Does Hemoperfusion Increase Survival in Acute Paraquat Poisoning? A Retrospective Multicenter Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Ethics Statement
2.3. Data Collection and Definition
2.4. Outcome Measures
2.5. Statistical Analysis
3. Results
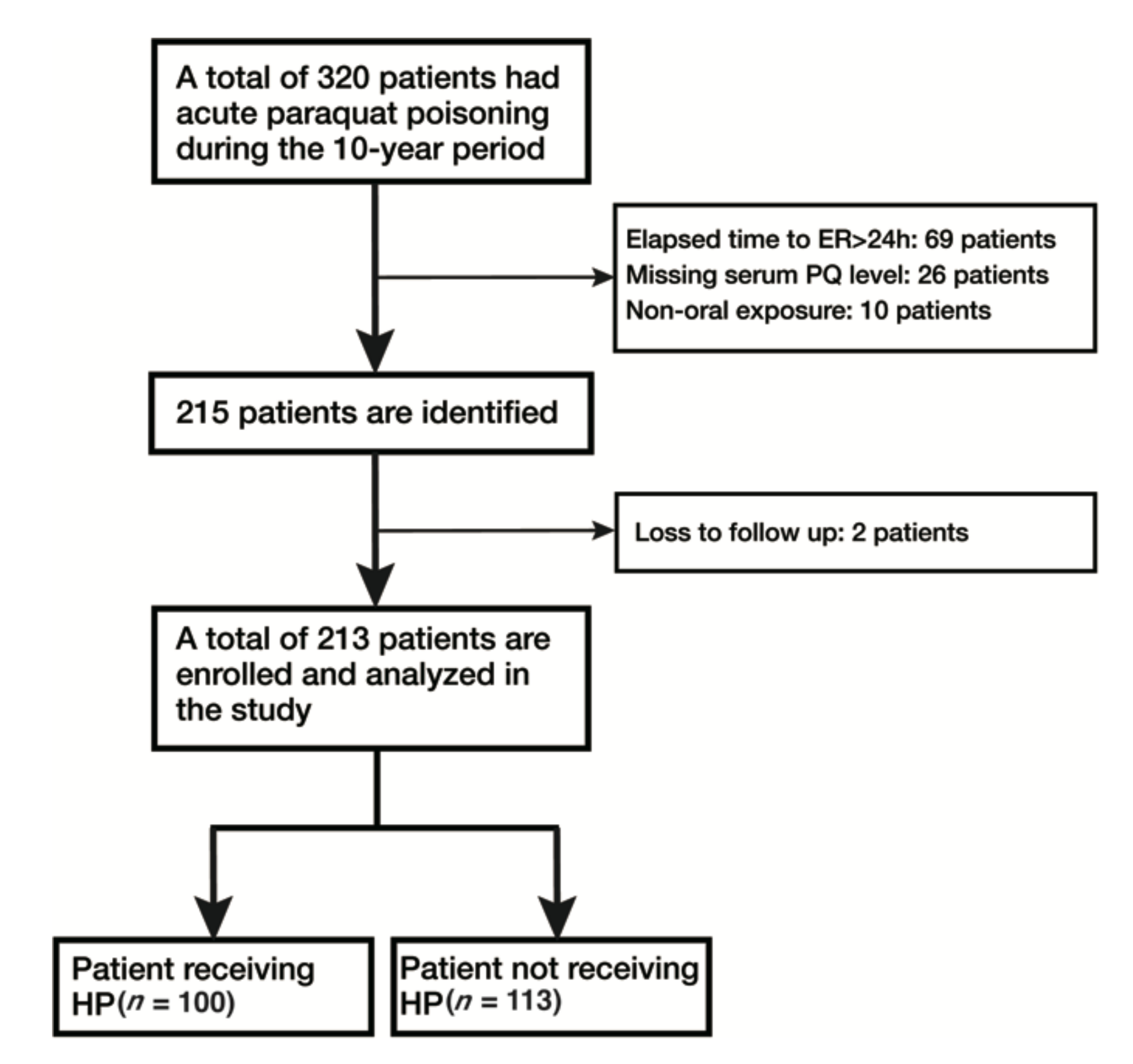
3.1. Enrollment
3.2. Baseline Characteristic
3.3. Differences between HP and Non-HP Groups
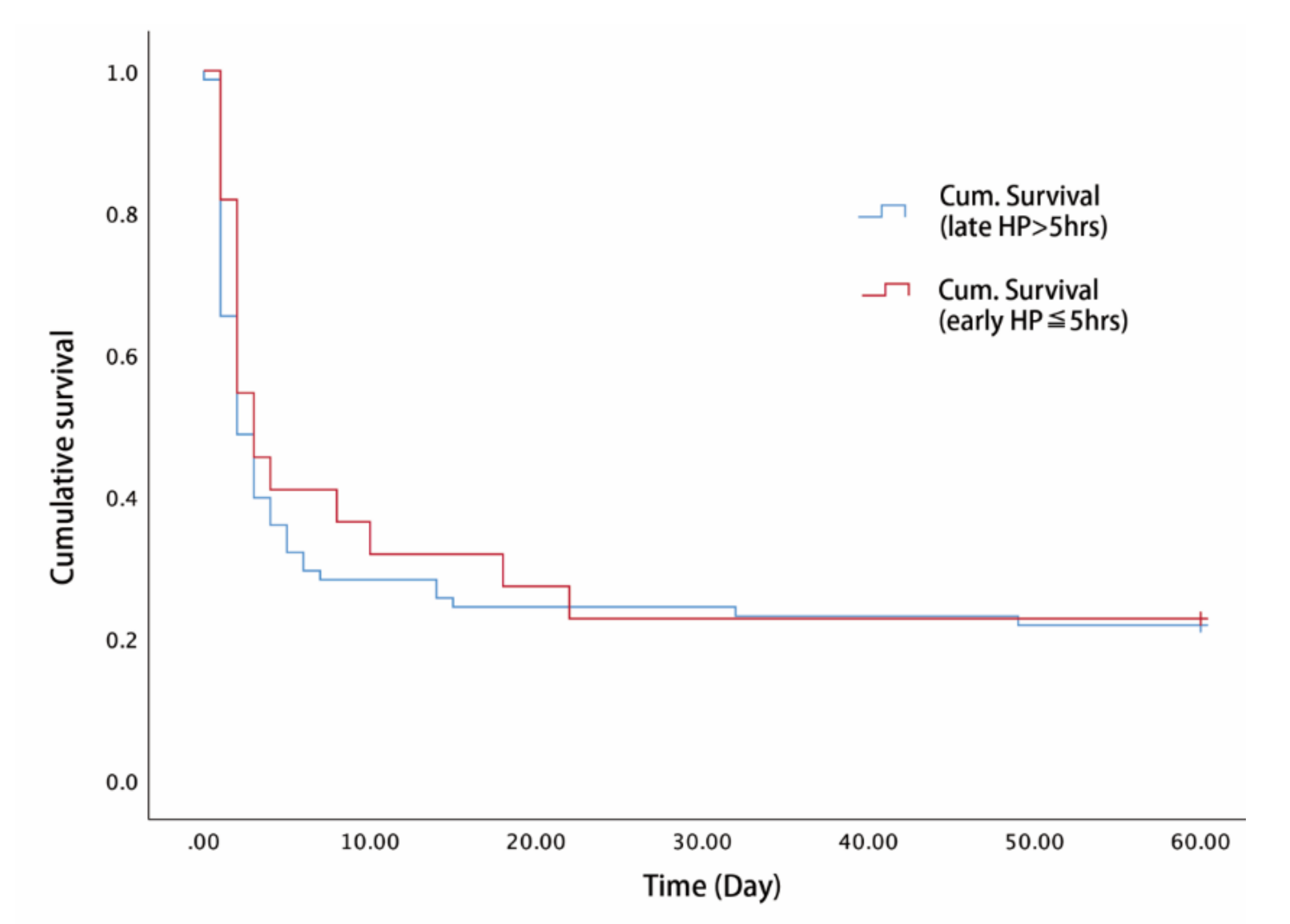
3.4. Survival Analysis
4. Discussion
5. Limitation
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dinis-Oliveira, R.J.; Duarte, J.A.; Sanchez-Navarro, A.; Remiao, F.; Bastos, M.L.; Carvalho, F. Paraquat poisonings: Mechanisms of lung toxicity, clinical features, and treatment. Crit. Rev. Toxicol. 2008, 38, 13–71. [Google Scholar] [CrossRef] [PubMed]
- Gunnell, D.; Eddleston, M.; Phillips, M.R.; Konradsen, F. The global distribution of fatal pesticide self-poisoning: Systematic review. BMC Public Health 2007, 7, 357. [Google Scholar] [CrossRef]
- Lee, H.L.; Lin, H.J.; Yeh, S.T.; Chi, C.H.; Guo, H.R. Presentations of patients of poisoning and predictors of poisoning-related fatality: Findings from a hospital-based prospective study. BMC Public Health 2008, 8, 7. [Google Scholar] [CrossRef] [PubMed]
- Ko, D.R.; Chung, S.P.; You, J.S.; Cho, S.; Park, Y.; Chun, B.; Moon, J.; Kim, H.; Kim, Y.H.; Kim, H.J.; et al. Effects of Paraquat Ban on Herbicide Poisoning-Related Mortality. Yonsei Med. J. 2017, 58, 859–866. [Google Scholar] [CrossRef] [PubMed]
- Kervégant, M.; Merigot, L.; Glaizal, M.; Schmitt, C.; Tichadou, L.; de Haro, L. Paraquat poisonings in France during the European ban: Experience of the Poison Control Center in Marseille. J. Med. Toxicol. 2013, 9, 144–147. [Google Scholar] [CrossRef]
- Hong, S.Y.; Yang, J.O.; Lee, E.Y.; Kim, S.H. Effect of haemoperfusion on plasma paraquat concentration in vitro and in vivo. Toxicol. Ind. Health 2003, 19, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Van de Vyver, F.L.; Giuliano, R.A.; Paulus, G.J.; Verpooten, G.A.; Franke, J.P.; de Zeeuw, R.A.; van Gaal, L.F.; de Broe, M.E. Hemoperfusion-hemodialysis ineffective for paraquat removal in life-threatening poisoning? J. Toxicol. Clin. Toxicol. 1985, 23, 117–131. [Google Scholar] [CrossRef]
