1. Introduction
Motor imagery (MI)-based classification remains one of the most challenging problems in brain–computer interfaces (BCIs), despite its central role in assistive control and neurorehabilitation. For example, Lin et al. [
1] developed a VR-integrated MI training system with real-time electromyography (EMG) feedback and reported promising outcomes in post-stroke rehabilitation. Similarly, Gordleeva et al. [
2] proposed an EEG–EMG hybrid human–machine interface (HMI) for lower-limb exoskeleton control, where combining leg MI with corresponding EMG signals improved both accuracy and reliability. These studies illustrate the potential of intelligent human–machine systems for motor restoration and assistive robotics. However, they also reflect a broader trend: most existing MI-BCI systems are highly engineered for specific scenarios, require extensive calibration, and often rely on controlled laboratory conditions that limit scalability in real-world applications.
In practice, MI-BCI systems still face several fundamental obstacles. Achieving stable and effective BCI control typically demands weeks or even months of user training [
3], substantially increasing time and resource costs and hindering wider clinical and home deployment. Moreover, EEG signal patterns vary considerably across individuals due to physiological and anatomical differences, meaning that even for nominally identical tasks, sensorimotor activation can differ markedly between users [
4]. Conventional MI paradigms—such as those used in benchmark datasets and competitions [
5]—usually employ abstract symbolic cues to prompt MI. For novice users in particular, these cue-based paradigms are difficult to interpret and execute consistently without prior experience, resulting in highly variable EEG patterns across sessions and participants and limiting both classification performance and system generalizability.
To reduce training costs and enhance the consistency of neural activation, researchers have increasingly turned to Action Observation (AO) as a complementary mechanism to MI. AO refers to the engagement of the mirror neuron system when individuals observe others performing specific motor actions [
6,
7,
8]. Prior work has demonstrated substantial overlap between the neural circuits recruited by AO and MI, even when each is executed in isolation [
9]. Building on this, combined AO + MI paradigms have been shown to produce stronger cortical activation and more robust engagement of the mirror neuron system compared with AO or MI alone [
10]. At the same time, virtual reality (VR) can further amplify these benefits by providing immersive, task-specific environments that reduce external distractions and promote sustained engagement [
11]. For instance, Choi et al. [
12] implemented a VR-based training scenario in which participants controlled a drone along a predefined path using AO + MI-driven training for an LDA classifier, followed by feedback-based control. Participants who received visual feedback of grasping hand movements significantly outperformed those without feedback, underscoring the potential of AO + MI paradigms combined with immersive feedback. More recent studies have further reported that combining VR-based action observation with kinesthetic motor imagery [
13] can enhance rhythmic modulation and improve task differentiation, supporting the neurophysiological plausibility of AO-in-VR priming. Additionally, VR-enhanced AO + MI has been increasingly integrated with rehabilitation-oriented systems (e.g., exoskeleton-related frameworks), highlighting the growing interest in ecologically valid training paradigms.
Despite these advances in training design and user engagement, the core signal-processing problem remains difficult. Due to the inherently non-stationary nature of EEG and the pronounced inter- and intra-subject variability, traditional feature extraction techniques—such as Common Spatial Patterns (CSP), Wavelet Transform (WT), and Short-Time Fourier Transform (STFT) [
14]—often struggle to capture discriminative, temporally stable features from complex neural data. Deep learning has therefore gained traction in the BCI community, as it can learn hierarchical, non-linear representations directly from raw or minimally preprocessed signals in an end-to-end manner. In particular, convolutional neural networks (CNNs) have demonstrated strong performance in MI classification, efficiently exploiting spatial–temporal structure in EEG and frequently outperforming handcrafted features in terms of both accuracy and generalization [
15]. More recently, transformer-based architectures have further improved offline decoding performance, especially in multi-class (e.g., four-class) MI settings [
16]. However, these gains are overwhelmingly reported in offline analyses under controlled conditions.
A further complication arises from the dynamic nature of the human brain. Sensorimotor rhythms (SMRs), which are widely used in MI-based BCIs, exhibit substantial variability over time and across users. Turi et al. [
3] reported that SMR characteristics can be strongly influenced by experimental environment, psychological state, and underlying neural circuitry. Saha et al. [
17] documented substantial intra- and inter-subject variability in EEG signals, while Morioka et al. [
4] observed that even within a single individual, EEG features may vary across phases or sessions, partly due to neural plasticity and repeated MI training. This continual adaptation of the nervous system—though beneficial for rehabilitation—poses a challenge for static classification models, which are typically trained once and then deployed without mechanisms to cope with gradual signal drift.
In response, continual learning (also known as incremental learning) [
18] has emerged as a promising paradigm for real-world BCIs. Continual-learning models are designed to assimilate new information from continuous data streams while preserving and refining previously acquired knowledge, thereby mitigating catastrophic forgetting. In the deep learning literature, van de Ven et al. [
19] categorize continual learning into three primary scenarios: (i) task-incremental learning, where task identities are known and a multi-headed output is used; (ii) domain-incremental learning, where the task remains fixed but the input distribution shifts over time; and (iii) class-incremental learning, where new classes are introduced sequentially and the model must retain performance on earlier classes. For MI-BCIs operating with evolving EEG distributions—such as those induced by subject learning, fatigue, or hardware variability—domain-incremental continual learning is particularly relevant, yet remains underexplored, especially in online, multi-class, and dry-electrode settings. Recent efforts have started to explore domain-/session-incremental [
20] learning strategies for MI-EEG across shifting distributions, but these studies are still predominantly evaluated under offline or limited deployment settings, and rarely under the combined constraints of dry electrodes, real-time closed-loop control, and multi-class (four-class) decoding. Therefore, the practical question of whether continual adaptation can reliably reduce the offline-to-online gap for dry-electrode, real-time four-class MI control remains insufficiently answered.
