Promising Recent Strategies with Potential Clinical Translational Value to Combat Antibacterial Resistant Surge
Abstract
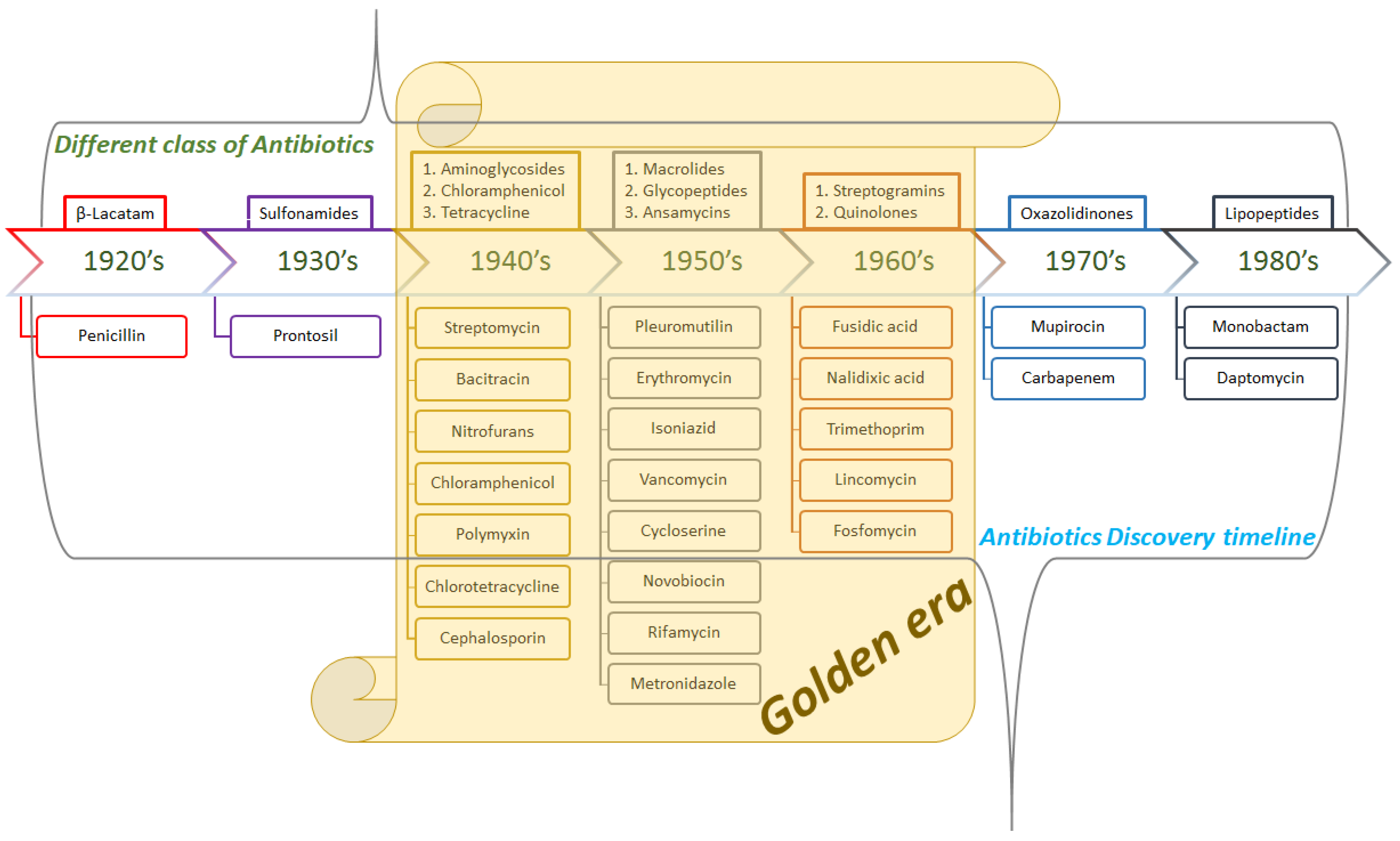
1. Introduction
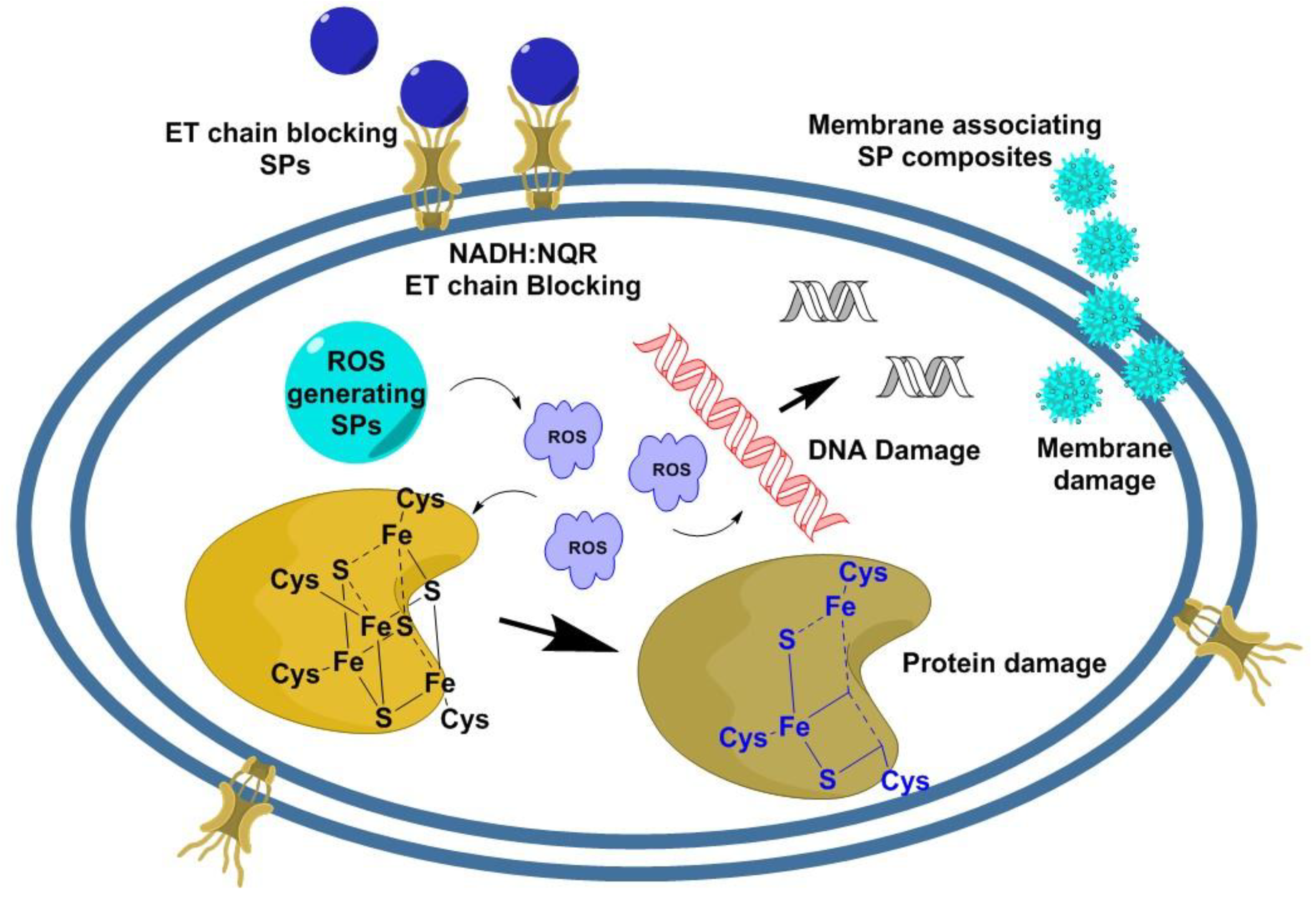
2. Mechanism of Action of the SPs
3. Small Particles as Delivery Vehicles for Small Molecule Antibiotics
3.1. Bio-Mimetic and Bio-Compatible SPs for Targeted Antibacterial Drug Delivery Strategies
3.2. Bio-Mimetic and Bio-Compatible SPs for Immune System-Targeted Antibacterial Drug Delivery Strategies
3.3. Small Particle Composites with Externally Triggerable Drug Release Mechanisms
3.4. Bio-Mimetic and Bio-Compatible SPs for Immune System-Targeted Antibacterial Drug Delivery Strategies with Externally Triggerable Drug Release Mechanisms
4. Small Particles as Antibiotics
4.1. Small Particle Applications in Wound Healing
4.2. Small Particles in Artificial Implants
4.3. Small Particle–Small Molecule Dual Functional Antibiotics; the Ultimate Weapon
5. Challenges and Prospects
6. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Fleming, A. On the antibacterial action of cultures of a penicillium, with special reference to their use in the isolation of b. Influenzae. Br. J. Exp. Pathol. 1929, 10, 226. [Google Scholar] [CrossRef]
- Brown, E.D.; Wright, G.D. Antibacterial drug discovery in the resistance era. Nature 2016, 529, 336. [Google Scholar] [CrossRef]
- World Health Organization. World Malaria Report 2014; World Health Organization: Geneva, Switzerland, 2015. [Google Scholar]
- World Health Organization. Global Antimicrobial Resistance Surveillance System (Glass) Report: Early Implementation 2016–2017; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- Fischbach, M.A.; Walsh, C.T. Antibiotics for emerging pathogens. Science 2009, 325, 1089–1093. [Google Scholar] [CrossRef] [PubMed]
- Levy, S.B.; Marshall, B. Antibacterial resistance worldwide: Causes, challenges and responses. Nat. Med. 2004, 10, S122. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.; Uehara, T.; Bernhardt, T.G. Beta-lactam antibiotics induce a lethal malfunctioning of the bacterial cell wall synthesis machinery. Cell 2014, 159, 1300–1311. [Google Scholar] [CrossRef] [PubMed]
- Maranan, M.C.; Moreira, B.; Boyle-Vavra, S.; Daum, R.S. Antimicrobial resistance in staphylococci: Epidemiology, molecular mechanisms, and clinical relevance. Infect. Dis. Clin. N. Am. 1997, 11, 813–849. [Google Scholar] [CrossRef]
- Wilson, D.N. Ribosome-targeting antibiotics and mechanisms of bacterial resistance. Nat. Rev. Microbiol. 2014, 12, 35. [Google Scholar] [CrossRef] [PubMed]
- Demirci, H.; Murphy, F., IV; Murphy, E.; Gregory, S.T.; Dahlberg, A.E.; Jogl, G. A structural basis for streptomycin-induced misreading of the genetic code. Nat. Commun. 2013, 4, 1355. [Google Scholar] [CrossRef]
- Abraham, E.P.; Chain, E. An enzyme from bacteria able to destroy penicillin. Nature 1940, 146, 837. [Google Scholar] [CrossRef]
- Infectious Diseases Society of America. The 10×’20 initiative: Pursuing a global commitment to develop 10 new antibacterial drugs by 2020. Clin. Infect. Dis. 2010, 50, 1081–1083. [Google Scholar] [CrossRef]
- Laxminarayan, R.; Brown, G.M. Economics of antibiotic resistance: A theory of optimal use. J. Environ. Econ. Manag. 2001, 42, 183–206. [Google Scholar] [CrossRef]
- Coates, A.; Hu, Y.; Bax, R.; Page, C. The future challenges facing the development of new antimicrobial drugs. Nat. Rev. Drug Discov. 2002, 1, 895. [Google Scholar] [CrossRef] [PubMed]
- Sköld, O. Antibiotics and Antibiotic Resistance; John Wiley & Sons: Hoboken, NJ, USA, 2011. [Google Scholar]
- Davies, J.; Davies, D. Origins and evolution of antibiotic resistance. Microbiol. Mol. Biol. Rev. 2010, 74, 417–433. [Google Scholar] [CrossRef] [PubMed]
- Amyes, S. The rise in bacterial resistance: Is partly because there have been no new classes of antibiotics since the 1960s. Br. Med. J. 2000, 320, 199. [Google Scholar] [CrossRef]
- Vadlapatla, R.K.; Vadlapudi, A.D.; Kwatra, D.; Pal, D.; Mitra, A.K. Differential effect of p-gp and mrp2 on cellular translocation of gemifloxacin. Int. J. Pharm. 2011, 420, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Khan, F.Z.; Nawaz, T.; Mirani, Z.A.; Khan, S.; Raza, Y.; Kazmi, S.U. Study of class 1 integrons in multidrug-resistant uropathogenic escherichia coli isolated from different hospitals in karachi. Iran. J. Basic Med. Sci. 2018, 21, 1079. [Google Scholar]
- Rostkowska, K.A.; Szymanek-Pasternak, A.; Simon, K.A. Spontaneous bacterial peritonitis–therapeutic challenges in the era of increasing drug resistance of bacteria. Clin. Exp. Hepatol. 2018, 4, 224. [Google Scholar] [CrossRef]
- Cottell, J.L.; Webber, M.A. Experiences in fosfomycin susceptibility testing and resistance mechanism determination in escherichia coli from urinary tract infections in the UK. J. Med. Microbiol. 2018. [Google Scholar] [CrossRef]
- Ge, J.; Yang, T.; Zhang, L.; Zhang, X.; Zhu, X.; Tang, B.; Wan, X.; Tong, J.; Song, K.; Yao, W. The incidence, risk factors and outcomes of early bloodstream infection in patients with malignant hematologic disease after unrelated cord blood transplantation: A retrospective study. BMC Infect. Dis. 2018, 18, 654. [Google Scholar] [CrossRef]
- Giske, C.G.; Monnet, D.L.; Cars, O.; Carmeli, Y. Clinical and economic impact of common multidrug-resistant gram-negative bacilli. Antimicrob. Agents Chemother. 2008, 52, 813–821. [Google Scholar] [CrossRef]
- Czaplewski, L.; Bax, R.; Clokie, M.; Dawson, M.; Fairhead, H.; Fischetti, V.A.; Foster, S.; Gilmore, B.F.; Hancock, R.E.; Harper, D. Alternatives to antibiotics—A pipeline portfolio review. Lancet Infect. Dis. 2016, 16, 239–251. [Google Scholar] [CrossRef]
- Payne, D.J.; Gwynn, M.N.; Holmes, D.J.; Pompliano, D.L. Drugs for bad bugs: Confronting the challenges of antibacterial discovery. Nat. Rev. Drug Discov. 2007, 6, 29. [Google Scholar] [CrossRef] [PubMed]
- Coates, A.R.; Halls, G.; Hu, Y. Novel classes of antibiotics or more of the same? Br. J. Pharmacol. 2011, 163, 184–194. [Google Scholar] [CrossRef] [PubMed]
- Okeke, I.N.; Peeling, R.W.; Goossens, H.; Auckenthaler, R.; Olmsted, S.S.; de Lavison, J.-F.; Zimmer, B.L.; Perkins, M.D.; Nordqvist, K. Diagnostics as essential tools for containing antibacterial resistance. Drug Resist. Updat. 2011, 14, 95–106. [Google Scholar] [CrossRef]
- McEwen, S.A.; Fedorka-Cray, P.J. Antimicrobial use and resistance in animals. Clin. Infect. Dis. 2002, 34, S93–S106. [Google Scholar] [CrossRef] [PubMed]
- Goossens, H.; Ferech, M.; Vander Stichele, R.; Elseviers, M.; Group, E.P. Outpatient antibiotic use in europe and association with resistance: A cross-national database study. Lancet 2005, 365, 579–587. [Google Scholar] [CrossRef]