- Bismuth, C.; Garnier, R.; Dally, S.; Fournier, P.E.; Scherrmann, J.M. Prognosis and treatment of paraquat poisoning: A review of 28 cases. J. Toxicol. Clin. Toxicol. 1982, 19, 461–474. [Google Scholar] [CrossRef]
- Hampson, E.C.; Pond, S.M. Failure of haemoperfusion and haemodialysis to prevent death in paraquat poisoning. A retrospective review of 42 patients. Med. Toxicol. Advers. Drug Exp. 1988, 3, 64–71. [Google Scholar]
- Hsu, C.W.; Lin, J.L.; Lin-Tan, D.T.; Chen, K.H.; Yen, T.H.; Wu, M.S.; Lin, S.C. Early hemoperfusion may improve survival of severely paraquat-poisoned patients. PLoS ONE 2012, 7, e48397. [Google Scholar] [CrossRef]
- Li, A.; Li, W.; Hao, F.; Wang, H. Early Stage Blood Purification for Paraquat Poisoning: A Multicenter Retrospective Study. Blood Purif. 2016, 42, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.R.; Pan, J.; Shang, A.D.; Lu, Y.Q. Time-dependent haemoperfusion after acute paraquat poisoning. Sci. Rep. 2017, 7, 2239. [Google Scholar] [CrossRef] [PubMed]
- Rao, R.; Bhat, R.; Pathadka, S.; Chenji, S.K.; Dsouza, S. Golden Hours in Severe Paraquat Poisoning—The Role of Early Haemoperfusion Therapy. J. Clin. Diagn. Res. 2017, 11, Oc06–Oc08. [Google Scholar] [CrossRef] [PubMed]
- Sawada, Y.; Yamamoto, I.; Hirokane, T.; Nagai, Y.; Satoh, Y.; Ueyama, M. Severity index of paraquat poisoning. Lancet 1988, 1, 1333. [Google Scholar] [CrossRef]
- Scherrmann, J.M.; Houze, P.; Bismuth, C.; Bourdon, R. Prognostic value of plasma and urine paraquat concentration. Hum. Toxicol. 1987, 6, 91–93. [Google Scholar] [CrossRef]
- Xu, S.; Hu, H.; Jiang, Z.; Tang, S.; Zhou, Y.; Sheng, J.; Chen, J.; Cao, Y. APACHE score, Severity Index of Paraquat Poisoning, and serum lactic acid concentration in the prognosis of paraquat poisoning of Chinese Patients. Pediatr. Emerg. Care 2015, 31, 117–121. [Google Scholar] [CrossRef]
- Bismuth, C.; Scherrmann, J.M.; Garnier, R.; Baud, F.J.; Pontal, P.G. Elimination of paraquat. Hum. Toxicol. 1987, 6, 63–67. [Google Scholar] [CrossRef]
