Quantifying In Vivo Arterial Deformation from CT and MRI: A Systematic Review of Segmentation, Motion Tracking, and Kinematic Metrics
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy and Information Sources
(“aorta” OR “aortic” OR “arterial” OR “vascular”) AND (“patient-specific” OR “in vivo”) AND (“strain” OR “deformation” OR “compliance” OR “motion tracking”) AND (“Computed Tomography” OR “CT” OR “Magnetic Resonance” OR “MRI”) AND NOT (“experimental” OR “Fluid-structure interaction” OR “computational fluid” OR “Finite element” OR “heart”).
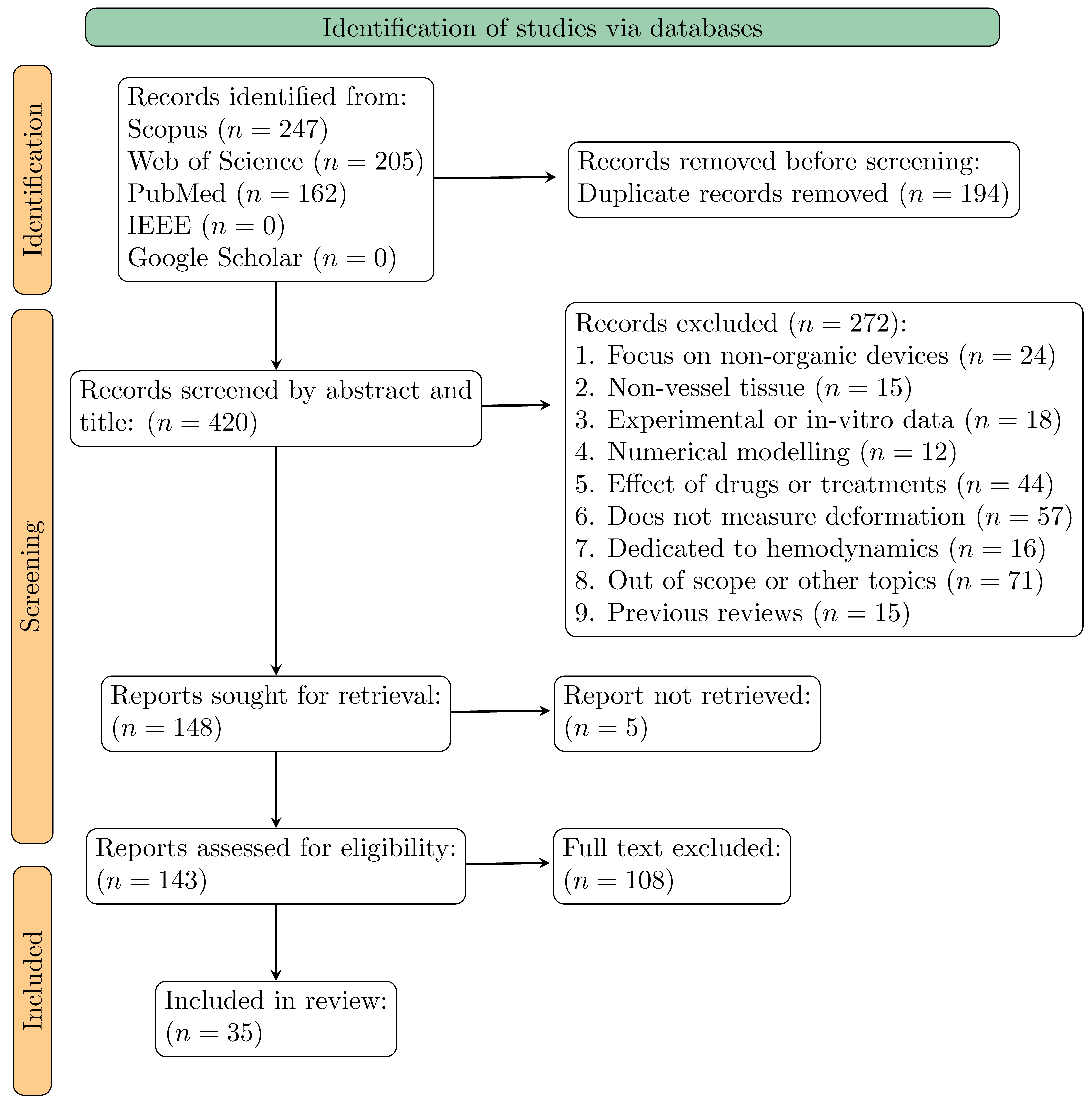
2.2. Study Selection and Data Extraction
2.3. Synthesis Approach
- Sample Size Adequacy: Studies with subjects were rated as adequate, n = 10–19 as moderate, and as limited.
