Abstract
Aqueous extracts (20% w/w) of dried berry fruits and skins were used as sources of phenolic compounds to fortify yogurt beverages. The total phenol and anthocyanin content of the reformulated yogurts were determined, and the antioxidant properties were compared to plain yogurt (C) during storage at 4 °C for a total period of four weeks. Yogurt beverages fortified with salal berry (SB) extracts contained higher amounts of phenolic compounds (>69.9 μg GAE/mL) and anthocyanins (>19.12 mg C3G/L) compared to drinks supplemented with blackcurrant pomace (BC) extract (>50.13 μg GAE/mL and >10.80 mg C3G/L respectively). Storage affected the stability of anthocyanins, whereas total phenol content remained unaffected. Yogurts with SB displayed the highest antioxidant capacity followed by samples with BC, which is attributed to the radical scavenging effect of the bioactive compounds present with antioxidant properties. The antioxidant capacity of the yogurt beverages fortified with fruit extracts was maintained during cold storage. Findings of this study indicate that SB and BC pomace can be used as functional ingredients to increase the antioxidant potential of yogurt beverages.
1. Introduction
Yogurt is a popular dairy product typically produced by the fermentation activity of Lactobacillus delbrueckii ssp. Bulgaricus and Streptococcus thermophilus. It is a major source of essential macro- and micronutrients, contributes to the daily energy intake and as such is an important part of the human diet [1]. Yogurt is commercially available in various forms and is highly appreciated for its sensory properties and ease of consumption [2]. Even though yogurt contains various health-promoting ingredients which contribute to the product’s high nutritional value, plain yogurt is considered a poor source of phenolic compounds and their impact on human health is of little significance. Diverse phenolic compounds have been detected in ruminant milk including carvacrol, thymol, phenol and cresol and their relative abundance varies depending on the mammalian species [1]. Sheep milk is known to contain the highest concentration of total phenolic compounds (168 mg GAE/L), whereas cow milk contains the lowest values (49 mg GAE/L) [3]. Interspecies differences in phenolic composition and amounts are attributed to differences in feeding patterns between ruminants [1]. To overcome this limitation and further enhance the nutritional value of the product, fruit-based additives are commonly used for yogurt fortification [4,5,6].
Salal (Gaultheria shallon) is a member of the Ericaceae family which is native to western North America and has been naturalized in some areas of Britain including Scotland [7]. The phytochemical profile of salal fruits has been recently analyzed, and literature indicates the plant is a promising source of polyphenols with anthocyanins and flavonols being the major phenolic components [8]. Small berries such as blackcurrants (Ribes nigrum spp.) are also known to be good sources of polyphenolic compounds including anthocyanins, proanthocyanidins, hydroxybenzoic acids, hydroxycinnamic acids, flavonols, flavanols [9]. Blackcurrant pomace is a major by-product of juice production, which is usually discarded into soil or landfills but could be a promising food ingredient thanks to its high fiber content [10].
The incorporation of fresh or processed fruits in yogurt is a popular approach to increase the phenolic content of the product and likewise enhance its antioxidant profile. The fortification of yogurt with naturally sourced antioxidants also meets the consumer demands for “clean label” foods [11]. Furthermore, there is increasing interest in applying fruit processing wastes as functional food ingredients since significant amounts of the beneficial bioactive compounds are retained in those byproducts [12]. The utilization of food processing waste products for human consumption is also a viable approach to address environmental, economic and sustainability challenges of the modern world.
The objective of this study was to fortify a yogurt beverage with polyphenol-rich extracts from plant sources. An underutilized berry (salal fruit) and a by-product of food production (blackcurrant pomace) were used to prepare yogurt formulations, and the antioxidant properties of the reformulated products were compared with plain yogurt. Both utilized sources of antioxidants remain underexploited from a commercial perspective, and thus there is scope to investigate their antioxidants properties. To investigate the potential synergistic effect of combining food ingredients and assess changes during shelf-life, the antioxidant capacity of the yogurt samples was monitored for 4 weeks of cold storage. To our knowledge, this is the first study on the feasibility of yogurt drinks fortified with salal berries, and blackcurrant pomace and results may have practical implications for the food and drink industry.
