Atypical Resting-State and Task-Evoked EEG Signatures in Children with Developmental Language Disorder
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
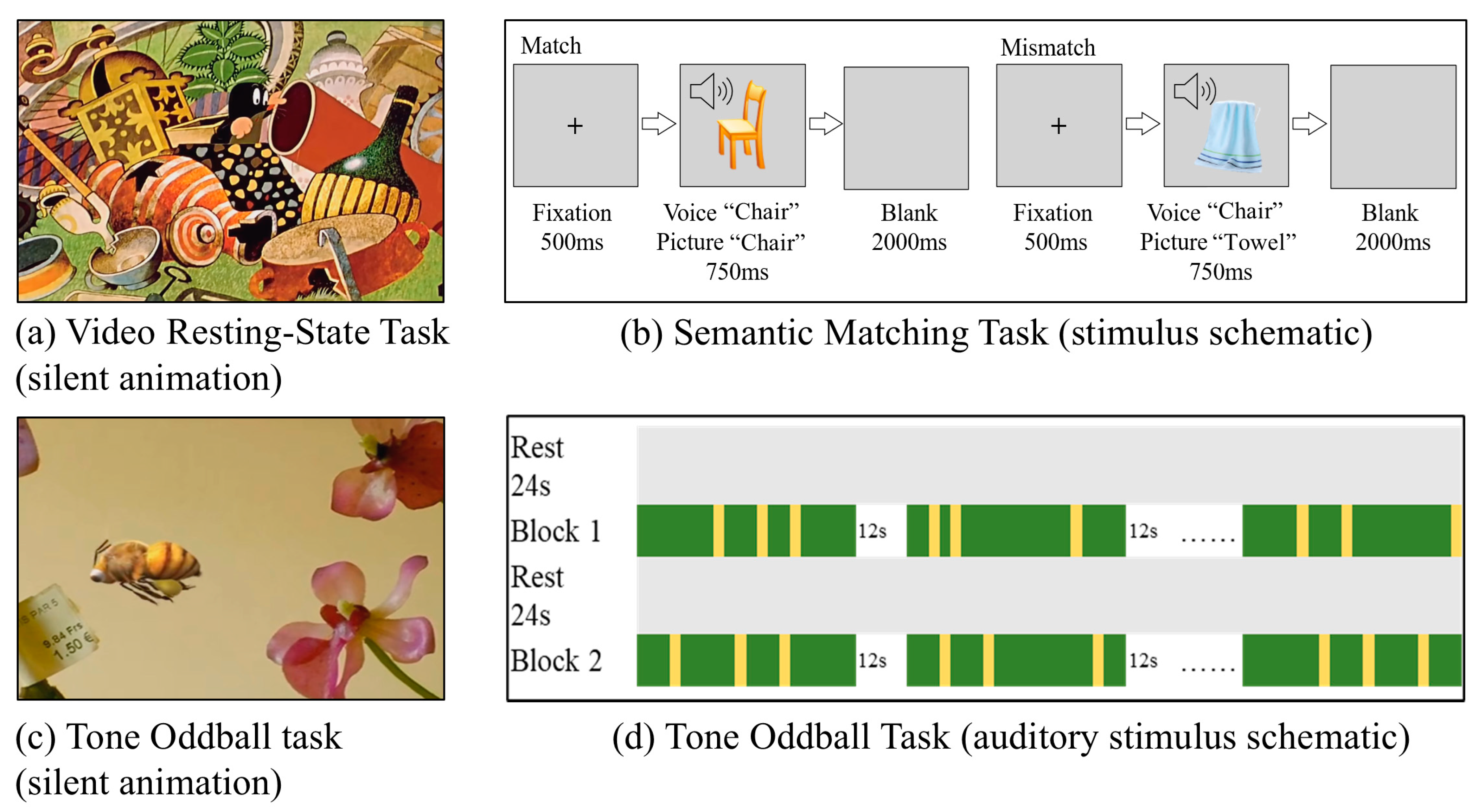
2.2. Experimental Procedure
2.3. EEG Data Acquisition and Preprocessing
2.4. Data Analysis
2.4.1. Resting-State Analysis
2.4.2. Semantic Matching Task Analysis
2.4.3. Tone Oddball Task Analysis
2.4.4. Machine Learning Classification
3. Results
3.1. Resting-State Neurodynamic Reveal Atypical Brain Network Organization in DLD
3.1.1. Frequency-Specific Functional Connectivity Alterations
3.1.2. Altered Microstate Dynamics
3.2. Task-Evoked Neural Responses Reveal Deficits in Language and Auditory Processing
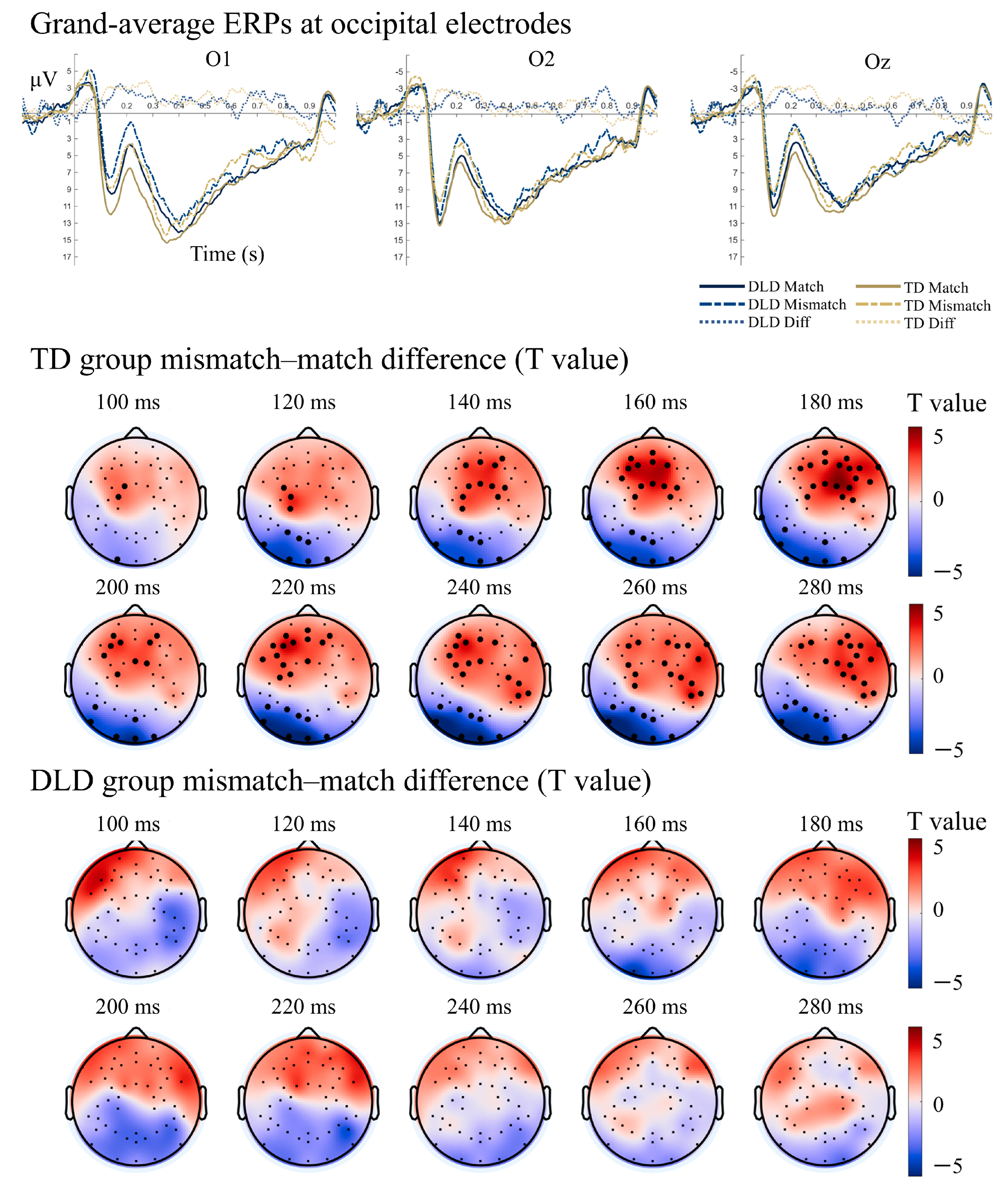
3.2.1. Semantic Matching Task: Attenuated ERP Components
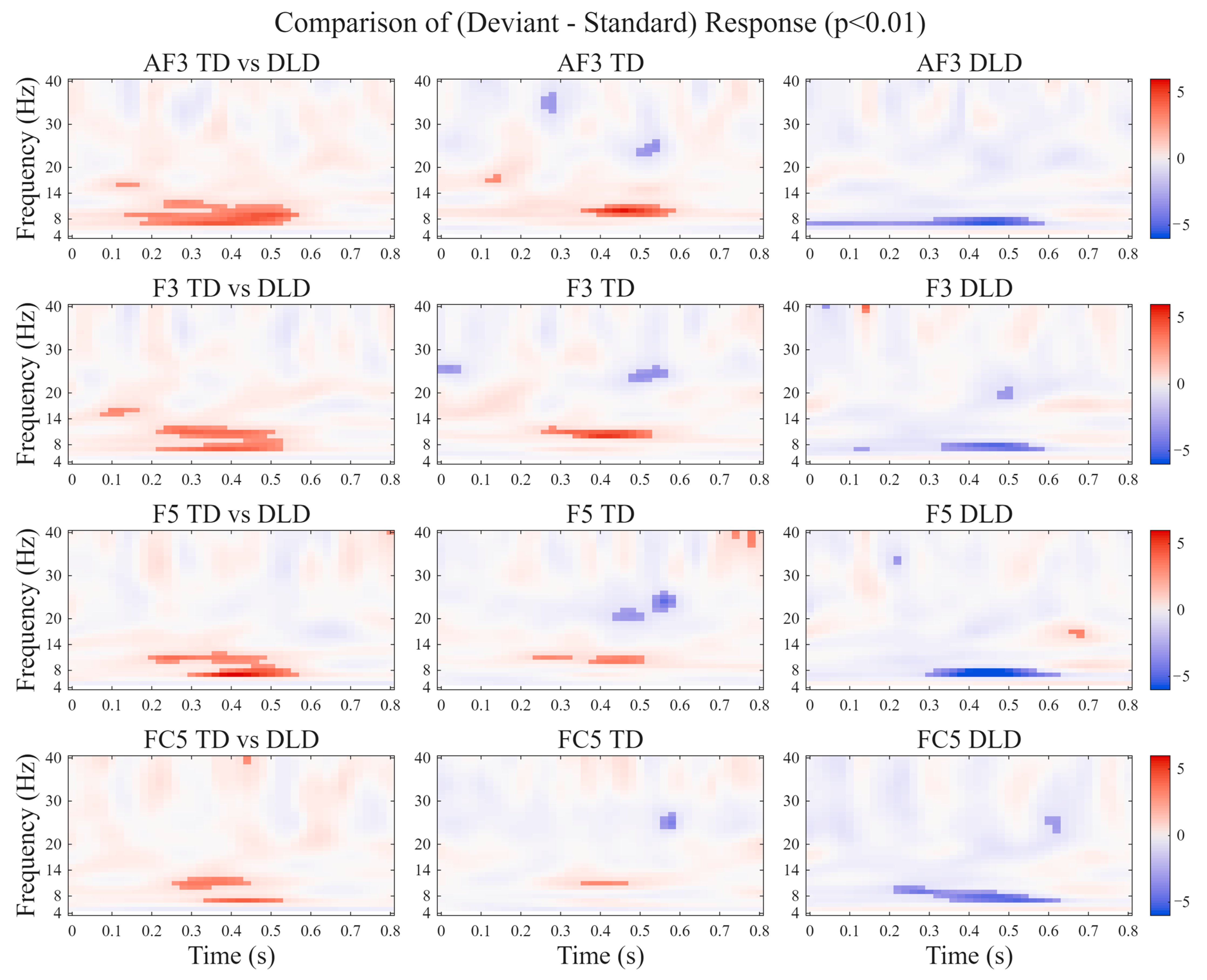
3.2.2. Tone Oddball Task: Impaired Oscillatory Power Modulation
3.3. Classification Accuracy Using a Machine Learning Model
4. Discussion
4.1. Atypical Intrinsic Network Architecture in DLD
4.2. Propagation to Task-Based Processing Deficits
4.3. Feasibility and Challenges of a Multi-Level Classifier
4.4. Implications for Neural Models of Language
4.5. Limitations and Strengths
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bishop, D.V.; Snowling, M.J.; Thompson, P.A.; Greenhalgh, T.; CATALISE-2 consortium. Phase 2 of CATALISE: A multinational and multidisciplinary delphi consensus study of problems with language development: Terminology. J. Child Psychol. Psychiatry 2017, 58, 1068–1080. [Google Scholar] [CrossRef]
- Norbury, C.F.; Gooch, D.; Wray, C.; Baird, G.; Charman, T.; Simonoff, E.; Vamvakas, G.; Pickles, A. The impact of nonverbal ability on prevalence and clinical presentation of language disorder: Evidence from a population study. J. Child Psychol. Psychiatry 2016, 57, 1247–1257. [Google Scholar] [CrossRef] [PubMed]