- Witte, W. Medical Consequences of Antibiotic Use in Agriculture; American Association for the Advancement of Science: Washington, DC, USA, 1998. [Google Scholar]
- Garau, J.; Xercavins, M.; Rodríguez-Carballeira, M.; Gómez-Vera, J.R.; Coll, I.; Vidal, D.; Llovet, T.; Ruíz-Bremón, A. Emergence and dissemination of quinolone-resistantescherichia coli in the community. Antimicrob. Agents Chemother. 1999, 43, 2736–2741. [Google Scholar] [CrossRef] [PubMed]
- Khlebtsov, N.; Dykman, L. Biodistribution and toxicity of engineered gold nanoparticles: A review of in vitro and in vivo studies. Chem. Soc. Rev. 2011, 40, 1647–1671. [Google Scholar] [CrossRef]
- Bondarenko, O.; Juganson, K.; Ivask, A.; Kasemets, K.; Mortimer, M.; Kahru, A. Toxicity of ag, cuo and zno nanoparticles to selected environmentally relevant test organisms and mammalian cells in vitro: A critical review. Arch. Toxicol. 2013, 87, 1181–1200. [Google Scholar] [CrossRef] [PubMed]
- Baun, A.; Hartmann, N.B.; Grieger, K.; Kusk, K.O. Ecotoxicity of engineered nanoparticles to aquatic invertebrates: A brief review and recommendations for future toxicity testing. Ecotoxicology 2008, 17, 387–395. [Google Scholar] [CrossRef] [PubMed]
- Reidy, B.; Haase, A.; Luch, A.; Dawson, K.; Lynch, I. Mechanisms of silver nanoparticle release, transformation and toxicity: A critical review of current knowledge and recommendations for future studies and applications. Materials 2013, 6, 2295–2350. [Google Scholar] [CrossRef] [PubMed]
- De Lima, R.; Seabra, A.B.; Durán, N. Silver nanoparticles: A brief review of cytotoxicity and genotoxicity of chemically and biogenically synthesized nanoparticles. J. Appl. Toxicol. 2012, 32, 867–879. [Google Scholar] [CrossRef] [PubMed]
- Albanese, A.; Chan, W.C. Effect of gold nanoparticle aggregation on cell uptake and toxicity. ACS Nano 2011, 5, 5478–5489. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Kaner, R.B. Shape and aggregation control of nanoparticles: Not shaken, not stirred. J. Am. Chem. Soc. 2006, 128, 968–975. [Google Scholar] [CrossRef] [PubMed]
- Moghimi, S.M.; Szebeni, J. Stealth liposomes and long circulating nanoparticles: Critical issues in pharmacokinetics, opsonization and protein-binding properties. Prog. Lipid Res. 2003, 42, 463–478. [Google Scholar] [CrossRef]
- Wang, L.; Hu, C.; Shao, L. The antimicrobial activity of nanoparticles: Present situation and prospects for the future. Int. J. Nanomed. 2017, 12, 1227. [Google Scholar] [CrossRef] [PubMed]
- Talebian, N.; Zavvare, H.S.H. Enhanced bactericidal action of sno2 nanostructures having different morphologies under visible light: Influence of surfactant. J. Photochem. Photobiol. B Biol. 2014, 130, 132–139. [Google Scholar] [CrossRef]
- Cha, S.-H.; Hong, J.; McGuffie, M.; Yeom, B.; VanEpps, J.S.; Kotov, N.A. Shape-dependent biomimetic inhibition of enzyme by nanoparticles and their antibacterial activity. ACS Nano 2015, 9, 9097–9105. [Google Scholar] [CrossRef]
- Actis, L.; Srinivasan, A.; Lopez-Ribot, J.L.; Ramasubramanian, A.K.; Ong, J.L. Effect of silver nanoparticle geometry on methicillin susceptible and resistant staphylococcus aureus, and osteoblast viability. J. Mater. Sci. Mater. Med. 2015, 26, 215. [Google Scholar] [CrossRef]
- Gao, M.; Sun, L.; Wang, Z.; Zhao, Y. Controlled synthesis of ag nanoparticles with different morphologies and their antibacterial properties. Mater. Sci. Eng. C 2013, 33, 397–404. [Google Scholar] [CrossRef]
- Hong, X.; Wen, J.; Xiong, X.; Hu, Y. Shape effect on the antibacterial activity of silver nanoparticles synthesized via a microwave-assisted method. Environ. Sci. Pollut. Res. 2016, 23, 4489–4497. [Google Scholar] [CrossRef]
- Prasannakumar, J.; Vidya, Y.; Anantharaju, K.; Ramgopal, G.; Nagabhushana, H.; Sharma, S.; Prasad, B.D.; Prashantha, S.; Basavaraj, R.; Rajanaik, H. Bio-mediated route for the synthesis of shape tunable y2o3: Tb3+ nanoparticles: Photoluminescence and antibacterial properties. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 151, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Rai, M.; Kon, K.; Gade, A.; Ingle, A.; Nagaonkar, D.; Paralikar, P.; da Silva, S. Antibiotic resistance: Can nanoparticles tackle the problem? Antibiot. Resist. 2016. [Google Scholar] [CrossRef]
- Chaloupka, K.; Malam, Y.; Seifalian, A.M. Nanosilver as a new generation of nanoproduct in biomedical applications. Trends Biotechnol. 2010, 28, 580–588. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, A.K.; Chakraborty, R.; Basu, T. Mechanism of antibacterial activity of copper nanoparticles. Nanotechnology 2014, 25, 135101. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.; Curtis, A.; Hoskins, C. Application of nanoparticle technologies in the combat against anti-microbial resistance. Pharmaceutics 2018, 10, 11. [Google Scholar] [CrossRef] [PubMed]
- Imlay, J.A.; Linn, S. DNA damage and oxygen radical toxicity. Science 1988, 240, 1302–1309. [Google Scholar] [CrossRef]
- Itoh, M.; Nakamura, M.; Suzuki, T.; Kawai, K.; Horitsu, H.; Takamizawa, K. Mechanism of chromium (vi) toxicity in escherichia coli: Is hydrogen peroxide essential in cr (vi) toxicity? J. Biochem. 1995, 117, 780–786. [Google Scholar] [CrossRef]
- Geslin, C.; Llanos, J.; Prieur, D.; Jeanthon, C. The manganese and iron superoxide dismutases protect escherichia coli from heavy metal toxicity. Res. Microbiol. 2001, 152, 901–905. [Google Scholar] [CrossRef]
- Parvatiyar, K.; Alsabbagh, E.M.; Ochsner, U.A.; Stegemeyer, M.A.; Smulian, A.G.; Hwang, S.H.; Jackson, C.R.; McDermott, T.R.; Hassett, D.J. Global analysis of cellular factors and responses involved in pseudomonas aeruginosa resistance to arsenite. J. Bacteriol. 2005, 187, 4853–4864. [Google Scholar] [CrossRef]
- Sumner, E.R.; Shanmuganathan, A.; Sideri, T.C.; Willetts, S.A.; Houghton, J.E.; Avery, S.V. Oxidative protein damage causes chromium toxicity in yeast. Microbiology 2005, 151, 1939–1948. [Google Scholar] [CrossRef] [PubMed]
- Lemire, J.A.; Harrison, J.J.; Turner, R.J. Antimicrobial activity of metals: Mechanisms, molecular targets and applications. Nat. Rev. Microbiol. 2013, 11, 371. [Google Scholar] [CrossRef] [PubMed]
- Fadeeva, M.; Bertsova, Y.; Euro, L.; Bogachev, A. Cys377 residue in nqrf subunit confers ag+ sensitivity of na+-translocating nadh: Quinone oxidoreductase from vibrio harveyi. Biochemistry 2011, 76, 186–195. [Google Scholar] [PubMed]
