Antiproliferative and Antioxidant Activities of Two Extracts of the Plant Species Euphorbia dendroides L.
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Plant Material
2.3. Extraction
2.4. HPLC-TOF/MS Analysis
2.5. Phenolic Contents Analysis
2.5.1. Total Phenolic Content (TPC)
2.5.2. Total Flavonoids Content (TFC)
2.6. Determination of Antioxidant Activity
2.6.1. DPPH Radical Scavenging Assay
2.6.2. Reducing Power Assay
2.6.3. The Inhibition of Linoleic Acid Peroxidation
2.7. Determination of In Vitro Antiproliferative Activity
2.7.1. Cell Culture
2.7.2. Cell Proliferation Assays
3. Results and Discussion
3.1. Extraction Yield
3.2. Total Phenolic Compounds and Total Flavonoids
3.3. High Performance Liquid Chromatography Analysis
3.4. Antioxidant Activity
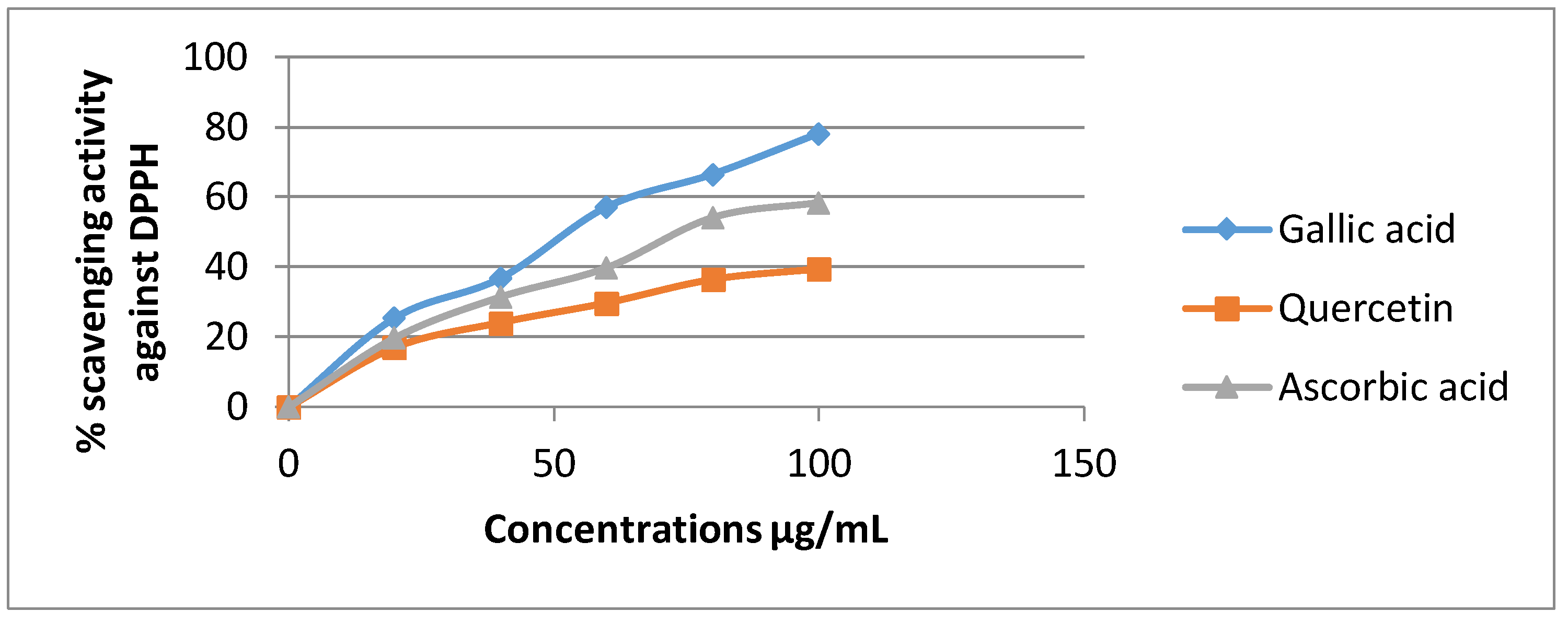
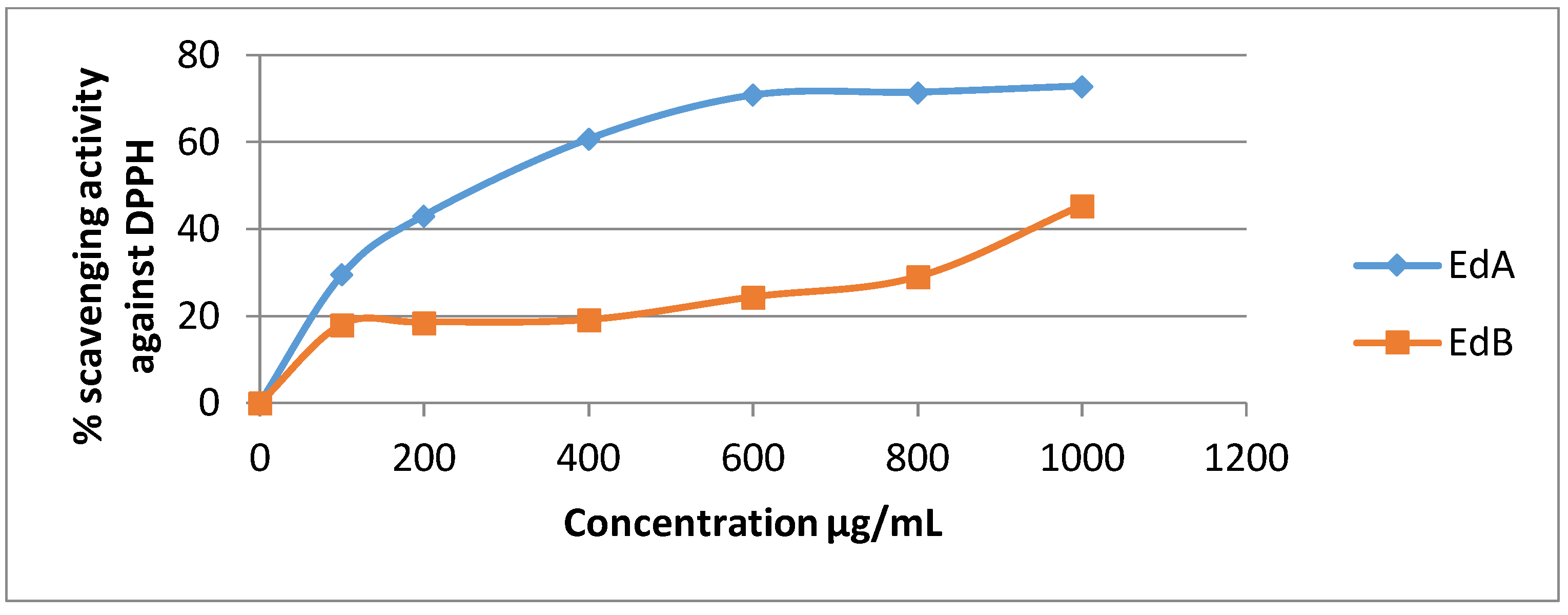
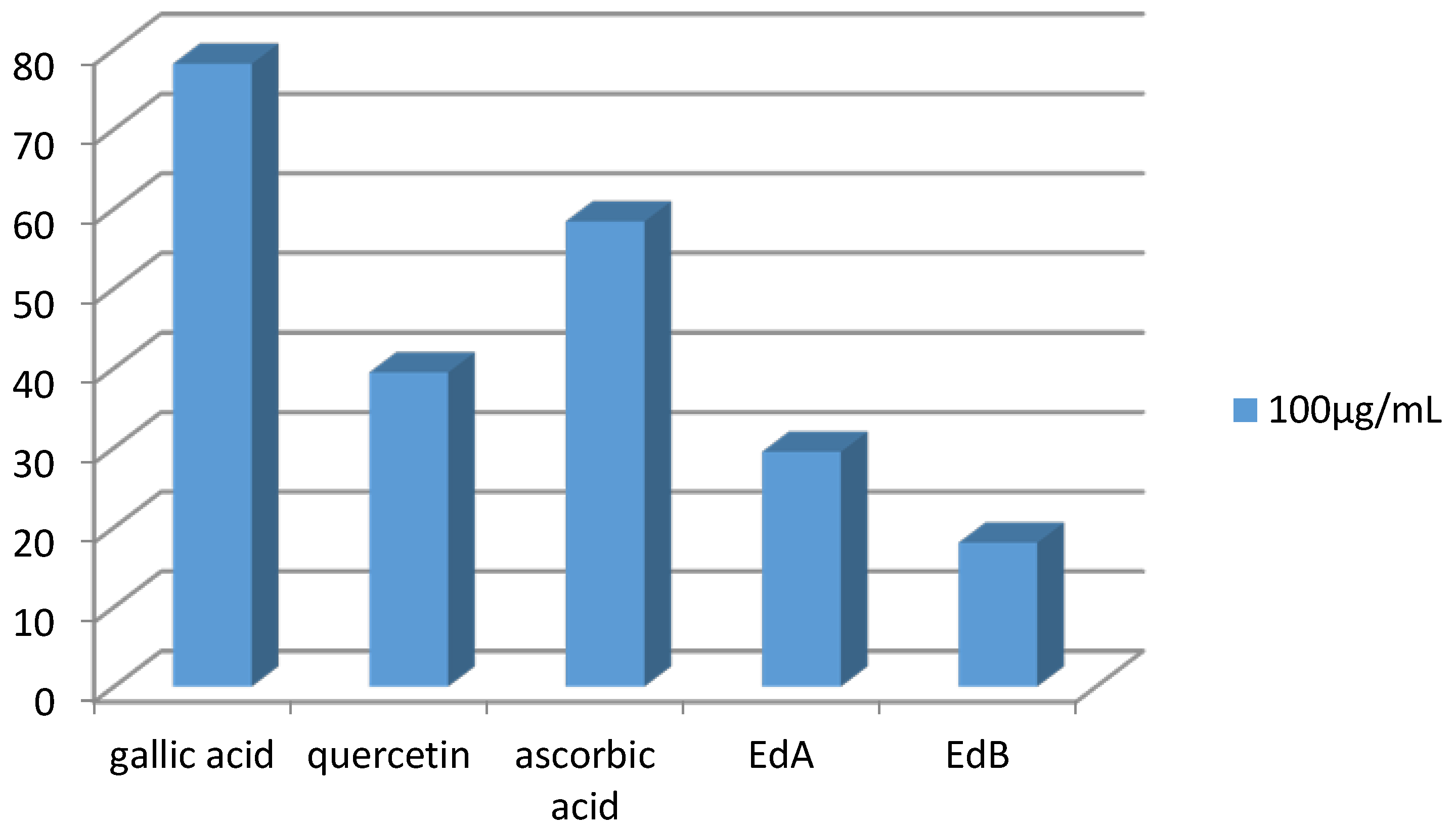
3.4.1. DPPH Radical Assay
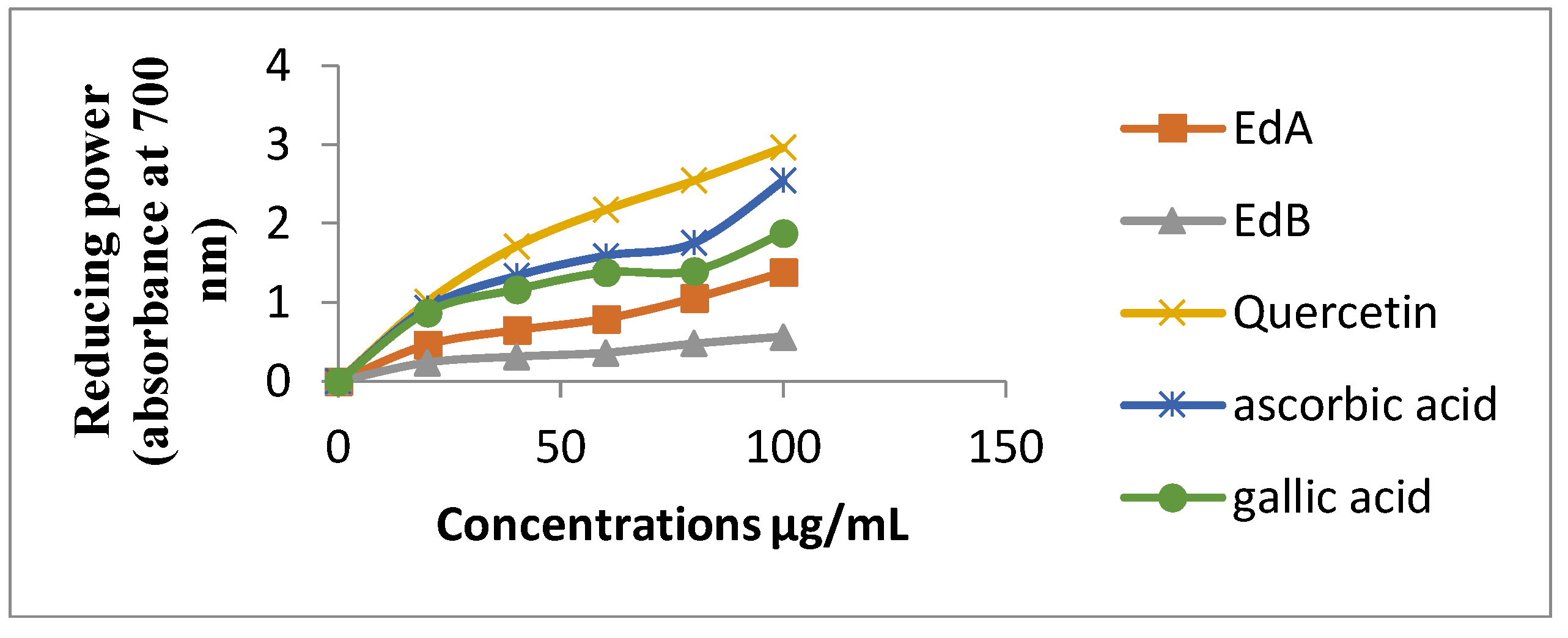
3.4.2. Reducing Power Assay
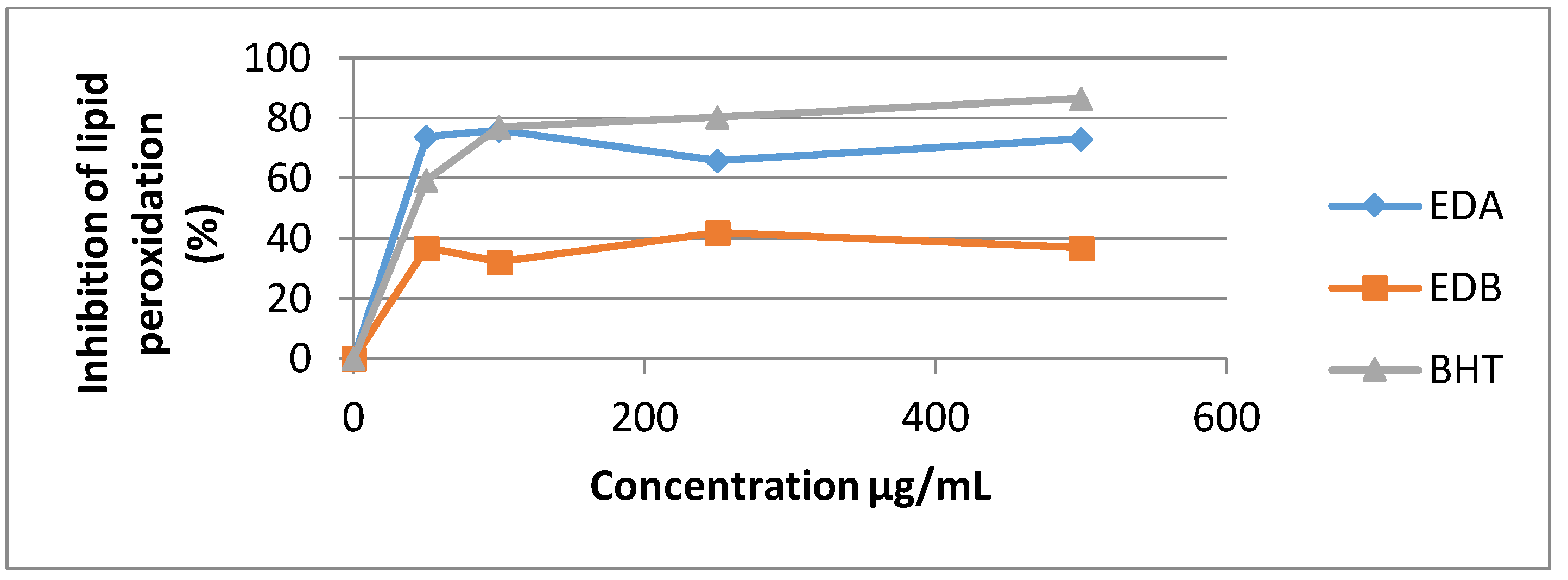
3.4.3. The Inhibition of Linoleic Acid Peroxidation
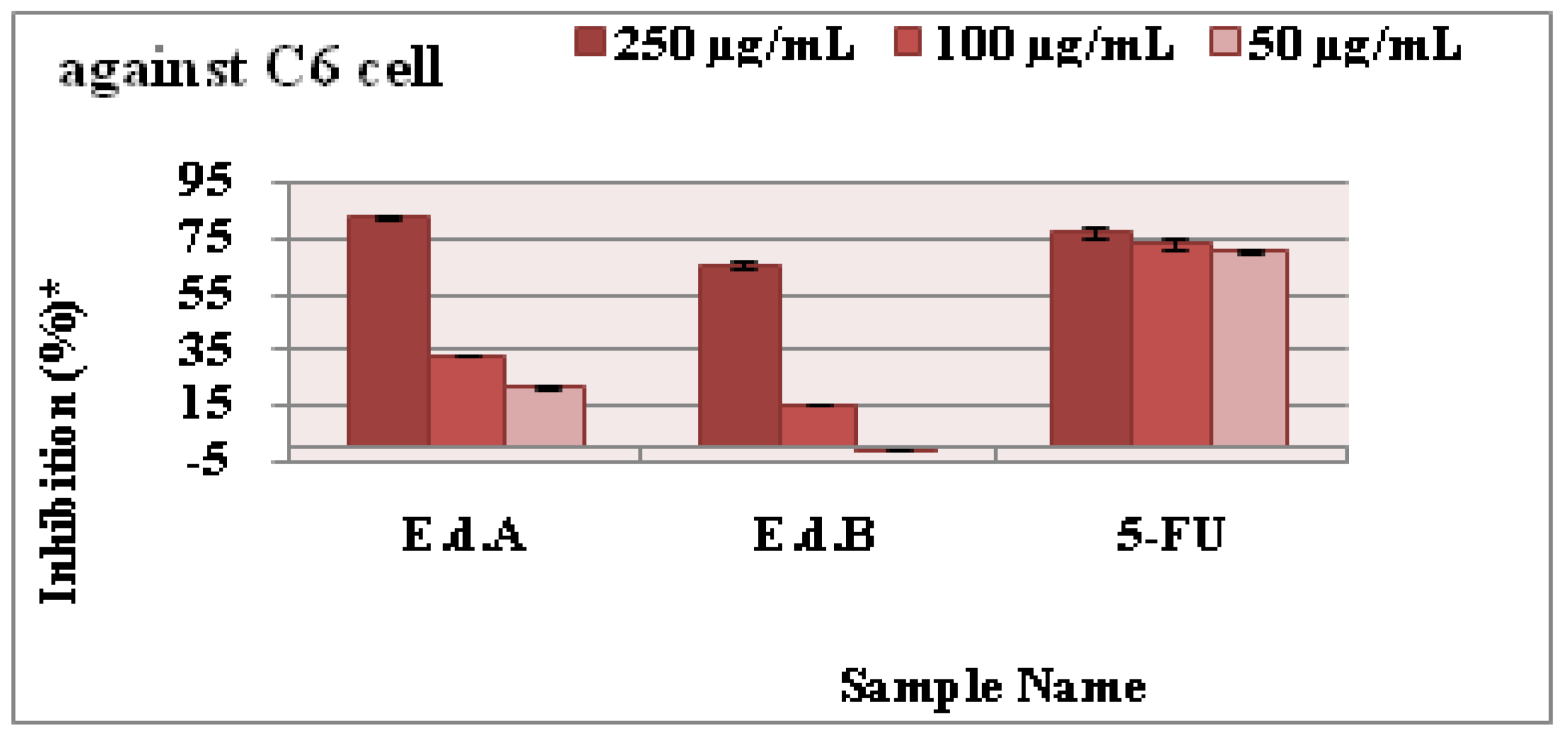
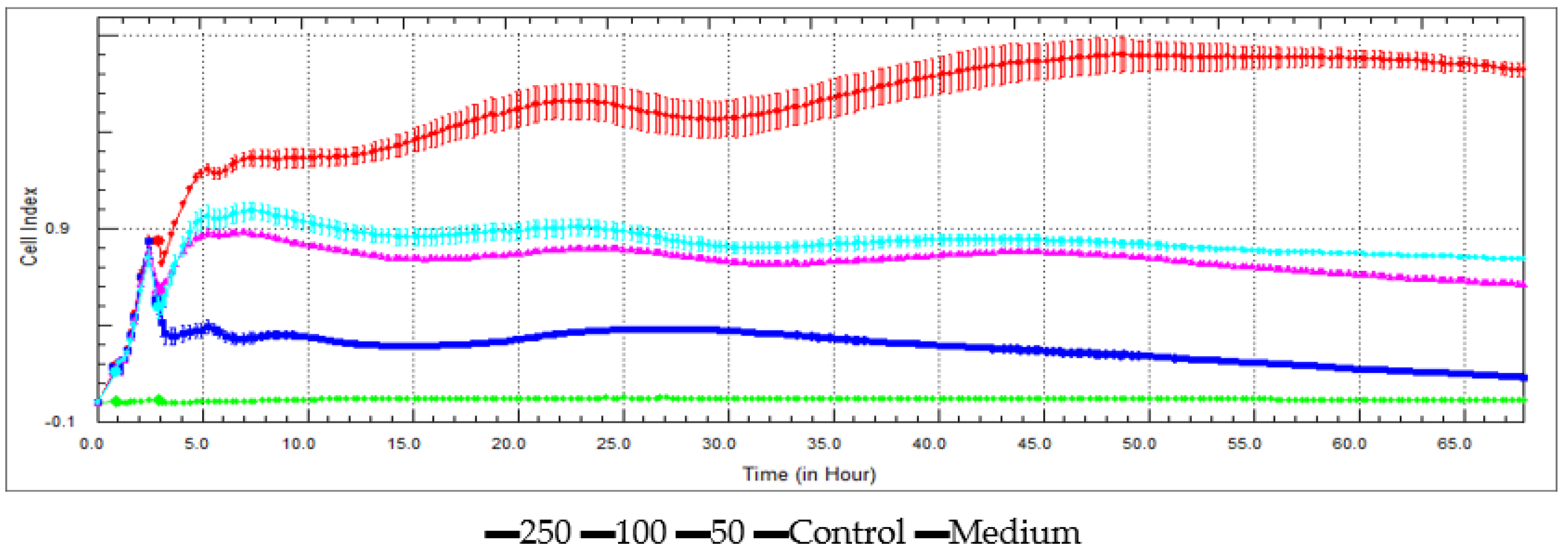
3.5. Antiproliferative Activity
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lobo, V.; Patil, A.; Phatak, A.; Chandra, N. Free radicals, antioxidants and functional foods: Impact on human health. Pharmacogn. Rev. 2010, 4, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Pham-Huy, L.A.; He, H.; Pham-Huy, C. Free Radicals, Antioxidants in Disease and Health. Int. J. Biomed. Sci. 2008, 4, 89–96. [Google Scholar] [PubMed]
- Rahman, K. Studies on free radicals, antioxidants, and co-factors. Clin. Interv. Aging 2007, 2, 219–236. [Google Scholar] [PubMed]