- Gawarammana, I.B.; Buckley, N.A. Medical management of paraquat ingestion. Br. J. Clin. Pharmacol. 2011, 72, 745–757. [Google Scholar] [CrossRef]
- Naka, T.; Shinozaki, M.; Akizawa, T.; Shima, Y.; Takaesu, H.; Nasu, H. The Effect of Continuous Veno-Venous Hemofiltration or Direct Hemoperfusion with Polymyxin B-Immobilized Fiber on Neutrophil Respiratory Oxidative Burst in Patients with Sepsis and Septic Shock. Ther. Apher. Dial. 2006, 10, 7–11. [Google Scholar] [CrossRef]
- De Broe, M.E.; Bismuth, C.; de Groot, G.; Heath, A.; Okonek, S.; Ritz, D.R.; Verpooten, G.A.; Volans, G.N.; Widdop, B. Haemoperfusion: A useful therapy for a severely poisoned patient? Hum. Toxicol. 1986, 5, 11–14. [Google Scholar] [CrossRef]
- Proudfoot, A.T.; Prescott, L.F.; Jarvie, D.R. Haemodialysis for Paraquat Poisoning. Hum. Toxicol. 1987, 6, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, K.; Takasu, N.; Okabe, T.; Ishimatsu, S.; Ueda, A.; Tanaka, S.; Fukuda, A.; Arita, S.; Kohama, A. Effect of Aggressive Haemoperfiusion on the Clinical Course of Patients with Paraquat Poisonin. Hum. Exp. Toxicol. 1993, 12, 323–327. [Google Scholar] [CrossRef] [PubMed]
- Houze, P.; Baud, F.J.; Mouy, R.; Bismuth, C.; Bourdon, R.; Scherrmann, J.M. Toxicokinetics of paraquat in humans. Hum. Exp. Toxicol. 1990, 9, 5–12. [Google Scholar] [CrossRef] [PubMed]
- Hampson, E.C.; Effeney, D.J.; Pond, S.M. Efficacy of single or repeated hemoperfusion in a canine model of paraquat poisoning. J. Pharmacol. Exp. Ther. 1990, 254, 732–740. [Google Scholar] [PubMed]
- Li, C.; Hu, D.; Xue, W.; Li, X.; Wang, Z.; Ai, Z.; Song, Y.; Liu, X.; Cheng, J.; Fan, S.; et al. Treatment Outcome of Combined Continuous Venovenous Hemofiltration and Hemoperfusion in Acute Paraquat Poisoning: A Prospective Controlled Trial. Crit. Care Med. 2018, 46, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chen, Y.; Mao, L.; Zhao, G.; Hong, G.; Li, M.; Wu, B.; Chen, X.; Tan, M.; Wang, N.; et al. Effects of hemoperfusion and continuous renal replacement therapy on patient survival following paraquat poisoning. PLoS ONE 2017, 12, e0181207. