Catalytic Oxidation of Alkanes and Cycloalkanes: Overview
Abstract
1. Introduction
2. Products of Oxidation of Alkanes and Cycloalkanes
3. Homogeneous Catalysis in the Oxidation of [cyclo-]alkanes
- The catalyst and substrate are in the same phase, most often in solution;
- The catalyst is a complex of transition metals and ligands;
- The reaction involves several molecular stages, each of which controls the selectivity of the process.
- Their C–H bonds have high breaking energy;
- Their molecules do not contain reaction centres;
- They are chemically and thermodynamically stable.
- Low stability of catalysts in an oxidative environment;
- Side processes leading to the formation of acidic and carbon dioxide products;
- Difficulty in controlling selectivity; i.e., many reactions occur in parallel;
- High cost of transition metals;
- Difficulty in transferring laboratory conditions to an industrial scale.
4. Heterogeneous Catalysis in the Oxidation of [cyclo-]alkanes
- High thermal and chemical stability of catalysts—Cr2O3, V2O5, MnO2 oxides, etc., withstand 300–600 °C and aggressive environments.
- Ease of separating the catalyst from the products, as the catalyst is in the solid phase. Hence, it is easy to separate, wash and regenerate.
- Reusability, as solid-phase oxides can operate for many cycles without significant loss of activity.
- Convenience for continuous industrial processes—fixed beds, fluidised beds, tubular reactors—technological simplicity.
- Possibility of fine-tuning surface properties by doping (Cr–Fe, V–Mo–W systems), changing morphology (nanoparticles, porous structures), and choosing a carrier (Al2O3, SiO2, zeolites).
- No need to use solvents (in the gas phase), i.e., environmental friendliness and cost-effectiveness.
- Ability to operate at high pressures and in the presence of oxygen because of the high mechanical strength of oxides.
5. Special Methods for Activating C–H Bonds in Alkanes
5.1. Microseconds Catalysed Oxidation
5.2. Photocatalysis
5.3. Ultrasonic Cavitation
5.4. Biocatalysis and Anaerobic Microbial Oxidation
5.5. Alkane Activation at Ambient Temperatures
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| CNT | Carbon nanotube |
| DFT | Density functional theory |
| ORR | Oxidation–reduction reaction |
| POE | Polyolefin elastomers |
| SWCNT | Single-walled carbon nanotube |
| 1H NMR | Proton nuclear magnetic resonance |
References
- Wang, Y.; Hu, P.; Yang, J.; Zhu, Y.-A.; Chen, D. C–H bond activation in light alkanes: A theoretical perspective. Chem. Soc. Rev. 2021, 50, 4299–4358. [Google Scholar] [CrossRef]
- Shul’pin, G.B.; Shul’pina, L.S. Oxidation of Organic Compounds with Peroxides Catalyzed by Polynuclear Metal Compounds. Catalysts 2021, 11, 186. [Google Scholar] [CrossRef]
- Calvert, J.; Derwent, R.; Orlando, J.; Tyndall, G.; Wallington, T. Mechanisms of Atmospheric Oxidation of the Alkanes, 2nd ed.; Oxford University Press: Oxford, UK, 2023. [Google Scholar] [CrossRef]