Recent deep learning approaches have reported high offline accuracies for four-class MI–BCI classification—for example, EEGNet [
15] and transformer-based models [
16] can reach approximately 75–80% or higher on public benchmark datasets under subject-dependent offline evaluation protocols. However, these results do not readily translate to online performance. In real-time settings, accuracy is frequently lower and less stable; in a four-class Cybathlon scenario, for instance, performance deteriorated near competition despite extensive training, suggesting high sensitivity to stress-induced EEG shifts and environmental changes [
21]. Reviews in stroke neurorehabilitation similarly point out that clinical translation is still limited by low online accuracy and the fact that many “high” performance reports are based on offline analyses rather than closed-loop control [
22]. Although virtually embodied feedback can improve subjective usability and engagement [
12], it still typically requires prolonged, user-specific calibration, and robust four-class online control remains highly variable across users.
Importantly, studies that are most comparable to the present work are those that evaluate MI decoding under deployment-relevant constraints—namely closed-loop/online operation, multi-class control (especially four-class), and/or wearable-friendly acquisition (e.g., dry electrodes). Across these settings, reported performance is typically lower than offline benchmarks and exhibits larger variability, highlighting a practical gap between “offline accuracy” and “online reliability”. Moreover, direct numerical comparison across studies can be misleading because electrode type (dry vs. wet), cue/feedback design, class cardinality, and closed-loop timing constraints differ substantially; therefore, comparability should be interpreted primarily in terms of setting realism and adaptation capability rather than peak offline scores alone.
Moreover, most prior MI–BCI studies rely on traditional wet electrodes, which are impractical for large-scale or daily use. Dry-electrode EEG provides a more convenient and user-friendly alternative for real-world systems; however, it also introduces practical challenges such as higher and time-varying contact impedance, increased susceptibility to motion/pressure artifacts, and reduced signal-to-noise ratio, which can exacerbate non-stationarity during prolonged use. Recent reviews of portable dry-electrode EEG systems [
23] further highlight that these hardware and contact-related factors remain key barriers to stable, repeatable decoding in real-world deployments. As a result, online multi-class (four-class or more) MI–BCIs with dry electrodes remain challenging and relatively underexplored.
To more explicitly address previously reported works, we position our study against three representative research directions. First, prior AO/VR-based MI-BCI systems primarily emphasize training design and user engagement (e.g., VR feedback and AO + MI paradigms) but typically rely on static decoders and do not explicitly handle online distribution shifts during closed-loop control. Second, recent deep models such as EEGNet and transformer-based architectures have achieved strong performance largely under offline evaluations and controlled protocols, leaving a gap to deployment-relevant online reliability. Third, real-world competitive/clinical reports highlight that four-class MI performance can deteriorate under stress and environmental changes and that offline accuracy often overestimates closed-loop performance. Motivated by these limitations, our work focuses on a realistic setting—dry-electrode acquisition with real-time four-class control—and introduces domain-incremental continual adaptation to reduce the offline-to-online gap under non-stationarity.
In this work, we propose a user-centered AO-in-VR MI-BCI framework that explicitly follows a two-phase learning pipeline, thereby addressing key limitations of related works that are often evaluated offline and/or do not explicitly mitigate dry-EEG non-stationarity during closed-loop operation. First, an immersive AO + MI pre-training stage with dry-electrode EEG is used for offline initialization of a CNN–Transformer hybrid decoder. Second, during closed-loop online operation, a continual-learning-enhanced adaptation stage incrementally updates the model using real-time data and feedback, targeting domain-incremental shifts in MI-related EEG. This rarely explored practical combination—dry-electrode EEG, immersive VR, real-time four-class decoding, and continual learning—enables robust multi-class control under realistic constraints. In a four-class MI paradigm (left hand, right hand, both feet, and rest), the proposed strategy improves online accuracy from 47.4% to 66.2% through continual adaptation, while ERD/ERS analyses further corroborate strengthened sensorimotor engagement under AO + MI. By jointly addressing neural activation, dry-electrode signal constraints, and online non-stationarity, the proposed framework narrows the gap between offline benchmarks and reliable real-time multi-class control, supporting practical deployment in neurorehabilitation and assistive BCI applications.
The remainder of this paper is organized as follows.
Section 2 reviews relevant literature and theoretical background on MI-BCI, AO + MI paradigms, dry-electrode EEG, and continual learning.
Section 3 describes the proposed system architecture, including the AO-in-VR training environment, experimental protocol, two-phase (offline + online) training pipeline, CNN–Transformer hybrid model, baseline methods, and ERD/ERS analysis.
Section 4 presents the experimental results, covering online classification performance and neurophysiological findings, followed by an in-depth discussion. Finally,
Section 5 concludes the study and outlines future directions for extending the proposed framework to broader real-world BCI and neuroadaptive applications.
2. Materials and Methods
2.1. Participants and EEG Acquisition
Five healthy, right-handed male participants (aged 20–22 years) were recruited for this study. None reported any history of neurological or psychiatric disorders. All procedures were approved by the local ethics committee, and written informed consent was obtained in accordance with institutional guidelines.
EEG signals were collected using a wireless dry-electrode EEG system (InMex; WellFulfill Ltd., Taoyuan, Taiwan), featuring eight measurement channels, a 24-bit analog-to-digital converter (ADC), and a 1 kHz sampling rate. The system includes integrated Bluetooth and hardware trigger interfaces. EEG signals were transmitted via Bluetooth and acquired in real time using a custom-developed backend built with Python 3.0, which provided a graphical user interface (GUI) for continuous signal monitoring and control.
Electrode positioning followed the international 10–20 system. Eight active electrodes were placed over key sensorimotor regions: F3, F4, C3, Cz, C4, P3, Pz, and P4. Ground and reference electrodes were located at the left and right mastoids, respectively. The electrode layout is shown in
Figure 1.
2.2. System Architecture and Experimental Setup
As illustrated in
Figure 2, participants were seated in a fixed-position ergonomic chair to minimize body movement during the experiment. Each participant wore a dry-electrode EEG cap and a VR headset. Prior to each session, electrode positions were calibrated and adjusted to ensure optimal contact and signal quality.
EEG data were continuously recorded from eight channels using a wireless EEG acquisition system. Raw EEG data were transmitted in real time to a host computer via Bluetooth. Upon reception, the signals were band-pass filtered between 8 and 30 Hz using a third-order Butterworth infinite impulse response (IIR) filter to retain relevant sensorimotor rhythms.
