Development of Antibacterial Dentures Using Titanium Apatite Peening
Abstract
1. Introduction
2. Materials and Methods
2.1. Effect of Peening Mass Flow Rate on Titanium Apatite Transfer
2.1.1. Peening Apparatus
2.1.2. Particles and Test Specimens
2.1.3. Experimental Conditions and Evaluation Methods
2.1.4. Antimicrobial Evaluation
2.2. Effect of Total Peening Mass on Titanium Apatite Transfer
2.2.1. Experimental Conditions and Evaluation Methods
2.2.2. Antimicrobial Evaluation
2.3. Statistical Analysis
3. Results
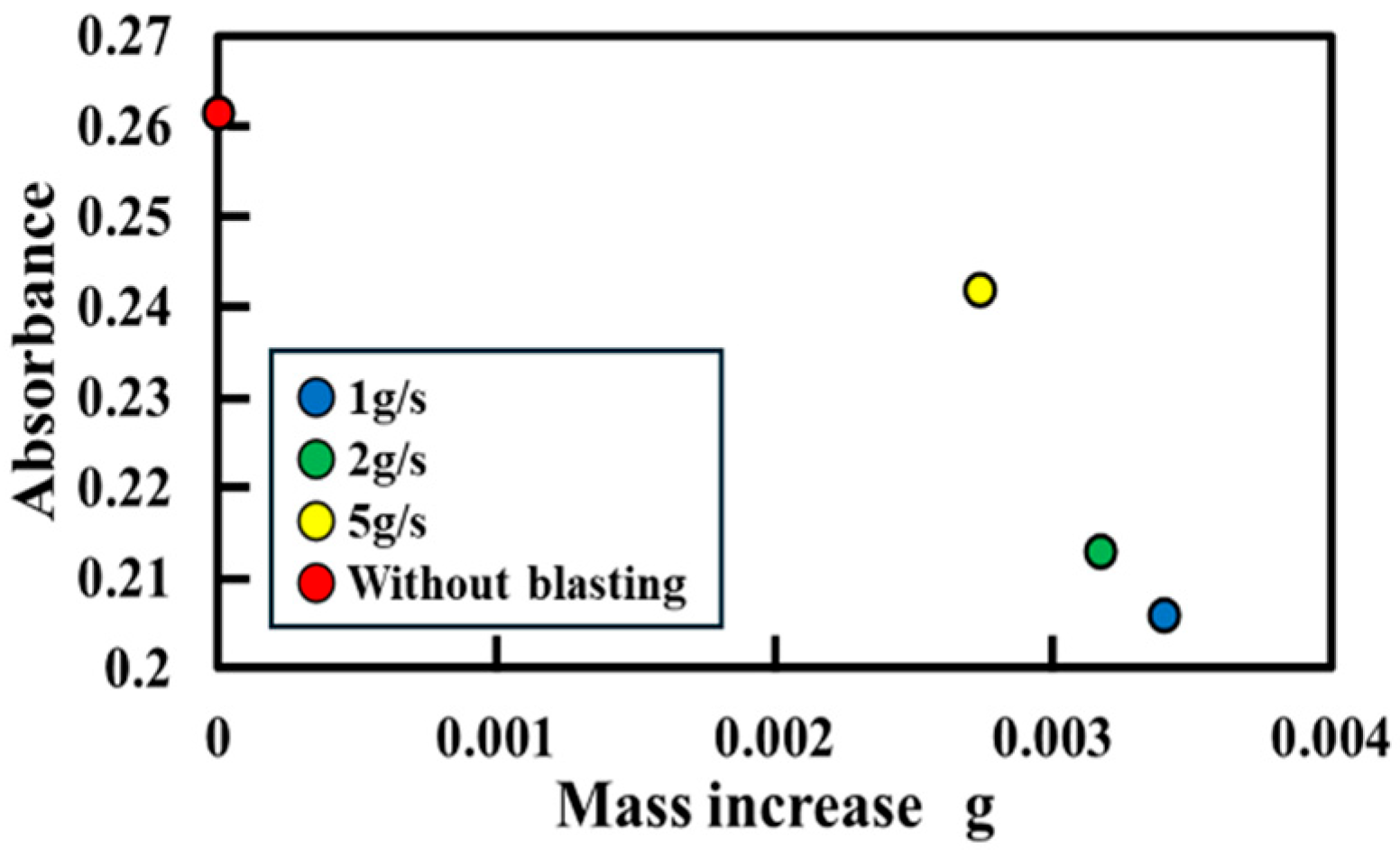
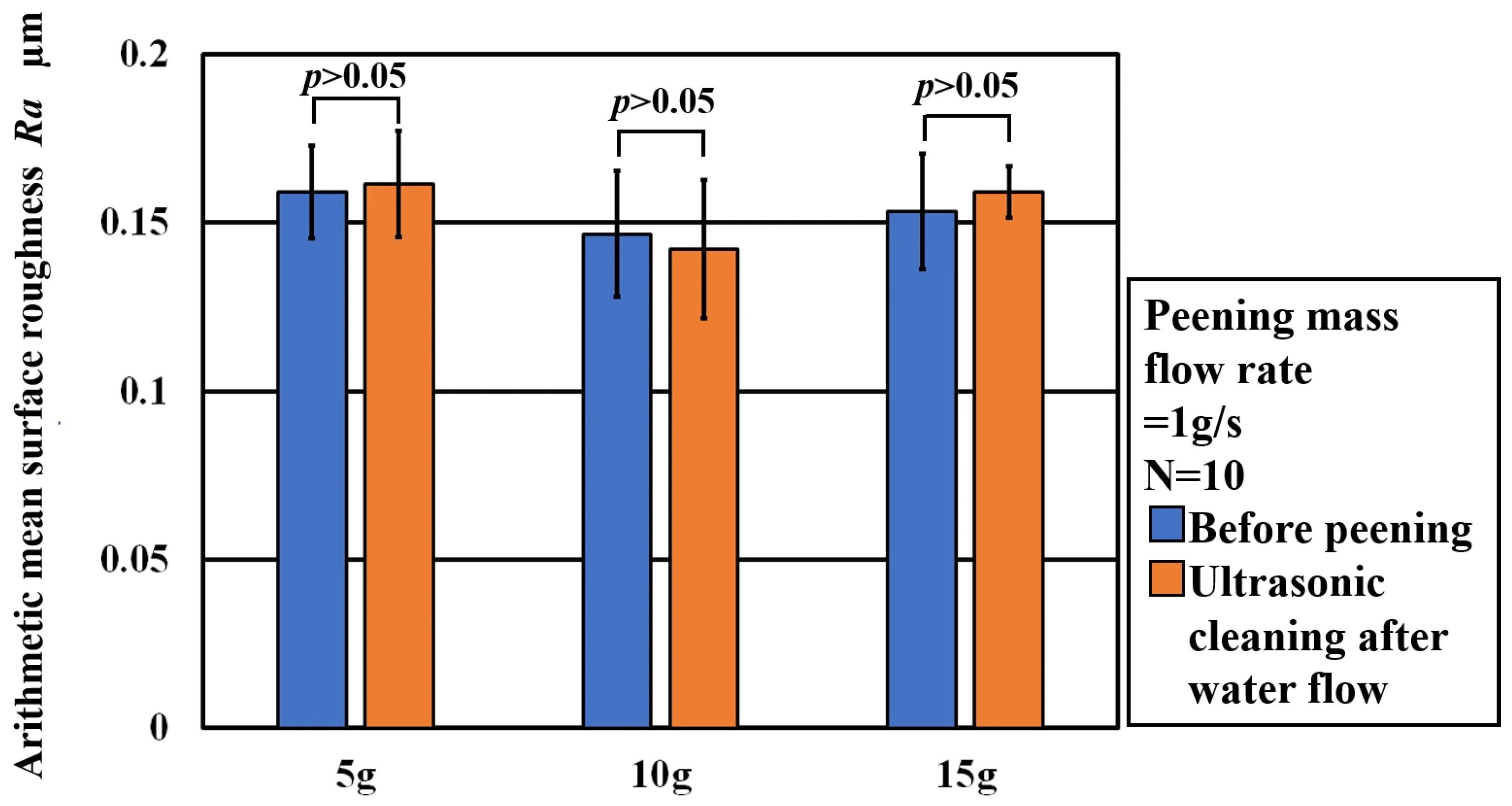
3.1. Effect of Peening Mass Flow Rate on Titanium Apatite Transfer
3.1.1. Surface and Mass Analysis After Water Flow Testing
3.1.2. Antimicrobial Evaluation
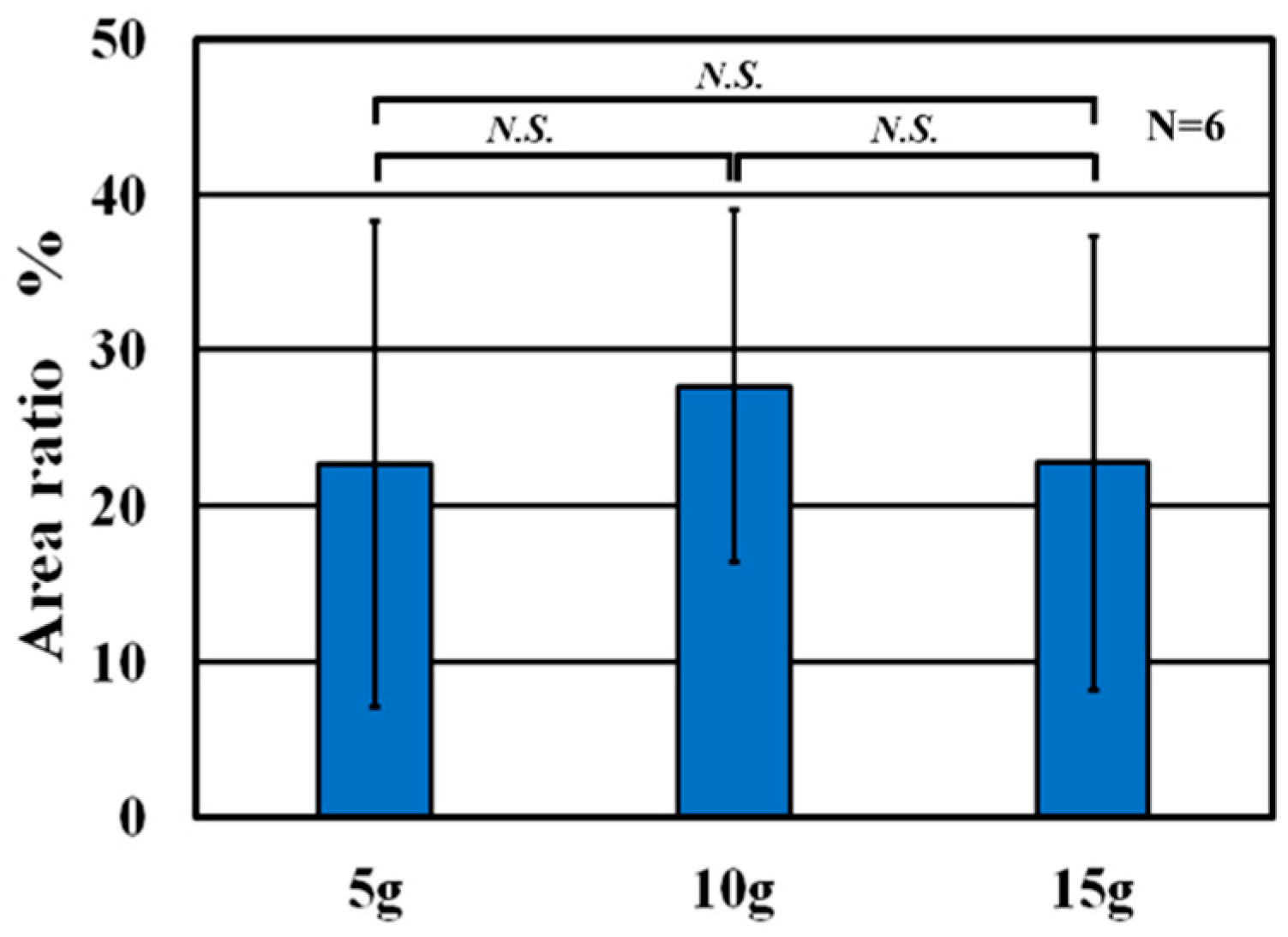
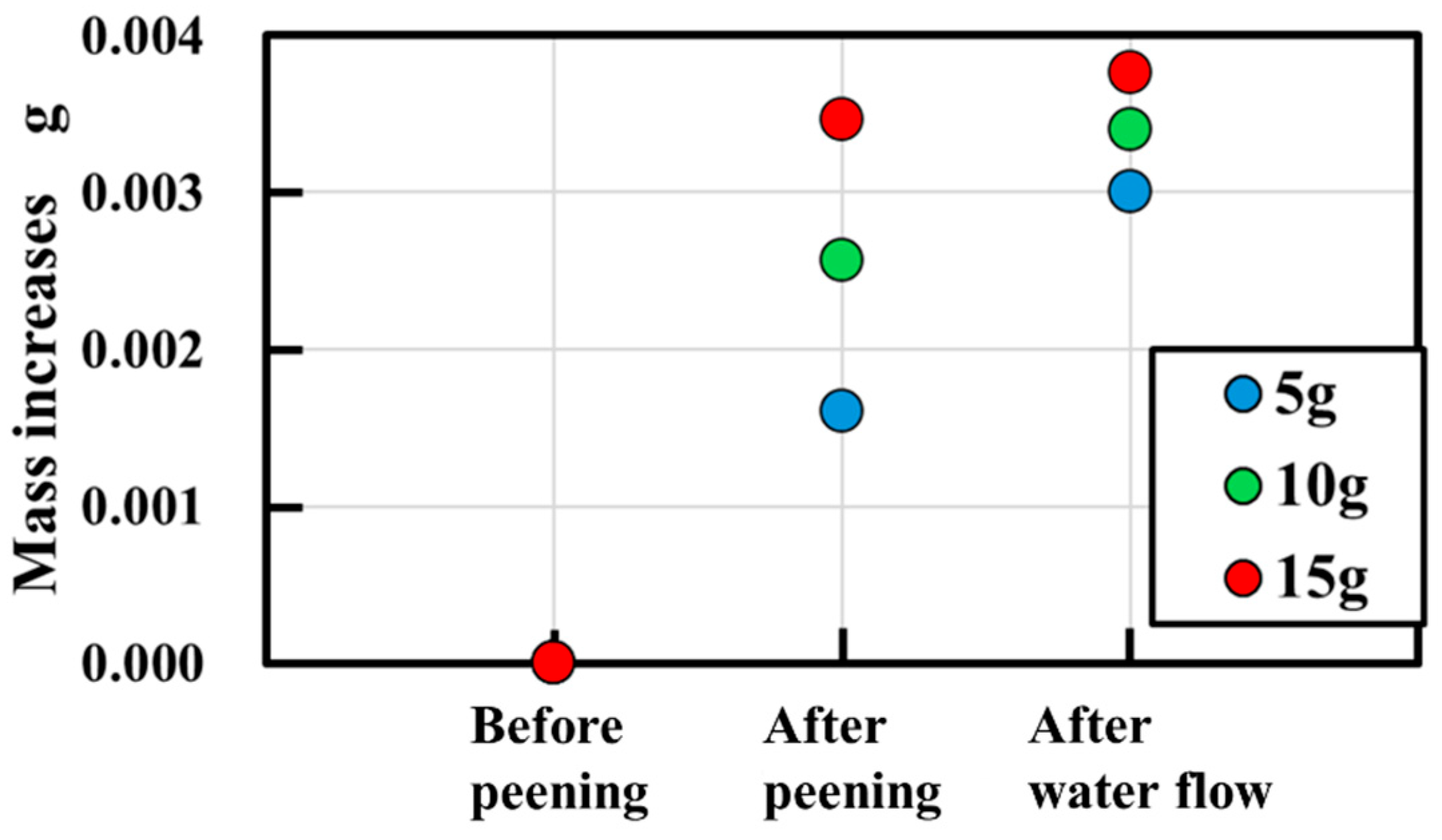
3.2. Effect of Total Peening Mass on Titanium Apatite Transfer
3.2.1. Experimental Conditions and Evaluation Methods
3.2.2. Antimicrobial Evaluation
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tuuliainen, E.; Nihtilä, A.; Nykänen, I.; Suominen, A.L.; Komulainen, K.; Tiihonen, M.; Hartikainen, S. The Association of Frailty with Oral Cleaning Habits and Oral Hygiene among Elderly Home Care Clients. Scand. J. Caring Sci. 2019, 34, 938–947. [Google Scholar] [CrossRef]
- Kanlı, A.; Demirel, F.; Sezgin, Y. Oral Candidosis, Denture Cleanliness and Hygiene Habits in an Elderly Population. Aging Clin. Exp. Res. 2005, 17, 502–507. [Google Scholar] [CrossRef]
