Bioengineered 3D Human Trabecular Meshwork Models for Outflow Physiology and Glaucoma Research
Abstract
1. Introduction
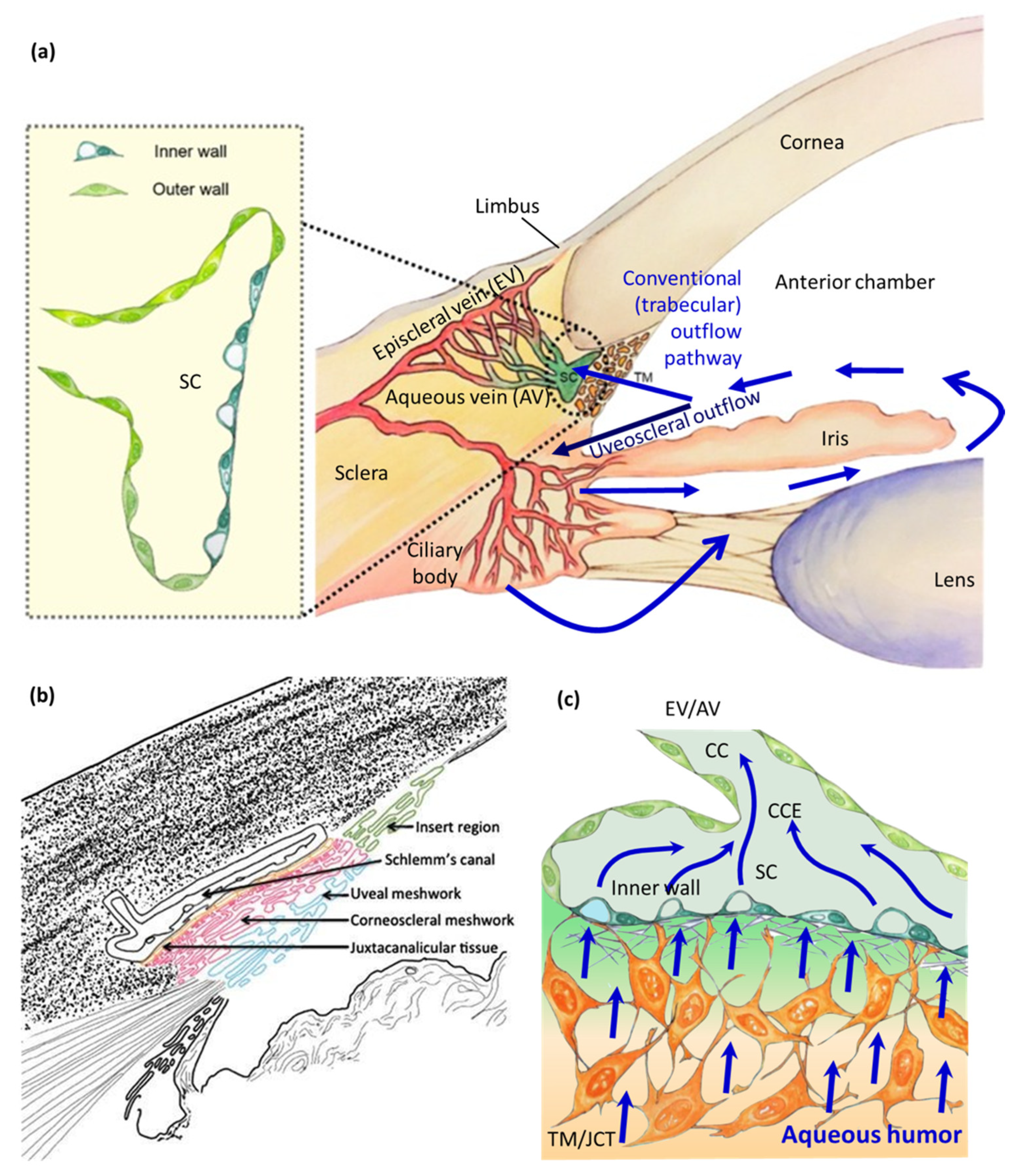
2. Hydrogel-Based HTM Models
2.1. HTM Culture on Synthetic Polyacrylamide Hydrogels
2.2. HTM Culture in Matrigel
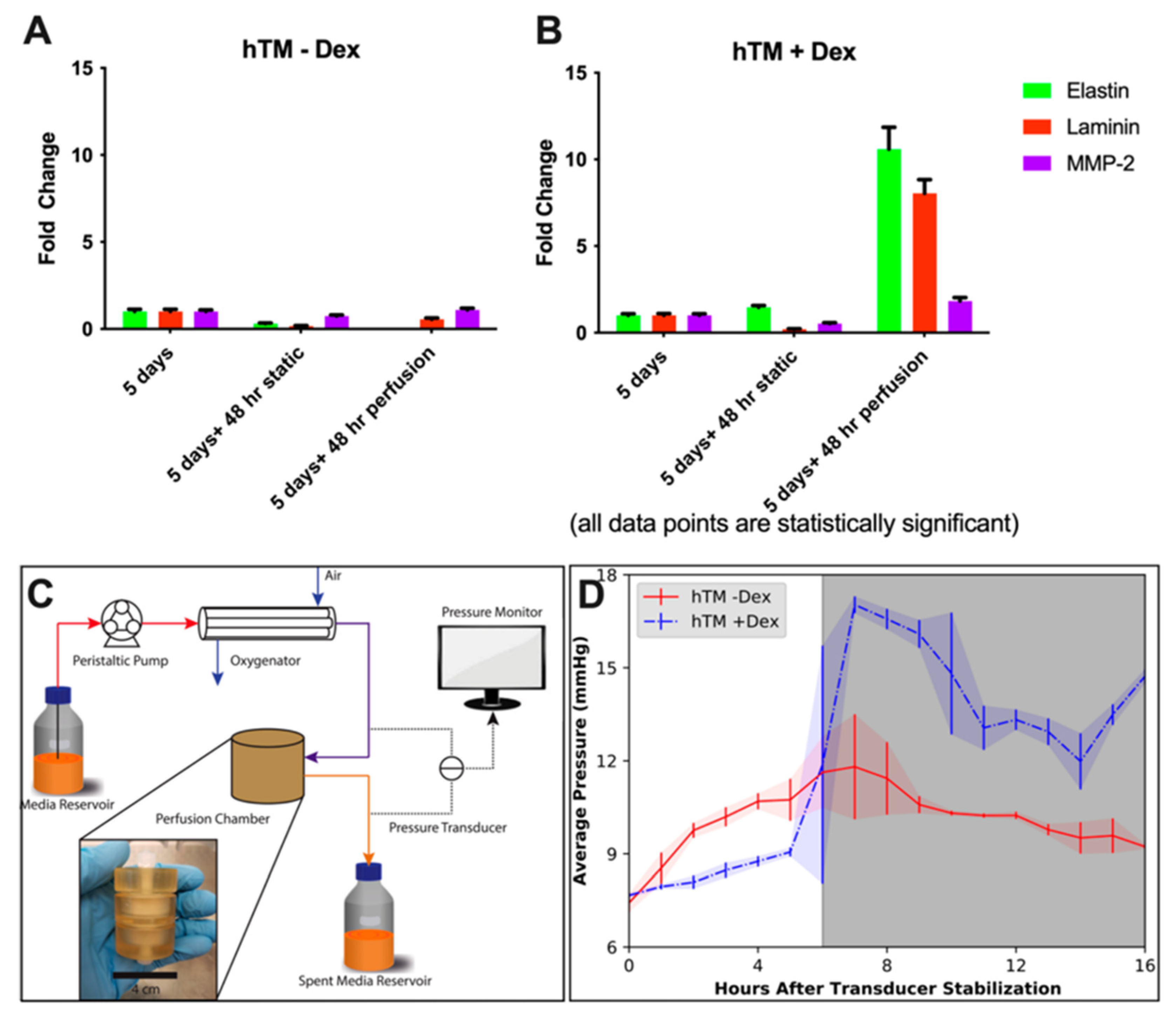
2.3. HTM Culture in Collagen-Based Hydrogels
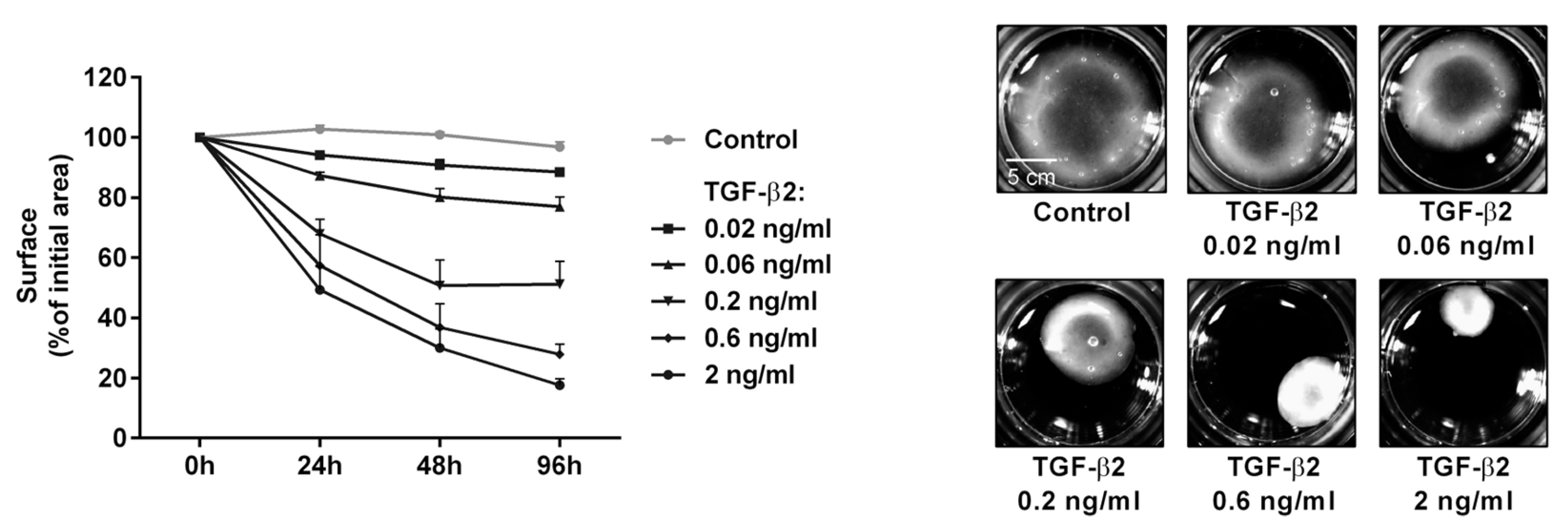
2.4. HTM Culture in Hybrid Mechanoelectric Transducing Hydrogels
2.5. HTM Culture in Self-Assembed Peptide Hydrogels
2.6. HTM Culture on Decellularized Extracellular Matrices
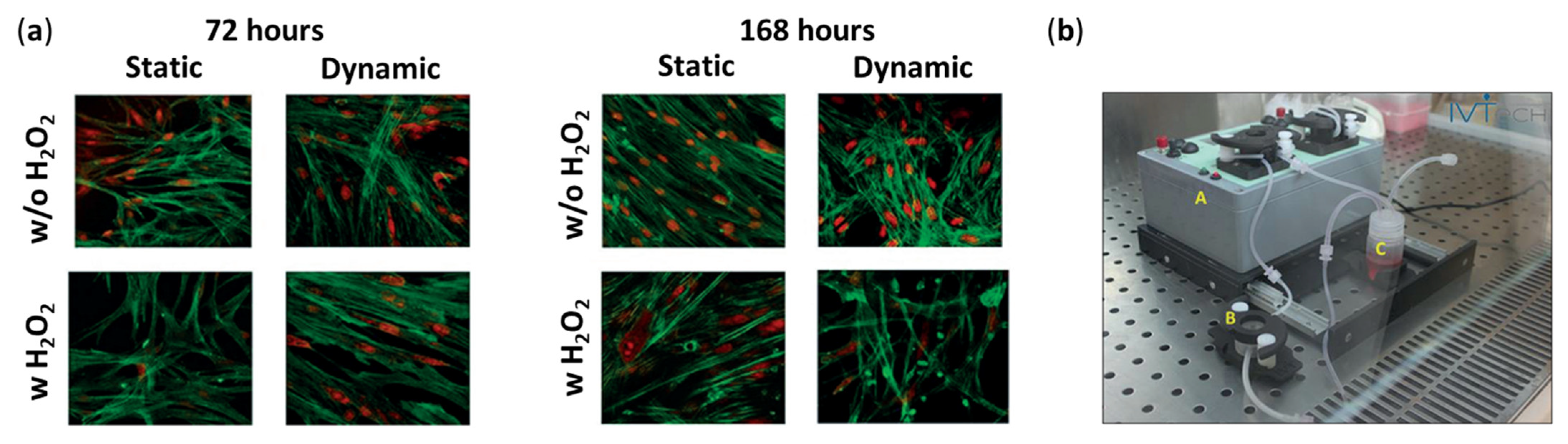
2.7. Limitations of Hydrogel-Based HTM Models for Perfusion Studies
3. Micro- and Nanofabricated Substrate-Based HTM Models
3.1. Micro- and Nanopatterned Nonporous Polyurethane Surfaces for HTM Culture
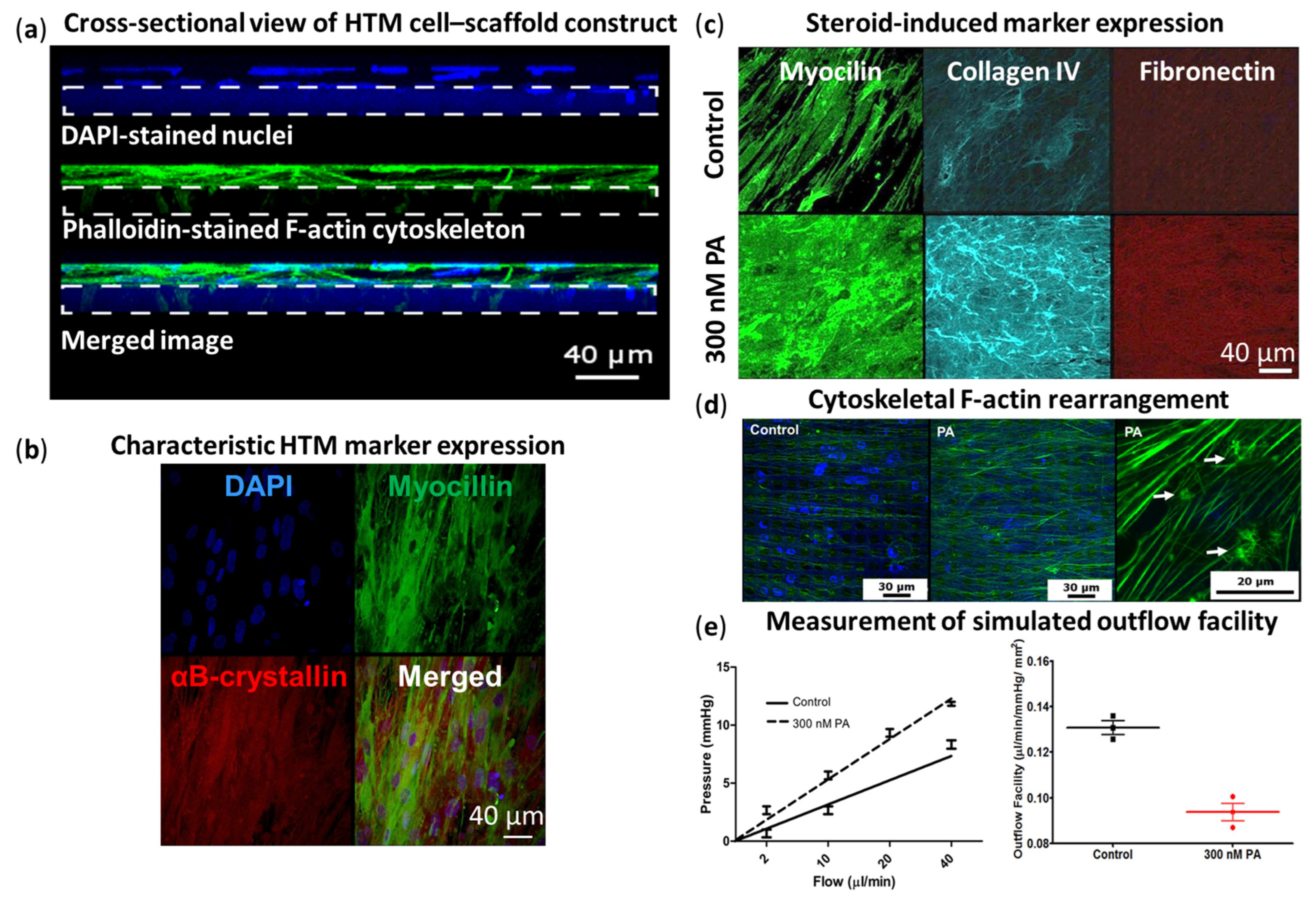
3.2. Micropatterned Porous SU-8 Scaffolds for HTM and HSC Culture to Develop Artificial Conventional Outflow Systems (ACOSs)
3.3. Porous PCL Scaffolds for HTM Culture
| HTM Structure | Fabrication Method | Thickness | Fiber Diameter | Pore Size/ Porosity | Mechanical Properties | Refs. |
|---|---|---|---|---|---|---|
| JCT | Native | 2–20 μm | 5–12 μm | 4–7 µm | [138,139,140] | |
| CSM | 40–60 µm | Lamellar | 30 µm | |||
| UVM | 15–20 μm | beams | 70–100 µm | |||
| HTM | 70–130 µm | ∼4 kPa (elastic modulus) | ||||
| 515 kPa (tensile testing) | ||||||
| JCT-like | Photolithography (SU-8 scaffolds) | 20 µm | 7 µm | 12 µm | [129] | |
| JCT-like | Electrospinning (PCL scaffolds) | 20.3 μm | 0.770 µm | 5.6 μm2 | 0.95 ± 0.05 MPa (elastic modulus) | [136] |
| JCT-like | Electrospinning (PCL scaffolds) | 30.4 µm | 0.600 µm | 3.3 µm/ 70.9% | Young’s modulus/tensile modulus 5.15 ± 0.55 MPa/0.17 ± 0.02 MPa | [137] |
| Cryoelectrospinning (PCL scaffolds) | 76.9 µm | 0.430 µm | 8.5 µm/ 91.9% | 0.79 ± 0.24 MPa/0.03 ± 0.01 MPa | ||
| Compression modulus/tensile | [138] | |||||
| JCT-like | MEW | 125 µm | 10.0 µm | 86.8% | 11.2 ± 3.3 kPa/13.0 ± 1.7 MPa | |
| CSM-like | (PCL scaffolds) | 299 µm | 10.2 µm | 84.7% | 87.9 ± 75.6 kPa/7.2 ± 2.1 MPa | |
| UVM-like | 140 µm | 11.8 µm | 91.2% | 63.8 ± 79.9 kPa/7.2 ± 2.1 MPa | ||
| Full HTM | 506 µm | 11.9 µm | 84.2% | 358 ± 235 kPa/6.9 ± 1.1 MPa | ||
| Elastic modulus/yield stress | [141] | |||||
| JCT-like | Electrospinning | 20 µm | 0.770 µm | 5.6 µm2 | 0.94 ± 0.05 MPa/2.84 ± 0.20 MPa | |
| CSM-like | MEW | 610 µm | 29.1 µm | 0.75 mm | 0.18 ± 0.01 MPa/0.39 ± 0.03 MPa | |
| UVM-like | Combined | 260 µm | 37.5 µm | 0.86 mm | 0.14 ± 0.01 MPa/0.24 ± 0.04 MPa | |
| Full HTM | (PCL scaffolds) | 510 µm | 0.29 ± 0.03 MPa/0.65 ± 0.22 MPa | |||
| JCT-like | MEW | 36 µm | 8.0 µm | 50 µm | 6 MPa Young’s modulus | [142] |
| CSM-like | (PCL scaffolds) | 52 µm | 10.2 µm | 88 µm | 23 MPa Young’s modulus | |
| UVM-like | 76 µm | 29.2 µm | 130 µm | 17 MPa Young’s modulus | ||
| Full HTM | 164 µm | 8.0–29.2 µm | 50.1–131 µm | 23 MPa Young’s modulus |

4. Scaffold-Free HTM Spheroids and Organoids
5. Clinically Relevant 3D Outflow Pathway Models Using Stem Cell-Derived HTM/HSC Cells
5.1. Need for Stem Cell-Derived HTM and HSC Cells
5.2. Role of Stem Cell-Derived HTM and HSC Cells for TM Function Regulation
5.3. Stem Cell-Derived ACOS for Simulated Outflow Facility
6. HTM Models for Outflow Physiology Studies
6.1. Outflow Resistance, Hydraulic Conductivity, and Outflow Facility
6.2. Bioengineered ACOS for Simulating Outflow Facility Responses to IOP-Modulating Agents
6.3. Limitations of PCL Scaffold-Based HTM Models for Perfusion Studies
6.4. Traditional HTM Cell Monolayer Culture on Filter Membranes for Perfusion Studies to Measure Hydraulic Conductivity
6.5. 3D HTM/HSC Cultures in Microfluidic Devices for Assessment of Outflow Rate
7. Summary and Future Outlook
- -
- Development of advanced multicellular co-culture systems that more accurately recapitulate the anatomy of the conventional outflow pathway.
- -
- Use of patient-specific, stem cell-derived HTM and HSC models.
- -
- Advancement of HTM models that replicate chronic ECM remodeling and aging.
- -
- Utilization of dynamic, pressure-controlled or flow-controlled hybrid scaffold-based or microfluidics-based platforms for evaluation of the HTM phenotype and simulated outflow facility.
- -
- Integration of omics with outflow physiology to elucidate mechanisms underlying HTM function.
- -
- Implementation of high-throughput drug screening with respect to changes in TM biomechanics and simulated outflow facility to reveal mechanisms of IOP-modulation and identify therapeutic targets.
- -
- Development of biomimetic, implantable, and regenerative scaffolds.
- -
- Standardization, benchmarking, and validation against ex vivo, preclinical, and clinical data.
- -
- Data integration and AI-enabled functional modeling.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 2D | Two-dimensional |
| 3D | Three-dimensional |
| αSMA | α-smooth muscle actin |
| ABCG2+ | ATP-binding cassette super-family G member 2-positive |
| ACOS | Artificial conventional outflow system |
| ADSC | Adipose-derived mesenchymal stem cell |
| AH | Aqueous humor |
| AM | Acrylamide |
| ANGPTL7 | Angiopoietin-like 7 |
| AQP | Aquaporin |
| AV | Aqueous vein |
| BM-MSC | Bone marrow-derived mesenchymal stem cell |
| CC | Collector channels |
| CCEs | Collector channel entrances |
| CCL | Chemokine |
| CHI3L1 | Chitinase-3-like protein 1 |
| CLAN | Crosslinked actin network |
| COL1A1 | Collagen type I alpha 1 chain |
| COL4A5 | Collagen type IV alpha 5 chain |
| COLI/IV | Collagen type I and collagen type IV |
| COLIV | Collagen type IV |
| CS | Chondroitin sulfate |
| CSM | Corneoscleral meshwork |
| CTGF | Connective tissue growth factor |
| CXCR4 | C-X-C chemokine receptor type 4 |
| CYP1B1 | Cytochrome P450 family 1 subfamily B member 1 |
| DEX | Dexamethasone |
| ECM | Extracellular matrix |
| EDTA | Ethylenediaminetetraacetic acid |
| EP2 | Prostaglandin E2 receptor 2 |
| ER | Endoplasmic reticulum |
| ERK | Extracellular signal regulated kinase |
| EV | Episcleral vein |
| FBS | Fetal bovine serum |
| FN | Fibronectin |
| FP | Prostaglandin F prostanoid receptor |
| GAG | Glycosaminoglycan |
| GelMA | Gelatin methacryloyl |
| GMPs | Good manufacturing practices |
| GTM | HTM cells derived from donors with glaucoma |
| HA | Hyaluronic acid |
| HNK1 | Human natural killer-1 antigen |
| HSC | Human Schlemm’s canal |
| HTM | Human trabecular meshwork |
| IL | Interleukin |
| IOP | Intraocular pressure |
| iPSC | Induced pluripotent stem cell |
| iPSC-TM | Induced pluripotent stem cell-derived trabecular meshwork |
| JCT | Juxtacanalicular connective tissue |
| Lat-B | Latrunculin B |
| LEC | Lymphatic endothelial cell |
| LTBP2 | Latent transforming growth factor beta binding protein 2 |
| MCP | Monocyte chemoattractant protein |
| MEW | Melt electrowriting |
| MGP | Matrix Gla protein |
| MSC | Mesenchymal stem cell |
| MYOC | Myocilin |
| NCCs | Neural crest cells |
| NGFR | Nerve growth factor receptor |
| PA | Prednisolone acetate |
| PCL | Poly-ε-caprolactone/polycaprolactone |
| PDMS | Polydimethylsiloxane |
| p-MLC | Phosphorylated myosin light chain |
| POAG | Primary open-angle glaucoma |
| RGCs | Retinal ganglion cells |
| ROCK | Rho-associated protein kinase |
| ROS | Reactive oxygen species |
| SC | Schlemm’s canal |
| SDF1 | Stromal cell-derived factor 1 |
| SEM | Scanning electron microscopy |
| sEVs | Small extracellular vehicles |
| siC | siRNA control |
| siRNA | Small interfering RNA |
| SPARC | Secreted protein acidic and rich in cysteine |
| STL | Stereolithography |
| TAZ | Transcriptional co-activator with PDZ-binding motif |
| TEER | Transepithelial electrical resistance |
| TGFβ | Transforming growth factor beta |
| TGM | Transglutaminase |
| TIMP3 | Tissue inhibitor of metalloproteinases-3 |
| TM | Trabecular meshwork |
| TMSCs | Trabecular meshwork stem cells |
| TRPV4 | Transient receptor potential vanilloid 4 |
| UVM | Uveal meshwork |
| VCAM1 | Vascular cell adhesion molecule 1 |
| VEGF-C | Vascular endothelial growth factor-C |
| YAP | Yes-associated protein |
References
- Kolovos, A.; Maxwell, G.; Souzeau, E.; Craig, J.E. Progress in Translating Glaucoma Genetics Into the Clinic: A Review. Clin. Exp. Ophthalmol. 2025, 53, 246–259. [Google Scholar] [CrossRef] [PubMed]
- Ehrlich, J.R.; Burke-Conte, Z.; Wittenborn, J.S.; Saaddine, J.; Omura, J.D.; Friedman, D.S.; Flaxman, A.D.; Rein, D.B. Prevalence of Glaucoma Among US Adults in 2022. JAMA Ophthalmol. 2024, 142, 1046–1053. [Google Scholar] [CrossRef]