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Bai, Y.; Zou, Y.; Cai, B.; Liu, F.; Fu, P.; Wang, L. The value of plasma paraquat concentration in predicting therapeutic effects of haemoperfusion in patients with acute paraquat poisoning. PLoS ONE 2012, 7, e40911. [Google Scholar] [CrossRef]
| Parameter | Patient Having sPQ, n = 213 | Patient Not Having sPQ, n = 24 | p-Value |
|---|---|---|---|
| Median age, years (SD) | 51 (17.44) | 54 (17.84) | 0.391 |
| Gender | 0.488 | ||
| Male, n (%) | 145 (68.1) | 18 (75.0) | |
| Female, n (%) | 68 (31.9) | 6 (25.0) | |
| GCS > 13, n (%) | 154 (72.3) | 14 (58.3) | 0.153 |
| Creatinine (mg/dl); Mean (SD) | 1.66 (1.29) | 2.68 (4.00) | 0.227 |
| Gastric lavage, n (%) | 171 (80.3) | 17 (70.3) | 0.291 |
| Activated charcoal, n (%) | 150 (70.4) | 11 (45.8) | 0.014 |
| Cyclophosphamide, n (%) | 32 (15.0) | 3 (12.5) | 1.0 |
| N-acetylcysteine, n (%) | 74 (34.7) | 10 (41.7) | 0.501 |
| Vitamin C, n (%) | 48 (22.5) | 6 (25.0) | 0.785 |
| Steroid, n (%) | 146 (68.5) | 18 (75) | 0.516 |
| HP, n (%) | 100 (46.9) | 5 (20.8) | 0.015 |
| Mortality, n (%) | 161 (75.6) | 22 (91.7) | 0.075 |
| Parameter | HP Group n = 100 | Non-HP Group n = 113 | p-Value |
|---|---|---|---|
| Median age, years (SD) | 47 (17.41) | 54 (16.96) | 0.006 |
| Gender | 0.365 | ||
| Male, n (%) | 65 (65.0) | 80 (70.8) | |
| Female, n (%) | 35 (35.0) | 33 (29.2) | |
| GCS > 13, n (%) | 82 (82) | 72 (63.7) | 0.003 |
| Creatinine (mg/dL); Mean (SD) | 1.36 (0.82) | 1.93 (1.56) | 0.001 |
| SIPP; Mean (SD) | 98.71 (399.10) | 158.53 (338.07) | 0.238 |
| SIPP < 10, n (%) | 35 (35.0) | 39 (34.5) | 0.941 |
| SIPP 10–50, n (%) | 33 (33.0) | 32 (28.3) | 0.459 |
| SIPP > 50, n (%) | 32 (32.0) | 42 (37.2) | 0.429 |
| Gastric lavage, n (%) | 83 (83.0) | 88 (77.9) | 0.348 |
| Activated charcoal, n (%) | 75 (75.0) | 75 (66.4) | 0.169 |
| Cyclophosphamide, n (%) | 23 (23.0) | 9 (8.0) | 0.002 |
| N-acetylcysteine, n (%) | 22 (22.0) | 52 (46.0) | <0.001 |
| Vitamin C, n (%) | 21 (21.0) | 27 (23.9) | 0.614 |