- Lok, C.-N.; Ho, C.-M.; Chen, R.; He, Q.-Y.; Yu, W.-Y.; Sun, H.; Tam, P.K.-H.; Chiu, J.-F.; Che, C.-M. Silver nanoparticles: Partial oxidation and antibacterial activities. JBIC J. Biol. Inorg. Chem. 2007, 12, 527–534. [Google Scholar] [CrossRef] [PubMed]
- Bragg, P.; Rainnie, D. The effect of silver ions on the respiratory chain of escherichia coli. Can. J. Microbiol. 1974, 20, 883–889. [Google Scholar] [CrossRef] [PubMed]
- Kandi, V.; Kandi, S. Antimicrobial properties of nanomolecules: Potential candidates as antibiotics in the era of multi-drug resistance. Epidemiol. Health 2015, 37, e2015020. [Google Scholar] [CrossRef] [PubMed]
- Simões, D.; Miguel, S.P.; Ribeiro, M.P.; Coutinho, P.; Mendonça, A.G.; Correia, I.J. Recent advances on antimicrobial wound dressing: A review. Eur. J. Pharm. Biopharm. 2018, 127, 130–141. [Google Scholar] [CrossRef]
- Lesniak, A.; Salvati, A.; Santos-Martinez, M.J.; Radomski, M.W.; Dawson, K.A.; Åberg, C. Nanoparticle adhesion to the cell membrane and its effect on nanoparticle uptake efficiency. J. Am. Chem. Soc. 2013, 135, 1438–1444. [Google Scholar] [CrossRef]
- Huh, A.J.; Kwon, Y.J. “Nanoantibiotics”: A new paradigm for treating infectious diseases using nanomaterials in the antibiotics resistant era. J. Control. Release 2011, 156, 128–145. [Google Scholar] [CrossRef]
- Begg, E.J.; Barclay, M.L.; Kirkpatrick, C.J. The therapeutic monitoring of antimicrobial agents. Br. J. Clin. Pharmacol. 1999, 47, 23–30. [Google Scholar] [CrossRef]
- Yount, N.Y.; Yeaman, M.R. Emerging themes and therapeutic prospects for anti-infective peptides. Annu. Rev. Pharmacol. Toxicol. 2012, 52, 337–360. [Google Scholar] [CrossRef] [PubMed]
- Koul, A.; Arnoult, E.; Lounis, N.; Guillemont, J.; Andries, K. The challenge of new drug discovery for tuberculosis. Nature 2011, 469, 483. [Google Scholar] [CrossRef] [PubMed]
- Triggs, E.; Charles, B. Pharmacokinetics and therapeutic drug monitoring of gentamicin in the elderly. Clin. Pharmacokinet. 1999, 37, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Kopecka, J.; Campia, I.; Olivero, P.; Pescarmona, G.; Ghigo, D.; Bosia, A.; Riganti, C. A ldl-masked liposomal-doxorubicin reverses drug resistance in human cancer cells. J. Control. Release 2011, 149, 196–205. [Google Scholar] [CrossRef] [PubMed]
- Kakkar Thukral, D.; Dumoga, S.; K Mishra, A. Solid lipid nanoparticles: Promising therapeutic nanocarriers for drug delivery. Curr. Drug Deliv. 2014, 11, 771–791. [Google Scholar] [CrossRef]
- Daeihamed, M.; Dadashzadeh, S.; Haeri, A.; Faghih Akhlaghi, M. Potential of liposomes for enhancement of oral drug absorption. Curr. Drug Deliv. 2017, 14, 289–303. [Google Scholar] [CrossRef] [PubMed]
- Abed, N.; Couvreur, P. Nanocarriers for antibiotics: A promising solution to treat intracellular bacterial infections. Int. J. Antimicrob. Agents 2014, 43, 485–496. [Google Scholar] [CrossRef]
- Naseri, N.; Valizadeh, H.; Zakeri-Milani, P. Solid lipid nanoparticles and nanostructured lipid carriers: Structure, preparation and application. Adv. Pharm. Bull. 2015, 5, 305. [Google Scholar] [CrossRef]
- Liu, Y.; K Tee, J.; NC Chiu, G. Dendrimers in oral drug delivery application: Current explorations, toxicity issues and strategies for improvement. Curr. Pharm. Des. 2015, 21, 2629–2642. [Google Scholar] [CrossRef]
- Ranghar, S.; Sirohi, P.; Verma, P.; Agarwal, V. Nanoparticle-based drug delivery systems: Promising approaches against infections. Braz. Arch. Biol. Technol. 2014, 57, 209–222. [Google Scholar] [CrossRef]
- Saha, B.; Bhattacharya, J.; Mukherjee, A.; Ghosh, A.; Santra, C.; Dasgupta, A.K.; Karmakar, P. In vitro structural and functional evaluation of gold nanoparticles conjugated antibiotics. Nanoscale Res. Lett. 2007, 2, 614. [Google Scholar] [CrossRef]
- Brown, A.; Smith, K.; Samuels, T.A.; Lu, J.; Obare, S.; Scott, M.E. Nanoparticles functionalized with ampicillin destroy multiple antibiotic resistant isolates of pseudomonas aeruginosa, enterobacter aerogenes and methicillin resistant staphylococcus aureus. Appl. Environ. Microbiol. 2012. [Google Scholar] [CrossRef] [PubMed]
- Vallet-Regí, M.; Arcos, D.; Navarrete, D.A. Biomimetic Nanoceramics in Clinical Use: From Materials to Applications; Royal Society of Chemistry: London, UK, 2008; Volume 5. [Google Scholar]
- Lowe, D.B. Drug discovery: Combichem all over again. Nat. Chem. 2014, 6, 851. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Zhang, K.; Shi, C.; Liu, L.; Yan, G.; Liu, C.; Zhou, Y.; Hu, Y.; Sun, H.; Yang, B. Small molecules modified biomimetic gelatin/hydroxyapatite nanofibers constructing an ideal osteogenic microenvironment with significantly enhanced cranial bone formation. Int. J. Nanomed. 2018, 13, 7167. [Google Scholar] [CrossRef] [PubMed]
- Tao, H.; Brenckle, M.A.; Yang, M.; Zhang, J.; Liu, M.; Siebert, S.M.; Averitt, R.D.; Mannoor, M.S.; McAlpine, M.C.; Rogers, J.A. Silk-based conformal, adhesive, edible food sensors. Adv. Mater. 2012, 24, 1067–1072. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; Zhang, L. Coating nanoparticles with cell membranes for targeted drug delivery. J. Drug Target. 2015, 23, 619–626. [Google Scholar] [CrossRef]
- Fang, R.H.; Hu, C.-M.J.; Luk, B.T.; Gao, W.; Copp, J.A.; Tai, Y.; O’Connor, D.E.; Zhang, L. Cancer cell membrane-coated nanoparticles for anticancer vaccination and drug delivery. Nano Lett. 2014, 14, 2181–2188. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.-M.J.; Fang, R.H.; Wang, K.-C.; Luk, B.T.; Thamphiwatana, S.; Dehaini, D.; Nguyen, P.; Angsantikul, P.; Wen, C.H.; Kroll, A.V. Nanoparticle biointerfacing by platelet membrane cloaking. Nature 2015, 526, 118. [Google Scholar] [CrossRef]
- Hu, Q.; Sun, W.; Qian, C.; Wang, C.; Bomba, H.N.; Gu, Z. Anticancer platelet-mimicking nanovehicles. Adv. Mater. 2015, 27, 7043–7050. [Google Scholar] [CrossRef]
- McColl, K.E. Helicobacter pylori infection. N. Engl. J. Med. 2010, 362, 1597–1604. [Google Scholar] [CrossRef]
- Yamaoka, Y. Mechanisms of disease: Helicobacter pylori virulence factors. Nat. Rev. Gastroenterol. Hepatol. 2010, 7, 629. [Google Scholar] [CrossRef] [PubMed]
- Salama, N.R.; Hartung, M.L.; Müller, A. Life in the human stomach: Persistence strategies of the bacterial pathogen helicobacter pylori. Nat. Rev. Microbiol. 2013, 11, 385. [Google Scholar] [CrossRef] [PubMed]
- O’connor, A.; O’morain, C.A.; Ford, A.C. Population screening and treatment of helicobacter pylori infection. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 230. [Google Scholar] [CrossRef] [PubMed]
- Sgouras, D.N.; Trang, T.T.H.; Yamaoka, Y. Pathogenesis of helicobacter pylori infection. Helicobacter 2015, 20, 8–16. [Google Scholar] [CrossRef]