- Ryabov, A.Y.; Kudryashov, S.V.; Ocheredko, A.N. Combined Oxidation of Gaseous and Liquid Alkanes in the Barrier Discharge Plasma. Chem. Sustain. Dev. 2024, 32, 190–195. [Google Scholar] [CrossRef]
- Aliyeva, A.Z.; Nuriev, L.G.; Zeynalov, E.B. Fullerene C60/C70 in the catalysis of liquid-phase oxidation of n-decane. Oil Refin. Petrochem. 2010, 11, 16–20. [Google Scholar]
- Aliyeva, A.Z. Identification of petroleum hydrocarbon oxidation products obtained in the presence of fullerene C60/C70 and metal-containing carbon nanotubes. Oil Refin. Petrochem. 2015, 2, 23–26. [Google Scholar]
- Aliyeva, A.Z.; Nuriyev, L.H.; Kuliyev, A.D.; Zeynalov, E.B. Oxidation of n-decane at the joint presence of fullerenes and manganese naphthenate. Process. Petrochem. Oil Refin. 2008, 1, 15–18. [Google Scholar]
- Zeynalov, E.B.; Aliyeva, A.Z.; Friedrich, J.F. Factors Affecting the Intrinsic Anti-Oxidative Activity of Carbon Nanofibers—Metallic Impurities. Mater. Test. 2009, 51, 209–214. [Google Scholar] [CrossRef]
- Kudryashov, S.V.; Ryabov, A.Y.; Shchegoleva, G.S.; Sirotkina, E.E.; Suslov, A.I. Oxidation of n-C5-C8 hydrocarbons and cyclohexane in a barrier discharge reactor. Part I. Experimental investigation results. Proc. Tomsk. Polytech. Univ. 2006, 309, 92–96. [Google Scholar]
- Yiu, S.-M.; Man, W.-L.; Lau, T.-C. Efficient Catalytic Oxidation of Alkanes by Lewis Acid/[OsVI(N)Cl4]−Using Peroxides as Terminal Oxidants Evidence for a Metal-Based Active Intermediate. J. Am. Chem. Soc. 2008, 130, 10821–10827. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhao, L.; Zhang, H.; Huang, R.; Zhao, J. Oxidation of alkanes and secondary alcohols to ketones with tert-butyl hydroperoxide catalyzed by a water-soluble ruthenium complex under solvent-free conditions. Appl. Organometal. Chem. 2017, 31, e3709. [Google Scholar] [CrossRef]
- Komiya, N.; Naota, T.; Oda, Y.; Murahashi, S.-I. Aerobic oxidation of alkanes and alkenes in the presence of aldehydes catalyzed by copper salts and copper-crown ether. J. Mol. Catal. A Chem. 1997, 117, 21–37. [Google Scholar] [CrossRef]
- Pullabhotla, R.V.S.R.; Southway, C.; Jonnalagadda, S.B. Ozone initiated oxidation of long chained n-alkanes in the presence of activated charcoal or silica gel. React. Kinet. Catal. Lett. 2008, 94, 289–299. [Google Scholar] [CrossRef]
- Shul’pin, G.B.; Kozlov, Y.N.; Shul’pina, L.S.; Petrovskiy, P.V. Oxidation of alkanes and alcohols with hydrogen peroxide catalyzed by complex Os3(CO)10(µ-H)2. Appl. Organomet. Chem. 2010, 24, 464–472. [Google Scholar] [CrossRef]
- Suzuki, Y.; Harada, E.; Nakamaru, K.; Takeda, Y.; Sano, M.; Hashimoto, K.; Miyake, T. Direct oxidation of cycloalkanes with molecular oxygen to dicarboxylic acids using isoamyl nitrite. J. Mol. Catal. A Chem. 2007, 276, 1–7. [Google Scholar] [CrossRef]
