Comparative Assessment of Tungsten Toxicity in the Absence or Presence of Other Metals
Abstract
1. Introduction
2. Tungsten: Uses and Routes of Exposure
3. Tungsten Toxicity
4. Toxicity of Tungsten Metal Mixtures
4.1. Tungsten and Cobalt Mixtures
4.2. Tungsten, Cobalt and Nickel Mixtures
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Agency for Toxic Substances and Disease Registry. Toxicological Profile for Tungsten; Department of Health and Human Services: Atlanta, GA, USA, 2005.
- Emond, C.A.; Vergara, V.B.; Lombardini, E.D.; Mog, S.R.; Kalinich, J.F. Induction of rhabdomyosarcoma by embedded military-grade tungsten/nickel/cobalt not by tungsten/nickel/iron in the B6C3F1 mouse. Int. J. Toxicol. 2015, 34, 44–54. [Google Scholar] [CrossRef] [PubMed]
- Tuna, G.S.; Braida, W.; Ogundipe, A.; Strickland, D. Assessing tungsten transport in the vadose zone: From dissolution studies to soil columns. Chemosphere 2012, 86, 1001–1007. [Google Scholar] [CrossRef] [PubMed]
- CDC. Exposure to Tungsten in Three Nevada Communities-CDC-2002. Available online: https://www.cdc.gov/nceh/clusters/fallon/tungsten_report.pdf (accessed on 19 August 2017).
- Sheppard, P.R.; Speakman, R.J.; Ridenour, G.; Witten, M.L. Temporal variability of tungsten and cobalt in Fallon, Nevada. Environ. Health Perspect. 2007, 115, 715–719. [Google Scholar] [CrossRef] [PubMed]
- Kalinich, J.F.; Emond, C.A.; Dalton, T.K.; Mog, S.R.; Coleman, G.D.; Kordell, J.E.; Miller, A.C.; McClain, D.E. Embedded weapons-grade tungsten alloy shrapnel rapidly induces metastatic high-grade rhabdomyosarcomas in F344 rats. Environ. Health Perspect. 2005, 113, 729–734. [Google Scholar] [CrossRef] [PubMed]
- Van der Voet, G.B.; Todorov, T.I.; Centeno, J.A.; Jonas, W.; Ives, J.; Mullick, F.G. Metals and health: A clinical toxicological perspective on tungsten and review of the literature. Mil. Med. 2007, 172, 1002–1005. [Google Scholar] [CrossRef] [PubMed]
- Keith, L.S.; Moffett, D.B.; Rosemond, Z.A.; Wohlers, D.W.; Disease, R. ATSDR evaluation of health effects of tungsten and relevance to public health. Toxicol. Ind. Health 2007, 23, 347–387. [Google Scholar] [CrossRef] [PubMed]
- Rizzato, G.; Lo Cicero, S.; Barberis, M.; Torre, M.; Pietra, R.; Sabbioni, E. Trace of metal exposure in hard metal lung disease. Chest 1986, 90, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Haddad, E.; Zikovsky, L. Determination of Al, As, Co, Cr, Cs, Fe, Mn, Sb, Sc, W and Zn in the workroom air by instrumental neutron activation analysis. J. Radioanal. Nucl. Chem. 1985, 93, 371–378. [Google Scholar] [CrossRef]
- Laulicht, F.; Brocato, J.; Cartularo, L.; Vaughan, J.; Wu, F.; Kluz, T.; Sun, H.; Oksuz, B.A.; Shen, S.; Peana, M.; et al. Tungsten-induced carcinogenesis in human bronchial epithelial cells. Toxicol. Appl. Pharmacol. 2015, 288, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Bey, E.A.; Bentle, M.S.; Reinicke, K.E.; Dong, Y.; Yang, C.R.; Girard, L.; Minna, J.D.; Bornmann, W.G.; Gao, J.; Boothman, D.A. An NQO1- and PARP-1-mediated cell death pathway induced in non-small-cell lung cancer cells by β-lapachone. Proc. Natl. Acad. Sci. USA 2007, 104, 11832–11837. [Google Scholar] [CrossRef] [PubMed]
