Advancements in Bio-Based Piezoelectric Composites for Antibacterial Applications
Abstract
1. Introduction
2. Piezocatalyzed Antibacterial Mechanisms
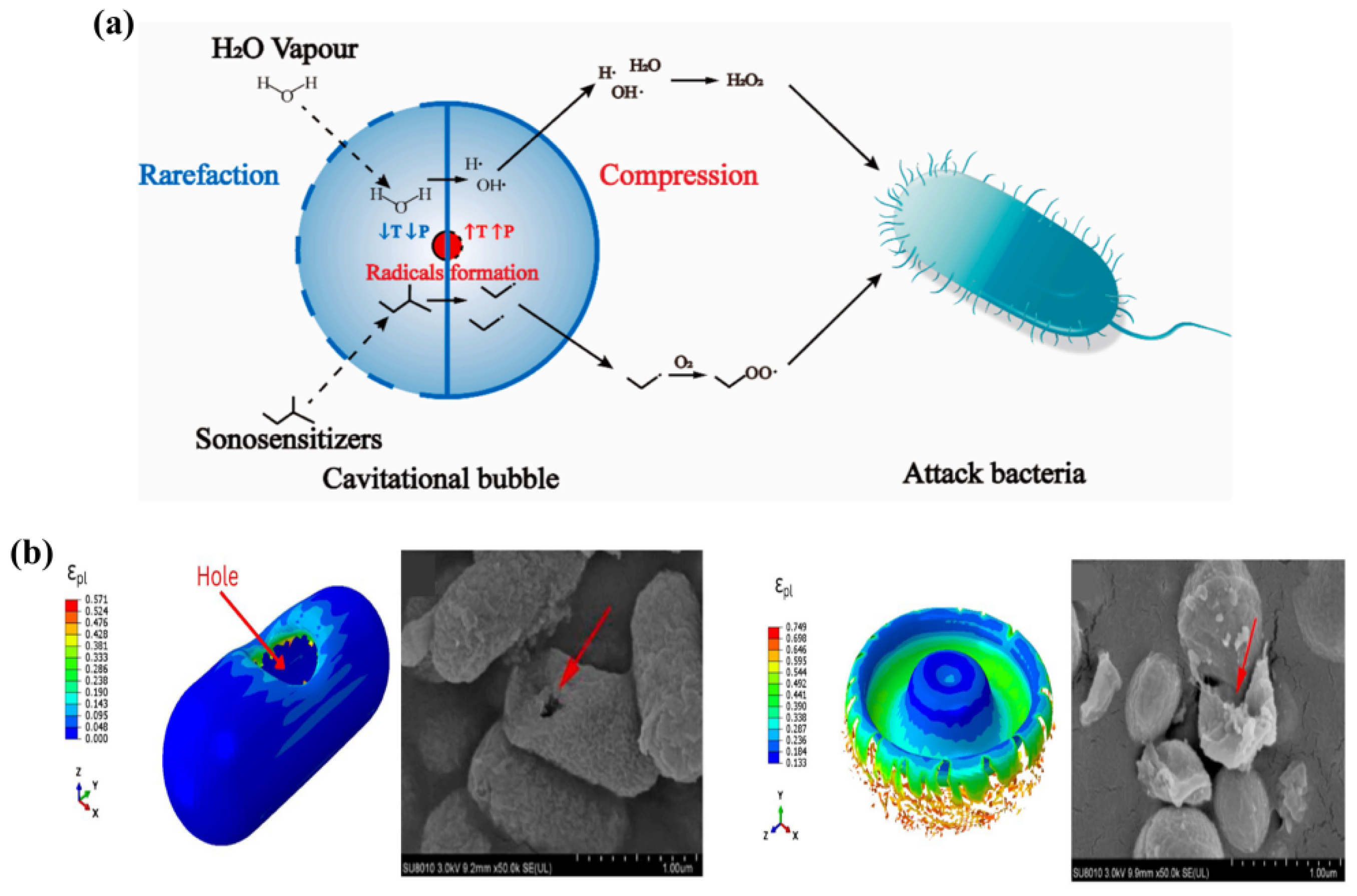
2.1. Ultrasonic-Induced Cavitation Effect
2.2. Piezoelectric Perforation
2.3. Bacterial Inactivation Mediated by ROS

3. Prevalent Biological Substances Exhibiting Piezoelectric Characteristics
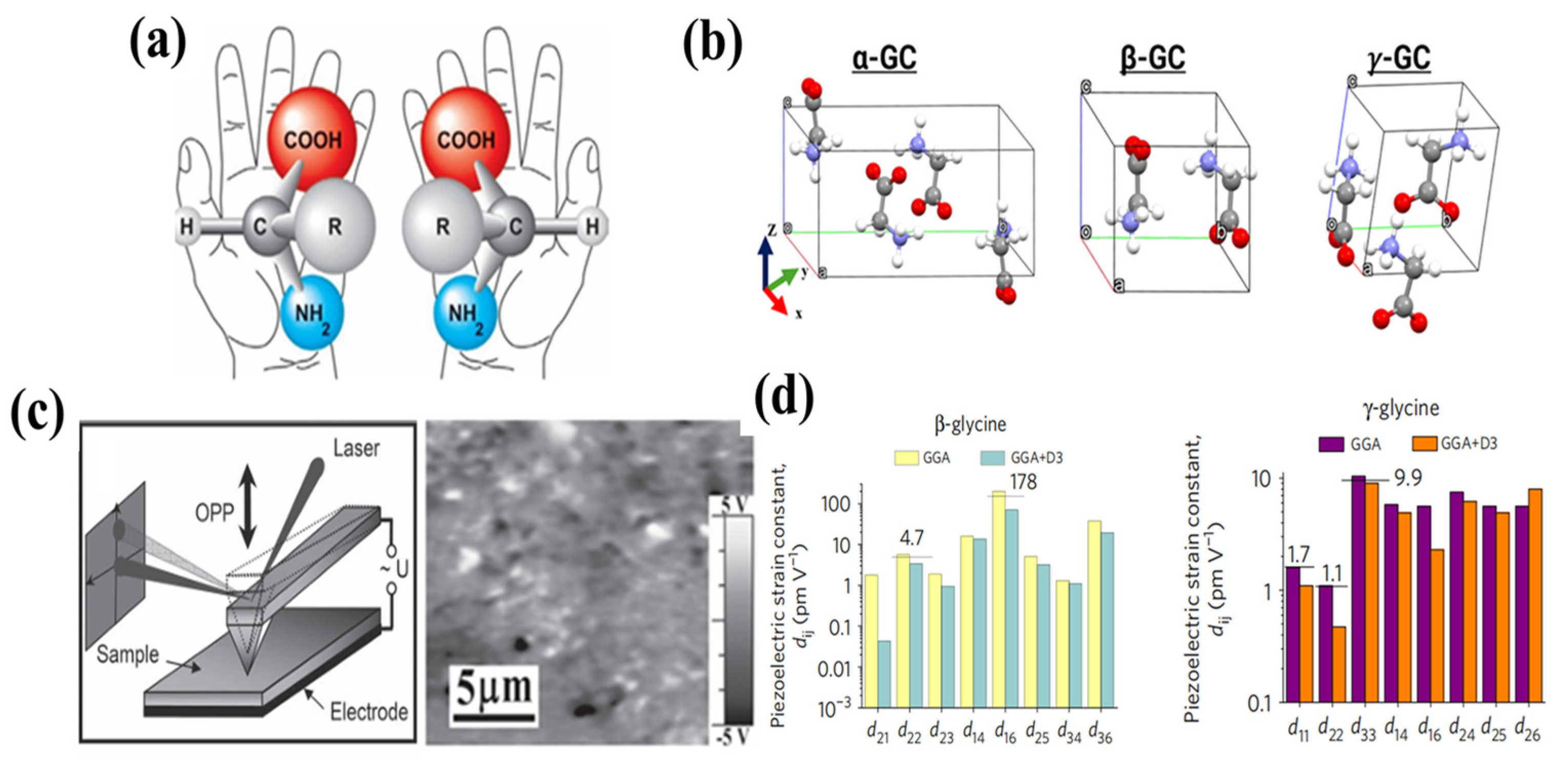
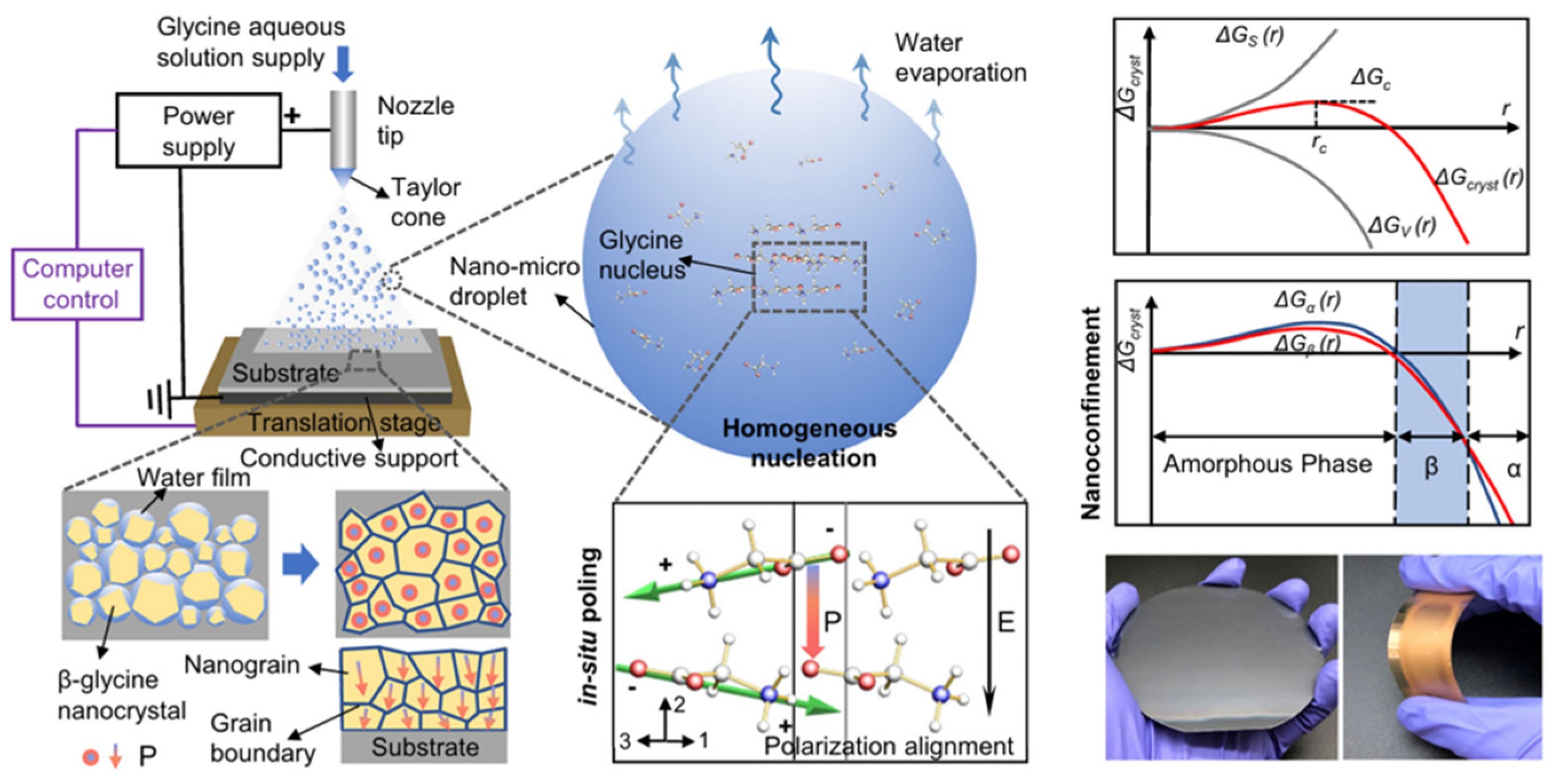
3.1. Amino Acids
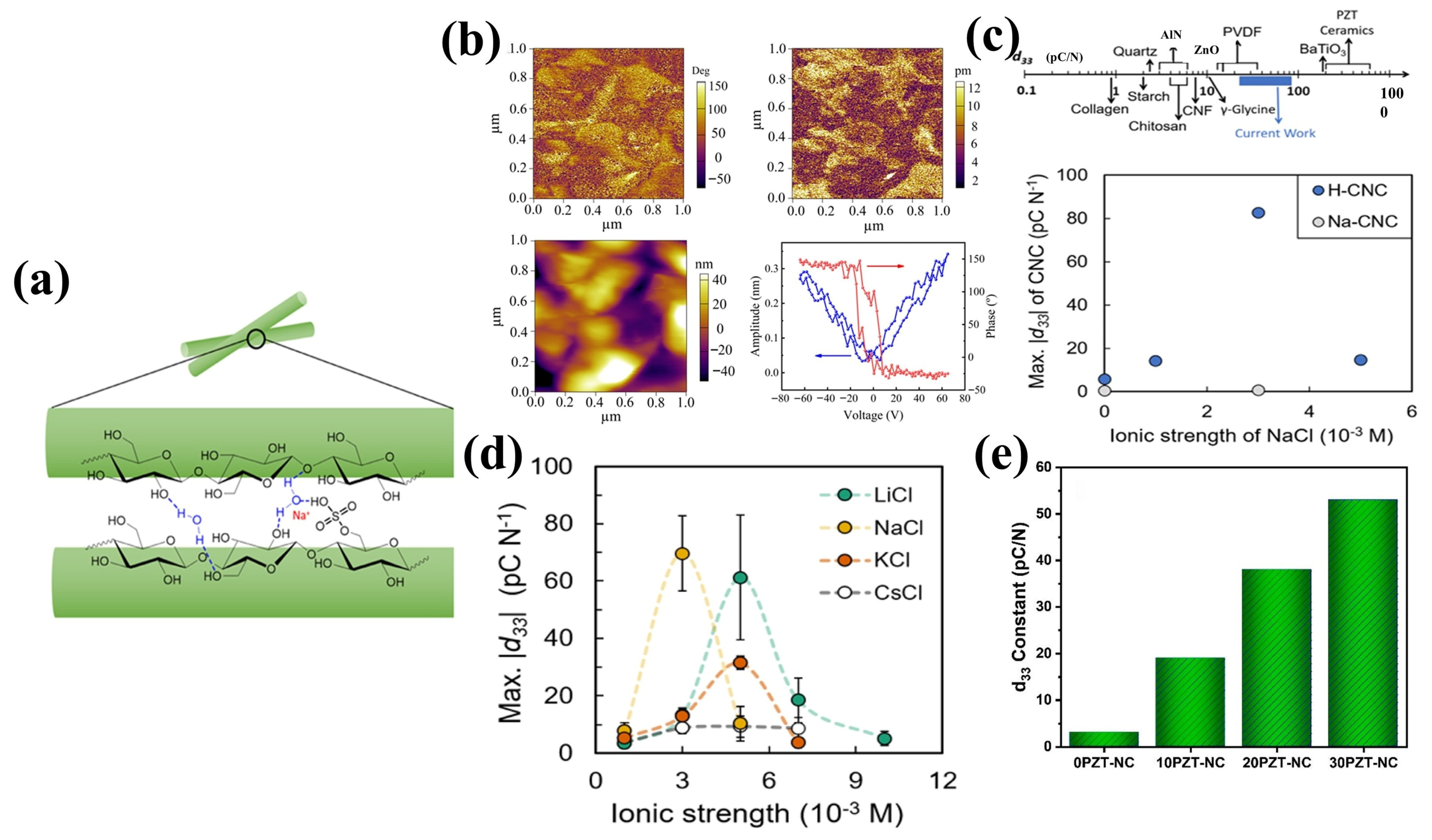
3.2. Cellulose
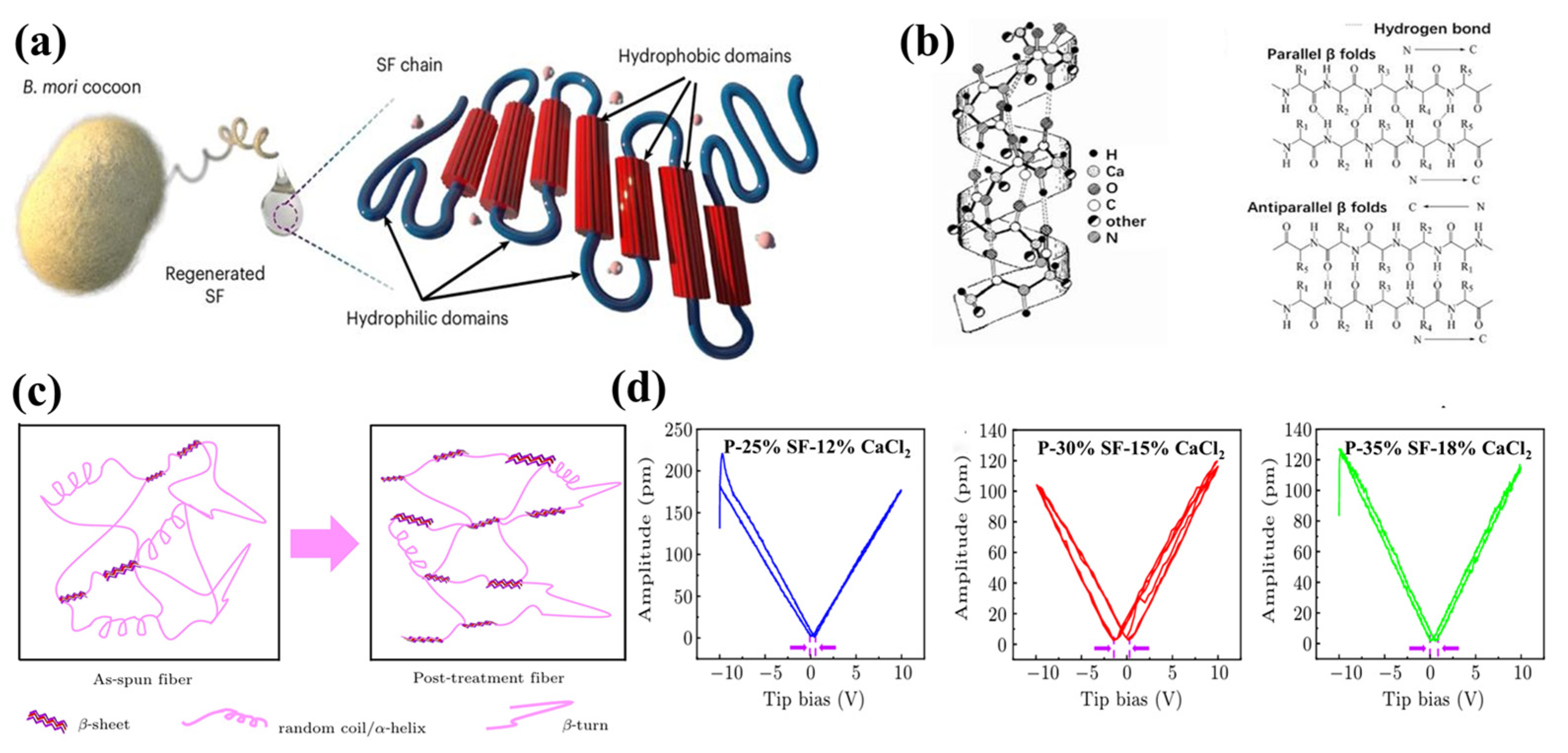
3.3. Proteins
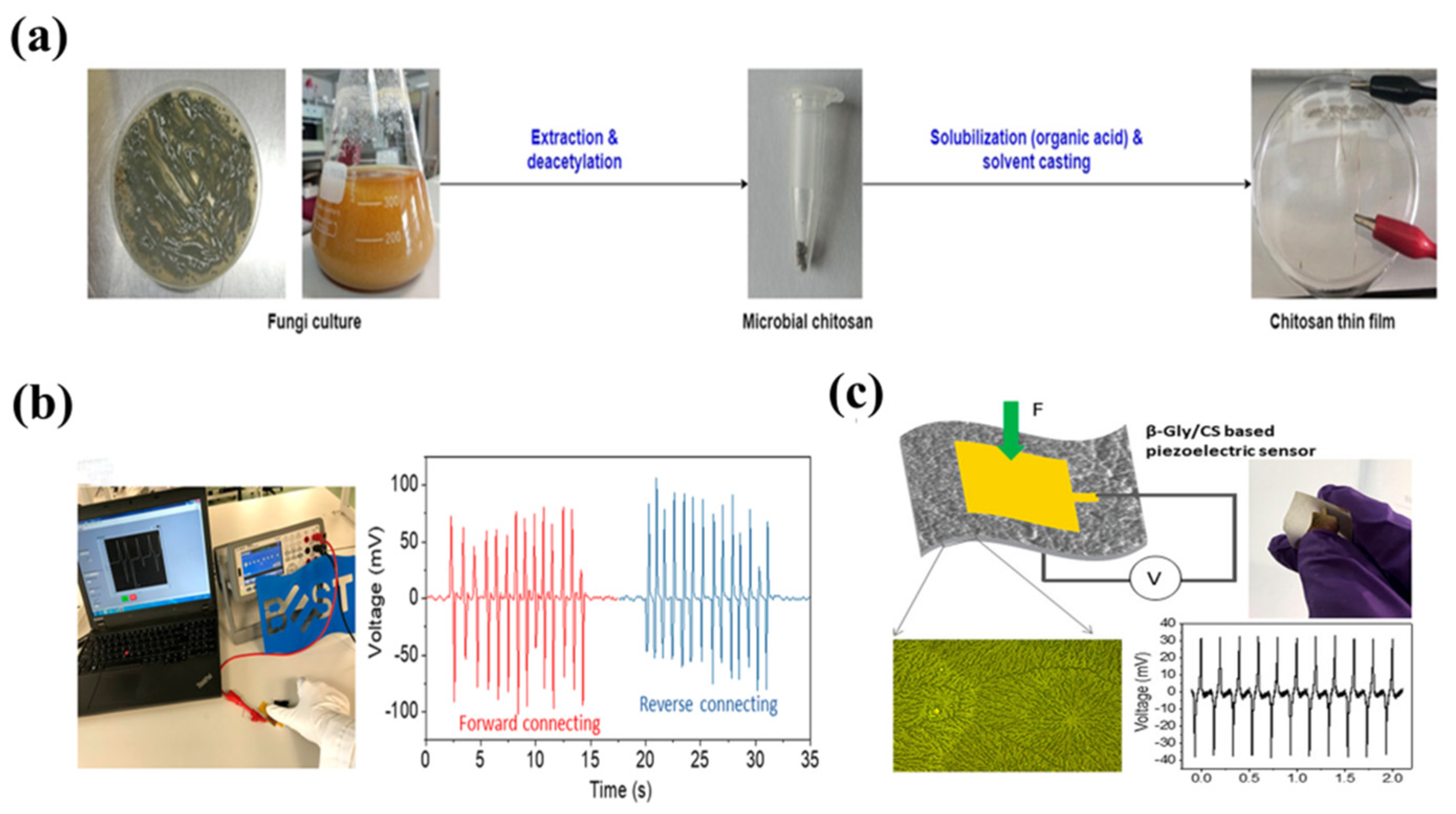
3.4. Chitosan
4. Fabrication Strategies for Piezoelectric Biocomposites
4.1. Spin Coating
4.2. Electrochemical Deposition
4.3. Self-Assembly Strategy
4.4. Electrospinning
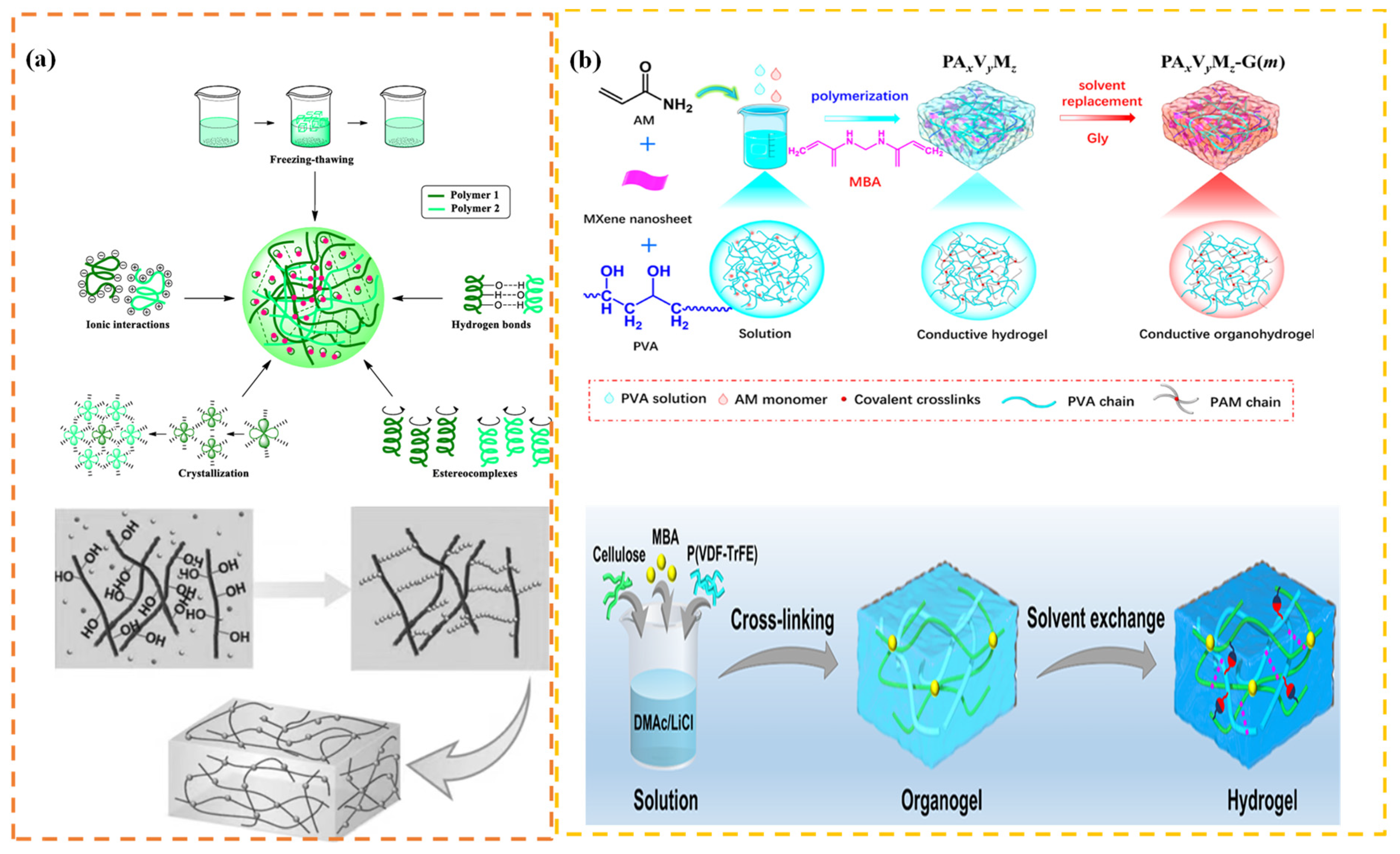
4.5. Physical-Chemical Crosslinking Method
5. Piezocatalysis in Biological Antibacterial Applications
5.1. Water Disinfection
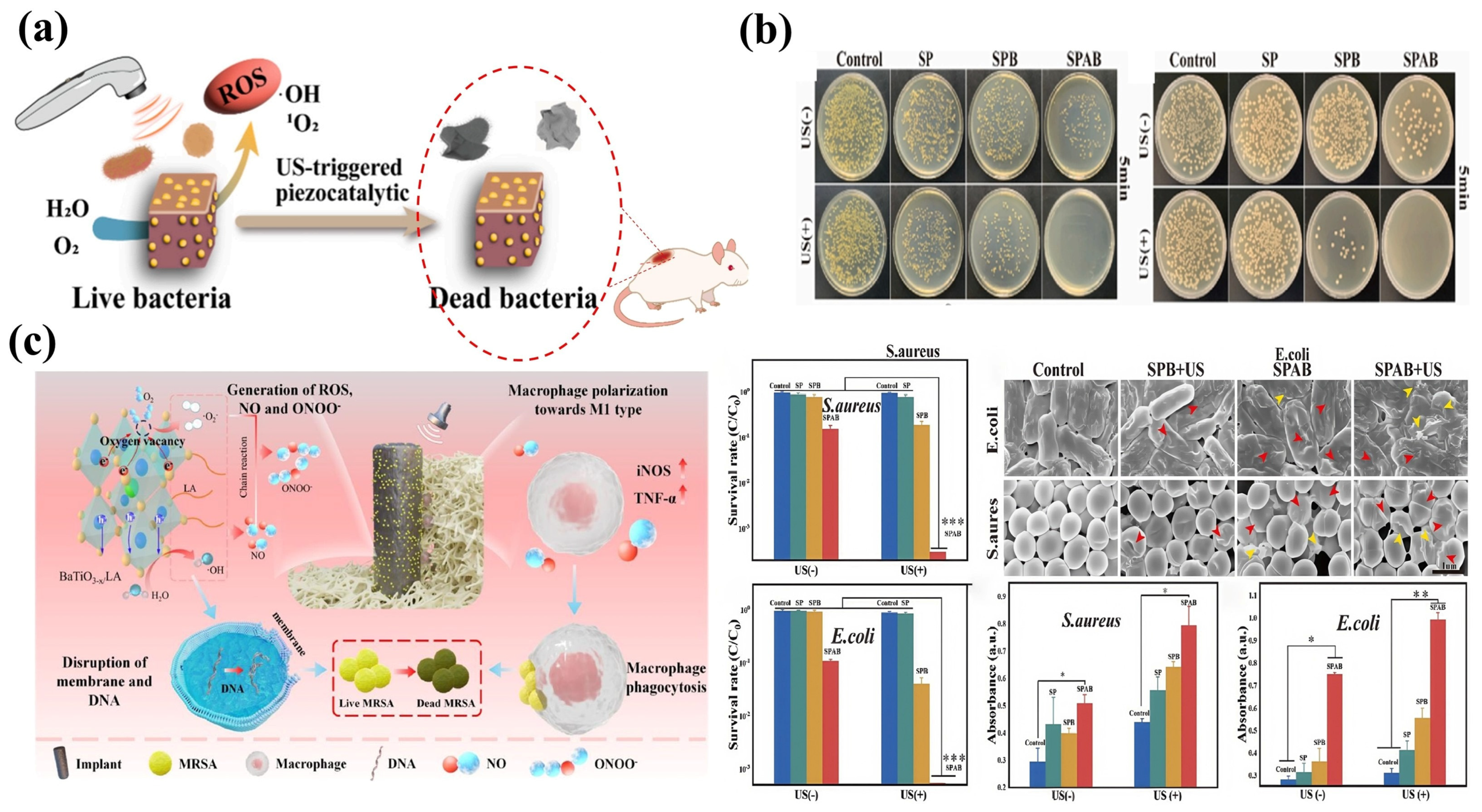
5.2. Biomedical Antimicrobial
6. Summary and Perspectives
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Juanola, A.; Mezzano, G.; Pose, E.; Moreta, M.J.; Incicco, S.; Gagliardi, R.; Johansen, S.; Torp, N.; Israelsen, M.; Jiménez-Esquivel, N.; et al. PD-L1 and the risk of bacterial infection in patients with chronic liver diseases: An international multicohort study. JHEP Rep. 2025, 7, 101597. [Google Scholar] [CrossRef] [PubMed]
- Javidnia, S.; Talebi, M.; Saifi, M.; Katouli, M.; Rastegar Lari, A.; Pourshafie, M.R. Clonal dissemination of methicillin-resistant Staphylococcus aureus in patients and the hospital environment. Int. J. Infect. Dis. 2013, 17, e691–e695. [Google Scholar] [CrossRef] [PubMed]
- Kehl, K.; Schallenberg, A.; Szekat, C.; Albert, C.; Sib, E.; Exner, M.; Zacharias, N.; Schreiber, C.; Parčina, M.; Bierbaum, G. Dissemination of carbapenem resistant bacteria from hospital wastewater into the environment. Sci. Total Environ. 2022, 806, 151339. [Google Scholar] [CrossRef]
- Lin, W.-S.; Chen, C.-L.; Liang, S.-W.; Wang, H.-C. Dementia Detection via Retinal Hyperspectral Imaging and Deep Learning: Clinical Dataset Analysis and Comparative Evaluation of Multiple Architectures. Bioengineering 2025, 12, 12121362. [Google Scholar] [CrossRef]
- Maniah, K.; Olyan Al-Otibi, F.; Mohamed, S.; Said, B.A.; Ragab AbdelGawwad, M.; Taha Yassin, M. Synergistic antibacterial activity of biogenic AgNPs with antibiotics against multidrug resistant bacterial strains. J. King Saud. Univ. Sci. 2024, 36, 103461. [Google Scholar] [CrossRef]
- Wolnicka-Glubisz, A.; Wisniewska-Becker, A. Dual Action of Curcumin as an Anti- and Pro-Oxidant from a Biophysical Perspective. Antioxidants 2023, 12, 12091725. [Google Scholar] [CrossRef]
- Yang, K.; He, Z. Formation of disinfection byproducts in electrochemical water disinfection. Curr. Opin. Environ. Sci. 2025, 48, 100685. [Google Scholar] [CrossRef]
- Zuma, F.; Lin, J.; Jonnalagadda, S.B. Ozone-initiated disinfection kinetics of Escherichia coli water. J. Environ. Sci. Health Part A 2012, 44, 48–56. [Google Scholar] [CrossRef]
- Ren, Y.; Li, S.; Lan, X.; Liu, P.; Li, Y.; Yin, W.; Du, W.; Yang, Q.; Ma, Y. Oxygen vacancy-rich N-doped Bi2WO6 for photocatalytic disinfection of Escherichia coli in juice. Appl. Surf. Sci. 2025, 705, 163513. [Google Scholar] [CrossRef]