- Shen, H.M.; Wang, X.; Guo, A.B.; Zhang, L.; She, Y.B. Catalytic oxidation of cycloalkanes by porphyrin cobalt(II) through efficient utilization of oxidation intermediates. J. Porphyr. Phthalocyanines. 2020, 24, 1166–1173. [Google Scholar] [CrossRef]
- Liu, J.; Su, P.; Dai, B.; Zhou, D.; Wang, C. Anomaly Detection Algorithm for Searching Selective Catalyst Differentiating Linear and Cyclic Alkanes in Oxidation. Chin. J. Chem. 2025, 43, 1685–1690. [Google Scholar] [CrossRef]
- Aliyeva, A.Z.; Nuriyev, L.H.; Zeynalov, E.B. Use of multi-walled metal-containing carbon nanotubes as catalyst for oil hydrocarbons aerobic oxidation. In International Conference ‘Functional Materials and Nanotechnologies’; Institute of Solid State Physics, University of Latvia: Riga, Latvia, 2011; p. 273. [Google Scholar]
- Aliyeva, L.I.; Efendiyeva, L.M.; Abbasov, V.M.; Veliyeva, F.M.; Babany, N.N.; Musaly, V.K.H.; Seidahmedova, F.N. Optimisation of deep aerobic oxidation of petroleum naphthenic-paraffin fraction. Oil Refin. Petrochem. 2018, 7, 41–46. [Google Scholar]
- Zeynalov, E.; Friedrich, J.; Meyer-Plath, A.; Hidde, G.; Nuriyev, L.; Aliyeva, A.; Cherepnova, Y. Plasma-chemically brominated single-walled carbon nanotubes as novel catalysts for oil hydrocarbons aerobic oxidation. Appl. Catal. A Gen. 2013, 454, 115–118. [Google Scholar] [CrossRef]
- Aliyeva, A.Z.; Abbasov, V.M.; Ibragimov, K.D.; Ismailov, E.G.; Nuriev, L.G.; Nasibova, G.G.; Suleimanova, S.A.; Musaeva, A.P. Liquid-Phase Oxidation of Oil Hydrocarbons in the Presence of Nanostructured Mn-Containing Catalyst. Pet. Chem. 2018, 58, 573–576. [Google Scholar] [CrossRef]
- Suleymanova, R.H.; Zeynalov, N.A.; Badalova, O.T.; Qulubayova, L.N.; Sharifzade, N.F.; Quliyeva, A.R. Liquid-phase oxidation of aliphatic hydrocarbons in a presence of metal-polymer complexes. Azərbaycan Kim. Jurnali 2016, 4, 80–84. [Google Scholar]
- Mao, S.; Verspeek, D.; Wen, X.-D.; Yang, Y.; Li, Y.-W.; Junge, K.; Beller, M. Homogeneous Iron-Catalysed Oxidation of Non-Activated Alkanes with Hydrogen Peroxide. ChemCatChem 2023, 15, e202300735. [Google Scholar] [CrossRef]
- Chepaikin, E.G. Homogeneous catalysis in the oxidative functionalization of alkanes in protic media. Russ. Chem. Rev. 2011, 80, 384–416. [Google Scholar] [CrossRef]
- Chen, M.; Pan, Y.; Kwong, H.-K.; Zeng, R.J.; Lau, K.-C.; Lau, T.-C. Catalytic oxidation of alkanes by a (salen)osmium(VI) nitrido complex using H2O2 as the terminal oxidant. Chem. Commun. 2015, 51, 13686–13689. [Google Scholar] [CrossRef]
- Garcia-Bosch, I.; Siegler, M.A. Copper-Catalyzed Oxidation of Alkanes with H2O2 under a Fenton-like Regime. Angew. Chem. 2016, 55, 12873–12876. [Google Scholar] [CrossRef]
- Komiya, N.; Naota, T.; Murahashi, S.-I. Aerobic oxidation of alkanes in the presence of acetaldehyde catalysed by copper-crown ether. Tetrahedron Lett. 1996, 37, 1633–1636. [Google Scholar] [CrossRef]
