Microbial and Chemical Diversity of Traditional Non-Cereal Based Alcoholic Beverages of Sub-Saharan Africa
Abstract
1. Introduction
2. Non-Cereal-Based Alcoholic Beverages
3. Commercialization of Non-Cereal-Based Alcoholic Beverages
4. Advancements in Fermentation Strategies
5. Microbial Consortia for Fermentation
5.1. Yeasts
5.2. Bacteria
6. Microbial Identification Methods
7. Culture Dependent Identification of Microbes
8. Culture-Independent Identification of Microbes
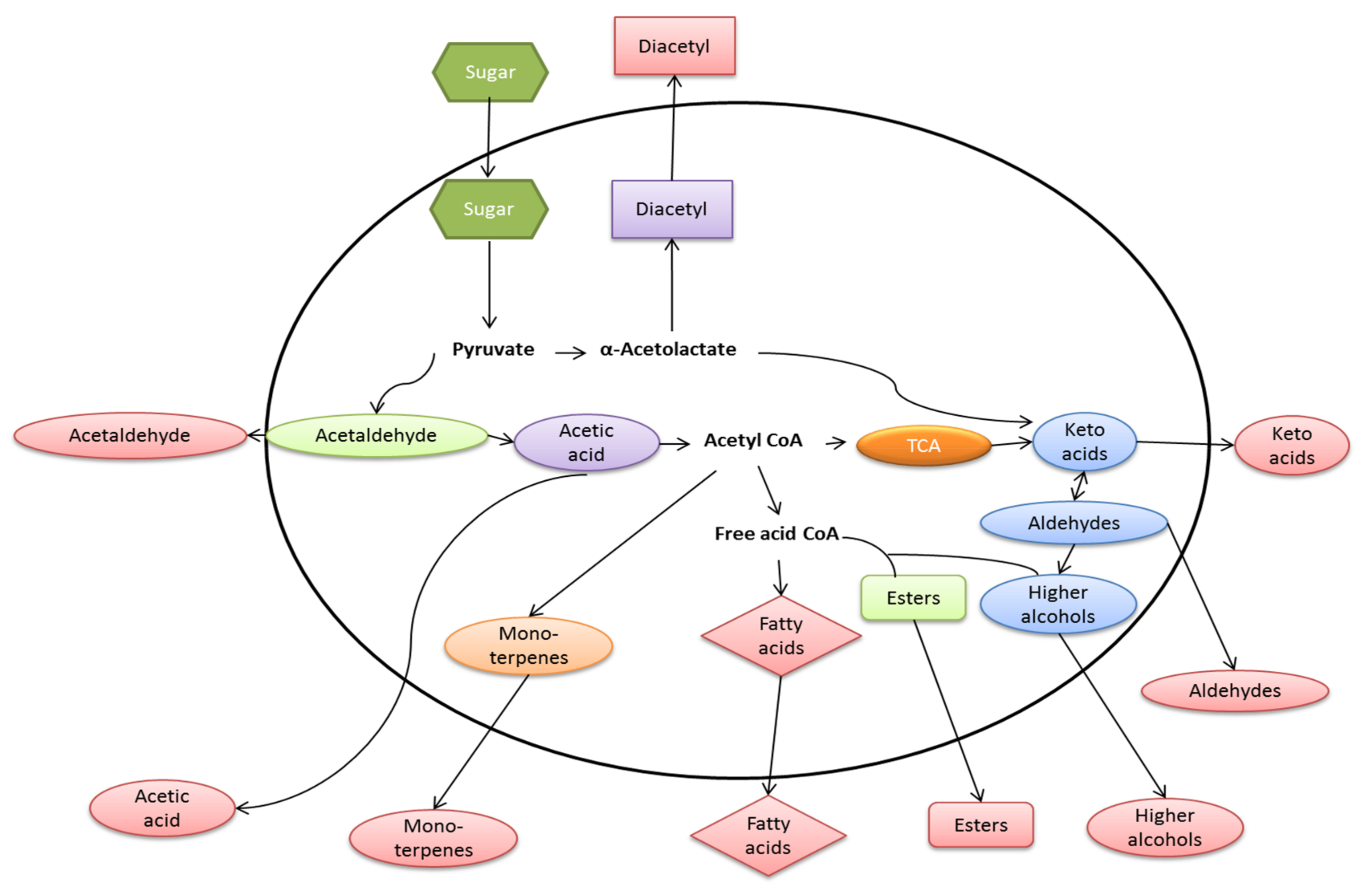
9. Chemical Products of Fermentation
10. Influence of Ecology on Fermentation Microbes
11. Microbial and Chemical Characteristics of Selected Alcoholic Beverages
12. Palm Wine
12.1. Urwagwa
12.2. Tej
12.3. Pineapple Wine
12.4. Muchema
12.5. Khadi
12.6. Setopoti
12.7. Mukumbi
12.8. Kachasu
13. Plantain Beverages
14. Cassava Brews
15. Conclusions
Conflicts of Interest
References
- Sekwati-Monang, B.; Gänzle, M.G. Microbiological and chemical characterisation of ting, a sorghum-based sourdough product from Botswana. Int. J. Food Microbiol. 2011, 150, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Kapoor, A. Levels of antinutritional factors in pearl millet as affected by processing treatments and various types of fermentation. Plant Foods Hum. Nutr. 1996, 49, 241–252. [Google Scholar] [CrossRef] [PubMed]
- Devi, P.B.; Vijayabharathi, R.; Sathyabama, S. Health benefits of finger millet (Eleusine coracana L.) polyphenols and dietary fiber: A review. Nutr. Food Sci. 2014, 51, 1021–1040. [Google Scholar] [CrossRef] [PubMed]
- Gimbi, D.M.; Kitabatake, N. Changes in alpha-and beta-amylase activities during seed germination of African finger millet. Int. J. Food Sci. Nutr. 2002, 53, 481–488. [Google Scholar] [CrossRef] [PubMed]
- Gadaga, T.H.; Mutukumira, A.N.; Narvhus, J.A.; Feresu, S.B. A review of traditional fermented foods and beverages of Zimbabwe. Int. J. Food Microbiol. 1999, 53, 1–11. [Google Scholar] [CrossRef]
- Kolawole, O.M.; Kayode, R.M.O.; Akinduyo, B. Proximate and microbial analyses of burukutu and pito produced in Ilorin, Nigeria. Afr. J. Biotechnol. 2007, 6, 587–590. [Google Scholar]
- Lee, M.; Regu, M.; Seleshe, S. Uniqueness of Ethiopian traditional alcoholic beverage of plant origin, tella. J. Ethn. Foods 2015, 2, 110–114. [Google Scholar] [CrossRef]
- Onyenekwe, P.C.; Erhabor, G.O.; Akande, S.A. Characterisation of aroma volatiles of indigenous alcoholic beverages: Burukutu and pito. Nat. Prod. Res. 2016, 30, 558–564. [Google Scholar] [CrossRef] [PubMed]
- Nyanga, L.K.; Nout, M.J.R.; Gadaga, T.H.; Theelen, B.; Boekhout, T.; Zwietering, M.H. Yeasts and lactic acid bacteria microbiota from masau (Ziziphus mauritiana) fruits and their fermented fruit pulp in Zimbabwe. Int. J. Food Microbiol. 2007, 120, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Chelule, P.; Mokoena, M.; Gqaleni, N. Advantages of traditional lactic acid bacteria fermentation of food in Africa. Curr. Res. Technol. Educ. Top. Appl. Microbiol. Microb. Biotechnol. 2010, 2, 1160–1167. [Google Scholar]
- Tamang, J.P.; Watanabe, K.; Holzapfel, W.H. Review: Diversity of microorganisms in global fermented foods and beverages. Front. Microbiol. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Tafere, G. A review on Traditional Fermented Beverages of Ethiopian. J. Nat. Scinces Res. 2015, 5, 94–103. [Google Scholar]
- Blandino, A.; Al-Aseeri, M.E.; Pandiella, S.S.; Cantero, D.; Webb, C. Cereal-based fermented foods and beverages. Food Res. Int. 2003, 36, 527–543. [Google Scholar] [CrossRef]
- Jane, M.M.; Lodewyk, K.; Elma, P.; Carlien, P.; Remigio, Z. Characterisation of yeasts isolated from traditional opaque beer beverages brewed in Zimbabwean households. Afr. J. Microbiol. Res. 2015, 9, 549–556. [Google Scholar] [CrossRef]
- Escalante, A.; Giles-Gómez, M.; Hernández, G.; Córdova-Aguilar, M.S.; López-Munguía, A.; Gosset, G.; Bolívar, F. Analysis of bacterial community during the fermentation of pulque, a traditional Mexican alcoholic beverage, using a polyphasic approach. Int. J. Food Microbiol. 2008, 124, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Figueroa, L.M.; Gutiérrez-Uribe, J.A.; Martín-del-Campo, S.T.; Escalante, A. Fermented Foods in Health and Disease Prevention; Frias, J., Martinez-Villaluenga, C., Penas, E., Eds.; Academic Press: London, UK, 2017; ISBN 9780128023099. [Google Scholar]
- Escalante, A.; Soto, D.R.L.; Gutiérrez, J.E.V.; Giles-gómez, M.; Bolívar, F.; López-munguía, A. Pulque, a Traditional Mexican Alcoholic Fermented Beverage: Historical, Microbiological, and Technical Aspects. Front. Microbiol. 2016, 7, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, A.; Arau, M.; Fietto, L.G.; Castro, I.D.M.; Coutrim, X.; Schu, D.; Alves, H.; Casal, M.; Santos, J.D.O.; Henrique, P. Biochemical and Molecular Characterization of Saccharomyces cerevisiae Strains Obtained from Sugar-Cane Juice Fermentations and Their Impact in Cachaca. Appl. Environ. Microbiol. 2008, 74, 693–701. [Google Scholar] [CrossRef] [PubMed]