- Time resolution: The number of time steps each article takes into account to analyse the deformation with adequate articles , partial with t = 5–15 and inadequate with .
- Tracking Method Validation: Whether the motion tracking algorithm was validated (e.g., against phantom data, in vitro experiments, or established ground truth).
- Reproducibility Reporting: Whether observer or automatic metrics were reported.
3. Results
3.1. Publications Overview
3.2. Image Segmentation Methods
3.3. Temporal 3D Deformation Tracking Methods
3.4. Metrics Used to Measure Deformation and Results
3.4.1. Kinematic Metrics
3.4.2. Geometric Metrics
3.4.3. Stiffness and Material Properties
3.4.4. Other Metrics
3.5. Dynamic Results
4. Discussion
Standardisation Recommendations and Proposed Roadmap
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AA | Abdominal Aorta |
| AAA | Abdominal Aorta Aneurysms |
| AoA | Aortic Arch |
| ATAA | Ascending Thoracic Aorta Aneurysm |
| BAV | Bicuspid Aortic Valve |
| CNN | Convolutional Neural Networks |
| CT | Computed Tomography |
| CTA | Computed Tomography Angiography |
| CVD | Cardiovascular Diseases |
| DTA | Descending Thoracic Aorta |
| ECG | Electrocardiographic |
| ICP | Iterative Closest Point |
| LDDMM | Large Deformation Diffeomorphic Metric Mapping |
| LESI | Local Extensional Stiffness Identification |
| MRI | Magnetic Resonance Imaging |
| OSF | Open Science Framework |
| PPA | Proximal Pulmonary Artery |
| PRISMA | Preferred Reporting Items for Systematic Reviews and Meta-Analyses |
| ROI | Region of Interest |
| SFA | superficial Femoral Artery |
| SyN | Symmetric Normalisation |
| TA | Thoracic Aortic Aneurysm |
| TAV | Tricuspid Aortic Valve |
| TEVAR | Thoracic Endovascular Aortic Repair |
| US | Ultrasound |
References
- Cheng, D.C.Y.; Climie, R.E.; Shu, M.; Grieve, S.M.; Kozor, R.; Figtree, G.A. Vascular ageing and cardiovascular disease: Pathophysiology and measurement in the coronary arteries. Front. Cardiovasc. Med. 2023, 10, 1206156. [Google Scholar] [CrossRef] [PubMed]
- Strain, W.D.; Paldánius, P.M. Diabetes, cardiovascular disease and the microcirculation. Cardiovasc. Diabetol. 2018, 17, 57. [Google Scholar] [CrossRef]
- Lu, Y.; Kiechl, S.J.; Wang, J.; Xu, Q.; Kiechl, S.; Pechlaner, R.; Aguilar, D.; Al-Hashmi, K.M.; Alvim, R.O.; Al-Zakwani, I.S.; et al. Global distributions of age- and sex-related arterial stiffness: Systematic review and meta-analysis of 167 studies with 509,743 participants. eBioMedicine 2023, 92, 104619. [Google Scholar] [CrossRef]
- Cecelja, M.; Ruijsink, B.; Puyol-Antón, E.; Li, Y.; Godwin, H.; King, A.P.; Razavi, R.; Chowienczyk, P. Aortic distensibility measured by automated analysis of Magnetic Resonance Imaging predicts adverse cardiovascular events in UK Biobank. J. Am. Heart Assoc. 2022, 11, e026361. [Google Scholar] [CrossRef]