- Takaki, K.; Yamamoto, J.; Matsushita, Y.; Morii, H.; Shishido, T.; Takehira, K. Oxidation of Alkanes with Dioxygen Induced by Visible Light and Cu(II) and Fe(III) Chlorides. Bull. Chem. Soc. Jpn. 2003, 76, 393–398. [Google Scholar] [CrossRef]
- Kozlov, Y.N.; Nizova, G.V.; Shul’pin, G.B. The mechanism of hydrogen peroxide-induced aerobic oxidation of alkanes in catalysis by a vanadium complex and pyrazine-2-carboxylic acid. Russ. J. Phys. Chem. 2001, 75, 770–774. [Google Scholar]
- Li, Z.; Xia, C.-G.; Xu, C.-Z. Oxidation of alkanes catalyzed by manganese(III) porphyrin in an ionic liquid at room temperature. Tetrahedron Lett. 2003, 44, 9229–9232. [Google Scholar] [CrossRef]
- Sinev, M.Y. Oxygen activation and pathways in high-temperature reactions of light alkane oxidation: A seeming simplicity of kinetic description. Kinet. Catal. 2019, 60, 420–431. [Google Scholar] [CrossRef]
- Kerimova, U.A. Reaction mechanism of liquid-phase aerobic oxidation of hydrocarbons in the presence of metal-containing catalysts. Oil Gas Technol. 2023, 145, 18–21. [Google Scholar] [CrossRef]
- Zeynalov, E. Academician Bahadur Zeynalov. His Life Journey as a Constant Exploration and Advancement Towards the Pinnacles of Science; Muallim: Baku, Azərbaycan, 2017. [Google Scholar]
- Zeynalov, E.B.; Abbasov, V.M.; Efendiyeva, L.M.; Nuriyev, L.H.; Agamaliyev, Z.Z.; Aliyeva, L.I.; Nasibova, G.Q. Catalytic activity of naphthenates of the transition metals in reaction aerobic oxidation of an oil fraction. World Pet. Prod. Newsl. Oil Co. 2012, 2, 23–26. [Google Scholar]
- Su, Y.; Li, Y.; Chen, Z.; Huang, J.; Wang, H.; Yu, H.; Cao, Y.; Peng, F. New Understanding of Selective Aerobic Oxidation of Ethylbenzene Catalyzed by Nitrogen-doped Carbon Nanotubes. ChemCatChem 2021, 13, 646–655. [Google Scholar] [CrossRef]
- Aliyeva, A.Z.; Karimova, U.A.; Yunusov, S.H.; Kayahan, E.; Agayev, U.I.; Zamanova, L.S. Liquid-phase oxidation of paraffin concentrate of the boiling fraction at 135–360 °C using ultrasonically treated fullerene soot. SOCAR Proc. 2025, 4, 240–246. [Google Scholar]
- Shul’pin, G.B.; Kozlov, Y.N.; Shul’pina, L.S. Metal Complexes Containing Redox-Active Ligands in Oxidation of Hydrocarbons and Alcohols: A Review. Catalysts 2019, 9, 1046. [Google Scholar] [CrossRef]
- Levitsky, M.M.; Bilyachenko, A.N.; Shul’pin, G.B. Oxidation of C-H compounds with peroxides catalyzed by polynuclear transition metal complexes in Si-or Ge-sesquioxane frameworks: A review. J. Organomet. Chem. 2017, 849–850, 201–218. [Google Scholar] [CrossRef]
- Kulakova, A.N.; Khrustalev, V.N.; Zubavichus, Y.V.; Shul’pina, L.S.; Shubina, E.S.; Levitsky, M.M.; Ikonnikov, N.S.; Bilyachenko, A.N.; Kozlov, Y.N.; Shul’pin, G.B. Palanquin-Like Cu4Na4 Silsesquioxane Synthesis (via Oxidation of 1,1-bis(Diphenylphosphino)methane), Structure and Catalytic Activity in Alkane or Alcohol Oxidation with Peroxides. Catalysts 2019, 9, 154. [Google Scholar] [CrossRef]