- Kaplan-Türköz, B.; Jiménez-Soto, L.F.; Dian, C.; Ertl, C.; Remaut, H.; Louche, A.; Tosi, T.; Haas, R.; Terradot, L. Structural insights into helicobacter pylori oncoprotein caga interaction with β1 integrin. Proc. Natl. Acad. Sci. USA 2012. [Google Scholar] [CrossRef]
- Königer, V.; Holsten, L.; Harrison, U.; Busch, B.; Loell, E.; Zhao, Q.; Bonsor, D.A.; Roth, A.; Kengmo-Tchoupa, A.; Smith, S.I. Helicobacter pylori exploits human ceacams via hopq for adherence and translocation of caga. Nat. Microbiol. 2017, 2, 16188. [Google Scholar] [CrossRef] [PubMed]
- Koelblen, T.; Bergé, C.; Cherrier, M.V.; Brillet, K.; Jimenez-Soto, L.; Ballut, L.; Takagi, J.; Montserret, R.; Rousselle, P.; Fischer, W. Molecular dissection of protein–protein interactions between integrin α5β1 and the helicobacter pylori cag type iv secretion system. FEBS J. 2017, 284, 4143–4157. [Google Scholar] [CrossRef] [PubMed]
- Parreira, P.; Shi, Q.; Magalhaes, A.; Reis, C.; Bugaytsova, J.; Borén, T.; Leckband, D.; Martins, M. Atomic force microscopy measurements reveal multiple bonds between helicobacter pylori blood group antigen binding adhesin and lewis b ligand. J. R. Soc. Interface 2014, 11, 20141040. [Google Scholar] [CrossRef] [PubMed]
- Fei, Y.; Schmidt, A.; Bylund, G.; Johansson, D.; Henriksson, S.; Lebrilla, C.; Solnick, J.; Borén, T.; Zhu, X. Use of real-time, label-free analysis in revealing low-affinity binding to blood group antigens by helicobacter pylori. Anal. Chem. 2011, 83, 6336–6341. [Google Scholar] [CrossRef] [PubMed]
- Angsantikul, P.; Thamphiwatana, S.; Zhang, Q.; Spiekermann, K.; Zhuang, J.; Fang, R.H.; Gao, W.; Obonyo, M.; Zhang, L. Coating nanoparticles with gastric epithelial cell membrane for targeted antibiotic delivery against helicobacter pylori infection. Adv. Ther. 2018, 1, 1800016. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M. High antibiotic resistance rate: A difficult issue for helicobacter pylori eradication treatment. World J. Gastroenterol. 2015, 21, 13432. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Zhang, M.; Lu, B.; Dai, J. Helicobacter pylori and antibiotic resistance, a continuing and intractable problem. Helicobacter 2016, 21, 349–363. [Google Scholar] [CrossRef] [PubMed]
- Alba, C.; Blanco, A.; Alarcón, T. Antibiotic resistance in helicobacter pylori. Curr. Opin. Infect. Dis. 2017, 30, 489–497. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Antimicrobial Resistance: Global Report on Surveillance; World Health Organization: Geneva, Switzerland, 2014. [Google Scholar]
- Appelbaum, P. The emergence of vancomycin-intermediate and vancomycin-resistant staphylococcus aureus. Clin. Microbiol. Infect. 2006, 12, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Hiramatsu, K. Vancomycin-resistant staphylococcus aureus: A new model of antibiotic resistance. Lancet Infect. Dis. 2001, 1, 147–155. [Google Scholar] [CrossRef]
- Gardete, S.; Tomasz, A. Mechanisms of vancomycin resistance in staphylococcus aureus. J. Clin. Investig. 2014, 124, 2836–2840. [Google Scholar] [CrossRef] [PubMed]
- Hong, H.-J.; Hutchings, M.I.; Buttner, M.J. Vancomycin resistance vans/vanr two-component systems. In Bacterial Signal Transduction: Networks and Drug Targets; Springer: New York, NY, USA, 2008; pp. 200–213. [Google Scholar]
- Arthur, M.; Courvalin, P. Genetics and mechanisms of glycopeptide resistance in enterococci. Antimicrob. Agents Chemother. 1993, 37, 1563. [Google Scholar] [CrossRef] [PubMed]
- Liakos, I.; Grumezescu, A.; Holban, A. Magnetite nanostructures as novel strategies for anti-infectious therapy. Molecules 2014, 19, 12710–12726. [Google Scholar] [CrossRef]
- Peng, B.; Zhang, X.; Aarts, D.G.; Dullens, R.P. Superparamagnetic nickel colloidal nanocrystal clusters with antibacterial activity and bacteria binding ability. Nat. Nanotechnol. 2018, 13, 478. [Google Scholar] [CrossRef]
- Cowger, T.A.; Yang, Y.; Rink, D.E.; Todd, T.; Chen, H.; Shen, Y.; Yan, Y.; Xie, J. Protein-adsorbed magnetic-nanoparticle-mediated assay for rapid detection of bacterial antibiotic resistance. Bioconjug. Chem. 2017, 28, 890–896. [Google Scholar] [CrossRef]
- Hassan, M.M.; Ranzoni, A.; Phetsang, W.; Blaskovich, M.A.; Cooper, M.A. Surface ligand density of antibiotic-nanoparticle conjugates enhances target avidity and membrane permeabilization of vancomycin-resistant bacteria. Bioconjug. Chem. 2016, 28, 353–361. [Google Scholar] [CrossRef] [PubMed]
- Miele, E.; Spinelli, G.P.; Miele, E.; Tomao, F.; Tomao, S. Albumin-bound formulation of paclitaxel (abraxane® abi-007) in the treatment of breast cancer. Int. J. Nanomed. 2009, 4, 99. [Google Scholar]
- Peters, T., Jr. All about Albumin: Biochemistry, Genetics, and Medical Applications; Academic press: Cambridge, MA, USA, 1995. [Google Scholar]
- Matsushita, S.; Chuang, V.T.G.; Kanazawa, M.; Tanase, S.; Kawai, K.; Maruyama, T.; Suenaga, A.; Otagiri, M. Recombinant human serum albumin dimer has high blood circulation activity and low vascular permeability in comparison with native human serum albumin. Pharm. Res. 2006, 23, 882–891. [Google Scholar] [CrossRef] [PubMed]
- Nunes, S.; Barros, A. The use of coating agents to enhance liposomes blood circulation time. J. Mol. Pharm. Org. Process. Res. 2015, 3, 1–2. [Google Scholar]
- Grumezescu, A.M.; Holban, A.M.; Andronescu, E.; Mogoşanu, G.D.; Vasile, B.S.; Chifiriuc, M.C.; Lazar, V.; Andrei, E.; Constantinescu, A.; Maniu, H. Anionic polymers and 10 nm fe3o4@ ua wound dressings support human foetal stem cells normal development and exhibit great antimicrobial properties. Int. J. Pharm. 2014, 463, 146–154. [Google Scholar] [CrossRef] [PubMed]
- Grumezescu, A.M.; Cotar, A.I.; Andronescu, E.; Ficai, A.; Ghitulica, C.D.; Grumezescu, V.; Vasile, B.S.; Chifiriuc, M.C. In vitro activity of the new water-dispersible fe 3 o 4@ usnic acid nanostructure against planktonic and sessile bacterial cells. J. Nanopart. Res. 2013, 15, 1766. [Google Scholar] [CrossRef]
- Anghel, I.; Grumezescu, A.M.; Holban, A.M.; Ficai, A.; Anghel, A.G.; Chifiriuc, M.C. Biohybrid nanostructured iron oxide nanoparticles and satureja hortensis to prevent fungal biofilm development. Int. J. Mol. Sci. 2013, 14, 18110–18123. [Google Scholar] [CrossRef] [PubMed]
- Grumezescu, A.M.; Gestal, M.C.; Holban, A.M.; Grumezescu, V.; Vasile, B.Ș.; Mogoantă, L.; Iordache, F.; Bleotu, C.; Mogoșanu, G.D. Biocompatible fe3o4 increases the efficacy of amoxicillin delivery against gram-positive and gram-negative bacteria. Molecules 2014, 19, 5013–5027. [Google Scholar] [CrossRef]
- Fairbairn, I.P.; Stober, C.B.; Kumararatne, D.S.; Lammas, D.A. Atp-mediated killing of intracellular mycobacteria by macrophages is a p2x7-dependent process inducing bacterial death by phagosome-lysosome fusion. J. Immunol. 2001, 167, 3300–3307. [Google Scholar] [CrossRef]
