Overview on Photoreforming of Biomass Aqueous Solutions to Generate H2 in the Presence of g-C3N4-Based Materials
Abstract
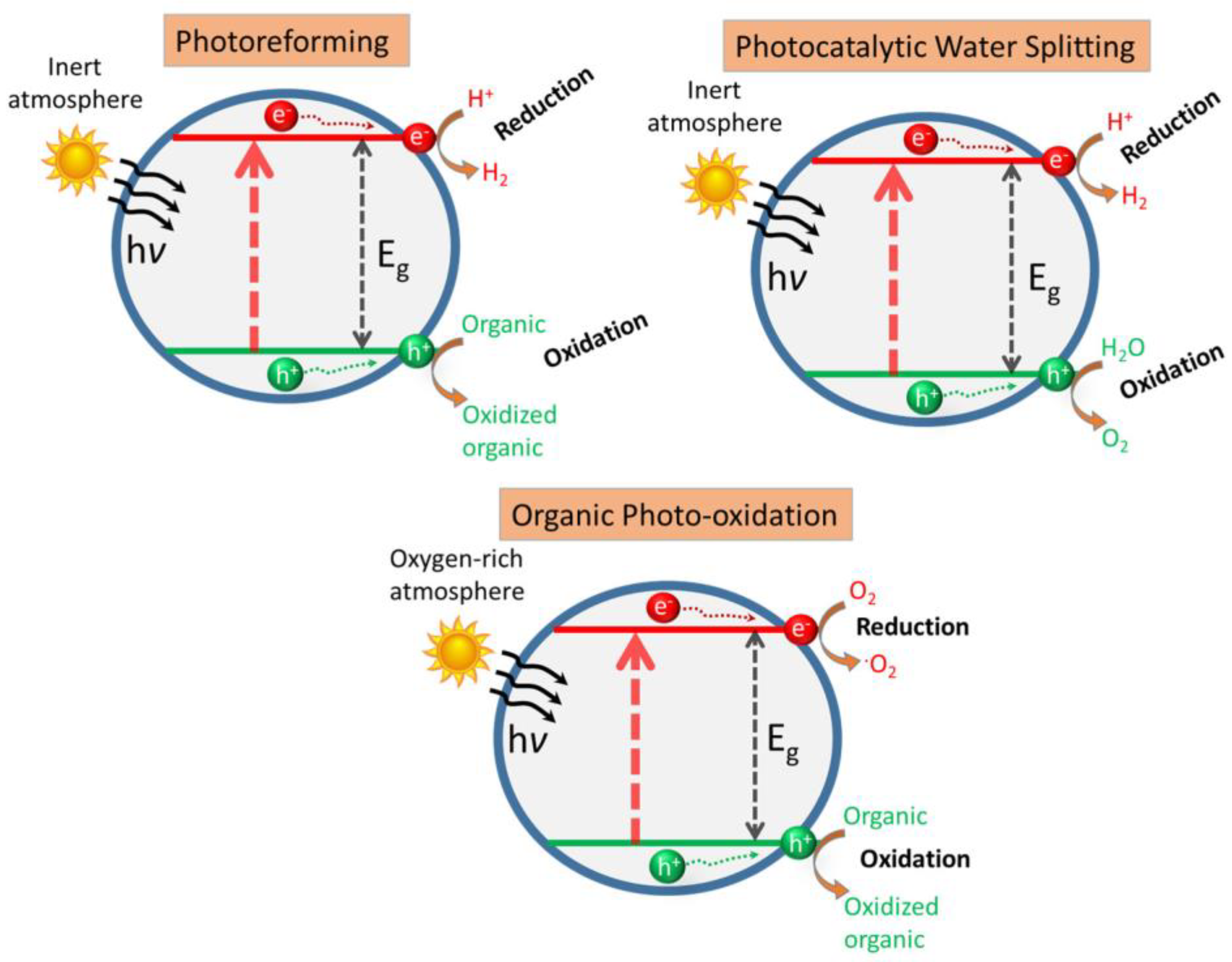
1. Introduction
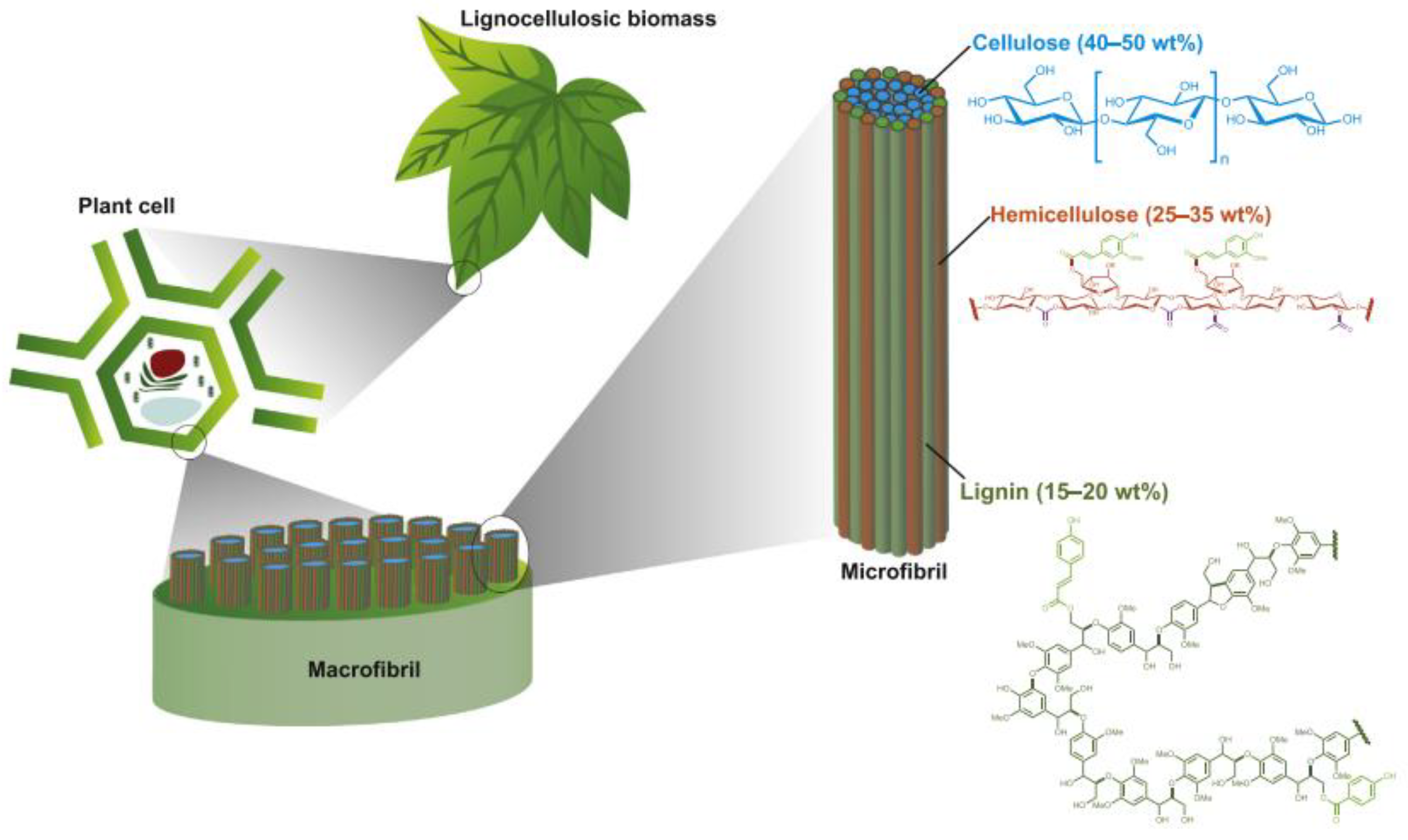
2. Organic Molecules as Hole Scavengers in Photoreforming: From Model Molecules to Biomass
3. Some Considerations and Details on g-C3N4-Based Photocatalysts for H2 Production
3.1. g-C3N4 as Photocatalyst
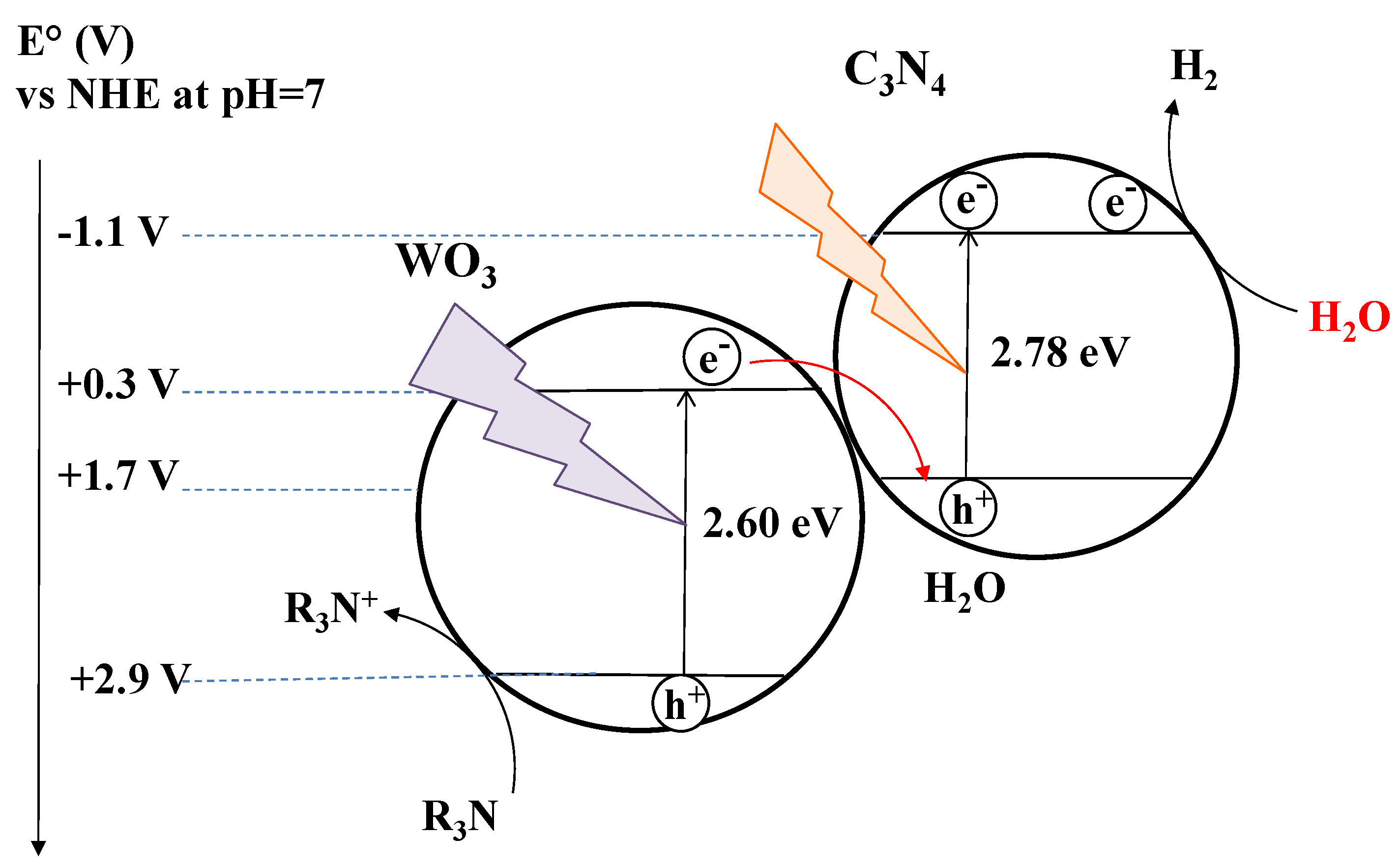
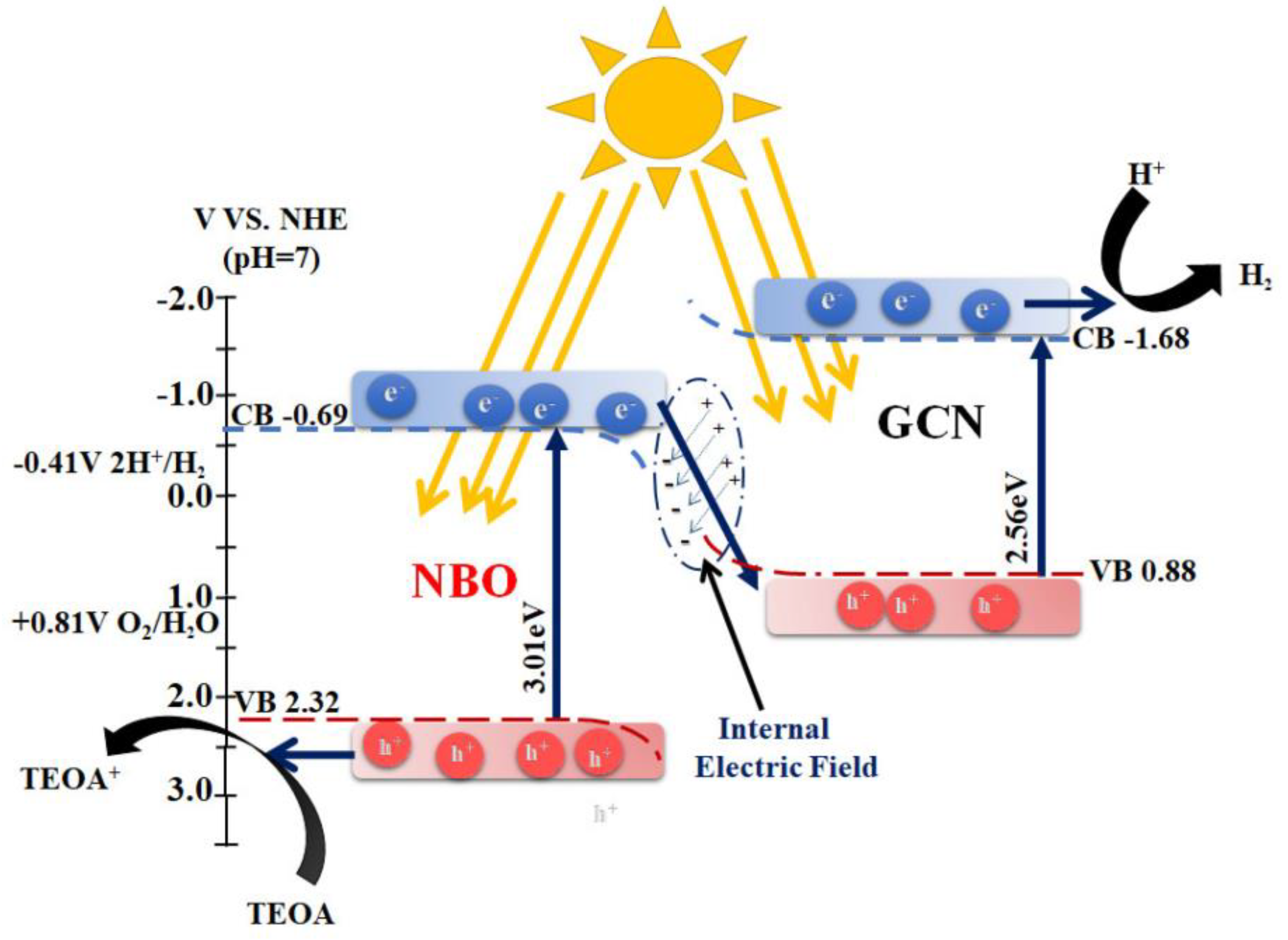
3.2. g-C3N4 in Anaerobic Conditions: Strategies to Improve Its Photocatalytic Activity
3.3. Real Wastes as Hole Scavengers in the Photoreforming Process in the Presence of g-C3N4-Based Semiconductors
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Ng, C.H.; Teo, S.H.; Mansir, N.; Islam, A.; Joseph, C.G.; Hayase, S.; Taufiq-Yap, Y.H.; Yap, T. Recent advancements and opportunities of decorated graphitic carbon nitride toward solar fuel production and beyond. Sustain. Energy Fuels 2021, 5, 4457–4511. [Google Scholar] [CrossRef]
- DOE Hydrogen and Fuel Cells Program: 2014 Annual Progress Report; US Department of Energy: Washington, DC, USA, 2014.
- Turner, J.A. Sustainable hydrogen production. Science 2004, 305, 972–974. [Google Scholar] [CrossRef] [PubMed]
- García-López, E.I.; Palmisano, L. Chapter 1: Fundamentals of photocatalysis: The role of the photocatalysts in heterogeneous photo-assisted reactions. In Materials Science in Photocatalysis, 1st ed.; García-López, E.I., Palmisano, L., Eds.; Elsevier: Amsterdam, The Netherlands, 2021; pp. 3–9. [Google Scholar]
- Fujishima, A.; Honda, K. Electrochemical Photolysis of Water at a Semiconductor Electrode. Nature 1972, 238, 37–38. [Google Scholar] [CrossRef] [PubMed]
- Samage, A.; Gupta, P.; Halakarni, M.A.; Nataraj, S.K.; Sinhamahapatra, A. Progress in the Photoreforming of Carboxylic Acids for Hydrogen Production. Photochemistry 2022, 2, 40. [Google Scholar] [CrossRef]
- Jitputti, J.; Pavasupree, S.; Suzuki, Y.; Yoshikawa, S. Synthesis and photocatalytic activity for water-splitting reaction of nanocrystalline mesoporous titania prepared by hydrothermal method. J. Solid State Chem. 2007, 180, 1743–1749. [Google Scholar] [CrossRef]