- Kumar, S. The importance of antioxidant and their role in pharmaceutical science—A review. Asian J. Res. Chem. Pharm. Sci. 2014, 1, 27–44. [Google Scholar]
- Carocho, M.; Barreiro, M.F.K.; Morales, P.; Ferreira Isabel, C.F.R. Adding Molecules to Food, Pros and Cons: A Review on Synthetic and Natural Food Additives. Compr. Rev. Food Sci. Food Saf. 2014, 13, 377–399. [Google Scholar] [CrossRef]
- Ksouri, R.; Ksouri, W.M.; Jallali, I.; Debez, A.; Magné, C.; Hiroko, I. Medicinal halophytes: Potent source of health promoting biomolecules with medical, nutraceutical and food applications. Crit. Rev. Biotechnol. 2012, 32, 289–326. [Google Scholar] [CrossRef] [PubMed]
- Martin-Bettolo, G.B. Present aspects of the use of medicinal plants in traditional medicine. J. Ethnopharmacol. 1980, 2, 5–7. [Google Scholar] [CrossRef]
- Othman, A.; Ismail, A.; Ghani, N.A.; Adenan, I. Antioxidant capacity and phenolic content of cocoa beans. Food Chem. 2007, 100, 1523–1530. [Google Scholar] [CrossRef]
- Gómez, C.; Espadaler, X. Curva de dispersión de semillasporhormigas en Euphorbiacharacias L. y Euphorbianicaeensis All. (Euphorbiaceae). Ecol. Mediterr. 1994, 20, 51–59. [Google Scholar]
- Rechinger, K.H.; Schiman-Czeika, H. Euphorbiaceae. In Flora Iranica; AkademischeDruck- und Verlagsanstalt: Graz, Austria, 1964; Volume 6, pp. 1–48. ISBN 978-3-201-00728-3. [Google Scholar]
- Appendino, G.; Szallasi, A. Euphorbium: Modern research on its active principle, resiniferatoxin, revives an ancient medicine. Life Sci. J. 1997, 60, 681–696. [Google Scholar] [CrossRef]
- Singla, A.K.; Pathak, K. Phytoconstituents of Euphorbia species. Fitoterapia 1990, 61, 483–516. [Google Scholar]