- Li, N.; Zhu, B.; Huang, L.Q.; Huo, L.L.; Dong, Q.; Ma, J.Q. Piezoelectric Polarization and Sulfur Vacancy Enhanced Photocatalytic Hydrogen Evolution Performance of Bi2S3/ZnSn(OH)6 Piezo-photocatalyst. Inorg. Chem. 2024, 63, 10011–10021. [Google Scholar] [CrossRef]
- Wang, K.; Li, B.; Zhao, C.; Yuan, S.; Zhang, C.; Liang, X.; Wang, J.; Wu, Y.; He, Y. A novel NiO/BaTiO3 heterojunction for piezocatalytic water purification under ultrasonic vibration. Ultrason. Sonochem. 2023, 92, 106285. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.X.; Dong, Y.L.; Zhang, J.Y.; Tao, F.F.; Xu, J.J. Construction of NiO/g-C3N4 p-n heterojunctions for enhanced photocatalytic CO2 reduction. J. Solid. State Chem. 2022, 308, 122878. [Google Scholar] [CrossRef]
- Tu, S.; Guo, Y.; Zhang, Y.; Hu, C.; Zhang, T.; Ma, T.; Huang, H. Piezocatalysis and piezo-photocatalysis: Catalysts classification and modification strategy, reaction mechanism, and practical application. Adv. Func. Mater. 2020, 30, 2005158. [Google Scholar] [CrossRef]
- Feng, K.; Zhang, Y.; Zhou, X.; Zhao, Y.; Gong, H.; Zhou, X.; Bowen, C. Tailoring of PVDF for retrieval of piezoelectric powders to optimize piezo-catalytic water treatment. J. Mater. Chem. A 2024, 12, 23518–23529. [Google Scholar] [CrossRef]
- Cao, J.; Zhou, H.; Huang, C.; Wu, Q.; Yao, W. ZIF-8-derived Zn, N-codoped porous carbon as a high-performance piezocatalyst for organic pollutant degradation and hydrogen production. J. Colloid. Interface Sci. 2023, 645, 794–805. [Google Scholar] [CrossRef]
- Wei, Y.; Zhang, Y.; Geng, W.; Su, H.; Long, M. Efficient bifunctional piezocatalysis of Au/BiVO4 for simultaneous removal of 4-chlorophenol and Cr(VI) in water. Appl. Catal. B Environ. 2019, 259, 118084. [Google Scholar] [CrossRef]
- Yingnakorn, T.; Gordon, R.; Florido, D.M.; Elgar, C.E.; Jacobson, B.; Li, S.; Prentice, P.; Abbott, A.P.; Yang, J.M. Fast Delamination of Fuel Cell Catalyst-Coated Membranes Using High-Intensity Ultrasonication. Ultrason. Sonochem. 2025, 116, 107330. [Google Scholar] [CrossRef]
- Ju, S.; Trujillo, F.J.; Gong, Z.; Rezaeimotlagh, A.; Cullen, P.J. Synergy of oxygen plasma bubbles with ultrasonic waves for Escherichia coli inactivation. Innov. Food Sci. Emerg. Technol. 2025, 104, 104106. [Google Scholar] [CrossRef]
- Luo, Y.; Du, Y.; Gu, Z. Rethinking bacterial membrane models in molecular modeling: Is the cell wall dispensable in nanomaterial-bacterial membrane interaction. Mater. Des. 2025, 260, 115245. [Google Scholar] [CrossRef]
- Alashrah, M.S.; Kouki, N.; Algreiby, A.; Tar, H.; Béji, L. Study of basic red 12 transport through a liquid membrane cell using polypropylene nonwoven membranes in aqueous solution. Chem. Eng. J. Adv. 2025, 24, 100903. [Google Scholar] [CrossRef]
- Fu, Y.; Cui, N.; Zhang, Q.; Gu, Y.; Zhang, J.; An, Y.; Hu, X.; Qu, Z. Attachment detection on the inner wall of gas-liquid two phase flow pipe based on ultrasonic guided waves. J. Build. Eng. 2024, 97, 110894. [Google Scholar] [CrossRef]
- Li, J.; Chen, S.; Luo, J.; Xu, W.; Tang, J.; Qu, T. Shockwaves from air bubbles within pits induced by nearby cavitation bubbles. Ultrason. Sonochem. 2025, 122, 107602. [Google Scholar] [CrossRef] [PubMed]
- Zevnik, J.; Dular, M. Cavitation bubble interaction with compliant structures on a microscale: A contribution to the understanding of bacterial cell lysis by cavitation treatment. Ultrason. Sonochem. 2022, 87, 106053. [Google Scholar] [CrossRef]