- Michelini, G.; Carlisi, C.O.; Eaton, N.R.; Elison, J.T.; Haltigan, J.D.; Kotov, R.; Krueger, R.F.; Latzman, R.D.; Li, J.J.; Levin-Aspenson, H.F.; et al. Where do neurodevelopmental conditions fit in transdiagnostic psychiatric frameworks? Incorporating a new neurodevelopmental spectrum. World Psychiatry 2024, 23, 333–357. [Google Scholar] [CrossRef] [PubMed]
- Camas, R.A.F.; Leon-Rojas, J.E. Specific Language Impairment and Executive Functions in School-Age Children: A Systematic Review. Cureus 2023, 15, e43163. [Google Scholar] [CrossRef] [PubMed]
- Chinn, L.K.; Zhukova, M.A.; Kroeger, R.J.; Ledesma, L.M.; Cavitt, J.E.; Grigorenko, E.L. Auditory brainstem response deficits in learning disorders and developmental language disorder: A systematic review and meta-analysis. Sci. Rep. 2022, 12, 20124. [Google Scholar] [CrossRef]
- Murphy, E. ROSE: A universal neural grammar. Cogn. Neurosci. 2025, 16, 49–80. [Google Scholar] [CrossRef]
- Fedorenko, E.; Ivanova, A.A.; Regev, T.I. The language network as a natural kind within the broader landscape of the human brain. Nat. Rev. Neurosci. 2024, 25, 289–312. [Google Scholar] [CrossRef]
- Matchin, W.; Hickok, G. The cortical organization of syntax. Cereb. Cortex 2019, 30, 1481–1498. [Google Scholar] [CrossRef]
- Campos, A.; Loyola-Navarro, R.; González, C.; Iverson, P. Resting-state electroencephalogram and speech perception in young children with developmental language disorder. Brain Sci. 2025, 15, 219. [Google Scholar] [CrossRef]
- Stanojević, N.; Fatić, S.; Jeličić, L.; Nenadović, V.; Stokić, M.; Bilibajkić, R.; Subotić, M.; Matić, T.B.; Konstantinović, L.; Ćirović, D. Resting-state EEG alpha rhythm spectral power in children with specific language impairment: A cross-sectional study. J. Appl. Biomed. 2023, 21, 113–120. [Google Scholar] [CrossRef]
