Systematic Assessment of Exposure Variations on Observed Bioactivity in Zebrafish Chemical Screening
Abstract
1. Introduction
2. Materials and Methods
2.1. Test Agent Selection
2.2. Zebrafish Husbandry and Embryo Collection
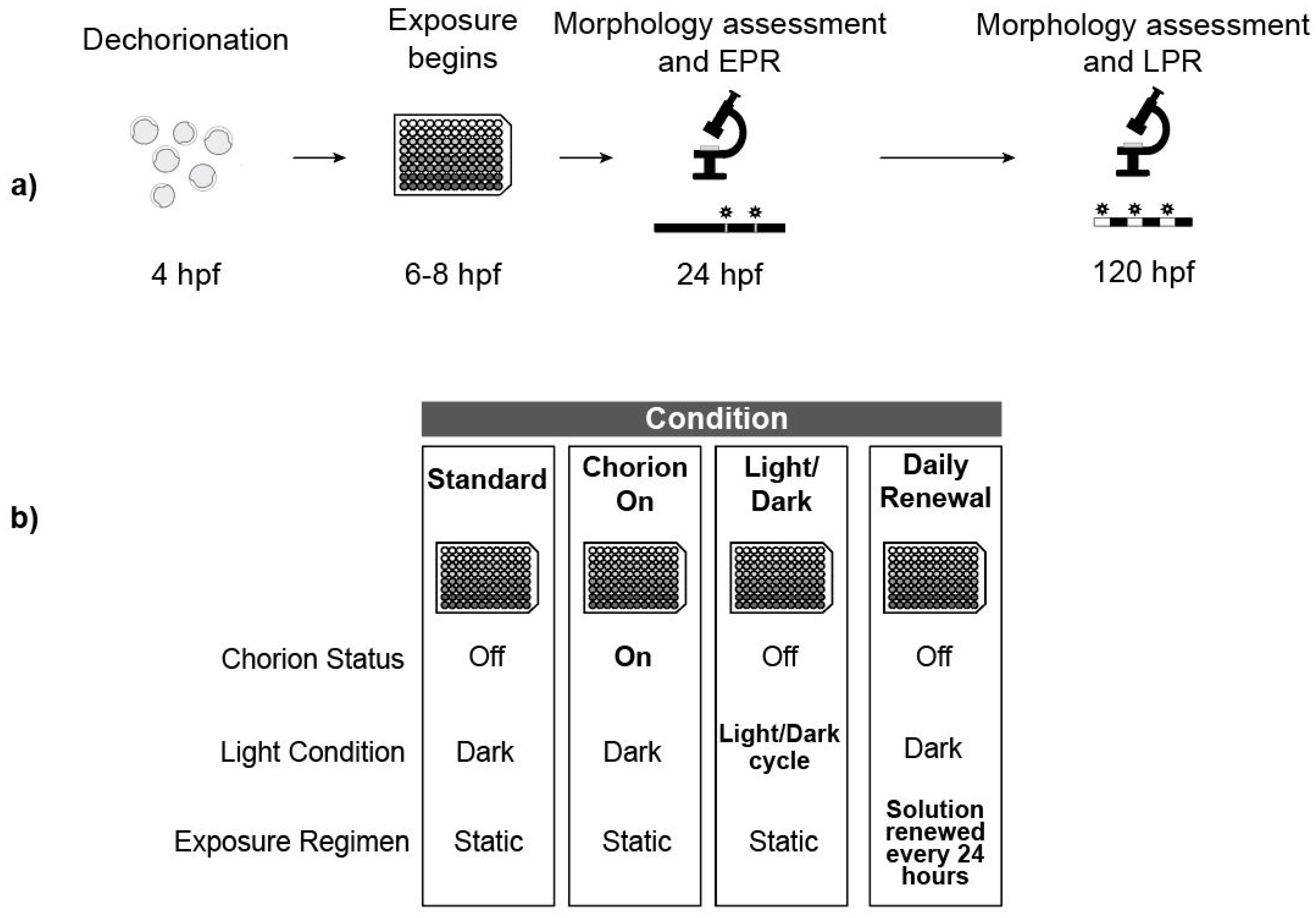
2.3. Embryo Dechorionation and Plating
2.4. Chemical Exposure
2.5. MWCNT Exposure
2.6. Standard Exposure Regimen
2.7. Photomotor Responses
2.8. Experiment Overview
2.9. Daily Solution Renewals
2.10. Data Analysis
3. Results and Discussion
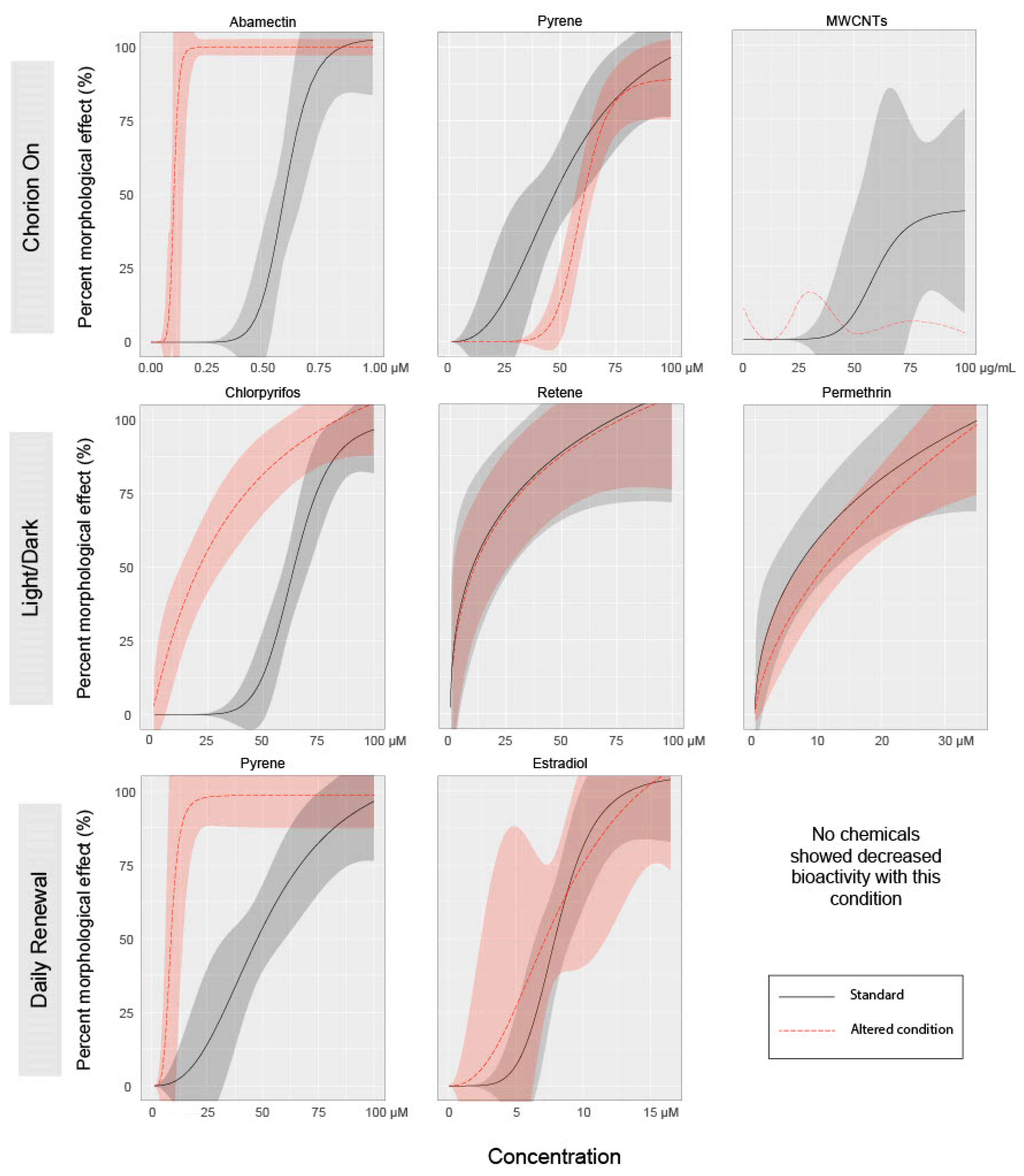
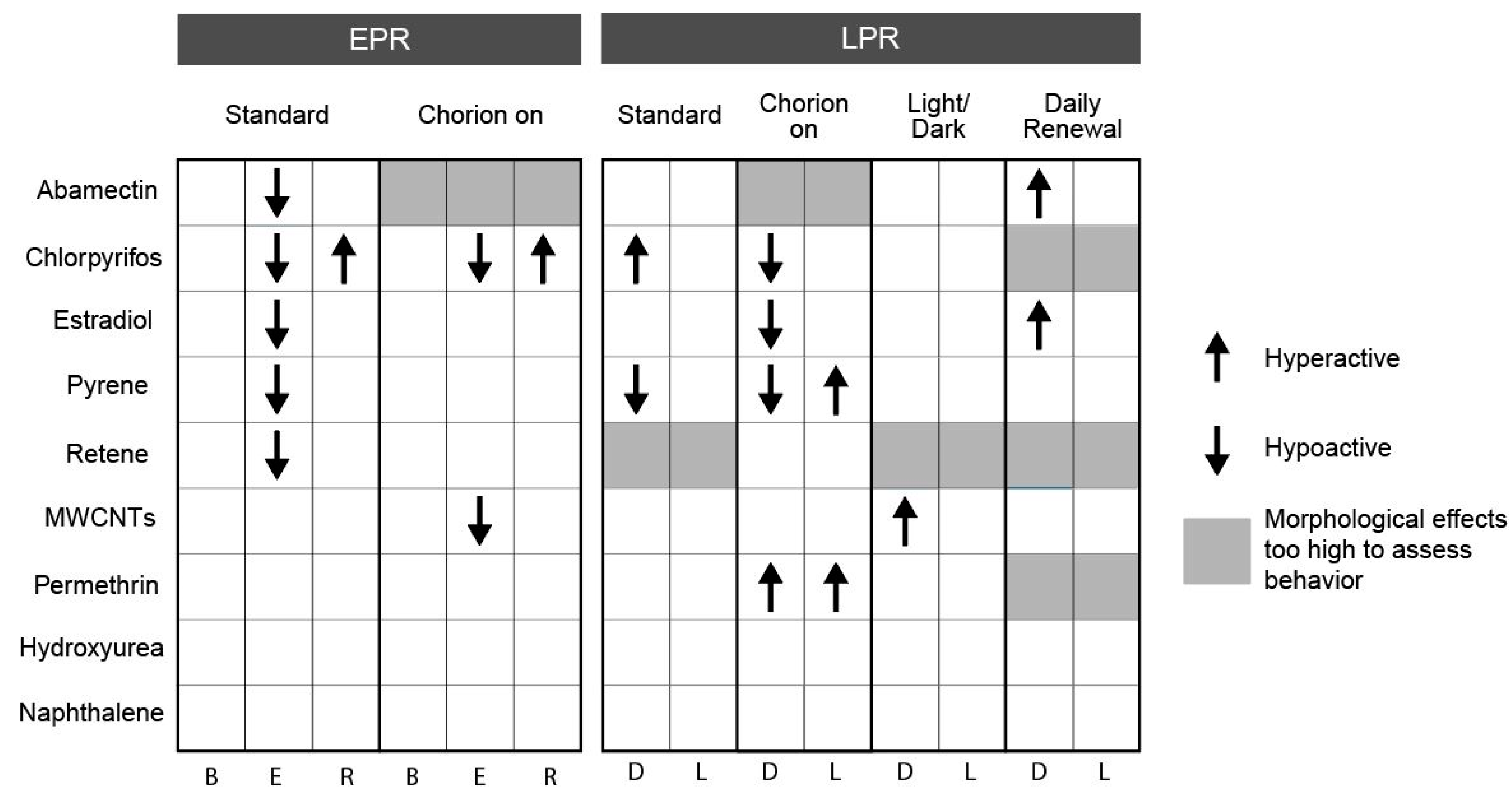
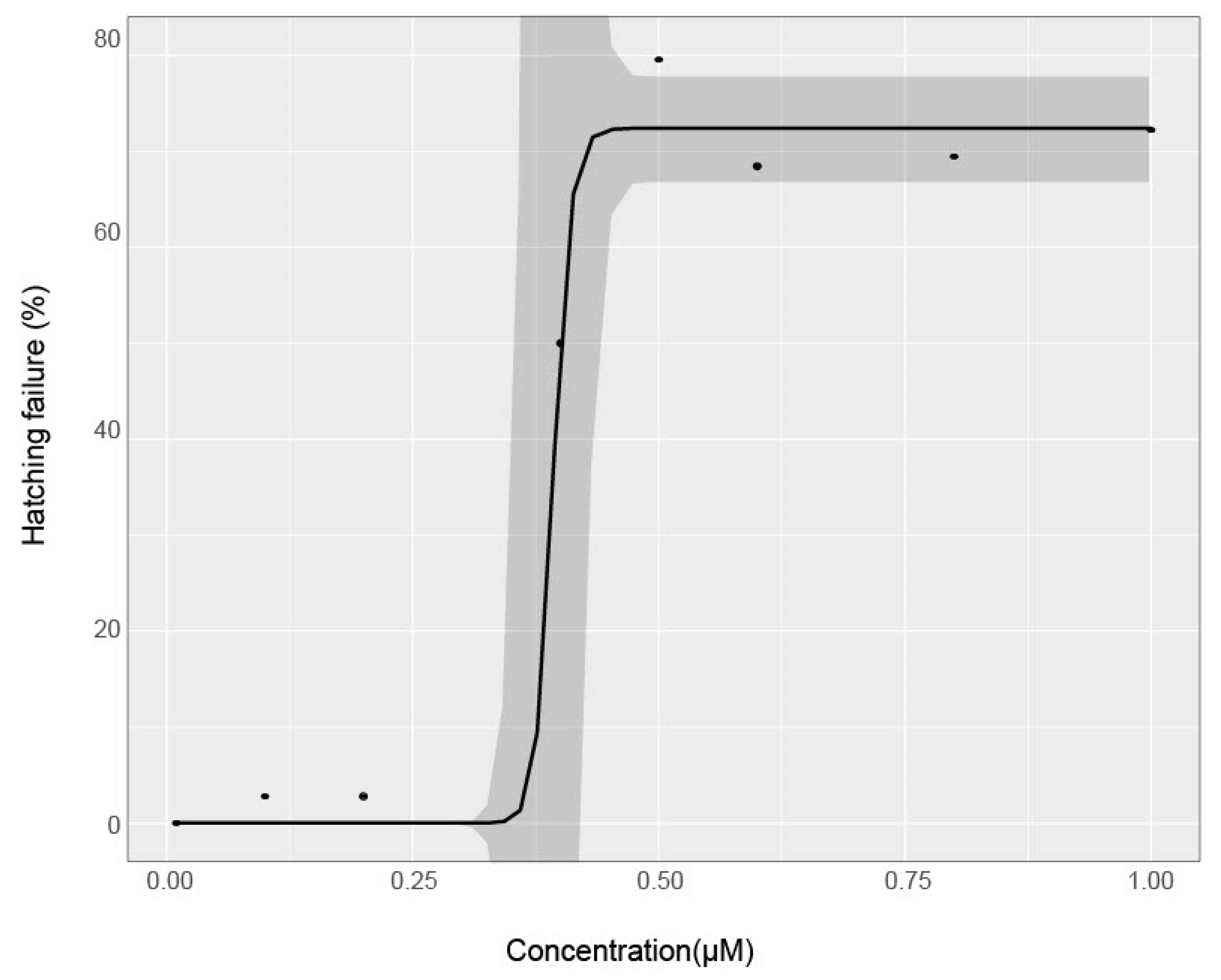
3.1. Effect of Chorion Status on Bioactivity
3.2. Effect of Light Status on Bioactivity
3.3. Effect of Exposure Regimen on Bioactivity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Judson, R.; Richard, A.; Dix, D.J.; Houck, K.; Martin, M.; Kavlock, R.; Dellarco, V.; Henry, T.; Holderman, T.; Sayre, P.; et al. The toxicity data landscape for environmental chemicals. Environ. Health Perspect. 2009, 117, 685–695. [Google Scholar] [CrossRef] [PubMed]
- Russell, W.M. The development of the three Rs concept. Altern. Lab. Anim. 1995, 23, 298–304. [Google Scholar] [CrossRef] [PubMed]