- Buvaylo, E.A.; Kokozay, V.N.; Vassilyeva, O.Y.; Skelton, B.W.; Nesterova, O.V.; Pombeiro, A.J.L. Copper(II) complex of the 2-pyridinecarbaldehyde aminoguanidine Schiff base: Crystal structure and catalytic behaviour in mild oxidation of alkanes. Inorg. Chem. Commun. 2017, 78, 85–90. [Google Scholar] [CrossRef]
- Li, C.; Zhang, X. Advances in Metal Complex-Catalyzed Oxidation of Alkanes. Sci. J. Technol. 2025, 7, 206–213. [Google Scholar] [CrossRef]
- Zheng, R.; Liu, Z.; Wang, Y.; Xie, Z. Industrial catalysis: Strategies to enhance selectivity. Chin. J. Catal. 2020, 41, 1032–1038. [Google Scholar] [CrossRef]
- Yao, Y.-F.Y. Oxidation of Alkanes over Noble Metal Catalysts. Ind. Eng. Chem. Prod. Res. Dev. 1980, 19, 293–298. [Google Scholar] [CrossRef]
- Vigdorowitsch, M. A surface-induced modification of the reaction order on a reactant. Colloids Surf. A Physicochem. Eng. Asp. 2025, 718, 136878. [Google Scholar] [CrossRef]
- Solsona, B.E.; Garcia, T.; Jones, C.; Taylor, S.H.; Carley, A.F.; Hutchings, G.J. Supported gold catalysts for the total oxidation of alkanes and carbon monoxide. Appl. Catal. A Gen. 2006, 312, 67–76. [Google Scholar] [CrossRef]
- Khouw, C.B.; Dartt, C.B.; Labinger, J.A.; Davis, M.E. Studies on the Catalytic-Oxidation of Alkanes and Alkenes by Titanium Silicates. J. Catal. 1994, 149, 195–205. [Google Scholar] [CrossRef]
- Clerici, M.G. Oxidation of saturated hydrocarbons with hydrogen peroxide, catalysed by titanium silicalite. Appl. Catal. 1991, 68, 249–261. [Google Scholar] [CrossRef]
- Nagarjun, N.; Dhakshinamoorthy, A. Liquid phase aerobic oxidation of cyclic and linear hydrocarbons using iron metal organic frameworks as solid heterogeneous catalyst. Mol. Catal. 2019, 463, 54–60. [Google Scholar] [CrossRef]
- Shibata, S.; Sugahara, K.; Kamata, K.; Hara, M. Liquid-phase oxidation of alkanes with molecular oxygen catalyzed by high valent iron-based perovskite. Chem. Commun. 2018, 54, 6772–6775. [Google Scholar] [CrossRef]
- Yamaguchia, K.; Mizuno, N. Heterogeneously catalyzed liquid-phase oxidation of alkanes and alcohols with molecular oxygen. New J. Chem. 2002, 26, 972–974. [Google Scholar] [CrossRef]
- Kholdeeva, O.A. Recent developments in liquid-phase selective oxidation using environmentally benign oxidants and mesoporous metal silicates. Catal. Sci. Technol. 2014, 4, 1869–1889. [Google Scholar] [CrossRef]
- Lunin, A.V.; Danov, S.М.; Fedosov, А.Е.; Fedosova, М.Е. Kinetics of liquid-phase oxidation of n-alkanes by hydrogen peroxide on the heterogeneous catalyst DP-1. Fine Chem. Technol. 2014, 9, 59–63. [Google Scholar]
- Labinger, J.A.; Bercaw, J.E. Understanding and exploiting C–H bond activation. Nature 2002, 417, 507–514. [Google Scholar] [CrossRef]
- Chen, L.; Su, X.-J.; Jurss, J.W. Selective Alkane C–H Bond Oxidation Catalyzed by a Non-Heme Iron Complex Featuring a Robust Tetradentate Ligand. Organometallics 2018, 37, 4535–4539. [Google Scholar] [CrossRef]