- Çankaya, Z.T.; Yurdakoş, A.; Kalabay, P.G. The Association between Denture Care and Oral Hygiene Habits, Oral Hygiene Knowledge and Periodontal Status of Geriatric Patients Wearing Removable Partial Dentures. Eur. Oral Res. 2020, 54, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Moussa, A.R.; Dehis, W.M.; Elboraey, A.N.; Elgabry, H.S. A Comparative Clinical Study of the Effect of Denture Cleansing on the Surface Roughness and Hardness of Two Denture Base Materials. Open Access Maced. J. Med. Sci. 2016, 4, 476–481. [Google Scholar] [CrossRef]
- Rocha, M.M.; Carvalho, A.M.; Coimbra, F.C.T.; De Arruda, C.N.F.; Oliveira, V.D.C.; Macedo, A.P.; Silva-Lovato, C.H.; Pagnano, V.O.; Paranhos, H.D.F.O. Complete Denture Hygiene Solutions: Antibiofilm Activity and Effects on Physical and Mechanical Properties of Acrylic Resin. J. Appl. Oral Sci. 2021, 29, e20200948. [Google Scholar] [CrossRef]
- Müller, F. Oral Hygiene Reduces the Mortality from Aspiration Pneumonia in Frail Elders. J. Dent. Res. 2014, 94, 14S–16S. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, Y.; Yamazaki, Y.; Okada, K.; Kondo, M.; Matsushita, T.; Nakazawa, S. Oral Health for Achieving Longevity. Geriatr. Gerontol. Int. 2020, 20, 526–538. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, A.; Miyake, Y.; Yoneyama, T.; Miyatake, K.; Hirota, K. Professional Oral Health Care Reduces the Number of Oropharyngeal Bacteria. J. Dent. Res. 2008, 87, 594–598. [Google Scholar] [CrossRef]