2. Materials and Methods
2.1. Materials
Dried skimmed cow’s milk powder (Marvel), was purchased from a Tesco supermarket (Aberdeen, UK). Freeze-dried yogurt starter culture containing Lactobacillus bulgaricus and Streptococcus thermophilus was purchased from Goat Nutrition Ltd. (Ashford, England). Dried and powdered Salal berries (Gaultheria shallon) and blackcurrant (Ribes nigrum) pomace were kindly donated by the James Hutton Institute (Dundee, Scotland). All reagents used were of analytical grade.
2.2. Preparation and Storage of Aqueous Fruit Extracts and Yogurt Beverages
Extracts and yogurt beverages were prepared according to a previously described method [13]. In brief, purified water was added to dried fruit at a 10:1 ratio (v/w) for preparing each aqueous extract. The extracts were mixed for 1 h on a Stuart SRT6 tube roller (Cole-Palmer, Staffordshire, UK) at 20 °C and then centrifuged at 2290× g for 15 min using an EppendorfTM 5702R (Fisher Scientific, Loughborough, UK). The supernatant was collected and filtered with butter muslin squares (Lakeland, Aberdeen, UK) to remove any residues. The extraction process was repeated 3 times and extracts were combined and stored at −20 °C.
Yogurt was made up to 0.5 kg for each sample using the following ingredients: milk powder, filtered water, freshly prepared yogurt starter and aqueous fruit extracts. Yogurt starter was prepared according to the manufacturer’s instructions by dissolving the freeze-dried culture (5 g) in 840 g of water and adding 155 g of dried milk powder. The recipe for all samples included 16% (w/w) of dried milk powder, 3% (w/w) of yogurt starter, 61% (w/w) filtered water and 20% (w/w) of aqueous fruit extract. The water content was adjusted accordingly for the control sample (81% w/w). Yogurt mixes (milk powder, water and fruit extracts if applicable) were heated to 80 °C for 10 min to denature milk proteins and ensure thorough mixing of ingredients. This step was followed by the addition of the yogurt starter to the mixes when the samples were cooled down to a temperature of approximately 45 °C. Samples were then poured in a sterile glass container and placed in a yogurt fermenter (Lakeland, Aberdeen, UK) set at 44 °C. A portable food and dairy pH meter (Hanna Instruments Ltd, Leighton Buzzard, UK) was used to measure the changes in pH of the samples during fermentation on an hourly basis until a pH of 4.5 was reached. The fortification of yogurt beverages with berry extracts had no effect on fermentation time (data not shown) or on the growth kinetics of the lactic acid bacteria population [13]. At the end of the fermentation process (4 h) samples were diluted (1:1) on a weight basis and samples were then processed twice at 50 bar in a single stage valve homogenizer (APV-1000, SPX Flow Technology, West Sussex, UK) to ensure the beverage is homogeneous. Two different batches for every yogurt beverage (500 g) were prepared, which were stored at 4 °C for 5 weeks. Sampling was performed on a weekly basis for subsequent analysis. The experimental plan is represented below:
2.3. Preparation of Yogurt Extracts (YE)
Yogurt samples (0.8 g) were freeze-dried and powdered prior to extraction with 19.2 mL ethanol (50%) for 2 h with shaking at room temperature. Samples were then centrifuged at 3220× g (Eppendorf® 5810R, Fisher Scientific, Loughborough, UK) for 1 h at 4 °C and the supernatant was collected. The extraction was repeated twice, the supernatants were combined and dried using a rotary evaporator (Rotavapor R-114, Büchi Labortechnik AG, Switzerland) and the remaining solution was freeze-dried (FreeZone, Labconco Corporation, MO, USA). The dried YE was reconstituted in DMSO (100%) at 200 mg/mL, aliquoted and stored at −20 °C until further analysis. The same procedure was followed for preparing extracts from the dried fruit powders (DFP). YE were used for all analyses unless otherwise stated.