- Mensel, B.; Mahnken, A.H.; Kaiser, E.; Völzke, H.; Dörr, M.; Felix, S.B.; Ittermann, T.; Lieb, W.; Lorbeer, R. Association of aortic diameters and mortality in the general population—An MRI-based study. Eur. Radiol. 2025, 35, 427–437. [Google Scholar] [CrossRef]
- Isselbacher, E.M.; Preventza, O.; Hamilton Black, J., III; Augoustides, J.G.; Beck, A.W.; Bolen, M.A.; Braverman, A.C.; Bray, B.E.; Brown-Zimmerman, M.M.; Chen, E.P.; et al. 2022 ACC/AHA Guideline for the Diagnosis and Management of Aortic Disease: A Report of the American Heart Association/American College of Cardiology Joint Committee on Clinical Practice Guidelines. J. Am. Coll. Cardiol. 2022, 80, e223–e393. [Google Scholar] [CrossRef] [PubMed]
- Van Disseldorp, E.; Petterson, N.; Rutten, M.; Van De Vosse, F.; Van Sambeek, M.; Lopata, R. Patient Specific Wall Stress Analysis and Mechanical Characterization of Abdominal Aortic Aneurysms Using 4D Ultrasound. Eur. J. Vasc. Endovasc. Surg. 2016, 52, 635–642. [Google Scholar] [CrossRef]
- Maas, E.J.; Nievergeld, A.H.M.; Fonken, J.H.C.; Thirugnanasambandam, M.; Van Sambeek, M.R.H.M.; Lopata, R.G.P. 3D-Ultrasound Based Mechanical and Geometrical Analysis of Abdominal Aortic Aneurysms and Relationship to Growth. Ann. Biomed. Eng. 2023, 51, 2554–2565. [Google Scholar] [CrossRef]
- Satriano, A.; Rivolo, S.; Martufi, G.; Finol, E.A.; Di Martino, E.S. In Vivo Strain Assessment of the Abdominal Aortic Aneurysm. J. Biomech. 2015, 48, 354–360. [Google Scholar] [CrossRef]
- Herold, V.; Wellen, J.; Ziener, C.H.; Weber, T.; Hiller, K.H.; Nordbeck, P.; Rommel, E.; Haase, A.; Bauer, W.R.; Jakob, P.M.; et al. In Vivo Comparison of Atherosclerotic Plaque Progression with Vessel Wall Strain and Blood Flow Velocity in apoE−/− Mice with MR Microscopy at 17.6 T. Magn. Reson. Mater. Physics, Biol. Med. 2009, 22, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Mourato, A.; Valente, R.; Xavier, J.; Brito, M.; Avril, S.; de Sá, J.C.; Tomás, A.; Fragata, J. Computational Modelling and Simulation of Fluid Structure Interaction in Aortic Aneurysms: A Systematic Review and Discussion of the Clinical Potential. Appl. Sci. 2022, 12, 8049. [Google Scholar] [CrossRef]
- Sotelo, J.; Dux-Santoy, L.; Guala, A.; Rodríguez-Palomares, J.; Evangelista, A.; Sing-Long, C.; Urbina, J.; Mura, J.; Hurtado, D.E.; Uribe, S. 3D Axial and Circumferential Wall Shear Stress from 4D Flow MRI Data Using a Finite Element Method and a Laplacian Approach. Magn. Reson. Med. 2018, 79, 2816–2823. [Google Scholar] [CrossRef]
- Pasta, S.; Agnese, V.; Di Giuseppe, M.; Gentile, G.; Raffa, G.M.; Bellavia, D.; Pilato, M. In Vivo Strain Analysis of Dilated Ascending Thoracic Aorta by CG-Gated CT Angiographic Imaging. Ann. Biomed. Eng. 2017, 45, 2911–2920. [Google Scholar] [CrossRef]
- Vander Linden, K.; Vanderveken, E.; Van Hoof, L.; Maes, L.; Fehervary, H.; Dreesen, S.; Hendrickx, A.; Verbrugghe, P.; Rega, F.; Meuris, B.; et al. Stiffness Matters: Improved Failure Risk Assessment of Ascending Thoracic Aortic Aneurysms. JTCVS Open 2023, 16, 66–83. [Google Scholar] [CrossRef]