- Senpuku, H.; Tsuha, Y.; Hanada, N.; Miyazaki, H.; Inoshita, E.; Sogame, A. Systemic Diseases in Association with Microbial Species in Oral Biofilm from Elderly Requiring Care. Gerontology 2003, 49, 301–309. [Google Scholar] [CrossRef]
- Salerno, C.; Esposito, V.; Guida, A.; Milillo, L.; Pascale, M.; Petruzzi, M.; Contaldo, M.; Busciolano, M.; Serpico, R. Candida-Associated Denture Stomatitis. Med. Oral 2011, 16, e139–e143. [Google Scholar] [CrossRef]
- Addy, M.; Bates, J.F. Plaque Accumulation Following the Wearing of Different Types of Removable Partial Dentures. J. Oral Rehabil. 1979, 6, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Baba, Y.; Sato, Y.; Owada, G.; Minakuchi, S. Effectiveness of a Combination Denture-Cleaning Method versus a Mechanical Method: Comparison of Denture Cleanliness, Patient Satisfaction, and Oral Health-Related Quality of Life. J. Prosthodont. Res. 2018, 62, 353–358. [Google Scholar] [CrossRef] [PubMed]
- Iosif, L.; Dimitriu, B.; Țâncu, A.M.C.; Amza, O.E.; Imre, M.; Pantea, M.; Ispas, A. Qualitative Assessment of the Removable Denture Microbiome. Germs 2024, 14, 28–37. [Google Scholar] [CrossRef]
- Hannah, V.E.; O’Donnell, L.; Robertson, D.; Ramage, G. Denture Stomatitis: Causes, Cures and Prevention. Prim. Dent. J. 2017, 6, 46–51. [Google Scholar] [CrossRef]
- Perić, M.; Miličić, B.; Živković, R.; Arsenijević, V.A.; Pfićer, J.K. A Systematic Review of Denture Stomatitis: Predisposing Factors, Clinical Features, Etiology, and Global Candida Spp. Distribution. JoF 2024, 10, 328. [Google Scholar] [CrossRef] [PubMed]