- Angulo-Ruiz, B.Y.; Rodríguez-Martínez, E.I.; Ruiz-Martínez, F.J.; Gómez-Treviño, A.; Muñoz, V.; Crespo, S.A.; Gómez, C.M. Brain Complexity and Parametrization of Power Spectral Density in Children with Specific Language Impairment. Entropy 2025, 27, 572. [Google Scholar] [CrossRef] [PubMed]
- O’Reilly, C.; Lewis, J.D.; Elsabbagh, M. Is functional brain connectivity atypical in autism? A systematic review of EEG and MEG studies. PLoS ONE 2017, 12, e0175870. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Yang, Y.; Liu, J.; Ren, H.; Bi, H. Functional connectivity alterations in developmental dyslexia: A meta-analysis of task-based and resting-state fMRI studies. Dev. Sci. 2025, 29, e70093. [Google Scholar] [CrossRef] [PubMed]
- Custo, A.; Van De Ville, D.; Wells, W.M.; Tomescu, M.I.; Brunet, D.; Michel, C.M. Electroencephalographic resting-state networks: Source localization of microstates. Brain Connect 2017, 7, 671–682. [Google Scholar] [CrossRef] [PubMed]
- Haydock, D.; Kadir, S.; Leech, R.; Nehaniv, C.L.; Antonova, E. EEG microstate syntax analysis: A review of methodological challenges and advances. NeuroImage 2025, 309, 121090. [Google Scholar] [CrossRef]
- Das, S.; Zomorrodi, R.; Enticott, P.G.; Kirkovski, M.; Blumberger, D.M.; Rajji, T.K.; Desarkar, P. Resting state electroencephalography microstates in autism spectrum disorder: A mini-review. Front. Psychiatry 2022, 13, 988939. [Google Scholar] [CrossRef]
- Kujala, T.; Leminen, M. Low-level neural auditory discrimination dysfunctions in specific language impairment—A review on mismatch negativity findings. Dev. Cogn. Neurosci. 2017, 28, 65–75. [Google Scholar] [CrossRef]
- Campos, A.; Tuomainen, J.; Tuomainen, O. Mismatch responses to speech contrasts in preschoolers with and without developmental language disorder. Brain Sci. 2023, 14, 42. [Google Scholar] [CrossRef]