- Franco, G.R.; Mourão, M.M.; Rosa, C.A. Identification of lactic acid bacteria associted with traditional cachaca fermentation. Braz. J. Microbiol. 2010, 41, 486–492. [Google Scholar]
- Behera, S.K. Kefir and Koumiss Origin. In Fermented Food—Part II: Technological Interventions; CRC Press: Boca Raton, FL, USA, 2017; pp. 401–417. [Google Scholar]
- Rosa, D.D.; Dias, M.M.S.; Grześkowiak, Ł.M.; Reis, S.A.; Conceição, L.L.; do Peluzio, M.C.G. Milk kefir: Nutritional, microbiological and health benefits. Nutr. Res. Rev. 2017, 30, 82–96. [Google Scholar] [CrossRef] [PubMed]
- Mu, Z.; Yang, X.; Yuan, H. Detection and identification of wild yeast in Koumiss. Food Microbiol. 2012, 31, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Azam, M.; Mohsin, M.; Ijaz, H.; Tulain, U.R.; Ashraf, M.A.; Fayyaz, A.; Abadeen, Z.; Kamran, Q. Lactic acid bacteria in traditional fermented Chinese foods. Food Res. Int. 2011, 44, 643–651. [Google Scholar] [CrossRef]
- Shamala, T.R.; Sreekantiah, K.R. Microbiological and biochemical studies on traditional Indian palm wine fermentation. Food Microbiol. 1988, 5, 157–162. [Google Scholar] [CrossRef]
- Choi, S.-H. Characterization of airag collected in Ulaanbaatar, Mongolia with emphasis on isolated lactic acid bacteria. J. Anim. Sci. Technol. 2016, 58, 10. [Google Scholar] [CrossRef] [PubMed]
- Von Rudloff, A.; Mogkatlhe, L.; Mookodi, G. Botswana: Summary of nationwide research, in International Center for Alcohol Policies (ed.), Producers, Sellers, and Drinkers: Studies of Noncommercial Alcohol in Nine Countries. In Global Actions on Harmful Drinking; ICAP Monograph: Washington, DC, USA, 2012; pp. 13–18. [Google Scholar]
- Bahiru, B.; Mehari, T.; Ashenafi, M. Yeast and lactic acid flora of tej, an indigenous Ethiopian honey wine: Variations within and between production units. Food Microbiol. 2006, 23, 277–282. [Google Scholar] [CrossRef] [PubMed]
- Yohannes, T.; Melak, F.; Siraj, K. Preparation and physicochemical analysis of some Ethiopian traditional alcoholic beverages. Afr. J. Food Sci. 2013, 7, 399–403. [Google Scholar] [CrossRef]
- Van Wyk, B.E.; Gericke, N. People’s Plants: A Guide to Useful Plants of Southern Africa; Briza Publications: Johannesburg, South Africa, 2000. [Google Scholar]
- Kubo, R.; Funakawa, S.; Araki, S.; Kitabatake, N. Production of indigenous alcoholic beverages in a rural village of Cameroon. J. Inst. Brewing. 2014, 133–141. [Google Scholar] [CrossRef]
- Miguel, J.; Tulipani, A.S.; Romandini, S.; Bertoli, E.; Battino, M. Contribution of honey in nutrition and human health: A review. Mediterr. J. Nutr. Metab. 2010, 3, 15–23. [Google Scholar] [CrossRef]
- Olaitan, P.B.; Adeleke, O.E. Honey: A reservoir for microorganisms and an inhibitory. Afr. Health Sci. 2007, 7, 159–165. [Google Scholar]
- Bogdanov, S.; Jurendic, T.; Sieber, R. Honey for Nutrition and Health: A Review. Am. J. Coll. Nutr. 2008, 27, 677–689. [Google Scholar] [CrossRef]
- Amoa-Awua, W.K.; Sampson, E.; Tano-Debrah, K. Growth of yeasts, lactic and acetic acid bacteria in palm wine during tapping and fermentation from felled oil palm (Elaeis guineensis) in Ghana. J. Appl. Microbiol. 2007, 102, 599–606. [Google Scholar] [CrossRef] [PubMed]
- Santiago-Urbina, J.A.; Rui’z-Tera’n, F. Microbiology and biochemistry of traditional palm wine produced around the world. Int. Food Res. J. 2014, 21, 1261–1269. [Google Scholar]
- Makhlouf-gafsi, I.; Mokni-ghribi, A.; Bchir, B.; Attia, H.; Blecker, C.; Besbes, S. Physico-chemical properties and amino acid profiles of sap from Tunisian date palm. Sci. Agric. 2016, 73, 85–90. [Google Scholar] [CrossRef]
- Nyanga, L.K.; Nout, M.J.R.; Gadaga, T.H.; Boekhout, T.; Zwietering, M.H. Traditional processing of masau fruits (Ziziphus mauritiana) in Zimbabwe. Ecol. Food Nutr. 2008, 47, 95–107. [Google Scholar] [CrossRef]
- Dellacassa, E.; Trenchs, O.; Fariña, L.; Debernardis, F.; Perez, G.; Boido, E.; Carrau, F. Pineapple (Ananas comosus L. Merr.) wine production in Angola: Characterisation of volatile aroma compounds and yeast native flora. Int. J. Food Microbiol. 2017, 241, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Haruenkit, R. Analysis of sugars and organic acids in pineapple, papaya and star fruit by HPLC using an Aminex HPx-87 H column. J. Anal. Chem. 1988, 22, 11–18. [Google Scholar]
- Makebe, C.W.; Desobgo, Z.S.C.; Nso, E.J. Optimization of the Juice Extraction Process and Investigation on Must Fermentation of Overripe Giant Horn Plantains. Beverages 2017, 3, 19. [Google Scholar] [CrossRef]
- Ketiku, O.A. Chemical composition of unripe (green) and ripe plantain (Musa paradiasca). J. Sci. Food Agric. 1973, 24, 703–707. [Google Scholar] [CrossRef] [PubMed]