- Bani, S.; Kaul, A.; Jaggi, B.S.; Suri, K.A.; Suri, O.P.; Sharma, O.P. Anti-inflammatory activity of the hydrosoluble fraction of Euphorbia royleana latex. Fitoterapia 2000, 71, 655–662. [Google Scholar] [CrossRef]
- Tona, L.; Kambu, K.; Ngimbi, N.; Meisa, K.; Penge, O.; Lusakibanza, M.; Cimanga, K.; de Bruyne, T.; Apers, S.; Totté, J.; et al. Antiamoebic and spasmolytic activities of extracts from some amtidiarrhoeal traditional preparations used in Kinshasa, Congo. Phytomedicine 2000, 7, 31–38. [Google Scholar] [CrossRef]
- Semple, S.J.; Reynolds, G.D.; O’Leary, M.C.; Flower, R.L. Screening of Australian medicinal plants for antiviral activity. J. Ethnopharmacol. 1998, 60, 163–172. [Google Scholar] [CrossRef]
- Shimura, H.; Watanabe, N.; Tamai, M.; Hanada, K.; Takahashi, A.; Tanaka, Y.; Arai, K.; Zhang, P.L.; Chang, R. Hepatoprotective compounds from Canarium album and Euphorbia nematocypha. Chem. Pharm. Bull. (Tokyo) 1990, 38, 2201–2203. [Google Scholar]
- Ernst, M.; Grace, O.M.; Saslis-Lagoudakis, C.H.; Nilsson, N.; Simonsen, H.T.; Rønsted, N. Global medicinal uses of Euphorbia L. (Euphorbiaceae). J. Ethnopharmacol. 2015, 176, 90–101. [Google Scholar] [CrossRef] [PubMed]
- Özbilgin, S.; Citoğlu, G.S.T. Uses of Some Euphorbia Species in Traditional Medicine in Turkey and Their Biological Activities. Turk. J. Pharm. Sci. 2012, 9, 241–256. [Google Scholar]
- Brussell, D.E. Medicinal plants of Mt. Pelion, Greece. Econ. Bot. Spec. Sect. Med. Plants 2004, 58, S174–S202. [Google Scholar] [CrossRef]