- Iskander, K.; Barrios, R.J.; Jaiswal, A.K. Disruption of NAD(P)H:quinone oxidoreductase 1 gene in mice leads to radiation-induced myeloproliferative disease. Cancer Res. 2008, 68, 7915–7922. [Google Scholar] [CrossRef] [PubMed]
- Yokoi, S.; Yasui, K.; Saito-Ohara, F.; Koshikawa, K.; Iizasa, T.; Fujisawa, T.; Terasaki, T.; Horii, A.; Takahashi, T.; Hirohashi, S. A novel target gene, SKP2, within the 5p13 amplicon that is frequently detected in small cell lung cancers. Am. J. Pathol. 2002, 161, 207–216. [Google Scholar] [CrossRef]
- Laulicht-Glick, F.; Wu, F.; Zhang, X.; Jordan, A.; Brocato, J.; Kluz, T.; Sun, H.; Costa, M. Tungsten exposure causes a selective loss of histone demethylase protein. Mol. Carcinog. 2017, 56, 1778–1788. [Google Scholar] [CrossRef] [PubMed]
- Bolt, A.M.; Sabourin, V.; Molina, M.F.; Police, A.M.; Negro Silva, L.F.; Plourde, D.; Lemaire, M.; Ursini-Siegel, J.; Mann, K.K. Tungsten targets the tumor microenvironment to enhance breast cancer metastasis. Toxicol. Sci. 2015, 143, 165–177. [Google Scholar] [CrossRef] [PubMed]
- Chinde, S.; Grover, P. Toxicological assessment of nano and micron-sized tungsten oxide after 28days repeated oral administration to Wistar rats. Mutat. Res. 2017, 819, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Srivatsan, T.; Woods, R.; Petraroli, M.; Sudarshan, T. An investigation of the influence of powder particle size on microstructure and hardness of bulk samples of tungsten carbide. Powder Technol. 2002, 122, 54–60. [Google Scholar] [CrossRef]
- Salikhodzhaev, S.; Vengerskaya, K.Y. Some industrial hygiene problems in the production of hard alloys. Sov. Powder Metall. Metal. Ceram. 1962, 1, 142–145. [Google Scholar] [CrossRef]
- Kaplun, Z.; Mezentseva, N. Industrial dusts encountered in powder metallurgy (hard alloys). In Toxicology of the Rare Metals; US Dept of Commerce, National Technical Information Service: Spingfield, VA, USA, 1967; pp. 155–163. [Google Scholar]
- Lison, D. Human toxicity of cobalt-containing dust and experimental studies on the mechanism of interstitial lung disease (hard metal disease). Crit. Rev. Toxicol. 1996, 26, 585–616. [Google Scholar] [CrossRef] [PubMed]
- Harding, H. Notes on the toxicology of cobalt metal. Br. J. Ind. Med. 1950, 7, 76. [Google Scholar] [CrossRef] [PubMed]
- Davison, A.; Haslam, P.; Corrin, B.; Coutts, I.; Dewar, A.; Riding, W.; Studdy, P.; Newman-Taylor, A. Interstitial lung disease and asthma in hard-metal workers: Bronchoalveolar lavage, ultrastructural, and analytical findings and results of bronchial provocation tests. Thorax 1983, 38, 119–128. [Google Scholar] [CrossRef] [PubMed]
- Sahle, W.; Laszlo, I.; Krantz, S.; Christensson, B. Airborne tungsten oxide whiskers in a hard-metal industry. Preliminary findings. Ann. Occup. Hyg. 1994, 38, 37–44. [Google Scholar]
- Sahle, W. Possible role of tungsten oxide whiskers in. Chest 1992, 102, 1310. [Google Scholar] [CrossRef] [PubMed]
- Rousseau, M.C.; Straif, K.; Siemiatycki, J. IARC carcinogen update. Environ. Health Perspect. 2005, 113, A580–A581. [Google Scholar] [CrossRef] [PubMed]
