accuracy and reliability of the aroma compound extraction method, AEDA combined with GC-O, quantitative analysis of complementary aroma active compounds, and the method for identifying the key aroma components based on OAV calculation used in this study.
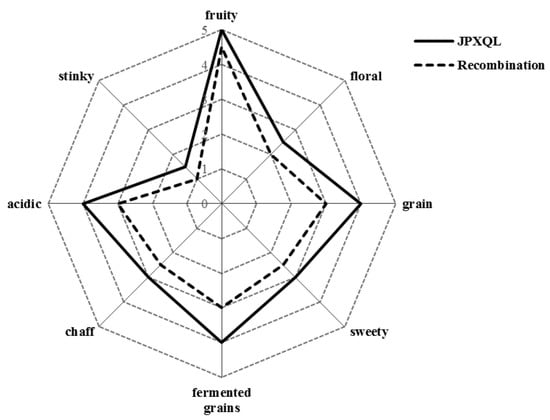
Figure 2.
Profile of aroma recombination of JPXQL.
Table 5 shows the results of the omission experiments. Among the 11 scenarios of omission experiments, six scenarios of omission would lead to significant differences in aroma profiles (p ≤ 0.05). The omission of all the ester compounds resulted in extremely significant differences in aroma profile, indicating the dominant role of ester compounds in the flavor of Xiaoqu liquor. Besides, omission of ethyl octanoate showed that this compound has great contribution to the aroma of Xiaoqu liquor. When ethyl acetate and ethyl lactate were omitted at the same time, there were obvious changes in the aroma profile, implying that these two compounds contribute greatly to the aroma of the liquor. Furthermore, when these two compounds were respectively omitted alone, the omission of ethyl acetate would lead to significant differences, while that of ethyl lactate would not, indicating that ethyl acetate has a greater contribution to the overall aroma than ethyl lactate. Although the content of ethyl lactate is high in Xiaoqu liquor, it may only affect the mouthfeel instead of the aroma of the liquor [24].

Table 5.
Omission experiments of the aroma compounds from JPXQL.
The omission of acetic acid and valeric acid led to extremely significant differences in aroma profile, indicating that these two acids contribute greatly to the sour taste in the overall aroma of Xiaoqu liquor. The omission of 1-octen-3-ol and all alcohol compounds did not lead to obvious changes in aroma profile, possibly due to the presence of a large amount of ethanol in the liquor, which is of very high volatility and a low threshold value and thus affects the judgment on the alcohol compounds.
4. Conclusions
Here, we took Xiaoqu liquor as the subject and employed SBSE-GC-MS to extract as well as qualitatively and quantitatively analyze the volatile compounds in Xiaoqu liquor. Besides, the key aroma compounds in Xiaoqu liquor were identified by GC-MS-O combined with AEDA and OAV, which were then evaluated and validated through aroma recombination and omission experiments. A total of 32 key aroma compounds were identified from Xiaoqu liquor, among which ethyl octanoate was found to be the most important aroma compounds with a FD value of 256, followed by 12 compounds such as acetaldehyde, 1,1-diethoxyethane, isoamyl acetate, and ethyl hexanoate. It was also found that Xiaoqu liquor has high contents of ethyl octanoate, acetaldehyde, isoamyl acetate, and ethyl laurate. The aroma recombination and omission experiments of the 32 compounds showed that the recombination sample had very similar sensory characteristics to the original liquor, and some compounds such as ethyl octanoate, ethyl lactate, acetic acid, and valeric acid play important roles in the flavor of Xiaoqu liquor.
Overall, Xiaoqu liquor has a smaller number of key aroma compounds than Daqu liquor, as well as some unique key aroma compounds such as 1-octene-3-ol and 1,1-diethoxyethane, and these aroma compounds have different contributions to the aroma compared with those of Daqu liquor. These factors together contribute to the unique aroma characteristics of Xiaoqu liquor. Our findings provide a full characterization of the flavor of Xiaoqu liquor and lay a foundation for further dissecting the mechanism that underlies its unique flavor.