- Ci, Y.; Tianzhi, W.; Peiyuan, L.; Manuel, F.; Suxia, G.; Yujie, Z. Disinfection mechanism of chlorine-resistant bacteria by micro-nano bubbles in drinking water: A case study of Bacillus cereus. Chem. Eng. J. 2025, 515, 163782. [Google Scholar] [CrossRef]
- Ci, Y.; Tianzhi, W.; Ziyi, L.; Guantong, Z.; Peiyuan, L. Disinfection mechanism of micro-nano bubbles on Bacillus cereus in drinking water under ultraviolet irradiation. Chem. Eng. J. 2026, 527, 171737. [Google Scholar] [CrossRef]
- Fitriyanti, M.; Tanuwijaya, A.; Hakim, B.; Herwanto, M.H.; Budiman, B.A.; Nurprasetio, I.P.; Sambegoro, P. Bacterial inactivation mechanisms under impact loading generated by ultrasonic cavitation. Mech. Res. Commun. 2025, 150, 104569. [Google Scholar] [CrossRef]
- Zevnik, J.; Dular, M. Cavitation bubble interaction with a rigid spherical particle on a microscale. Ultrason. Sonochem. 2020, 69, 105252. [Google Scholar] [CrossRef] [PubMed]
- Liang, T.; Lan, Y.-T.; Liang, M.-Y. Review of microstructure evolution and force-electric coupling behavior of piezoelectric materials. Mater. Today Commun. 2025, 47, 113027. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, J.; Su, Y.; Gong, H.; Han, X.; Ma, S.; Sun, D.; Liu, Y.; Chen, W.; Zhang, M.; et al. Polydopamine-mediated interfacial engineering of piezoelectric materials: A review of physicochemical mechanisms and applications. Mater. Today Commun. 2026, 50, 114484. [Google Scholar] [CrossRef]
- Freire, V.; Lattanzio, G.; Orera, I.; Mañas, P.; Cebrián, G. Component release after exposure of Staphylococcus aureus cells to pulsed electric fields. Innov. Food Sci. Emerg. Technol. 2021, 74, 102838. [Google Scholar] [CrossRef]
- Roy, D.; Michalet, X.; Miller, E.W.; Bharadwaj, K.; Weiss, S. Toward measurements of absolute membrane potential in Bacillus subtilis using fluorescence lifetime. Biophys. Rep. 2025, 5, 100196. [Google Scholar] [CrossRef]
- Ali, T.; Warsi, M.F.; Zulfiqar, S.; Sami, A.; Ullah, S.; Rasheed, A.; Alsafari, I.A.; Agboola, P.O.; Shakir, I.; Baig, M.M. Green nickel/nickel oxide nanoparticles for prospective antibacterial and environmental remediation applications. Ceram. Int. 2022, 48, 8331–8340. [Google Scholar] [CrossRef]
- Truong Hoang, Q.; Ravichandran, V.; Nguyen Cao, T.G.; Kang, J.H.; Ko, Y.T.; Lee, T.I.; Shim, M.S. Piezoelectric Au-decorated ZnO nanorods: Ultrasound-triggered generation of ROS for piezocatalytic cancer therapy. Chem. Eng. J. 2022, 435, 135039. [Google Scholar] [CrossRef]
- Huang, J.; Dong, H.; An, L.; Zhu, M.; Qin, J. Al-based metal-organic framework for piezocatalytic hydrogen peroxide production: Efficiency, pathway, and mechanism. Chem. Eng. J. 2025, 504, 158555. [Google Scholar] [CrossRef]
- Yue, J.; Wu, R.; Zhang, Y.; Zhang, N.; Jing, H.; Wei, S.; Ouyang, F. The piezoelectric field-induced rearrangement of free carriers unlocks the high redox ability of 1T@2H-MoS2/Bi2S3 piezoelectric catalyst. Appl. Surf. Sci. 2023, 623, 157033. [Google Scholar] [CrossRef]
- Shuai, C.; Pan, G.; Wang, Z.; He, T.; Shuai, X.; Zhong, Q.; Peng, S. Bifunctional MoS2@Cu2O heterojunction within scaffold for dual-mode synergistic antibacterial effects. Appl. Surf. Sci. 2025, 686, 162154. [Google Scholar] [CrossRef]
- Zhai, S.; Guo, H.; Sun, T.; Chen, J.; Guo, M.; Chen, G. 3-Methyl-1-butanol inhibited gray mold of red grape by damaging cell membrane integrity and the antioxidant capacity of Botrytis cinerea under oxidative stress. LWT 2025, 231, 118328. [Google Scholar] [CrossRef]
