Discrimination between Arabica and Robusta Coffees Using Hydrosoluble Compounds: Is the Efficiency of the Parameters Dependent on the Roast Degree?
Abstract
:1. Introduction
2. Experimental Section
2.1. Coffee Samples
2.2. Standards and Solvents
2.3. Color Evaluation
2.4. Determination of Nicotinic Acid, Caffeine, Trigonelline, and 5-CQA
2.5. Statistical Analysis
3. Results and Discussion
3.1. Influence of Roast Degree on the Composition and on Color of Arabica and Robusta Coffees
| Values | Species\Roast degree | Light ** | Medium ** | Dark ** |
|---|---|---|---|---|
| L* | A 100% | 28.0 A,d ± 0.4 | 15.9 B,c ± 0.4 | 13.0 C,b ± 0.4 |
| R 20% | 31.0 A,c ± 0.6 | 17.9 B,b ± 0.4 | 13.4 C,b ± 0.3 | |
| R 30% | 29.3 A,bc ± 0.5 | 17.9 B,b ± 0.4 | 13.5 C,b ± 0.2 | |
| R 50% | 31.3 A,b ± 0.7 | 18.4 B,b ± 0.3 | 13.8 C,b ± 0.4 | |
| R 100% | 37.6 A,a ± 0.3 | 24.7 B,a ± 0.4 | 17.4 C,a ± 0.4 | |
| h° | A 100% | 57.4 A,c ± 0.1 | 45.1 B,d ± 0.4 | 41.6 C,d ± 1.0 |
| R 20% | 58.0 A,c ± 0.3 | 47.8 B,c ± 0.2 | 41.2 C,d ± 0.3 | |
| R 30% | 58.6 A,bc ± 1.2 | 48.3 B,c ± 0.3 | 43.0 C,c ± 0.6 | |
| R 50% | 59.8 A,b ± 0.2 | 50.7 B,b ± 0.3 | 44.9 C,b ± 0.2 | |
| R 100% | 62.7 A,a ± 0.2 | 56.0 B,a ± 0.2 | 48.6 C,a ± 0.2 |
| Compounds | Species\Roast degree | Light ** | Medium ** | Dark ** | ||||
|---|---|---|---|---|---|---|---|---|
| 5-CQA | A 100% | 1.786 A,c ± 0.056 | 0.475 B,a ± 0.021 | 0.263 C,a ± 0.010 | ||||