2.4. Total Anthocyanin and Total Phenol Assays
The total anthocyanin concentration was estimated by the pH differential absorbance method [14]. YE were diluted with pH 1.0 buffer (potassium chloride, 0.025 M) until absorbance at 520 nm was within the linear range of the spectrophotometer, and the same dilution factor was used to prepare all samples for pH 4.5 buffer (sodium acetate, 0.4 M). Diluted samples were turbid and were therefore centrifuged prior to measuring absorbance at 520 and 700 nm (20–50 min after preparation). The diluted samples were read versus a blank cell filled with distilled water. To calculate the anthocyanin pigment concentration, (expressed as cyanidin-3-glucoside equivalents), the following formula was used:
where A = (A520 nm − A700 nm)pH 1.0 − (A520 nm − A700 nm)pH 4.5; MW (molecular weight) = 449.2 g/mol for cyanidin-3-glucoside (cyd-3-glu); DF = dilution factor established in D; l = pathlength in cm; ε = 26,900 molar extinction coefficient for cyd-3-glu in L × mol–1 × cm–1 and 103 = factor for conversion from g to mg.
Phenol content was measured using a modified Folin–Ciocalteau method [15]. In brief, 50 μL of YE and DFP were mixed with 250 μL of Folin–Ciocalteu reagent and incubated at room temperature for 1 min. Following the addition of 200 μL of 7.5% (w/v) sodium carbonate to the mixture, total polyphenols were determined after 1 h of incubation in the dark at room temperature. The absorbance of the reaction mixture was determined at 765 nm against a blank sample using a SpectraMax 190 microplate reader (Molecular Devices Limited, Berkshire, UK). Quantification was done with respect to the standard curve of Gallic acid and results are expressed as Gallic acid equivalents (GAE).
2.5. Antioxidant Properties
2.5.1. Ferric Reducing Power (FRP)
The reducing power of yogurt beverages was measured as described by Siddhuraju and Becker [16] with slight modifications. One hundred μL of YE (and blank) were dissolved in 250 μL phosphate buffer (0.2 M, pH 6.6), mixed with 250 μL potassium ferricyanide (1% w/v) and incubated at 50 °C for 20 min. At the end of the incubation period, 250 μL of trichloroacetic acid (10% w/v) was added to stop the reaction, and the mixtures were centrifuged at 650× g for 10 min. One hundred μL of distilled water and 25 μL of ferric chloride (0.1% w/v) were added to 100 μL of the supernatant in a 96 well plate, and mixed for 10 s on a plate mixer. The absorbance of the reaction mixture was determined at 700 nm after 10 min, against a blank sample using a SpectraMax 190 microplate reader (Molecular Devices Limited, Berkshire, UK). An increased absorbance of the reaction mixture indicated the increased reducing power.
2.5.2. Ferric Reducing Ability of Plasma Assay (FRAP)
The total antioxidant potential of the yogurt beverage samples was determined using the FRAP assay adapted from Benzie and Strain [17] as a measure of antioxidant power. The FRAP reagent was freshly prepared by mixing 25 mL of acetate buffer (300 mM, pH 3.6), 2.5 mL of a solution of 10 mM TPTZ in 40 mM HCl, and 2.5 mL of 20 mM FeCl3 and once prepared was kept in a water bath at 37 °C. The following amounts were added directly in a 96 well plate and absorbance was measured using a SpectraMax 190 microplate reader (Molecular Devices Limited, Berkshire, UK) thermostated at 37 °C: 180 μL FRAP reagent and 24 μL of YE, standard or H2O as a blank. Butylated hydroxytoluene (BHT) was used as positive control. Absorbance readings were taken at 593 nm after 10 min at 37 °C. Serial dilutions of a 1 mM FeSO4 solution were used to prepare the standard curve. The results were corrected for dilution and expressed in μM Fe+2 per g.
2.5.3. Color Measurement
Color analysis of the yogurt beverages was performed using a Konica Minolta CR1 10 colorimeter (Konica Minolta Solutions Ltd., Basildon, UK). The measurements were conducted under artificial light to minimize daylight effects. The color parameters L* (lightness), a* (red/greenness), and b* (yellow/blueness) of the yogurt samples were evaluated according to the International Commission on Illumination (CIE) L*a*b* system.
2.6. Statistical Analysis
Results are expressed as mean±standard deviation (SD) of at least three replicates. Statistical analysis of the data was performed using the statistical software SPSS Statistics 25 (IBM Corp., Armonk, NY, USA). The normality of data distribution was tested by the Shapiro-Wilk method. Statistical significance values of groups’ means were made by repeated measures analysis of variance (rmANOVA) and Bonferroni post hoc test. The statistical analysis performed were considered significant when p < 0.05.