The filtered EEG data were segmented using a 2 s sliding window with a step size of 0.1 s, resulting in an overlap of 1.9 s between consecutive segments. Each EEG segment was input into a deep learning-based classification model to identify one of four motor imagery (MI) states: left turn, right turn, forward movement, or idle. The classification output was then transmitted to a Unity-based VR environment, where the corresponding avatar action was rendered in real time based on the predicted MI state.
2.3. Model Framework and Training Strategy
The proposed experimental model integrates Action Observation (AO) and Motor Imagery (MI) strategies to construct a personalized, pre-trained BCI classification model for each participant. Because dry-electrode EEG is prone to time-varying non-stationarity (e.g., contact changes, motion artifacts, fatigue), to adapt to the dynamic changes in the user’s brain state, a continual learning mechanism is incorporated to enable automatic updates of model parameters over time. This approach aims to sustain and progressively enhance MI classification accuracy in real-world applications. All update operations are performed within the same fixed task and class set, i.e., a domain-incremental setting.
Consistent with the framework outlined in
Section 1, the proposed MI-BCI system follows a two-phase training pipeline: (i) an offline AO + MI pre-training phase using dry-electrode EEG to initialize a CNN–Transformer hybrid decoder, and (ii) an online continual-learning phase, in which the pre-trained model is incrementally adapted based on real-time feedback data. The goal is to implement a domain-incremental continual learning scheme that tracks gradual distribution shifts in MI-related dry-EEG while preserving previously acquired knowledge. In this domain-incremental scenario, the MI classes remain unchanged across sessions, whereas the input distribution may drift over time.
The overall system architecture consists of the following components.
2.3.1. Pre-Training Phase (Offline Training)
In the offline pre-training phase, participants performed AO + MI and pure MI tasks while wearing the VR headset, so that all visual stimuli (fixation cross, directional cues, and avatar movements) were presented in an immersive virtual environment. Following the experimental paradigm described in [
24], each participant underwent three consecutive days of training, completing five experimental runs per day (15 runs in total). Each run consisted of randomized trials involving three types of motor imagery (MI) tasks: left hand, right hand, and both feet. Each MI type was presented in three sets, and each set included three trials: two Action Observation + Motor Imagery (AO + MI) trials followed by one pure MI trial.
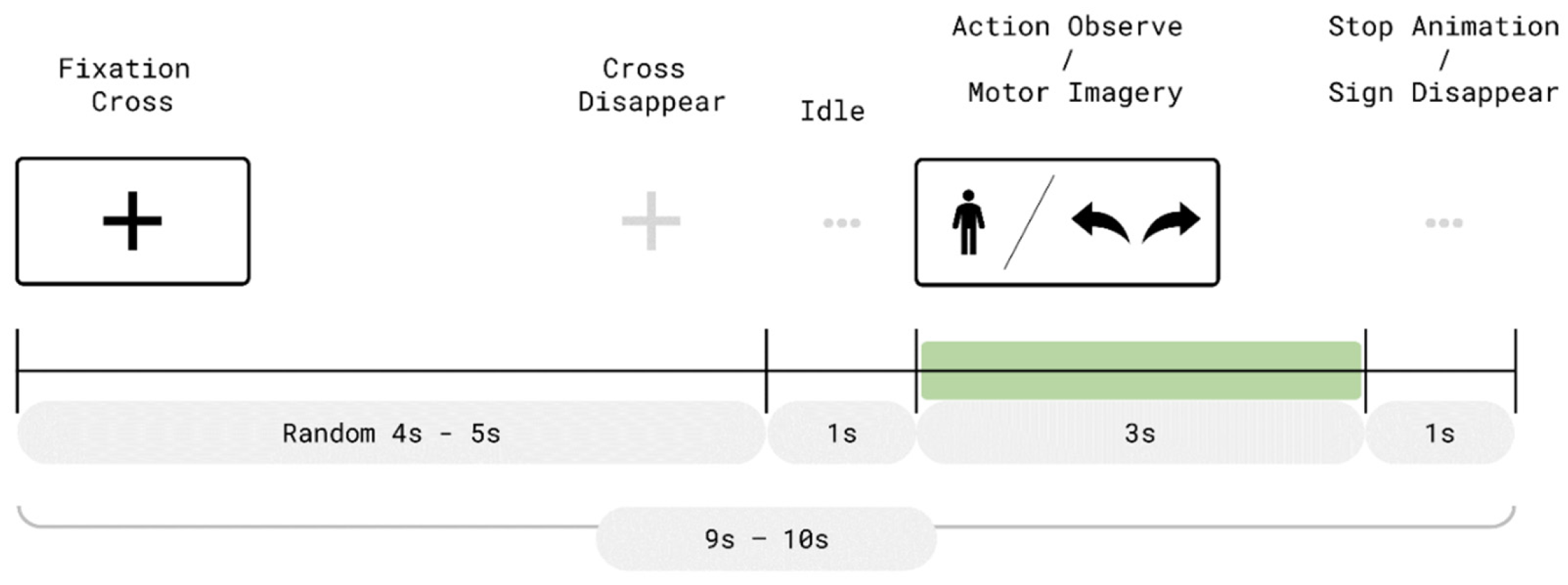
As illustrated in
Figure 3, each trial consisted of two time periods: a resting period (randomly lasting 4–5 s) and a 3 s movement period. At the beginning of each trial, a fixation cross was presented at the center of the VR display during the resting phase. After the fixation period, a 1 s preparatory interval was given before entering the movement phase.
In AO + MI trials, a virtual avatar in the VR environment performed the designated movement while a corresponding directional cue was displayed. For example, in a right-hand trial, the avatar raised its right arm and a right-pointing arrow appeared above its right shoulder, as shown in
Figure 4. Participants were instructed to simultaneously imagine performing the same movement.
In contrast, during pure MI trials (
Figure 5), the avatar was not shown. Only the directional cue appeared within the VR scene, prompting the participant to perform motor imagery without any concurrent visual movement observation. Each movement phase lasted 3 s. Afterward, the avatar ceased movement (if present) and the directional cue disappeared. A 1 s interval followed before the next trial began with the reappearance of the fixation cross. Each run lasted approximately 4.5 min, and each movement class was presented in a total of 27 trials across the session.