- Kornilov, S.A.; Landi, N.; Rakhlin, N.; Fang, S.-Y.; Grigorenko, E.L.; Magnuson, J.S. Attentional but not pre-attentive neural measures of auditory discrimination are atypical in children with developmental language disorder. Dev. Neuropsychol. 2014, 39, 543–567. [Google Scholar] [CrossRef]
- Kornilov, S.A.; Magnuson, J.S.; Rakhlin, N.; Landi, N.; Grigorenko, E.L. Lexical processing deficits in children with developmental language disorder: An event-related potentials study. Dev. Psychopathol. 2015, 27, 459–476. [Google Scholar] [CrossRef]
- Pijnacker, J.; Davids, N.; van Weerdenburg, M.; Verhoeven, L.; Knoors, H.; van Alphen, P. Semantic processing of sentences in preschoolers with specific language impairment: Evidence from the N400 effect. J. Speech Lang. Hear. Res. 2017, 60, 627–639. [Google Scholar] [CrossRef] [PubMed]
- Wechsler, D. Wechsler Intelligence Scale for Children–Fourth Edition (WISC-IV) Manual, 4th ed.; The Psychological Corporation: San Antonio, TX, USA, 2003. [Google Scholar]
- Sun, X. Hearing Impairment Evaluation Criteria and Methods of Auditory Language Ability; The Video Library Sunchime Audiovisual Press: Beijing, China, 2009. [Google Scholar]
- Jin, C.; Li, R.-L.; Zhang, L.-L.; Zhang, Y.; Li, N.; Wang, J.-H.; Wang, X.-Y.; Liang, W.-L.; Zhang, Y.-X.; Wei, Z.; et al. The revision and according validity research of China developmental scale for China. Chin. J. Child Health Care 2014, 22, 1242–1246. [Google Scholar] [CrossRef]
- Fox, N.A.; Pérez-Edgar, K.; Morales, S.; Brito, N.H.; Campbell, A.M.; Cavanagh, J.F.; Gabard-Durnam, L.J.; Hudac, C.M.; Key, A.P.; Larson-Prior, L.J.; et al. The development and structure of the healthy brain and child development (HBCD) study EEG protocol. Dev. Cogn. Neurosci. 2024, 69, 101447. [Google Scholar] [CrossRef] [PubMed]
- Delorme, A.; Makeig, S. EEGLAB: An open source toolbox for analysis of single-trial EEG dynamics including independent component analysis. J. Neurosci. Methods 2004, 134, 9–21. [Google Scholar] [CrossRef]