- MacArthur Clark, J. The 3Rs in research: A contemporary approach to replacement, reduction and refinement. Br. J. Nutr. 2018, 120 (Suppl. 1), S1–S7. [Google Scholar] [CrossRef] [PubMed]
- Falcao, M.A.P.; de Souza, L.S.; Dolabella, S.S.; Guimaraes, A.G.; Walker, C.I.B. Zebrafish as an alternative method for determining the embryo toxicity of plant products: A systematic review. Environ. Sci. Pollut. Res. Int. 2018, 25, 35015–35026. [Google Scholar] [CrossRef] [PubMed]
- Bugel, S.M.; Tanguay, R.L.; Planchart, A. Zebrafish: A marvel of high-throughput biology for 21(st) century toxicology. Curr. Environ. Health Rep. 2014, 1, 341–352. [Google Scholar] [CrossRef] [PubMed]
- Lammer, E.; Carr, G.J.; Wendler, K.; Rawlings, J.M.; Belanger, S.E.; Braunbeck, T. Is the fish embryo toxicity test (FET) with the zebrafish (Danio rerio) a potential alternative for the fish acute toxicity test? Comp. Biochem. Physiol. C Toxicol. Pharm. 2009, 149, 196–209. [Google Scholar] [CrossRef]
- Truong, L.; Bugel, S.M.; Chlebowski, A.; Usenko, C.Y.; Simonich, M.T.; Simonich, S.L.; Tanguay, R.L. Optimizing multi-dimensional high throughput screening using zebrafish. Reprod. Toxicol. 2016, 65, 139–147. [Google Scholar] [CrossRef]
- Truong, L.; Harper, S.L.; Tanguay, R.L. Evaluation of embryotoxicity using the zebrafish model. Methods Mol. Biol. 2011, 691, 271–279. [Google Scholar]