- Rosnel, C.; Sivera, R.; Cervi, E.; Danton, M.; Schievano, S.; Capelli, C.; Aggarwal, A. Are Aortic Biomechanical Properties Early Markers of Dilatation in Patients with Marfan Syndrome? A Systematic Review and Meta-Analysis. Biomech. Model. Mechanobiol. 2024, 23, 2043–2061. [Google Scholar] [CrossRef]
- Leemans, E.L.; Willems, T.P.; Van Der Laan, M.J.; Slump, C.H.; Zeebregts, C.J. Biomechanical Indices for Rupture Risk Estimation in Abdominal Aortic Aneurysms. J. Endovasc. Ther. 2017, 24, 254–261. [Google Scholar] [CrossRef]
- Simon, A. Stratification of Vascular Risk in Hypertension and Therapeutic Perspective. Am. J. Hypertens. 1995, 8, 45S–48S. [Google Scholar] [CrossRef]
- Oluwole, B.O.; Du, W.; Mills, I.; Sumpio, B.E. Gene Regulation by Mechanical Forces. Endothelium 1997, 5, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Reneman, R.S.; Arts, T.; Hoeks, A.P. Wall Shear Stress—An Important Determinant of Endothelial Cell Function and Structure—In the Arterial System in Vivo. Cardiovasc. Res. 2006, 43, 251–269. [Google Scholar] [CrossRef]
- De Korte, C.L.; Fekkes, S.; Nederveen, A.J.; Manniesing, R.; Hansen, H.R.H.G. Review: Mechanical Characterization of Carotid Arteries and Atherosclerotic Plaques. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2016, 63, 1613–1623. [Google Scholar] [CrossRef] [PubMed]
- Rong, L.Q.; Kim, J.; Gregory, A.J. Speckle Tracking Echocardiography: Imaging Insights into the Aorta. Curr. Opin. Cardiol. 2020, 35, 116–122. [Google Scholar] [CrossRef]
- Musch, G. A Window on the Lung: Molecular Imaging as a Tool to Dissect Pathophysiologic Mechanisms of Acute Lung Disease. Contrast Media Mol. Imaging 2019, 2019, 1510507. [Google Scholar] [CrossRef]
- Wells, P.N.T.; Liang, H.D. Medical Ultrasound: Imaging of Soft Tissue Strain and Elasticity. J. R. Soc. Interface 2011, 8, 1521–1549. [Google Scholar] [CrossRef]
- Linninger, A.A.; Tangen, K.; Hsu, C.Y.; Frim, D. Cerebrospinal Fluid Mechanics and Its Coupling to Cerebrovascular Dynamics. Annu. Rev. Fluid Mech. 2016, 48, 219–257. [Google Scholar] [CrossRef]
- Jain, V.; Duda, J.; Avants, B.; Giannetta, M.; Xie, S.X.; Roberts, T.; Detre, J.A.; Hurt, H.; Wehrli, F.W.; Wang, D.J.J. Longitudinal Reproducibility and Accuracy of Pseudo-Continuous Arterial Spin–Labeled Perfusion MR Imaging in Typically Developing Children. Radiology 2012, 263, 527–536. [Google Scholar] [CrossRef]
- Cardoso, L.; Fritton, S.P.; Gailani, G.; Benalla, M.; Cowin, S.C. Advances in Assessment of Bone Porosity, Permeability and Interstitial Fluid Flow. J. Biomech. 2013, 46, 253–265. [Google Scholar] [CrossRef] [PubMed]
- Zaloszyc, A.; Bernardor, J.; Bacchetta, J.; Laverny, G.; Schmitt, C.P. Mouse Models of Mineral Bone Disorders Associated with Chronic Kidney Disease. Int. J. Mol. Sci. 2023, 24, 5325. [Google Scholar] [CrossRef] [PubMed]