- Bailey, N.; Biabani, M.; Hill, A.; Miljevic, A.; Rogasch, N.; McQueen, B.; Murphy, O.; Fitzgerald, P. Introducing RELAX: An automated pre-processing pipeline for cleaning EEG data—Part 1: Algorithm and application to oscillations. Clin. Neurophysiol. 2023, 149, 178–201. [Google Scholar] [CrossRef]
- Hill, A.T.; Enticott, P.G.; Fitzgerald, P.B.; Bailey, N.W. RELAX-jr: An automated pre-processing pipeline for developmental EEG recordings. Hum. Brain Mapp. 2024, 45, e70034. [Google Scholar] [CrossRef]
- Kalburgi, S.N.; Kleinert, T.; Aryan, D.; Nash, K.; Schiller, B.; Koenig, T. MICROSTATELAB: The EEGLAB toolbox for resting-state microstate analysis. Brain Topogr. 2024, 37, 621–645. [Google Scholar] [CrossRef]
- Oostenveld, R.; Fries, P.; Maris, E.; Schoffelen, J.-M. FieldTrip: Open source software for advanced analysis of MEG, EEG, and invasive electrophysiological data. Comput. Intell. Neurosci. 2011, 2011, 156869. [Google Scholar] [CrossRef]
- Gao, Y.; Linke, A.; Keehn, R.J.J.; Punyamurthula, S.; Jahedi, A.; Gates, K.; Fishman, I.; Müller, R. The language network in autism: Atypical functional connectivity with default mode and visual regions. Autism Res. Off. J. Int. Soc. Autism Res. 2019, 12, 1344–1355. [Google Scholar] [CrossRef]
- Rubia, K. Cognitive neuroscience of attention deficit hyperactivity disorder (ADHD) and its clinical translation. Front. Hum. Neurosci. 2018, 12, 100. [Google Scholar] [CrossRef]
- Buzsáki, G.; Watson, B.O. Brain rhythms and neural syntax: Implications for efficient coding of cognitive content and neuropsychiatric disease. Dialogues Clin. Neurosci. 2012, 14, 345–367. [Google Scholar] [CrossRef]
- Xue, H.; Wang, Z.; Tan, Y.; Yang, H.; Fu, W.; Xue, L.; Zhao, J. Resting-state EEG reveals global network deficiency in dyslexic children. Neuropsychologia 2020, 138, 107343. [Google Scholar] [CrossRef]