- Vajaranant, T.S.; Wu, S.; Torres, M.; Varma, R. The Changing Face of Primary Open-Angle Glaucoma in the United States: Demographic and Geographic Changes from 2011 to 2050. Am. J. Ophthalmol. 2012, 154, 303–314.e3. [Google Scholar] [CrossRef]
- Varma, R.; Lee, P.P.; Goldberg, I.; Kotak, S. An Assessment of the Health and Economic Burdens of Glaucoma. Am. J. Ophthalmol. 2011, 152, 515–522. [Google Scholar] [CrossRef]
- Kwon, Y.H.; Fingert, J.H.; Greenlee, E.C. A Patient’s Guide to Glaucoma; FEP International: Brussels, Belgium, 2008; p. 172. [Google Scholar]
- Zhang, N.; Wang, J.; Chen, B.; Li, Y.; Jiang, B. Prevalence of Primary Angle Closure Glaucoma in the Last 20 Years: A Meta-Analysis and Systematic Review. Front. Med. 2020, 7, 624179. [Google Scholar] [CrossRef]
- Vision Loss Expert Group of the Global Burden of Disease Study; the GBD 2019 Blindness and Vision Impairment Collaborators. Global Estimates on the Number of People Blind or Visually Impaired by Glaucoma: A Meta-Analysis from 2000 to 2020. Eye 2024, 38, 2036–2046. [CrossRef]
- Shan, S.; Wu, J.; Cao, J.; Feng, Y.; Zhou, J.; Luo, Z.; Song, P.; Rudan, I. Global Incidence and Risk Factors for Glaucoma: A Systematic Review and Meta-Analysis of Prospective Studies. J. Glob. Health 2024, 14, 04252. [Google Scholar] [CrossRef] [PubMed]
- Tham, Y.C.; Li, X.; Wong, T.Y.; Quigley, H.A.; Aung, T.; Cheng, C.Y. Global Prevalence of Glaucoma and Projections of Glaucoma Burden through 2040: A Systematic Review and Meta-Analysis. Ophthalmology 2014, 121, 2081–2090. [Google Scholar] [CrossRef]
- Allison, K.; Patel, D.; Alabi, O. Epidemiology of Glaucoma: The Past, Present, and Predictions for the Future. Cureus 2020, 12, e11686. [Google Scholar] [CrossRef] [PubMed]
- Weinreb, R.N.; Aung, T.; Medeiros, F.A. The Pathophysiology and Treatment of Glaucoma: A Review. JAMA 2014, 311, 1901–1911. [Google Scholar] [CrossRef]
- Stein, J.D.; Khawaja, A.P.; Weizer, J.S. Glaucoma in Adults-Screening, Diagnosis, and Management: A Review. JAMA 2021, 325, 164–174. [Google Scholar] [CrossRef]
- Weinreb, R.N.; Leung, C.K.; Crowston, J.G.; Medeiros, F.A.; Friedman, D.S.; Wiggs, J.L.; Martin, K.R. Primary Open-Angle Glaucoma. Nat. Rev. Dis. Primers 2016, 2, 16067. [Google Scholar] [CrossRef]
- Jayaram, H.; Kolko, M.; Friedman, D.S.; Gazzard, G. Glaucoma: Now and Beyond. Lancet 2023, 402, 1788–1801. [Google Scholar] [CrossRef]
- Wójcik-Gryciuk, A.; Skup, M.; Waleszczyk, W.J. Glaucoma-State of the Art and Perspectives on Treatment. Restor. Neurol. Neurosci. 2016, 34, 107–123. [Google Scholar] [CrossRef]
- Leske, M.C.; Heijl, A.; Hyman, L.; Bengtsson, B.; Dong, L.; Yang, Z. Predictors of Long-Term Progression in the Early Manifest Glaucoma Trial. Ophthalmology 2007, 114, 1965–1972. [Google Scholar] [CrossRef]
- Jayaram, H. Intraocular Pressure Reduction in Glaucoma: Does Every mmHg Count? Taiwan J. Ophthalmol. 2020, 10, 255–258. [Google Scholar] [CrossRef]
- Heijl, A.; Leske, M.C.; Bengtsson, B.; Hyman, L.; Bengtsson, B.; Hussein, M. Reduction of Intraocular Pressure and Glaucoma Progression: Results from the Early Manifest Glaucoma Trial. Arch. Ophthalmol. 2002, 120, 1268–1279. [Google Scholar] [CrossRef] [PubMed]
- Collaborative Normal-Tension Glaucoma Study Group. Comparison of Glaucomatous Progression between Untreated Patients with Normal-Tension Glaucoma and Patients with Therapeutically Reduced Intraocular Pressures. Am. J. Ophthalmol. 1998, 126, 487–497. [Google Scholar] [CrossRef] [PubMed]
- Miller-Ellis, E.; Fleming, G.P. Then and Now: Medical Therapy for Glaucoma. Ophthalmol. Glaucoma 2025, 8, S33–S37. [Google Scholar] [CrossRef]
- Kolko, M.; Tatham, A.J.; Lim, K.S.; Wells, A.P.; Shiu, M.; Uy, H.S.; Sarkisian, S.R., Jr.; Ho, Q.; Jiao, J.; Kim, K.; et al. Phase 3, Randomized, Comparison Study of Intracameral Bimatoprost Implant 10 µg and Selective Laser Trabeculoplasty. Am. J. Ophthalmol. 2025, 272, 19–37. [Google Scholar] [CrossRef]
- Fechtner, R.; Mansberger, S.; Branch, J.; Mulaney, J.; Ziebell, S.; Lopez, K.; Hubatsch, D. A Randomized, Controlled Comparison of NCX 470, a Nitric Oxide-Donating Bimatoprost, and Latanoprost in Subjects with Open-Angle Glaucoma or Ocular Hypertension: The MONT BLANC Study. Am. J. Ophthalmol. 2024, 264, 66–74. [Google Scholar] [CrossRef]
- Tanihara, H.; Yamamoto, T.; Aihara, M.; Koizumi, N.; Fukushima, A.; Kawakita, K.; Kojima, S.; Nakamura, T.; Suganami, H. Long-term Intraocular Pressure-Lowering Efficacy and Safety of Ripasudil-Brimonidine Fixed-Dose Combination for Glaucoma and Ocular Hypertension: A Multicentre, Open-Label, Phase 3 Study. Graefes Arch. Clin. Exp. Ophthalmol. 2024, 262, 2579–2591. [Google Scholar] [CrossRef]
- Garway-Heath, D.F.; Crabb, D.P.; Bunce, C.; Lascaratos, G.; Amalfitano, F.; Anand, N.; Azuara-Blanco, A.; Bourne, R.R.; Broadway, D.C.; Cunliffe, I.A.; et al. Latanoprost for Open-Angle Glaucoma (UKGTS): A Randomised, Multicentre, Placebo-Controlled Trial. Lancet 2015, 385, 1295–1304. [Google Scholar] [CrossRef]
- Kass, M.A.; Heuer, D.K.; Higginbotham, E.J.; Johnson, C.A.; Keltner, J.L.; Miller, J.P.; Parrish, R.K., 2nd; Wilson, M.R.; Gordon, M.O. The Ocular Hypertension Treatment Study: A Randomized Trial Determines that Topical Ocular Hypotensive Medication Delays or Prevents the Onset of Primary Open-Angle Glaucoma. Arch. Ophthalmol. 2002, 120, 701–713. [Google Scholar] [CrossRef]
- Mai, A.P.; Ferguson, T.J.; Radcliffe, N. New Treatments for Glaucoma. Curr. Opin. Ophthalmol. 2025, 36, 135–139. [Google Scholar] [CrossRef]
- Mann, E.; Kammer, J.A.; Sawhney, G.; An, J.; Werts, E.C.; Vera, V.; Rivas, M.; Lai, H.; Sonparote, S.; Craven, E.R. Prospective 18-Month Study of Bimatoprost Intracameral Implant in Patients with Open-Angle Glaucoma or Ocular Hypertension in US Clinical Practice. Drugs 2025, 85, 397–414. [Google Scholar] [CrossRef]
- Ferguson, T.J.; Samuelson, T.W.; Herndon, L.; Terveen, D.C.; Bacharach, J.; Brubaker, J.W.; Berdahl, J.P.; Radcliffe, N.M. Negative Pressure Application by the Ocular Pressure Adjusting Pump to Lower Intraocular Pressure in Normal-Tension Glaucoma: HERCULES Study. Am. J. Ophthalmol. 2025, 275, 121–134. [Google Scholar] [CrossRef] [PubMed]
- Sarkisian, S.R., Jr.; Ang, R.E.; Lee, A.M.; Berdahl, J.P.; Heersink, S.B.; Burden, J.H.; Doan, L.V.; Stephens, K.G.; Kothe, A.C.; Usner, D.W.; et al. Phase 3 Randomized Clinical Trial of the Safety and Efficacy of Travoprost Intraocular Implant in Patients with Open-Angle Glaucoma or Ocular Hypertension. Ophthalmology 2024, 131, 1021–1032. [Google Scholar] [CrossRef]
- Confalonieri, F.; Casarini, B.; Papapicco, A.; Stiro, F.; Piscopo, R.; D’Andrea, L.; Petrovski, G.; Verdina, T. A Systematic Review of Micropulse Laser Trabeculoplasty (MLT) in Primary Open-Angle Glaucoma (POAG) Management: Efficacy, Safety, and Future Perspectives. Biomedicines 2025, 13, 211. [Google Scholar] [CrossRef] [PubMed]
- Fahy, E.T.; Montesano, G.; Garg, A.; Vickerstaff, V.; Konstantakopoulou, E.; Gazzard, G. The Impact of Baseline Intraocular Pressure on Initial Treatment Response in the LiGHT Trial: Selective Laser Trabeculoplasty versus Medication. Ophthalmology 2024, 131, 1366–1376. [Google Scholar] [CrossRef] [PubMed]
- Latina, M.A.; Sibayan, S.A.; Shin, D.H.; Noecker, R.J.; Marcellino, G. Q-switched 532-nm Nd:YAG Laser Trabeculoplasty (Selective Laser Trabeculoplasty): A Multicenter, Pilot, Clinical Study. Ophthalmology 1998, 105, 2082–2088; discussion 2089–2090. [Google Scholar] [CrossRef]
- Moghimi, S.; Girkin, C.; Weinreb, R.N. Intraocular Pressure Matters. Ophthalmol. Glaucoma 2025, 8, S6–S13. [Google Scholar] [CrossRef] [PubMed]
- Acott, T.S.; Fautsch, M.P.; Mao, W.; Ethier, C.R.; Huang, A.S.; Kelley, M.J.; Aga, M.; Bhattacharya, S.K.; Borras, T.; Bovenkamp, D.; et al. Consensus Recommendations for Studies of Outflow Facility and Intraocular Pressure Regulation Using Ex Vivo Perfusion Approaches. Investig. Ophthalmol. Vis. Sci. 2024, 65, 32. [Google Scholar] [CrossRef]
- Grant, W.M. Further Studies on Facility of Flow through the Trabecular Meshwork. AMA Arch. Ophthalmol. 1958, 60, 523–533. [Google Scholar] [CrossRef] [PubMed]
- Llobet, A.; Gasull, X.; Gual, A. Understanding Trabecular Meshwork Physiology: A Key to the Control of Intraocular Pressure? News Physiol. Sci. 2003, 18, 205–209. [Google Scholar] [CrossRef] [PubMed]
- Yun, H.; Zhou, Y.; Wills, A.; Du, Y. Stem Cells in the Trabecular Meshwork for Regulating Intraocular Pressure. J. Ocul. Pharmacol. Ther. 2016, 32, 253–260. [Google Scholar] [CrossRef]
- Speakman, J. Aqueous Outflow Channels in the Trabecular Meshwork in Man. Br. J. Ophthalmol. 1959, 43, 129–138. [Google Scholar] [CrossRef]