- Miyake, A.; Komasa, Y.; Hashimoto, Y.; Okazaki, J.; Komasa, S. Adsorption of Saliva Related Protein on Denture Materials: An X-Ray Photoelectron Spectroscopy and Quartz Crystal Microbalance Study. Adv. Mater. Sci. Eng. 2016, 2016, 5478326. [Google Scholar] [CrossRef]
- Komasa, S.; Tashiro, Y.; Miyake, A.; Matsumoto, T.; Wang, X.; Nakai, M.; Sato, H.; Hashimoto, Y. Development of a New Self-Cleaning Denture Base Material Using Titanium Apatite. Dent. Mater. J. 2025, 44, 365–374. [Google Scholar] [CrossRef]
- Sato, W.; Hasegawa, Y.; Yoshida, Y.; Okazaki, J.; Komasa, S. Antimicrobial Effect of Titanium Hydroxyapatite in Denture Base Resin. Appl. Sci. 2018, 8, 963. [Google Scholar] [CrossRef]
- Kulak-Ozkan, Y.; Arikan, A.; Kazazoglu, E. Oral Hygiene Habits, Denture Cleanliness, Presence of Yeasts and Stomatitis in Elderly People. J. Oral Rehabil. 2002, 29, 300–304. [Google Scholar] [CrossRef]
- Choo, A.; Delac, D.M.; Messer, L.B. Oral Hygiene Measures and Promotion: Review and Considerations. Aust. Dent. J. 2001, 46, 166–173. [Google Scholar] [CrossRef]
- Takahashi, Y.; Hamanaka, I.; Shimizu, H. Effect of Thermal Shock on Mechanical Properties of Injection-Molded Thermoplastic Denture Base Resins. Acta Odontol. Scand. 2011, 70, 297–302. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, H.; Kawaguchi, T.; Hamanaka, I.; Takahashi, Y. Effect of Long-Term Water Immersion or Thermal Shock on Mechanical Properties of High-Impact Acrylic Denture Base Resins. Dent. Mater. J. 2016, 35, 204–209. [Google Scholar] [CrossRef]