For model training, EEG segments corresponding to the three MI tasks and the inter-trial resting periods were labeled as four classes: left-hand MI, right-hand MI, both-feet MI, and rest. During the subsequent online phase, these three MI classes were mapped to left-turn, right-turn, and forward navigation commands, respectively, while the rest class served as the idle state (see
Section 2.3.2).
2.3.2. Online Continual-Learning
As shown in
Figure 6, the online continual-learning phase involved real-time navigation through a serpentine VR maze using motor imagery (MI). Participants controlled a virtual avatar from a fixed starting point to the goal by responding to directional cues—left turn, right turn, or forward movement—with the corresponding imagined movement. During each imagery period, EEG signals were classified by the pre-trained model. If the predicted class matched the instructed direction, the avatar executed the intended movement; otherwise, the avatar remained stationary, and the trial was repeated until successful classification.
This phase was designed as a domain-incremental continual learning extension of the pre-training protocol, enabling further refinement of the decoder under realistic, closed-loop conditions. Each feedback session was treated as a new domain in a domain-incremental continual learning scenario: the model was initialized from the offline pre-trained weights and then incrementally updated with data collected in the current and previous sessions, instead of being retrained from scratch. By progressively fine-tuning the model on accumulated feedback trials, the system aimed to track session-to-session shifts in the dry-EEG distribution while retaining prior knowledge, thus mitigating catastrophic forgetting. The online continual-learning experiment was conducted over three consecutive days, with three sessions per day, resulting in a total of nine adaptation runs per participant.
To minimize environmental interference and preserve consistency with the pre-training phase, participants remained seated and followed the same posture and trial timing structure. This design ensured that EEG variability was primarily attributed to neural adaptation rather than physical artifacts.
The trial timeline for the online continual-learning phase is illustrated in
Figure 7a,b. At the beginning of each trial, a fixation cross was displayed for a randomly selected duration of 2–3 s, followed by a 1 s preparatory interval. A directional cue (e.g., left turn, right turn, or forward) then appeared on the VR display for 2 s, during which participants were instructed to perform the corresponding MI task.
Immediately afterward, the system entered a 4 s classification window, continuously analyzing EEG data in real time. If the predicted class matched the presented cue, the avatar executed the intended movement and visual feedback was displayed (see
Figure 7a). Otherwise, the avatar remained stationary and the trial was repeated following a 1 s preparatory interval (
Figure 7b). To minimize participant fatigue and mitigate performance anxiety resulting from repeated failed attempts, the system limited the number of retries per instruction to three. If all three attempts failed, the system force-passed the trial and proceeded to the next command.
For real-time classification, a 2 s sliding window was employed with a 1.9 s overlap, yielding a new prediction every 0.1 s. To improve output stability, a temporal smoothing mechanism was applied: five consecutive identical predictions were required within the MI period to confirm a valid classification. If this consistency criterion was not met, the model output defaulted to a “rest” state. While this method enhanced classification stability, it introduced an approximate 0.5 s response latency.
2.4. EEG Conformer Model Architecture
The proposed decoder adopts a lightweight CNN–Transformer hybrid design to balance representational power and real-time feasibility. The CNN front-end serves as an efficient local feature extractor that captures short-range temporal dynamics and spatially distributed sensorimotor patterns (e.g., ERD/ERS-related signatures) with low computational overhead, which is desirable for dry-electrode EEG, where signal quality is noisier and latency constraints exist. However, convolution alone can be limited in modeling longer-range temporal dependencies and global inter-channel relationships that may shift across sessions. Therefore, a Transformer encoder is introduced to learn context-aware representations via attention, enabling the model to better integrate distributed evidence across time and channels under non-stationary conditions. A qualitative, implementation-oriented comparison of practical considerations across representative architectures (EEGNet, S3T, and the proposed model) is summarized in
Supplementary Table S1.
Figure 8 illustrates the architecture of the proposed model, which consists of two primary components: the preprocessing pipeline and the core classification model.
2.4.1. Preprocessing Pipeline
The preprocessing pipeline was designed to convert raw EEG signals into a structured format suitable for classification. This process consisted of a series of steps: band-pass filtering, artifact suppression, epoch extraction, downsampling, and normalization.
Initially, a band-pass filter (1–100 Hz) was applied to isolate the relevant EEG frequency components, and a 60 Hz band-stop filter was used to eliminate power line interference. To suppress transient artifacts caused by body movement or cable tension, the signal amplitudes were clipped to a range of ±50 µV (i.e., ±5 × 10−5 V).
Following filtering and clipping, the signal was further restricted to the 4–40 Hz band to emphasize motor-related activity. Event triggers were then used to segment the continuous EEG stream into 2 s epochs, each comprising 2000 time points at the original sampling rate of 1000 Hz.
To reduce computational load, the epochs were downsampled to 250 Hz, yielding 500 samples per segment. Each channel was subsequently normalized using Z-score normalization, defined as:
where
is the normalized signal,
is the signal after filtering and downsampling, and
and
denote the mean and standard deviation calculated over the training dataset. This normalization step reduced inter-channel variability and mitigated nonstationarity, enhancing model stability and generalization.
After preprocessing, the raw EEG data originally shaped as 8 × samples (eight channels) were converted into a 3D tensor of dimensions trials × 8 × 500, which was used as input for subsequent model training and classification.
2.4.2. Core Classification Model (CNN–Transformer Hybrid/EEG Conformer)
The core classification model adopts a CNN–Transformer hybrid architecture based on the EEG Conformer proposed by Song et al. [
25]. It comprises three main modules: a convolutional module, a self-attention module, and a classifier module. The convolutional and self-attention components together serve as a feature extractor, capturing both local and global patterns in the EEG signals. The resulting feature vectors are subsequently passed to the classifier module to generate four-class motor imagery (MI) predictions. For reproducibility, the complete architecture hyperparameters derived from
Figure 8 and the released implementation (including the Transformer configuration, e.g., N = 3 encoder blocks, h = 10 heads, d_model = 40, FFN hidden dim = 160, and dropout
p = 0.5) are summarized in
Supplementary Table S2.
