A Preliminary Evaluation to Establish Bath Pasteurization Guidelines for Hard Cider
Abstract
1. Introduction
2. Materials and Methods
2.1. Cider Fermentation
2.2. Microbial Inoculation
2.3. Pasteurization
2.4. Enumeration
2.5. Statistical Analysis and Replicates
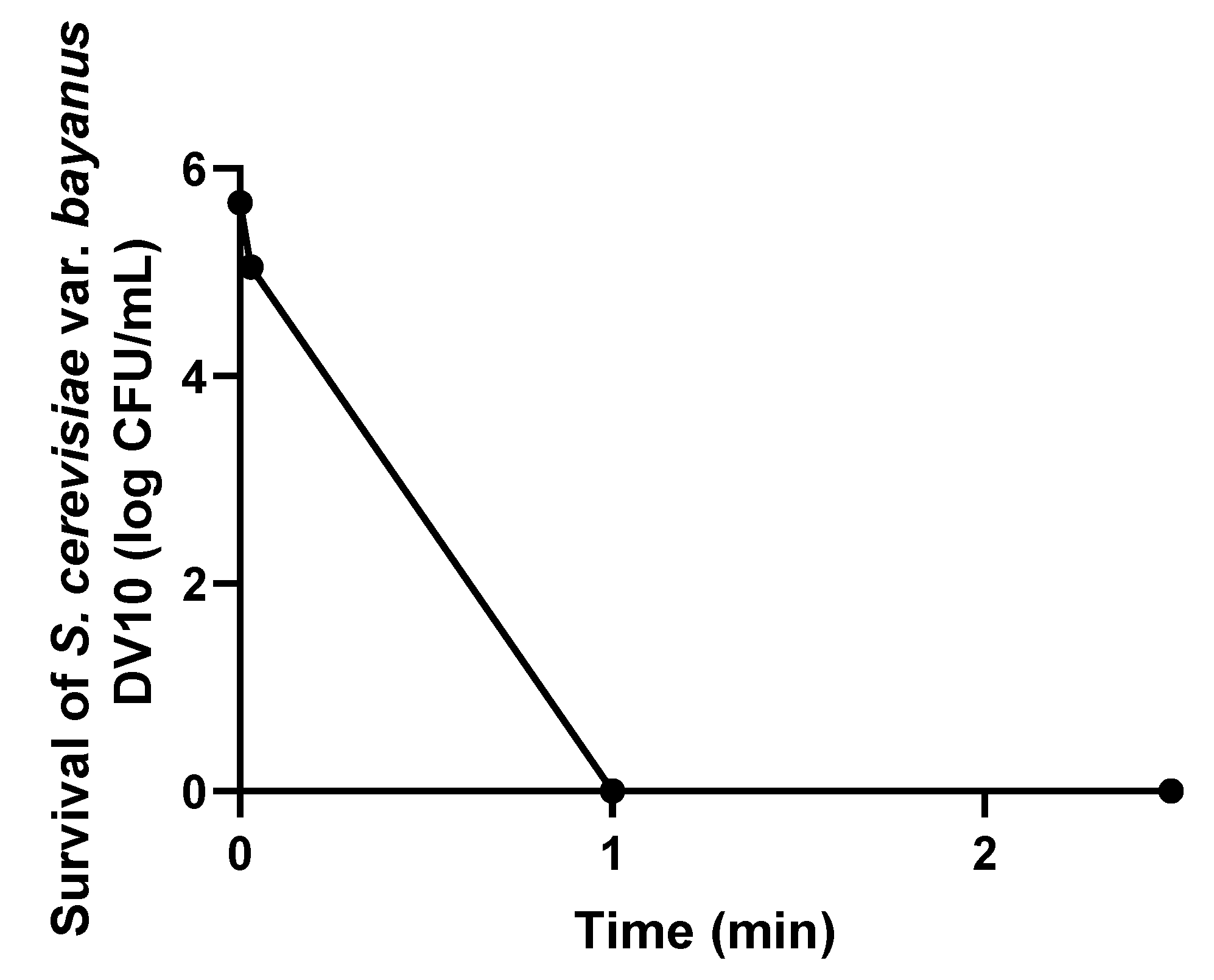
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Grinbaum, A.; Ashkenazi, I.; Treister, G.; Goldschmied-Reouven, A.; Block, C.S. Exploding bottles: Eye injury due to yeast fermentation of an uncarbonated soft drink. Br. J. Ophthalmol. 1994, 78, 883. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fugelsang, K.; Edwards, C. Wine Microbiology: Practical Applications and Procedures, 2nd ed.; Springer Science: New York, NY, USA, 2007; p. 168. [Google Scholar]
- Vecchio, H.D.; Dayharsh, C.A.; Baselt, F.C. Thermal death time studies on beer spoilage organisms–I. In Proceedings of Annual Meeting—American Society of Brewing Chemists; Taylor & Francis: London, UK, 1951; Volume 9, pp. 45–50. [Google Scholar]
- Tsang, E.W.T.; Ingledew, W.M. Studies on the heat resistance of wild yeasts and bacteria in beer. J. Am. Soc. Brew. Chem. 1982, 40, 1–8. [Google Scholar] [CrossRef]
- Buzrul, S. A suitable model of microbial survival curves for beer pasteurization. LWT-Food Sci. Technol. 2007, 40, 1330–1336. [Google Scholar] [CrossRef]
- Campbell, I. Microbiological Methods in Brewing Analysis. In Brewing Microbiology, 3rd ed.; Priest, F.G., Campbell, I., Eds.; Springer US: Boston, MA, USA, 2003; pp. 367–392. [Google Scholar]
- Mitchell, P. Cider & Perry Production-A Foundation; The Cider & Perry Academy: Gloucestershire, UK, 2017. [Google Scholar]
- Gao, C.; Fleet, G.H. The Effects of Temperature and PH on the Ethanol Tolerance of the Wine Yeasts, Saccharomyces Cerevisiae, Candida Stellata and Kloeckera Apiculata. J. Appl. Bacteriol. 1988, 65, 405–409. [Google Scholar] [CrossRef]
- Malletroit, V.; Guinard, J.-X.; Kunkee, R.E.; Lewis, M.J. Effect of pasteurization on microbiological and sensory quality of white grape juice and wine. J. Food. Process. Pres. 1991, 15, 19–29. [Google Scholar] [CrossRef]