- Slauch, J.M. How does the oxidative burst of macrophages kill bacteria? Still an open question. Mol. Microbiol. 2011, 80, 580–583. [Google Scholar] [CrossRef]
- Abbas, A.K.; Lichtman, A.H.; Pillai, S. Basic Immunology: Functions and Disorders of the Immune System; Elsevier Health Sciences: Amsterdam, The Netherlands, 2014. [Google Scholar]
- Foster, T.J. Immune evasion by staphylococci. Nat. Rev. Microbiol. 2005, 3, 948. [Google Scholar] [CrossRef] [PubMed]
- Proctor, R.A.; Von Eiff, C.; Kahl, B.C.; Becker, K.; McNamara, P.; Herrmann, M.; Peters, G. Small colony variants: A pathogenic form of bacteria that facilitates persistent and recurrent infections. Nat. Rev. Microbiol. 2006, 4, 295. [Google Scholar] [CrossRef] [PubMed]
- Bakker-Woudenberg, I.A. Delivery of antimicrobials to infected tissue macrophages. Adv. Drug Deliv. Rev. 1995, 17, 5–20. [Google Scholar] [CrossRef]
- Briones, E.; Colino, C.I.; Lanao, J.M. Delivery systems to increase the selectivity of antibiotics in phagocytic cells. J. Control. Release 2008, 125, 210–227. [Google Scholar] [CrossRef] [PubMed]
- Toti, U.S.; Guru, B.R.; Hali, M.; McPharlin, C.M.; Wykes, S.M.; Panyam, J.; Whittum-Hudson, J.A. Targeted delivery of antibiotics to intracellular chlamydial infections using plga nanoparticles. Biomaterials 2011, 32, 6606–6613. [Google Scholar] [CrossRef] [PubMed]
- Deysine, M.; Chua, A.; Gerboth, A. Selective delivery of antibiotics to experimental infection by autologous white blood cells. Surg. Forum 1979, 30, 38–39. [Google Scholar] [PubMed]
- Mehta, R.T.; McQueen, T.J.; Keyhani, A.; López-Berestein, G. Phagocyte transport as mechanism for enhanced therapeutic activity of liposomal amphotericin b. Chemotherapy 1994, 40, 256–264. [Google Scholar] [CrossRef]
- Irache, J.M.; Salman, H.H.; Gamazo, C.; Espuelas, S. Mannose-targeted systems for the delivery of therapeutics. Expert Opin. Drug Deliv. 2008, 5, 703–724. [Google Scholar] [CrossRef]
- Mosser, D.M.; Edwards, J.P. Exploring the full spectrum of macrophage activation. Nat. Rev. Immunol. 2008, 8, 958. [Google Scholar] [CrossRef]
- Kawai, T.; Akira, S. Toll-like receptors and their crosstalk with other innate receptors in infection and immunity. Immunity 2011, 34, 637–650. [Google Scholar] [CrossRef]
- Kelly, C.; Jefferies, C.; Cryan, S.-A. Targeted liposomal drug delivery to monocytes and macrophages. J. Drug Deliv. 2011, 2011, 727241. [Google Scholar] [CrossRef] [PubMed]
- Karmakar, P.; Lee, K.; Sarkar, S.; Wall, K.A.; Sucheck, S.J. Synthesis of a liposomal muc1 glycopeptide-based immunotherapeutic and evaluation of the effect of l-rhamnose targeting on cellular immune responses. Bioconjug. Chem. 2015, 27, 110–120. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, S.; Salyer, A.C.; Wall, K.A.; Sucheck, S.J. Synthesis and immunological evaluation of a muc1 glycopeptide incorporated into l-rhamnose displaying liposomes. Bioconjug. Chem. 2013, 24, 363–375. [Google Scholar] [CrossRef] [PubMed]
- Kamath, A.T.; Rochat, A.-F.; Christensen, D.; Agger, E.M.; Andersen, P.; Lambert, P.-H.; Siegrist, C.-A. A liposome-based mycobacterial vaccine induces potent adult and neonatal multifunctional t cells through the exquisite targeting of dendritic cells. PLoS ONE 2009, 4, e5771. [Google Scholar] [CrossRef]
- Rukavina, Z.; Klarić, M.Š.; Filipović-Grčić, J.; Lovrić, J.; Vanić, Ž. Azithromycin-loaded liposomes for enhanced topical treatment of methicillin-resistant staphyloccocus aureus (mrsa) infections. Int. J. Pharm. 2018, 553, 109–119. [Google Scholar] [CrossRef]
- Gabizon, A.; Shmeeda, H.; Barenholz, Y. Pharmacokinetics of pegylated liposomal doxorubicin. Clin. Pharm. 2003, 42, 419–436. [Google Scholar] [CrossRef] [PubMed]
- Wijagkanalan, W.; Kawakami, S.; Higuchi, Y.; Yamashita, F.; Hashida, M. Intratracheally instilled mannosylated cationic liposome/nfκb decoy complexes for effective prevention of lps-induced lung inflammation. J. Control. Release 2011, 149, 42–50. [Google Scholar] [CrossRef]
- Xiong, M.H.; Li, Y.J.; Bao, Y.; Yang, X.Z.; Hu, B.; Wang, J. Bacteria-responsive multifunctional nanogel for targeted antibiotic delivery. Adv. Mater. 2012, 24, 6175–6180. [Google Scholar] [CrossRef]
- Xiong, M.-H.; Wu, J.; Wang, Y.-C.; Li, L.-S.; Liu, X.-B.; Zhang, G.-Z.; Yan, L.-F.; Wang, J. Synthesis of peg-armed and polyphosphoester core-cross-linked nanogel by one-step ring-opening polymerization. Macromolecules 2009, 42, 893–896. [Google Scholar] [CrossRef]
- Faron, M.L.; Ledeboer, N.A.; Buchan, B.W. Resistance mechanisms, epidemiology, and approaches to screening for vancomycin resistant enterococcus (vre) in the health care setting. J. Clin. Microbiol. 2016. [Google Scholar] [CrossRef]
- Eliopoulos, G.M.; Gold, H. Vancomycin-resistant enterococci: Mechanisms and clinical observations. Clin. Infect. Dis. 2001, 33, 210–219. [Google Scholar]
- DeVinney, R.; Steele-Mortimer, O.; Finlay, B.B. Phosphatases and kinases delivered to the host cell by bacterial pathogens. Trends Microbiol. 2000, 8, 29–33. [Google Scholar] [CrossRef]
- Schaffner, T.; Keller, H.U.; Hess, M.W.; Cottier, H. Macrophage functions in antimicrobial defense. Klinische Wochenschr. 1982, 60, 720–726. [Google Scholar] [CrossRef]
- Lu, Z.-Z.; Wu, J.; Sun, T.-M.; Ji, J.; Yan, L.-F.; Wang, J. Biodegradable polycation and plasmid DNA multilayer film for prolonged gene delivery to mouse osteoblasts. Biomaterials 2008, 29, 733–741. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.-Z.; Sun, T.-M.; Dou, S.; Wu, J.; Wang, Y.-C.; Wang, J. Block copolymer of polyphosphoester and poly (l-lactic acid) modified surface for enhancing osteoblast adhesion, proliferation, and function. Biomacromolecules 2009, 10, 2213–2220. [Google Scholar] [CrossRef]
- Wachiralarpphaithoon, C.; Iwasaki, Y.; Akiyoshi, K. Enzyme-degradable phosphorylcholine porous hydrogels cross-linked with polyphosphoesters for cell matrices. Biomaterials 2007, 28, 984–993. [Google Scholar] [CrossRef]
- Huang, C.; Cabot, M. Vasopressin-induced polyphosphoinositide and phosphatidylcholine degradation in fibroblasts. Temporal relationship for formation of phospholipase c and phospholipase d hydrolysis products. J. Biol. Chem. 1990, 265, 17468–17473. [Google Scholar]
- Lakes, A.L.; Peyyala, R.; Ebersole, J.L.; Puleo, D.A.; Hilt, J.Z.; Dziubla, T.D. Synthesis and characterization of an antibacterial hydrogel containing covalently bound vancomycin. Biomacromolecules 2014, 15, 3009–3018. [Google Scholar] [CrossRef]