The convolutional module consists of two sequential convolutional layers designed to extract temporal and spatial features. The first layer applies 20 kernels of size 1 × 25 with a stride of 1 × 1 to model temporal dependencies within each channel. The second layer uses 40 kernels of size 8 × 1 (stride 1 × 1) to capture inter-channel spatial relationships across the eight input channels. This hierarchical design enables the model to learn a robust set of local representations across time and space.
Each convolutional layer is followed by batch normalization and an exponential linear unit (ELU) activation function to accelerate convergence and reduce overfitting. To further lower computational complexity, an average pooling layer with a kernel size of 1 × 75 and a stride of 1 × 10 is applied, performing temporal downsampling and dimensionality reduction. The resulting output is reshaped into a matrix of shape (samples × number of convolutional filters), which serves as input to the subsequent self-attention module.
The self-attention module is designed to capture long-range temporal dependencies. Input features are linearly projected into query (
), key (
), and value (
) matrices. Attention weights are computed via scaled dot-product attention, where the dot product of
Q and
K is scaled by the square root of the key dimension, followed by a softmax activation to yield normalized attention scores. These weights are applied to the
V matrix to produce a context vector:
The attention output is then passed through a two-layer feed-forward network to further enhance feature expressiveness. This self-attention block is repeated times to increase temporal modeling depth.
To enhance representational diversity, a multi-head attention mechanism is employed.
,
, and
matrices are divided into
attention heads, each processed independently and subsequently concatenated. Here
denotes the head index (i.e., the
-th attention head), where
∈ {0, 1, …, h − 1}, and
is the total number of heads. For each head, the projected query, key, and value matrices (
,
,
) are obtained by applying head-specific linear projections to
, and
, respectively. The output of each head is computed via scaled dot-product attention, and the final MHA output is formed by concatenating all head outputs (followed by an output projection, if applicable):
Finally, the classifier module consists of two fully connected layers. The first layer includes 126 units with ELU activation, followed by a second layer with 16 units and a four-class softmax output corresponding to the MI categories. Dropout regularization is applied between layers to mitigate overfitting and improve generalization.
2.5. Model Training and Optimization Pipeline
During the pre-training data processing phase, stratified 5-fold cross-validation was initially performed to assess the generalization performance of the model. Following this evaluation, the entire dataset was used to train the final version of the model, which was subsequently deployed in the online feedback experiments. Stratification ensured that each fold preserved the same class distribution as the original dataset, thereby improving the reliability of performance estimation across motor imagery (MI) categories. The overall training procedure—offline AO + MI pre-training to initialize the CNN–Transformer decoder, followed by progressive fine-tuning across three online feedback sessions (Online session 1–Online session 3)—is summarized in
Figure 9, which illustrates the offline–online training and domain-incremental continual-learning pipeline used in this study.
Unlike standard cross-validation schemes, the dataset in this study consisted of two trial types: Action Observation (AO) and Motor Imagery (MI). Since both the experimental focus and real-time feedback control were exclusively based on MI, a custom partitioning strategy was employed. Specifically, only the MI data were partitioned into five folds for training and testing. All AO trials were incorporated into the training set of each fold, ensuring that the model had full access to AO data during training, while the test set consisted exclusively of MI trials. This partitioning strategy ensured that the validation setting more accurately reflected the conditions encountered during the online MI-based feedback phase.
The original dataset comprised 90 AO and 45 MI trials, yielding a 2:1 ratio. After partitioning, each fold contained 126 training samples and 9 testing samples. To enhance model robustness, data augmentation was applied to both training and test sets after the folds were defined to ensure independence. For each movement class (left hand, right hand, both feet), five overlapping 2 s EEG segments were extracted from the event trigger point at 0.1 s intervals. For the rest class, segments were extracted starting 3 s before the event. Because rest trials were more abundant, random subsampling was applied post-augmentation to ensure class balance. This process expanded each class to 675 trials. Performing augmentation only after data partitioning avoided sample overlap between training and testing, thereby preserving statistical independence.
During the online continual-learning phase, each participant completed three adaptation sessions, during which the model underwent two rounds of fine-tuning. A pre-trained model was initially developed using all available offline data and subsequently updated after the first and second sessions based on the successfully triggered trials collected in each session. The fine-tuning process involved adjustments to key hyperparameters—such as learning rate, batch size, and number of epochs—to optimize model performance and enhance real-time feedback accuracy. This progressive fine-tuning procedure corresponds to a domain-incremental continual learning strategy: newly collected feedback trials from each session are combined with previously acquired data to update the model, allowing it to adapt to evolving dry-EEG statistics while preserving earlier knowledge learned during offline AO + MI pre-training.
Two data handling strategies were employed based on the outcome of each triggering attempt. In successful trials, where the model accurately predicted the intended movement and the avatar responded accordingly in the VR environment, the movement onset was aligned with the classifier’s decision derived from the sliding window. The motor imagery (MI) trigger point was defined as 2.5 s prior to this motion onset. From that point, five overlapping 2 s EEG segments were extracted at 0.1 s intervals to augment the dataset. Rest-class data were processed using the same approach as in the pre-training phase, with five segments extracted from 3 s before the event trigger.
In contrast, for unsuccessful trials where the model failed to trigger the correct movement after three consecutive attempts, it was assumed that the participant initiated imagery upon cue presentation. In such cases, the cue onset was treated as the MI trigger point, and five 2 s segments were extracted using the same sliding-window method. These augmented samples were incorporated into the training set to ensure that the model could learn from both successful and unsuccessful control attempts.
Before each fine-tuning iteration, the updated dataset was evaluated using the existing model to assess current performance. The data were then split into training and testing sets with an 8:2 ratio. All data augmentation was performed after this split to preserve the independence of the test set, following the same precautions as in offline training.