- Hong, C.E.; Oh, M.S.; An, S.; Song, K.H.; Lee, S. Cobalt-Catalyzed C-H Oxidation of Alkanes and Postfunctionalization of Polyolefin Elastomers. Chem. Eur. J. 2025, 31, e202500510. [Google Scholar] [CrossRef]
- Rogge, T.; Kaplaneris, N.; Chatani, N.; Kim, J.; Chang, S.; Punji, B.; Schafer, L.L.; Musaev, D.G.; Wencel-Delord, J.; Roberts, C.A.; et al. C–H activation. Nat. Rev. Methods Primers 2021, 1, 43. [Google Scholar] [CrossRef]
- Goetsch, D.A.; Schmidt, L.D. Microsecond Catalytic Partial Oxidation of Alkanes. Science 1996, 271, 1560–1562. [Google Scholar] [CrossRef]
- Anikin, N.B.; Starikovskaia, S.M.; Starikovskii, A.Y. Oxidation of saturated hydrocarbons under the effect of nanosecond pulsed space discharge. J. Phys. D Appl. Phys. 2006, 39, 3244–3258. [Google Scholar] [CrossRef]
- Coutard, N.; Goldberg, J.M.; Valle, H.U.; Cao, Y.; Jia, X.; Jeffrey, P.D.; Gunnoe, T.B.; Groves, J.T. Aerobic Partial Oxidation of Alkanes Using Photodriven Iron Catalysis. Inorg. Chem. 2022, 61, 759–766. [Google Scholar] [CrossRef] [PubMed]
- Amaniampong, P.N.; Jérôme, F. Catalysis under ultrasonic irradiation: A sound synergy. Curr. Opin. Green Sustain. Chem. 2020, 22, 7–12. [Google Scholar] [CrossRef]
- Soudagar, S.R.; Samant, S.D. Investigation of ultrasound catalyzed oxidation of arylalkanes using aqueous potassium permanganate. Ultrason. Sonochem. 1995, 2, S15–S18. [Google Scholar] [CrossRef]
- Aliyeva, A.Z.; Kerimova, U.A.; Yunusov, S.G.; Aliyeva, A.A.; Aleskerova, S.M.; Tagiyev, F.F. Ultrasound in Catalysis. Chem. Sustain. Dev. 2024, 32, 258–263. [Google Scholar] [CrossRef]
- Abbasov, V.M.; Aliyeva, A.Z.; Kerimova, U.A.; Kayahan, E.; Yunusov, S.G.; Mamedov, A.M.; Zamanova, L.S. Use of ultrasonic pretreatment in the reaction of liquid aerobic oxidation of C11, C14 paraffin hydrocarbons in the presence of (RCOO)3CR and (RCOO)2NI catalysts. Iran. J. Chem. Chem. Eng. 2024, 43, 707–713. [Google Scholar] [CrossRef]
- Singh, R.; Guzman, M.S.; Bose, A. Anaerobic Oxidation of Ethane, Propane, and Butane by Marine Microbes: A Mini Review. Front. Microbiol. 2017, 8, 2056. [Google Scholar] [CrossRef] [PubMed]
- Chan, S.I.; Yu, S.S.-F.; Liu, C.-C.; Mou, C.-Y. Selective oxidation of light alkanes under mild conditions. Curr. Opin. Green Sustain. Chem. 2020, 22, 39–46. [Google Scholar] [CrossRef]
- Trionfetti, C.; Ağiral, A.; Gardeniers, H.J.G.E.; Lefferts, L.; Seshan, K. Alkane activation at ambient temperatures: Unusual selectivities, C-C, C-H bond scission versus C-C bond coupling. ChemPhysChem 2008, 9, 533–537. [Google Scholar] [CrossRef] [PubMed]
- Abbasov, V.M.; Ibragimov, K.D.; Dzhafarov, R.P.; Kerimova, U.A.; Alieva, A.Z. Mathematical description of the aerobic oxidation process of n-undcan un the presence of chromium salt treated with ultrasound. Oil Refin. Petrochem. 2020, 9, 19–23. Available online: https://www.elibrary.ru/item.asp?id=44400164 (accessed on 26 January 2026).