- Zhang, X.; Xia, L.-Y.; Chen, X.; Chen, Z.; Wu, F.-G. Hydrogel-based phototherapy for fighting cancer and bacterial infection. Sci. China Mater. 2017, 60, 487–503. [Google Scholar] [CrossRef]
- Lu, Y.; Wu, Y.; Liang, J.; Libera, M.R.; Sukhishvili, S.A. Self-defensive antibacterial layer-by-layer hydrogel coatings with ph-triggered hydrophobicity. Biomaterials 2015, 45, 64–71. [Google Scholar] [CrossRef]
- McMahon, S.; Kennedy, R.; Duffy, P.; Vasquez, J.M.; Wall, J.G.; Tai, H.; Wang, W. Poly (ethylene glycol)-based hyperbranched polymer from raft and its application as a silver-sulfadiazine-loaded antibacterial hydrogel in wound care. ACS Appl. Mater. Interfaces 2016, 8, 26648–26656. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Wu, H.; Guo, B.; Dong, R.; Qiu, Y.; Ma, P.X. Antibacterial anti-oxidant electroactive injectable hydrogel as self-healing wound dressing with hemostasis and adhesiveness for cutaneous wound healing. Biomaterials 2017, 122, 34–47. [Google Scholar] [CrossRef] [PubMed]
- Peppas, N.A.; Hilt, J.Z.; Khademhosseini, A.; Langer, R. Hydrogels in biology and medicine: From molecular principles to bionanotechnology. Adv. Mater. 2006, 18, 1345–1360. [Google Scholar] [CrossRef]
- Seif-Naraghi, S.B.; Singelyn, J.M.; Salvatore, M.A.; Osborn, K.G.; Wang, J.J.; Sampat, U.; Kwan, O.L.; Strachan, G.M.; Wong, J.; Schup-Magoffin, P.J. Safety and efficacy of an injectable extracellular matrix hydrogel for treating myocardial infarction. Sci. Transl. Med. 2013, 5, ra125–ra173. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Fang, L.; Zhu, Z.; Ma, Y.; Zhou, L.; Chen, X.; Xu, D.; Yang, C. Design and synthesis of target-responsive hydrogel for portable visual quantitative detection of uranium with a microfluidic distance-based readout device. Biosens. Bioelectron. 2016, 85, 496–502. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Wu, M.; Zeng, Y.; Wu, L.; Wang, Q.; Han, X.; Liu, X.; Liu, J. Chlorin e6 conjugated poly (dopamine) nanospheres as pdt/ptt dual-modal therapeutic agents for enhanced cancer therapy. ACS Appl. Mater. Interfaces 2015, 7, 8176–8187. [Google Scholar] [CrossRef] [PubMed]
- Lynge, M.E.; van der Westen, R.; Postma, A.; Städler, B. Polydopamine—A nature-inspired polymer coating for biomedical science. Nanoscale 2011, 3, 4916–4928. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Cao, J.; Li, H.; Li, J.; Jin, Q.; Ren, K.; Ji, J. Mussel-inspired polydopamine: A biocompatible and ultrastable coating for nanoparticles in vivo. ACS Nano 2013, 7, 9384–9395. [Google Scholar] [CrossRef]
- Li, H.; Jia, Y.; Peng, H.; Li, J. Recent developments in dopamine-based materials for cancer diagnosis and therapy. Adv. Colloid Interface Sci. 2018, 252, 1–20. [Google Scholar] [CrossRef]
- Huang, L.; Li, Z.; Zhao, Y.; Zhang, Y.; Wu, S.; Zhao, J.; Han, G. Ultralow-power near infrared lamp light operable targeted organic nanoparticle photodynamic therapy. J. Am. Chem. Soc. 2016, 138, 14586–14591. [Google Scholar] [CrossRef]
- Jiang, Y.; Li, J.; Zhen, X.; Xie, C.; Pu, K. Dual-peak absorbing semiconducting copolymer nanoparticles for first and second near-infrared window photothermal therapy: A comparative study. Adv. Mater. 2018, 30, 1705980. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Huang, L.; Zhang, Y.; Zhao, Y.; Yang, H.; Han, G. Near-infrared light activated persistent luminescence nanoparticles via upconversion. Nano Res. 2017, 10, 1840–1846. [Google Scholar] [CrossRef]
- Liu, F.; He, X.; Lei, Z.; Liu, L.; Zhang, J.; You, H.; Zhang, H.; Wang, Z. Facile preparation of doxorubicin-loaded upconversion@ polydopamine nanoplatforms for simultaneous in vivo multimodality imaging and chemophotothermal synergistic therapy. Adv. Healthc. Mater. 2015, 4, 559–568. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhang, J.; Wang, Y.; Wang, C.; Xiao, J.; Zhang, Q.; Cheng, Y. Multi-responsive photothermal-chemotherapy with drug-loaded melanin-like nanoparticles for synergetic tumor ablation. Biomaterials 2016, 81, 114–124. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, C.; Wang, X.; Wang, Y.; Zhang, Q.; Cheng, Y. A polydopamine nanoparticle-knotted poly (ethylene glycol) hydrogel for on-demand drug delivery and chemo-photothermal therapy. Chem. Mater. 2017, 29, 1370–1376. [Google Scholar] [CrossRef]
- Gao, G.; Jiang, Y.-W.; Jia, H.-R.; Wu, F.-G. Near-infrared light-controllable on-demand antibiotics release using thermo-sensitive hydrogel-based drug reservoir for combating bacterial infection. Biomaterials 2019, 188, 83–95. [Google Scholar] [CrossRef] [PubMed]
- Sakudo, A. Near-infrared spectroscopy for medical applications: Current status and future perspectives. Clin. Chim. Acta 2016, 455, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Wang, Y.; Zhang, L.; Miron, R.J.; Liang, J.; Shi, M.; Mo, W.; Zheng, S.; Zhao, Y.; Zhang, Y. Pretreated macrophage-membrane-coated gold nanocages for precise drug delivery for treatment of bacterial infections. Adv. Mater. 2018, 30, 1804023. [Google Scholar] [CrossRef]
- Taylor, P.R.; Martinez-Pomares, L.; Stacey, M.; Lin, H.-H.; Brown, G.D.; Gordon, S. Macrophage receptors and immune recognition. Annu. Rev. Immunol. 2005, 23, 901–944. [Google Scholar] [CrossRef]
- Cao, H.; Dan, Z.; He, X.; Zhang, Z.; Yu, H.; Yin, Q.; Li, Y. Liposomes coated with isolated macrophage membrane can target lung metastasis of breast cancer. ACS Nano 2016, 10, 7738–7748. [Google Scholar] [CrossRef]
- Yavuz, M.S.; Cheng, Y.; Chen, J.; Cobley, C.M.; Zhang, Q.; Rycenga, M.; Xie, J.; Kim, C.; Song, K.H.; Schwartz, A.G. Gold nanocages covered by smart polymers for controlled release with near-infrared light. Nat. Mater. 2009, 8, 935. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Cai, X.; Kim, C.; Sun, G.; Zhang, Y.; Deng, R.; Yang, M.; Chen, J.; Achilefu, S.; Wang, L.V. Gold nanocages covered with thermally-responsive polymers for controlled release by high-intensity focused ultrasound. Nanoscale 2011, 3, 1724–1730. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Global Tuberculosis Control: Epidemiology, Strategy, Financing: Who Report 2009; World Health Organization: Geneva, Switzerland, 2009. [Google Scholar]
- Theuretzbacher, U. Accelerating resistance, inadequate antibacterial drug pipelines and international responses. Int. J. Antimicrob. Agents 2012, 39, 295–299. [Google Scholar] [CrossRef]
- Bowler, P.; Duerden, B.; Armstrong, D.G. Wound microbiology and associated approaches to wound management. Clin. Microbiol. Rev. 2001, 14, 244–269. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.; Hu, Z.-Q.; Peng, R.-Y. Effects and mechanisms of a microcurrent dressing on skin wound healing: A review. Mil. Med. Res. 2014, 1, 24. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Who Publishes List of Bacteria for Which New Antibiotics Are Urgently Needed; WHO: Geneva, Switzerland, 2017. [Google Scholar]