- Aldegheishem, A.; Aldeeb, M.; Al-Ahdal, K.; Helmi, M.; Alsagob, E.I. Influence of Reinforcing Agents on the Mechanical Properties of Denture Base Resin: A Systematic Review. Polymers 2021, 13, 3083. [Google Scholar] [CrossRef]
- Polychronakis, N.C.; Polyzois, G.L.; Lagouvardos, P.E.; Papadopoulos, T.D. Effects of Cleansing Methods on 3-D Surface Roughness, Gloss and Color of a Polyamide Denture Base Material. Acta Odontol. Scand. 2014, 73, 353–363. [Google Scholar] [CrossRef]
- Khan, A.A.; Fareed, M.A.; Alshehri, A.H.; Aldegheishem, A.; Alharthi, R.; Saadaldin, S.A.; Zafar, M.S. Mechanical Properties of the Modified Denture Base Materials and Polymerization Methods: A Systematic Review. IJMS 2022, 23, 5737. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, H.; Suenaga, H.; Suzuki, O.; Takahashi, N.; Takahashi, M.; Sasaki, K. Deterioration of Polymethyl Methacrylate Dentures in the Oral Cavity. Dent. Mater. J. 2015, 34, 234–239. [Google Scholar] [CrossRef]
- Vallittu, P.K. A Review of Methods Used to Reinforce Polymethyl Methacrylate Resin. J. Prosthodont. 1995, 4, 183–187. [Google Scholar] [CrossRef] [PubMed]
- Heimer, S.; Stawarczyk, B.; Schmidlin, P.R. Discoloration of PMMA, Composite, and PEEK. Clin. Oral Investig. 2016, 21, 1191–1200. [Google Scholar] [CrossRef]
- Arai, T.; Sugiyama, T.; Ueda, T.; Sakurai, K. Inhibiting Microbial Adhesion to Denture Base Acrylic Resin by Titanium Dioxide Coating. J. Oral Rehabil. 2009, 36, 902–908. [Google Scholar] [CrossRef]