- Esposito, M.; Nothias, L.F.; Nedev, H.; Gallard, J.F.; Leyssen, P.; Retailleau, P.; Costa, J.; Roussi, F.; Iorga, B.I.; Paolini, J.; et al. Euphorbia dendroides Latex as a Source of Jatrophane Esters: Isolation, Structural Analysis, Conformational Study, and Anti-CHIKV Activity. J. Nat. Prod. 2016, 79, 2873–2882. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Weller, C.L. Recent Advances in Extraction of Nutraceuticals from Plants. Trends Food Sci. Technol. 2006, 17, 300–312. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Aruoma, O.I. Free radicals, oxidative stress and antioxidants in human health and disease. J. Am. Oil Chem. Soc. 1989, 75, 199–212. [Google Scholar] [CrossRef]
- Masuda, T.; Yonemory, S.; Ouyama, Y.; Takeda, Y.; Tanaka, T.; Andoh, T. Evaluation of the antioxidant activity of environmental plants: Activity of the leaf extracts from seashore plants. J. Agric. Food Chem. 1999, 47, 1749–1754. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.P.; Lu, Y.H.; Wei, D.Z. Antioxidant activity of a flavonoid-rich extract of Hypericum perforatum L. in vitro. J. Agric. Food Chem. 2004, 52, 5032–5039. [Google Scholar] [CrossRef] [PubMed]
- Oyaizu, M. Studies on products of the browning reaction prepared from glucose amine. Jpn. J. Nutr. 1986, 44, 307–315. [Google Scholar] [CrossRef]
- Choi, C.W.; Kim, S.C.; Hwang, S.S.; Choi, B.K.; Ahn, H.J.; Lee, M.Y.; Park, S.H.; Kim, S.K. Antioxidant activity and free radical scavenging capacity between Korean medicinal plants and flavonoids by assay-guided comparison. Plant Sci. 2002, 163, 1161–1168. [Google Scholar] [CrossRef]
- Yaglioglu, A.S.; Demirtas, I.; Goren, N. Bioactivity-guided isolation of antiproliferative compounds from Centaurea carduiformis DC. Phytochem. Lett. 2014, 8, 213–219. [Google Scholar] [CrossRef]