- He, J.; Hong, M.; Xie, W.; Chen, Z.; Chen, D.; Xie, S. Progress and prospects of nanomaterials against resistant bacteria. J. Control. Release 2022, 351, 301–323. [Google Scholar] [CrossRef]
- Han, S.W.; Kim, Y.; Lee, K.H.; Chae, S. Biomimetic piezocatalysts: A novel approach to water purification using TiO2/PVDF-TrFE. Results Eng. 2025, 28, 107438. [Google Scholar] [CrossRef]
- Zhang, T.; Jin, Y.; Yu, R.; Li, Y.; Wu, H. Ultrasonic cavitation in medicine: Current status and applications. WFUMB Ultrasound Open 2025, 36, 100100. [Google Scholar] [CrossRef]
- Liu, Y.; Luo, J.; Bai, L.; Hu, J. Ball motion and bubble ripples in the interaction of cavitation bubble-elastic ball-curved wall. Ultrason. Sonochem. 2025, 117, 107348. [Google Scholar] [CrossRef]
- Meng, J.; Yang, B.; Ren, L.; Yang, F.; Li, J.; Wang, J. Amino acid structure-oriented pyrolysis nitrogen evolution mechanism: Multi-scale experiment and DFT simulation. Chem. Eng. J. 2025, 519, 10732. [Google Scholar] [CrossRef]
- Du, W.; Xu, X.; Yang, R.; Qiao, Y.; Yu, Y.; Tao, L.; Hu, J.; Huang, Y.; Zhu, M. Amino-enhanced MIL-101(Fe) piezocatalysis for in-situ hydrogen peroxide generation and selective production of 1O2 for organic pollutants degradation. Chem. Eng. J. 2025, 519, 345. [Google Scholar] [CrossRef]
- Piñeros, W.D.; Tlusty, T. Spontaneous chiral symmetry breaking in a random driven chemical system. Nat. Commun. 2022, 13, 2244. [Google Scholar] [CrossRef] [PubMed]
- Lemanov, V.V. Piezoelectric and pyroelectric properties of protein amino acids as basic materials of Soft State Physics. Ferroelectrics 2000, 238, 211–218. [Google Scholar] [CrossRef]
- Takashima, S. Computation of the dipole moment of protein molecules using protein databases.: Bacteriophage T4 lysozyme and its mutants. Colloids Surf. A Physicochem. Eng. Asp. 1999, 148, 95–106. [Google Scholar] [CrossRef]
- Bohmbach, K.; Bauer, V.; Henneberger, C. Glycine and glycine transport control dendritic excitability and spiking. Prog. Neurobiol. 2026, 256, 11325. [Google Scholar] [CrossRef] [PubMed]
- Heredia, A.; Meunier, V.; Bdikin, I.K.; Gracio, J.; Balke, N.; Jesse, S.; Tselev, A.; Agarwal, P.K.; Sumpter, B.G.; Kalinin, S.V.; et al. Nanoscale Ferroelectricity in Crystalline γ-Glycine. Adv. Funct. Mater. 2012, 22, 2996–3003. [Google Scholar] [CrossRef]
- Guerin, S.; Stapleton, A.; Chovan, D.; Mouras, R.; Gleeson, M.; McKeown, C.; Noor, M.R.; Silien, C.; Rhen, F.M.F.; Kholkin, A.L.; et al. Control of piezoelectricity in amino acids by supramolecular packing. Nat. Mater. 2017, 17, 180–186. [Google Scholar] [CrossRef]
- Nascimento, L.; Richardson, G.; Melo, P.; Barroca, N. The hidden power of glycine: A small amino acid with huge potential for piezoelectric and piezo-triboelectric nanogenerators. Chem. Eng. J. 2025, 510, 34236. [Google Scholar] [CrossRef]
- Cheng, Y.; Wang, T.; Zhu, H.; Hu, X.; Mi, J.; Li, L.; Zhang, Y.; Yang, J.; Dong, L.; Li, Y.; et al. Molecular Engineering of Amino Acid Crystals with Enhanced Piezoelectric Performance for Biodegradable Sensors. Angew. Chem. Int. Ed. 2025, 64, 12897. [Google Scholar]
- Salama, A.; Al Kiey, S.A.; Saleh, A.K. Pyridinium@cellulose as a green antimicrobial material with tailored dielectric behavior for bioelectronic interfaces. React. Funct. Polym. 2026, 219, 106566. [Google Scholar] [CrossRef]