- Splittstoesser, D.F.; Lienk, L.L.; Wilkison, M.; Stamer, J.R. Influence of wine composition on the heat resistance of potential spoilage organisms. Appl. Microbiol. 1975, 30, 369. [Google Scholar] [CrossRef] [PubMed]
- Adams, M.R.; O’Brien, P.; Taylor, G.T. Effect of the ethanol content of beer on the heat resistance of a spoilage Lactobacillus. J. Appl. Bacteriol. 1989, 66, 491–495. [Google Scholar] [CrossRef]
| Parameter | Part I | Part II |
|---|---|---|
| Chemistries of juice prior to fermentation. | ||
| SSC (°Brix) | 13.0 ± 1.0 | 13.0 ± 1.0 |
| Titratable Acidity (g/L) | 4.0 ± 0.0 | 3.9 ± 0.1 |
| pH | 3.5 ± 0.0 | 3.5 ± 0.0 |
| Free SO2 (mg/L) | 13.5 ± 0.6 | 8.3 ± 1.7 |
| Total SO2 (mg/L) | 28.0 ± 2.6 | 14.8 ± 1.3 |
| Chemistries of fermented cider prior to pasteurization treatments. | ||
| Titratable Acidity (g/L) | 5.6 ± 0.1 | 5.8 ± 0.0 |
| pH | 3.6 ± 0.0 | 3.6 ± 0.0 |
| Free SO2 (mg/L) | 8.3 ± 1.3 | 12.3 ± 0.5 |
| Total SO2 (mg/L) | 21.0 ± 1.4 | 33.8 ± 3.0 |
| Organism | PUs Applied | Incubation Time (Days) | Mean (CFU/mL) | Standard Deviation (CFU/mL) |
|---|---|---|---|---|
| S. cerevisiae | 0 | 2 | 4.6 × 105 | 1.7 × 104 |
| 0.03 | 2 | 1.1 × 105 | 1.4 × 104 | |
| 1 | 2 | <1 | 0 | |
| 2.5 | 2 | <1 | 0 | |
| 5 | 2 | <1 | 0 | |
| 13 | 2 | <1 | 0 | |
| 23 | 2 | <1 | 0 | |
| Z. rouxii | 0 | 2 | 4.8 × 103 | 2.5 × 103 |
| 1 | 2 | <1 | 0 | |
| 5 | 2 | <1 | 0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valliere, B.; Harkins, S. A Preliminary Evaluation to Establish Bath Pasteurization Guidelines for Hard Cider. Beverages 2020, 6, 24. https://doi.org/10.3390/beverages6020024
Valliere B, Harkins S. A Preliminary Evaluation to Establish Bath Pasteurization Guidelines for Hard Cider. Beverages. 2020; 6(2):24. https://doi.org/10.3390/beverages6020024
Chicago/Turabian StyleValliere, Brianna, and Sarah Harkins. 2020. "A Preliminary Evaluation to Establish Bath Pasteurization Guidelines for Hard Cider" Beverages 6, no. 2: 24. https://doi.org/10.3390/beverages6020024
APA StyleValliere, B., & Harkins, S. (2020). A Preliminary Evaluation to Establish Bath Pasteurization Guidelines for Hard Cider. Beverages, 6(2), 24. https://doi.org/10.3390/beverages6020024