- D’cRoz-Baron, D.F.; Baker, M.; Michel, C.M.; Karp, T. EEG microstates analysis in young adults with autism spectrum disorder during resting-state. Front. Hum. Neurosci. 2019, 13, 173. [Google Scholar] [CrossRef]
- Michel, C.M.; Koenig, T. EEG microstates as a tool for studying the temporal dynamics of whole-brain neuronal networks: A review. Neuroimage 2018, 180, 577–593. [Google Scholar] [CrossRef] [PubMed]
- Haebig, E.; Leonard, L.; Usler, E.; Deevy, P.; Weber, C. An initial investigation of the neural correlates of word processing in preschoolers with specific language impairment. J. Speech Lang. Hear. Res. 2018, 61, 729–739. [Google Scholar] [CrossRef] [PubMed]
- Montgomery, J.W. Sentence comprehension in children with specific language impairment: The role of phonological working memory. J. Speech Hear. Res. 1995, 38, 187–199. [Google Scholar] [CrossRef] [PubMed]
- Brouwer, H.; Hoeks, J.C.J. A time and place for language comprehension: Mapping the N400 and the P600 to a minimal cortical network. Front. Hum. Neurosci. 2013, 7, 758. [Google Scholar] [CrossRef]
- Bishop, D.V.M. Using mismatch negativity to study central auditory processing in developmental language and literacy impairments: Where are we, and where should we be going? Psychol. Bull. 2007, 133, 651–672. [Google Scholar] [CrossRef]
- Klimesch, W. Alpha-band oscillations, attention, and controlled access to stored information. Trends Cogn. Sci. 2012, 16, 606–617. [Google Scholar] [CrossRef]
- ter Wal, M.; Linde-Domingo, J.; Lifanov, J.; Roux, F.; Kolibius, L.D.; Gollwitzer, S.; Lang, J.; Hamer, H.; Rollings, D.; Sawlani, V.; et al. Theta rhythmicity governs human behavior and hippocampal signals during memory-dependent tasks. Nat. Commun. 2021, 12, 7048. [Google Scholar] [CrossRef]
- Liang, A.; Cui, Z.; Ding, J.; Lu, B.; Qu, C.; Li, S.; Yin, M.; Ning, X.; Fang, J. Classification of children with developmental language disorder using task fNIRS data and convolutional neural network. IEEE J. Sel. Top. Quantum Electron. 2025, 31, 7700109. [Google Scholar] [CrossRef]