Pilot runs and optimizer settings. Model development and training were implemented using PyTorch 1.8.1 and executed on an NVIDIA GeForce RTX 2060 GPU (12 GB). A fixed random seed was applied across all runs to ensure reproducibility. We optimized the cross-entropy loss using Adam with .5, .999, and weight decay = 1 ×. These hyperparameters were selected based on established practice and pilot validation to balance convergence stability and robustness under noisy dry-electrode EEG. Specifically, = 0.999 provides a stable second-moment estimate, while the smaller .5 makes the first-moment (momentum) update more responsive to distribution shifts and trial-to-trial variability, which is particularly relevant during closed-loop online adaptation. The weight decay term acts as regularization to mitigate overfitting given the limited sample size and model capacity.
To make the pilot-run procedure explicit, we conducted pilot runs within the training data using a training-only inner validation split under the offline 5-fold protocol and evaluated validation accuracy and weighted F1 together with convergence stability across folds/runs. The pilot search covered representative choices of learning rate, warmup length/schedule, Adam betas, weight decay, and batch size. For offline pre-training, we adopted a warm-up learning rate schedule (Equation (4)) with
= 10 to stabilize early optimization and reduce instabilities commonly observed in attention-based architectures, using a base learning rate of 3 ×
and a total of
= 100 epochs. For online fine-tuning, we used a smaller learning rate 2 ×
and fewer epochs (20) to reduce overfitting to limited feedback data while meeting real-time constraints (short adaptation horizon and low latency). The detailed hyperparameters for the offline pre-training and online feedback adaptation phases are summarized in
Table 1. Representative pilot-run comparisons of optimizer and learning-rate schedule configurations are summarized in
Supplementary Table S3.
2.6. Baseline Models
This section introduces the four baseline models used for pre-training evaluation. The selected models include one Transformer-based architecture—Spatial-Temporal Tiny Transformer (S3T)—and three well-established convolutional neural network (CNN) models: EEGNet, DeepConvNet, and ShallowConvNet.
S3T [
26] is a compact model specifically designed for motor imagery (MI) EEG classification, aiming to simultaneously capture spatial and temporal dependencies in EEG signals. The input signals are first transformed using a one-to-many mapping via Common Spatial Pattern (CSP) filtering. The resulting features are stacked and fed into the model. A channel attention mechanism is applied to prioritize task-relevant EEG channels while suppressing irrelevant ones, enhancing the model’s representational precision. After spatial processing, the data are compressed and segmented along the temporal dimension, followed by a temporal attention mechanism to extract global sequential features critical to MI decoding. Final classification is performed via global average pooling and a fully connected layer.
EEGNet [
15] is a lightweight end-to-end model for EEG-based classification, composed of three distinct convolutional operations. The first layer is a standard 2D convolution that extracts initial spatial and temporal features. The second is a depthwise convolution, which learns frequency-specific spatial patterns across channels. The third is a pointwise convolution, forming a separable convolution structure that integrates temporal features efficiently. A fully connected layer is used at the end for classification.
DeepConvNet [
27] is a deeper end-to-end CNN architecture tailored for MI EEG classification. It consists of four convolution–max pooling blocks. The first block includes two convolutional layers that handle raw EEG inputs, extracting low-level temporal and spatial features. The remaining three blocks progressively increase the depth of learned representations, improving the model’s discriminative capability. Final predictions are made through a fully connected output layer.
ShallowConvNet [
27] is a simplified variant of DeepConvNet, featuring a lighter architecture composed of two convolutional layers followed by a classifier. While more compact, it also emphasizes the extraction of key spatial and temporal patterns. Functionally, its architecture mimics the Filter Bank Common Spatial Pattern (FBCSP) approach: the early convolution layers serve as substitutes for bandpass and spatial filters, while the later stages apply squaring, average pooling, and a log activation function to estimate log-variance features across trials. Classification is completed via a fully connected layer.
2.7. ERD/ERS Analysis
To examine changes in neural activity following training, event-related desynchronization (ERD) and event-related synchronization (ERS) analyses were performed on trials that were correctly classified by the model during each motor imagery feedback (MI-FB) session. An ensemble averaging approach was applied to these successfully triggered trials to assess whether the model reliably identified physiologically relevant EEG patterns and to evaluate potential enhancements in cortical activation after training.
ERD is characterized by a decrease in power within specific frequency bands (e.g., alpha or beta) in response to motor-related events or stimuli, typically indicating cortical activation. In contrast, ERS represents an increase in band power following an event, often associated with cortical deactivation or inhibition.
In the context of motor imagery tasks, desynchronization (ERD) in the alpha (8–14 Hz) and beta bands is commonly observed over the sensorimotor cortex during imagined hand or foot movements. This is typically followed by resynchronization (ERS) after the movement concludes or during short rest intervals.
The ERD/ERS analysis followed the method proposed by Pfurtscheller et al. [
28]. EEG signals were first band-pass filtered in the alpha range (8–14 Hz) to isolate motor-related rhythms. The power of the filtered signal was then computed by squaring the signal amplitude, resulting in a trial-wise power estimate denoted as
for each motor imagery class and trial.
To compute the mean band power across trials for each motor-imagery (MI) class, we first extracted the trial-wise band power
and then averaged it over all trials within that class, as defined in Equation (5). Here,
denotes the band power of the
-th trial from the specified MI class, and
is the total number of trials belonging to that class. In this study,
. The resulting class-wise mean power
was used to characterize MI-related oscillatory dynamics and to provide neurophysiological validation (e.g., ERD/ERS) of the decoding results.
To compute the baseline power reference, we used the rest condition and averaged the power over both time samples within each trial and all rest trials, as shown in Equation (6). The baseline
is a scalar constant used as the reference for subsequent ERD/ERS percentage calculations. Here,
denotes the time-resolved power in the target frequency band at the
sample of the
rest trial,
is the number of rest trials, and
is the number of samples per trial.
Subsequently, the ERD/ERS percentage for each MI class was computed using Equation (7):
This equation quantifies the relative power change between MI trials and the resting-state baseline. Negative values (ERD) indicate power attenuation (desynchronization), reflecting cortical activation associated with motor imagery, whereas positive values (ERS) indicate power enhancement (synchronization), depending on the frequency band of interest.