- Kawai, T.; Sakata, T. Conversion of carbohydrate into hydrogen fuel by a photocatalytic process. Nature 1980, 286, 474–476. [Google Scholar] [CrossRef]
- Toe, C.Y.; Tsounis, C.; Zhang, J.; Masood, H.; Gunawan, D.; Scott, J.; Amal, R. Advancing photoreforming of organics: Highlights on photocatalyst and system designs for selective oxidation reactions. Energy Environ. Sci. 2021, 14, 1140–1175. [Google Scholar] [CrossRef]
- Al-Mazroai, L.S.; Bowker, M.; Davies, P.; Dickinson, A.; Greaves, J.; James, D.; Millard, L. The photocatalytic reforming of methanol. Catal. Today 2007, 122, 46–50. [Google Scholar] [CrossRef]
- Romero Ocana, I.; Beltram, A.; Delgado Jaen, J.J.; Adami, G.; Montini, T.; Fornasiero, P. Photocatalytic H2 production by ethanol photodehydrogenation: Effect of anatase/brookite nanocomposites composition. Inorg. Chim. Acta 2015, 431, 197–205. [Google Scholar] [CrossRef]
- Daskalaki, V.M.; Kondarides, D.I. Efficient production of hydrogen by photo-induced reforming of glycerol at ambient conditions. Catal. Today 2009, 144, 75–80. [Google Scholar] [CrossRef]
- Bahruji, H.; Bowker, M.; Davies, P.R.; Pedrono, F. New insights into the mechanism of photocatalytic reforming on Pd/TiO2. Appl. Catal. B 2011, 107, 205–209. [Google Scholar] [CrossRef]
- Bowker, M.; Morton, C.; Kennedy, J.; Bahruji, H.; Greaves, J.; Jones, W.; Davies, P.R.; Brookes, C.; Wells, P.P.; Dimitratos, N. Hydrogen production by photoreforming of biofuels using Au, Pd and Au-Pd/TiO2 photocatalysts. J. Catal. 2014, 10, 10–15. [Google Scholar] [CrossRef]
- Bowker, M.; Bahruji, H.; Kennedy, J.; Jones, W.; Hartley, G.; Morton, C. Photocatalytic Window: Photo-reforming of organics and water splitting for sustainable hydrogen production. Catal. Lett. 2015, 145, 214–219. [Google Scholar] [CrossRef]
- Caravaca, A.; Jones, W.; Hardacre, C.; Bowker, M. H2 production by the photocatalytic reforming of cellulose and raw biomass using Ni, Pd, Pt and Au on titania. Proc. R. Soc. A 2016, 472, 20160054. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Liu, K.; Yang, X.; Jin, D.; Li, Y.; Jiao, G.; Zhou, J.; Sun, R. Recent Advances and Challenges in Photoreforming of Biomass-Derived Feedstocks into Hydrogen, Biofuels, or Chemicals by Using Functional Carbon Nitride Photocatalysts. ChemSusChem 2021, 14, 4903–4922. [Google Scholar] [CrossRef]
