E-Cigarette Liquid Provokes Significant Embryotoxicity and Inhibits Angiogenesis
Abstract
1. Introduction
2. Materials and Methods
2.1. E-Cigarette Liquid
2.2. Embryos and In Ovo E-Cigarette Treatment
2.3. Macroscopic and Microscopic Analysis
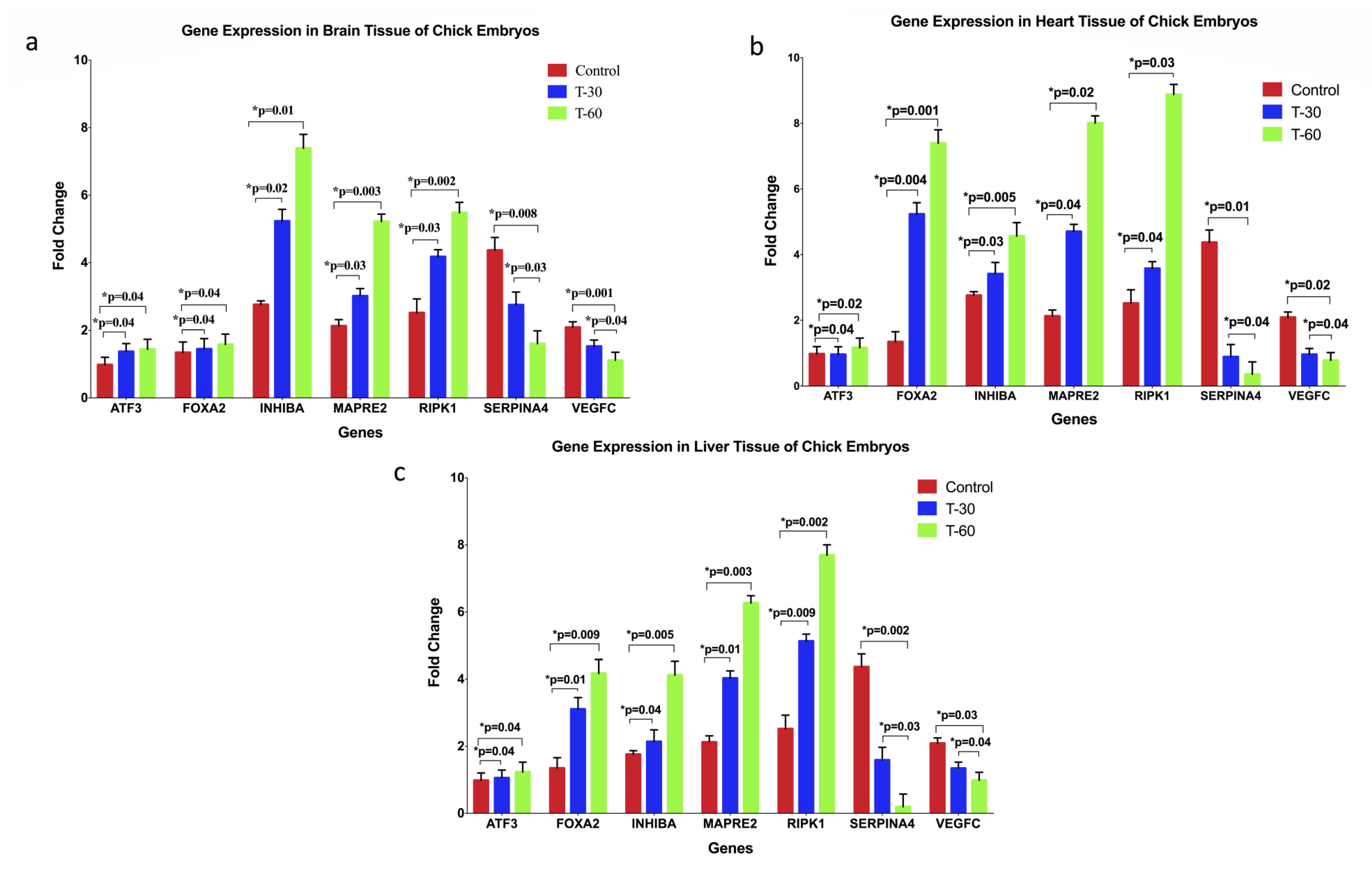
2.4. RNA Isolation and Reverse Transcription (RT)-PCR Analysis
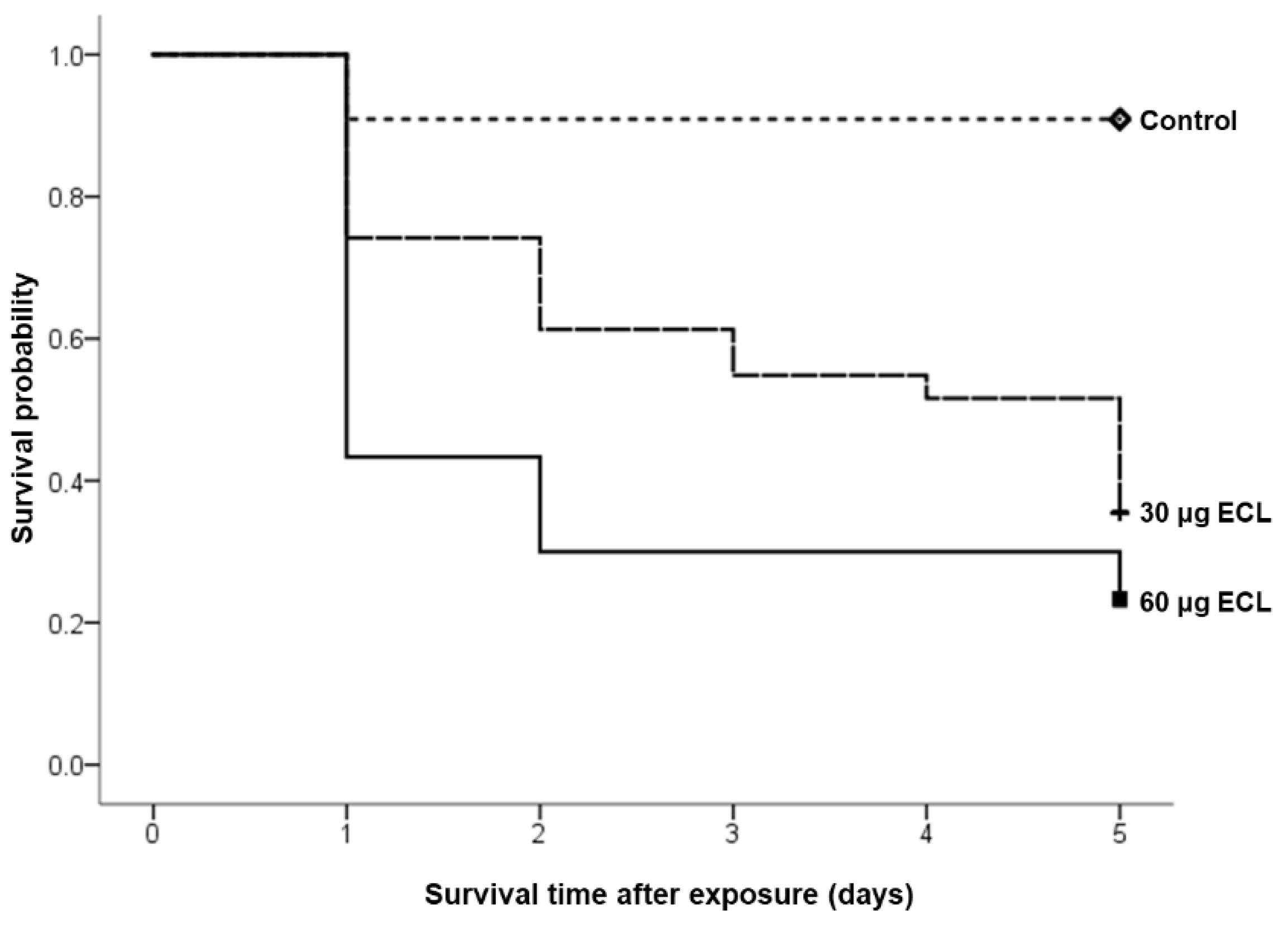
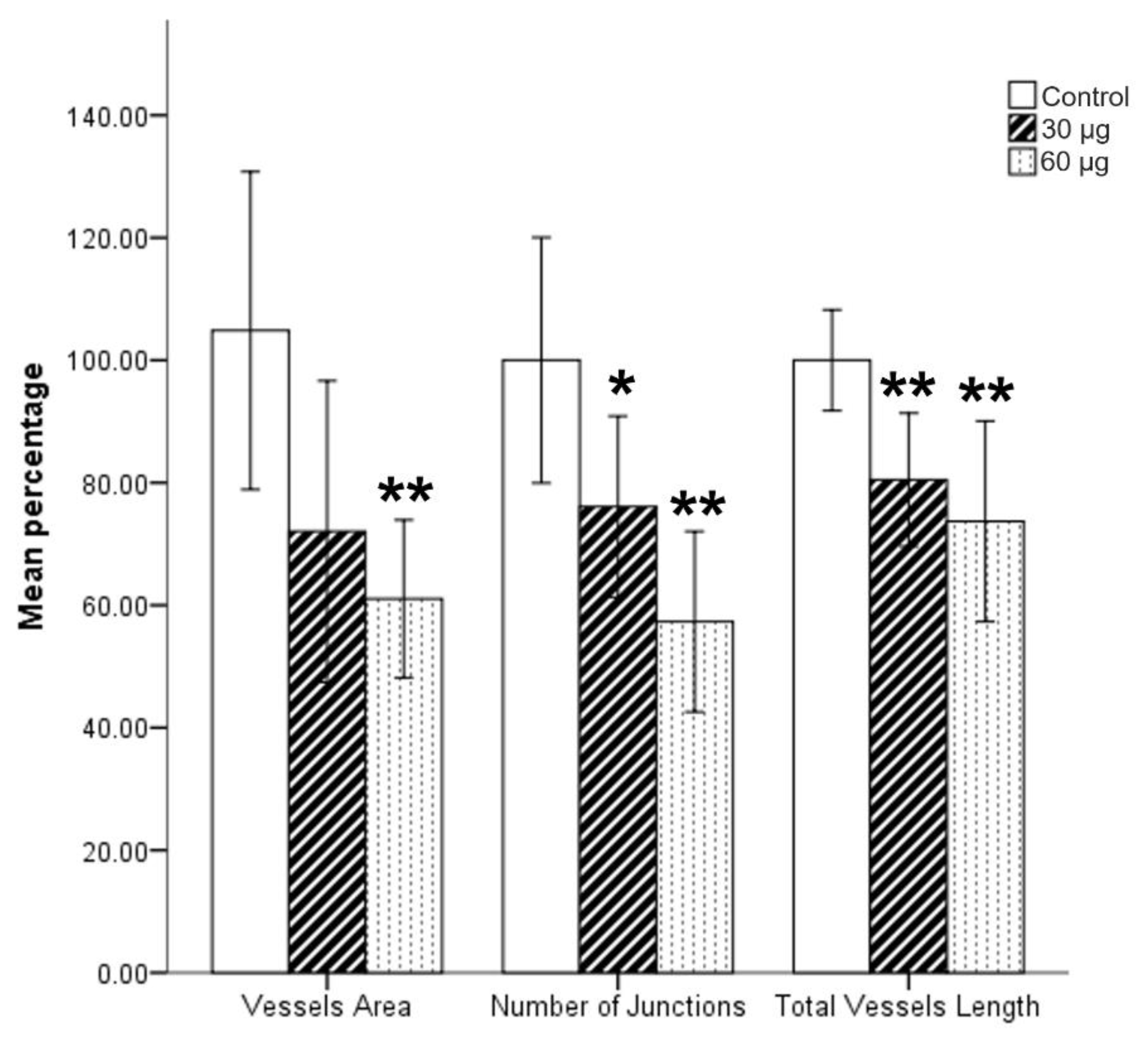
2.5. Angiogenesis Quantification, Statistical and Survival Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- WHO. Tobacco. Available online: https://www.who.int/news-room/fact-sheets/detail/tobacco (accessed on 1 December 2019).