- Wilson, P.; David, T.; Sam, B. Microbial and Biochemical Changes Occurring during Production of Traditional Rwandese Banana Beer “Urwagwa”. Ferment. Technol. 2015, 1, 1–5. [Google Scholar] [CrossRef]
- Pareek, S. Nutritional Composition of Fruit Cultivars; Simmonds, M., Preed, V., Eds.; Academic Press: London, UK, 2016; ISBN 978-0-12-408117-8. [Google Scholar]
- Colehour, A.M.; Meadow, J.F.; Liebert, M.A.; Cepon-robins, T.J.; Gildner, T.E.; Urlacher, S.S.; Bohannan, B.J.M.M.; Snodgrass, J.J.; Sugiyama, L.S. Local domestication of lactic acid bacteria via cassava beer fermentation. PeerJ 2014, 2, e479. [Google Scholar] [CrossRef] [PubMed]
- Coulin, P.; Farah, Z.; Assanvo, J.; Spillmann, H.; Puhan, Z. Characterisation of the microflora of attieke, a fermented cassava product, during traditional small-scale preparation. Int. J. Food Microbiol. 2006, 106, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Freire, A.L.; Ramos, C.L.; Schwan, R.F. Microbiological and chemical parameters during cassava based-substrate fermentation using potential starter cultures of lactic acid bacteria and yeast. Food Res. Int. 2015, 76, 787–795. [Google Scholar] [CrossRef] [PubMed]
- Salvador, E.M.; Steenkamp, V.; McCrindle, C.M.E. Production, consumption and nutritional value of cassava (Manihot esculenta, Crantz) in Mozambique: An overview. J. Agric. Biotechnol. Sustain. Dev. 2014, 6, 29–38. [Google Scholar] [CrossRef]
- Afoakwa, E.O.; Budu, A.S.; Asiedu, C.; Chiwona-karltun, L.; Nyirenda, D.B. Nutrition & Food Application of Multivariate Techniques for Characterizing Composition of Starches and Sugars in Six High Yielding CMD Resistant Cassava (Manihot esculenta Crantz) Varieties. Nutr. Food Sci. 2011, 1, 1–5. [Google Scholar] [CrossRef]
- Mariod, A.A.; Abdelwahab, S.I. Sclerocarya birrea (Marula), an African Tree of Nutritional and Medicinal Uses: A Review. Food Rev. Int. 2012, 28, 375–388. [Google Scholar] [CrossRef]
- Dlamini, N.R.; Dube, S. Studies on the physico-chemical, nutritional and microbiological changes during the traditional preparation of Marula wine in Gwanda, Zimbabwe. Nutr. Food Sci. 2008. [Google Scholar] [CrossRef]
- Hal, P.H.; George, P. A review of the proximate composition and nutritional value of Marula (Sclerocarya birrea subsp. caffra). Phytochem. Rev. 2014, 13, 881–892. [Google Scholar] [CrossRef]
- Teshome, D.A.; Rainer, M.; Gerhard, J.N.; Fuchs, S.D.; Bliem, H.R.; Günther, B.K. Chemical compositions of traditional alcoholic beverages and consumers’ characteristics, Ethiopia. Afr. J. Food Sci. 2017, 11, 234–245. [Google Scholar] [CrossRef]
- Shale, K.; Mukamugema, J.; Lues, R.J.; Venter, P.; Mokoena, K.K. Characterisation of selected volatile organic compounds in Rwandan indigenous beer “Urwagwa” by dynamic headspace gas chromatography-mass spectrometry. Afr. J. Biotechnol. 2013, 12, 2990–2996. [Google Scholar] [CrossRef]
- Nwachukwu, I.; Ekaiko, M.U.; Stephen, C. Microbiological quality of palm wine (Elaeis guineensis and Raphia hookeri) sold within Aba Metropolis, Abia State, South Eastern Nigeria. Eur. J. Biotechnol. Genet. Eng. 2016, 3, 38–44. [Google Scholar]
- Steinkraus, H.K. Fermentations in world food processing. Compr. Rev. Food Sci. Food Saf. 2002, 1, 23–32. [Google Scholar] [CrossRef]
- Mapitse, R.; Okatch, H.; Moshoeshoe, E. Analysis of Volatile Compounds in Khadi (an Unrecorded Alcohol Beverage) from Botswana by Gas Chromatography-Flame Ionization Detection (GC-FID). S. Afr. J. Chem. 2014, 67, 184–188. [Google Scholar]
- Misihairabgwi, J.; Cheikhyoussef, A. Traditional fermented foods and beverages of Namibia. J. Ethn. Foods 2017, 1–9. [Google Scholar] [CrossRef]
- Ayogu, T.E. Evaluation of the performance of a yeast isolate from Nigerian palm wine in wine production from pineapple fruits. Bioresour. Technol. 1999, 69, 189–190. [Google Scholar] [CrossRef]
- Roda, A.; Lucini, L.; Torchio, F.; Dordoni, R.; Marco, D.; Faveri, D.; Lambri, M. Metabolite profiling and volatiles of pineapple wine and vinegar obtained from pineapple waste. Food Chem. 2017, 229, 734–742. [Google Scholar] [CrossRef] [PubMed]
- Pino, J.A.; Queris, O. Analysis of volatile compounds of pineapple wine using solid-phase microextraction techniques. Food Chem. 2010, 122, 1241–1246. [Google Scholar] [CrossRef]
- Canonico, L.; Comitini, F.; Ciani, M. Torulaspora delbrueckii contribution in mixed brewing fermentations with different Saccharomyces cerevisiae strains. Int. J. Food Microbiol. 2017, 259, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Marshall, E.; Mejia, D. Traditional Fermented Food and Beverages for Improved Livelihoods; FAO: Rome, Italy, 2011; ISBN 9789251070741. [Google Scholar]
- Dashko, S.; Zhou, N.; Tinta, T.; Sivilotti, P. Use of non-conventional yeast improves the wine aroma profile of Ribolla Gialla. J. Ind. Microbiol. Biotechnol. 2015, 997–1010. [Google Scholar] [CrossRef] [PubMed]