- Bellardita, M.; García-López, E.I.; Marcì, G.; Palmisano, L. Photocatalytic formation of H2 and value-added chemicals in aqueous glucose (Pt)-TiO2 suspension. Int. J. Hydrogen Energy 2016, 41, 5934–5947. [Google Scholar] [CrossRef]
- Huang, C.W.; Nguyen, B.S.; Wu, J.C.S.; Nguyen, V.H. A current perspective for photocatalysis towards the hydrogen production from biomass-derived organic substances and water. Int. J. Hydrogen Energy 2020, 45, 18144–18159. [Google Scholar] [CrossRef]
- Wang, J.; Zhao, H.; Lui, P.; Yasri, N.; Zhong, N.; Kibria, M.G.; Hu, J. Selective superoxide radical generation for glucose photoreforming into arabinose. J. Energy Chem. 2022, 74, 324–331. [Google Scholar] [CrossRef]
- Marcì, G.; García-López, E.I.; Palmisano, L. Polymeric carbon nitride (C3N4) as heterogeneous photocatalyst for selective oxidation of alcohols to aldehydes. Catal. Today 2018, 315, 126–137. [Google Scholar] [CrossRef]
- Bowker, M.; O’Rourke, C.; Mills, A. The Role of Metal Nanoparticles in Promoting Photocatalysis by TiO2. Top. Curr. Chem. 2022, 380, 17. [Google Scholar] [CrossRef]
- Anastas, P.T.; Warner, J.C. Green Chemistry: Theory and Practice; Oxford University Press: Oxford, UK, 1998. [Google Scholar]
- Sheldon, R.A. Fundamentals of green chemistry: Efficiency in reaction design. Chem Soc. Rev. 2012, 41, 1437–1451. [Google Scholar] [CrossRef] [PubMed]
- Butburee, T.; Chakthranont, P.; Phawa, C.; Faungnawakij, K. Beyond Artificial Photosynthesis: Prospects on Photobiorefinery. ChemCatChem 2020, 12, 1873–1890. [Google Scholar] [CrossRef]
- Rodríguez-Padrón, D.; Puente-Santiago, A.R.; Balu, A.M. Environmental catalysis: Present and future. ChemCatChem 2019, 11, 18–38. [Google Scholar] [CrossRef]
- Huber, G.W.; Iborra, S.; Corma, A. Synthesis of transportation fuels from biomass: Chemistry, catalysts, and engineering. Chem. Rev. 2006, 106, 4044–4098. [Google Scholar] [CrossRef] [PubMed]