- Gowing, L.R.; Ali, R.L.; Allsop, S.; Marsden, J.; Turf, E.E.; West, R.; Witton, J. Global statistics on addictive behaviours: 2014 status report. Addiction 2015, 110, 904–919. [Google Scholar] [CrossRef] [PubMed]
- Onor, I.O.; Stirling, D.L.; Williams, S.R.; Bediako, D.; Borghol, A.; Harris, M.B.; Darensburg, T.B.; Clay, S.D.; Okpechi, S.C.; Sarpong, D.F. Clinical Effects of Cigarette Smoking: Epidemiologic Impact and Review of Pharmacotherapy Options. Int. J. Environ. Res. Public Health 2017, 14, 1147. [Google Scholar] [CrossRef] [PubMed]
- Caponnetto, P.; Campagna, D.; Papale, G.; Russo, C.; Polosa, R. The emerging phenomenon of electronic cigarettes. Expert Rev. Respir. Med. 2012, 6, 63–74. [Google Scholar] [CrossRef] [PubMed]
- Lempert, L.K.; Grana, R.; Glantz, S.A. The importance of product definitions in US e-cigarette laws and regulations. Tob. Control 2016, 25, e44–e51. [Google Scholar] [CrossRef]
- Laverty, A.A.; Filippidis, F.T.; Vardavas, C.I. Patterns, trends and determinants of e-cigarette use in 28 European Union Member States 2014–2017. Prev. Med. 2018, 116, 13–18. [Google Scholar] [CrossRef]
- Goniewicz, M.L.; Knysak, J.; Gawron, M.; Kosmider, L.; Sobczak, A.; Kurek, J.; Prokopowicz, A.; Jablonska-Czapla, M.; Rosik-Dulewska, C.; Havel, C.; et al. Levels of selected carcinogens and toxicants in vapour from electronic cigarettes. Tob. Control 2014, 23, 133–139. [Google Scholar] [CrossRef]
- Olmedo, P.; Goessler, W.; Tanda, S.; Grau-Perez, M.; Jarmul, S.; Aherrera, A.; Chen, R.; Hilpert, M.; Cohen, J.E.; Navas-Acien, A.; et al. Metal Concentrations in e-Cigarette Liquid and Aerosol Samples: The Contribution of Metallic Coils. Environ. Health Perspect. 2018, 126, 027010. [Google Scholar] [CrossRef]
- Farsalinos, K.E.; Romagna, G.; Allifranchini, E.; Ripamonti, E.; Bocchietto, E.; Todeschi, S.; Tsiapras, D.; Kyrzopoulos, S.; Voudris, V. Comparison of the cytotoxic potential of cigarette smoke and electronic cigarette vapour extract on cultured myocardial cells. Int. J. Environ. Res. Public Health 2013, 10, 5146–5162. [Google Scholar] [CrossRef]
- Hua, M.; Talbot, P. Potential health effects of electronic cigarettes: A systematic review of case reports. Prev. Med. Rep. 2016, 4, 169–178. [Google Scholar] [CrossRef]
- Benowitz, N.L.; Fraiman, J.B. Cardiovascular effects of electronic cigarettes. Nat. Rev. Cardiol. 2017, 14, 447–456. [Google Scholar] [CrossRef]
- Hess, I.M.; Lachireddy, K.; Capon, A. A systematic review of the health risks from passive exposure to electronic cigarette vapour. Public Health Res. Pract. 2016, 26, e2621617. [Google Scholar] [CrossRef] [PubMed]
- Lerner, C.A.; Sundar, I.K.; Yao, H.; Gerloff, J.; Ossip, D.J.; McIntosh, S.; Robinson, R.; Rahman, I. Vapors produced by electronic cigarettes and e-juices with flavorings induce toxicity, oxidative stress, and inflammatory response in lung epithelial cells and in mouse lung. PLoS ONE 2015, 10, e0116732. [Google Scholar] [CrossRef] [PubMed]
