1. Introduction
Rubus species are an excellent source of dietary phytochemicals and comprise berries with a high content of polyphenols. In particular, red raspberry (
Rubus ideaus) has a high content of phenolic acids and flavonoids, with mean contents as high as 113 mg/100 g and 88 mg/100 g, respectively [
1]. Phenolic compounds and vitamin C have been implicated in the antioxidant capacity of fruits and derived beverages [
2]. The phenolic compounds from
Rubus sp. contribute to the total antioxidant capacity of the raspberry [
3,
4].
Polyphenols are considered to play a protective role in several chronic diseases associated with oxidative stress. Studies suggest that diets rich in plant polyphenols offer some protection against the development of cancers, cardiovascular diseases, diabetes, osteoporosis, and neurodegenerative diseases [
5]. This embodies one of the main reasons for consumers’ growing interest in antioxidant-rich products, alongside an interest in products with reduced sugar content and of natural origin.
Stevia rebaudiana Bertoni is a perennial herb native to South America, where it has been used for centuries to sweeten food and beverages. Stevia leaves are several times sweeter than table sugar but have no calories [
6,
7,
8]; steviol glycosides are sweet secondary metabolites that mainly occur in the leaves. Besides its sweetening properties, stevia is a good source of carbohydrates, fiber, proteins, and essential amino acids [
9,
10] and contains antioxidant compounds with diverse biochemical roles, including ascorbic acid [
11], phenolic compounds [
12,
13,
14,
15,
16], and flavonoids [
17,
18,
19]. Nowadays, stevia’s crude extracts, as well as its steviol glycosides, are used to sweeten foods and beverages. Moreover, with a recent update of the European Commission’s Novel-Food Catalogue, now recognizing
Stevia rebaudiana Bertoni as a traditional food when prepared with or contained in fruits, tea, or herbal infusions [
20], stevia has been of growing importance to the European Union (EU) beverage market.
Previous work with the oxygen radical absorbance capacity (ORAC) assay has shown that crude water extracts from stevia leaves exhibit promising antioxidant activity, superior to that of the stem’s extract, ranging from about 960 to 1070 μmol Trolox equivalents per gram of dried leaves [
19]. Moreover, the crude extracts from stevia exhibit a high antioxidant potential when compared to its purified sweet metabolites [
19].
Recently, the outcome of the addition of stevia to high-pressure processed fruits was reported [
21]. The authors showed that the addition of up to 2.5%
w/
v of stevia extract increases the total phenolic content and ORAC values, inhibits the absorption of ABTS, and contributes to the inactivation of polyphenol oxidase and peroxidase, two key enzymes involved in the oxidation of phenolic compounds, while also reducing the microbial load of the processed fruits. More recently, a study from our group showed that the heat treatment of berries-mash at 85 °C, followed by an enzymatic treatment and by heating treatments, positively affects the phenolic yield of the berry juices and leads to an increase in the antioxidant capacity [
22]. Therefore, we employed the knowledge acquired on this issue to produce natural raspberry juices, without the use of preservatives, artificial colors, or synthetic substances and sweetened with a natural source. The goal of the investigation has been to rate the effect of the addition, at two different production steps, of dry-grinded stevia leaves or their crude extract on the antioxidant potential of raspberry juices. Regarding the antioxidant potential, the study was conducted purely chemically (e.g., ORAC, total phenolics), as well as by a cell-based assay (CAA). The latter approach shows great progress over the merely chemical methods, as it takes account of the biological system’s complexity [
23]. The CAA assay has been widely used so far to assess the antioxidant potential of foods; however, to the best of our knowledge, there is no literature describing a similar approach to studying the effect of the addition of stevia on the antioxidant potential of fruit juices.
2. Methods
2.1. Plant Material
Frozen raspberries (Rubus idaeus L.) were obtained from Mainfrucht GmbH & Co. KG (Gochsheim, Germany) and stored at −20 °C until use.
Dried powders from S. rebaudiana leaves were obtained from Organic Stevia GmbH (Hannover, Germany). Extracts were prepared fresh by infusing stevia powder via stirring for 10 min in 100 °C distilled water at a final concentration of 100 g/L. The extract was centrifuged for 10 min at 14,000× g, and then, 100 mL of the resulting supernatant were immediately added to 900 mL of raspberry juice.
2.2. Juice Production
Raspberry juices were produced from 2 kg packages belonging to the same lot.