- Nyah, F.; Ridzuan, N.; Epelle, E.; Bin Abd Aziz, M.A.; Money, B.; Abutu, D.; Agi, A. Cellulose Bionanomaterial Design for Enhanced Oil Recovery: A Review of Existing, Emerging Technologies and Future Outlook. Pet. Res. 2025, 60, 122768. [Google Scholar] [CrossRef]
- Bao, Y.; Si, Q.; Buck, D.; Wang, C.; Wang, S.; Zhang, F.; Gu, Q.; Feng, X.; Ma, H.; Wang, L.; et al. Sustainable regeneration of cellulose gels and fibration of powder materials via eco-friendly dissolution–regeneration process. Ind. Crops Prod. 2026, 239, 122486. [Google Scholar] [CrossRef]
- Cao, Z.; Luo, X.; Lv, J.; Wang, L. Space group informed transformer for crystalline materials generation. Sci. Bull. 2025, 70, 3522–3533. [Google Scholar] [CrossRef]
- Fukada, E. Piezoelectricity as a fundamental property of wood. Wood Sci. Technol. 1968, 2, 299–307. [Google Scholar] [CrossRef]
- Moon, R.J.; Martini, A.; Nairn, J.; Simonsen, J.; Youngblood, J. Cellulose nanomaterials review: Structure, properties and nanocomposites. Chem. Soc. Rev. 2011, 40, 15643. [Google Scholar] [CrossRef]
- Miao, C.; Reid, L.; Hamad, W.Y. Moisture-tunable, ionic strength-controlled piezoelectric effect in cellulose nanocrystal films. Appl. Mater. Today 2021, 24, 59. [Google Scholar] [CrossRef]
- Yoshimoto, S.; Sasahara, J.; Suzuki, A.; Kanie, J.; Koiwai, K.; Lupas, A.N.; Hori, K. Insights into the complex formation of a trimeric autotransporter adhesin with a peptidoglycan-binding periplasmic protein. Cell Surf. 2025, 14, 1223–1238. [Google Scholar] [CrossRef]
- Mauck, T.A.; Zacharias, M. Influence of Methionine Oxidation on Protein Stability and Association Studied by Free Energy Simulations. J. Mol. Biol. 2026, 438, 169576. [Google Scholar] [CrossRef]
- Loureiro, R.J.; Maiti, S.; Mondal, K.; Mukherjee, S.; Bujnicki, J.M. Modeling flexible RNA 3D structures and RNA-protein complexes. Curr. Opin. Struct. Biol. 2025, 94, 4365–4384. [Google Scholar] [CrossRef]
- Bagriantsev, S.N.; Gracheva, E.O.; Gallagher, P.G. Piezo Proteins: Regulators of Mechanosensation and Other Cellular Processes. J. Biol. Chem. 2014, 289, 31673–31681. [Google Scholar] [CrossRef]
- Gupta, S.; Sharma, S.; Kapoor, B. From waste to biomaterial: Valorization of keratin for 3D printing and bioprinting. Mater. Sci. Eng. B 2026, 325, 119135. [Google Scholar] [CrossRef]
- Huang, J.; Li, Y.; Huang, R.K.; He, C.T.; Gong, L.; Hu, Q.; Wang, L.; Xu, Y.T.; Tian, X.Y.; Liu, S.Y.; et al. Electrochemical Exfoliation of Pillared-Layer Metal–Organic Framework to Boost the Oxygen Evolution Reaction. Angew. Chem. Int. Ed. 2018, 57, 4632–4636. [Google Scholar] [CrossRef]
- Akdag, Z.; Izgordu, M.S.; Ayran, M.; Uzuner, A.; Sancakli, A.; Arican, F.; Ahi, Z.B.; Yildirim, R.; Gunduz, O. Methacrylated keratin biopolymer with tunable properties for advanced biomedical applications. Mater. Lett. 2026, 405, 139713. [Google Scholar] [CrossRef]
- Long, D.; Yang, Z.; Zhang, X.; Kundu, S.C.; Du, Y.; Zhang, Z.; Yang, C.; Dai, F. Advances in Silkworm Silk Proteins: From Textile to Biomedical Innovations. J. Resour. Insects 2025, 12, 100005. [Google Scholar] [CrossRef]
- Liang, X.; Xu, H.; Cong, H.; Wan, X.; Liu, L.; Li, Y.; Liu, C.; Chen, C.; Jiang, G.; Asadi, K.; et al. Robust Piezoelectric Biomolecular Membranes from Eggshell Protein for Wearable Sensors. ACS Appl. Mater. Interfaces 2023, 15, 55790–55802. [Google Scholar] [CrossRef]