- Scheffler, S.; Dieken, H.; Krischenowski, O.; Forster, C.; Branscheid, D.; Aufderheide, M. Evaluation of E-cigarette liquid vapor and mainstream cigarette smoke after direct exposure of primary human bronchial epithelial cells. Int. J. Environ. Res. Public Health 2015, 12, 3915–3925. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Li, G.; Chan, Y.L.; Chapman, D.G.; Sukjamnong, S.; Nguyen, T.; Annissa, T.; McGrath, K.C.; Sharma, P.; Oliver, B.G. Maternal E-Cigarette Exposure in Mice Alters DNA Methylation and Lung Cytokine Expression in Offspring. Am. J. Respir. Cell Mol. Biol. 2018, 58, 366–377. [Google Scholar] [CrossRef]
- Nguyen, T.; Li, G.E.; Chen, H.; Cranfield, C.G.; McGrath, K.C.; Gorrie, C.A. Maternal E-Cigarette Exposure Results in Cognitive and Epigenetic Alterations in Offspring in a Mouse Model. Chem. Res. Toxicol. 2018, 31, 601–611. [Google Scholar] [CrossRef]
- Raez-Villanueva, S.; Ma, C.; Kleiboer, S.; Holloway, A.C. The effects of electronic cigarette vapor on placental trophoblast cell function. Reprod. Toxicol. 2018, 81, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Ashour, A.A.; Haik, M.Y.; Sadek, K.W.; Yalcin, H.C.; Bitharas, J.; Aboulkassim, T.; Batist, G.; Yasmeen, A.; Al Moustafa, A.E. Substantial Toxic Effect of Water-Pipe Smoking on the Early Stage of Embryonic Development. Nicotine Tob. Res. 2018, 20, 502–507. [Google Scholar] [CrossRef] [PubMed]
- Sadek, K.W.; Haik, M.Y.; Ashour, A.A.; Baloch, T.; Aboulkassim, T.; Yasmeen, A.; Vranic, S.; Zeidan, A.; Al Moustafa, A.-E. Water-pipe smoking promotes epithelial-mesenchymal transition and invasion of human breast cancer cells via ERK1/ERK2 pathways. Cancer Cell Int. 2018, 18, 180. [Google Scholar] [CrossRef] [PubMed]
- Roman, D.; Yasmeen, A.; Mireuta, M.; Stiharu, I.; Al Moustafa, A.E. Significant toxic role for single-walled carbon nanotubes during normal embryogenesis. Nanomedicine 2013, 9, 945–950. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Zudaire, E.; Gambardella, L.; Kurcz, C.; Vermeren, S. A computational tool for quantitative analysis of vascular networks. PLoS ONE 2011, 6, e27385. [Google Scholar] [CrossRef] [PubMed]
- Ejaz, S.; Ejaz, A.; Sohail, A.; Lim, C.W. Vascular and morphogenetic abnormalities associated with exposure of cigarette smoke condensate during chicken and murine embryogenesis. Biomed. Environ. Sci. 2010, 23, 305–311. [Google Scholar] [CrossRef]
- Palpant, N.J.; Hofsteen, P.; Pabon, L.; Reinecke, H.; Murry, C.E. Cardiac development in zebrafish and human embryonic stem cells is inhibited by exposure to tobacco cigarettes and e-cigarettes. PLoS ONE 2015, 10, e0126259. [Google Scholar] [CrossRef] [PubMed]
- Massarsky, A.; Abdel, A.; Glazer, L.; Levin, E.D.; Di Giulio, R.T. Exposure to 1,2-Propanediol Impacts Early Development of Zebrafish (Danio rerio) and Induces Hyperactivity. Zebrafish 2017, 14, 216–222. [Google Scholar] [CrossRef]
- Canistro, D.; Vivarelli, F.; Cirillo, S.; Babot Marquillas, C.; Buschini, A.; Lazzaretti, M.; Marchi, L.; Cardenia, V.; Rodriguez-Estrada, M.T.; Lodovici, M.; et al. E-cigarettes induce toxicological effects that can raise the cancer risk. Sci. Rep. 2017, 7, 2028. [Google Scholar] [CrossRef]
- Martin, E.M.; Clapp, P.W.; Rebuli, M.E.; Pawlak, E.A.; Glista-Baker, E.; Benowitz, N.L.; Fry, R.C.; Jaspers, I. E-cigarette use results in suppression of immune and inflammatory-response genes in nasal epithelial cells similar to cigarette smoke. Am. J. Physiol. Lung Cell. Mol. Physiol. 2016, 311, L135–L144. [Google Scholar] [CrossRef]