- Genet, N.; Bhatt, N.; Bourdieu, A.; Hirschi, K.K. Multifaceted Roles of Connexin 43 in Stem Cell Niches. Curr. Stem Cell Rep. 2018, 4, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Houben, I.B.; Nama, N.; Moll, F.L.; Van Herwaarden, J.A.; Nordsletten, D.A.; Williams, D.M.; Patel, H.J.; Figueroa, C.A.; Burris, N.S. Mapping Pre-Dissection Aortic Wall Abnormalities: A Multiparametric Assessment. Eur. J. Cardio-Thorac. Surg. 2020, 57, 1061–1067. [Google Scholar] [CrossRef]
- Farzaneh, S.; Trabelsi, O.; Chavent, B.; Avril, S. Identifying Local Arterial Stiffness to Assess the Risk of Rupture of Ascending Thoracic Aortic Aneurysms. Ann. Biomed. Eng. 2019, 47, 1038–1050. [Google Scholar] [CrossRef]
- Farzaneh, S.; Trabelsi, O.; Avril, S. Inverse Identification of Local Stiffness across Ascending Thoracic Aortic Aneurysms. Biomech. Model. Mechanobiol. 2019, 18, 137–153. [Google Scholar] [CrossRef] [PubMed]
- Hirotsu, K.; Suh, G.Y.; Lee, J.T.; Dake, M.D.; Fleischmann, D.; Cheng, C.P. Changes in Geometry and Cardiac Deformation of the Thoracic Aorta after Thoracic Endovascular Aortic Repair. Ann. Vasc. Surg. 2018, 46, 83–89. [Google Scholar] [CrossRef]
- Suh, G.Y.K.; Bondesson, J.; Zhu, Y.D.; Nilson, M.C.; Roselli, E.E.; Cheng, C.P. Ascending Aortic Endograft and Thoracic Aortic Deformation After Ascending Thoracic Endovascular Aortic Repair. J. Endovasc. Ther. 2025, 32, 7–17. [Google Scholar] [CrossRef]
- Suh, G.Y.; Beygui, R.E.; Fleischmann, D.; Cheng, C.P. Aortic Arch Vessel Geometries and Deformations in Patients with Thoracic Aortic Aneurysms and Dissections. J. Vasc. Interv. Radiol. 2014, 25, 1903–1911. [Google Scholar] [CrossRef]
- Derwich, W.; Schönborn, M.; Blase, C.; Wittek, A.; Oikonomou, K.; Böckler, D.; Erhart, P. Correlation of Four-Dimensional Ultrasound Strain Analysis with Computed Tomography Angiography Wall Stress Simulations in Abdominal Aortic Aneurysms. JVS-Vasc. Sci. 2024, 5, 100199. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.; Qi, X.; Cao, L.; Yang, M.; Luo, H.; Li, Q.; Qian, P.; Lu, J.; Lei, Z.; Luo, Y.; et al. Regional Stiffness and Hardening Indices: New Indicators Derived from Multidimensional Dynamic CTA for Aneurysm Risk Assessment. Adv. Sci. 2024, 11, 2400653. [Google Scholar] [CrossRef]
- Satriano, A.; Guenther, Z.; White, J.A.; Merchant, N.; Di Martino, E.S.; Al-Qoofi, F.; Lydell, C.P.; Fine, N.M. Three-Dimensional Thoracic Aorta Principal Strain Analysis from Routine ECG-gated Computerized Tomography: Feasibility in Patients Undergoing Transcatheter Aortic Valve Replacement. BMC Cardiovasc. Disord. 2018, 18, 76. [Google Scholar] [CrossRef]