- Achi, O.K. The potential for upgrading traditional fermented foods through biotechnology. Afr. J. Biotechnol. 2005, 4, 375–380. [Google Scholar]
- Lencioni, L.; Romani, C.; Gobbi, M.; Comitini, F.; Ciani, M.; Domizio, P. Controlled mixed fermentation at winery scale using Zygotorulaspora florentina and Saccharomyces cerevisiae. Int. J. Food Microbiol. 2016, 234, 36–44. [Google Scholar] [CrossRef] [PubMed]
- Dashko, S.; Zhou, N.; Compagno, C.; Piskur, J. Why, when, and how did yeast evolve alcoholic fermentation? FEMS Yeast Res. 2014, 14, 826–832. [Google Scholar] [CrossRef] [PubMed]
- Walker, G.; Stewart, G. Saccharomyces cerevisiae in the Production of Fermented Beverages. Beverages 2016, 2, 30. [Google Scholar] [CrossRef]
- Garofalo, C.; Arena, M.P.; Laddomada, B.; Cappello, M.S.; Bleve, G.; Grieco, F.; Beneduce, L.; Berbegal, C.; Spano, G.; Capozzi, V. Starter Cultures for Sparkling Wine. Fermentation 2016, 2, 21. [Google Scholar] [CrossRef]
- Drumonde-Neves, J.; Franco-Duarte, R.; Lima, T.; Schuller, D.; Pais, C. Association between grape yeast communities and the vineyard ecosystems. PLoS ONE 2017, 12, e0169883. [Google Scholar] [CrossRef] [PubMed]
- Barata, A.; Malfeito-Ferreira, M.; Loureiro, V. The microbial ecology of wine grape berries. Int. J. Food Microbiol. 2012, 153, 243–259. [Google Scholar] [CrossRef] [PubMed]
- De Ponzzes-Gomes, C.M.P.B.S.; de Mélo, D.L.F.M.; Santana, C.A.; Pereira, G.E.; Mendonça, M.O.C.; Gomes, F.C.O.; Oliveira, E.S.; Barbosa, A.M.; Trindade, R.C.; Rosa, C.A. Saccharomyces cerevisiae and non-Saccharomyces yeasts in grape varieties of the São Francisco Valley. Braz. J. Microbiol. 2014, 45, 411–416. [Google Scholar] [CrossRef] [PubMed]
- Swiegers, J.H.; Kievit, R.L.; Siebert, T.; Lattey, K.A.; Bramley, B.R.; Francis, I.L.; King, E.S.; Pretorius, I.S. The influence of yeast on the aroma of Sauvignon Blanc wine. Food Microbiol. 2009, 26, 204–211. [Google Scholar] [CrossRef] [PubMed]
- Schifferdecker, A.J.; Dashko, S.; Ishchuk, O.P.; Piškur, J. The wine and beer yeast Dekkera bruxellensis. Yeast 2014, 31, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Varela, C. The impact of non-Saccharomyces yeasts in the production of alcoholic beverages. Appl. Microbiol. Biotechnol. 2016, 9861–9874. [Google Scholar] [CrossRef] [PubMed]
- Pretorius, I.S. Tailoring wine yeast for the new millennium: Novel approaches to the ancient art of winemaking. Yeast 2000, 16, 675–729. [Google Scholar] [CrossRef]
- Hansen, E.H.; Nissen, P.; Sommer, P.; Nielsen, J.C.; Arneborg, N.; Di Maro, E.; Ercolini, D.; Coppola, S.; Pretorius, I.S. The effect of oxygen on the survival of non-Saccharomyces yeasts during mixed culture fermentations of grape juice with Saccharomyces cerevisiae. J. Appl. Microbiol. 2001, 91, 541–547. [Google Scholar] [CrossRef]
- Di Maro, E.; Ercolini, D.; Coppola, S. Yeast dynamics during spontaneous wine fermentation of the Catalanesca grape. Int. J. Food Microbiol. 2007, 117, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Hagman, A.; Sall, T.; Compagno, C.; Piskur, J. Yeast “Make-Accumulate-Consume” Life Strategy Evolved as a Multi-Step Process That Predates the Whole Genome Duplication. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Zhou, N.; Swamy, K.B.S.; Leu, J.Y.; McDonald, M.J.; Galafassi, S.; Compagno, C.; Piškur, J. Coevolution with bacteria drives the evolution of aerobic fermentation in Lachancea kluyveri. PLoS ONE 2017, 12. [Google Scholar] [CrossRef] [PubMed]
- De Deken, R.H. The Crabtree Effect: A Regulatory System in Yeast. J. Gen. Microbiol. 1966, 44, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Stringini, M.; Comitini, F.; Taccari, M.; Ciani, M. Yeast diversity during tapping and fermentation of palm wine from Cameroon. Food Microbiol. 2009, 26, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.J.; Choi, Y.R.; Lee, S.Y.; Park, J.T.; Shim, J.H.; Park, K.H.; Kim, J.W. Screening wild yeast strains for alcohol fermentation from various fruits. Mycobiology 2011, 39, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Lleixà, J.; Manzano, M.; Mas, A.; del Portillo, M.C. Saccharomyces and non-Saccharomyces competition during microvinification under different sugar and nitrogen conditions. Front. Microbiol. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Fei, Q.; Zhang, Y.; Contreras, L.M.; Utturkar, S.M.; Brown, S.D.; Himmel, M.E.; Zhang, M. Zymomonas mobilis as a model system for production of biofuels and biochemicals. Microb. Biotechnol. 2016, 9, 699–717. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Wu, B.; Qin, H.; Ruan, Z.; Tan, F.; Wang, J.; Shui, Z.; Dai, L.; Zhu, Q.; Pan, K.; et al. Zymomonas mobilis: A novel platform for future biorefineries. Biotechnol. Biofuels 2014, 7, 101. [Google Scholar] [CrossRef] [PubMed]