- Sussan, T.E.; Gajghate, S.; Thimmulappa, R.K.; Ma, J.; Kim, J.H.; Sudini, K.; Consolini, N.; Cormier, S.A.; Lomnicki, S.; Hasan, F.; et al. Exposure to electronic cigarettes impairs pulmonary anti-bacterial and anti-viral defenses in a mouse model. PLoS ONE 2015, 10, e0116861. [Google Scholar] [CrossRef]
- Chatterjee, S.; Tao, J.Q.; Johncola, A.; Guo, W.; Caporale, A.; Langham, M.C.; Wehrli, F.W. Acute exposure to e-cigarettes causes inflammation and pulmonary endothelial oxidative stress in nonsmoking, healthy young subjects. Am. J. Physiol. Lung Cell. Mol. Physiol. 2019, 317, L155–L166. [Google Scholar] [CrossRef]
- Ejaz, S.; Ashraf, M.; Nawaz, M.; Lim, C.W.; Kim, B. Cigarette smoke condensate and total particulate matter severely disrupts physiological angiogenesis. Food Chem. Toxicol. 2009, 47, 601–614. [Google Scholar] [CrossRef]
- Song, Q.; Chen, Q.; Wang, Q.; Yang, L.; Lv, D.; Jin, G.; Liu, J.; Li, B.; Fei, X. ATF-3/miR-590/GOLPH3 signaling pathway regulates proliferation of breast cancer. BMC Cancer 2018, 18, 255. [Google Scholar] [CrossRef]
- Dondelinger, Y.; Delanghe, T.; Rojas-Rivera, D.; Priem, D.; Delvaeye, T.; Bruggeman, I.; Van Herreweghe, F.; Vandenabeele, P.; Bertrand, M.J.M. MK2 phosphorylation of RIPK1 regulates TNF-mediated cell death. Nat. Cell Biol. 2017, 19, 1237–1247. [Google Scholar] [CrossRef] [PubMed]
- National Academies of Sciences, Engineering, and Medicine; Health and Medicine Division; Board on Population Health and Public Health Practice; Committee on the Review of the Health Effects of Electronic Nicotine Delivery Systems. Public Health Practice; Committee on the Review of the Health Effects of Electronic Nicotine Delivery Systems. Public Health Consequences of E-Cigarettes. In E-Cigarette Devices, Uses, and Exposures; Eaton, D.L., Kwan, L.Y., Stratton, K., Eds.; National Academies Press (US): Washington, DC, USA, 2018; Volume 3. [Google Scholar]
- Mulder, H.A.; Patterson, J.L.; Halquist, M.S.; Kosmider, L.; Turner, J.B.M.; Poklis, J.L.; Poklis, A.; Peace, M.R. The Effect of Electronic Cigarette User Modifications and E-liquid Adulteration on the Particle Size Profile of an Aerosolized Product. Sci. Rep. 2019, 9, 10221. [Google Scholar] [CrossRef] [PubMed]
- Farsalinos, K.E.; Kistler, K.A.; Gillman, G.; Voudris, V. Evaluation of electronic cigarette liquids and aerosol for the presence of selected inhalation toxins. Nicotine Tob. Res. 2015, 17, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Romagna, G.; Allifranchini, E.; Bocchietto, E.; Todeschi, S.; Esposito, M.; Farsalinos, K.E. Cytotoxicity evaluation of electronic cigarette vapor extract on cultured mammalian fibroblasts (ClearStream-LIFE): Comparison with tobacco cigarette smoke extract. Inhal. Toxicol. 2013, 25, 354–361. [Google Scholar] [CrossRef]
- Misra, M.; Leverette, R.D.; Cooper, B.T.; Bennett, M.B.; Brown, S.E. Comparative in vitro toxicity profile of electronic and tobacco cigarettes, smokeless tobacco and nicotine replacement therapy products: E-liquids, extracts and collected aerosols. Int. J. Environ. Res. Public Health 2014, 11, 11325–11347. [Google Scholar] [CrossRef]