- Brown, R.; Nguyen, T.D.; Spincemaille, P.; Prince, M.R.; Wang, Y. In Vivo Quantification of Femoral-popliteal Compression during Isometric Thigh Contraction: Assessment Using MR Angiography. J. Magn. Reson. Imaging Off. J. Int. Soc. Magn. Reson. Med. 2009, 29, 1116–1124. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.P.; Wilson, N.M.; Hallett, R.L.; Herfkens, R.J.; Taylor, C.A. In Vivo MR Angiographic Quantification of Axial and Twisting Deformations of the Superficial Femoral Artery Resulting from Maximum Hip and Knee Flexion. J. Vasc. Interv. Radiol. 2006, 17, 979–987. [Google Scholar] [CrossRef]
- Cheng, C.P.; Choi, G.; Herfkens, R.J.; Taylor, C.A. The Effect of Aging on Deformations of the Superficial Femoral Artery Resulting from Hip and Knee Flexion: Potential Clinical Implications. J. Vasc. Interv. Radiol. 2010, 21, 195–202. [Google Scholar] [CrossRef]
- Mandal, K.; Parent, F.; Martel, S.; Kashyap, R.; Kadoury, S. Vessel-Based Registration of an Optical Shape Sensing Catheter for MR Navigation. Int. J. Comput. Assist. Radiol. Surg. 2016, 11, 1025–1034. [Google Scholar] [CrossRef] [PubMed]
- Ronneberger, O.; Fischer, P.; Brox, T. U-Net: Convolutional Networks for Biomedical Image Segmentation. In Medical Image Computing and Computer-Assisted Intervention—MICCAI: Proceedings of the 18th International Conference, Munich, Germany, 5–9 October 2015; Navab, N., Hornegger, J., Wells, W.M., Frangi, A.F., Eds.; Springer International Publishing: Cham, Switzerland, 2015; pp. 234–241. [Google Scholar] [CrossRef]
- Tjahjadi, N.; Campello Jorge, C.; Marway, P.S.; Kim, T.; Baker, T.; Hazenberg, C.; Van Herwaarden, J.A.; Figueroa, C.A.; Patel, H.J.; Burris, N.S. Three-Dimensional Characterization of Ascending Aortic Strain, Motion and Growth in Patients Undergoing Thoracic Endovascular Aortic Repair. JVS-Vasc. Sci. 2025, 6, 100293. [Google Scholar] [CrossRef] [PubMed]
- Parent, F.; Gerard, M.; Monet, F.; Loranger, S.; Soulez, G.; Kashyap, R.; Kadoury, S. Intra-Arterial Image Guidance with Optical Frequency Domain Reflectometry Shape Sensing. IEEE Trans. Med. Imaging 2018, 38, 482–492. [Google Scholar] [CrossRef]
- Markodimitrakis, E.; Lin, S.; Koutoulakis, E.; Marín-Castrillón, D.M.; Tovar Sáez, F.A.; Leclerc, S.; Bernard, C.; Boucher, A.; Presles, B.; Bouchot, O.; et al. Comparison of In-Vivo and Ex-Vivo Ascending Aorta Elastic Properties through Automatic Deep Learning Segmentation of Cine-MRI and Biomechanical Testing. J. Clin. Med. 2023, 12, 402. [Google Scholar] [CrossRef]
- Du, P.; An, D.; Wang, C.; Wang, J.X. AI-powered automated model construction for patient-specific CFD simulations of aortic flows. Sci. Adv. 2025, 11, eadw2825. [Google Scholar] [CrossRef]
- Choi, G.; Cheng, C.P.; Wilson, N.M.; Taylor, C.A. Methods for Quantifying Three-Dimensional Deformation of Arteries Due to Pulsatile and Nonpulsatile Forces: Implications for the Design of Stents and Stent Grafts. Ann. Biomed. Eng. 2009, 37, 14–33. [Google Scholar] [CrossRef] [PubMed]
- Martin, C.; Sun, W.; Primiano, C.; McKay, R.; Elefteriades, J. Age-Dependent Ascending Aorta Mechanics Assessed Through Multiphase CT. Ann. Biomed. Eng. 2013, 41, 2565–2574. [Google Scholar] [CrossRef]