- Lombaert, N.; De Boeck, M.; Decordier, I.; Cundari, E.; Lison, D.; Kirsch-Volders, M. Evaluation of the apoptogenic potential of hard metal dust (WC-Co), tungsten carbide and metallic cobalt. Toxicol. Lett. 2004, 154, 23–34. [Google Scholar] [CrossRef] [PubMed]
- Kominami, K.; Nakabayashi, J.; Nagai, T.; Tsujimura, Y.; Chiba, K.; Kimura, H.; Miyawaki, A.; Sawasaki, T.; Yokota, H.; Manabe, N.; et al. The molecular mechanism of apoptosis upon caspase-8 activation: Quantitative experimental validation of a mathematical model. Biochim. Biophys. Acta 2012, 1823, 1825–1840. [Google Scholar] [CrossRef] [PubMed]
- Moche, H.; Chevalier, D.; Barois, N.; Lorge, E.; Claude, N.; Nesslany, F. Tungsten carbide-cobalt as a nanoparticulate reference positive control in in vitro genotoxicity assays. Toxicol. Sci. 2014, 137, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Lombaert, N.; Castrucci, E.; Decordier, I.; Van Hummelen, P.; Kirsch-Volders, M.; Cundari, E.; Lison, D. Hard-metal (WC-Co) particles trigger a signaling cascade involving p38 MAPK, HIF-1α, HMOX1, and p53 activation in human PBMC. Arch. Toxicol. 2013, 87, 259–268. [Google Scholar] [CrossRef] [PubMed]
- Brüne, B.; Zhou, J.; Von Knethen, A. Nitric oxide, oxidative stress, and apoptosis. Kidney Int. 2003, 63, S22–S24. [Google Scholar] [CrossRef] [PubMed]
- Chandel, N.S.; Vander Heiden, M.G.; Thompson, C.B.; Schumacker, P.T. Redox regulation of p53 during hypoxia. Oncogene 2000, 19, 3840. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Mohamed, M.A.A.; Park, S.Y.; Yi, T.H. Fucosterol protects cobalt chloride induced inflammation by the inhibition of hypoxia-inducible factor through PI3K/Akt pathway. Int. Immunopharmacol. 2015, 29, 642–647. [Google Scholar] [CrossRef] [PubMed]
- Chachami, G.; Simos, G.; Hatziefthimiou, A.; Bonanou, S.; Molyvdas, P.A.; Paraskeva, E. Cobalt induces hypoxia-inducible factor-1α expression in airway smooth muscle cells by a reactive oxygen species- and PI3K-dependent mechanism. Am. J. Respir. Cell Mol. Biol. 2004, 31, 544–551. [Google Scholar] [CrossRef] [PubMed]
- Chandel, N.S.; McClintock, D.S.; Feliciano, C.E.; Wood, T.M.; Melendez, J.A.; Rodriguez, A.M.; Schumacker, P.T. Reactive oxygen species generated at mitochondrial complex III stabilize hypoxia-inducible factor-1α during hypoxia: A mechanism of O2 sensing. J. Biol. Chem. 2000, 275, 25130–25138. [Google Scholar] [CrossRef] [PubMed]
- Stohs, S.J.; Bagchi, D. Oxidative mechanisms in the toxicity of metal ions. Free Radic. Biol. Med. 1995, 18, 321–336. [Google Scholar] [CrossRef]
- Lison, D.; Carbonnelle, P.; Mollo, L.; Lauwerys, R.; Fubini, B. Physicochemical mechanism of the interaction between cobalt metal and carbide particles to generate toxic activated oxygen species. Chem. Res. Toxicol. 1995, 8, 600–606. [Google Scholar] [CrossRef] [PubMed]
- Bastian, S.; Busch, W.; Kuhnel, D.; Springer, A.; Meissner, T.; Holke, R.; Scholz, S.; Iwe, M.; Pompe, W.; Gelinsky, M.; et al. Toxicity of tungsten carbide and cobalt-doped tungsten carbide nanoparticles in mammalian cells in vitro. Environ. Health Perspect. 2009, 117, 530–536. [Google Scholar] [CrossRef] [PubMed]