3. Results and Discussion
3.1. Total Anthocyanin and Total Phenol Content of Different Yogurt Beverages during Cold Storage
Salal berries and blackcurrants are both good sources of phenolic compounds, especially anthocyanins. Therefore, total anthocyanin and total phenol content of yogurt samples containing salal berries and blackcurrant pomace extract were analyzed during their shelf-life period. As presented in Figure 1, total phenol contents in yogurt beverages containing SB and BC were both higher than the plain yogurt (Control). This effect was significant (p < 0.05) for all storage times, except for week 2 (p = 0.058 for BC and C) and week 5 (p = 0.416 for BC and C, and p = 0.343 for SB and BC). The difference in total phenol content of beverages fortified with fruits extracts (SB > BC) reflects the observed difference between the amounts detected in the fruit’s powders (Figure 2). An increase of total phenols was observed for all yogurts during the first four weeks of storage, which was significant (p < 0.05) for C and BC yogurts. Similar fluctuations in the phenolic content during refrigerated storage has been documented for fruit juices [18,19] and was attributed to the formation of compounds that react with the Folin-Ciocalteu reagent [20]. For instance, proteolysis of milk proteins may release amino acids with phenolic side chains, such as tyrosine, which could contribute to the increase in total phenol content. In addition, metabolism of phenolic compounds by the yogurt cultures may include flavonoid glycoside hydrolysis or C-ring cleavage and the release of simple phenolics such as phenolic acids [21,22]. Following storage for four weeks, total phenol content in yogurt beverage containing SB had a significant reduction, which was similar to other yogurts containing different fruit extracts [4,6,21]. Polyphenols are known to interact with milk proteins and form insoluble complexes which reduce the total free polyphenol content [23]. However, significant levels of polyphenols were detected even after five weeks of storage.
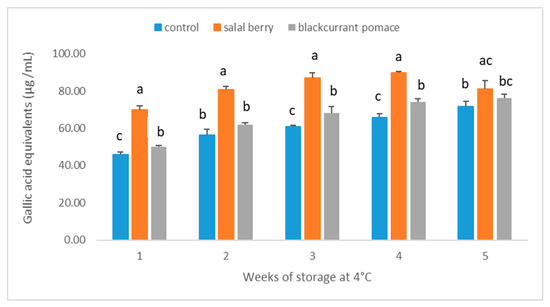
Figure 1.
Total phenol content of yogurt beverages during cold storage. Results are presented as means ± SD. Different low case letters denote significant differences at each time point (p < 0.05).
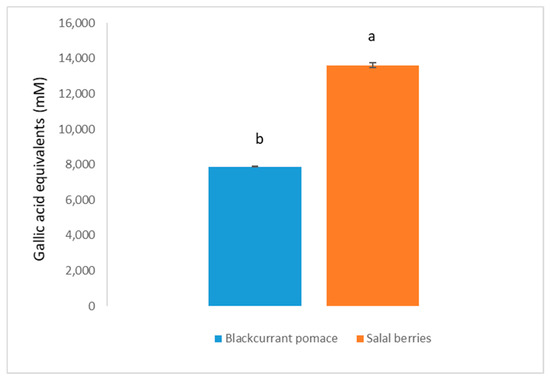
Figure 2.
Total phenol content of dried fruit powders (DFP). Results are presented as means ± SD. Different low case letters denote significant differences (p < 0.05).
Anthocyanins are of interest to the food industry because of their antioxidant power, attractive color, and stability in high acid foods [24]. The total anthocyanin content of different types of yogurt beverages was analyzed. As shown in Figure 3, the addition of SB and BC extracts increased the total anthocyanin content significantly (p < 0.05) in yogurt beverages compared to the plain yogurt, and the yogurt containing SB had higher total anthocyanin content than that containing BC (p < 0.05). The anthocyanin content of the yogurt beverages is also reflected on the color properties of the samples (Figure 4). Yogurt beverages fortified with SB show a positive a* value which signifies red color, whereas for both control and BC samples the axis coordinate is negative. As expected, the control beverage has the highest L* value which suggests it has the brightest color followed by BC and SB. The total anthocyanin content decreased gradually with the extension of storage time, which is in accord with other research [23,24]. This effect was significant (p < 0.05) for SB and BC yogurts, whereas the anthocyanin content of the plain yogurt remained stable. Anthocyanins are highly reactive pigments and readily degrade during storage depending on environmental factors, such as pH, temperature, enzymatic and microbial activity. At the end of shelf-life, around 75% of the anthocyanins in the yogurt beverages containing SB and BC remained intact.