- Usman, M.; Lin, W.-Y.; Lin, Y.-Y.; Hsieh, S.-T.; Chen, Y.-T.; Lo, Y.-C.; Lin, C.-L. Classifying developmental delays with EEG: A comparative study of machine learning and deep learning approaches. Biocybern. Biomed. Eng. 2025, 45, 229–246. [Google Scholar] [CrossRef]
- Varoquaux, G. Cross-validation failure: Small sample sizes lead to large error bars. Neuroimage 2018, 180, 68–77. [Google Scholar] [CrossRef]




| Band | Channel A | Channel B | T | p |
|---|---|---|---|---|
| delta | O1 | P8 | −3.92 | 0.0001 |
| (1–3 Hz) | AFz | P4 | −3.28 | 0.0008 |
| AF4 | P3 | −3.22 | 0.0010 | |
| Oz | P10 | −3.07 | 0.0007 | |
| Oz | P8 | −2.63 | 0.0003 | |
| theta | CP5 | F8 | 4.57 | 0.0002 |
| (4–7 Hz) | P3 | F8 | 4.33 | 0.0003 |
| CP5 | FT8 | 4.27 | 0.0001 | |
| P1 | FT8 | 4.15 | 0.0002 | |
| CP2 | CP6 | 4.02 | 0.0003 | |
| P3 | FT8 | 3.85 | 0.0001 | |
| TP7 | F8 | 3.49 | 0.0007 | |
| TP7 | FT8 | 3.48 | 0.0003 | |
| FC1 | TP10 | 3.30 | 0.0008 | |
| FCz | TP10 | 3.15 | 0.0009 | |
| C5 | FT8 | 3.10 | 0.0007 | |
| T9 | T10 | −2.97 | 0.0004 | |
| alpha | F9 | P4 | 5.48 | 0.0001 |
| (7–10 Hz) | FC1 | Pz | 5.02 | 0.0001 |
| P4 | E64 | 4.48 | 0.0003 | |
| P10 | FC4 | 4.46 | 0.0002 | |
| F9 | P2 | 4.45 | 0.0002 | |
| TP7 | FC4 | 4.27 | 0.0003 | |
| P10 | C2 | 4.16 | 0.0002 | |
| F7 | P4 | 4.16 | 0.0001 | |
| P5 | T10 | 3.91 | 0.0002 | |
| Fp2 | T9 | 3.75 | 0.0002 | |
| O2 | FC4 | 3.75 | 0.0003 | |
| O2 | C2 | 3.72 | 0.0009 | |
| P8 | FC4 | 3.64 | 0.0004 | |
| F9 | P6 | 3.60 | 0.0007 | |
| FCz | Pz | 3.60 | 0.0010 | |
| T7 | FC2 | 3.58 | 0.0010 | |
| P3 | T10 | 3.58 | 0.0010 | |
| FC1 | POz | 3.54 | 0.0010 | |
| T9 | FC4 | 3.48 | 0.0009 | |
| Pz | FC2 | 3.47 | 0.0008 | |
| C5 | T8 | 3.46 | 0.0010 | |
| C5 | FT8 | 3.44 | 0.0009 | |
| Fp2 | FC6 | 3.35 | 0.0010 | |