- Wu, Q.; Jiang, D.; Minor, M.; Chu, H.W. Electronic cigarette liquid increases inflammation and virus infection in primary human airway epithelial cells. PLoS ONE 2014, 9, e108342. [Google Scholar] [CrossRef]
- Lim, H.B.; Kim, S.H. Inhallation of e-Cigarette Cartridge Solution Aggravates Allergen-induced Airway Inflammation and Hyper-responsiveness in Mice. Toxicol. Res. 2014, 30, 13–18. [Google Scholar] [CrossRef]
- Omaiye, E.E.; McWhirter, K.J.; Luo, W.; Pankow, J.F.; Talbot, P. High-Nicotine Electronic Cigarette Products: Toxicity of JUUL Fluids and Aerosols Correlates Strongly with Nicotine and Some Flavor Chemical Concentrations. Chem. Res. Toxicol. 2019, 32, 1058–1069. [Google Scholar] [CrossRef]
- Manson, J.; Brabec, M.J.; Buelke-Sam, J.; Carlson, G.P.; Chapin, R.E.; Favor, J.B.; Fischer, L.J.; Hattis, D.; Lees, P.S.; Perreault-Darney, S.; et al. NTP-CERHR expert panel report on the reproductive and developmental toxicity of acrylamide. Birth Defects Res. B Dev. Reprod. Toxicol. 2005, 74, 17–113. [Google Scholar] [CrossRef]
| Gene Name | Primers |
|---|---|
| ATF-3 | 5′ - AAAAGCGAAGAAGGGAAAGG - 3′ |
| 5′ - ATACAGGTGGGCCTGTGAAG - 3′ | |
| FOXA2 | 5′ - GACCTCTTCCCCTTCTACCG - 3′ |
| 5′ - AGGTAGCAGCCGTTCTCAAA - 3′ | |
| INHBA | 5′ - GCCACCAAGAAACTCCATGT - 3′ |
| 5′ - GCAACGTTTTCTTGGGTGTT - 3′ | |
| MAPRE-2 | 5′ - CAAAGGAGCCTTCCACAGAG - 3′ |
| 5′ - GTCACTTCTGATGGCAGCAA - 3′ | |
| RIPK-1 | 5′ - CCGTACAGAATTGCAGCAGA - 3′ |
| 5′ - TTCCATTAGCACACGAGCTG - 3′ | |
| SERPINA-4 | 5′ - CCAGCAAAAGGGAAAATGAA - 3′ |
| 5′ - CACCACTGATGCCAGAGAGA - 3′ | |
| VEGF-C | 5′- AGGGAACACTCCAGCTCTGA - 3′ |
| 5′ - CTCCAAACTCTTTCCCCACA - 3′ | |
| GAPDH | 5′ - CCTCTCTGGCAAAGTCCAAG - 3′ |
| 5′ - CATCTGCCCATTTGATGTTG - 3′ |
| Embryos | Number of Cases | Number of Embryos Dead Before 8 Days of Incubation (%) |
|---|---|---|
| 60μg ECL | 30 | 23 (76) |
| 30μg ECL | 31 | 20 (64) |
| Control | 11 | 1 (9) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ashour, A.A.; Alhussain, H.; Bin Rashid, U.; Abughazzah, L.; Gupta, I.; Malki, A.; Vranic, S.; Al Moustafa, A.-E. E-Cigarette Liquid Provokes Significant Embryotoxicity and Inhibits Angiogenesis. Toxics 2020, 8, 38. https://doi.org/10.3390/toxics8020038
Ashour AA, Alhussain H, Bin Rashid U, Abughazzah L, Gupta I, Malki A, Vranic S, Al Moustafa A-E. E-Cigarette Liquid Provokes Significant Embryotoxicity and Inhibits Angiogenesis. Toxics. 2020; 8(2):38. https://doi.org/10.3390/toxics8020038
Chicago/Turabian StyleAshour, Anas A., Hashim Alhussain, Umar Bin Rashid, Labiba Abughazzah, Ishita Gupta, Ahmed Malki, Semir Vranic, and Ala-Eddin Al Moustafa. 2020. "E-Cigarette Liquid Provokes Significant Embryotoxicity and Inhibits Angiogenesis" Toxics 8, no. 2: 38. https://doi.org/10.3390/toxics8020038
APA StyleAshour, A. A., Alhussain, H., Bin Rashid, U., Abughazzah, L., Gupta, I., Malki, A., Vranic, S., & Al Moustafa, A.-E. (2020). E-Cigarette Liquid Provokes Significant Embryotoxicity and Inhibits Angiogenesis. Toxics, 8(2), 38. https://doi.org/10.3390/toxics8020038