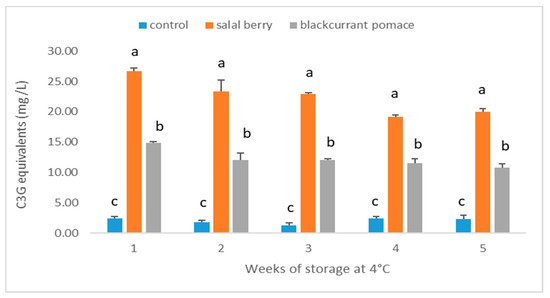
Figure 3.
Total anthocyanin content of yogurt beverages during cold storage. Results are presented as means ± SD. Different low case letters denote significant differences at each time point (p < 0.05).

Figure 4.
Color properties of yogurt beverages. Numerical findings represent color differences between samples using the CIE L*a*b* (International Commission on Illumination) color scale ± SD.
3.2. Antioxidant Properties of Different Types of Yogurt Beverages
Phenolic compounds could act as antioxidants via their ability to donate hydrogen or electrons which results in the termination of a chain reaction or by chelating transition metal ions. Many fruit extracts rich in phenolic compounds have been added to dairy products to improve their antioxidant capacity [21]. In this research, FRP and FRAP assays were used to comprehensively evaluate the antioxidant capacities of different types of yogurt beverages. Their principles, advantages and limitations are presented below [25].
3.2.1. Ferric Reducing Power of Yogurt Beverages
It has been reported that the reducing power is associated with the antioxidant activity and this relationship has been established with numerous bioactive compounds isolated from natural products [26]. The FRAP assay measures the presence of reducers (i.e., antioxidants) which result in the reduction of the Fe3+/ferricyanide complex to the ferrous form. An increase in the absorbance of the reaction mixtures indicates an increase in antioxidant activity. In this method, the antioxidant compounds present form a colored complex with potassium ferricyanide, trichloro acetic acid and ferric chloride. The method offers the advantage that measurements are recorded at 700 nm, which is distant from the typical absorption spectrum of anthocyanins (500–600 nm) and thus reduces the risk of spectral interference [27]. Figure 5 shows that the yogurt beverage containing SB had significantly stronger reducing power compared with the other beverages (C, BC) included in this study. BC yogurts were also significantly more potent for reducing ferric to ferrous ions compared to plain yogurt, except for weeks 3 (p = 0.8) and 5 (p = 0.6). During the storage period at 4 °C, the reducing power of the yogurt containing SB gradually reduced at significant levels (p = 0.036), whereas the plain yogurt and that containing BC remained stable between weeks 1 and 5. Siddhuraju and Becker reported that the reducing power is correlated with the concentration of phenolic compounds [16]. The reduction of the reducing power of the yogurt containing SB may be explained by the degradation of polyphenols in SB extract and/or the precipitation of phenolic compounds due to milk protein-polyphenol interactions. Previous studies have reported that the antioxidant activity of polyphenols decreases with increasing interactions between milk proteins and polyphenols [28].
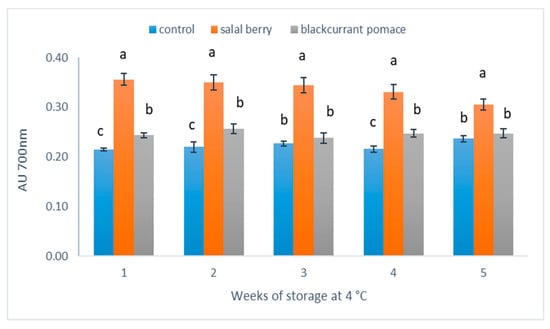
Figure 5.
Reducing power of different yogurt beverages. Results are presented as means ± SD. Different low case letters denote significant differences at each time point (p < 0.05).