| R 20% | 1.854 A,bc ± 0.040 | 0.499 B,a ± 0.011 | 0.249 C,a ± 0.002 | |||||
| R 30% | 1.871 A,bc ± 0.003 | 0.511 B,a ± 0.016 | 0.211 C,a ± 0.002 | |||||
| R 50% | 1.953 A,ab ± 0.078 | 0.574 B,a ± 0.013 | 0.172 C,b ± 0.001 | |||||
| R 100% | 2.015 A,a ± 0.081 | 0.518 B,a ± 0.029 | 0.094 C,b ± 0.000 | |||||
| Trigonelline | A 100% | 0.928 A,a ± 0.005 | 0.489 B,a ± 0.007 | 0.297 C,a ± 0.010 | ||||
| R 20% | 0.894 A,b ± 0.007 | 0.462 B,a ± 0.009 | 0.262 C,b ± 0.001 | |||||
| R 30% | 0.863 A,b ± 0.004 | 0.458 B,a ± 0.022 | 0.239 C,bc ± 0.005 | |||||
| R 50% | 0.865 A,c ± 0.032 | 0.456 B,a ± 0.005 | 0.206 C,c ± 0.002 | |||||
| R 100% | 0.683 A,d ± 0.011 | 0.380 B,b ± 0.015 | 0.119 C,d ± 0.003 | |||||
| Nicotinic acid | A 100% | 0.091 A,a ± 0.001 | 0.012 B,a ± 0.000 | 0.010 B,a ± 0.001 | ||||
| R 20% | 0.072 A,b ± 0.001 | 0.011 B,a ± 0.002 | 0.010 B,a ± 0.000 | |||||
| R 30% | 0.072 A,b ± 0.004 | 0.011 B,a ± 0.000 | 0.008 B,a ± 0.001 | |||||
| R 50% | 0.063 A,c ± 0.001 | 0.012 B,a ± 0.000 | 0.007 C,a ± 0.000 | |||||
| R 100% | 0.032 A,d ± 0.000 | 0.009 B,a ± 0.000 | 0.000 C,b ± 0.000 | |||||
| Compound | Species\Roast degree | Light | Medium | Dark | Mean values ** | |||
| Caffeine | A 100% | 1.33 ± 0.02 | 1.35 ± 0.05 | 1.36 ± 0.06 | 1.35 d ± 0.02 | |||
| R 20% | 1.53 ± 0.05 | 1.51 ± 0.00 | 1.52 ± 0.06 | 1.52 c ± 0.02 | ||||