- Cardona, A.F.; Wilson, S.E. Skin and soft-tissue infections: A critical review and the role of telavancin in their treatment. Clin. Infect. Dis. 2015, 61, S69–S78. [Google Scholar] [CrossRef] [PubMed]
- Negut, I.; Grumezescu, V.; Grumezescu, A. Treatment strategies for infected wounds. Molecules 2018, 23, 2392. [Google Scholar] [CrossRef]
- Vijayakumar, V.; Samal, S.K.; Mohanty, S.; Nayak, S.K. Recent advancements in biopolymer and metal nanoparticle-based materials in diabetic wound healing management. Int. J. Biol. Macromol. 2018, 122, 137–148. [Google Scholar] [CrossRef]
- Verma, J.; Kanoujia, J.; Parashar, P.; Tripathi, C.B.; Saraf, S.A. Wound healing applications of sericin/chitosan-capped silver nanoparticles incorporated hydrogel. Drug Deliv. Transl. Res. 2017, 7, 77–88. [Google Scholar] [CrossRef]
- Zhou, Y.; Chen, R.; He, T.; Xu, K.; Du, D.; Zhao, N.; Cheng, X.; Yang, J.; Shi, H.; Lin, Y. Biomedical potential of ultrafine ag/agcl nanoparticles coated on graphene with special reference to antimicrobial performances and burn wound healing. ACS Appl. Mater. Interfaces 2016, 8, 15067–15075. [Google Scholar] [CrossRef] [PubMed]
- Ye, H.; Cheng, J.; Yu, K. In situ reduction of silver nanoparticles by gelatin to obtain porous silver nanoparticle/chitosan composites with enhanced antimicrobial and wound-healing activity. Int. J. Biol. Macromol. 2019, 121, 633–642. [Google Scholar] [CrossRef] [PubMed]
- Manigandan, V.; Karthik, R.; Ramachandran, S.; Rajagopal, S. Chitosan applications in food industry. In Biopolymers for Food Design; Elsevier: Amsterdam, The Netherlands, 2018; pp. 469–491. [Google Scholar]
- Mariod, A.A.; Adam, H.F. Gelatin, source, extraction and industrial applications. Acta Sci. Pol. Technol. Aliment. 2013, 12, 135–147. [Google Scholar]
- Chifiriuc, C.M.; Grumezescu, A.M.; Saviuc, C.; Croitoru, C.; Mihaiescu, D.E.; Lazar, V. Improved antibacterial activity of cephalosporins loaded in magnetic chitosan microspheres. Int. J. Pharm. 2012, 436, 201–205. [Google Scholar] [CrossRef]
- Grumezescu, A.M.; Andronescu, E.; Holban, A.M.; Ficai, A.; Ficai, D.; Voicu, G.; Grumezescu, V.; Balaure, P.C.; Chifiriuc, C.M. Water dispersible cross-linked magnetic chitosan beads for increasing the antimicrobial efficiency of aminoglycoside antibiotics. Int. J. Pharm. 2013, 454, 233–240. [Google Scholar] [CrossRef]
- Balaure, P.C.; Andronescu, E.; Grumezescu, A.M.; Ficai, A.; Huang, K.-S.; Yang, C.-H.; Chifiriuc, C.M.; Lin, Y.-S. Fabrication, characterization and in vitro profile based interaction with eukaryotic and prokaryotic cells of alginate–chitosan–silica biocomposite. Int. J. Pharm. 2013, 441, 555–561. [Google Scholar] [CrossRef] [PubMed]
- Mamaeva, V.; Sahlgren, C.; Lindén, M. Mesoporous silica nanoparticles in medicine—recent advances. Adv. Drug Deliv. Rev. 2013, 65, 689–702. [Google Scholar] [CrossRef]
- Wang, C.; Zhou, H.; Niu, H.; Ma, X.; Yuan, Y.; Hong, H.; Liu, C. Tannic acid-loaded mesoporous silica for rapid hemostasis and antibacterial activity. Biomater. Sci. 2018, 6, 3318–3331. [Google Scholar] [CrossRef]
- Xia, W.; Grandfield, K.; Hoess, A.; Ballo, A.; Cai, Y.; Engqvist, H. Mesoporous titanium dioxide coating for metallic implants. J. Biomed. Mater. Res. Part B Appl. Biomater. 2012, 100, 82–93. [Google Scholar] [CrossRef]
- Galiano, K.; Pleifer, C.; Engelhardt, K.; Brössner, G.; Lackner, P.; Huck, C.; Lass-Flörl, C.; Obwegeser, A. Silver segregation and bacterial growth of intraventricular catheters impregnated with silver nanoparticles in cerebrospinal fluid drainages. Neurol. Res. 2008, 30, 285–287. [Google Scholar] [CrossRef]
- Della Valle, C.; Visai, L.; Santin, M.; Cigada, A.; Candiani, G.; Pezzoli, D.; Arciola, C.R.; Imbriani, M.; Chiesa, R. A novel antibacterial modification treatment of titanium capable to improve osseointegration. Int. J. Artif. Organs 2012, 35, 864–875. [Google Scholar] [CrossRef]
- Samuel, U.; Guggenbichler, J. Prevention of catheter-related infections: The potential of a new nano-silver impregnated catheter. Int. J. Antimicrob. Agents 2004, 23, 75–78. [Google Scholar] [CrossRef] [PubMed]
- Ramazanzadeh, B.; Jahanbin, A.; Yaghoubi, M.; Shahtahmassbi, N.; Ghazvini, K.; Shakeri, M.; Shafaee, H. Comparison of antibacterial effects of zno and cuo nanoparticles coated brackets against streptococcus mutans. J. Dent. 2015, 16, 200. [Google Scholar]
- Aboelzahab, A.; Azad, A.M.; Dolan, S.; Goel, V. Mitigation of staphylococcus aureus-mediated surgical site infections with ir photoactivated tio2 coatings on ti implants. Adv. Healthc. Mater. 2012, 1, 285–291. [Google Scholar] [CrossRef] [PubMed]
- Oprea, A.E.; Pandel, L.M.; Dumitrescu, A.M.; Andronescu, E.; Grumezescu, V.; Chifiriuc, M.C.; Mogoantă, L.; Bălşeanu, T.-A.; Mogoşanu, G.D.; Socol, G. Bioactive zno coatings deposited by maple—An appropriate strategy to produce efficient anti-biofilm surfaces. Molecules 2016, 21, 220. [Google Scholar] [CrossRef] [PubMed]
- Vaishya, R.; Chauhan, M.; Vaish, A. Bone cement. J. Clin. Orthop. Trauma 2013, 4, 157–163. [Google Scholar] [CrossRef]
- Dunne, N. Bone cement fixation: Acrylic cements. In Joint Replacement Technology; Elsevier: Amsterdam, The Netherlands, 2008; pp. 212–251. [Google Scholar]
- Clyburn, T.A.; Cui, Q. Antibiotic Laden Cement: Current State of the Art. Available online: https://www.jointreplacementassociates.com/pdf/antibiotic-laden-cement.pdf (accessed on 5 December 2018).
- Breusch, S.; Aldinger, P.; Thomsen, M.; Ewerbeck, V.; Lukoschek, M. Anchoring principles in hip endoprostheses. I: Prosthesis stem. Der Unfallchirurg 2000, 103, 918–931. [Google Scholar] [CrossRef]
- Alt, V.; Bechert, T.; Steinrücke, P.; Wagener, M.; Seidel, P.; Dingeldein, E.; Domann, E.; Schnettler, R. An in vitro assessment of the antibacterial properties and cytotoxicity of nanoparticulate silver bone cement. Biomaterials 2004, 25, 4383–4391. [Google Scholar] [CrossRef] [PubMed]
- Shen, S.-C.; Ng, W.K.; Shi, Z.; Chia, L.; Neoh, K.G.; Tan, R.B.H. Mesoporous silica nanoparticle-functionalized poly(methyl methacrylate)-based bone cement for effective antibiotics delivery. J. Mater. Sci. Mater. Med. 2011, 22, 2283. [Google Scholar] [CrossRef]
- Voicu, G.; Grumezescu, V.; Andronescu, E.; Grumezescu, A.M.; Ficai, A.; Ficai, D.; Ghitulica, C.D.; Gheorghe, I.; Chifiriuc, M.C. Caprolactam-silica network, a strong potentiator of the antimicrobial activity of kanamycin against gram-positive and gram-negative bacterial strains. Int. J. Pharm. 2013, 446, 63–69. [Google Scholar] [CrossRef]