3.2.2. Ferric Reducing Antioxidant Power Assay of Different Yogurt Beverages
The FRAP assay measures total electron transfer and in combination with other methods can be useful for distinguishing dominant mechanisms with different antioxidants [27]. FRAP assay measures the ability of antioxidants to reduce ferric (Fe3+) to ferrous (Fe2+) ion at low pH through the donation of an electron. Although the results generated by the FRAP assay are not mechanistically or physiologically relevant, it is widely used for in vitro experiments thanks to its robustness and convenience. In agreement to the results of the FRP assay, the yogurt beverage containing SB showed stronger FRAP antioxidant potential than the other samples (Figure 6). Storage time throughout 4 weeks had significant effects on FRAP antioxidant potential of the yogurt beverage containing SB (p = 0.007 between week 1 and 4); however, the same trend was not observed for the other two samples. After storage for four weeks the FRAP antioxidant potential of the yogurt containing SB reduced by around 18%, whereas the plain and BC yogurts showed no significant change. Ashwani and Lawin reported that the decrease in antioxidant activity of yogurts fortified with fruit extract were attributed to the loss of anthocyanin activity [29,30]. The decrease of FRAP antioxidant potential of the yogurt beverage containing SB may be due to the decrease in the anthocyanin content (Figure 7), which is more profound for the samples supplemented with the SB extract (Figure 3).
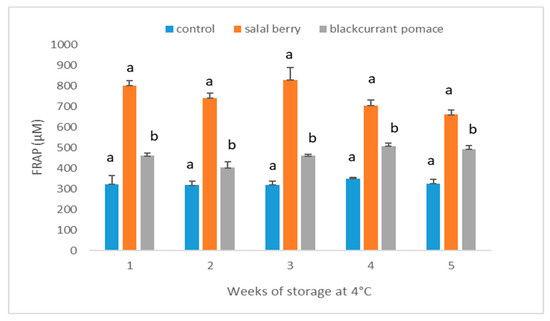
Figure 6.
FRAP (ferric reducing antioxidant power) assay of different yogurt beverages. Results are presented as means ± SD. Different low case letters denote significant differences at each time point (p < 0.05).
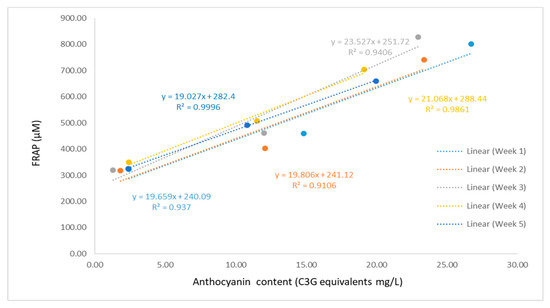
Figure 7.
Correlation between FRAP and total anthocyanin content.
4. Conclusions
Fruit extracts containing polyphenols are often added to yogurt and other dairy products to improve their antioxidant activity. In this research, the fortified yogurt beverages containing SB and BC extract had higher amounts of polyphenolic compounds and anthocyanins compared to the plain control and showed higher antioxidant activities. Storage time affected the stability of anthocyanins, FRP and FRAP antioxidant potentials of the yogurt containing SB. The effect of storage was less profound for control samples and beverages fortified with BC due to their low content of anthocyanins, and thus degradation losses were not significant. This study suggests that the antioxidant potential of yogurt remains moderately stable during shelf-life. Furthermore, SB and BC pomace could be used as functional ingredients to enhance the phytochemical profile of dairy products and increase their antioxidant potential and health-promoting properties. Future studies to determine the organoleptic properties of the reformulated samples should be conducted to assess the commercial viability of the yogurt beverages.
Author Contributions
V.R. (Vassilios Raikos) conceptualization; V.R. (Vassilios Raikos), H.H, methodology, V.R. (Vassilios Raikos) and H.H. data analysis, V.R. (Vassilios Raikos), V.R. (Viren Ranawana) and H.N. writing and reviewing.
Funding
This work is part of the Strategic Research Programme 2016–2021 and is funded by the Scottish Government’s Rural and Environment Science and Analytical Services Division (RESAS).
Conflicts of Interest
The authors declare no conflict of interest.