- Kimmel, C.B.; Ballard, W.W.; Kimmel, S.R.; Ullmann, B.; Schilling, T.F. Stages of embryonic development of the zebrafish. Dev. Dyn. Off. Publ. Am. Assoc. Anat. 1995, 203, 253–310. [Google Scholar] [CrossRef]
- Tseng, H.P.; Hseu, T.H.; Buhler, D.R.; Wang, W.D.; Hu, C.H. Constitutive and xenobiotics-induced expression of a novel CYP3A gene from zebrafish larva. Toxicol. Appl. Pharmacol. 2005, 205, 247–258. [Google Scholar] [CrossRef]
- Cassar, S.; Adatto, I.; Freeman, J.L.; Gamse, J.T.; Iturria, I.; Lawrence, C.; Muriana, A.; Peterson, R.T.; Van Cruchten, S.; Zon, L.I. Use of Zebrafish in Drug Discovery Toxicology. Chem. Res. Toxicol. 2020, 33, 95–118. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, J.L.; Oris, J.T. Altered gene expression: A mechanism for reproductive toxicity in zebrafish exposed to benzo[a]pyrene. Aquat. Toxicol. 2006, 78, 332–340. [Google Scholar] [CrossRef] [PubMed]
- Parsons, A.; Lange, A.; Hutchinson, T.H.; Miyagawa, S.; Iguchi, T.; Kudoh, T.; Tyler, C.R. Molecular mechanisms and tissue targets of brominated flame retardants, BDE-47 and TBBPA, in embryo-larval life stages of zebrafish (Danio rerio). Aquat. Toxicol. 2019, 209, 99–112. [Google Scholar] [CrossRef] [PubMed]