Author Contributions
H.Y. conceived and designed the experiments; Z.W. and X.S. performed the experiments and analyzed the data; Z.W. and X.S. wrote the paper; Y.L. supervised the experiments. All authors have read and agreed to the published version of the manuscript.
Funding
We gratefully acknowledge financial support from Jing Brand Research Institute, Jing Brand Co., Ltd.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Yu, M.; Yongguang, H. Volatile components and aroma characteristics of Maotai Fen—Flavored liquor. Food Sci. 2019, 40, 241–248. [Google Scholar]
- Mingxia, Z.; Xuna, Z.; Tianyou, Y.; Zhengyang, H. Optimization of Headspace Solid Phase Microextraction Conditions for GC-MS Analysis of Aromatic Constituents in White Liquor. Food Sci. 2011, 32, 49–53. [Google Scholar] [CrossRef]
- Bin, C.; Hongkui, H.; Guoying, L.; Fengjie, Z.; Anjun, L.; Xinglin, H. The influence of the Daqu, Xiaoqu and the fermentation way on the flavor of Fen-flavor Baijiu (Chinese liquor). Food Ferment. Ind. 2018, 8, 166–171. [Google Scholar] [CrossRef]
- Kong, Y.; Wu, Q.; Zhang, Y.; Xu, Y. In Situ Analysis of Metabolic Characteristics Reveals the Key Yeast in the Spontaneous and Solid-State Fermentation Process of Chinese Light-Style Liquor. Appl. Environ. Microbiol. 2014, 80, 3667–3676. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Fan, S.; Yan, Y.; Yang, L.; Chen, S.; Xu, Y. Characterization of Potent Odorants Causing a Pickle-like Off-Odor in Moutai-Aroma Type Baijiu by Comparative Aroma Extract Dilution Analysis, Quantitative Measurements, Aroma Addition, and Omission Studies. J. Agric. Food Chem. 2020, 68, 1666–1677. [Google Scholar] [CrossRef]
- Lingling, W.; Wenlai, F.; Yan, X. Analysis of capillary chromatographic skeleton compounds in Chinese soy sauce aroma type liquor by liquid-liquid microextraction and aroma recombination. Sci. Technol. Food Ind. 2012, 33, 304–308. [Google Scholar] [CrossRef]
- Wenlai, F.; Qingqing, N.; Yan, X. Key aroma compounds of Yanghe Supple and Mellow Aroma Style Liquors. Food Sci. 2013, 34, 135–139. [Google Scholar]
- Nguyen, H.; Campi, E.M.; Jackson, W.; Patti, A.F. Effect of oxidative deterioration on flavour and aroma components of lemon oil. Food Chem. 2009, 112, 388–393. [Google Scholar] [CrossRef]
- Siru, J.; Ting, Q.; Xichang, W.; Qiyan, S. Identification of the key odorant compounds in the gonad of steamed Chinese mitten crab. Mod. Food Sci. Technol. 2019, 2, 323–329. [Google Scholar] [CrossRef]
- Grosch, W. Evaluation of the key odorants of foods by dilution experiments, aroma models and omission. Chem. Senses 2001, 26, 533–545. [Google Scholar] [CrossRef]
- Gao, W.; Fan, W.; Xu, Y. Characterization of the Key Odorants in Light Aroma Type Chinese Liquor by Gas Chromatography–Olfactometry, Quantitative Measurements, Aroma Recombination, and Omission Studies. J. Agric. Food Chem. 2014, 62, 5796–5804. [Google Scholar] [CrossRef] [PubMed]
- Wenlai, F.; Qian, M.C. Headspace solid phase microextraction and gas chromatography-olfactometry dilution analysis of young and aged Chinese “Yanghe Daqu” liquors. J. Agric. Food Chem. 2005, 53, 7931–7938. [Google Scholar]