- Carreon, T.; van der Merwe, E.; Fellman, R.L.; Johnstone, M.; Bhattacharya, S.K. Aqueous Outflow—A Continuum from Trabecular Meshwork to Episcleral Veins. Prog. Retin. Eye Res. 2017, 57, 108–133. [Google Scholar] [CrossRef]
- Grant, W.M. Facility of Flow through the Trabecular Meshwork. AMA Arch. Ophthalmol. 1955, 54, 245–248. [Google Scholar] [CrossRef]
- Rosenquist, R.; Epstein, D.; Melamed, S.; Johnson, M.; Grant, W.M. Outflow Resistance of Enucleated Human Eyes at Two Different Perfusion Pressures and Different Extents of Trabeculotomy. Curr. Eye Res. 1989, 8, 1233–1240. [Google Scholar] [CrossRef]
- Johnson, M. What Controls Aqueous Humour Outflow Resistance? Exp. Eye Res. 2006, 82, 545–557. [Google Scholar] [CrossRef]
- Tamm, E.R. The Trabecular Meshwork Outflow Pathways: Structural and Functional Aspects. Exp. Eye Res. 2009, 88, 648–655. [Google Scholar] [CrossRef] [PubMed]
- Keller, K.E.; Acott, T.S. The Juxtacanalicular Region of Ocular Trabecular Meshwork: A Tissue with a Unique Extracellular Matrix and Specialized Function. J. Ocul. Biol. 2013, 1, 3. [Google Scholar] [CrossRef]
- Swaminathan, S.S.; Oh, D.J.; Kang, M.H.; Rhee, D.J. Aqueous Outflow: Segmental and Distal Flow. J. Cataract. Refract. Surg. 2014, 40, 1263–1272. [Google Scholar] [CrossRef]
- Acott, T.S.; Vranka, J.A.; Keller, K.E.; Raghunathan, V.; Kelley, M.J. Normal and Glaucomatous Outflow Regulation. Prog. Retin. Eye Res. 2021, 82, 100897. [Google Scholar] [CrossRef] [PubMed]
- Overby, D.R.; Stamer, W.D.; Johnson, M. The Changing Paradigm of Outflow Resistance Generation: Towards Synergistic Models of the JCT and Inner Wall Endothelium. Exp. Eye Res. 2009, 88, 656–670. [Google Scholar] [CrossRef]
- Polansky, J.R.; Wood, I.S.; Maglio, M.T.; Alvarado, J.A. Trabecular Meshwork Cell Culture in Glaucoma Research: Evaluation of Biological Activity and Structural Properties of Human Trabecular Cells in vitro. Ophthalmology 1984, 91, 580–595. [Google Scholar] [CrossRef]
- Stamer, W.D.; Clark, A.F. The Many Faces of the Trabecular Meshwork Cell. Exp. Eye Res. 2017, 158, 112–123. [Google Scholar] [CrossRef]
- Patel, G.; Fury, W.; Yang, H.; Gomez-Caraballo, M.; Bai, Y.; Yang, T.; Adler, C.; Wei, Y.; Ni, M.; Schmitt, H.; et al. Molecular Taxonomy of Human Ocular Outflow Tissues Defined by Single-Cell Transcriptomics. Proc. Natl. Acad. Sci. USA 2020, 117, 12856–12867. [Google Scholar] [CrossRef]
- Tolman, N.; Li, T.; Balasubramanian, R.; Li, G.; Bupp-Chickering, V.; Kelly, R.A.; Simón, M.; Peregrin, J.; Montgomery, C.; Stamer, W.D.; et al. Single-Cell Profiling of Trabecular Meshwork Identifies Mitochondrial Dysfunction in a Glaucoma Model that is Protected by Vitamin B3 Treatment. eLife 2025, 14, RP107161. [Google Scholar] [CrossRef]
- Balasubramanian, R.; Kizhatil, K.; Li, T.; Tolman, N.; Bhandari, A.; Clark, G.; Bupp-Chickering, V.; Kelly, R.A.; Zhou, S.; Peregrin, J.; et al. Transcriptomic Profiling of Schlemm’s Canal Cells Reveals a Lymphatic-Biased Identity and three Major Cell States. eLife 2024, 13, RP96459. [Google Scholar] [CrossRef]
- van Zyl, T.; Yan, W.; McAdams, A.; Peng, Y.R.; Shekhar, K.; Regev, A.; Juric, D.; Sanes, J.R. Cell Atlas of Aqueous Humor Outflow Pathways in Eyes of Humans and Four Model Species Provides Insight into Glaucoma Pathogenesis. Proc. Natl. Acad. Sci. USA 2020, 117, 10339–10349. [Google Scholar] [CrossRef]
- Dautriche, C.N.; Tian, Y.; Xie, Y.; Sharfstein, S.T. A Closer Look at Schlemm’s Canal Cell Physiology: Implications for Biomimetics. J. Funct. Biomater. 2015, 6, 963–985. [Google Scholar] [CrossRef]
- Grierson, I.; Hogg, P. The Proliferative and Migratory Activities of Trabecular Meshwork Cells. Prog. Retin. Eye Res. 1995, 15, 33–67. [Google Scholar] [CrossRef]
- Fautsch, M.P.; Howell, K.G.; Vrabel, A.M.; Charlesworth, M.C.; Muddiman, D.C.; Johnson, D.H. Primary Trabecular Meshwork Cells Incubated in Human Aqueous Humor Differ from Cells Incubated in Serum Supplements. Investig. Ophthalmol. Vis. Sci. 2005, 46, 2848–2856. [Google Scholar] [CrossRef]
- Alvarado, J.; Murphy, C.; Polansky, J.; Juster, R. Age-Related Changes in trabecular Meshwork Cellularity. Investig. Ophthalmol. Vis. Sci. 1981, 21, 714–727. [Google Scholar]
- Alvarado, J.; Murphy, C.; Juster, R. Trabecular Meshwork Cellularity in Primary Open-Angle Glaucoma and Nonglaucomatous Normals. Ophthalmology 1984, 91, 564–579. [Google Scholar] [CrossRef]
- Grierson, I.; Howes, R.C. Age-Related Depletion of the Cell Population in the Human Trabecular Meshwork. Eye 1987, 1, 204–210. [Google Scholar] [CrossRef]
- Gabelt, B.T.; Kaufman, P.L. Changes in Aqueous Humor Dynamics with Age and Glaucoma. Prog. Retin. Eye Res. 2005, 24, 612–637. [Google Scholar] [CrossRef]
- Tektas, O.Y.; Lütjen-Drecoll, E. Structural Changes of the Trabecular Meshwork in Different Kinds of Glaucoma. Exp. Eye Res. 2009, 88, 769–775. [Google Scholar] [CrossRef]
- Li, G.; van Batenburg-Sherwood, J.; Safa, B.N.; Fraticelli Guzmán, N.S.; Wilson, A.; Bahrani Fard, M.R.; Choy, K.; de Ieso, M.L.; Cui, J.S.; Feola, A.J.; et al. Aging and Intraocular Pressure Homeostasis in Mice. Aging Cell 2024, 23, e14160. [Google Scholar] [CrossRef]
- Rao, A.; Rao, T.; Banka, N.; Senthil, S.; Jaketi, S. Trabecular Meshwork Ultrastructural Changes in Primary and Secondary Glaucoma. Sci. Rep. 2025, 15, 138. [Google Scholar] [CrossRef]
- Brumley, S.; Xian, S.; Kuehn, M.H. Loss of Trabecular Meshwork Stem Cells is Correlated with Open Angle Glaucoma. Exp. Eye Res. 2025, 253, 110296. [Google Scholar] [CrossRef]
- Gong, H.; Swain, D.L. The Histopathological Changes in the Trabecular Outflow Pathway and their Possible Effects on Aqueous Outflow in Eyes with Primary Open-Angle Glaucoma. In Glaucoma Research and Clinical Advances; Knepper, P.A., Samples, J.R., Eds.; Kugler: Amsterdam, The Netherlands, 2016; pp. 17–40. [Google Scholar]
- Stamer, W.D.; Acott, T.S. Current Understanding of Conventional Outflow Dysfunction in Glaucoma. Curr. Opin. Ophthalmol. 2012, 23, 135–143. [Google Scholar] [CrossRef]
- Braunger, B.M.; Fuchshofer, R.; Tamm, E.R. The Aqueous Humor Outflow Pathways in Glaucoma: A Unifying Concept of Disease Mechanisms and Causative Treatment. Eur. J. Pharm. Biopharm. 2015, 95, 173–181. [Google Scholar] [CrossRef]
- Tang, L.T.C.; Wang, Y.; Shi, X. The Molecular Mechanisms of Trabecular Meshwork Damage in POAG and Treatment Advances; IntechOpen: London, UK, 2023. [Google Scholar]
- Roy, S. Emerging Strategies Targeting Genes and Cells in Glaucoma. Vision. Res. 2025, 227, 108533. [Google Scholar] [CrossRef]
- Maddala, R.; Rao, P.V. Global Phosphotyrosinylated Protein Profile of Cell-Matrix Adhesion Complexes of trabecular Meshwork Cells. Am. J. Physiol. Cell Physiol. 2020, 319, C288–C299. [Google Scholar] [CrossRef]
- Lütjen-Drecoll, E.; Rittig, M.; Rauterberg, J.; Jander, R.; Mollenhauer, J. Immunomicroscopical Study of Type VI Collagen in the Trabecular Meshwork of Normal and Glaucomatous Eyes. Exp. Eye Res. 1989, 48, 139–147. [Google Scholar] [CrossRef]
- Babizhayev, M.A.; Brodskaya, M.W. Fibronectin Detection in Drainage Outflow System of Human Eyes in Ageing and Progression Of Open-Angle Glaucoma. Mech. Ageing Dev. 1989, 47, 145–157. [Google Scholar] [CrossRef]
- Faralli, J.A.; Filla, M.S.; Peters, D.M. Role of Fibronectin in Primary Open Angle Glaucoma. Cells 2019, 8, 1518. [Google Scholar] [CrossRef]
- Clark, A.F. The Cell and Molecular Biology of Glaucoma: Biomechanical Factors in Glaucoma. Investig. Ophthalmol. Vis. Sci. 2012, 53, 2473–2475. [Google Scholar] [CrossRef]
- Kasetti, R.B.; Maddineni, P.; Millar, J.C.; Clark, A.F.; Zode, G.S. Increased Synthesis and Deposition of Extracellular Matrix Proteins Leads to Endoplasmic Reticulum Stress in the Trabecular Meshwork. Sci. Rep. 2017, 7, 14951. [Google Scholar] [CrossRef]
- Yang, Y.F.; Holden, P.; Sun, Y.Y.; Faralli, J.A.; Peters, D.M.; Keller, K.E. Fibrosis-Related Gene and Protein Expression in Normal and Glaucomatous Trabecular Meshwork Cells. Investig. Ophthalmol. Vis. Sci. 2025, 66, 48. [Google Scholar] [CrossRef]
- Li, H.; Harvey, D.H.; Dai, J.; Swingle, S.P.; Compton, A.M.; Sugali, C.K.; Dhamodaran, K.; Yao, J.; Lin, T.Y.; Sulchek, T.; et al. Characterization, Enrichment, and Computational Modeling of Cross-Linked Actin Networks in Transformed Trabecular Meshwork Cells. Investig. Ophthalmol. Vis. Sci. 2025, 66, 65. [Google Scholar] [CrossRef]
- Watanabe, M.; Sato, T.; Tsugeno, Y.; Higashide, M.; Furuhashi, M.; Umetsu, A.; Suzuki, S.; Ida, Y.; Hikage, F.; Ohguro, H. All-trans Retinoic Acids Synergistically and Beneficially Affect In Vitro Glaucomatous Trabecular Meshwork (TM) Models Using 2D and 3D Cell Cultures of Human TM Cells. Int. J. Mol. Sci. 2022, 23, 9912. [Google Scholar] [CrossRef]