- Bertella, S.; Luterbacher, J.S. Lignin Functionalization for the Production of Novel Materials. Trends Chem. 2020, 2, 440–453. [Google Scholar] [CrossRef]
- Zhao, H.; Li, C.; Yong, X.; Kumar, P.; Palma, B.; Hu, Z.; Tendeloo, G.; Siahrostami, S.; Larter, S.; Zheng, D.; et al. Coproduction of hydrogen and lactic acid from glucose photocatalysis on band-engineered Zn1-xCdxS homojunction. iScience 2021, 24, 102109. [Google Scholar] [CrossRef]
- Bellardita, M.; García-López, E.I.; Marcì, G.; Megna, B.; Pomilla, F.R.; Palmisano, L. Photocatalytic conversion of glucose in aqueous suspensions of heteropolyacid-TiO2 composites. RSC Adv. 2015, 5, 59037. [Google Scholar] [CrossRef]
- Kuehnel, M.F.; Reisner, E. Solar Hydrogen Generation from Lignocellulose. Angew. Chem. Int. Ed. 2018, 57, 3290–3296. [Google Scholar] [CrossRef]
- Uekert, T.; Pichler, C.M.; Schubert, T.; Reisner, E. Solar-driven reforming of solid waste for a sustainable future. Nat. Sustain. 2021, 4, 383–391. [Google Scholar] [CrossRef]
- Muhmood, T.; Xia, M.; Lei, W.; Wang, F. Erection of duct-like graphitic carbon nitride with enhanced photocatalytic activity for ACB photodegradation. J. Phys. D Appl. Phys. 2018, 51, 065501. [Google Scholar] [CrossRef]
- Zhurenok, A.V.; Vasilchenko, D.B.; Kozlova, E.A. Comprehensive Review on g-C3N4-Based Photocatalysts for the Photocatalytic Hydrogen Production under Visible Light. Int. J. Mol. Sci. 2023, 24, 346. [Google Scholar] [CrossRef] [PubMed]
- Alaghmandfard, A.; Ghandi, K. A Comprehensive Review of Graphitic Carbon Nitride (g-C3N4)-Metal Oxide-Based Nanocomposites: Potential for Photocatalysis and Sensing. Nanomaterials 2022, 12, 294. [Google Scholar] [CrossRef] [PubMed]
- Goettmann, F.; Fischer, A.; Antonietti, M.; Thomas, A. Metal-free catalysis of sustainable Friedel-Crafts reactions: Direct activation of benzene by carbon nitrides to avoid the use of metal chlorides and halogenated compounds. Chem. Commun. 2006, 5, 4530–4532. [Google Scholar] [CrossRef]
- Wang, X.C.; Maeda, K.; Thomas, A.; Takanabe, K.; Xin, G.; Domen, K.; Antonietti, M. A metal-free polymeric photocatalyst for hydrogen production from water under visible light. Nat. Mater. 2009, 8, 76–80. [Google Scholar] [CrossRef]