- Demirtas, I.; Yaglioglu, A.S. Bioactive Volatile Content of the Stem and Root of Centaurea carduiformis DC. subsp. carduiformis var. carduiformis. J. Chem. 2012, 2013. [Google Scholar] [CrossRef]
- Koldaş, S.; Demirtas, I.; Ozen, T.; Demirci, M.A.; Behçet, L. Phytochemical screening, anticancer and antioxidant activities of Origanum vulgare L. ssp. viride (Boiss.) Hayek, a plant of traditional usage. J. Sci. Food Agric. 2015, 95, 786–1482. [Google Scholar] [CrossRef] [PubMed]
- Maisuthisakul, P.; Suttajit, M.; Pongsawatmanit, R. Assessment of phenolic content and free radical-scavenging capacity of some Thai indigenous plants. Food Chem. 2007, 4, 1409–1418. [Google Scholar] [CrossRef]
- Prior, R.L.; Cao, G.H. In vivo total antioxidant capacity: Comparison of different analytical methods. Free Radic. Biol. Med. 1999, 27, 1173–1181. [Google Scholar] [CrossRef]
- Xu, B.J.; Chang, S.K. A comparative study on phenolic profiles and antioxidant activities of legumes as affected by extraction solvents. J. Food Sci. 2007, 72, S159–S166. [Google Scholar] [CrossRef] [PubMed]
- Hodgson, J.M.; Croft, K.D. Tea flavonoids and cardiovascular health. Mol. Asp. Med. 2010, 31, 495–502. [Google Scholar] [CrossRef] [PubMed]
- Kanatt, S.R.; Chander, R.; Radhakrishna, P.; Sharma, A. Potato peel extract—A natural antioxidant for retarding lipid peroxidation in radiation processed lamb meat. J. Agric. Food Chem. 2005, 53, 1499–1504. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.Y.; Liu, H.P.; Zhang, Y.C.; Guo, L.Q.; Li, Z.X.; Shi, X.F. Anticancer potential of Euphorbia helioscopia L extracts against human cancer cells. Anat. Rec. (Hoboken) 2012, 295, 223–233. [Google Scholar] [CrossRef] [PubMed]