- Watanabe, M.; Ida, Y.; Furuhashi, M.; Tsugeno, Y.; Hikage, F.; Ohguro, H. Pan-ROCK and ROCK2 Inhibitors Affect Dexamethasone-Treated 2D- and 3D-Cultured Human Trabecular Meshwork (HTM) Cells in Opposite Manners. Molecules 2021, 26, 6382. [Google Scholar] [CrossRef]
- Suzuki, S.; Furuhashi, M.; Tsugeno, Y.; Umetsu, A.; Ida, Y.; Hikage, F.; Ohguro, H.; Watanabe, M. Comparison of the Drug-Induced Efficacies between Omidenepag Isopropyl, an EP2 Agonist and PGF2α toward TGF-β2-Modulated Human Trabecular Meshwork (HTM) Cells. J. Clin. Med. 2022, 11, 1652. [Google Scholar] [CrossRef]
- Zhang, Y.; Tseng, S.C.G.; Zhu, Y.T. Suppression of TGF-β1 Signaling by Matrigel via FAK Signaling in Cultured Human Trabecular Meshwork Cells. Sci. Rep. 2021, 11, 7319. [Google Scholar] [CrossRef]
- Kumon, M.; Fuwa, M.; Shimazaki, A.; Odani-Kawabata, N.; Iwamura, R.; Yoneda, K.; Kato, M. Downregulation of COL12A1 and COL13A1 by a Selective EP2 Receptor Agonist, Omidenepag, in Human Trabecular Meshwork Cells. PLoS ONE 2023, 18, e0280331. [Google Scholar] [CrossRef]
- Watanabe, M.; Sato, T.; Tsugeno, Y.; Higashide, M.; Furuhashi, M.; Umetsu, A.; Suzuki, S.; Ida, Y.; Hikage, F.; Ohguro, H. An α2-Adrenergic Agonist, Brimonidine, Beneficially Affects the TGF-β2-Treated Cellular Properties in an In Vitro Culture Model. Bioengineering 2022, 9, 310. [Google Scholar] [CrossRef]
- Vernazza, S.; Tirendi, S.; Scarfi, S.; Passalacqua, M.; Oddone, F.; Traverso, C.E.; Rizzato, I.; Bassi, A.M.; Sacca, S.C. 2D- and 3D-Cultures of Human Trabecular Meshwork Cells: A Preliminary Assessment of an in vitro Model for Glaucoma Study. PLoS ONE 2019, 14, e0221942. [Google Scholar] [CrossRef]
- Bikuna-Izagirre, M.; Aldazabal, J.; Extramiana, L.; Moreno-Montanes, J.; Carnero, E.; Paredes, J. Technological advances in ocular trabecular meshwork in vitro models for glaucoma research. Biotechnol. Bioeng. 2022, 119, 2698–2714. [Google Scholar] [CrossRef]
- Ghosh, S.; Herberg, S. ECM Biomaterials for Modeling of Outflow Cell Biology in Health and Disease. Biomater. Biosyst. 2024, 13, 100091. [Google Scholar] [CrossRef]
- Lamont, H.C.; Masood, I.; Grover, L.M.; El Haj, A.J.; Hill, L.J. Fundamental Biomaterial Considerations in the Development of a 3D Model Representative of Primary Open Angle Glaucoma. Bioengineering 2021, 8, 147. [Google Scholar] [CrossRef]
- Buffault, J.; Brignole-Baudouin, F.; Labbé, A.; Baudouin, C. An Overview of Current Glaucomatous Trabecular Meshwork Models. Curr. Eye Res. 2023, 48, 1089–1099. [Google Scholar] [CrossRef]
- Torrejon, K.Y.; Unser, A.M.; Ramesh, P. Protocol for Studying TM Cell Function in a 3-D Matrix. In Research Protocols for Ophthalmic Disease Mechanisms and Therapeutics: Glaucoma-Ocular Hypertension; Bentham Science Publishers: Singapore, 2025. [Google Scholar]
- Schlunck, G.n.; Han, H.; Wecker, T.; Kampik, D.; Meyer-ter-Vehn, T.; Grehn, F. Substrate Rigidity Modulates Cell–Matrix Interactions and Protein Expression in Human Trabecular Meshwork Cells. Investig. Ophthalmol. Vis. Sci. 2008, 49, 262–269. [Google Scholar] [CrossRef]
- Wood, J.A.; McKee, C.T.; Thomasy, S.M.; Fischer, M.E.; Shah, N.M.; Murphy, C.J.; Russell, P. Substratum Compliance Regulates Human Trabecular Meshwork Cell Behaviors and Response to Latrunculin B. Investig. Ophthalmol. Vis. Sci. 2011, 52, 9298–9303. [Google Scholar] [CrossRef]
- McKee, C.T.; Wood, J.A.; Shah, N.M.; Fischer, M.E.; Reilly, C.M.; Murphy, C.J.; Russell, P. The Effect of Biophysical Attributes of the Ocular Trabecular Meshwork Associated with Glaucoma on the Cell Response to Therapeutic Agents. Biomaterials 2011, 32, 2417–2423. [Google Scholar] [CrossRef]
- Thomasy, S.M.; Wood, J.A.; Kass, P.H.; Murphy, C.J.; Russell, P. Substratum Stiffness and Latrunculin B Regulate Matrix Gene and Protein Expression in Human Trabecular Meshwork Cells. Investig. Ophthalmol. Vis. Sci. 2012, 53, 952–958. [Google Scholar] [CrossRef]
- Thomasy, S.M.; Morgan, J.T.; Wood, J.A.; Murphy, C.J.; Russell, P. Substratum Stiffness and Latrunculin B Modulate the Gene Expression of the Mechanotransducers YAP and TAZ in Human Trabecular Meshwork Cells. Exp. Eye Res. 2013, 113, 66–73. [Google Scholar] [CrossRef]
- Karimi, A.; Aga, M.; Khan, T.; D’Costa, S.D.; Thaware, O.; White, E.; Kelley, M.J.; Gong, H.; Acott, T.S. Comparative Analysis of Traction Forces in Normal and Glaucomatous Trabecular Meshwork Cells within a 3D, Active Fluid-Structure Interaction Culture Environment. Acta Biomater. 2024, 180, 206–229. [Google Scholar] [CrossRef]
- Karimi, A.; Aga, M.; Stanik, A.; Franca, C.; Siadat, S.M.; White, E.; Kelley, M.; Acott, T. Matrix stiffness Regulates Traction Forces, Cytoskeletal Dynamics, and Collagen Reorganization in Trabecular Meshwork Cells in Glaucoma. Matter 2025, 8, 102094. [Google Scholar] [CrossRef]
- Wolff, L.; Hendrix, S. Rethinking Matrigel: The Complex Journey to Matrix Alternatives in Organoid Culture. Adv. Sci. 2025, 12, e08734. [Google Scholar] [CrossRef]
- Zhang, Y.; Cai, S.; Tseng, S.C.G.; Zhu, Y.T. Isolation and Expansion of Multipotent Progenitors from Human Trabecular Meshwork. Sci. Rep. 2018, 8, 2814. [Google Scholar] [CrossRef]
- Bouchemi, M.; Roubeix, C.; Kessal, K.; Riancho, L.; Raveu, A.L.; Soualmia, H.; Baudouin, C.; Brignole-Baudouin, F. Effect of Benzalkonium Chloride on Trabecular Meshwork Cells in a New In Vitro 3D Trabecular Meshwork Model for Glaucoma. Toxicol. In Vitro 2017, 41, 21–29. [Google Scholar] [CrossRef]
- Buffault, J.; Brignole-Baudouin, F.; Reboussin, É.; Kessal, K.; Labbé, A.; Mélik Parsadaniantz, S.; Baudouin, C. The Dual Effect of Rho-Kinase Inhibition on Trabecular Meshwork Cells Cytoskeleton and Extracellular Matrix in an In Vitro Model of Glaucoma. J. Clin. Med. 2022, 11, 1001. [Google Scholar] [CrossRef]
- Tirendi, S.; Saccà, S.C.; Vernazza, S.; Traverso, C.; Bassi, A.M.; Izzotti, A. A 3D Model of Human Trabecular Meshwork for the Research Study of Glaucoma. Front. Neurol. 2020, 11, 591776. [Google Scholar] [CrossRef]
- Saccà, S.C.; Tirendi, S.; Scarfì, S.; Passalacqua, M.; Oddone, F.; Traverso, C.E.; Vernazza, S.; Bassi, A.M. An Advanced In Vitro Model to Assess Glaucoma Onset. Altex 2020, 37, 265–274. [Google Scholar] [CrossRef]
- Saccà, S.C.; Izzotti, A.; Vernazza, S.; Tirendi, S.; Scarfì, S.; Gandolfi, S.; Bassi, A.M. Can Polyphenols in Eye Drops Be Useful for Trabecular Protection from Oxidative Damage? J. Clin. Med. 2020, 9, 3584. [Google Scholar] [CrossRef]
- Vernazza, S.; Passalacqua, M.; Tirendi, S.; Marengo, B.; Domenicotti, C.; Sbardella, D.; Oddone, F.; Bassi, A.M. Citicoline Eye Drops Protect Trabecular Meshwork Cells from Oxidative Stress Injury in a 3D In Vitro Glaucoma Model. Int. J. Mol. Sci. 2022, 23, 11375. [Google Scholar] [CrossRef]
- Frantz, C.; Stewart, K.M.; Weaver, V.M. The Extracellular Matrix at a Glance. J. Cell Sci. 2010, 123, 4195–4200. [Google Scholar] [CrossRef]
- Kalouche, G.; Beguier, F.; Bakria, M.; Melik-Parsadaniantz, S.; Leriche, C.; Debeir, T.; Rostène, W.; Baudouin, C.; Vigé, X. Activation of Prostaglandin FP and EP2 Receptors Differently Modulates Myofibroblast Transition in a Model of Adult Primary Human Trabecular Meshwork Cells. Investig. Ophthalmol. Vis. Sci. 2016, 57, 1816–1825. [Google Scholar] [CrossRef]
- Osmond, M.; Bernier, S.M.; Pantcheva, M.B.; Krebs, M.D. Collagen and Collagen-Chondroitin Sulfate Scaffolds with Uniaxially Aligned Pores for the Biomimetic, Three Dimensional Culture of Trabecular Meshwork Cells. Biotechnol. Bioeng. 2017, 114, 915–923. [Google Scholar] [CrossRef]
- Osmond, M.J.; Krebs, M.D.; Pantcheva, M.B. Human Trabecular Meshwork Cell Behavior is Influenced by collagen Scaffold Pore Architecture and Glycosaminoglycan Composition. Biotechnol. Bioeng. 2020, 117, 3150–3159. [Google Scholar] [CrossRef]
- Adhikari, B.; Osmond, M.J.; Pantcheva, M.B.; Krebs, M.D. Glycosaminoglycans Influence Extracellular Matrix of Human Trabecular Meshwork Cells Cultured on 3D Scaffolds. ACS Biomater. Sci. Eng. 2022, 8, 5221–5232. [Google Scholar] [CrossRef]
- Singh, A.; Ghosh, R.; Li, H.; Geiss, M.P.; Yoo, H.; Strat, A.N.; Ganapathy, P.S.; Herberg, S. Three-Dimensional Extracellular Matrix Protein Hydrogels for Human Trabecular Meshwork Cell Studies. Methods Mol. Biol. 2025, 2858, 17–29. [Google Scholar] [CrossRef]
- Li, H.; Bagué, T.; Kirschner, A.; Strat, A.N.; Roberts, H.; Weisenthal, R.W.; Patteson, A.E.; Annabi, N.; Stamer, W.D.; Ganapathy, P.S.; et al. A Tissue-Engineered Human Trabecular Meshwork Hydrogel for Advanced Glaucoma Disease Modeling. Exp. Eye Res. 2021, 205, 108472. [Google Scholar] [CrossRef]