- Obire, O. Activity of Zymomonas species in palm-sap obtained from three areas in Edo State, Nigeria. J. Appl. Sci. Environ. Manag. 2005, 9, 25–30. [Google Scholar]
- Lyumugabe, F.; Gros, J.; Nzungize, J.; Bajyana, E.; Thonart, P. Characteristics of African traditional beers brewed with sorghum malt: A review. Biotechnol. Agron. Soc. Environ. 2012, 16, 509–530. [Google Scholar]
- Lütke-Eversloh, T.; Bahl, H. Metabolic engineering of Clostridium acetobutylicum: Recent advances to improve butanol production. Curr. Opin. Biotechnol. 2011, 22, 634–647. [Google Scholar] [CrossRef] [PubMed]
- Sreekumar, S.; Baer, Z.C.; Pazhamalai, A.; Gunbas, G.; Grippo, A.; Blanch, H.W.; Clark, D.S.; Toste, F.D. Production of an acetone-butanol-ethanol mixture from Clostridium acetobutylicum and its conversion to high-value biofuels. Nat. Protoc. 2015, 10, 528–537. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Cao, C.; Wang, Y.; Li, C.; Wu, M.; Chen, Y.; Zhang, C.; Pei, H.; Xiao, D. Effect of the inactivation of lactate dehydrogenase, ethanol dehydrogenase, and phosphotransacetylase on 2,3-butanediol production in Klebsiella pneumoniae strain. Biotechnol. Biofuels 2014, 7, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Tamang, J.P.; Shin, D.H.; Jung, S.J.; Chae, S.W. Functional properties of microorganisms in fermented foods. Front. Microbiol. 2016, 7, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Jespersen, L. Occurrence and taxonomic characteristics of strains of Saccharomyces cerevisiae predominant in African indigenous fermented foods and beverages. FEMS Yeast Res. 2003, 3, 191–200. [Google Scholar] [CrossRef]
- Barukčić, I.; Gracin, L.; Jambrak, A.R.; Božanić, R. Comparison of chemical, rheological and sensory properties of kefir produced by kefir grains and commercial kefir starter. Mljekarstvo 2017, 67, 163–176. [Google Scholar] [CrossRef]
- Muyanja, C.M.B.K.; Narvhus, J.A.; Treimo, J.; Langsrud, T. Isolation, characterisation and identification of lactic acid bacteria from bushera: A Ugandan traditional fermented beverage. Food Microbiol. 2003, 80, 201–210. [Google Scholar] [CrossRef]
- Kamaruzaman, M.R.B. Isolation of Microorganism from oil Palm Sap; University of Malaysia Pahang: Gambang, Malaysia, 2011. [Google Scholar]
- Hittinger, C.T. Saccharomyces diversity and evolution: A budding model genus. Trends Genet. 2013, 29, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Jeffries, T.; Hittinger, C.T.; Rokas, A.; Bai, F.; Boekhout, T.; Gonc, P.; Jeffries, T.W.; Libkind, D.; Sampaio, P.; Kurtzman, C.P.; Rosa, C.A. Genomics and the making of yeast biodiversity. Genet. Dev. 2015, 35, 100–109. [Google Scholar] [CrossRef]
- Nwakakanma, C.; Unachukwu, N.M.; Onah, P.; Engwa, A.G. Isolation and sensory evaluation of Saccharomyces cerevisiae from palm wine (Elaeis guinneensis) gotten from different sites in Enugu. Eur. J. Biomed. Pharm. Sci. 2015, 2, 19–26. [Google Scholar]
- Shale, K.; Mukamugema, J.; Lues, R.J.; Venter, P. Possible microbial and biochemical contaminants of an indigenous banana beer “Urwagwa”: A mini review. Afr. J. Food Sci. Rev. 2014, 8, 376–389. [Google Scholar] [CrossRef]
- Fleet, G.H. Yeasts in foods and beverages: Impact on product quality and safety. Food Biotechnol. 2007, 18, 170–175. [Google Scholar] [CrossRef] [PubMed]
- Mpofu, A.; Pretorious, E.E.; Pohl, C.H.; Zvauya, R. Identification of yeasts isolated from Mukumbi, a Zimbabwe traditional wine. J. Sustain. Dev. Africa 2008, 10, 88–102. [Google Scholar]
- Iwuoha, C.I.; Eke, O.S. Nigerian indigenous fermented foods: Their traditional process operation, inherent problems, improvements and current status. Food Res. Int. 1996, 29, 527–540. [Google Scholar] [CrossRef]
- Brock, T.D. The study of microorganisms in situ: Progress and problems. In Symposium of the Society for General Microbiology; Cambridge University Press: New York, NY, USA, 1987; Volume 41, pp. 1–17. [Google Scholar]
- Wade, W. Unculturable bacteria: The uncharacterized organisms that cause oral infections. J. R. Soc. Med. 2002, 95, 81–83. [Google Scholar] [PubMed]
- Styger, G.; Prior, B.; Bauer, F.F. Wine flavor and aroma. J. Ind. Microbiol. Biotechnol. 2011, 38, 1145–1159. [Google Scholar] [CrossRef] [PubMed]
- Kong, Q.X.; Zhang, A.L.; Cao, L.M.; Chen, X. Over-expressing GLT1 in a gpd2∆ mutant of Saccharomyces cerevisiae to improve ethanol production. Appl. Microbiol. Biotechnol. 2007, 75, 1361–1366. [Google Scholar] [CrossRef] [PubMed]
- Navarrete, C.; Nielsen, J.; Siewers, V. Enhanced ethanol production and reduced glycerol formation in fps1∆ mutants of Saccharomyces cerevisiae engineered for improved redox balancing. AMB Express 2014, 4, 2–8. [Google Scholar] [CrossRef] [PubMed]