- Frankel, E. Nutritional benefits of flavonoids. In Proceedings of the International Conference on Food Factors: Chemistry and Cancer Prevention, Hamamatsu, Japan, 10–15 December 1995. [Google Scholar]
- Yildrim, A.; Mavi, A.; Oktay, M.; Kara, A.A.; Algur, F.; Bilaloglu, V. Comparison of antioxidant and antimicrobial activities of tilia (TiliaargenteaDesf ex DC), sage (Salvia triloba L.), and black tea (Camellia sinensis) extracts. J. Agric. Food Chem. 2000, 48, 5030. [Google Scholar] [CrossRef]
- Aktumsek, A.; Zengin, G.; Guler, G.O.; Cakmak, Y.S.; Duran, A. Antioxidant potentials and anticholinesterase activities of methanolic and aqueous extracts of three endemic Centaurea L. species. Food Chem. Toxicol. 2013, 55, 290–296. [Google Scholar] [CrossRef] [PubMed]
- Sarikurkcu, C.; Tepe, B.; Semiz, D.K.; Solak, M.H. Evaluation of metal concentration and antioxidant activity of three edible mushrooms from Mugla, Turkey. Food Chem. Toxicol. 2010, 48, 1230–1233. [Google Scholar] [CrossRef] [PubMed]
- Katsube, T.; Tabata, H.; Ohta, Y.; Yamasaki, Y.; Anuurad, E.; Shiwaku, K.; Yamane, Y. Screening for antioxidant activity in edible plant products: Comparison of low-density lipoprotein oxidation assay, DPPH radical scavenging assay, and Folin-Ciocalteu assay. J. Agric. Food Chem. 2004, 52, 2391–2396. [Google Scholar] [CrossRef] [PubMed]
- Cao, G.H.; Prior, R.L. Comparison of different analytical methods for assessing total antioxidant capacity of human serum. Clin. Chem. 1998, 44, 1309–1315. [Google Scholar] [PubMed]