4. Discussion
4.1. Addressing Limitations of Conventional MI-BCI Systems Through Action Observation
Conventional motor imagery-based brain–computer interface (MI–BCI) systems and datasets typically rely on simple visual or auditory cues to prompt motor imagery execution. However, such abstract and minimal instructions often lack concrete and intuitive movement guidance, making it difficult for users—especially those new to BCI—to understand how to effectively generate motor imagery. This can lead to increased cognitive load and reduced training efficiency. Moreover, in the early stages of training, when the classification model has not yet been established, the system is unable to provide real-time feedback. This absence of feedback can result in frustration, reduced motivation, and lower engagement among users. Critically, the lack of action-specific clarity and consistent feedback can also contribute to variability in EEG patterns across sessions, thereby undermining model stability and generalization.
To address these challenges, the present study employed action observation (AO) to assist motor imagery (MI), offering participants concrete and dynamic visual references to enhance the vividness and interpretability of the imagery task [
30]. In the AO + MI experiments, we observed clear event-related desynchronization (ERD) in sensorimotor regions associated with right-hand and both-feet imagery, indicating that AO-augmented MI effectively activates the corresponding cortical areas. However, the effectiveness of AO appeared to vary across motor classes. Specifically, for left-hand imagery, AO + MI alone was insufficient to elicit stable and robust ERD responses. This class-dependent variability may be related to factors such as hand dominance, individual imagery strategy, and the limited dry-electrode montage, and therefore should not be interpreted as an inherent limitation of AO itself. These observations motivate the need for personalized adaptation to stabilize class-specific representations during closed-loop use. Consistent desynchronization in the relevant cortical areas only emerged after additional training through an online continual-learning phase, during which the system provided real-time feedback and model updates based on the user’s imagery performance. These findings suggest that while AO serves as a useful priming tool for MI–BCI training, motor-specific adaptation via online learning remains essential for achieving reliable cortical engagement, particularly for non-dominant or less frequently imagined movements. In our framework, AO-primed offline training in an immersive VR environment provides a more consistent initial representation of MI-related activity, which is then refined during the online continual-learning phase. By coupling AO + MI pre-training with closed-loop feedback on dry-electrode signals, the system directly addresses the lack of intuitive guidance and unstable online performance frequently reported in conventional four-class MI–BCIs.
4.2. Neurophysiological Implications of AO + MI and Training Effects Across Sessions
As summarized in
Table 4, action observation (AO) consistently elicited stronger event-related desynchronization (ERD) responses than pure motor imagery (MI), particularly in the C3 and Cz channels during left-hand imagery and in the C3 channel during both-feet imagery. These differences reached statistical significance (
p < 0.05, Wilcoxon signed-rank test) and align with prior findings [
10], which indicates that AO + MI strategies can significantly enhance cortical activation in sensorimotor regions.
In addition to its neurophysiological benefits, AO + MI also helps novice users more easily engage in motor imagery [
31], serving as an intuitive scaffold for acquiring MI skills. Notably, mirror neuron activation elicited by AO is highly resilient to repetition, showing minimal fatigue effects even with frequent stimulation [
32], making AO especially suitable for long-term BCI training protocols.
While most prior studies adopt either AO or MI in isolation, our method uniquely integrates alternating AO and MI phases, where participants recall the observed AO movement during the subsequent MI task. This reinforced mental representation facilitates more vivid and consistent MI execution, and allows the offline-trained model to capture stronger and more stable ERD features, thereby laying a solid foundation for reliable online adaptation. From an algorithmic perspective, these AO-induced ERD patterns provide informative training examples for the CNN–Transformer decoder, strengthening its initial feature space before domain-incremental adaptation. This is particularly important in dry-electrode settings, where signal-to-noise ratios are lower and robust initialization can substantially reduce the burden on subsequent continual learning.
The importance of real-time BCI feedback in enhancing user control within immersive environments was also evident in this study. Each participant completed three VR-based feedback sessions, each containing three route trials. While trial durations within a single session remained relatively consistent, average task completion time decreased across sessions, indicating that participants were able to navigate the same virtual paths more efficiently in later sessions.
The across-session reduction in VR task completion time suggests improved closed-loop controllability with practice. However, given the relatively short training duration per session, we interpret this behavioral trend cautiously and do not claim direct evidence of neuroplasticity. The improvement could reflect a combination of factors, including better familiarity with the VR interface, more consistent imagery strategy execution, and the model’s progressive alignment to the user-specific EEG distribution through continual adaptation. Importantly, this behavioral trend is consistent with the session-wise decoding gains and reduced confusion observed in the online results, while the ERD/ERS analyses provide complementary physiological evidence of sensorimotor engagement during AO priming.
To examine how participants’ EEG representations evolved across online sessions during closed-loop training, we employed t-distributed stochastic neighbor embedding (t-SNE) as a qualitative visualization tool. The purpose of this analysis was to explore whether the continual adaptation process was associated with increased within-class compactness and clearer between-class separation in the learned feature space for representative participants. t-SNE was selected because it provides an intuitive visualization of local neighborhood structures in high-dimensional embeddings produced by the deep model. Our goal was not to benchmark dimensionality-reduction techniques, but rather to obtain an interpretable view of how internal feature representations changed across sessions. Principal component analysis (PCA) was therefore not used as the primary visualization approach because its linear projection may not adequately reveal potentially nonlinear structures in deep EEG feature embeddings. Although alternative nonlinear methods such as UMAP could also be applied, the objective of this analysis was not to compare visualization algorithms, but to use a commonly adopted and widely interpretable technique to complement the quantitative evaluation. Accordingly, the t-SNE plots should be interpreted as qualitative and supportive visualizations rather than definitive evidence of performance improvement. The primary conclusions of this study are supported by the quantitative closed-loop results, including classification accuracy, weighted F1-score, row-normalized confusion matrices, and neurophysiological analyses. In our implementation, PCA was used only for initialization of the t-SNE embedding, and the perplexity parameter was empirically set to 20 to ensure stable and reproducible visualization under a fixed random seed.
In our implementation, AO stimuli were presented from a third-person perspective, featuring a robot-like virtual avatar. While first-person perspectives are generally considered to promote stronger subjective agency [
33], the observed ERD magnitudes induced by third-person AO remained significantly higher than those from pure MI. This suggests that even without first-person visual feedback during MI, the third-person AO still provided sufficient visual context and functional embodiment to enhance cortical activation.