- Bague, T.; Singh, A.; Ghosh, R.; Yoo, H.; Kelly, C.; deLong, M.A.; Kopczynski, C.C.; Herberg, S. Effects of Netarsudil-Family Rho Kinase Inhibitors on Human Trabecular Meshwork Cell Contractility and Actin Remodeling Using a Bioengineered ECM Hydrogel. Front. Ophthalmol. 2022, 2, 948397. [Google Scholar] [CrossRef]
- Li, H.; Raghunathan, V.; Stamer, W.D.; Ganapathy, P.S.; Herberg, S. Extracellular Matrix Stiffness and TGFβ2 Regulate YAP/TAZ Activity in Human Trabecular Meshwork Cells. Front. Cell Dev. Biol. 2022, 10, 844342. [Google Scholar] [CrossRef]
- Karimi, A.; Stanik, A.; Golchin, H.; Fuller, D.; Aga, M.; White, E.; Kelley, M.J.; Acott, T.S. Actin-Microtubule Synergy Dominates Force Transmission and Collagen Strain in Human Trabecular Meshwork. Acta Biomater. 2025, 203, 478–491. [Google Scholar] [CrossRef]
- Lamont, H.C.; Wright, A.L.; Devries, K.; Okur, K.E.; Jones, M.; Masood, I.; Hill, L.J.; Nazhat, S.N.; Grover, L.M.; Haj, A.J.E.; et al. Trabecular Meshwork Cell Differentiation in Response to Collagen and TGFβ-2 Spatial Interactions. Acta Biomater. 2024, 189, 217–231. [Google Scholar] [CrossRef]
- Adhikari, B.; Stinson, B.S.; Osmond, M.J.; Pantcheva, M.B.; Krebs, M.D. Photoinduced Gelatin-Methacrylate Scaffolds to Examine the Impact of Extracellular Environment on Trabecular Meshwork Cells. Ind. Eng. Chem. Res. 2021, 60, 17417–17428. [Google Scholar] [CrossRef]
- Adhikari, B.; Barakoti, P.; Pantcheva, M.B.; Krebs, M.D. 3D Printed Gelatin Methacryloyl Hydrogels for Perfusion Culture of Human Trabecular Meshwork Cells and Glaucoma Studies. Biotechnol. Bioeng. 2025, 122, 69–79. [Google Scholar] [CrossRef]
- Wang, R.; Wei, H.; Shi, Y.; Wang, C.; Yu, Z.; Zhang, Y.; Lai, Y.; Chen, J.; Wang, G.; Tian, W. Self-Generating Electricity System Driven by Aqueous Humor Flow and Trabecular Meshwork Contraction Motion Activated BCKa for Glaucoma Intraocular Pressure Treatment. Mater. Horiz. 2025, 12, 434–450. [Google Scholar] [CrossRef]
- Waduthanthri, K.D.; He, Y.; Montemagno, C.; Cetinel, S. An Injectable Peptide Hydrogel for Reconstruction of the Human Trabecular Meshwork. Acta Biomater. 2019, 100, 244–254. [Google Scholar] [CrossRef]
- Crouch, D.J.; Sheridan, C.M.; Behnsen, J.G.; Bosworth, L.A. An Optimized Method to Decellularize Human Trabecular Meshwork. Bioengineering 2022, 9, 194. [Google Scholar] [CrossRef]
- Raghunathan, V.K.; Morgan, J.T.; Chang, Y.R.; Weber, D.; Phinney, B.; Murphy, C.J.; Russell, P. Transforming Growth Factor Beta 3 Modifies Mechanics and Composition of Extracellular Matrix Deposited by Human Trabecular Meshwork Cells. ACS Biomater. Sci. Eng. 2015, 1, 110–118. [Google Scholar] [CrossRef]
- Raghunathan, V.K.; Morgan, J.T.; Park, S.A.; Weber, D.; Phinney, B.S.; Murphy, C.J.; Russell, P. Dexamethasone Stiffens Trabecular Meshwork, Trabecular Meshwork Cells, and Matrix. Investig. Ophthalmol. Vis. Sci. 2015, 56, 4447–4459. [Google Scholar] [CrossRef]
- Yemanyi, F.; Vranka, J.; Raghunathan, V. Generating Cell-Derived Matrices from Human Trabecular Meshwork Cell Cultures for Mechanistic Studies. Methods Cell Biol. 2020, 156, 271–307. [Google Scholar] [CrossRef]
- Raghunathan, V.K.; Benoit, J.; Kasetti, R.; Zode, G.; Salemi, M.; Phinney, B.S.; Keller, K.E.; Staverosky, J.A.; Murphy, C.J.; Acott, T.; et al. Glaucomatous Cell Derived Matrices Differentially Modulate Non-Glaucomatous Trabecular Meshwork Cellular Behavior. Acta Biomater. 2018, 71, 444–459. [Google Scholar] [CrossRef]
- Li, X.; Gong, J.P. Design Principles for Strong and Tough Hydrogels. Nat. Rev. Mater. 2024, 9, 380–398. [Google Scholar] [CrossRef]
- Russell, P.; Gasiorowski, J.Z.; Nealy, P.F.; Murphy, C.J. Response of Human Trabecular Meshwork Cells to Topographic Cues on the Nanoscale Level. Investig. Ophthalmol. Vis. Sci. 2008, 49, 629–635. [Google Scholar] [CrossRef]
- Mochizuki, H.; Murphy, C.J.; Brandt, J.D.; Kiuchi, Y.; Russell, P. Altered Stability of mRNAs Associated with Glaucoma Progression in Human Trabecular Meshwork Cells Following Oxidative Stress. Investig. Ophthalmol. Vis. Sci. 2012, 53, 1734–1741. [Google Scholar] [CrossRef]
- Kim, B.; Roberts, C.J.; Mahmoud, A.M.; Weber, P.L.; Zhao, Y. Effect of Topographic Cues on In Vitro Cultured Trabecular Meshwork Endothelial Cells. In Proceedings of the 15th International Conference on Miniaturized Systems for Chemistry and Life Sciences 2011, MicroTAS 2011, Seattle, WA, USA, 2–6 October 2011; pp. 801–803. [Google Scholar]
- Torrejon, K.Y.; Pu, D.; Bergkvist, M.; Danias, J.; Sharfstein, S.T.; Xie, Y. Recreating a Human Trabecular Meshwork Outflow System on Microfabricated Porous Structures. Biotechnol. Bioeng. 2013, 110, 3205–3218. [Google Scholar] [CrossRef]
- Dautriche, C.N.; Szymanski, D.; Kerr, M.; Torrejon, K.Y.; Bergkvist, M.; Xie, Y.; Danias, J.; Stamer, W.D.; Sharfstein, S.T. A Biomimetic Schlemm’s Canal Inner Wall: A Model to Study Outflow Physiology, Glaucoma Pathology and High-Throughput Drug Screening. Biomaterials 2015, 65, 86–92. [Google Scholar] [CrossRef]
- Tian, Y.I.; Zhang, X.; Torrejon, K.; Danias, J.; Du, Y.; Xie, Y. A Biomimetic, Stem Cell-Derived In Vitro Ocular Outflow Model. Adv. Biosyst. 2020, 4, e2000004. [Google Scholar] [CrossRef]
- Tian, Y.I.; Zhang, X.; Torrejon, K.; Danias, J.; Gindina, S.; Nayyar, A.; Du, Y.; Xie, Y. A Bioengineering Approach to Schlemm’s Canal-Like Stem Cell Differentiation for In Vitro Glaucoma Drug Screening. Acta Biomater. 2020, 105, 203–213. [Google Scholar] [CrossRef]
- Torrejon, K.Y.; Papke, E.L.; Halman, J.R.; Stolwijk, J.; Dautriche, C.N.; Bergkvist, M.; Danias, J.; Sharfstein, S.T.; Xie, Y. Bioengineered Glaucomatous 3D Human Trabecular Meshwork as an In Vitro Disease Model. Biotechnol. Bioeng. 2016, 113, 1357–1368. [Google Scholar] [CrossRef]
- Torrejon, K.Y.; Papke, E.L.; Halman, J.R.; Bergkvist, M.; Danias, J.; Sharfstein, S.T.; Xie, Y. TGFβ2-induced Outflow Alterations in a Bioengineered Trabecular Meshwork are Offset by a Rho-Associated Kinase Inhibitor. Sci. Rep. 2016, 6, 38319. [Google Scholar] [CrossRef]
- Beardslee, L.A.; Halman, J.R.; Unser, A.M.; Xie, Y.; Danias, J.; Bergkvist, M.; Sharfstein, S.T.; Torrejon, K.Y. Recreating the Trabecular Outflow Tissue on Implantable, Micropatterned, Ultrathin, Porous Polycaprolactone Scaffolds. Bioengineering 2023, 10, 679. [Google Scholar] [CrossRef]
- Bikuna-Izagirre, M.; Aldazabal, J.; Extramiana, L.; Moreno-Montanes, J.; Carnero, E.; Paredes, J. Nanofibrous PCL-Based Human Trabecular Meshwork for Aqueous Humor Outflow Studies. ACS Biomater. Sci. Eng. 2023, 9, 6333–6344. [Google Scholar] [CrossRef]
- Crouch, D.J.; Sheridan, C.M.; Behnsen, J.G.; D’Sa, R.A.; Bosworth, L.A. Cryo-Electrospinning Generates Highly Porous Fiber Scaffolds Which Improves Trabecular Meshwork Cell Infiltration. J. Funct. Biomater. 2023, 14, 490. [Google Scholar] [CrossRef]
- Wlodarczyk-Biegun, M.K.; Villiou, M.; Koch, M.; Muth, C.; Wang, P.; Ott, J.; Del Campo, A. Melt Electrowriting of Graded Porous Scaffolds to Mimic the Matrix Structure of the Human Trabecular Meshwork. ACS Biomater. Sci. Eng. 2022, 8, 3899–3911. [Google Scholar] [CrossRef]
- Tan, J.C.; Gonzalez, J.M., Jr.; Hamm-Alvarez, S.; Song, J. In Situ Autofluorescence Visualization of Human Trabecular Meshwork Structure. Investig. Ophthalmol. Vis. Sci. 2012, 53, 2080–2088. [Google Scholar] [CrossRef]
- Camras, L.J.; Stamer, W.D.; Epstein, D.; Gonzalez, P.; Yuan, F. Circumferential Tensile Stiffness of Glaucomatous Trabecular Meshwork. Investig. Ophthalmol. Vis. Sci. 2014, 55, 814–823. [Google Scholar] [CrossRef]
- Bikuna-Izagirre, M.; Aldazabal, J.; Moreno-Montanes, J.; De-Juan-Pardo, E.; Carnero, E.; Paredes, J. Artificial Trabecular Meshwork Structure Combining Melt Electrowriting and Solution Electrospinning. Polymers 2024, 16, 2162. [Google Scholar] [CrossRef]
- Gładysz, M.Z.; Fernandes, M.; Li, X.; Koch, M.; Marchena, F.; Hofman, A.; de Graaf, M.; Wolters, J.C.; Kamperman, M.; Nagelkerke, A.; et al. Multilayered Trabecular Meshwork for Dynamic In Vitro Studies in Glaucoma Research. Acta Biomater. 2025, 202, 262–275. [Google Scholar] [CrossRef]
- Ota, C.; Ida, Y.; Ohguro, H.; Hikage, F. ROCK Inhibitors Beneficially Alter the Spatial Configuration of TGFβ2-treated 3D Organoids from a Human Trabecular Meshwork (HTM). Sci. Rep. 2020, 10, 20292. [Google Scholar] [CrossRef]
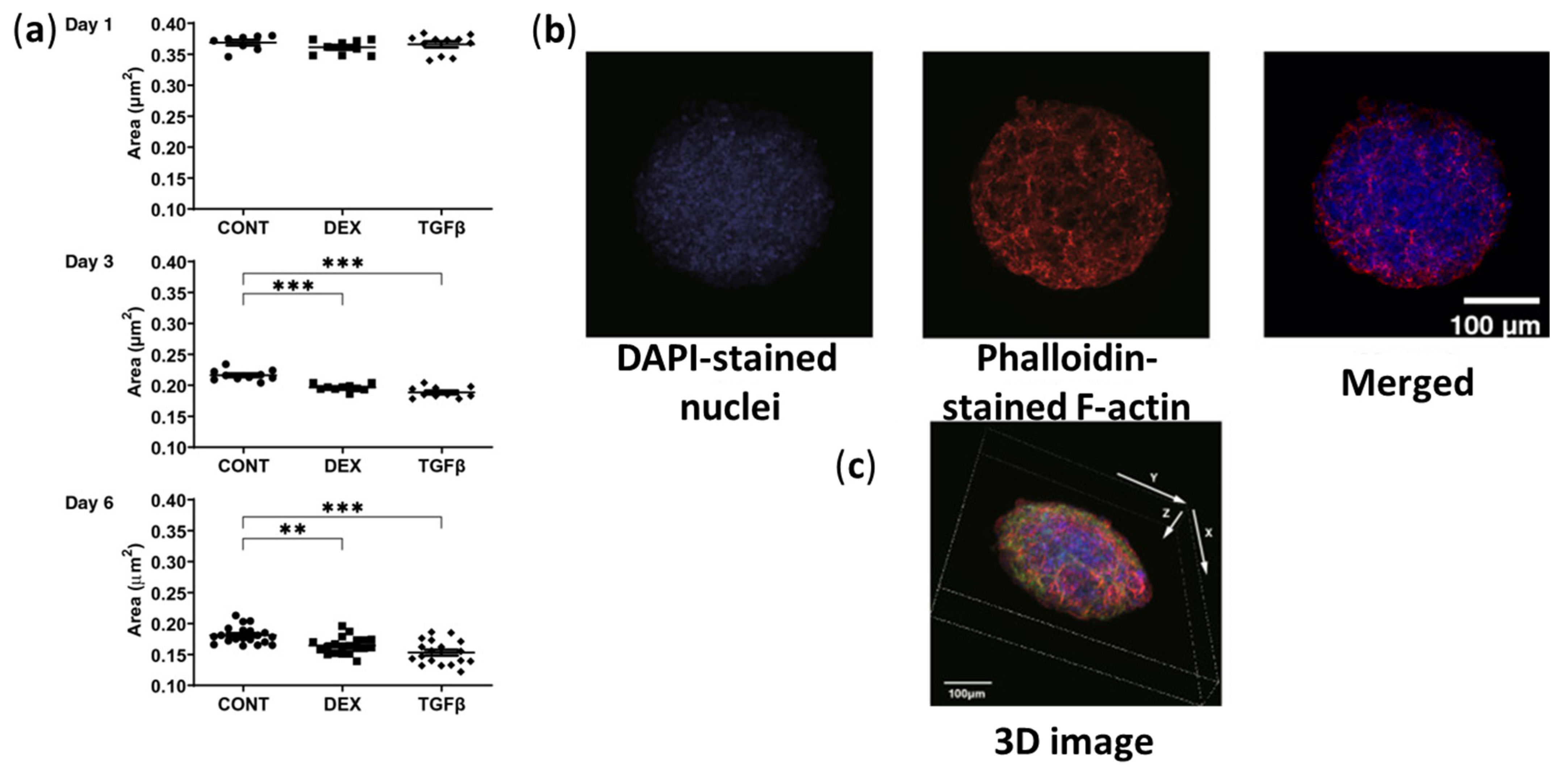
- Watanabe, M.; Ida, Y.; Ohguro, H.; Ota, C.; Hikage, F. Establishment of Appropriate Glaucoma Models Using Dexamethasone or TGFβ2 Treated Three-Dimension (3D) Cultured Human Trabecular Meshwork (HTM) Cells. Sci. Rep. 2021, 11, 19369. [Google Scholar] [CrossRef]