- Morrison, T.M.; Choi, G.; Zarins, C.K.; Taylor, C.A. Circumferential and Longitudinal Cyclic Strain of the Human Thoracic Aorta: Age-related Changes. J. Vasc. Surg. 2009, 49, 1029–1036. [Google Scholar] [CrossRef]
- Iffrig, E.; Wilson, J.S.; Zhong, X.; Oshinski, J.N. Demonstration of Circumferential Heterogeneity in Displacement and Strain in the Abdominal Aortic Wall by Spiral Cine DENSE MRI. J. Magn. Reson. Imaging 2019, 49, 731–743. [Google Scholar] [CrossRef]
- Hauguel, A.; Bondesson, J.; Azarine, A.; Kasani, K.; Barakat, A.I.; Haulon, S.; Cheng, C.P. Impacts of Thoracic Endovascular Aortic Repair and Thrombus Load on Aortic Radial Deformation. J. Endovasc. Ther. 2025, 0, 15266028251339347. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.S.; Taylor, W.R.; Oshinski, J. Assessment of the Regional Distribution of Normalized Circumferential Strain in the Thoracic and Abdominal Aorta Using DENSE Cardiovascular Magnetic Resonance. J. Cardiovasc. Magn. Reson. 2019, 21, 59. [Google Scholar] [CrossRef]
- Phan, L.; Courchaine, K.; Azarbal, A.; Vorp, D.; Grimm, C.; Rugonyi, S. A Geodesics-Based Surface Parameterization to Assess Aneurysm Progression. J. Biomech. Eng. 2016, 138, 054503. [Google Scholar] [CrossRef]
- Metaxa, E.; Iordanov, I.; Maravelakis, E.; Papaharilaou, Y. A Novel Approach for Local Abdominal Aortic Aneurysm Growth Quantification. Med. Biol. Eng. Comput. 2017, 55, 1277–1286. [Google Scholar] [CrossRef]
- Wilson, J.S.; Zhong, X.; Hair, J.; Robert Taylor, W.; Oshinski, J.N. In Vivo Quantification of Regional Circumferential Green Strain in the Thoracic and Abdominal Aorta by Two-Dimensional Spiral Cine DENSE MRI. J. Biomech. Eng. 2019, 141, 060901. [Google Scholar] [CrossRef] [PubMed]
- Manning, E.P.; Mishall, P.; Ramachandra, A.B.; Hassab, A.H.M.; Lamy, J.; Peters, D.C.; Murphy, T.E.; Heerdt, P.; Singh, I.; Downie, S.; et al. Stiffening of the Human Proximal Pulmonary Artery with Increasing Age. Physiol. Rep. 2024, 12, e16090. [Google Scholar] [CrossRef] [PubMed]
- Wedding, K.L.; Draney, M.T.; Herfkens, R.J.; Zarins, C.K.; Taylor, C.A.; Pelc, N.J. Measurement of Vessel Wall Strain Using Cine Phase Contrast MRI. J. Magn. Reson. Imaging Off. J. Int. Soc. Magn. Reson. Med. 2002, 15, 418–428. [Google Scholar] [CrossRef] [PubMed]
- Simmering, J.A.; Leeuwerke, S.J.G.; Meerwaldt, R.; Zeebregts, C.J.; Slump, C.H.; Geelkerken, R.H. In Vivo Quantification of Cardiac-Pulsatility-Induced Motion Before and After Double-Branched Endovascular Aortic Arch Repair. J. Endovasc. Ther. 2023, 30, 510–519. [Google Scholar] [CrossRef]
- Franquet, A.; Avril, S.; Le Riche, R.; Badel, P.; Schneider, F.C.; Li, Z.Y.; Boissier, C.; Favre, J.P. A New Method for the In Vivo Identification of Mechanical Properties in Arteries From Cine MRI Images: Theoretical Framework and Validation. IEEE Trans. Med. Imaging 2013, 32, 1448–1461. [Google Scholar] [CrossRef]