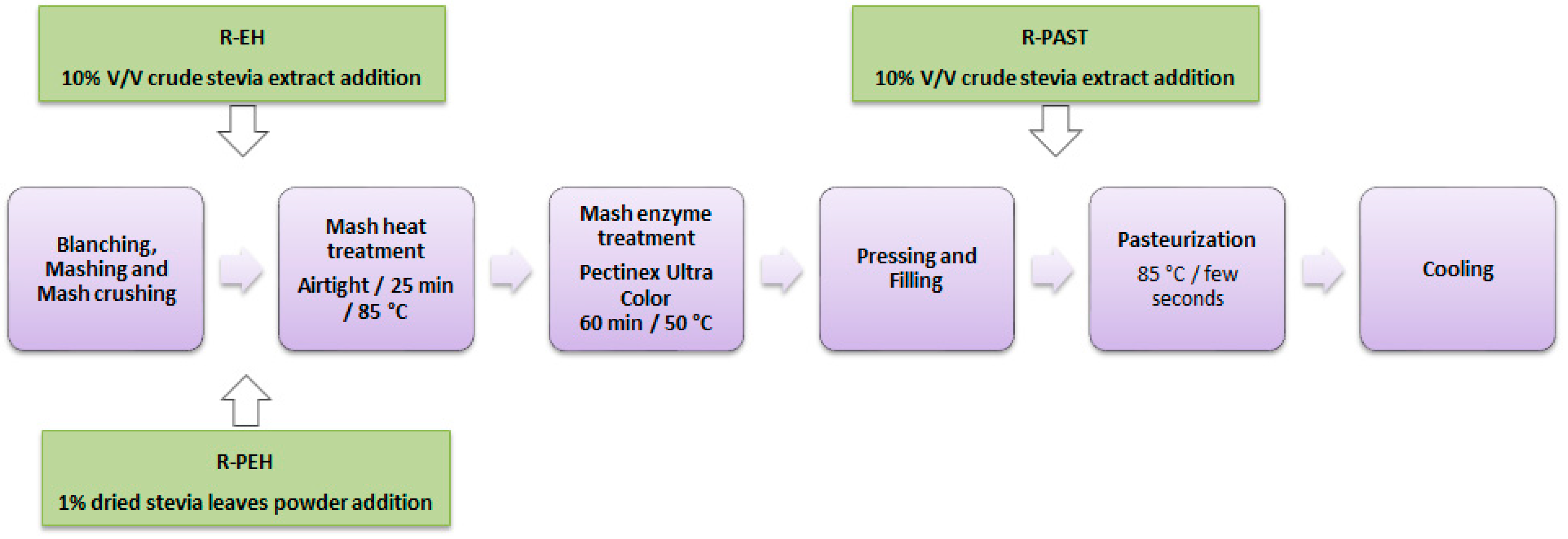
Figure 1 shows the flowchart of the red raspberry juice production. Fruit packages were defrosted by blanching with boiling distilled water for 2 min and then mashed by crushing with a blender for 1 min. To favor the phenolic yield and the antioxidant properties, the juices were produced as previously described [
22]. Briefly, the mash was heated to 85 °C in an airtight bag, held for 25 min at 85 °C, cooled open to 50 °C, and incubated at 50 °C with Pectinex
® Ultra Color (Novozymes, Bagsværd, Denmark) enzyme preparation for 60 min (dosage recommended by the manufacturer) before juice pressing.
At different production steps, each juice was sweetened to a final concentration of 10 g stevia/L. R-EH and R-PEH were sweetened immediately before the heat treatment with stevia crude extract or stevia leaves powder, respectively. R-PAST was sweetened with stevia crude extract immediately before pasteurization, and R-Std represented the unsweetened raspberry juice.
The juices were extracted using nylon filter bags and a stainless-steel Para-Press (Arauner, Kitzingen, Germany), in which the pressure was gradually raised to 5.6 bars. The pressed juices were put into 110 mL bottles, pasteurized by heating to 85 °C for a few seconds in a water bath, and then allowed to cool at room temperature. Two independent production batches from the same raspberry lot were bottled in duplicate. The juices were stored at room temperature in the dark for up to 4 weeks before analysis.
2.3. Characteristic Properties of Raspberry Juices
The yield was determined by weighing and is given as percentage.
The ascorbic acid was determined in triplicate using the Reflectoquant system RQflex 10 (Merck, Darmstadt, Germany), and the results are expressed as mg of ascorbic acid/L ± RSD %.