- Li, S.; Li, Y.; Zhang, X.; Jiang, D.; Kong, L. Electrospinning of Chitosan-based Nanofibers: Innovations in Fabrication and Applications. J. Agric. Food Res. 2025, 13, 102599. [Google Scholar] [CrossRef]
- Dodangeh, F.; Pontillo, A.R.N.; Chen, S.; Welton, T.; Xu, C.; Rohani, S. Ionic liquid-regenerated cellulose–chitosan coatings: A Bio-based coating for enhancing paper packaging performance. Surf. Interfaces 2025, 80, 108342. [Google Scholar] [CrossRef]
- Derraz, M.; Radoine, H.; Boumegnane, A.; Ben Achour, M.A.; Ennawaoui, C.; Hajjaji, A. Development of a novel Bi4Ti3O12/chitosan/rGO piezoelectric bio-composite for mechanical energy harvesting: Output energy optimization using response surface methodology modelling. Ceram. Int. 2025, 51, 2660–2673. [Google Scholar] [CrossRef]
- Praveen, E.; Murugan, S.; Jayakumar, K. Investigations on the existence of piezoelectric property of a bio-polymer—Chitosan and its application in vibration sensors. RSC Adv. 2017, 7, 35490–35495. [Google Scholar] [CrossRef]
- Amran, A.; Ahmad, F.B.; Akmal, M.H.M.; Ralib, A.A.M.; Bin Suhaimi, M.I. Biosynthesis of thin film derived from microbial chitosan for piezoelectric application. Mater. Today Commun. 2021, 29, 102919. [Google Scholar] [CrossRef]
- Boon-on, P.; Raksa, P.; Ponhan, W.; Chaiworn, P.; Tubtimtae, A. Effect of Sn concentration on antimony oxide thin films via using spin coating method: Analysis of structural and optical properties. Mater. Lett. 2025, 404, 139579. [Google Scholar] [CrossRef]
- Sabriantie Mulus, D.A.; Permana, M.D.; Hayaa’ Prawiranegara, S.P.; Tustika, C.Z.; Putri, S.P.; Zahra, S.A.; Deawati, Y.; Eddy, D.R. Enhanced performance of spin-coated silver-modified titanium dioxide thin films over dip coating method for metformin photodegradation. Results Opt. 2025, 21, 100838. [Google Scholar] [CrossRef]
- Joseph, J.; Singh, S.G.; Vanjari, S.R.K. Leveraging Innate Piezoelectricity of Ultra-Smooth Silk Thin Films for Flexible and Wearable Sensor Applications. IEEE Sens. J. 2017, 17, 8306–8313. [Google Scholar] [CrossRef]
- Pandiyarajan, S.; Marimuthu, A.; Liao, A.-H.; Selvaraj, M.; Assiri, M.A.; Savarimuthu, S.; Treeratanaphitak, T.; Chuang, H.-C. Fabrication of corrosion resistant nickel/egg-shell derived hydroxyapatite composite coating using supercritical-CO2 electrodeposition: Influence of pressure and corrosion durability. Inorg. Chem. Commun. 2025, 184, 115896. [Google Scholar] [CrossRef]
- Song, R.; Zhu, Y.; Xue, K.; Deng, X.; Wang, R.; Su, Y.; Chen, X.; Yuan, H. A “controlled assembly” strategy for constructing Coptidis Rhizoma self-assembled nanoparticles to ameliorate inflammatory damage in Atopic dermatitis. Bioact. Mater. 2026, 56, 234–247. [Google Scholar] [CrossRef]
- Yuan, H.; Lei, T.; Qin, Y.; He, J.-H.; Yang, R. Design and application of piezoelectric biomaterials. J. Phys. D Appl. Phys. 2019, 52, 194002. [Google Scholar] [CrossRef]
- Zhang, Z.; Li, X.; Peng, Z.; Yan, X.; Liu, S.; Hong, Y.; Shan, Y.; Xu, X.; Jin, L.; Liu, B.; et al. Active self-assembly of piezoelectric biomolecular films via synergistic nanoconfinement and in-situ poling. Nat. Commun. 2023, 14, 4094. [Google Scholar] [CrossRef]
- Xiao, X.; Xie, B.; Ouyang, L.; Yang, J.; Li, C.; Zhao, J.; Lv, Y.; Zeng, X.; Li, C. Electrospinning of thermal interface materials. Adv. Colloid Interface Sci. 2025, 349, 103754. [Google Scholar] [CrossRef] [PubMed]