- Teng, M.; Zhu, W.; Wang, D.; Yan, J.; Qi, S.; Song, M.; Wang, C. Acute exposure of zebrafish embryo (Danio rerio) to flutolanil reveals its developmental mechanism of toxicity via disrupting the thyroid system and metabolism. Environ. Pollut. 2018, 242 Pt B, 1157–1165. [Google Scholar] [CrossRef]
- Alsop, D.; Wood, C.M. Metal and pharmaceutical mixtures: Is ion loss the mechanism underlying acute toxicity and widespread additive toxicity in zebrafish? Aquat. Toxicol. 2013, 140–141, 257–267. [Google Scholar] [CrossRef]
- Dooley, K.; Zon, L.I. Zebrafish: A model system for the study of human disease. Curr. Opin. Genet. Dev. 2000, 10, 252–256. [Google Scholar] [CrossRef]
- BARUT, B.A.; ZON, L.I. Realizing the potential of zebrafish as a model for human disease. Physiol. Genom. 2000, 2, 49–51. [Google Scholar] [CrossRef]
- Zon, L.I. Zebrafish: A New Model for Human Disease. Genome Res. 1999, 9, 99–100. [Google Scholar]
- OECD. Test No. 236: Fish Embryo Acute Toxicity (FET) Test, OECD Guidelines for the Testing of Chemicals; OECD Publishing: Paris, France, 2013. [Google Scholar]
- Hamm, J.; Behl, M.; Ceger, P.; Haendel, M.; Marvel, S.; Maull, E.A.; Muriana, A.; Reif, D.; Tanguay, R.L.; Thessen, A.; et al. The National Toxicology Program’s Systematic Evaluation of the Application of Zebrafish in Toxicology: SEAZIT. Birth Defects Res. 2019, 111, 520. [Google Scholar]