- Hollmann, D.; Karnahl, M.; Tschierlei, S.; Kailasam, K.; Schneider, M.; Radnik, J.; Grabow, K.; Bentrup, U.; Junge, H.; Beller, M.; et al. Structure-Activity Relationships in Bulk Polymeric and Sol–Gel-Derived Carbon Nitrides during Photocatalytic Hydrogen Production. Chem. Mater. 2014, 26, 1727–1733. [Google Scholar] [CrossRef]
- Cui, Y.J.; Ding, Z.X.; Liu, P.; Antonietti, M.; Fu, X.Z.; Wang, X.C. Metal-free activation of H2O2 by g-C3N4 under visible light irradiation for the degradation of organic pollutants. Phys. Chem. Chem. Phys. 2012, 14, 1455–1462. [Google Scholar] [CrossRef] [PubMed]
- Niu, P.; Zhang, L.; Liu, G.; Cheng, H. Graphene-like carbon nitride nanosheets for improved photocatalytic activities. Adv. Funct. Mater. 2012, 22, 4763–4770. [Google Scholar] [CrossRef]
- Wen, J.; Xie, J.; Chen, X.; Li, X. A review on g-C3N4-based photocatalysts. Appl. Surf. Sci. 2017, 391, 72–123. [Google Scholar] [CrossRef]
- Ong, W.J.; Tan, L.L.; Ng, Y.H.; Yong, S.T.; Chai, S.P. Graphitic Carbon Nitride (g-C3N4)-Based Photocatalysts for Artificial Photosynthesis and Environmental Remediation: Are We a Step Closer To Achieving Sustainability? Chem. Rev. 2016, 116, 7159–7329. [Google Scholar] [CrossRef]
- Sui, Y.; Liu, J.; Zhang, Y.; Tian, X.; Chen, W. Dispersed conductive polymer nanoparticles on graphitic carbon nitride for enhanced solar-driven hydrogen evolution from pure water. Nanoscale 2013, 5, 9150–9155. [Google Scholar] [CrossRef]
- Liu, J.; Liu, Y.; Liu, N.; Han, Y.; Zhang, X.; Huang, H.; Lifshitz, Y.; Lee, S.; Zhong, J.; Kang, Z. Metal-free efficient photocatalyst for stable visible water splitting via a two-electron pathway. Science 2015, 347, 970–974. [Google Scholar] [CrossRef]
- Naseri, A.; Samadi, M.; Pourjavadi, A.; Moshfegh, A.Z.; Ramakrishna, S. Graphitic carbon nitride (g-C3N4)-based photocatalysts for solar hydrogen generation: Recent advances and future development directions. J. Mater. Chem. A 2017, 5, 23406–23433. [Google Scholar] [CrossRef]