| R 30% | 1.62 ± 0.06 | 1.55 ± 0.07 | 1.57 ± 0.01 | 1.58 c ± 0.04 | ||||
| R 50% | 1.82 ± 0.11 | 1.79 ± 0.04 | 1.78 ± 0.06 | 1.79 b ± 0.02 | ||||
| R 100% | 2.25 ± 0.13 | 2.10 ± 0.14 | 2.20 ± 0.05 | 2.18 a ± 0.08 | ||||
| Mean value ** | 1.71 A ± 0.35 | 1.66 A ± 0.30 | 1.69 A ± 0.32 | |||||
3.2. Discrimination of the Coffee Species
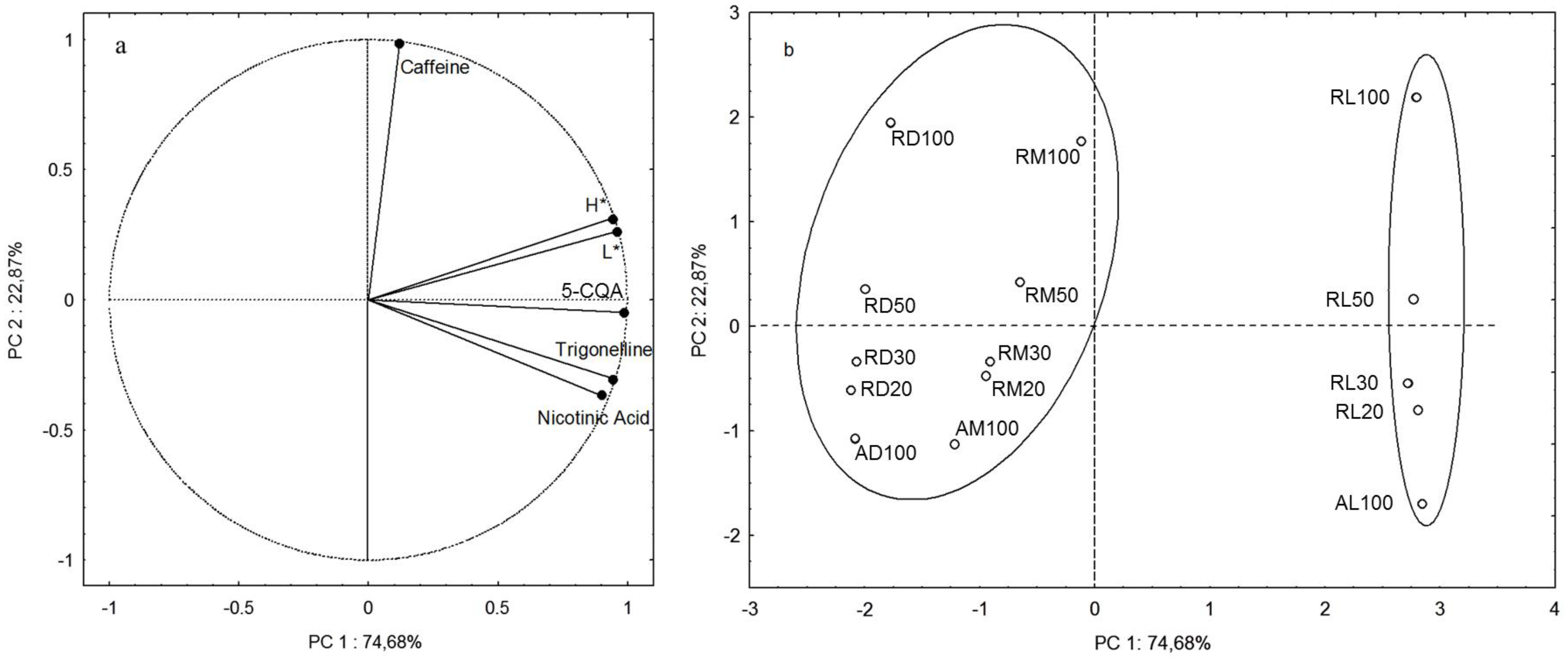
3.3. The Use of Combined Parameters
| Sum | Species\Roast Degree | Light ** | Medium ** | Dark ** |
| A 100% | 1.02 A,a ± 0.01 | 0.50 B,a ± 0.01 | 0.31 C,a ± 0.01 | |
| R 20% | 0.97 A,b ± 0.01 | 0.47 B,ab ± 0.01 | 0.27 C,ab ± 0.00 | |
| R 30% | 0.93 A,b ± 0.01 | 0.47 B,b ± 0.02 | 0.25 C,bc ± 0.01 | |
| R 50% | 0.87 A,c ± 0.03 | 0.47 B,b ± 0.01 | 0.21 C,c ± 0.00 | |
| R 100% | 0.72 A,d ± 0.01 | 0.39 B,c ± 0.02 | 0.12 C,d ± 0.00 | |
| Ratio | A 100% | 1.34 A,a ± 0.02 | 0.35 B,a ± 0.00 | 0.19 C,a ± 0.00 |
| R 20% | 1.21 A,b ± 0.01 | 0.33 B,a ± 0.01 | 0.16 C,ab ± 0.01 | |
| R 30% | 1.16 A,c ± 0.04 | 0.33 B,a ± 0.00 | 0.13 C,bc ± 0.00 | |
| R 50% | 1.08 A,d ± 0.02 | 0.32 B,a ± 0.01 | 0.10 C,c ± 0.00 | |
| R 100% | 0.90 A,e ± 0.01 | 0.25 B,a ± 0.00 | 0.04 C,d ± 0.00 |
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- ICO-International Coffee Organization. Exporting Countries: Total Production. Available online: http://www.ico.org/prices/po.htm (accessed on 29 June 2015).