| Steroid, n (%) | 73 (73.0) | 73 (64.6) | 0.188 |
| Mortality, n (%) | 78 (78.0) | 83 (73.5) | 0.441 |
| Variables | Adjusted HR (95% CI) | p-Value |
|---|---|---|
| Age (years) | 1.01 (1.00–1.02) | 0.024 |
| Gender as female (Yes) | 0.97 (0.69–1.37) | 0.888 |
| Creatinine (mg/dL) | 1.17 (1.05–1.29) | 0.003 |
| SIPP | 2.61 (2.08–3.28) | <0.001 |
| Cyclophosphamide (Yes) | 0.80 (0.50–1.33) | 0.384 |
| N-acetylcysteine (Yes) | 1.00 (0.71–1.41) | 0.995 |
| HP group (Yes) | 1.17 (0.84–1.63) | 0.363 |
| Variables | Adjusted HR (95% CI) | p-Value |
|---|---|---|
| Age (years) | 1.01 (0.99–1.03) | 0.083 |
| Gender as female (Yes) | 0.84 (0.50–1.39) | 0.490 |
| Creatinine (mg/dL) | 1.15 (0.88–1.50) | 0.320 |
| SIPP | 2.49 (1.75–3.56) | <0.001 |
| Cyclophosphamide (Yes) | 0.71 (0.39–1.31) | 0.277 |
| N-acetylcysteine (Yes) | 0.68 (0.36–1.27) | 0.224 |
| Secession of HP | 0.78 (0.56–1.07) | 0.124 |
| Interval to first HP ≤ 5 h | 0.96 (0.54–1.69) | 0.880 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yeh, Y.-T.; Chen, C.-K.; Lin, C.-C.; Chang, C.-M.; Lan, K.-P.; How, C.-K.; Yen, H.-T.; Chen, Y.-C. Does Hemoperfusion Increase Survival in Acute Paraquat Poisoning? A Retrospective Multicenter Study. Toxics 2020, 8, 84. https://doi.org/10.3390/toxics8040084
Yeh Y-T, Chen C-K, Lin C-C, Chang C-M, Lan K-P, How C-K, Yen H-T, Chen Y-C. Does Hemoperfusion Increase Survival in Acute Paraquat Poisoning? A Retrospective Multicenter Study. Toxics. 2020; 8(4):84. https://doi.org/10.3390/toxics8040084
Chicago/Turabian StyleYeh, Ying-Tse, Chun-Kuei Chen, Chih-Chuan Lin, Chia-Ming Chang, Kai-Ping Lan, Chorng-Kuang How, Hung-Tsang Yen, and Yen-Chia Chen. 2020. "Does Hemoperfusion Increase Survival in Acute Paraquat Poisoning? A Retrospective Multicenter Study" Toxics 8, no. 4: 84. https://doi.org/10.3390/toxics8040084
APA StyleYeh, Y.-T., Chen, C.-K., Lin, C.-C., Chang, C.-M., Lan, K.-P., How, C.-K., Yen, H.-T., & Chen, Y.-C. (2020). Does Hemoperfusion Increase Survival in Acute Paraquat Poisoning? A Retrospective Multicenter Study. Toxics, 8(4), 84. https://doi.org/10.3390/toxics8040084