- Guerin, S.; Tofail, S.A.; Thompson, D. Organic piezoelectric materials: Milestones and potential. NPG Asia Mater. 2019, 11, 10. [Google Scholar] [CrossRef]
- Dani, S.S.; Sundaray, B.; Nayak, S.K.; Mohanty, S. Development of electrospinning-assisted flexible PVDF-Ba(1−x) Sr(x)TiO3 nanocomposite membrane for piezoelectric energy harvesting application. Surf. Interfaces 2025, 71, 312–356. [Google Scholar] [CrossRef]
- Guerin, S. Getting the lead out: Biomolecular crystals as low-cost, high-performance piezoelectric components. Acc. Mater. Res. 2022, 38, 782–784. [Google Scholar] [CrossRef]
- Zhu, G.; Yan, Z.; Yao, W.; Zhai, H.; Li, Q.; Fan, D. Scalable manufacture of ultratough physical cross-linked hybrid films for durable radiative cooling. Chem. Eng. J. 2025, 518, 345–367. [Google Scholar] [CrossRef]
- Zhang, H.; You, Z.; Wang, R.; Liang, J.; Song, Y.; Gao, Y.; Duan, Y.; Li, Z. Double cross-linked hydrogel electrolytes for flexible supercapacitors with mechanical stability, wide temperature adaptability and self-healing property. J. Energy Storage 2025, 130, 117333. [Google Scholar] [CrossRef]
- Zhang, C.; Kwon, S.H.; Huerta, A.; Jiang, Z.; Sun, M.; Dong, L. Self-Polarized Piezoelectric Hydrogels with Flexible-Rigid Networks for Ultrasensitive Multifunctional Sensors. ACS Mater. Lett. 2025, 7, 2607–2616. [Google Scholar] [CrossRef]
- Laxmanan, K.; Yadav, I.; Barani, P.K.; Meena, M.; Kang, H.W.; Dhanka, M. Structurally interlinked multi-crosslinking bioactive hydrogel network with enhanced antioxidant, antibiofilm, and antibacterial functionalities. Mater. Today Commun. 2025, 46, 112936. [Google Scholar] [CrossRef]
- Du, Y.; Wu, T.; Liu, C.; Che, H.; Liu, B.; Ao, Y. Breaking activity-transport limits in photocatalysis: 3D cyanide-rich hydrogel membranes via radical polymerization for solar-powered wastewater purification. Chem. Eng. J. 2025, 521, 166682. [Google Scholar] [CrossRef]
- Dumitrescu, C.R.; Neacsu, I.A.; Trusca, R.; Popescu, R.C.; Raut, I.; Constantin, M.; Andronescu, E. Piezoelectric Biocomposites for Bone Grafting in Dentistry. Polymers 2023, 15, 234–245. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.-C.; Lu, J.-Q.; Zhang, T.-W.; Liang, H.-F.; Yuan, H.; Su, D.-H.; Ding, W.; Lian, R.-X.; Ge, Y.-X.; Liang, B.; et al. Piezoresistive MXene/Silk fibroin nanocomposite hydrogel for accelerating bone regeneration by Re-establishing electrical microenvironment. Bioact. Mater. 2023, 22, 1–17. [Google Scholar] [CrossRef]
- Mondal, D.; Bardhan, S.; Das, N.; Roy, J.; Ghosh, S.; Maity, A.; Roy, S.; Basu, R.; Das, S. Natural clay-based reusable piezo-responsive membrane for water droplet mediated energy harvesting, degradation of organic dye and pathogenic bacteria. Nano Energy 2022, 104, 107893. [Google Scholar] [CrossRef]
- Xu, M.; Yang, S.; Guo, C.; DuBois, D.; Chen, S.; Meng, F. Bubble-triggered piezocatalytic generation of hydrogen peroxide by copper nanosheets-modified polyvinylidene fluoride films for organic pollutant degradation and water disinfection. Water Res. 2025, 283, 123865. [Google Scholar] [CrossRef]
- Sardana, S.; Agrawal, S.; George, B.P.; Abrahamse, H.; Sarbadhikary, P. Applications of synthetic non-porphyrinoid photosensitizers for antimicrobial photodynamic disinfection of natural water systems. Dye. Pigment. 2026, 246, 113308. [Google Scholar] [CrossRef]