- Büker, J.; Huang, X.; Bitzer, J.; Kleist, W.; Muhler, M.; Peng, B. Synthesis of Cu Single Atoms Supported on Mesoporous Graphitic Carbon Nitride and Their Application in Liquid-Phase Aerobic Oxidation of Cyclohexene. ACS Catal. 2021, 11, 7863–7875. [Google Scholar] [CrossRef]
- Sutradhar, M.; Alegria, E.C.B.A.; Guedes da Silva, M.F.C.; Liu, C.-M.; Pombeiro, A.J.L. Peroxidative Oxidation of Alkanes and Alcohols under Mild Conditions by Di- and Tetranuclear Copper (II) Complexes of Bis (2-Hydroxybenzylidene) Isophthalohydrazide. Molecules 2018, 23, 2699. [Google Scholar] [CrossRef] [PubMed]
- Cai, Z.; Liu, D.; Huang, J.; Feng, J.; Wang, H.; Yang, G.; Peng, F.; Cao, Y.; Yu, H. Solvent-free production of ε-caprolactone from oxidation of cyclohexanone catalyzed by nitrogen-doped carbon nanotubes. Ind. Eng. Chem. Res. 2022, 61, 2037–2044. [Google Scholar] [CrossRef]
- Kunene, A.; Leteba, G.; van Steen, E. Liquid Phase Oxidation of Benzyl Alcohol over Pt and Pt–Ni Alloy Supported on TiO2: Using O2 or H2O2 as Oxidant? Catal. Lett. 2022, 152, 1760–1768. [Google Scholar] [CrossRef]
- Chen, L.; Zhang, X.; Zhang, S.; Xu, L.; Yuan, Y.; Xu, L. A highly efficient Fe/Beta catalyst for the liquid-phase oxidation of naphthalene: The influence of Fe species and zeolite acidity. Mol. Catal. 2021, 514, 111646. [Google Scholar] [CrossRef]
- Mishra, S.; Sangma, S.W.; Bal, R.; Dey, R.K. Preparation and characterization of a copper oxide nanoparticle-supported red-mud catalyst for liquid phase oxidation of ethyl benzene to acetophenone. New J. Chem. 2021, 45, 13070–13079. [Google Scholar] [CrossRef]
- Zeynalov, E.B.; Huseynov, E.R.; Salmanova, N.I.; Abdurahmanova, N.A. Nano-grade titanium dioxide on the nanocarbon carrier as catalyst in aerobic oxidation of alkyl aromatic hydrocarbons. Chem. Probl. 2020, 18, 351–360. [Google Scholar] [CrossRef]
- Shul’pina, L.S.; Vinogradov, M.M.; Kozlov, Y.N.; Nelyubina, Y.V.; Ikonnikov, N.S.; Shul’pin, G.B. Copper complexes with 1,10-phenanthrolines as efficient catalysts for oxidation of alkanes by hydrogen peroxide. Inorg. Chim. Acta 2020, 512, 119889. [Google Scholar] [CrossRef]
- Cousin, T.; Chatel, G.; Kardos, N.; Andrioletti, B.; Draye, M. Recent trends in the development of sustainable catalytic systems for the oxidative cleavage of cycloalkenes by hydrogen peroxide. Catal. Sci. Technol. 2019, 9, 5256–5278. [Google Scholar] [CrossRef]
- Ma, Z.; Gurbanov, A.V.; Maharramov, A.M.; Guseinov, F.I.; Kopylovich, M.N.; Zubkov, F.I.; Mahmudov, K.T.; Pombeiro, A.J.L. Copper(II) arylhydrazone complexes as catalysts for CH activation in the Henry reaction in water. J. Mol. Catal. A Chem. 2017, 426, 526–533. [Google Scholar] [CrossRef]
- Ryabov, A.Y.; Kudryashov, S.V. Oxidation of gaseous alkanes into hydroxyl and carbonyl compounds. ChemChemTech 2025, 68, 84–91. [Google Scholar] [CrossRef]