| beta | TP9 | P9 | 3.58 | 0.0002 |
| (11–20 Hz) | ||||
| gamma | C5 | TP7 | 4.66 | 0.0001 |
| (21–40 Hz) | FT7 | P8 | 4.01 | 0.0004 |
| FC6 | F4 | −2.45 | 0.0007 | |
| Fz | Fp1 | −2.99 | 0.0003 |
| Classifier | Training Set | Test Set | ||||||
|---|---|---|---|---|---|---|---|---|
| Accuracy | Weighted Precision | Weighted Recall | Weighted Recall | Accuracy | Weighted Precision | Weighted Recall | Weighted Recall | |
| LDA | 88.46% | 88.83% | 88.46% | 88.51% | 80.00% | 80.00% | 80.00% | 80.00% |
| SVM | 88.46% | 88.56% | 88.46% | 88.37% | 80.00% | 80.00% | 80.00% | 80.00% |
| KNN | 76.92% | 80.65% | 76.92% | 76.92% | 70.00% | 69.52% | 70.00% | 69.01% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Liang, A.; Cui, Z.; Shi, Y.; Qu, C.; Wei, Z.; Wang, H.; Zhang, X.; Ning, X.; Ni, X.; Fang, J. Atypical Resting-State and Task-Evoked EEG Signatures in Children with Developmental Language Disorder. Bioengineering 2026, 13, 119. https://doi.org/10.3390/bioengineering13010119
Liang A, Cui Z, Shi Y, Qu C, Wei Z, Wang H, Zhang X, Ning X, Ni X, Fang J. Atypical Resting-State and Task-Evoked EEG Signatures in Children with Developmental Language Disorder. Bioengineering. 2026; 13(1):119. https://doi.org/10.3390/bioengineering13010119
Chicago/Turabian StyleLiang, Aimin, Zhijun Cui, Yang Shi, Chunyan Qu, Zhuang Wei, Hanxiao Wang, Xu Zhang, Xiaolin Ning, Xin Ni, and Jiancheng Fang. 2026. "Atypical Resting-State and Task-Evoked EEG Signatures in Children with Developmental Language Disorder" Bioengineering 13, no. 1: 119. https://doi.org/10.3390/bioengineering13010119
APA StyleLiang, A., Cui, Z., Shi, Y., Qu, C., Wei, Z., Wang, H., Zhang, X., Ning, X., Ni, X., & Fang, J. (2026). Atypical Resting-State and Task-Evoked EEG Signatures in Children with Developmental Language Disorder. Bioengineering, 13(1), 119. https://doi.org/10.3390/bioengineering13010119