References
- O’Connell, J.E.; Fox, P.F. Significance and applications of phenolic compounds in the production and quality of milk and dairy products: A review. Int. Dairy J. 2001, 11, 103–120. [Google Scholar] [CrossRef]
- Serafeimidou, A.; Zlatanos, S.; Kritikos, G.; Tourianis, A. Change of fatty acid profile, including conjugated linoleic acid (CLA) content, during refrigerated storage of yogurt made of cow and sheep milk. J. Food Compos. Anal. 2013, 31, 24–30. [Google Scholar] [CrossRef]
- Velázquez Vásquez, C.; Villa Rojas, M.G.; Alvarez Ramírez, C.; Chávez-Servín, J.L.; García-Gasca, T.; Ferriz Martínez, R.A.; Garcia, O.P.; Rosado, J.L.; López-Sabater, C.M.; Castellote, A.I.; et al. Total phenolic compounds in milk from different species. Design of an extraction technique for quantification using the Foline-Ciocalteu method. Food Chem. 2015, 176, 480–486. [Google Scholar] [CrossRef] [PubMed]
- Karaaslan, M.; Ozden, M.; Vardin, H.; Turkoglu, H. Phenolic fortification of yogurt using grape and callus extracts. LWT—Food Sci. Technol. 2011, 44, 1065–1072. [Google Scholar] [CrossRef]
- Trigueros, L.; Pérez-Alvarez, J.A.; Viuda-Martos, M.; Sendra, E. Production of low-fat yoghurt with quince (Cydonia oblonga Mill.) scalding water. LWT—Food Sci. Technol. 2011, 44, 1388–1395. [Google Scholar] [CrossRef]
- Chouchouli, V.; Kalogeropoulus, N.; Konteles, S.J.; Karvela, E.; Makris, D.P.; Karathanos, V.T. Fortification of yogurts with grapes (Vitis vinifera) seed extracts. LWT—Food Sci. Technol. 2013, 53, 522–529. [Google Scholar] [CrossRef]
- Keely, P.B.; Martinsen, C.S.; Hunn, E.S.; Norton, H.H. Composition of native American fruits in the Pacific Northwest. J. Am. Diet. Assoc. 1982, 81, 568–572. [Google Scholar] [PubMed]
- McDougall, G.J.; Austin, C.; Van Schayk, E.; Martin, P. Salal (Gaultheria shallon) and aronia (Aronia melanocarpa) fruits from Orkney: Phenolic content, composition and effect of wine-making. Food Chem. 2013, 205, 239–247. [Google Scholar] [CrossRef]
- Landbo, A.K.; Meyer, A.S. Enzyme-assisted extraction of antioxidative phenols from black currant juice residue (Ribes nigrum). J. Agric. Food Chem. 2001, 49, 3169–3177. [Google Scholar] [CrossRef]
- Rohm, H.; Brennan, C.S.; Turner, C.; Günther, E.; Campbell, G.M.; Hernando, I.; Struck, S.; Kontogiorgos, V. Adding value to fruit processing waste: Innovative ways to incorporate fibers from berry pomace in baked and extruded cereal-based foods e a SUSFOOD project. Foods 2015, 4, 690–697. [Google Scholar] [CrossRef]
- Granato, D.; Nunes, D.S.; Barba, F.J. An integrated strategy between food chemistry, biology, nutrition, pharmacology, and statistics in the development of functional foods: A proposal. Trends Food Sci. Technol. 2017, 62, 13–22. [Google Scholar] [CrossRef]
- Balasundram, N.; Sundram, K.; Samman, S. Phenolic compounds in plants and agri-industrial by-products: Antioxidant activity, occurrence, and potential uses. Food Chem. 2006, 99, 191–203. [Google Scholar] [CrossRef]
- Ni, H.; Hayes, H.E.; Stead, D.; Raikos, V. Incorporating salal berry (Gaultheria shallon) and blackcurrant (Ribes nigrum) pomace in yogurt for the development of a beverage with antidiabetic properties. Heliyon 2018, 4, e00875. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Durst, R.W.; Wrolstad, R.E. Determination of total monomeric anthocyanin pigment content of fruit juices, beverages, natural colorants, and wines by the pH differential method: Collaborative study. J. AOAC Int. 2005, 88, 1269–1278. [Google Scholar] [PubMed]