- Kemsley, E.K.; Ruault, S.; Wilson, R.H. Discrimination between Coffea arabica and Coffea canephora variant robusta beans using infrared spectroscopy. Food Chem. 1995, 54, 321–326. [Google Scholar] [CrossRef]
- Martín, M.J.; González, A.G.; Pablos, F. Discrimination between arabica and robusta green coffee varieties according to their chemical composition. Talanta 1998, 46, 1259–1264. [Google Scholar] [CrossRef]
- Casal, S.; Oliveira, M.B.; Ferreira, M.A. Development of an HPLC/diode-array detector method for simultaneous determination of trigonelline, nicotinic acid and caffeine in coffee. J. Liq. Chrom. Relat. Tech. 1998, 21, 3187–3195. [Google Scholar] [CrossRef]
- Casal, S.; Oliveira, M.B.; Alves, M.R.; Ferreira, M.A. Discriminate analysis of roasted coffee varieties for trigonelline, nicotinic acid, and caffeine content. J. Agric. Food Chem. 2000, 48, 3420–3424. [Google Scholar] [CrossRef] [PubMed]
- Ky, C.L.; Louarn, J.; Dussert, S.; Guyot, B.; Hamon, S.; Noirot, M. Caffeine, trigonelline, chlorogenic acid and sucrose diversity in wild Coffea arabica L. and C. canephora P. accessions. Food Chem. 2001, 75, 223–230. [Google Scholar] [CrossRef]
- González, A.G.; Pablos, F.; Martín, M.J.; León-Camacho, M.; Valdenebro, M.S. HPLC analysis of tocopherols and triglycerides in coffee and their use as authentication parameters. Food Chem. 2001, 73, 93–101. [Google Scholar] [CrossRef]
- Martínez, L.L.; López-de-Alba, P.L.; García-Campos, R.; León-Rodríguez, L.M. Simultaneous determination of methylxanthines in coffees and teas by UV-VIS spectrophotometry and partial least squares. Anal. Chim. Acta 2003, 493, 83–94. [Google Scholar] [CrossRef]
- Daglia, M.; Racchi, M.; Papetti, A.; Lanni, C.; Govoni, S.; Gazzani, G. In vitro and ex vivo antihydroxyl radical activity of green and roasted coffee. J. Agric. Food Chem. 2004, 52, 1700–1704. [Google Scholar] [CrossRef] [PubMed]
- Rubayiza, A.B.; Meurens, M. Chemical discrimination of arabica and robusta coffees by Fourier Transform Raman Spectroscopy. J. Agric. Food Chem. 2005, 53, 4654–4659. [Google Scholar] [CrossRef] [PubMed]
- Wermelinger, T.; D’Ambrosio, L.; Klopprogge, B.; Yeretzian, C. Quantification of the Robusta fraction in a coffee blend via Raman spectroscopy: Proof of principle. J. Agric. Food Chem. 2011, 59, 9074–9079. [Google Scholar] [CrossRef] [PubMed]
- Craig, A.P.; Franca, A.S.; Oliveira, L.S.; Irudayaraj, J.; Ileleji, K. Application of elastic net and infrared spectroscopy in the discrimination between defective and non-defective roasted coffees. Talanta 2014, 128, 393–400. [Google Scholar] [CrossRef] [PubMed]
- Dias, R.C.E.; de Faria, A.F.; Mercadante, A.Z.; Bragagnolo, N.; Benassi, M.T. Roasting process affects the profile of diterpenes in coffee. Eur. Food Res. Technol. 2014, 239, 961–970. [Google Scholar] [CrossRef]
- Craig, A.P.; Franca, A.S.; Oliveira, L.S.; Irudayaraj, J.; Ileleji, K. Fourier transform infrared spectroscopy and near infrared spectroscopy for the quantification of defects in roasted coffees. Talanta 2015, 134, 379–386. [Google Scholar] [CrossRef] [PubMed]
- Campa, C.; Ballester, J.F.; Doulbeau, S.; Dussert, S.; Hamon, S.; Nairot, M. Trigonelline and sucrose diversity in wild Coffea species. Food Chem. 2004, 88, 39–43. [Google Scholar] [CrossRef]
- Urgert, R.; van der Weg, G.; Kosmeijer-Schuil, T.G.; van de Bovenkamp, P.; Hovenier, R.; Katan, M.B. Levels of the cholesterol-elevating diterpenes cafestol and kahweol in various coffee brews. J. Agric. Food Chem. 1995, 43, 2167–2172. [Google Scholar] [CrossRef]