- Ayadi, A.; Sahtout, W.; Balédent, O. A Non-invasive Method for Determining Biomechanical Properties of the Internal Carotid Artery. IRBM 2020, 41, 125–132. [Google Scholar] [CrossRef]
- Liu, H.; Canton, G.; Yuan, C.; Yang, C.; Billiar, K.; Teng, Z.; Hoffman, A.H.; Tang, D. Using In Vivo Cine and 3D Multi-Contrast MRI to Determine Human Atherosclerotic Carotid Artery Material Properties and Circumferential Shrinkage Rate and Their Impact on Stress/Strain Predictions. J. Biomech. Eng. 2012, 134, 011008. [Google Scholar] [CrossRef]
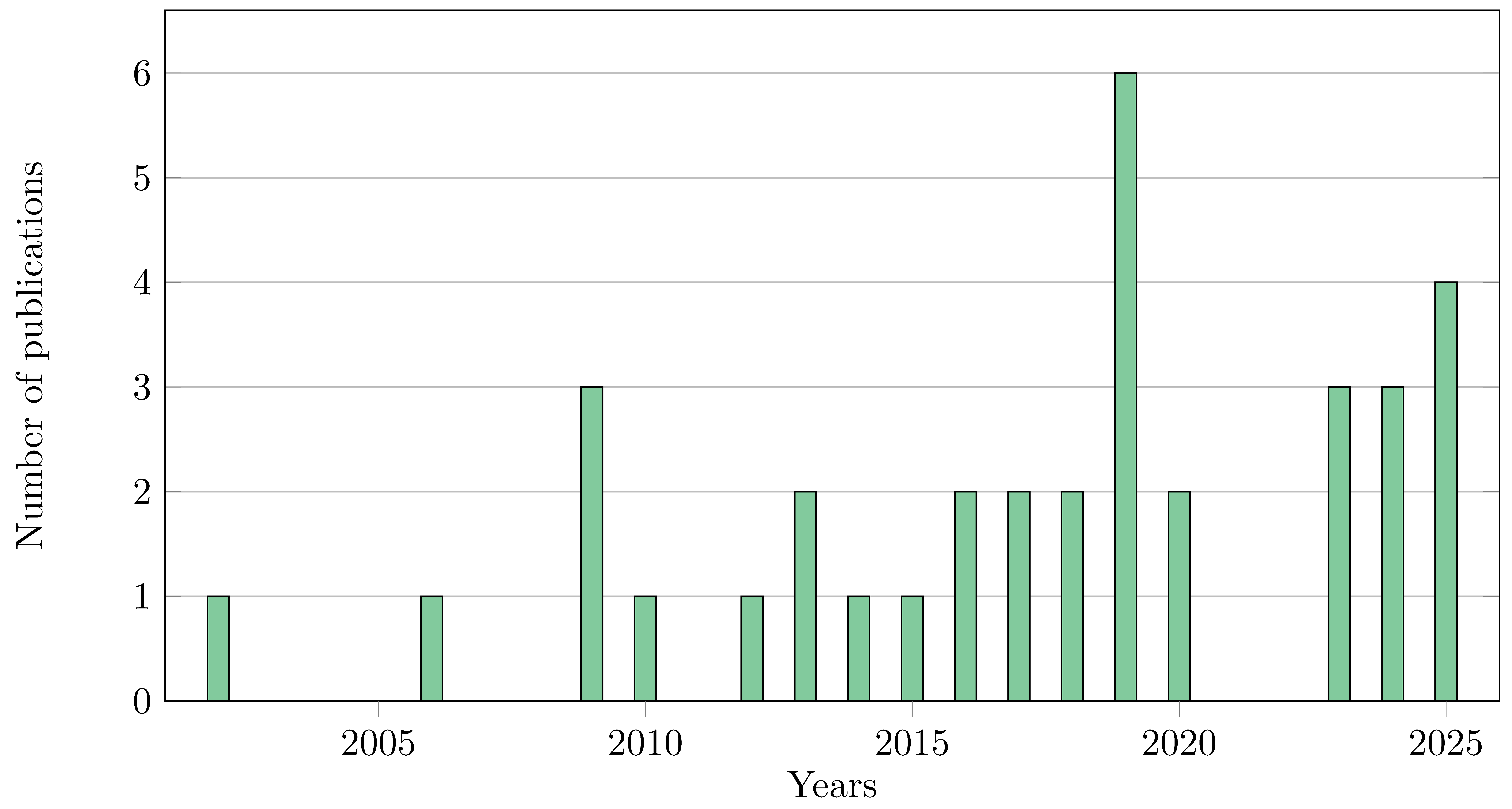
| Quality Domain | Adq. | Partial | Inadq. | Key Concerns |
|---|---|---|---|---|
| Sample Size Adequacy | 11 | 9 | 15 | Number of pilot studies |
| Time resolution | 15 | 7 | 13 | Not consistent acquisitions |
| Tracking Method Validation | 15 | - | 20 | Limited validation |
| Reproducibility reporting | 19 | - | 16 | Not automatized in some |
| Age | 30–49 | 50–59 | 60–69 | 70–85 |
|---|---|---|---|---|
| Ascending Thoracic Aorta (ATA) | 0.092±0.03 [48]; ≈0.108 ± 0.056 [49] | 0.072 ± 0.030 [48] | 0.056 ± 0.03 [48]; ≈0.030 ± 0.014 [49] | 0.102 ± 0.06 [37] |
| AoA | ≈0.082 ± 0.031 [49]; 0.08 ± 0.05 [55] | ≈0.023 ± 0.016 [49] | 0.061 ± 0.029 [37] | |
| AA | 0.14±0.05 [50]; 0.081 ± 0.033 [49] | ≈0.065 ± 0.016 [55] | 0.042 ± 0.019 [49] | 0.033 ± 0.017 [37] |
| AAA | 0.0570 ± 0.0226 [9] | 0.0102 ± 0.0063 (Circ.) [35] | ||
| 0.0091 ± 0.005 (Axial) [35] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Valente, R.; Henriques, B.; Mourato, A.; Xavier, J.; Brito, M.; Avril, S.; Tomás, A.; Fragata, J. Quantifying In Vivo Arterial Deformation from CT and MRI: A Systematic Review of Segmentation, Motion Tracking, and Kinematic Metrics. Bioengineering 2026, 13, 121. https://doi.org/10.3390/bioengineering13010121
Valente R, Henriques B, Mourato A, Xavier J, Brito M, Avril S, Tomás A, Fragata J. Quantifying In Vivo Arterial Deformation from CT and MRI: A Systematic Review of Segmentation, Motion Tracking, and Kinematic Metrics. Bioengineering. 2026; 13(1):121. https://doi.org/10.3390/bioengineering13010121
Chicago/Turabian StyleValente, Rodrigo, Bernardo Henriques, André Mourato, José Xavier, Moisés Brito, Stéphane Avril, António Tomás, and José Fragata. 2026. "Quantifying In Vivo Arterial Deformation from CT and MRI: A Systematic Review of Segmentation, Motion Tracking, and Kinematic Metrics" Bioengineering 13, no. 1: 121. https://doi.org/10.3390/bioengineering13010121
APA StyleValente, R., Henriques, B., Mourato, A., Xavier, J., Brito, M., Avril, S., Tomás, A., & Fragata, J. (2026). Quantifying In Vivo Arterial Deformation from CT and MRI: A Systematic Review of Segmentation, Motion Tracking, and Kinematic Metrics. Bioengineering, 13(1), 121. https://doi.org/10.3390/bioengineering13010121