- Hamm, J.T.; Ceger, P.; Allen, D.; Stout, M.; Maull, E.A.; Baker, G.; Zmarowski, A.; Padilla, S.; Perkins, E.; Planchart, A.; et al. Characterizing sources of variability in zebrafish embryo screening protocols. ALTEX 2019, 36, 103–120. [Google Scholar] [CrossRef]
- Kent, M.L.; Buchner, C.; Watral, V.G.; Sanders, J.L.; Ladu, J.; Peterson, T.S.; Tanguay, R.L. Development and maintenance of a specific pathogen-free (SPF) zebrafish research facility for Pseudoloma neurophilia. Dis. Aquat. Organ. 2011, 95, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Barton, C.L.; Johnson, E.W.; Tanguay, R.L. Facility Design and Health Management Program at the Sinnhuber Aquatic Research Laboratory. Zebrafish 2016, 13 (Suppl. 1), S39–S43. [Google Scholar] [CrossRef] [PubMed]
- Westerfield, M. The Zebrafish Book: A Guide for the Laboratory Use of Zebrafish. 2000. Available online: http://zfin.org/zf_info/zfbook/zfbk.html (accessed on 18 September 2020).
- Mandrell, D.; Truong, L.; Jephson, C.; Sarker, M.R.; Moore, A.; Lang, C.; Simonich, M.T.; Tanguay, R.L. Automated zebrafish chorion removal and single embryo placement: Optimizing throughput of zebrafish developmental toxicity screens. J. Lab. Autom. 2012, 17, 66–74. [Google Scholar] [CrossRef]