The total acid measurement was done in duplicate by potentiometric titration with 0.33 M NaOH (Merck, Darmstadt, Germany) to the titration endpoint of pH 7.0, calculated as acidity of wine and expressed as g/L. The pH was measured using a pH meter EcoScan (EUTECH Instruments, Nijkerk, The Netherlands). The total soluble solids (°brix) content was measured at 20 °C using a refractometer HANNA HI 96801 (Hanna Instruments, Vöhringen, Germany).
The viscosity was measured at 20 °C in duplicate with a MarsIII rheometer (Thermo Fisher Scientific, Karlsruhe, Germany). A concentric cylinder geometry was employed and rotational test was performed under variation of the shear rate between 0.5–500 s−1. The viscosity was evaluated by Newtonian fit of the shear rate range 1–100 s−1 and expressed as mPa s.
2.4. Total Phenolic Content (TPC)
The total phenolics were determined by the Folin–Ciocalteu method according to Singleton and Rossi [
24]. The total phenols are expressed as the mean of three experiments ± RSD % and as mg of gallic acid equivalents (GAE)/L of juice.
2.5. Oxygen Radical Absorbance Capacity (ORAC) Assay
The ROS scavenging capacities of the raspberry juices were determined by means of the ORAC test, according to Prior et al. [
25] with minor modifications. The juice samples were centrifuged at 3000×
g for 10 min; the supernatants were filtered through a 0.45 µm syringe filter. Serial dilutions were immediately prepared in a phosphate buffer (75 mM, pH 7.4) and analyzed in duplicate wells. The raw data were analyzed with MARS 2.0 Optima data analysis software (BMG Labtech, Offenburg, Germany). The integrated area under the fluorescence curve (AUC) was calculated for each sample and corrected with the AUC of a blank sample. Trolox equivalents (TE) were calculated based on the reference AUCs, and the ORAC values were expressed as the mean of two batches ± RSD % and as µmol TE/L of juice.
2.6. Cell Culture
HepG2 cells (Biobanking of veterinary resources-IZSLER, Brescia, Italy) were grown in DMEM high glucose medium supplemented with 10% FBS, 2 mM of L-Glutamine, 50 μg/mL of Penicillin, and 50 μg/mL of Streptomycin. The cells were maintained at 37 °C under a humidified atmosphere of 5% CO2.
2.7. Cellular Antioxidant Activity (CAA) Assay
For each experiment, the cell viability was determined by trypan blue exclusion. The CAA assay was conducted according to Wolfe and Liu [
23] with minor modifications. The HepG2 leaver cells were seeded in a black 96-well plate with transparent bottom (Greiner Bio-One, Frickenhausen, Germany) and incubated until confluence at 37 °C and 5% CO
2. The juice samples were centrifuged at 3000×
g for 10 min, and the resulting supernatants were filtered through 0.2 µm syringe filters. Subsequently, 200 µL of each filtrate were diluted in 800 µL of DMEM high glucose medium. The diluted samples were immediately tested in four replicate wells. The raw data were analyzed with MARS 2.0 Optima software (BMG Labtech, Offenburg, Germany). The AUC was calculated for each sample and corrected with the AUC of a blank sample. Quercetin equivalents (QE) were calculated based on the reference standard curves, and the CAA values were expressed as the mean ± RSD % of two independent production batches and as μmol QE/L of juice.
2.8. Statistics
The data are presented as the mean ± relative standard deviation (RSD %) of two independent production batches and compared by one-way analysis of variance (ANOVA) and the Tukey’s test. The level of statistical significance was set at
p < 0.05. Pearson’s test was used to assess the linearities of the calibration curves and the correlation among different parameters. The statistical analysis was performed with the aid of PAST 3.2 software [
26].
3. Results and Discussion
Table 1 shows the characteristic properties of the raspberry juices. The values represent an average of the two batches manufactured independently on a semi-industrial scale. The obtained pH, between 3.07 and 3.12, showed relatively acidic but typical values for fruit juices without significant differences between the samples. The juice yield was not significantly different between the samples, ranging from 81.2 to 82.5%. The viscosity was also constant between the samples, showing typical values for juices between 2.332 and 2.476 mPa s. The obtained extract contents between 8.99 and 9.77 °brix also did not differ significantly.