- Banakar, M.C.; Paramasivan, S.K.; Chattopadhyay, M.B.; Datta, S.; Chakraborty, P.; Chatterjee, M.; Kannan, K.; Thygarajan, E. 1-alpha, 25-dihydroxyvitamin D3 prevents DNA damage and restores antioxidant enzymes in rat hepatocarcinogenesis induced by diethylnitrosamine and promoted by phenobarbital. World J. Gastroenterol. 2004, 10, 1268–1275. [Google Scholar] [CrossRef] [PubMed]
- Esterbauer, H.; Chaur, R.J.; Zollner, H. Chemistry and biochemistry of 4-hydroxynonenal, malonaldehyde and related aldehydes. Free Radic. Biol. Med. 1991, 11, 81–128. [Google Scholar] [CrossRef]
- Miyoshi, N.; Nakamura, Y.; Ueda, Y.; Abe, M.; Ozawa, Y.; Uchida, K.; Osawa, T. Dietary ginger constituents, galanals A and B, are potent apoptosis inducers in Human T lymphoma Jurkat cells. Cancer Lett. 2003, 199, 113–119. [Google Scholar] [CrossRef]
- Wang, J.L.; Liu, K.; Gong, W.Z.; Wang, Q.; Xu, D.T.; Liu, M.F.; Bi, K.L.; Song, Y.F. Anticancer, antioxidant, and antimicrobial activities of anemone (Anemone cathayensis). Food Sci. Biotechnol. 2012, 21, 551–557. [Google Scholar] [CrossRef]

| Extract | Yield (%) |
|---|---|
| Ethyl acetate extract | 0.42% |
| n-Butanol extract | 2.12% |
| Extract | TPC(mg GAE/g of Extract) | TF(mg QE/g of Extract) |
|---|---|---|
| Ethyl acetate | 929.51 ± 20.1 | 26.04 ± 0.32 |
| n-Butanol | 164.25 ± 16.40 | 12.16 ± 0.2 |
| Compounds | RT | EtOAc | n-Butanol |
|---|---|---|---|
| Gallic acid | 2.831 | 614.12 | 481.94 |
| Gentisic acid | 4.358 | 33.61 | 1.25 |
| 4-hydroxybenzoic acid | 5.531 | 9.35 | |
| chlorogenic acid | 5.984 | 642.01 | 4505.78 |
| protocatechuic acid | 6.959 | 7.74 | 3.08 |
| caffeic acid | 7.623 | 67.79 | 2.51 |
| Vanillic acid | 7.796 | 10.88 | 4.05 |
| Rutin | 9.081 | 0.03 | - |