- Kitzberger, C.S.G.; Scholz, M.B.S.; Benassi, M.T. Bioactive compounds content in roasted coffee from traditional and modern Coffea arabica cultivars grown under the same edapho-climatic conditions. Food Res. Int. 2014, 61, 61–66. [Google Scholar] [CrossRef]
- Nogueira, M.; Trugo, L.C. Distribuição de isômeros de ácido clorogênico e teores de cafeína e trigonelina em cafés solúveis brasileiros. Ciênc. Tecnol. Aliment. 2003, 23, 296–299. [Google Scholar] [CrossRef]
- Daglia, M.; Cuzzoni, M.T.; Decano, C. Antibacterial activity of Coffee: Relationship between biological activity and chemical markers. J. Agric. Food Chem. 1994, 42, 2273–2277. [Google Scholar] [CrossRef]
- Alonso-Salces, R.M.; Serra, F.; Reniero, F.; Héberger, K. Botanical and geographical characterization of green coffee (Coffea arabica and Coffea canephora): Chemometric evaluation of phenolic and methylxanthine contents. J. Agric. Food Chem. 2009, 57, 4224–4235. [Google Scholar] [CrossRef] [PubMed]
- Kuhnert, N.; Jaiswal, R.; Eravuchira, P.; El-Abassy, R.M.; von der Kammer, B.; Materny, A. Scope and limitations of principal component analysis of high resolution LC-TOF-MS data: The analysis of the chlorogenic acid fraction in green coffee beans as a case study. Anal. Methods 2011, 3, 144–155. [Google Scholar] [CrossRef]
- Maeztu, L.; Andreza, S.; Ibanêz, C.; Peña, M.P.; Bello, J.; Cid, C. Multivariate methods for characterization and classification of espresso coffees from different botanical varieties and types of roast by foam, taste, and mouthfeel. J. Agric. Food Chem. 2001, 49, 4743–4747. [Google Scholar] [CrossRef] [PubMed]
- De Souza, R.M.N.; Canuto, G.A.B.; Dias, R.C.E.; Benassi, M.T. Teores de compostos bioativos em cafés torrados e moídos comerciais. Quim. Nova 2010, 33, 885–890. [Google Scholar] [CrossRef]
- França, A.S.; Oliveira, L.S.; Oliveira, R.C.S.; Agresti, P.C.M.; Augusti, R. A preliminary evaluation of the effect of processing temperature on coffee roasting degree assessment. J. Food Eng. 2009, 92, 345–352. [Google Scholar] [CrossRef]
- Wei, F.; Tanokura, M. Chemical changes in the components of coffee beans during roasting. In Coffee in Health and Disease Prevention, 1st ed.; Preedy, V.R., Ed.; Academic Press-Elsevier: London, UK, 2015; Single Volume, pp. 83–91. [Google Scholar]
- Trugo, L.C.; Macrae, R. A study of the effect of roasting on the chlorogenic acid composition of coffee using HPLC. Food Chem. 1984, 15, 219–227. [Google Scholar] [CrossRef]
- Abrahao, S.A.; Pereira, R.G.F.A.; Duarte, S.M.S.; Lima, A.R.; Alvarenga, D.J.; Ferreira, E.B. Compostos bioativos e atividade antioxidante do café (Coffea arabica L.). Ciênc. Agrotec. 2010, 34, 414–420. [Google Scholar] [CrossRef]
- Dawidowicz, A.L.; Typek, R. Thermal stability of 5-O-caffeoylquinic acid in aqueous solutions at different heating conditions. J. Agric. Food Chem. 2010, 58, 12578–12584. [Google Scholar] [CrossRef] [PubMed]
- Dawidowicz, A.L.; Typek, R. Thermal transformation of trans-5-O-caffeoylquinic acid (trans-5-CQA) in alcoholic solutions. Food Chem. 2015, 167, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Lei n° 13519, 08 DE ABRIL DE 2002. Súmula: Estabelece Obrigatoriedade de Informação, Conforme Especifica, nos Rótulos de Embalagens de Café Comercializado no Paraná. Available online: http://www.deloitte.com.br/publicacoes/2002all/052002/icmspr/lei13519.pdf (accessed on 28 April 2015).