- Cao, S.; Low, J.; Yu, J.; Jaroniec, M. Polymeric photocatalysts based on graphitic carbon nitride. Adv. Mater. 2015, 27, 2150–2176. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Wang, D.; Han, H.; Li, C. Roles of cocatalysts in photocatalysis and photoelectrocatalysis. Acc. Chem. Res. 2013, 46, 1900–1909. [Google Scholar] [CrossRef] [PubMed]
- Maeda, K.; Wang, X.; Nishihara, Y.; Lu, D.; Antonietti, M.; Domen, K. Photocatalytic activities of graphitic carbon nitride powder for water reduction and oxidation under visible light. J. Phys. Chem. C 2009, 113, 4940–4947. [Google Scholar] [CrossRef]
- Battula, V.R.; Jaryal, A.; Kailasam, K. Visible light-driven simultaneous H2 production by water splitting coupled with selective oxidation of HMF to DFF catalyzed by porous carbon nitride. J. Mater. Chem. A 2019, 7, 5643–5649. [Google Scholar] [CrossRef]
- Hou, Y.; Laursen, A.B.; Zhang, J.; Zhang, G.; Zhu, Y.; Wang, X.; Dahl, S.; Chorkendorff, I. Layered nanojunctions for hydrogen-evolution catalysis. Angew. Chem. Int. Ed. 2013, 52, 3621–3625. [Google Scholar] [CrossRef]
- Xiang, Q.; Yu, J.; Jaroniec, M. Preparation and Enhanced Visible-Light Photocatalytic H2-Production Activity of Graphene/C3N4 Composites. J. Phys. Chem. C 2011, 115, 7355–7363. [Google Scholar] [CrossRef]
- Sun, Q.; Wang, P.; Yu, H.G.; Wang, X.F. In situ hydrothermal synthesis and enhanced photocatalytic H2-evolution performance of suspended rGO/g-C3N4 photocatalysts. J. Mol. Catal. A 2016, 424, 369–376. [Google Scholar] [CrossRef]
- Yan, J.Q.; Peng, W.; Zhang, S.S.; Lei, D.P.; Huang, J.H. Ternary Ni2P/reduced graphene oxide/g-C3N4 nanotubes for visible light-driven photocatalytic H2 production. Int. J. Hydrogen Energy 2020, 45, 16094–16104. [Google Scholar] [CrossRef]
- Zhang, X.H.; Peng, B.S.; Zhang, S.; Peng, T.Y. Robust Wide Visible-Light-Responsive Photoactivity for H2 Production over a Polymer/Polymer Heterojunction Photocatalyst: The Significance of Sacrificial Reagent. ACS Sustain. Chem. Eng. 2015, 3, 1501–1509. [Google Scholar] [CrossRef]
- García-López, E.I.; Lo Meo, P.; Megna, B.; Palmisano, L.; Marcì, G. C3N4/reduced graphene oxide based photocatalysts for H2 evolution from aqueous solutions of oxygenated organic molecules. Catal. Today 2022, in press. [Google Scholar] [CrossRef]