- Jiang, Y.; Che, Y.; Zhang, Y.; Zhu, X.; Wu, C.; Lin, L.; Yu, M.; Yang, Y. Small Extracellular Vesicle Treatment of Trabecular Meshwork Fibrosis: 2D/3D In Vitro and In Vivo Analyses. Investig. Ophthalmol. Vis. Sci. 2025, 66, 48. [Google Scholar] [CrossRef]
- Watanabe, M.; Sato, T.; Yano, T.; Higashide, M.; Ogawa, T.; Nishikiori, N.; Furuhashi, M.; Ohguro, H. mTOR Inhibitors Modulate the Biological Nature of TGF-β2-Treated or -Untreated Human Trabecular Meshwork Cells in Different Manners. Biomedicines 2024, 12, 2604. [Google Scholar] [CrossRef]
- Watanabe, M.; Furuhashi, M.; Tsugeno, Y.; Ida, Y.; Hikage, F.; Ohguro, H. Autotaxin May Have Lysophosphatidic Acid-Unrelated Effects on Three-Dimension (3D) Cultured Human Trabecular Meshwork (HTM) Cells. Int. J. Mol. Sci. 2021, 22, 12039. [Google Scholar] [CrossRef]
- Sundaresan, Y.; Manivannan, L.P.; Radhakrishnan, S.; Ramasamy, K.S.; Veerappan, M.; Chidambaranathan, G.P. Reduction in Trabecular Meshwork Stem Cell Content in Donor Eyes with Primary Open Angle Glaucoma. Sci. Rep. 2021, 11, 24518. [Google Scholar] [CrossRef]
- Sundaresan, Y.; Ramasamy, K.S.; Veerappan, M.; Chidambaranathan, G.P. Functional Characterization of Adult Human Trabecular Meshwork Stem Cells. Exp. Cell Res. 2021, 405, 112709. [Google Scholar] [CrossRef]
- Kuehn, M.H.; Vranka, J.A.; Wadkins, D.; Jackson, T.; Cheng, L.; Ledolter, J. Circumferential Trabecular Meshwork Cell Density in the Human Eye. Exp. Eye Res. 2021, 205, 108494. [Google Scholar] [CrossRef]
- Du, Y.; Roh, D.S.; Mann, M.M.; Funderburgh, M.L.; Funderburgh, J.L.; Schuman, J.S. Multipotent Stem Cells from Trabecular Meshwork Become Phagocytic TM Cells. Investig. Ophthalmol. Vis. Sci. 2012, 53, 1566–1575. [Google Scholar] [CrossRef]
- Du, Y.; Yun, H.; Yang, E.; Schuman, J.S. Stem Cells From Trabecular Meshwork Home to TM Tissue In Vivo. Investig. Ophthalmol. Vis. Sci. 2013, 54, 1450–1459. [Google Scholar] [CrossRef]
- Yun, H.; Wang, Y.; Zhou, Y.; Wang, K.; Sun, M.; Stolz, D.B.; Xia, X.; Ethier, C.R.; Du, Y. Human Stem Cells Home to and Repair Laser-Damaged Trabecular Meshwork in a Mouse Model. Commun. Biol. 2018, 1, 216. [Google Scholar] [CrossRef]
- Xiong, S.; Kumar, A.; Tian, S.; Taher, E.E.; Yang, E.; Kinchington, P.R.; Xia, X.; Du, Y. Stem Cell Transplantation Rescued a Primary Open-Angle Glaucoma Mouse Model. eLife 2021, 10, e63677. [Google Scholar] [CrossRef]
- Iswarya, R.; Krishnadas, S.; Dharmalingam, K.; Gowri Priya, C. Human Trabecular Meshwork Stem Cell-Derived Small Extracellular Vesicles Enhance Trabecular Meshwork Cell Survival and Proliferation. Exp. Eye Res. 2025, 253, 110281. [Google Scholar] [CrossRef]
- Moujane, F.; Zhang, C.; Knight, R.; Lee, J.Y.; Deng, S.X.; Zheng, J.J. Corneal Stromal Stem Cell-Derived Extracellular Vesicles Attenuate ANGPTL7 Expression in the Human Trabecular Meshwork. Transl. Vis. Sci. Technol. 2025, 14, 21. [Google Scholar] [CrossRef]
- Xiong, S.; Xu, Y.; Wang, Y.; Kumar, A.; Peters, D.M.; Du, Y. α5β1 Integrin Promotes Anchoring and Integration of Transplanted Stem Cells to the Trabecular Meshwork in the Eye for Regeneration. Stem. Cells. Dev. 2020, 29, 290–300. [Google Scholar] [CrossRef]
- Zhou, Y.; Xia, X.; Yang, E.; Wang, Y.; Marra, K.G.; Ethier, C.R.; Schuman, J.S.; Du, Y. Adipose-Derived Stem Cells Integrate into Trabecular Meshwork with Glaucoma Treatment Potential. FASEB J. 2020, 34, 7160–7177. [Google Scholar] [CrossRef]
- Mallick, S.; Sharma, M.; Kumar, A.; Du, Y. Cell-Based Therapies for Trabecular Meshwork Regeneration to Treat Glaucoma. Biomolecules 2021, 11, 1258. [Google Scholar] [CrossRef]
- Abu-Hassan, D.W.; Li, X.; Ryan, E.I.; Acott, T.S.; Kelley, M.J. Induced Pluripotent Stem Cells Restore Function in a Human Cell Loss Model of Open-Angle Glaucoma. Stem Cells 2015, 33, 751–761. [Google Scholar] [CrossRef]
- Zhu, W.; Gramlich, O.W.; Laboissonniere, L.; Jain, A.; Sheffield, V.C.; Trimarchi, J.M.; Tucker, B.A.; Kuehn, M.H. Transplantation of iPSC-Derived TM Cells Rescues Glaucoma Phenotypes In Vivo. Proc. Natl. Acad. Sci. USA 2016, 113, E3492–E3500. [Google Scholar] [CrossRef]
- Zhu, W.; Jain, A.; Gramlich, O.W.; Tucker, B.A.; Sheffield, V.C.; Kuehn, M.H. Restoration of Aqueous Humor Outflow Following Transplantation of iPSC-Derived Trabecular Meshwork Cells in a Transgenic Mouse Model of Glaucoma. Investig. Ophthalmol. Vis. Sci. 2017, 58, 2054–2062. [Google Scholar] [CrossRef]
- Zhu, W.; Godwin, C.R.; Cheng, L.; Scheetz, T.E.; Kuehn, M.H. Transplantation of iPSC-TM Stimulates Division of Trabecular Meshwork Cells in Human Eyes. Sci. Rep. 2020, 10, 2905. [Google Scholar] [CrossRef]
- Sui, S.; Yu, H.; Wang, X.; Wang, W.; Yang, X.; Pan, X.; Zhou, Q.; Xin, C.; Du, R.; Wu, S.; et al. iPSC-Derived Trabecular Meshwork Cells Stimulate Endogenous TM Cell Division through Gap Junction in a Mouse Model of Glaucoma. Investig. Ophthalmol. Vis. Sci. 2021, 62, 28. [Google Scholar] [CrossRef]
- Xi, G.; Feng, P.; Zhang, X.; Wu, S.; Zhang, J.; Wang, X.; Xiang, A.; Xu, W.; Wang, N.; Zhu, W. iPSC-Derived Cells Stimulate ABCG2(+)/NES(+) Endogenous Trabecular Meshwork Cell Proliferation and Tissue Regeneration. Cell Prolif. 2024, 57, e13611. [Google Scholar] [CrossRef]
- Manuguerra-Gagné, R.; Boulos, P.R.; Ammar, A.; Leblond, F.A.; Krosl, G.; Pichette, V.; Lesk, M.R.; Roy, D.C. Transplantation of Mesenchymal Stem Cells Promotes Tissue Regeneration in a Glaucoma Model through Laser-Induced Paracrine Factor Secretion and Progenitor Cell Recruitment. Stem Cells 2013, 31, 1136–1148. [Google Scholar] [CrossRef]
- Roubeix, C.; Godefroy, D.; Mias, C.; Sapienza, A.; Riancho, L.; Degardin, J.; Fradot, V.; Ivkovic, I.; Picaud, S.; Sennlaub, F.; et al. Intraocular Pressure Reduction and Neuroprotection Conferred by Bone Marrow-Derived Mesenchymal Stem Cells in an Animal Model of Glaucoma. Stem Cell Res. Ther. 2015, 6, 177. [Google Scholar] [CrossRef]
- Chang, Y.F.; Cheng, Y.H.; Ko, Y.C.; Chiou, S.H.; Liu, C.J. Anti-Apoptotic and Autophagic Effect: Using Conditioned Medium from Human Bone Marrow Mesenchymal Stem Cells to Treat Human Trabecular Meshwork Cells. Regen. Ther. 2023, 22, 50–58. [Google Scholar] [CrossRef]
- Sihota, R.; Sen, S.; Mohanty, S.; Ahmad, M.; Ravi, A.; Gupta, V.; Bhatla, N. Effect of Intracameral Human Cord Blood-Derived Stem Cells on Lasered Rabbit Trabecular Meshwork. Int. Ophthalmol. 2019, 39, 2757–2766. [Google Scholar] [CrossRef]
- Kumar, A.; Xu, Y.; Du, Y. Stem Cells from Human Trabecular Meshwork Hold the Potential to Develop into Ocular and Non-Ocular Lineages After Long-Term Storage. Stem Cells Dev. 2020, 29, 49–61. [Google Scholar] [CrossRef]
- Ding, Q.J.; Zhu, W.; Cook, A.C.; Anfinson, K.R.; Tucker, B.A.; Kuehn, M.H. Induction of Trabecular Meshwork Cells from Induced Pluripotent Stem Cells. Investig. Ophthalmol. Vis. Sci. 2014, 55, 7065–7072. [Google Scholar] [CrossRef]
- Kumar, A.; Cheng, T.; Song, W.; Cheuk, B.; Yang, E.; Yang, L.; Xie, Y.; Du, Y. Two-Step Induction of Trabecular Meshwork Cells from Induced Pluripotent Stem Cells for Glaucoma. Biochem. Biophys. Res. Commun. 2020, 529, 411–417. [Google Scholar] [CrossRef]
- Wang, W.; Miao, Y.; Sui, S.; Wang, Y.; Wu, S.; Cao, Q.; Duan, H.; Qi, X.; Zhou, Q.; Pan, X.; et al. Xeno- and Feeder-Free Differentiation of Human iPSCs to Trabecular Meshwork-Like Cells by Recombinant Cytokines. Transl. Vis. Sci. Technol. 2021, 10, 27. [Google Scholar] [CrossRef]
- Feng, P.; Yu, C.; Zhang, X.; Xu, B.; Wu, S.; Xin, C.; Littleton, S.; Kang, J.; Wang, X.; Liang, S.; et al. iPSC-Derived ITGA6-Positive Cells Restore Aqueous Humor Outflow in Glaucoma Eyes. Nat. Commun. 2025, 16, 9441. [Google Scholar] [CrossRef]
- Snider, E.J.; Kubelick, K.P.; Tweed, K.; Kim, R.K.; Li, Y.; Gao, K.; Read, A.T.; Emelianov, S.; Ethier, C.R. Improving Stem Cell Delivery to the Trabecular Meshwork Using Magnetic Nanoparticles. Sci. Rep. 2018, 8, 12251. [Google Scholar] [CrossRef]
- Bahranifard, M.R.; Chan, J.; Read, A.T.; Li, G.; Cheng, L.; Safa, B.N.; Siadat, S.M.; Jhunjhunwala, A.; Grossniklaus, H.E.; Emelianov, S.Y.; et al. Magnetically Steered Cell Therapy for Reduction of Intraocular Pressure as a Treatment Strategy for Open-Angle Glaucoma. eLife 2025, 13, RP103256. [Google Scholar] [CrossRef]
- Goel, M.; Picciani, R.G.; Lee, R.K.; Bhattacharya, S.K. Aqueous Humor Dynamics: A Review. Open Ophthalmol. J. 2010, 4, 52–59. [Google Scholar] [CrossRef]
- Johnstone, M. Outflow Resistance: An Analysis of Early and Recent Research. Glaucoma Today (Bench Bedside) 2015, 13–15. Available online: https://glaucomatoday.com/articles/2015-jan-feb/outflow-resistance (accessed on 24 February 2026).