- Bajunaid, S.O. How Effective Are Antimicrobial Agents on Preventing the Adhesion of Candida Albicans to Denture Base Acrylic Resin Materials? A Systematic Review. Polymers 2022, 14, 908. [Google Scholar] [CrossRef]
- Mohammed, N.B.; Daily, Z.A.; Alsharbaty, M.H.; Abullais, S.S.; Arora, S.; Lafta, H.A.; Jalil, A.T.; Almulla, A.F.; Ramírez-Coronel, A.A.; Aravindhan, S.; et al. Effect of PMMA Sealing Treatment on the Corrosion Behavior of Plasma Electrolytic Oxidized Titanium Dental Implants in Fluoride-Containing Saliva Solution. Mater. Res. Express 2022, 9, 125401. [Google Scholar] [CrossRef]
- Yamada, M.; Fukumoto, M.; Nakano, H.; Isago, H. Cold Spraying of TiO2 Photocatalyst Coating with Nitrogen Process Gas. J. Therm. Spray Technol. 2010, 19, 1218–1223. [Google Scholar] [CrossRef]
- Darwish, G.; Barão, V.A.; Wu, C.D.; Knoernschild, K.; Huang, S.; Taukodis, C.G.; Bishal, A.K.; Sukotjo, C.; Yang, B.; Campbell, S. Improving Polymethyl Methacrylate Resin Using a Novel Titanium Dioxide Coating. J. Prosthodont. 2019, 28, 1011–1017. [Google Scholar] [CrossRef] [PubMed]
- Nobre, C.M.G.; Hannig, M.; Pütz, N. Adhesion of Hydroxyapatite Nanoparticles to Dental Materials under Oral Conditions. Scanning 2020, 2020, 6065739. [Google Scholar] [CrossRef] [PubMed]
- Carbaugh, D.J.; Parthiban, R.; Rahman, F.; Wright, J.T. Photolithography with Polymethyl Methacrylate (PMMA). Semicond. Sci. Technol. 2015, 31, 025010. [Google Scholar] [CrossRef]
- Li, Z.; Qi, Q.; Sun, J.; Lan, J. Effect of a Denture Base Acrylic Resin Containing Silver Nanoparticles on Candida Albicans Adhesion and Biofilm Formation. Gerodontology 2014, 33, 209–216. [Google Scholar] [CrossRef]