Under the tested conditions, the addition of 10% stevia crude extract before mash heating or juice pasteurization and the addition of 1% stevia leaf powder before mash heating had no impact on the characteristic properties of the yield, pH, °brix, or viscosity compared with the control. Similarly, it was recently reported that pH and °brix are not significantly affected in Chayote (
Sechium edule (Jacq.) Sw.) juices when mixed with dried and milled leaves of stevia (0.7–0.8%
w/
v) [
27]. The unsweetened raspberry juice (R-Std) showed 19.38 g/L of total acids. The addition of 10 g/L of stevia significantly lowered the total acid value in the raspberry juices to between 17.50 and 18.20 g/L. No significant differences were recorded between the different sweetened samples.
The ascorbic acid content of R-Std was 437 ± 26 mg/L. This value was higher in all the tested juices with stevia additions, precisely 505 ± 29 mg/L (R-PEH), 515 ± 15 mg/L (R-PAST), and 523 ± 12 mg/L (R-EH), all with the same level of significance. Therefore, the addition of 10 g/L of stevia as crude extract before mash heating or juice pasteurization or as leaves powder increases the ascorbic acid content. In this regard, Zablur et al. found that black chokeberry (
Aronia melanocarpa) juices sweetened with stevia dry-grinded leaves contain up to twice the vitamin C of chokeberry juices with added sucrose [
28]. Such an increment seems higher than that in our results with raspberry juices; however, the data for the unsweetened chokeberry juice were not reported, and thus, the effective increment through the addition of stevia cannot be figured for that berry juice.
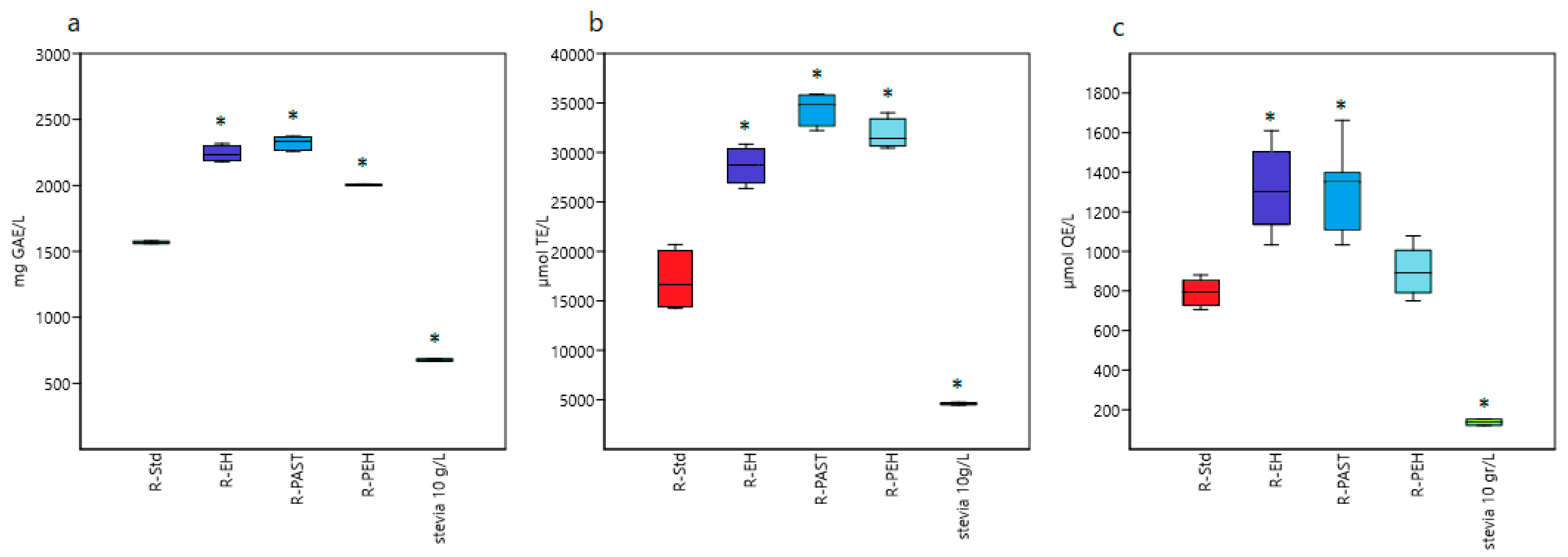
The effect of the stevia addition on the antioxidant activity of the processed raspberry juices was evaluated in vitro. As shown in
Figure 2a, the addition of 1%
w/
v stevia increased the total phenolic content of the raspberry juices. The highest content was found in the raspberry juice sweetened with stevia crude water extract immediately before pasteurization (R-PAST), being 2325 mg GAE/L ± 54, which is the mean of the two production days. Overall, higher phenolic content was observed when supplementing with the crude water extract (R-EH and R-PAST) than with the stevia powder (R-PEH). Similar results have previously been reported with the addition of stevia in juice [
29] or processed fruit pulp [
21] containing papaya, mango, and orange, as well as in chayote juice [
27]. These studies report that the addition of stevia extracts significantly increased the total phenolic content of the fruit-based preparations. In our study, the addition of stevia (grinded leaves or crude extract) increased the total phenolic content of the raspberry juices by between 27.8% and 48.3%.