- Reif, D.M.; Truong, L.; Mandrell, D.; Marvel, S.; Zhang, G.; Tanguay, R.L. High-throughput characterization of chemical-associated embryonic behavioral changes predicts teratogenic outcomes. Arch. Toxicol. 2016, 90, 1459–1470. [Google Scholar] [CrossRef]
- Kokel, D.; Dunn, T.W.; Ahrens, M.B.; Alshut, R.; Cheung, C.Y.; Saint-Amant, L.; Bruni, G.; Mateus, R.; van Ham, T.J.; Shiraki, T.; et al. Identification of nonvisual photomotor response cells in the vertebrate hindbrain. J. Neurosci. 2013, 33, 3834–3843. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Truong, L.; Tanguay, R.L.; Reif, D.M. A New Statistical Approach to Characterize Chemical-Elicited Behavioral Effects in High-Throughput Studies Using Zebrafish. PLoS ONE 2017, 12, e0169408. [Google Scholar] [CrossRef]
- Ritz, C.; Baty, F.; Streibig, J.C.; Gerhard, D. Dose-Response Analysis Using R. PLoS ONE 2015, 10, e0146021. [Google Scholar] [CrossRef]
- Slowikowski, K. ggrepel: Automatically Position Non-Overlapping Text Labels with ‘ggplot2’. R Package Version 0.8.2. 2020. Available online: https://CRAN.R-project.org/package=ggrepel (accessed on 18 September 2020).