- Matsui, T.; Guth, H.; Grosch, W. A comparative study of potent odorants in peanut, hazelnut, and pumpkin seed oils on the basis of aroma extract dilution analysis (AEDA) and gas chromatography olfactometry of headspace samples (GCOH). Fett Lipid 1998, 100, 51–56. [Google Scholar] [CrossRef]
- Zheng, Y.; Sun, B.; Zhao, M.; Zheng, F.; Huang, M.; Sun, J.; Sun, X.; Li, H. Characterization of the Key Odorants in Chinese Zhima Aroma-Type Baijiu by Gas Chromatography–Olfactometry, Quantitative Measurements, Aroma Recombination, and Omission Studies. J. Agric. Food Chem. 2016, 64, 5367–5374. [Google Scholar] [CrossRef] [PubMed]
- Guth, H. Quantitation and sensory studies of character impact odorants of different white wine varieties. J. Agric. Food Chem. 1997, 45, 3027–3032. [Google Scholar] [CrossRef]
- Qian, Z.; Qinya, L.; Minquan, H.; Jihong, W.; Jinyuan, S.; Xiaotao, S.; Fuping, Z.; Baoguo, S. Analysis of odor-active compounds in 2 sesame-flavor Chinese Baijiu. Food Sci. 2019, 40, 214–222. [Google Scholar]
- Zhao, D.; Shi, D.; Sun, J.; Li, A.; Sun, B.; Zhao, M.; Chen, F.; Sun, X.; Li, H.; Huang, M.; et al. Characterization of key aroma compounds in Gujinggong Chinese Baijiu by gas chromatography–olfactometry, quantitative measurements, and sensory evaluation. Food Res. Int. 2018, 105, 616–627. [Google Scholar] [CrossRef]
- Noguerol-Pato, R.; González-Álvarez, M.; González-Barreiro, C.; Cancho-Grande, B.; Simal-Gandara, J. Evolution of the aromatic profile in Garnacha Tintorera grapes during raisining and comparison with that of the naturally sweet wine obtained. Food Chem. 2013, 139, 1052–1061. [Google Scholar] [CrossRef]
- Zhu, S.; Lü, X.; Ji, K.; Guo, K.; Li, Y.; Wu, C.; Xua, G. Characterization of flavor compounds in Chinese liquor Moutai by comprehensive two-dimensional gas chromatography/time-of-flight mass spectrometry. Anal. Chim. Acta 2007, 597, 340–348. [Google Scholar] [CrossRef]
- Jinyuan, S.; Lili, G.; Guoying, L.; Hehe, L.; Xiaotao, S.; Mingquan, H.; Fuping, Z.; Baoguo, S. Analysis of volatile compounds in fermented grains of Chinese Gujinggong liquor by solvent-assisted flavor evaporation combined with GC-MS and GC-O. Food Sci. 2016, 37, 87–93. [Google Scholar]
- Zhang, W.-X.; Wu, Z.-Y.; Zhang, Q.-S.; Wang, R.; Li, H. Combination of newly developed high quality Fuqu with traditional Daqu for Luzhou-flavor liquor brewing. World J. Microbiol. Biotechnol. 2009, 25, 1721–1726. [Google Scholar] [CrossRef]
- Langos, D.; Granvogl, M.; Schieberle, P. Characterization of the Key Aroma Compounds in Two Bavarian Wheat Beers by Means of the Sensomics Approach. J. Agric. Food Chem. 2013, 61, 11303–11311. [Google Scholar] [CrossRef] [PubMed]
- Marcq, P.; Schieberle, P. Characterization of the Key aroma compounds in a Commercial Amontillado Sherry Wine by Means of the Sensomics Approach. J. Agric. Food Chem. 2015, 63, 4761–4770. [Google Scholar] [CrossRef] [PubMed]
- Jiahui, D.; Jianxue, L.; Sihai, H.; Xuan, L.; Jinke, L.; Xinluo, D.; Peiyan, L.; Baocheng, X.; Denglin, L. Effect of microwave radiation on acetic acid and ethyl acetate in Luzhou-flavor liquor. Food Mach. 2019, 2, 33–54. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).