- Deighton, N.; Brennan, R.; Finn, C.; Davies, H.V. Antioxidant properties of domesticated and wild Rubus species. J. Sci. Food Agric. 2000, 80, 1307–1313. [Google Scholar] [CrossRef]
- Siddhuraju, P.; Becker, K. Antioxidant properties of various solvent extracts of total phenolic constituents from three different agroclimatic origins of drumstick tree (Moringa oleifera Lam.) leaves. J. Agric. Food Chem. 2003, 51, 2144–2155. [Google Scholar] [CrossRef] [PubMed]
- Benzie, I.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Analy. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef]
- Cilla, A.; Perales, S.; Lagarda, M.J.; Barberá, R.; Clemente, G.; Farré, R. Influence of storage and in vitro gastrointestinal digestion on total antioxidant capacity of fruit beverages. J. Food Compos. Anal. 2011, 24, 87–94. [Google Scholar] [CrossRef]
- González-Larena, M.; Cilla, A.; García-Llatas, G.; Barberá, R.; Jesús Lagarda, M. Plant Sterols and Antioxidant Parameters in Enriched Beverages: Storage Stability. J. Agric. Food Chem. 2012, 60, 4725–4734. [Google Scholar] [CrossRef]
- Piljac-Žegarac, J.; Valek, L.; Martinez, S.; Belščak, A. Fluctuations in the phenolic content and antioxidant capacity of dark fruit juices in refrigerated storage. Food Chem. 2009, 113, 394–400. [Google Scholar] [CrossRef]
- Premalatha, M.; Amal, B.S.; Ahmad, S.B. Influence of green, white and black tea addition on the antioxidant activity of probiotic yogurt during refrigerated storage. Food Packag. Shelf Life 2016, 8, 1–8. [Google Scholar]
- Korhonen, H. Milk derived bioactive peptides: From science to applications. J. Funct. Foods 2009, 1, 177–187. [Google Scholar] [CrossRef]
- Oliveira, A.; Alexandre, E.M.C.; Coelho, M.; Lopes, C.; Almeida, D.P.F.; Pintado, M. Incorporation of strawberries preparation in yoghurt: Impact on phytochemicals and milk proteins. Food Chem. 2015, 171, 370–378. [Google Scholar] [CrossRef] [PubMed]
- Wallace, T.C.; Giusti, M.M. Determination of color, pigment, and phenolic stability in yogurt systems colored with nonacylated anthocyanins from Berberis boliviana L. as compared to other natural/synthetic colorants. J. Food Sci. 2010, 73, C241–C248. [Google Scholar] [CrossRef] [PubMed]
- Prior, R.L.; Wu, X.; Schaich, K. Standardised methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. J. Agric. Food Chem. 2005, 53, 4290–4302. [Google Scholar] [CrossRef] [PubMed]
- Siddhurajua, P.; Mohanb, P.S.; Becker, K. Studies on the antioxidant activity of Indian Laburnum (Cassia fistula L.): A preliminary assessment of crude extracts from stem bark, leaves, flowers and fruit pulp. Food Chem. 2002, 79, 61–67. [Google Scholar] [CrossRef]
- Merzlyak, M.N.; Chinkunova, O.B.; Solovchenko, A.E.; Navqi, K.R. Light absorption by anthocyanins in juvenile, stressed and senescing leaves. J. Exp. Bot. 2008, 59, 3903–3911. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Mao, F.; Yang, F.; Zhao, Y.; Zhang, C.; Yamamoto, K. Interaction of dietary polyphenols with bovine milk proteins: Molecular structure-affinity relationship and influencing bioactivity aspects. Mol. Nutr. Food Res. 2011, 55, 1637–1645. [Google Scholar] [CrossRef]
- Ashwani, K.; Dinesh, K. Development of antioxidant rich fruit supplemented probiotic yogurts using free and microencapsulated Lactobacillus rhamnosus culture. J. Food Sci. Technol. 2016, 53, 667–675. [Google Scholar]
- Lawin, P.; Kongbangkerd, T. Development of probiotic yoghurt mixed with roselle syrup. KKU Res. J. 2010, 15, 803–808. [Google Scholar]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).