- Dussap, C.G.; Poughon, L. Microbiology of Alcoholic Fermentation. In Current Developments in Biotechnology and Bioengineering: Food and Beverages Industry; Marinakis, K., Ed.; Elsevier B.V.: Amsterdam, The Netherlands, 2016; pp. 263–279. ISBN 978-0-444-63666-9. [Google Scholar]
- Carrau, F.M.; Medina, K.; Farina, L.; Boido, E.; Henschke, P.A.; Dellacassa, E. Production of fermentation aroma compounds by Saccharomyces cerevisiae wine yeasts: Effects of yeast assimilable nitrogen on two model strains. FEMS Yeast Res. 2008, 8, 1196–1207. [Google Scholar] [CrossRef] [PubMed]
- Pino, J.A.; Queris, O. Analysis of volatile compounds of mango wine. Food Chem. 2011, 125, 1141–1146. [Google Scholar] [CrossRef]
- Benés, I.; Furdíková, K.; Šmogrovičová, D. Influence of Saccharomyces cerevisiae strain on the profile of volatile organic compounds of blossom honey mead. Czech. J. Food Sci. 2015, 33, 334–339. [Google Scholar] [CrossRef]
- Michel, M.; Meier-Dörnberg, T.; Jacob, F.; Methner, F.J.; Wagner, R.S.; Hutzler, M. Review: Pure non-Saccharomyces starter cultures for beer fermentation with a focus on secondary metabolites and practical applications. J. Inst. Brew. 2016, 122, 569–587. [Google Scholar] [CrossRef]
- Karamoko, D.; Djeni, N.T.; N’guessan, K.F.; Bouatenin, K.M.J.P.; Dje, K.M. The biochemical and microbiological quality of palm wine samples produced at different periods during tapping and changes which occured during their storage. Food Control 2012, 26, 504–511. [Google Scholar] [CrossRef]
- Magalhães, K.T.; Dragone, G.; De Melo Pereira, G.V.; Oliveira, J.M.; Domingues, L.; Teixeira, J.A.; E Silva, J.B.A.; Schwan, R.F. Comparative study of the biochemical changes and volatile compound formations during the production of novel whey-based kefir beverages and traditional milk kefir. Food Chem. 2011, 126, 249–253. [Google Scholar] [CrossRef]
- Arslan, M.M.; Zeren, C.; Aydin, Z.; Akcan, R.; Dokuyucu, R.; Keten, A.; Cekin, N. Journal of Forensic and Legal Medicine Analysis of methanol and its derivatives in illegally produced alcoholic beverages. J. Forensic Leg. Med. 2015, 33, 56–60. [Google Scholar] [CrossRef] [PubMed]
- Aloys, N.; Angeline, N. Traditional fermented foods and beverages in Burundi. Food Res. Int. 2009, 42, 588–594. [Google Scholar] [CrossRef]
- Manel, Z.; Sana, M.; Nedia, K.; Moktar, H.; Ali, F. Microbiological analysis and screening of lactic acid bacteria from Tunisian date palm sap. Afr. J. Microbiol. Res. 2011, 5, 2929–2935. [Google Scholar] [CrossRef]
- Regodón Mateos, J.A.; Pérez-Nevado, F.; Ramírez Fernández, M. Influence of Saccharomyces cerevisiae yeast strain on the major volatile compounds of wine. Enzyme Microb. Technol. 2006, 40, 151–157. [Google Scholar] [CrossRef]
- Naydenova, V.; Iliev, V.; Kaneva, M.; Kostov, G.; Koprinkova-Hristova, P.; Popova, S. Modeling of alcohol fermentation in brewing—Carbonyl compounds synthesis and reduction. In Proceedings of the 28th European Conference on Modelling and Simulation (ECMS 2014), Brescia, Italy, 27–30 May 2014; pp. 279–284. [Google Scholar]
- Pereira, A.P.; Oliveira, J.M.; Mendes-Ferreira, A.; Estevinho, L.M.; Mendes-Faia, A. Mead and Other Fermented Beverages. In Current Developments in Biotechnology and Bioengineering: Food and Beverages Industry; Marinakis, K., Ed.; Elsevier B.V.: Amsterdam, The Netherlands, 2016; pp. 407–434. ISBN 9780444636775. [Google Scholar]
- Cao, L.; Kong, Q.; Zhang, A.; Chen, X. Overexpression of SYM1 in a gpdDelta mutant of Saccharomyces cerevisiae with modified ammonium assimilation for optimization of ethanol production. J. Taiwan Inst. Chem. Eng. 2010, 41, 2–7. [Google Scholar] [CrossRef]
- Escudero, A.; Campo, E.; Fariña, L.; Cacho, J.; Ferreira, V. Analytical characterization of the aroma of five premium red wines. Insights into the role of odor families and the concept of fruitiness of wines. J. Agric. Food Chem. 2007, 55, 4501–4510. [Google Scholar] [CrossRef] [PubMed]
- Okafor, N. Microbiology and biochemistry of oil-palm wine. Adv. Appl. Microbiol. 1978, 24, 237–256. [Google Scholar]
- Okagbue, R.N.; Siwela, M. Yeasts and related microorganisms isolated from ripe marula fruits (Sclerocarya caffra) in Zimbabwe. S. Afr. J. Bot. 2002, 98, 551–552. [Google Scholar]
- Snowdon, J.A.; Cliver, D.O. Microorganisms in honey. Int. J. Food Microbiol. 1996, 31, 1–26. [Google Scholar] [CrossRef]
- Lasekan, O.; Buettner, A.; Christlbauer, M. Investigation of important odorants of palm wine (Elaeis guineensis). Food Chem. 2007, 105, 15–23. [Google Scholar] [CrossRef]
- Mwesigye, P.K.; Okurut, T.O. A Survey of the Production and Consumption of Traditional Alcoholic Beverages in Uganda. Process Biochem. 1995, 30, 497–501. [Google Scholar] [CrossRef]
- Shanel, K.; Matemu, A. Microbial Quality of Traditional Banana Alcoholic Beverages in Arusha, Tanzania. Food Sci. Qual. Manag. 2015, 38, 28–39. [Google Scholar]