- Lee, D.-K.; Kim, S.V.; Limansubroto, A.N.; Yen, A.; Soundia, A.; Wang, C.-Y.; Shi, W.; Hong, C.; Tetradis, S.; Kim, Y. Nanodiamond–gutta percha composite biomaterials for root canal therapy. ACS Nano 2015, 9, 11490–11501. [Google Scholar] [CrossRef]
- Kahlenberg, J.M.; Kaplan, M.J. Little peptide, big effects: The role of ll-37 in inflammation and autoimmune disease. J. Immunol. 2013, 191, 4895–4901. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Yan, C.; Zhang, X.; Shi, D.; Chi, L.; Luo, G.; Deng, J. Antimicrobial peptide modification enhances the gene delivery and bactericidal efficiency of gold nanoparticles for accelerating diabetic wound healing. Biomater. Sci. 2018, 6, 2757–2772. [Google Scholar] [CrossRef] [PubMed]
- Petchiappan, A.; Chatterji, D. Antibiotic resistance: Current perspectives. ACS Omega 2017, 2, 7400–7409. [Google Scholar] [CrossRef] [PubMed]
- Olar, R.; Badea, M.; Marinescu, D.; Chifiriuc, C.-M.; Bleotu, C.; Grecu, M.N.; Iorgulescu, E.E.; Bucur, M.; Lazar, V.; Finaru, A. Prospects for new antimicrobials based on n, n-dimethylbiguanide complexes as effective agents on both planktonic and adhered microbial strains. Eur. J. Med. Chem. 2010, 45, 2868–2875. [Google Scholar] [CrossRef] [PubMed]
- Olar, R.; Badea, M.; Cristurean, E.; Lazar, V.; Cernat, R.; Balotescu, C. Thermal behavior, spectroscopic and biological characterization of co (ii), zn (ii), pd (ii) and pt (ii) complexes with n, n-dimethylbiguanide. J. Therm. Anal. Calorim. 2005, 80, 451–455. [Google Scholar] [CrossRef]
- Badea, M.; Crasanda, A.-M.; Chifiriuc, M.C.; Marutescu, L.; Lazar, V.; Marinescu, D.; Olar, R. Synthesis, spectral and thermal study on new fe (iii) complexes with n, n-dimethylbiguanide as antibacterial agents. J. Therm. Anal. Calorimetry 2013, 111, 1743–1751. [Google Scholar] [CrossRef]
- World Health Organization. Global Tuberculosis Report 2016; WHO: Geneva, Switzerland, 2016. [Google Scholar]
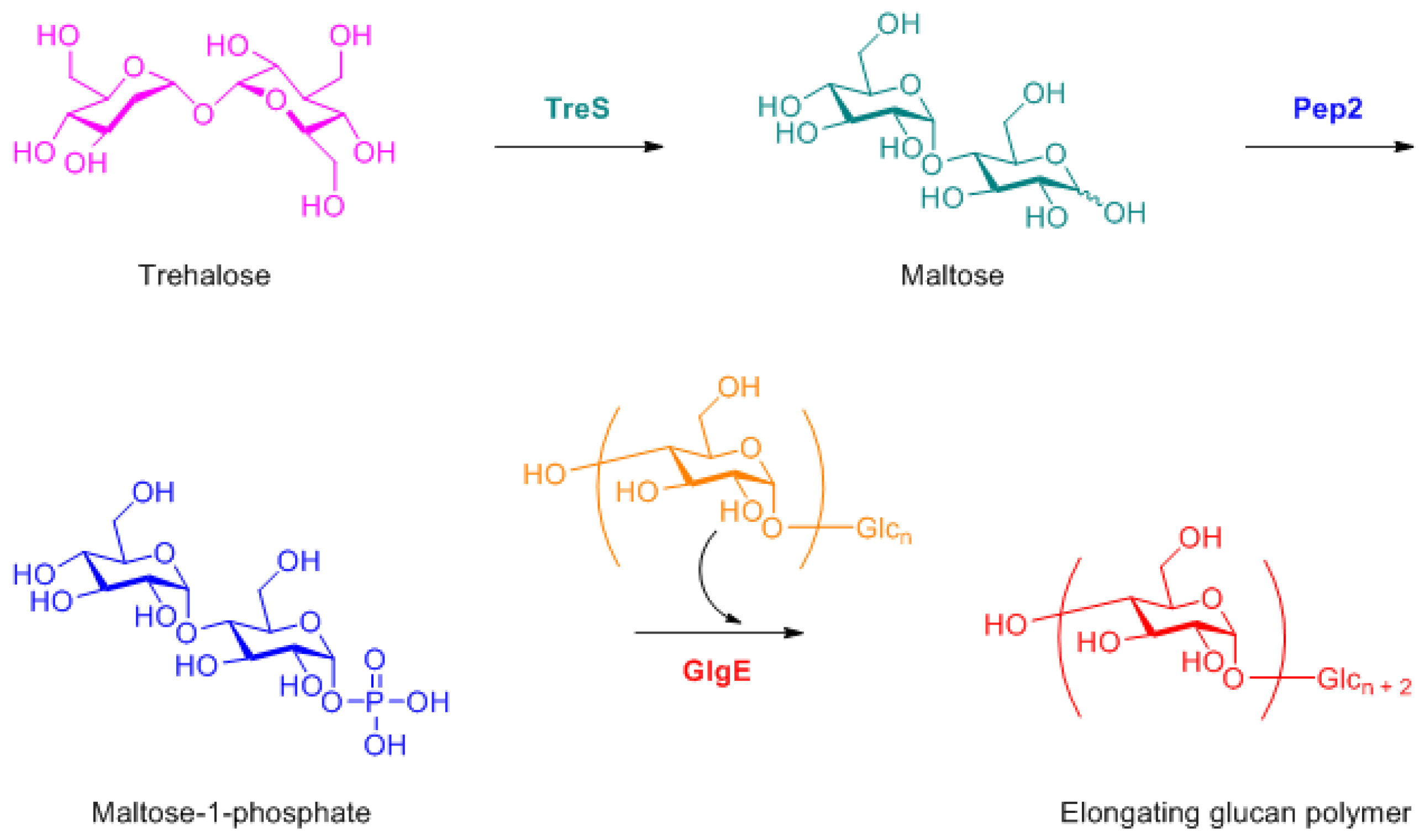
- Kalscheuer, R.; Syson, K.; Veeraraghavan, U.; Weinrick, B.; Biermann, K.E.; Liu, Z.; Sacchettini, J.C.; Besra, G.; Bornemann, S.; Jacobs, W.R., Jr. Self-poisoning of mycobacterium tuberculosis by targeting glge in an α-glucan pathway. Nat. Chem. Biol. 2010, 6, 376. [Google Scholar] [CrossRef] [PubMed]
- Syson, K.; Stevenson, C.E.; Rejzek, M.; Fairhurst, S.A.; Nair, A.; Bruton, C.J.; Field, R.A.; Chater, K.F.; Lawson, D.M.; Bornemann, S. Structure of a streptomyces maltosyltransferase glge: A homologue of a genetically validated anti-tuberculosis target. J. Biol. Chem. 2011. [Google Scholar] [CrossRef] [PubMed]
- Stam, M.R.; Danchin, E.G.; Rancurel, C.; Coutinho, P.M.; Henrissat, B. Dividing the large glycoside hydrolase family 13 into subfamilies: Towards improved functional annotations of α-amylase-related proteins. Protein Eng. Des. Sel. 2006, 19, 555–562. [Google Scholar] [CrossRef] [PubMed]
- Elbein, A.D.; Pastuszak, I.; Tackett, A.J.; Wilson, T.; Pan, Y.T. The last step in the conversion of trehalose to glycogen: A mycobacterial enzyme that transfers maltose from maltose-1-phosphate to glycogen. J. Biol. Chem. 2010. [Google Scholar] [CrossRef] [PubMed]
- Sassetti, C.M.; Boyd, D.H.; Rubin, E.J. Genes required for mycobacterial growth defined by high density mutagenesis. Mol. Microbiol. 2003, 48, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Thanna, S.; Lindenberger, J.J.; Gaitonde, V.V.; Ronning, D.R.; Sucheck, S.J. Synthesis of 2-deoxy-2, 2-difluoro-α-maltosyl fluoride and its x-ray structure in complex with streptomyces coelicolor glgei-v279s. Organ. Biomol. Chem. 2015, 13, 7542–7550. [Google Scholar] [CrossRef] [PubMed]
- Gaitonde, V.; Sucheck, S.J. Antitubercular drugs based on carbohydrate derivatives. In Carbohydrate Chemistry: State of the Art and Challenges for Drug Development: An Overview on Structure, Biological Roles, Synthetic Methods and Application as Therapeutics; World Scientific: Singapore, 2016; pp. 441–478. [Google Scholar]
- Veleti, S.K.; Petit, C.; Ronning, D.R.; Sucheck, S.J. Zwitterionic pyrrolidene-phosphonates: Inhibitors of the glycoside hydrolase-like phosphorylase streptomyces coelicolor glgei-v279s. Organ. Biomol. Chem. 2017, 15, 3884–3891. [Google Scholar] [CrossRef] [PubMed]
- Kapil, S.; Petit, C.; Drago, V.N.; Ronning, D.R.; Sucheck, S.J. Synthesis and in vitro characterization of trehalose-based inhibitors of mycobacterial trehalose 6-phosphate phosphatases. ChemBioChem 2018, 20, 260–269. [Google Scholar] [CrossRef] [PubMed]
- Hassan, A.S.; Masoud, D.M.; Sroor, F.M.; Askar, A.A. Synthesis and biological evaluation of pyrazolo [1, 5-a] pyrimidine-3-carboxamide as antimicrobial agents. Med. Chem. Res. 2017, 26, 2909–2919. [Google Scholar] [CrossRef]
- Abdallah, A.E.; Elgemeie, G.H. Design, synthesis, docking, and antimicrobial evaluation of some novel pyrazolo [1, 5-a] pyrimidines and their corresponding cycloalkane ring-fused derivatives as purine analogs. Drug Des. Dev. Ther. 2018, 12, 1785. [Google Scholar] [CrossRef]
- Catalano, J.G.; Gaitonde, V.; Beesu, M.; Leivers, A.L.; Shotwell, J.B. Phenoxide leaving group s n ar strategy for the facile preparation of 7-amino-3-aryl pyrazolo [1, 5-a] pyrimidines from a 3-bromo-7-phenoxypyrazolo [1, 5-a] pyrimidine intermediate. Tetrahedron Lett. 2015, 56, 6077–6079. [Google Scholar] [CrossRef]
- Bekale, R.B.; Du Plessis, S.-M.; Hsu, N.-J.; Sharma, J.R.; Sampson, S.L.; Jacobs, M.; Meyer, M.; Morse, G.D.; Dube, A. Mycobacterium tuberculosis and interactions with the host immune system: Opportunities for nanoparticle based immunotherapeutics and vaccines. Pharm. Res. 2019, 36, 8. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karmakar, P.; Gaitonde, V. Promising Recent Strategies with Potential Clinical Translational Value to Combat Antibacterial Resistant Surge. Medicines 2019, 6, 21. https://doi.org/10.3390/medicines6010021
Karmakar P, Gaitonde V. Promising Recent Strategies with Potential Clinical Translational Value to Combat Antibacterial Resistant Surge. Medicines. 2019; 6(1):21. https://doi.org/10.3390/medicines6010021
Chicago/Turabian StyleKarmakar, Partha, and Vishwanath Gaitonde. 2019. "Promising Recent Strategies with Potential Clinical Translational Value to Combat Antibacterial Resistant Surge" Medicines 6, no. 1: 21. https://doi.org/10.3390/medicines6010021
APA StyleKarmakar, P., & Gaitonde, V. (2019). Promising Recent Strategies with Potential Clinical Translational Value to Combat Antibacterial Resistant Surge. Medicines, 6(1), 21. https://doi.org/10.3390/medicines6010021