- DuBois, D.B.; Tressel, J.; Hergenroeder, D.; Jones, C.; Yu, B.; Singewald, K.; Millhauser, G.L.; Saltikov, C.; Chen, S. Antibacterial activity of bismuth tungstate against Escherichia coli: Enhanced piezocatalysis by morphological engineering. J. Environ. Chem. Eng. 2025, 13, 118871. [Google Scholar] [CrossRef]
- Fadhil, A.; Sriwidodo; Elamin, K.M.; Mohammed, A.F.A.; Mahmoud, S.A.; Wathoni, N. Study of various forms of propolis nanoparticles and their antibacterial effectiveness. OpenNano 2026, 27, 100276. [Google Scholar] [CrossRef]
- Huo, B.; Wang, J.; Wang, Z.; Liu, C.; Hao, W.; Wang, Y.; Cui, P.; Qi, J.; Gao, J.; Yang, J. Ni-doped MoS2 embedded in natural wood containing porous cellulose for piezo-catalytic degradation of tetracycline. Int. J. Biol. Macromol. 2023, 233, 123589. [Google Scholar] [CrossRef]
- Liu, Y.; Roy, J.; Roy, S.; Hoque, N.A.; Guo, B. Highly efficient piezocatalytic composite with chitosan biopolymeric membranes and bismuth ferrite nanoparticles for dye decomposition and pathogenic S. aureus bacteria killing. Front. Chem. 2024, 12, 1420040. [Google Scholar] [CrossRef] [PubMed]
- Mondal, D.; Roy, S.; Sau, A.; Roy, J.; Bag, N.; Ullah, Z.; Ghosh, S.; Gong, T.; Madni, M.; Chakraborty, I. Chitosan cloaked MWCNT-kaolinite bio-nanocomposite for energy generation and ultrasound driven ROS induced degradation of organic dyes and pathogen. Ceram. Int. 2025, 51, 45744–45754. [Google Scholar] [CrossRef]
- El-Sayed, S.E.; Messiha, A.A.; Zafer, M. Effective alternative strategies to combat challenges associated with MDR bacterial infections: Drug repurposing, role of artificial intelligence, and novel therapeutic options. J. Infect. Public Health 2026, 19, 103058. [Google Scholar] [CrossRef]
- Vinikoor, T.; Dzidotor, G.K.; Le, T.T.; Liu, Y.; Kan, H.-M.; Barui, S.; Chorsi, M.T.; Curry, E.J.; Reinhardt, E.; Wang, H. Injectable and biodegradable piezoelectric hydrogel for osteoarthritis treatment. Nat. Commun. 2023, 14, 8694. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Wang, J.; Huang, X.; Hang, R.; Han, P.; Guo, J.; Yao, X.; Chu, P.K.; Zhang, X. Ultrasound-driven radical chain reaction and immunoregulation of piezoelectric-based hybrid coating for treating implant infection. Biomaterials 2024, 307, 122532. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wang, C.; Zhang, Z.; Yu, F.; Wang, Y.; Ding, J.; Zhao, Z.; Liu, Y. 3D-printed piezocatalytic hydrogels for effective antibacterial treatment of infected wounds. Int. J. Biol. Macromol. 2024, 268, 131637. [Google Scholar] [CrossRef] [PubMed]





Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Mu, R.; Shi, X.; Chen, W.; Zhang, K. Advancements in Bio-Based Piezoelectric Composites for Antibacterial Applications. Bioengineering 2026, 13, 290. https://doi.org/10.3390/bioengineering13030290
Mu R, Shi X, Chen W, Zhang K. Advancements in Bio-Based Piezoelectric Composites for Antibacterial Applications. Bioengineering. 2026; 13(3):290. https://doi.org/10.3390/bioengineering13030290
Chicago/Turabian StyleMu, Ruihua, Xiaoqian Shi, Wenzhuo Chen, and Kaige Zhang. 2026. "Advancements in Bio-Based Piezoelectric Composites for Antibacterial Applications" Bioengineering 13, no. 3: 290. https://doi.org/10.3390/bioengineering13030290
APA StyleMu, R., Shi, X., Chen, W., & Zhang, K. (2026). Advancements in Bio-Based Piezoelectric Composites for Antibacterial Applications. Bioengineering, 13(3), 290. https://doi.org/10.3390/bioengineering13030290