- Roger, D.D.; Daoudou, B.; James, B.; Etoa, F. Artisanal production of “kuri” an honey made alcoholic beverage from Adamaoua, Cameroon. Glob. J. Sci. Res. 2014, 2, 65–70. [Google Scholar]
- Pereira, A.P.; Dias, T.; Andrade, J.; Ramalhosa, E.; Estevinho, L.M. Mead production: Selection and characterization assays of Saccharomyces cerevisiae strains. Food Chem. Toxicol. 2009, 47, 2057–2063. [Google Scholar] [CrossRef] [PubMed]
- Wyk, B. Van The potential of South African plants in the development of new food and beverage products. S. Afr. J. Bot. 2011, 77, 857–868. [Google Scholar] [CrossRef]
- World Health Organization. Global Status Report on Alcohol; World Health Organization: Geneva, Switzerland, 2004; pp. 4–6. [Google Scholar]
- Tredgold, M.H. Food Plants of Zimbabwe: With Old and New Ways of Preparation; Mambo Press: Gweru, Zimbabwe, 1986. [Google Scholar]
- Simatende, P. Microbial Ecology and Diversity of Swazi Traditional Fermented Foods; University of KwaZulu-Natal: Pietermaritzburg, South Africa, 2016. [Google Scholar]
- Sanni, A.; Lonner, C. Identification of yeasts isolated from Nigerian traditional alcoholic beverages. Food Microbiol. 1993, 10, 517–523. [Google Scholar] [CrossRef]
- Ramos, C.L.; Sousa, E.S.O.D.; Ribeiro, J.; Almeida, T.M.M.; Santos, C.C.A.D.A.; Abegg, M.A.; Schwan, R.F. Microbiological and chemical characteristics of tarubá, an indigenous beverage produced from solid cassava fermentation. Food Microbiol. 2015, 49, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Kouakou, J.; Nanga, S.N.; Plagne-Ismail, C.; Pali, A.M.; Ognakossan, K.E. Cassava Production and Processing; Cameron: London, UK, 2016. [Google Scholar]
- Cristina, C.; Santos, A.; De Almeida, E.G.; Vinícius, G.; De Melo, P.; Schwan, R.F. Microbiological and physicochemical characterisation of caxiri, an alcoholic beverage produced by the indigenous Juruna people of Brazil. Int. J. Food Microbiol. 2012, 156, 112–121. [Google Scholar] [CrossRef]
- Eze, V.C.; Eleke, O.I.; Omeh, Y.S. Microbiological and nutritional qualities of burukutu sold in mammy. Am. Food J. Nutr. 2011, 1, 141–146. [Google Scholar] [CrossRef]





| Substrate | Total Sugar Content * | Sugars | pH | Nitrogen Sources | References |
|---|---|---|---|---|---|
| Honey | 79.5% | Glucose (38.19%), fructose (31.28%), sucrose (1.31%), maltose, isomaltose, trehalose, trisaccharideserlose, raffinose, melezitose, tetrasaccharides and pentasaccharides | 3.4–6.1 | Proline, glutamic acid, aspartic acid, glutamine, histidine, glycine, arginine, tryptophan, and cysteine | [31,32,33] |
| Palm sap (Palmae family) | 10–18% | Sucrose (36%), glucose (33%), fructose, cellobiose, dextran, maltose, xylose, hamnose, arabinose and galacturonic acid | 7–7.4 | Valine, threonine, lysine and phenylalanine | [34,35,36] |
| Masau (Ziziphus mauritiana) | 13.7% | Glucose (6.7%) and fructose (6.8%) | 5.6–6.6 | [37] | |
| Pineapple (Ananas comosus) | 7.98% | Sucrose, fructose and glucose | 3.5 | [38,39] | |
| Plantain (Musa paradisiaca) | 20–27% | Glucose, fructose and sucrose | 4.26 | Arginine, aspartic acid, glutamic acid and methionine | [40,41] |
| Banana (Musa acuminata) | 14.20–20.18% | Glucose, fructose and sucrose | 4.78 | Aspartic acid, histidine, leucine and valine | [42,43] |
| Cassava (Manihot esculenta) | 4.04–18.47% | Sucrose (1.98–15.40%), maltose, fructose, and glucose | 6.2–6.9 | Valine, leucine, isoleucine, phenylalanine, tryptophan, methionine, threonine, histidine and lysine | [44,45,46,47,48] |
| Marula juice (Sclerocarya birrea) | 8.2% | Sucrose (5.9%), fructose and glucose | 4.10 | Methionine, cysteine, leucine, phenylalanine, lysine, and threonine | [49,50,51] |
| Alcoholic Beverage | Raw Materials/Substrate | Sensory Properties | Nature | Fermentative Microbes | Secondary Metabolites | Country | References |
|---|---|---|---|---|---|---|---|
| Tej | Honey | Sweet | Effervescent, yellow andcloudy | Saccharomyces cerevisiae, Kluyveromyces bulgaricus, Debaromyces phaffi, K. veronae, Zygosaccharomyces rouxii, Hanensula subpeliculosa, S. norbensis, K. vanudenii, Endomycopsis burtonii, Lactobacillus spp., Streptococcus spp., Leuconostoc spp., and Pediococcus spp. | Ethanol, methanol, esters and organic acids | Ethiopia | [11,27,52] |
| Urwagwa | Banana pulp | Pungent, fruity, herbaceous and astringent/acidic taste | Cloudy | Saccharomyces cerevisiae | Ethanol, 1-propanol, 2-hexanol, acetic acid, 5-hexanoic acid, benzoic acid, propanoic acid, formic acid, ethyl acetate, butanoic acid, ethyl ester, 1-Butanol 3-methyl-ethyl ester and hexanoic acid ethyl ester | Rwanda, Burundi, Uganda, Tanzania and Kenya | [53] |