- Acharya, R.; Pati, S.; Parida, K. A review on visible light driven spinel ferrite-g-C3N4 photocatalytic systems with enhanced solar light utilization. J. Mol. Liq. 2022, 357, 119105. [Google Scholar] [CrossRef]
- Jo, W.K.; Moru, S.; Tonda, S. Magnetically responsive SnFe2O4/g-C3N4 hybrid photocatalysts with remarkable visible-light-induced performance for degradation of environmentally hazardous substances and sustainable hydrogen production. Appl. Surf. Sci. 2020, 506, 144939. [Google Scholar] [CrossRef]
- Bard, A.J. Photoelectrochemistry and heterogeneous photocatalysis at semiconductors. J. Photochem. 1979, 10, 59–75. [Google Scholar] [CrossRef]
- Zhou, P.; Yu, J.; Jaroniec, M. All-Solid-State Z-Scheme Photocatalytic Systems. Adv. Mater. 2014, 26, 4920–4935. [Google Scholar] [CrossRef]
- Yu, Z.B.; Xie, Y.P.; Liu, G.; Lu, G.Q.; Ma, X.L.; Cheng, H.M. Self-assembled CdS/Au/ZnO heterostructure induced by surface polar charges for efficient photocatalytic hydrogen evolution. J. Mater. Chem. A 2013, 1, 2773–2776. [Google Scholar] [CrossRef]
- Fu, N.; Jin, Z.; Wu, Y.; Lu, G.; Li, D. Z-Scheme Photocatalytic System Utilizing Separate Reaction Centers by Directional Movement of Electrons. J. Phys. Chem. C 2011, 115, 8586–8593. [Google Scholar] [CrossRef]
- Yu, W.; Chen, J.; Shang, T.; Chen, L.; Gu, L.; Peng, T. Direct Z-scheme g-C3N4/WO3 photocatalyst with atomically defined junction for H2 production. Appl. Catal. B 2017, 219, 693–704. [Google Scholar] [CrossRef]
- Idrees, F.; Dillert, R.; Bahnemann, D.; Butt, F.K.; Tahir, M. In-Situ Synthesis of Nb2O5/gC3N4 Heterostructures as Highly Efficient Photocatalysts for Molecular H2 Evolution under Solar Illumination. Catalysts 2019, 9, 169. [Google Scholar] [CrossRef]
- Han, M.; Zhu, S.; Xia, C.; Yang, B. Photocatalytic upcycling of poly(ethylene terephthalate) plastic to high-value chemicals. Applied Catal. B 2022, 316, 121662. [Google Scholar] [CrossRef]
- Uekert, T.; Kasap, H.; Reisner, E. Photoreforming of nonrecyclable plastic waste over a carbon nitride/nickel phosphide catalyst. J. Am. Chem. Soc. 2019, 141, 15201–15210. [Google Scholar] [CrossRef]