- Murata, K.; Conte, F.S.; McInnis, E.; Fong, T.H.; Cherr, G.N. Identification of the origin and localization of chorion (egg envelope) proteins in an ancient fish, the white sturgeon, Acipenser transmontanus. Biol. Reprod. 2014, 90, 132. [Google Scholar] [CrossRef]
- Panzica-Kelly, J.M.; Zhang, C.X.; Augustine-Rauch, K.A. Optimization and Performance Assessment of the Chorion-Off [Dechorinated] Zebrafish Developmental Toxicity Assay. Toxicol. Sci. 2015, 146, 127–134. [Google Scholar] [CrossRef]
- Kim, K.T.; Tanguay, R.L. The role of chorion on toxicity of silver nanoparticles in the embryonic zebrafish assay. Environ. Health Toxicol. 2014, 29, e2014021. [Google Scholar] [CrossRef]
- Tran, C.M.; Lee, H.; Lee, B.; Ra, J.S.; Kim, K.T. Effects of the chorion on the developmental toxicity of organophosphate esters in zebrafish embryos. J. Hazard. Mater. 2020, 401, 123389. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.J.; Nallathamby, P.D.; Browning, L.M.; Osgood, C.J.; Xu, X.H. In vivo imaging of transport and biocompatibility of single silver nanoparticles in early development of zebrafish embryos. ACS Nano 2007, 1, 133–143. [Google Scholar] [CrossRef] [PubMed]
- Pelka, K.E.; Henn, K.; Keck, A.; Sapel, B.; Braunbeck, T. Size does matter—Determination of the critical molecular size for the uptake of chemicals across the chorion of zebrafish (Danio rerio) embryos. Aquat. Toxicol. 2017, 185, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Fent, K.; Weisbrod, C.J.; Wirth-Heller, A.; Pieles, U. Assessment of uptake and toxicity of fluorescent silica nanoparticles in zebrafish (Danio rerio) early life stages. Aquat. Toxicol. 2010, 100, 218–228. [Google Scholar] [CrossRef]
- Ali, M.K.; Saber, S.P.; Taite, D.R.; Emadi, S.; Iriving, R. The Protective Layer of Zebrafish Embryo Changes Continuously with Advancing Age of Embryo Development (AGED). J. Toxicol. Pharmacol. 2017, 1, 009. [Google Scholar]
- Finn, R.N. The physiology and toxicology of salmonid eggs and larvae in relation to water quality criteria. Aquat. Toxicol. 2007, 81, 337–354. [Google Scholar] [CrossRef]
- Mizell, M.; Romig, E.S. The aquatic vertebrate embryo as a sentinel for toxins: Zebrafish embryo dechorionation and perivitelline space microinjection. Int. J. Dev. Biol. 1997, 41, 411–423. [Google Scholar]
- Horzmann, K.A.; Portales, A.M.; Batcho, K.G.; Freeman, J.L. Developmental toxicity of trichloroethylene in zebrafish (Danio rerio). Environ. Sci. Process. Impacts 2020, 22, 728–739. [Google Scholar] [CrossRef]
- Huang, D.; Li, H.; He, Q.; Yuan, W.; Chen, Z.; Yang, H. Developmental Toxicity of Diethylnitrosamine in Zebrafish Embryos/Juveniles Related to Excessive Oxidative Stress. Water Air Soil Pollut. 2018, 229, 81. [Google Scholar] [CrossRef]
- Hurd, M.W.; Cahill, G.M. Entraining Signals Initiate Behavioral Circadian Rhythmicity in Larval Zebrafish. J. Biol. Rhythm. 2002, 17, 307–314. [Google Scholar] [CrossRef]
- Cahill, G.M.; Hurd, M.W.; Batchelor, M.M. Circadian rhythmicity in the locomotor activity of larval zebrafish. Neuroreport 1998, 9, 3445–3449. [Google Scholar] [CrossRef] [PubMed]
- Peyric, E.; Moore, H.A.; Whitmore, D. Circadian clock regulation of the cell cycle in the zebrafish intestine. PLoS ONE 2013, 8, e73209. [Google Scholar] [CrossRef] [PubMed]
- Laranjeiro, R.; Tamai, T.K.; Letton, W.; Hamilton, N.; Whitmore, D. Circadian Clock Synchronization of the Cell Cycle in Zebrafish Occurs through a Gating Mechanism Rather Than a Period-phase Locking Process. J. Biol. Rhythm. 2018, 33, 137–150. [Google Scholar] [CrossRef] [PubMed]
- Whitmore, D.; Foulkes, N.S.; Sassone-Corsi, P. Light acts directly on organs and cells in culture to set the vertebrate circadian clock. Nature 2000, 404, 87–91. [Google Scholar] [CrossRef] [PubMed]