- Johnson, D.H.; Tschumper, R.C. Human Trabecular Meshwork Organ Culture. A New Method. Investig. Ophthalmol. Vis. Sci. 1987, 28, 945–953. [Google Scholar]
- Comes, N.; Borrás, T. Individual Molecular Response to Elevated Intraocular Pressure in Perfused Postmortem Human Eyes. Physiol. Genom. 2009, 38, 205–225. [Google Scholar] [CrossRef]
- Erickson-Lamy, K.; Rohen, J.W.; Grant, W.M. Outflow Facility Studies in the Perfused Human Ocular Anterior Segment. Exp. Eye Res. 1991, 52, 723–731. [Google Scholar] [CrossRef]
- Bárány, E.H. Simultaneous Measurement of Changing Intraocular Pressure and Outflow Facility in the Vervet Monkey by Constant Pressure Infusion. Investig. Ophthalmol. Vis. Sci. 1964, 3, 135–143. [Google Scholar]
- Hu, Y.; Gabelt, B.T.; Kaufman, P.L. Monkey Organ-Cultured Anterior Segments: Technique and Response to H-7. Exp. Eye Res. 2006, 82, 1100–1108. [Google Scholar] [CrossRef]
- Tian, B.; Hu, Y.; Gabelt, B.T.; Kaufman, P.L. Factors Affecting Outflow Facility Calculations. Exp. Eye Res. 2006, 83, 1515–1520. [Google Scholar] [CrossRef]
- Sherwood, J.M.; Reina-Torres, E.; Bertrand, J.A.; Rowe, B.; Overby, D.R. Measurement of Outflow Facility Using iPerfusion. PLoS ONE 2016, 11, e0150694. [Google Scholar] [CrossRef]
- Kizhatil, K.; Chlebowski, A.; Tolman, N.G.; Freeburg, N.F.; Ryan, M.M.; Shaw, N.N.; Kokini, A.D.; Marchant, J.K.; John, S.W. An In Vitro Perfusion System to Enhance Outflow Studies in Mouse Eyes. Investig. Ophthalmol. Vis. Sci. 2016, 57, 5207–5215. [Google Scholar] [CrossRef]
- Millar, J.C.; Clark, A.F.; Pang, I.-H. Assessment of Aqueous Humor Dynamics in the Mouse by a Novel Method of Constant-Flow Infusion. Investig. Ophthalmol. Vis. Sci. 2011, 52, 685–694. [Google Scholar] [CrossRef]
- Perkins, T.W.; Alvarado, J.A.; Polansky, J.R.; Stilwell, L.; Maglio, M.; Juster, R. Trabecular Meshwork Cells Grown on Filters. Conductivity and Cytochalasin Effects. Investig. Ophthalmol. Vis. Sci. 1988, 29, 1836–1846. [Google Scholar]
- Roberts, C.J.; Rivera, B.K.; Grzybowski, D.M.; Mahmoud, A.M.; Weber, P.A. Effect of Low Fluence Diode Laser Irradiation on the Hydraulic Conductivity of Perfused Trabecular Meshwork Endothelial Cell Monolayers. Curr. Eye Res. 2007, 32, 625–638. [Google Scholar] [CrossRef]
- Dai, W.; Li, M. Experimental Study of Growth of Trabecular Cells on the Filters and Hydraulic Conductivity. Chin. Med. J. 1999, 112, 207–210. [Google Scholar]
- Alvarado, J.A.; Murphy, C.G.; Franse-Carman, L.; Chen, J.; Underwood, J.L. Effect of Beta-Adrenergic Agonists on Paracellular Width and Fluid Flow Across Outflow Pathway Cells. Investig. Ophthalmol. Vis. Sci. 1998, 39, 1813–1822. [Google Scholar]
- Burke, A.G.; Zhou, W.; O’Brien, E.T.; Roberts, B.C.; Stamer, W.D. Effect of Hydrostatic Pressure Gradients and Na2EDTA on Permeability of Human Schlemm’s Canal Cell Monolayers. Curr. Eye Res. 2004, 28, 391–398. [Google Scholar] [CrossRef]
- Lee, O.T.; Wong, J.; Liepmann, D.; Lang, T.; Lin, S. Ultrastructure and Fluid Flow Physiology of Fetal Trabecular Meshwork Cells. Curr. Eye Res. 2008, 33, 849–856. [Google Scholar] [CrossRef]
- Lu, R.; Kolarzyk, A.M.; Stamer, W.D.; Lee, E. Human Ocular Fluid Outflow On-Chip Reveals Trabecular Meshwork-Mediated Schlemm’s Canal Endothelial Dysfunction in Steroid-Induced Glaucoma. Nat. Cardiovasc. Res. 2025, 4, 1066–1076. [Google Scholar] [CrossRef]
- Ryskamp, D.A.; Frye, A.M.; Phuong, T.T.; Yarishkin, O.; Jo, A.O.; Xu, Y.; Lakk, M.; Iuso, A.; Redmon, S.N.; Ambati, B.; et al. TRPV4 Regulates Calcium Homeostasis, Cytoskeletal Remodeling, Conventional Outflow and Intraocular Pressure in The Mammalian Eye. Sci. Rep. 2016, 6, 30583. [Google Scholar] [CrossRef]
- Redmon, S.N.; Lakk, M.; Tseng, Y.T.; Rudzitis, C.N.; Searle, J.E.; Ahmed, F.; Unser, A.M.; Borrás, T.; Torrejon, K.; Križaj, D. TRPV4 Controls Circadian and Pathological Ocular Hypertension. J. Physiol. 2025, 603, 4091–4111. [Google Scholar] [CrossRef]
- Li, G.; Torrejon, K.Y.; Unser, A.M.; Ahmed, F.; Navarro, I.D.; Baumgartner, R.A.; Albers, D.S.; Stamer, W.D. Trabodenoson, an Adenosine Mimetic With A1 Receptor Selectivity Lowers Intraocular Pressure by Increasing Conventional Outflow Facility in Mice. Investig. Ophthalmol. Vis. Sci. 2018, 59, 383–392. [Google Scholar] [CrossRef]
- Bastia, E.; Toris, C.B.; Brambilla, S.; Galli, C.; Almirante, N.; Bergamini, M.V.W.; Masini, E.; Sgambellone, S.; Unser, A.M.; Ahmed, F.; et al. NCX 667, a Novel Nitric Oxide Donor, Lowers Intraocular Pressure in Rabbits, Dogs, and Non-Human Primates and Enhances TGFβ2-Induced Outflow in HTM/HSC Constructs. Investig. Ophthalmol. Vis. Sci. 2021, 62, 17. [Google Scholar] [CrossRef]
- Galli, C.; Bastia, E.; Hubatsch, D.A.; Toris, C.; Fan, S.; Unser, A.; Ahmed, F.; Torrejon, K.Y.; Impagnatiello, F. NCX 470 Reduces Intraocular Pressure More Effectively Than Lumigan in Dogs and Enhances Conventional and Uveoscleral Outflow in Non-Human Primates and Human Trabecular Meshwork/Schlemm’s Canal Constructs. J. Ocul. Pharmacol. Ther. 2024, 40, 389–396. [Google Scholar] [CrossRef]
- Brown, S.F.; Nguyen, H.; Mzyk, P.; De Ieso, M.L.; Unser, A.M.; Brown, I.; Ramesh, P.; Afzaal, H.; Ahmed, F.; Torrejon, K.Y.; et al. ANGPTL7 and Its Role in IOP and Glaucoma. Investig. Ophthalmol. Vis. Sci. 2024, 65, 22. [Google Scholar] [CrossRef]
- Lu, R.; Soden, P.A.; Lee, E. Tissue-Engineered Models for Glaucoma Research. Micromachines 2020, 11, 612. [Google Scholar] [CrossRef]
- Johnson, M.; Overby, D.R.; Stamer, W.D.; Ethier, C.R. Scientific Concerns About Recent Fluid-Structure Interaction Models of the Aqueous Humor Outflow Pathway. Investig. Ophthalmol. Vis. Sci. 2025, 66, 18. [Google Scholar] [CrossRef] [PubMed]
- Dautriche, C.N.; Xie, Y.; Sharfstein, S.T. Walking through Trabecular Meshwork Biology: Toward Engineering Design of Outflow Physiology. Biotechnol. Adv. 2014, 32, 971–983. [Google Scholar] [CrossRef] [PubMed]
- Maus, T.L.; Brubaker, R.F. Measurement of Aqueous Humor Flow by Fluorophotometry in the Presence of a Dilated Pupil. Investig. Ophthalmol. Vis. Sci. 1999, 40, 542–546. [Google Scholar]
- Moses, R.A. Circumferential Flow in Schlemm’s Canal. Am. J. Ophthalmol. 1979, 88, 585–591. [Google Scholar] [CrossRef]
- Buller, C.; Johnson, D. Segmental Variability of the Trabecular Meshwork in Normal and Glaucomatous Eyes. Investig. Ophthalmol. Vis. Sci. 1994, 35, 3841–3851. [Google Scholar]
- Johnson, M.; Chan, D.; Read, A.T.; Christensen, C.; Sit, A.; Ethier, C.R. The Pore Density in the Inner Wall Endothelium of Schlemm’s Canal of Glaucomatous Eyes. Investig. Ophthalmol. Vis. Sci. 2002, 43, 2950–2955. [Google Scholar]
- McEwen, W.K. Application of Poiseuille’s Law To Aqueous Outflow. AMA Arch. Ophthalmol. 1958, 60, 290–294. [Google Scholar] [CrossRef] [PubMed]
- HOLMBERG, A. The Fine Structure of the Inner Wall of Schlemm’s Canal. AMA Arch. Ophthalmol. 1959, 62, 956–958. [Google Scholar] [CrossRef]
- Vass, C.; Hirn, C.; Unger, E.; Mayr, W.; Georgopoulos, M.; Rainer, G.; Richter–Mueksch, S. Human Aqueous Humor Viscosity in Cataract, Primary Open Angle Glaucoma and Pseudoexfoliation Syndrome. Investig. Ophthalmol. Vis. Sci. 2004, 45, 5030. [Google Scholar]



| Cell Source | Strategy | Duration | MYOC | CHI3L1 | Key TM Markers Reported | Differentiation Efficiency | Functional Validation | Refs. |
|---|---|---|---|---|---|---|---|---|
| Human TMSC | SP/clone isolation → AH or FBS | ≤10 d | DEX-inducible | Inducible | AQP1, MGP, TIMP3 | NQ | Phagocytosis; TM marker induction | [170] |
| Human ADSC | TM-ECM + TM-CM or co-culture TM induction | ~10 d | DEX-inducible | Positive | AQP1, CHI3L1 | Phagocytosis-positive cells ~65–86% | Phagocytosis; DEX response (MYOC, CLAN formation) | [158] |
| Mouse iPSC | Co-culture with human TM cells (HTM-5; Transwell inserts) → TM-like induction | 7–21 d | DEX-inducible | NR | CAV1, COL4A5, MGP, TIMP3, VCAM1, MYOC | NQ | Phagocytosis; MMP3 secretion; DEX-induced MYOC secretion | [171] |
| Mouse iPSC | TM-CM media–induced TM-like differentiation | ~7–14 d | DEX-inducible | NR | LAMA4, TIMP3; | NQ | CLAN formation; TM-like phenotype; TM proliferation support (Cx43-dependent) | [161,164] |
| Human iPSC | Co-culture with primary TM cells | ~60 d | Positive | Positive | Vimentin, AQP1, MGP, COL I/IV, TIMP3 | NQ | Phagocytosis; transcriptomic similarity to TM | [163] |
| Human iPSC | Two-step: iPSC → NCCs (NGFR/HNK1+) → TM induction on TM-ECM + TM-CM | NCCs ~10 d; TM ~10–14 d | DEX-inducible | Positive (DEX-responsive) | CHI3L1; DEX ↑MYOC, ↑ANGPTL7 | NCC induction efficiency ~85% (NGFR+) | DEX response (MYOC, ANGPTL7); CLAN formation | [172] |
| Human iPSC | EB differentiation on TM-ECM + TM-CM | ~30 d | NR | Positive | CHI3L1, WNT1, α3-integrin, AQP1 | NR | Phagocytosis | [160] |