- Kasap, H.; Achilleos, D.S.; Huang, A.; Reisner, E. Photoreforming of Lignocellulose into H2 Using Nanoengineered Carbon Nitride under Benign Conditions. J. Am. Chem. Soc. 2018, 140, 11604–11607. [Google Scholar] [CrossRef] [PubMed]
- Uekert, T.; Kuehnel, M.F.; Wakerley, D.W.; Reisner, E. Plastic waste as a feedstock for solar-driven H2 generation. Energy Environ. Sci. 2018, 11, 2853–2857. [Google Scholar] [CrossRef]
- Zakzeski, J.; Bruijnincx, P.C.A.; Jongerius, A.L.; Weckhuysen, B.M. The Catalytic Valorization of Lignin for the Production of Renewable Chemicals. Chem. Rev. 2010, 110, 3552–3599. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.; Kang, F.; Zhu, Y.; Teng, M.; Shi, J.; Qi, H.; Huang, Z.; Si, C.; Jiang, F.; Hu, J. Photoreforming lignocellulosic biomass for hydrogen production: Optimized design of photocatalyst and photocatalytic system. Chem. Eng. J. 2023, 452, 138980. [Google Scholar] [CrossRef]
- Liu, X.; Duan, X.; Wei, W.; Wang, S.; Ni, B.J. Photocatalytic conversion of lignocellulosic biomass to valuable products. Green Chem. 2019, 21, 4266–4289. [Google Scholar] [CrossRef]





Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
García-López, E.I.; Palmisano, L.; Marcì, G. Overview on Photoreforming of Biomass Aqueous Solutions to Generate H2 in the Presence of g-C3N4-Based Materials. ChemEngineering 2023, 7, 11. https://doi.org/10.3390/chemengineering7010011
García-López EI, Palmisano L, Marcì G. Overview on Photoreforming of Biomass Aqueous Solutions to Generate H2 in the Presence of g-C3N4-Based Materials. ChemEngineering. 2023; 7(1):11. https://doi.org/10.3390/chemengineering7010011
Chicago/Turabian StyleGarcía-López, E. I., L. Palmisano, and G. Marcì. 2023. "Overview on Photoreforming of Biomass Aqueous Solutions to Generate H2 in the Presence of g-C3N4-Based Materials" ChemEngineering 7, no. 1: 11. https://doi.org/10.3390/chemengineering7010011
APA StyleGarcía-López, E. I., Palmisano, L., & Marcì, G. (2023). Overview on Photoreforming of Biomass Aqueous Solutions to Generate H2 in the Presence of g-C3N4-Based Materials. ChemEngineering, 7(1), 11. https://doi.org/10.3390/chemengineering7010011