- Sivakumar, I.; Rao, B.; Kamaraj, B.; Sajjan, S.; Arunachalam, K.S.; Ramaraju, A.V. Incorporation of Antimicrobial Macromolecules in Acrylic Denture Base Resins: A Research Composition and Update. J. Prosthodont. 2013, 23, 284–290. [Google Scholar] [CrossRef]
- Kamonwannasit, S.; Phatai, P.; Butburee, T.; Karaphun, A.; Khemthong, P.; Futalan, C.M. Synthesis of Copper-Silver Doped Hydroxyapatite via Ultrasonic Coupled Sol-Gel Techniques: Structural and Antibacterial Studies. J. Sol-Gel Sci. Technol. 2020, 96, 452–463. [Google Scholar] [CrossRef]
- Maleki-Ghaleh, H.; Omidi, Y.; Beygi-Khosrowshahi, Y.; Kavanlouei, M.; Moradpur-Tari, E.; Adibkia, K.; Khademi-Azandehi, P.; Koc, B.; Barar, J.; Kumar, A.P.; et al. Antibacterial and Cellular Behaviors of Novel Zinc-Doped Hydroxyapatite/Graphene Nanocomposite for Bone Tissue Engineering. Int. J. Mol. Sci. 2021, 22, 9564. [Google Scholar] [CrossRef]
- Fatimah, I.; Nugroho, B.H.; Hidayat, H.; Purwiandono, G.; Yahya, A.; Ibrahim, S.; Sagadevan, S.; Citradewi, P.W.; Mohd Ghazali, S.A.I.S. Biosynthesized Gold Nanoparticles-Doped Hydroxyapatite as Antibacterial and Antioxidant Nanocomposite. Mater. Res. Express 2021, 8, 115003. [Google Scholar] [CrossRef]


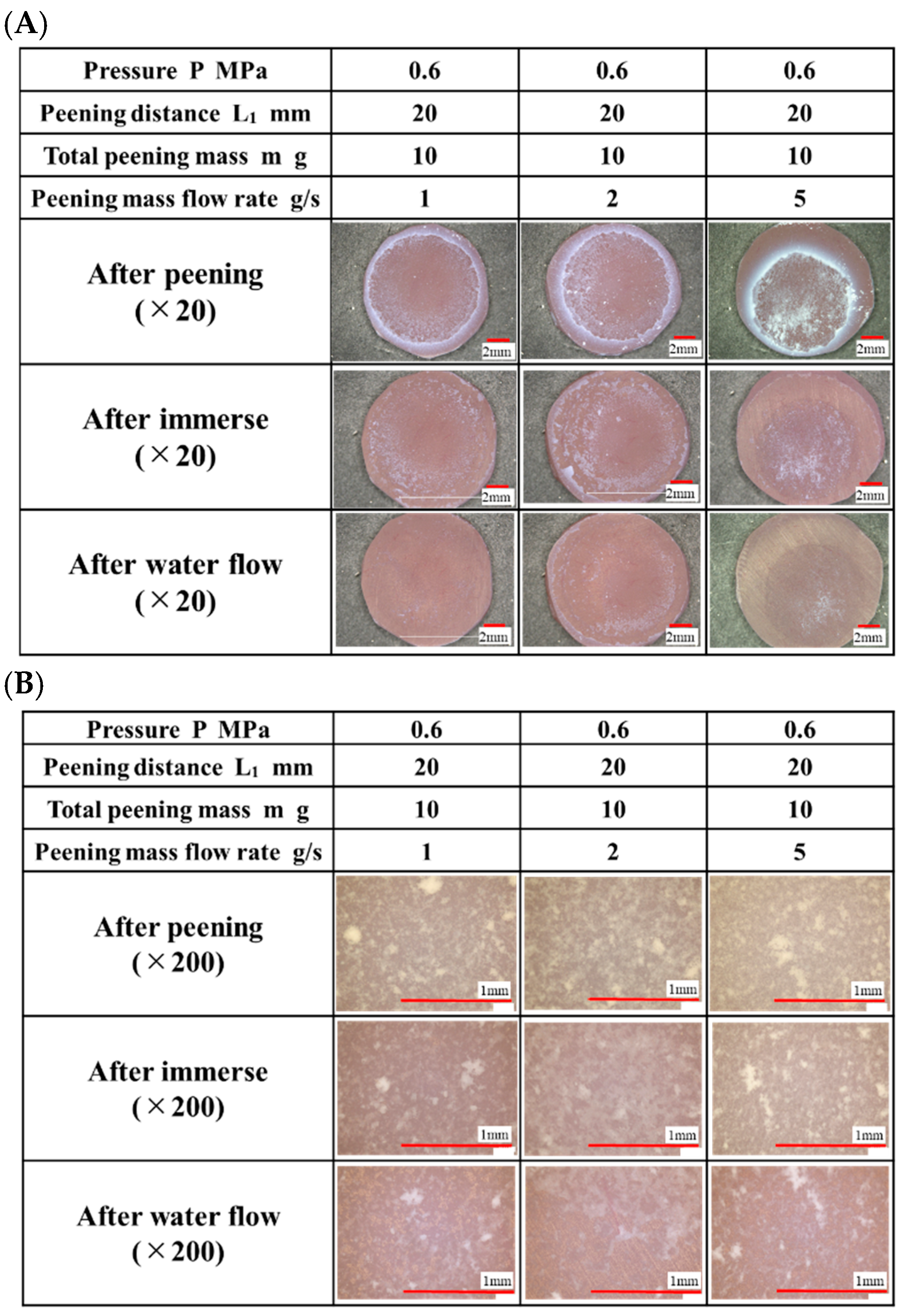


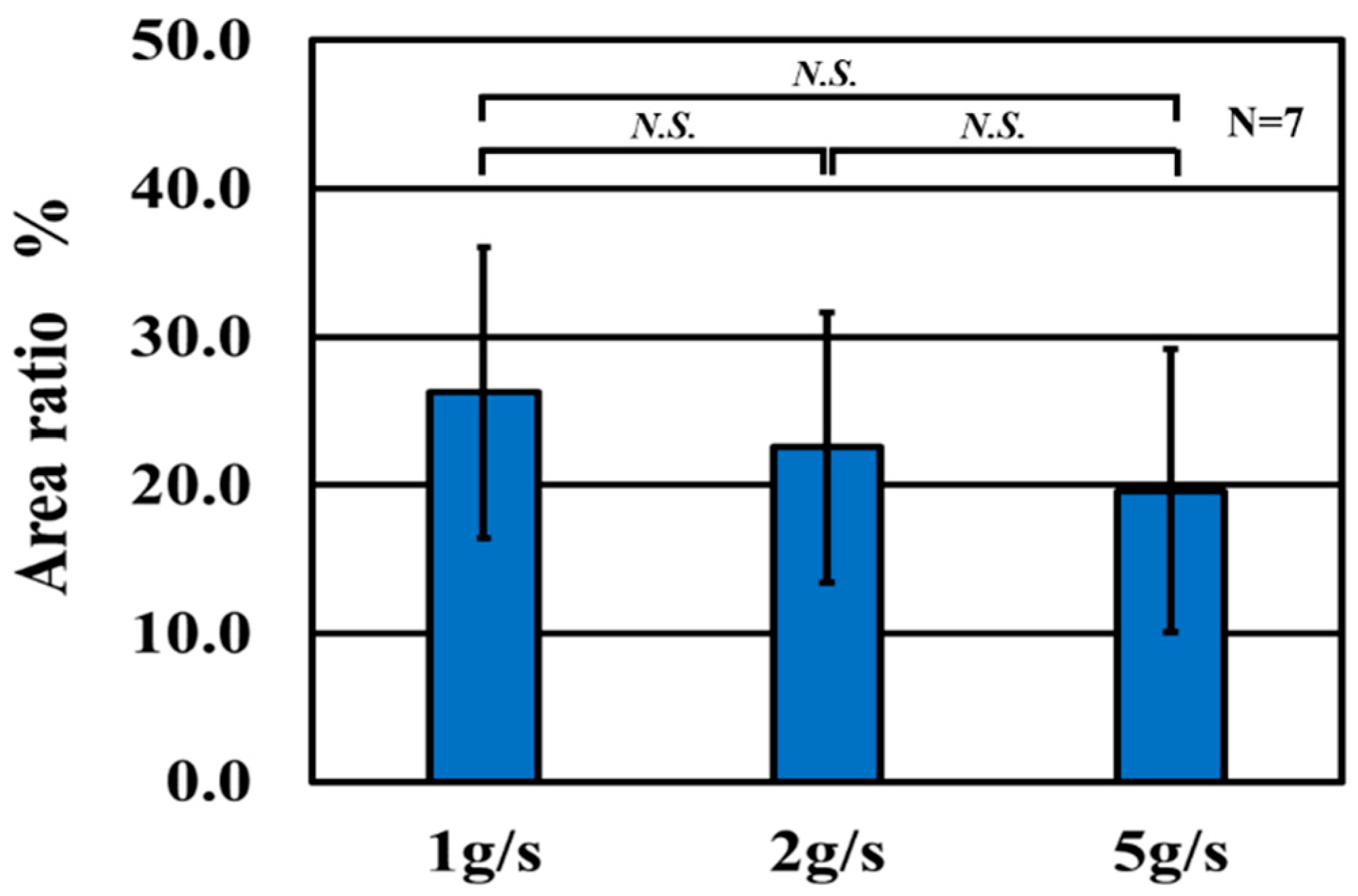

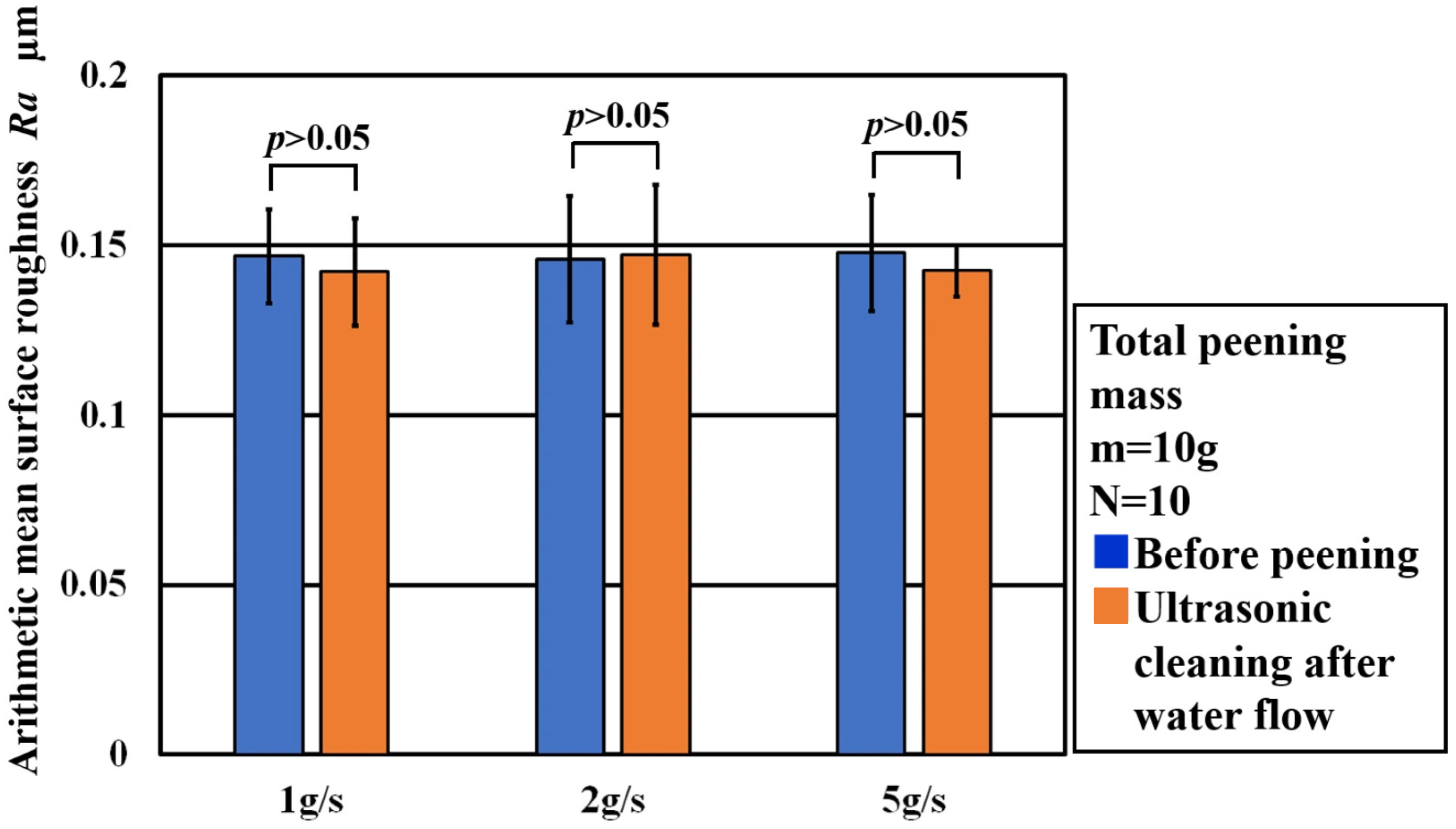

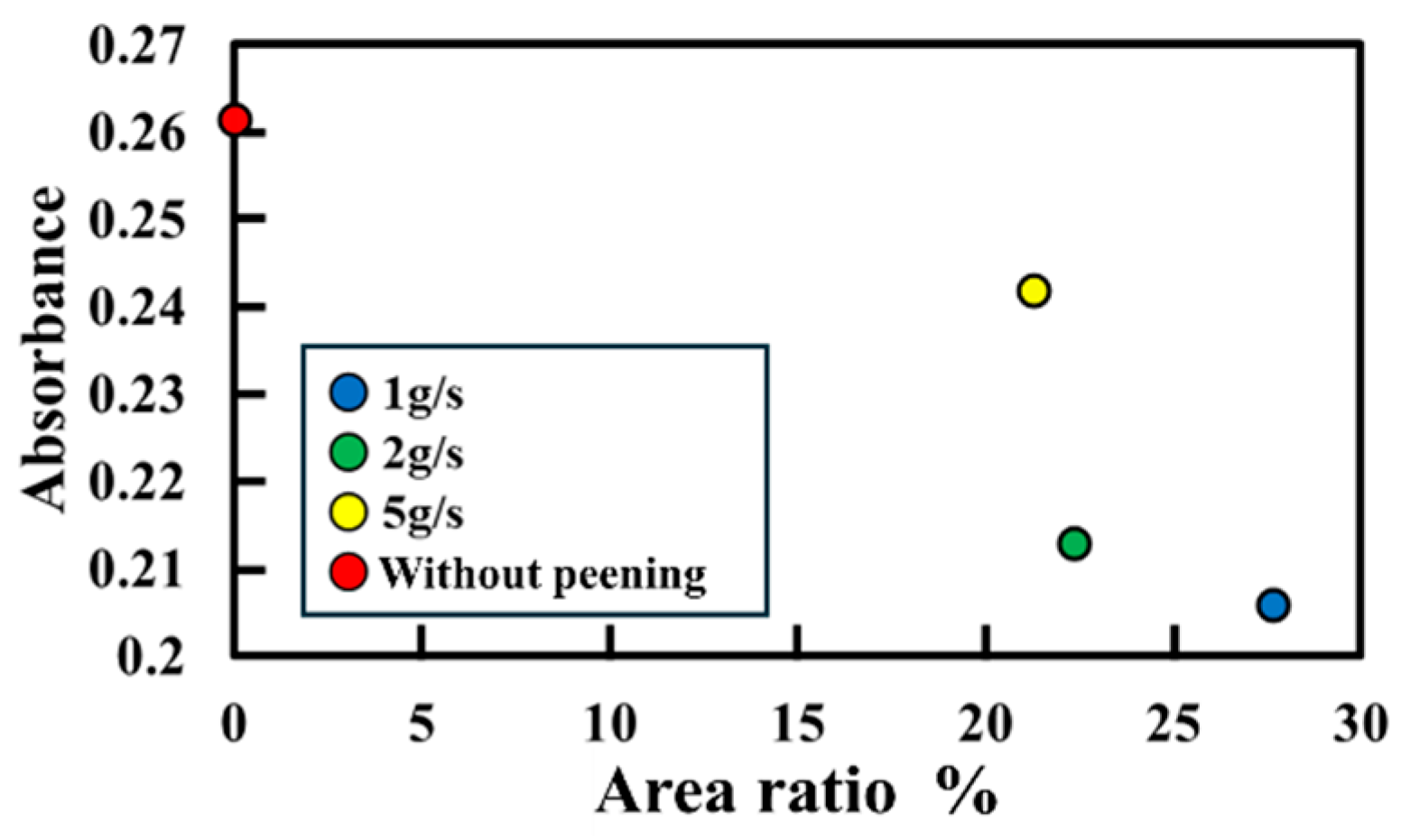





| Pressure P MPa | 0.6 |
| Peening distance L1 mm | 20 |
| Total peening mass m g | 10 |