| P-coumaric acid | 9.917 | 11.64 | 10.07 |
| chicoric acid | 10.982 | 1.02 | 3.22 |
| Ferulic acid | 11.082 | 8.81 | 6.84 |
| Hesperidin | 12.284 | - | 1.49 |
| Salicylic acid | 13.284 | -- | 9.70 |
| Resveratrol | 14.682 | - | 42.31 |
| Cinnamic acid | 15.779 | -- | 10.70 |
| Inhibition Concentration | Ethyl Acetate | N-Butanol |
|---|---|---|
| IC50 (µg/mL) | 119.49 | 151.18 |
| IC75 (µg/mL) | 185.74 | 200.62 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghout, A.; Zellagui, A.; Gherraf, N.; Demirtas, I.; Ayse Sahin, Y.; Boukhenaf, M.; Lahouel, M.; Nieto, G.; Akkal, S. Antiproliferative and Antioxidant Activities of Two Extracts of the Plant Species Euphorbia dendroides L. Medicines 2018, 5, 36. https://doi.org/10.3390/medicines5020036
Ghout A, Zellagui A, Gherraf N, Demirtas I, Ayse Sahin Y, Boukhenaf M, Lahouel M, Nieto G, Akkal S. Antiproliferative and Antioxidant Activities of Two Extracts of the Plant Species Euphorbia dendroides L. Medicines. 2018; 5(2):36. https://doi.org/10.3390/medicines5020036
Chicago/Turabian StyleGhout, Agena, Amar Zellagui, Noureddine Gherraf, Ibrahim Demirtas, Yaglioglu Ayse Sahin, Meriem Boukhenaf, Mesbah Lahouel, Gema Nieto, and Salah Akkal. 2018. "Antiproliferative and Antioxidant Activities of Two Extracts of the Plant Species Euphorbia dendroides L." Medicines 5, no. 2: 36. https://doi.org/10.3390/medicines5020036
APA StyleGhout, A., Zellagui, A., Gherraf, N., Demirtas, I., Ayse Sahin, Y., Boukhenaf, M., Lahouel, M., Nieto, G., & Akkal, S. (2018). Antiproliferative and Antioxidant Activities of Two Extracts of the Plant Species Euphorbia dendroides L. Medicines, 5(2), 36. https://doi.org/10.3390/medicines5020036