The ability of the raspberry juices to scavenge peroxyl radicals was evaluated. As illustrated in
Figure 2b, all the raspberry juices have a remarkable antioxidant capacity assessed by the ORAC test. The R-Std reference sample showed intermediate to high corrected values with an average of 17,027 ± 1470 µmol TE/L. The ORAC value of the juice was significantly enhanced (
p < 0.0001) with the addition of stevia. The strongest ORAC was reached with the raspberry juice sweetened with stevia crude extract immediately before pasteurization (R-PAST), being 34,433 ± 839 µmol TE/L, and followed by the R-PEH and R-EH samples (31,818 ± 765 and 28,679 ± 918 µmol TE/L, respectively). In all preparations, it was found that the addition of stevia at a final concentration of 10 g/L (4597 ± 133 µmol TE/L) increased the antioxidant capacity compared with the unsweetened juice by at least 68.4%. Elsewhere it has been reported that the addition of stevia positively influences the ORAC values of high-pressure processed fruit-mixture containing papaya, mango, and orange pulps [
21].
The intracellular antioxidant capacity of the raspberry juices was determined through CAA assay. This method measures the inhibition of intracellular ROS, imitating the reaction between antioxidants and lipids. In addition, it considers some key cellular features (e.g., cellular uptake, metabolism, and distribution) that are not taken into account with other in vitro tests. As shown in
Figure 2c, all the raspberry juices are absorbed within the HepG2 leaver cells, and an intracellular antioxidant potential was recorded. The CAA values ranged from 900 ± 24 μmol QE/L to 1305 ± 129 μmol QE/L for the raspberry juices sweetened with stevia, being lower in the unsweetened R-Std sample (794 ± 57 μmol QE/L). This increment is significantly higher (
p < 0.0001) with the addition of stevia crude extract, but not with the leaf powder. Of note is that the stevia crude extract itself showed a CAA value of 136 ± 16, roughly 17% of that of the reference sample R-Std, but its addition to the raspberry juices increased the CAA values to up to 64%. Analogous to the results here obtained by ORAC, such reduced oxidation effect is likely to be explained by a synergistic mechanism involving the multiple components present in the juice samples.
Overall it was found that the addition of stevia at a final concentration of 10 g/L increases the antioxidant capacity of the raspberry juices in the tested industrial processes compared with the unsweetened juice, as shown by the total phenolics, ORAC, and CAA values. Under the described experimental conditions, the ORAC, CAA, and total phenolics values are positively correlated, as per a regression analysis (
Table 2).
4. Conclusions
The improvement of processing methods to obtain antioxidant-rich juices and beverages is of ever increasing interest. Most existing literature on the use of stevia is based on data regarding purified steviol glycosides, while few studies address the use of stevia leaves as an ingredient, and even fewer studies point out stevia’s antioxidant potential in foods. This study describes the effect on the antioxidant potential of raspberry juices as a result of the addition of stevia extracts or leaf powder at different production steps. It was shown that the addition of 10 g/L of stevia (relative to the final volume) to raspberry juice does not significantly influence the yield, pH value, total soluble solids, or the viscosity. In contrast, the addition can increase parameters that are associated with antioxidant potential, such as ascorbic acid, total phenolic content, ORAC and CAA values. As far as we know, this is the first report considering a cellular model to measure the antioxidant potential of a stevia-containing food. In view of the results presented here, it can be concluded that the addition of Stevia rebaudiana crude extract or its grinded leaves enhance the global antioxidant potential of raspberry juices.
Overall, stevia crude extracts show good potential to be employed as a natural ingredient for the food industry, capable of sweetening and increasing the antioxidant quality of fruit beverages, while also serving as a food preservative and adding nutritional value.