- Whitmore, D.; Foulkes, N.S.; Strahle, U.; Sassone-Corsi, P. Zebrafish Clock rhythmic expression reveals independent peripheral circadian oscillators. Nat. Neurosci. 1998, 1, 701–707. [Google Scholar] [CrossRef]
- Carr, A.J.; Whitmore, D. Imaging of single light-responsive clock cells reveals fluctuating free-running periods. Nat. Cell Biol. 2005, 7, 319–321. [Google Scholar] [CrossRef]
- Yang, D.; Lauridsen, H.; Buels, K.; Chi, L.H.; La Du, J.; Bruun, D.A.; Olson, J.R.; Tanguay, R.L.; Lein, P.J. Chlorpyrifos-oxon disrupts zebrafish axonal growth and motor behavior. Toxicol. Sci. 2011, 121, 146–159. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |

| Test Material | Category | CAS # | Original Supplier | Purity (%) | MW (g/mol) | Log KOW |
|---|---|---|---|---|---|---|
| Abamectin | Pesticide | 71751–41–2 | Sigma-Aldrich | 94.16 | 873.1 | 4.400 |
| Chlorpyrifos | Pesticide | 2921–88–2 | Toronto Research Chemicals | 99.57 | 350.6 | 4.960 |
| Estradiol | Hormone | 50–28–2 | Spectrum Chemical Mfg. Corp. * | 99.15 | 272.4 | 4.010 |
| Hydroxyurea | Pharmaceutical | 127–07–1 | Sigma-Aldrich | 100.0 | 76.06 | −1.800 |
| Naphthalene | Polycyclic aromatic hydrocarbon | 91–20–3 | Sigma-Aldrich † | 99.90 | 128.2 | 3.300 |
| Permethrin | Pesticide | 52645–53–1 | Chem Service, Inc. * | 100.0 | 391.3 | 6.500 |
| Pyrene | Polycyclic aromatic hydrocarbon | 129–00–0 | Thermo Fisher * | 98.10 | 202.3 | 4.880 |
| Retene | Polycyclic aromatic hydrocarbon | 483–65–8 | Santa Cruz Biotech † | 93.00 | 234.3 | 6.400 |
| Multi-walled carbon nanotubes < 7 nm (MWCNT) | 2D Nanomaterial | 99685–96–8 | US Research Nanomaterials, Inc. | >97 | ------ | NA |
| Chemical | Concentrations Tested |
|---|---|
| Abamectin | 0, 0.1, 0.2, 0.4, 0.5, 0.6, 0.8, 1 µM |
| Chlorpyrifos | 0, 2.54, 10, 20, 40, 60, 80, 100 µM |
| Estradiol | 0, 1, 2.54, 5, 7, 9, 12, 16.4 µM |
| Hydroxyurea | 0, 1, 2.54, 6.45, 16.4, 35, 74.8, 100 µM |
| Naphthalene | 0, 1, 2.54, 6.45, 16.4, 35, 74.8, 100 µM |
| Permethrin | 0, 1, 2.54, 5, 8, 12, 16.4, 35 µM |
| Pyrene | 0, 1, 5, 16.5, 30, 50, 65, 100 µM |
| Retene | 0, 1, 5, 20, 30, 45, 65, 100 µM |
| MWCNT | 0, 10, 23.2, 50, 75, 100 µg/mL |
| Morphological Endpoints Assessed | |
|---|---|
| 24 hpf | mortality, delayed progression, spontaneous movement, notochord malformations |
| 120 hpf | mortality, yolk sac edema, pericardial edema, bent body axis, touch response, hatching failure (chorion-on condition only), and malformations of the eye, snout, jaw, otic vesicle, brain, somite, pectoral fin, caudal fin, trunk, swim bladder, notochord, pigment, and circulatory system |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wilson, L.B.; Truong, L.; Simonich, M.T.; Tanguay, R.L. Systematic Assessment of Exposure Variations on Observed Bioactivity in Zebrafish Chemical Screening. Toxics 2020, 8, 87. https://doi.org/10.3390/toxics8040087
Wilson LB, Truong L, Simonich MT, Tanguay RL. Systematic Assessment of Exposure Variations on Observed Bioactivity in Zebrafish Chemical Screening. Toxics. 2020; 8(4):87. https://doi.org/10.3390/toxics8040087
Chicago/Turabian StyleWilson, Lindsay B., Lisa Truong, Michael T. Simonich, and Robyn L. Tanguay. 2020. "Systematic Assessment of Exposure Variations on Observed Bioactivity in Zebrafish Chemical Screening" Toxics 8, no. 4: 87. https://doi.org/10.3390/toxics8040087
APA StyleWilson, L. B., Truong, L., Simonich, M. T., & Tanguay, R. L. (2020). Systematic Assessment of Exposure Variations on Observed Bioactivity in Zebrafish Chemical Screening. Toxics, 8(4), 87. https://doi.org/10.3390/toxics8040087