| Peening mass flow rate g/s | 1, 2, 5 |
| Number of peening n times | 1 |
| Distance Between the Tap and the Test Specimen L2 mm | 200 |
| Angle θ° | 90 |
| Flow rate Q m3/s | 3.0 × 10−5 |
| Water flow time min | 40 |
| Pressure P MPa | 0.6 |
| Peening distance L1 mm | 20 |
| Total peening mass m g | 5, 10, 15 |
| Peening mass flow rate g/s | 1 |
| Number of peening n times | 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Sato, H.; Miyake, A.; Harakawa, N.; Shoji, I.; Kameyama, Y.; Kodama, S.; Tashiro, Y.; Ogata, C.; Komasa, S. Development of Antibacterial Dentures Using Titanium Apatite Peening. Bioengineering 2026, 13, 230. https://doi.org/10.3390/bioengineering13020230
Sato H, Miyake A, Harakawa N, Shoji I, Kameyama Y, Kodama S, Tashiro Y, Ogata C, Komasa S. Development of Antibacterial Dentures Using Titanium Apatite Peening. Bioengineering. 2026; 13(2):230. https://doi.org/10.3390/bioengineering13020230
Chicago/Turabian StyleSato, Hideaki, Akiko Miyake, Nichika Harakawa, Issei Shoji, Yutaka Kameyama, Shuhei Kodama, Yuichiro Tashiro, Chizuko Ogata, and Satoshi Komasa. 2026. "Development of Antibacterial Dentures Using Titanium Apatite Peening" Bioengineering 13, no. 2: 230. https://doi.org/10.3390/bioengineering13020230
APA StyleSato, H., Miyake, A., Harakawa, N., Shoji, I., Kameyama, Y., Kodama, S., Tashiro, Y., Ogata, C., & Komasa, S. (2026). Development of Antibacterial Dentures Using Titanium Apatite Peening. Bioengineering, 13(2), 230. https://doi.org/10.3390/bioengineering13020230