| Human iPSC | Two-stage cytokine-driven TM-like differentiation (xeno-free) | Stage1: 7 d; Stage2: 14 d | DEX-inducible | NR | LAMA4, TIMP3, AQP1, MYOC, COLIV | NQ | CLAN formation; TM-like morphology; transcriptomic similarity to TM | [173,174] |
| Model | Cells/Components | Matrix/ Material | Flow and Mechanical Control | Primary Readouts | Advantages | Limitations | Refs. |
|---|---|---|---|---|---|---|---|
| Measurement of outflow facility | |||||||
| Ex vivo anterior segment perfusion | Whole anterior segment (donor) | Native corneal/limbal tissue ECM | Constant flow perfusion or constant pressure perfusion | Whole-tissue outflow facility, IOP response, histology | Closest to the in vivo physiology | Low throughput; donor variability; tissue availability | [34] |
| ACOS | Primary and stem cell-derived HTM and HSC cells | Micropatterned porous SU-8 scaffolds in perfusion chamber | Constant flow perfusion at 2–16 or 2–40 µL/min while monitoring pressure | Simulated outflow facility, cell phenotype, drug response | Reflecting pressure-dependent outflow resistance and dynamic HTM physiology; high throughput; high producibility; consistent; perfusion ready | Cleanroom needed for fabrication; non-physiologically relevant elastic modulus of SU-8 scaffolds (much higher than HTM ECM) | [129,131,132,133,134] |
| Nanofibrous scaffold-based JCT model | Acellular scaffold | Electrospun nanofibrous PCL scaffolds in perfusion chamber | Constant flow perfusion at 10, 20, 40, 80, and 160 μL/min while measuring pressure | Simulated outflow facility of the scaffold alone | Design flexibility; perfusion ready | Elastic modulus of PCL scaffolds higher than HTM ECM; high flow rates not suitable for cell perfusion | [136] |
| Measurement of hydraulic conductivity | |||||||
| Filter membrane-based perfusion | HTM cells HSC cells HSC cells Fetal HTM Adult HTM | 0.45 µm Millicell filters 0.45 µm Millicell filters 0.4 µm Snapwell filter 0.45 µm Millicell filters | Constant pressure perfusion at 5 mm Hg; under an elevated pressure with 0.5 mmHg/µL/min resistor; at 6 mmHg with gradual decrease; constant flow perfusion at 20 µL/min | Hydraulic conductivity, drug response | Easy access; perfusion ready | Limiting to a basic permeability readout rather than a full functional assessment of HTM outflow behavior; lack of 3D ECM context; mismatched stiffness and pore size with HTM | [188,189,190] [191] [192] [193] |
| Measurement of outflow rate | |||||||
| Ocular fluid outflow on-chip | HTM cells | Microfluidic PDMS chip with 2-parallel cylindrical microchannels in collagen hydrogel | Constant pressure perfusion under elevated hydraulic pressure | Average flow velocity (i.e., dividing total outflow volume by duration of time) | Physiologically relevant ECM mimics and flow velocity | Complicated fabrication protocol; lack of outflow facility measurement | [194] |
| Demonstration of pressure-dependent outflow resistance (pressure vs. time graph) at only one single flow rate | |||||||
| Hydrogel-based HTM model | HTM cells | Lyophilized collagen–GAG scaffolds | Constant flow perfusion at 61.09 µL/min | Pressure vs. time, cell phenotype, drug response | ECM mimics biochemically and biomechanically | Flow instability; inability to calculate outflow facility | [109] |
| Primary HTM cells | MAX8B peptide hydrogels | Constant flow perfusion at 3 µL/min | [119] | ||||
| Multilayer HTM model | Primary HTM cells | MEW-printed PCL stacks | Constant flow perfusion at 4 μL/min | Mimicry of JCT, CSM, and UVM | [142] | ||
| 3D Model | Treatment | Duration | Outflow Facility (μL/min/mmHg/mm2) | Reference |
|---|---|---|---|---|
| 3D HTM cell–scaffold constructs | ||||
| Primary HTM cells grown on SU-8 scaffolds for 14 days | Vehicle control 300 nM PA | >7 days | 0.131 ± 0.003 0.093 ± 0.004 | [133] |
| Vehicle control 2.5 ng/mL TGFβ2 10 µM Y27632 TGFβ2 + Y27632 | 9 days | ~0.17 ~0.02 ~0.24 ~0.16 | [134] | |
| Vehicle control 25 nM GSK101 1 μM HC-067047 | 24 h | ~0.25 ~0.18 ~0.4 | [195] | |
| Primary HTM cells grown on SU-8 scaffolds for 12–14 days, treated with 500 nM DEX for 3 days (3D glaucomatous model) | Vehicle control 1% PA 1% PA + 1 µm HC-067047 (TRPV4 antagonist) | 6 days | 0.32–0.37 0.08–0.12 0.45–0.66 | [196] |
| 3D HTM/HSC cell–scaffold constructs | ||||
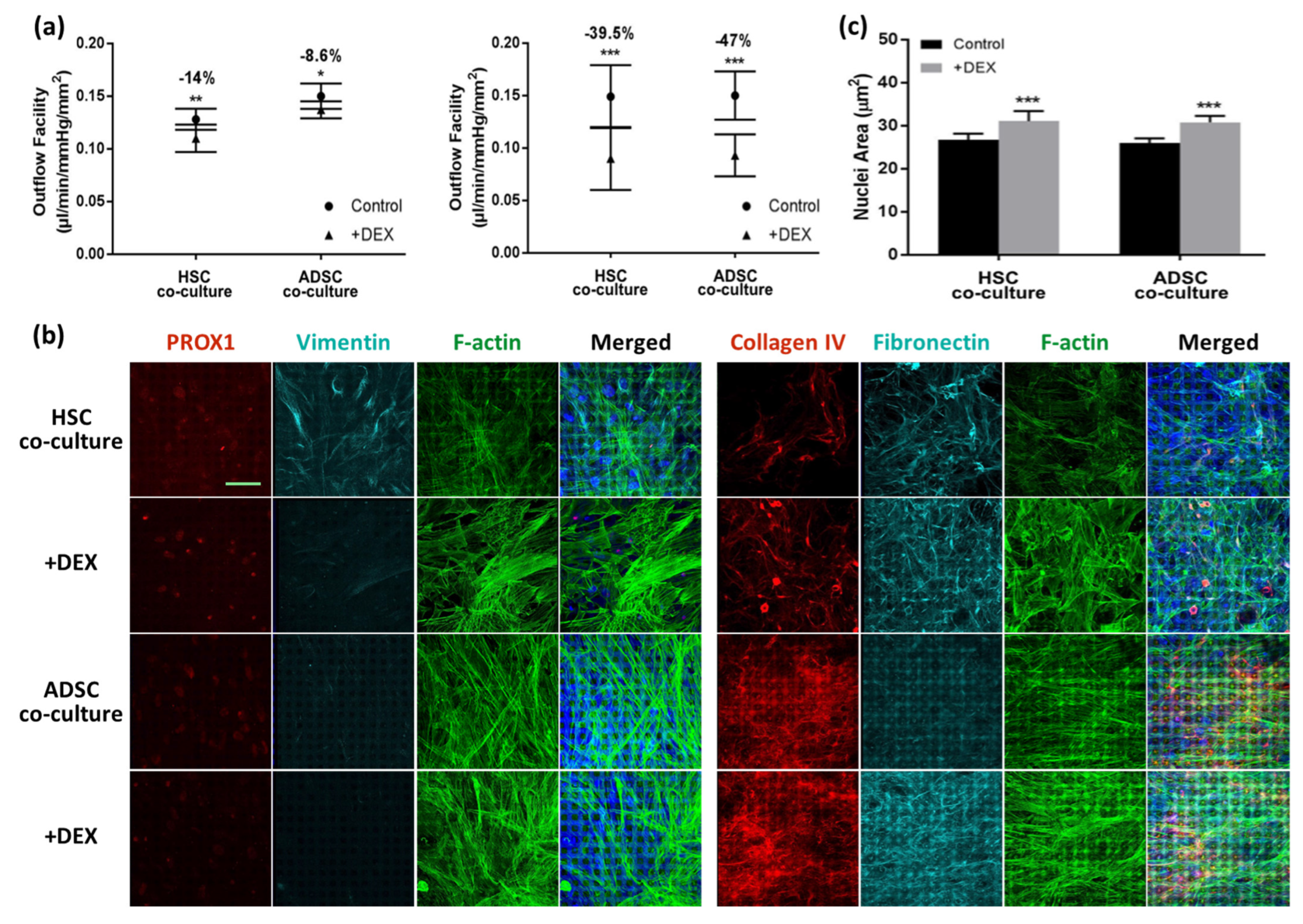
| Primary HTM cells grown on SU-8 until confluency and co-cultured with HSC cells for 7–10 days | Vehicle control 500 nM DEX 0.56 nM ANGPTL7 1.1 nM ANGPTL7 3.3. nM ANGPTL7 | 9 days | 0.35–0.90 0.075–0.18 0.51–1.0 0.26–0.74 0.16–0.63 | [200] |
| Primary HTM cells grown on SU-8 until confluency and co-cultured with HSC cells for 10 days, and then treated with TGFβ2 (2.5 ng/mL) for 6 days (3D glaucomatous model) | Vehicle control 100 µM NCX 667 | Perfused for 20 h (5 h at each of 4 flow rates) | 0.08 ± 0.04 0.31 ± 0.10 | [198] |
| Primary HTM cells grown on SU-8 until confluency and co-cultured with HSC cells for 10 days, and then treated with TGFβ2 (5 ng/mL) for 6 days (3D glaucomatous model) | Naive (no TGFβ2 pretreatment) Vehicle control 10 µM NCX 470 10 µM bimatoprost | Perfused for 20 h (5 h at each of 4 flow rates) | 0.73 ± 0.04 0.47 ± 0.02 0.76 ± 0.03 0.67 ± 0.04 | [199] |
| Key Features | Scaffold-Free Organoid | Hydrogel-Based | SU-8 Scaffold-Based | PCL Scaffold-Based | Outflow On-Chip |
|---|---|---|---|---|---|
| Tissue Architecture | JCT-like | JCT | JCT | JCT/CSM/UVM | JCT |
| Cellular Phenotype and Function | |||||
| - Marker expression | Yes | Yes | Yes | Yes | Yes |
| - ECM deposition | Yes | Yes | Yes | Yes | |
| - Cytoskeletal organization | Yes | Yes | Yes | Yes | |
| - Phagocytic activity | Yes | Yes | |||
| - Steroid responsiveness | Yes | Yes | Yes | Yes | |
| - Regulation of ECM turnover | Yes | Yes | |||
| Biomechanics | |||||
| - ECM composition | Yes | Gelatin-coated | Gelatin-coated | collagen | |
| - Stiffness | Yes | Yes | |||
| - Mechanosensitivity | Yes | Yes | |||
| - Contractility | Yes | Yes | |||
| Hydrodynamics | |||||
| - Outflow facility | Yes [131,132,133,134] | Scaffold only [136] | |||
| - Pressure-dependent outflow resistance | Yes | ||||
| - Hydraulic conductivity | Yes [130] | ||||
| - Outflow rate | Yes [194] | ||||
| HTM-HSC interaction | Yes [132] | Yes [194] | |||
| Limitations | Scaffold-free organoid | Hydrogel-based | SU-8 Scaffold-based | PCL Scaffold-based | Outflow on-chip |
| - Long-term stability | + | ++ to +++ | ++++ | ++++ | ++++ |
| - Reproducibility | + | ++ | +++++ | ++++ | ++++ |
| - Scalability | +++++ | ++++ | +++ | +++ | ++ |
| Parameters | Estimated Values | |
|---|---|---|
| Outflow rate (Q) | 2.5 µL/min [204] | |
| Flow velocity through TM (v) | 10 µm/s [202] | v = Q/(π d h ε) |
| HTM dimensions | ||
| Average height in the anterior–posterior direction (h) Internal limbal diameter (d) | 300 µm [205] 11.6 mm [205] | |
| Circumference (π d) | 36.4 mm [205] | |
| Porosity (ε) | 37.4 ± 5.9% [206] | |
| Flow velocity through the inner wall of SC (v) | ~4000–5000 µm/s [202] | v = 4 Q/(n Aiw π D2) |
| Inner wall pore density (n) | 835 pores/mm2 [207] | |
| Inner wall area (Aiw) | 11 mm2 [208] | |
| Pore diameter (D) | 1.2 µm [202] (0.5–1.5 µm) [209] | |
| Hydrodynamic shear stress (τ) | 0.01 Pa [202] | τ = µv/D |
| Viscosity of aqueous humor (µ) Characteristic length scale of the flow (D) | 7 × 10−4 Pa s [202,210] 0.5 µm [202] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Valarezo, A.; Ramesh, P.; Du, R.; Sharma, R.; Davis, E.; Sharfstein, S.T.; Danias, J.; Du, Y.; Xie, Y. Bioengineered 3D Human Trabecular Meshwork Models for Outflow Physiology and Glaucoma Research. Bioengineering 2026, 13, 291. https://doi.org/10.3390/bioengineering13030291
Valarezo A, Ramesh P, Du R, Sharma R, Davis E, Sharfstein ST, Danias J, Du Y, Xie Y. Bioengineered 3D Human Trabecular Meshwork Models for Outflow Physiology and Glaucoma Research. Bioengineering. 2026; 13(3):291. https://doi.org/10.3390/bioengineering13030291
Chicago/Turabian StyleValarezo, Andrea, Pujhitha Ramesh, Rong Du, Rohit Sharma, Evan Davis, Susan T. Sharfstein, John Danias, Yiqin Du, and Yubing Xie. 2026. "Bioengineered 3D Human Trabecular Meshwork Models for Outflow Physiology and Glaucoma Research" Bioengineering 13, no. 3: 291. https://doi.org/10.3390/bioengineering13030291
APA StyleValarezo, A., Ramesh, P., Du, R., Sharma, R., Davis, E., Sharfstein, S. T., Danias, J., Du, Y., & Xie, Y. (2026). Bioengineered 3D Human Trabecular Meshwork Models for Outflow Physiology and Glaucoma Research. Bioengineering, 13(3), 291. https://doi.org/10.3390/bioengineering13030291