4.3. Model Architecture Analysis and Training Progression
Beyond the neurophysiological findings, the proposed system can also be viewed as a domain-incremental continual-learning engine operating on dry-electrode, four-class MI data in real time. From an expert-systems perspective, this corresponds to an adaptive inference module that continuously updates its internal representation as new evidence (feedback trials) becomes available.
The proposed hybrid CNN–Transformer architecture, inspired by the design introduced by Song et al. [
25], demonstrated superior classification performance compared to traditional CNN models and CSP-based Transformer structures, as summarized in
Table 2. A complementary, implementation-oriented qualitative summary of practical considerations (e.g., training stability, overfitting tendency, computational/maintenance overhead, and online suitability) across representative architectures (EEGNet, S3T, and the proposed model) is provided in
Supplementary Table S1. In this model, the convolutional module automatically extracted low-level temporal and spatial features from EEG signals, while the self-attention module captured long-range temporal dependencies and facilitated global feature integration. This configuration outperformed models that relied solely on localized CNN filters.
While manually engineered features such as common spatial patterns (CSPs) have proven effective in extracting task-relevant cortical information [
14], the S3T model [
26], which integrates CSP with Transformer layers, lacked hierarchical representation learning for localized signal variations. As a result, its generalization capability was inferior to the end-to-end learning pipeline adopted in this study.
However, a potential limitation of the proposed architecture lies in the choice of convolutional kernel sizes. Smaller kernels are advantageous for capturing fine-grained features but may amplify noise, whereas larger kernels offer broader context at the cost of reduced sensitivity to subtle signal fluctuations. Future research may explore adaptive kernel sizing strategies to better balance temporal resolution and robustness in EEG decoding.
Across the offline and online evaluations (
Table 2,
Table 3,
Table 4 and
Table 5), the results consistently indicate that performance gains arise from the interaction between a strong initialization and deployment-time adaptation. The CNN–Transformer decoder (
Table 2) provides a representation that is better suited to capture both localized SMR-related features and longer-range dependencies than purely local convolutional baselines, which is beneficial under dry-electrode noise and distribution drift. Building on this initialization, session-wise continual adaptation yields progressively improved closed-loop decoding (
Table 3) and more coherent class-wise separability in the learned feature space (
Table 5, qualitative). Importantly, the physiological analyses (
Table 4) show that AO priming elicits stronger SMR modulation in several channel–task pairs, and the sessions/classes exhibiting clearer ERD patterns tend to align with higher decoding reliability. We emphasize that this correspondence provides convergent evidence rather than a causal proof; nevertheless, it strengthens the interpretation that improved online performance reflects better capture of functionally meaningful sensorimotor dynamics rather than solely overfitting to noise.
In our study, the decoder performs four-class motor imagery (MI) classification, where the theoretical chance level is 25%. Therefore, the obtained accuracies, such as 52.78% in offline cross-validation (offline CV) and 66.20% in the online Session 3 experiment, are substantially above chance and demonstrate meaningful decoding capability under a challenging task setting. It is important to note that multi-class MI-BCI systems (e.g., four-command/four-class control), particularly in online closed-loop operation, are considerably more difficult than the commonly studied binary MI paradigms. This is mainly because different imagined movements often produce overlapping EEG features across classes, making reliable classification more challenging. Consequently, stable performance in multi-class MI-BCI systems typically requires substantial user training, system adaptation, and multiple experimental sessions. Furthermore, EEG patterns themselves may evolve during the training process as users gradually learn to modulate their sensorimotor rhythms more effectively. To address these challenges, the present study adopts an action observation (AO) pretraining strategy combined with online fine-tuning, allowing the model to progressively adapt to subject-specific neural patterns and continuously improve decoding performance during online sessions. To further contextualize the contribution of this work, we summarize representative studies on online four-command MI/SMR-based BCI systems. In the work of Hehenberger et al. (2021) [
34], a tetraplegic pilot underwent 14 months of mutual training across 26 sessions to perform four-class motor imagery for controlling a racing avatar in the CYBATHLON BCI Race, ultimately achieving an online accuracy of approximately 53%. In another large-scale study, Stieger et al. (2021) [
35] released a dataset involving 62 participants, with more than 600 h of EEG recordings, 598 sessions, and over 250,000 trials. In their study, each participant performed four motor imagery tasks (left hand, right hand, both hands, and feet) across multiple sessions to control a 2D cursor task, with an average final control accuracy of approximately 49%. Compared with these studies, the proposed framework incorporates AO-based pretraining data to initialize the model, followed by three online sessions for continual model adaptation. As a result, the online performance shows a progressive improvement from 47.42% in Session 1 to 66.20% in Session 3, demonstrating that the proposed framework effectively narrows the offline-to-online performance gap in a practical real-time four-class MI-BCI scenario.
4.4. Limitations and Future Directions
Despite these promising findings, several limitations should be acknowledged. First, the study involved a small cohort of healthy participants and an 8-channel dry-electrode montage, which limits statistical power and generalizability to broader and clinical populations. Second, we observed notable inter-subject variability and class-dependent difficulty (e.g., uneven separability across motor classes), indicating that both user-specific factors and movement-specific characteristics influence closed-loop performance. Third, the online evaluation spanned only three feedback sessions, which is insufficient to establish cross-session generalization across longer time scales or to quantify catastrophic forgetting under extended deployment. Fourth, the current validation focused on a four-class MI paradigm in a controlled VR navigation task, and generalization to richer command sets or less structured environments remains to be demonstrated.
Future work will prioritize (i) larger and more diverse cohorts, including motor-impaired users, (ii) longitudinal and cross-session deployments to quantify drift, robustness, and forgetting, (iii) enhanced personalization (e.g., adaptive kernel sizing or data-driven receptive-field selection) to balance temporal resolution and robustness under dry-EEG, (iv) more robust continual-learning strategies (e.g., regularization- and replay-based updates) to stabilize adaptation under non-stationarity, and (v) optional multimodal extensions (e.g., EMG/kinematics) to further improve reliability.