| Palm wine | Palm tree sap | Sour, heavy and astringent taste | Whitish, milk-white opalescent liquid | Staphylococcus spp., Micrococcus spp., Serratia spp., Bacillus spp., Streptococcus spp., Saccharomyces cerevisae, S. pombe, Candida tropicalis, C. krusei, Candida spp., Pichia spp., S. chevalieri, Zymomonas mobilis, Hanseniaspora guilliermondii, H. uvarum, Lactobacillus plantarum, Leuconostoc mesenteroides, L. nagelii, L. sucicola, Acetobacter pasteurianus, A. indonesiensis and Gluconobacter spp. | Lactic acid, citric acid, tartaric acids, ethanol and acetic acid, esters, carbonyls, higher alcohols, phenols, sulphur compounds, terpenes, hydrocarbons, acetals, nitrogen compounds and lactone | Nigeria, Bukina Faso and Cameroon | [34,35,54,55] |
| Khadi | Grewia flava, Grewia occidentalis, Grewia flavascens, Kedrostis hirtella and Khadia acutipetala | Sweet | Reddish color | No literature on fermentative microbes | Ethanol, 2-methyl-1-propanol, 2/3-methyl-1-butanol, ethyl lactate and ethyl acetate | Botswana | [56] |
| Mukumbi | Marula fruits | Thick creamy liquor | Yellowish-brown | Saccharomyces cerevisiae, Pichia anomala, P. guilliermondii, Candida tropicalis, and C. intermedia | Ethanol | Namibia, Botswana, Swaziland, Zimbabwe and Zambia | [9,57] |
| Muchema | Palm tree sap | Sour | Brownish to colorless | No literature on the microbial diversity | Ethanol | Botswana and Namibia | [57] |
| Pineapple wine | Pineapples | Fruity, sweetand cream/fatty notes | Golden yellowish | Hanseniaspora guilliermondii, Pichia anomala, Meyerozyma guilliermondii, H. uvarum, Wickerhamomyces anomalus and H. opuntiae | Acetic acid, ethyl acetate, isobutanol, 3-methyl-1-butanol, 2-methyl-1-butanol, ethyl 2-methylpropanoate, 3-methyl-1-butyl acetate, ethyl hexanoate, methyl benzoate, 2-phenylethanol, methyl 2-methylheptanoate, ethyl benzoate, diethyl succinate, ethyl octanoate, ethyl phenylacetate, 2-phenylethyl acetate, ethyl 9-decenoate and ethyl decanoate | Angola | [38,58,59,60] |
| Kachasu | Masau fruits (Ziziphus mauritiana), Adansonia digitate and Tamarindus indica | Sour, acidic | Clear distillate | Saccharomyces cerevisiae, Issatchenkia orientalis, Pichia fabianii, Aureobasidium pullulans, Lactobacillus agilis, L. minor, L. confusus, L. fructosus, L. bifermentans, L. divergens, L. fermentum, L. hilgardii and Streptococcus spp. | Isoamyl alcohol, isobutanol, methanol, acetaldehyde, acetone, ethyl acetate, and furfurals | Zimbabwe | [5,9] |
| Metabolite Class | Examples of Compounds | Contribution to Flavor and Aroma | References |
|---|---|---|---|
| Organic acids | Succinic acid, acetic acid, lactic acid, citric acid | These compounds contribute to the astringency of fermented beverages. The presence of some acids, notably lactic acid, can indicate undesirable bacterial spoilage. | [72,73,117] |
| Higher alcohols | Isoamyl alcohol, phenylethanol, isopropanol | These impart desirable flavor and aromas to fermented beverages but only within certain limits | [114] |
| Carbonyl compounds | Acetaldehyde, aldehydes, ketones | Above its flavor threshold in beverages, this compound can impart a “grassy” or “green apple” (related to acetaldehyde) flavor but this can be removed by secondary yeast fermentation during conditioning. | [72,76] |
| Vicinal diketones | Diacetyl, pentane-2,3-dione | Diacetyl in most beverages is undesirable, imparting a rancid-butter or “butterscotch” flavor. | [118,119,120] |
| Polyols | Glycerol | This compound is produced during normal yeast metabolism or when yeasts are confronted with osmotic stress. Glycerol contributes desirable viscosity to fermented beverages, notably wines, the body of the wine per se. | [21,76,121] |
| Sulfur compounds | Hydrogen sulphide, dimethyl sulphide, sulphur dioxide, Thiols | These are important beverage flavor and aroma compounds. For example, in beer, dimethyl sulphide (DMS) in low concentration is a desirable attribute of lagers, but higher concentration imparts off-flavors. | [67,122] |
| Esters | Ethyl acetate | Associated with a fruity aroma and floral flavors and aromas to fermented beverages. | [79,80] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Motlhanka, K.; Zhou, N.; Lebani, K. Microbial and Chemical Diversity of Traditional Non-Cereal Based Alcoholic Beverages of Sub-Saharan Africa. Beverages 2018, 4, 36. https://doi.org/10.3390/beverages4020036
Motlhanka K, Zhou N, Lebani K. Microbial and Chemical Diversity of Traditional Non-Cereal Based Alcoholic Beverages of Sub-Saharan Africa. Beverages. 2018; 4(2):36. https://doi.org/10.3390/beverages4020036
Chicago/Turabian StyleMotlhanka, Koketso, Nerve Zhou, and Kebaneilwe Lebani. 2018. "Microbial and Chemical Diversity of Traditional Non-Cereal Based Alcoholic Beverages of Sub-Saharan Africa" Beverages 4, no. 2: 36. https://doi.org/10.3390/beverages4020036
APA StyleMotlhanka, K., Zhou, N., & Lebani, K. (2018). Microbial and Chemical Diversity of Traditional Non-Cereal Based Alcoholic Beverages of Sub-Saharan Africa. Beverages, 4(2), 36. https://doi.org/10.3390/beverages4020036