- Isazade, A.F.; Mammadova, U.A.; Asadov, M.M.; Mammadova, S.A.; Zeynalov, N.A.; Tagiyev, D.B. Description of initial reactions in kinetic modeling of low-temperature oxidation of n-alkanes. Azerbaijan Chem. J. 2025, 3, 71–80. [Google Scholar] [CrossRef]






















| 5-ethyl dihydro 2(3H)-furanone (C6H10O2): | : |
| 5-propyl dihydro 2(3H)-furanone (C7H12O2) | |
| 5-pentyl-2(3H)-furanone (C9H16O2) | |
| 5-hexyl-2(3H)-furanone (C10H18O2) | |
4-hexadecyl ester of hexanoic acid (C22H44O2) | Caprylic anhydride (C16H30O3) |
5-undecanol (C11H24O) | 1-octanol, 2-butyl (C12H26O) |
2-tridecyl ester of octanoic acid (C21H42O2)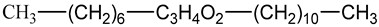 | : |
2-tridecyl ester of valeric acid (C18H36O2)  | |
nonane, 5-methyl, 5-propyl (C13H28) | 4-octyl ester of heptanoic acid (C15 H30O2) |
3,6-nonadecadione (C19H36O2) | Nonenoic acid |
Dodecane (C12H26) | Heptanoic acid (C7H14O2) |
2-undecanol (C11H24O):  | : |
2-undecanone (C11H22O):  | |
5-undecanone, 2-methyl (C12H24O):  | : |
7-tridecanone (C13H26O):  | |
3-undecanol (C11H24O):  | : |
3-undecanone (C11H22O):  | |
1,1-dimethoxy octadecan (C20H42O2) | 2,3-ethyl, 5-butyl octadecan (C26H54) |
2,3-heptadecadione (C17H32O) | 8-pentadecanone (C15H30O) |
| Catalyst Type | Typical Active Species | Main Mechanism | Advantages | Disadvantages |
|---|---|---|---|---|
| Metal complexes | Fe, Cu, Ru, Os, Mn complexes | Radical or metal–oxo pathways | High activity, tunable selectivity, mechanistic insight | Low stability, difficult separation, high cost |
| Metalloporphyrins | Co, Mn porphyrins | Controlled radical oxidation | High selectivity, biomimetic | Limited lifetime, oxidative degradation |
| Metal oxides | V2O5, Cr2O3, MnO2, Fe-based catalysts | Mars– van Krevelen | Thermal stability, industrial applicability | Tendency to deep oxidation |
| Zeolites (TS-1) | Ti–O sites | Peroxide activation | Mild conditions, H2O2 compatibility | Diffusion limitations |
| Carbon nanomaterials | CNTs, fullerenes | Radical initiation, O2 activation | Enhanced mass transfer, stability | Mechanism complexity |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Aliyeva, A.Z.; Karimova, U.A.; Yunusov, S.G.; Vigdorowitsch, M.; Mammadkhanova, S.A. Catalytic Oxidation of Alkanes and Cycloalkanes: Overview. ChemEngineering 2026, 10, 25. https://doi.org/10.3390/chemengineering10020025
Aliyeva AZ, Karimova UA, Yunusov SG, Vigdorowitsch M, Mammadkhanova SA. Catalytic Oxidation of Alkanes and Cycloalkanes: Overview. ChemEngineering. 2026; 10(2):25. https://doi.org/10.3390/chemengineering10020025
Chicago/Turabian StyleAliyeva, Aygun Zabit, Ulviyya Aliman Karimova, Sahib Gadji Yunusov, Michael Vigdorowitsch, and Sevinj Abdulhamid Mammadkhanova. 2026. "Catalytic Oxidation of Alkanes and Cycloalkanes: Overview" ChemEngineering 10, no. 2: 25. https://doi.org/10.3390/chemengineering10020025
APA StyleAliyeva, A. Z., Karimova, U. A., Yunusov, S. G., Vigdorowitsch, M., & Mammadkhanova, S. A. (2026). Catalytic Oxidation of Alkanes and Cycloalkanes: Overview. ChemEngineering, 10(2), 25. https://doi.org/10.3390/chemengineering10020025