- TRAMITAҪÃO DE PROJETOS. PL 4703 2013-PROJETO DE LEI. Available online: http://www.almg.gov.br/atividade_parlamentar/tramitacao_projetos/interna.html?a=2013&n=4703&t=PL (accessed on 28 April 2015).
- Instituto Nacional de Meteorologia-INMET. Precipitaҫão Acumulada em 24 horas. Available online: http://www.inmet.gov.br/ (accessed on 27 May 2015).
- Alves, S.T.; Dias, R.C.E.; Benassi, M.T. Metodologia para análise simultânea de ácido nicotínico, trigonelina, ácido clorogênico e cafeína em café torrado por cromatografia líquida de alta eficiência. Quim. Nova 2006, 29, 1164–1168. [Google Scholar] [CrossRef]
- Borges, M.L.A.; França, A.S.; Oliveira, L.S.; Corrêa, P.C.; Gloria, M.B.A. Estudo da variação da coloração de café arábica durante a torra em diferentes condições de aquecimento. Rev. Bras. Armaz. 2002, 5, 3–8. [Google Scholar]
- Dias, R.C.E.; de Faria, A.F.; Mercadante, A.Z.; Bragagnolo, N.; Benassi, M.T. Comparison of extraction methods for kahweol and cafestol analysis in roasted coffee. J. Braz. Chem. Soc. 2013, 24, 492–499. [Google Scholar] [CrossRef]
- Vignoli, J.A.; Viegas, M.C.; Bassoli, D.G.; Benassi, M.T. Roasting process affects differently the bioactive compounds and the antioxidant activity of arabica and robusta coffees. Food Res. Int. 2014, 61, 279–285. [Google Scholar] [CrossRef]
- Mendes, L.C.; Menezes, H.C.; da Silva, M.A. Optimization of the roasting of robusta coffee (C. canephora conilon) using acceptabitility tests and RSM. Food Qual. Prefer. 2001, 12, 153–162. [Google Scholar] [CrossRef]
- Monteiro, M.C.; Trugo, L.C. Determinação de compostos bioativos em amostras comerciais de café torrado. Quim. Nova 2005, 28, 637–641. [Google Scholar] [CrossRef]
- Vitorino, M.D.; França, A.S. Variação de características físicas e químicas de café durante a torra. In Proceedings of the II Simpósio de Pesquisa dos Cafés do Brasil, Vitória, Brazil, 24–27 September 2001; Embrapa Café: Brasília, Brazil, 2001; pp. 1588–1604. [Google Scholar]
- Casal, S.; Oliveira, M.B.; Ferreira, M.A. HPLC/diode-array applied to the thermal degradation of trigonelline, nicotinic acid and caffeine in coffee. Food Chem. 2000, 68, 481–485. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dias, R.C.E.; Benassi, M.D.T. Discrimination between Arabica and Robusta Coffees Using Hydrosoluble Compounds: Is the Efficiency of the Parameters Dependent on the Roast Degree? Beverages 2015, 1, 127-139. https://doi.org/10.3390/beverages1030127
Dias RCE, Benassi MDT. Discrimination between Arabica and Robusta Coffees Using Hydrosoluble Compounds: Is the Efficiency of the Parameters Dependent on the Roast Degree? Beverages. 2015; 1(3):127-139. https://doi.org/10.3390/beverages1030127
Chicago/Turabian StyleDias, Rafael C. E., and Marta De T. Benassi. 2015. "Discrimination between Arabica and Robusta Coffees Using Hydrosoluble Compounds: Is the Efficiency of the Parameters Dependent on the Roast Degree?" Beverages 1, no. 3: 127-139. https://doi.org/10.3390/beverages1030127
APA StyleDias, R. C. E., & Benassi, M. D. T. (2015). Discrimination between Arabica and Robusta Coffees Using Hydrosoluble Compounds: Is the Efficiency of the Parameters Dependent on the Roast Degree? Beverages, 1(3), 127-139. https://doi.org/10.3390/beverages1030127