- Limbach, L.K.; Wick, P.; Manser, P.; Grass, R.N.; Bruinink, A.; Stark, W.J. Exposure of engineered nanoparticles to human lung epithelial cells: Influence of chemical composition and catalytic activity on oxidative stress. Environ. Sci. Technol. 2007, 41, 4158–4163. [Google Scholar] [CrossRef] [PubMed]
- Lison, D.; Lauwerys, R. In vitro cytotoxic effects of cobalt-containing dusts on mouse peritoneal and rat alveolar macrophages. Environ. Res. 1990, 52, 187–198. [Google Scholar] [CrossRef]
- Busch, W.; Kuhnel, D.; Schirmer, K.; Scholz, S. Tungsten carbide cobalt nanoparticles exert hypoxia-like effects on the gene expression level in human keratinocytes. BMC Genom. 2010, 11, 65. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.D.; Zhao, J.; Bowman, L.; Shi, X.; Castranova, V.; Ding, M. Tungsten carbide-cobalt particles activate Nrf2 and its downstream target genes in JB6 cells possibly by ROS generation. J. Environ. Pathol. Toxicol. Oncol. 2010, 29, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Davies, K.J.A.; Forman, H.J. Oxidative stress response and Nrf2 signaling in aging. Free Radic. Biol. Med. 2015, 88, 314–336. [Google Scholar] [CrossRef] [PubMed]
- Fenoglio, I.; Corazzari, I.; Francia, C.; Bodoardo, S.; Fubini, B. The oxidation of glutathione by cobalt/tungsten carbide contributes to hard metal-induced oxidative stress. Free Radic. Res. 2008, 42, 437–745. [Google Scholar] [CrossRef] [PubMed]
- Armstead, A.L.; Arena, C.B.; Li, B. Exploring the potential role of tungsten carbide cobalt (WC-Co) nanoparticle internalization in observed toxicity toward lung epithelial cells in vitro. Toxicol. Appl. Pharmacol. 2014, 278, 1–8. [Google Scholar] [CrossRef] [PubMed]
- A-Rahman, A.R.; Spencer, D. Totally implantable vascular access devices for cystic fibrosis. Cochrane Database Syst. Rev. 2003. [Google Scholar] [CrossRef]
- Ding, M.; Kisin, E.R.; Zhao, J.; Bowman, L.; Lu, Y.; Jiang, B.; Leonard, S.; Vallyathan, V.; Castranova, V.; Murray, A.R.; et al. Size-dependent effects of tungsten carbide-cobalt particles on oxygen radical production and activation of cell signaling pathways in murine epidermal cells. Toxicol. Appl. Pharmacol. 2009, 241, 260–268. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Bowman, L.; Magaye, R.; Leonard, S.S.; Castranova, V.; Ding, M. Apoptosis induced by tungsten carbide-cobalt nanoparticles in JB6 cells involves ROS generation through both extrinsic and intrinsic apoptosis pathways. Int. J. Oncol. 2013, 42, 1349–1359. [Google Scholar] [CrossRef] [PubMed]
- Kuhnel, D.; Scheffler, K.; Wellner, P.; Meissner, T.; Potthoff, A.; Busch, W.; Springer, A.; Schirmer, K. Comparative evaluation of particle properties, formation of reactive oxygen species and genotoxic potential of tungsten carbide based nanoparticles in vitro. J. Hazard. Mater. 2012, 227–228, 418–426. [Google Scholar] [CrossRef] [PubMed]
- Wigger, H.; Steinfeldt, M.; Bianchin, A. Environmental benefits of coatings based on nano-tungsten-carbide cobalt ceramics. J. Clean.Prod. 2017, 148, 212–222. [Google Scholar] [CrossRef]
- Lombaert, N.; Lison, D.; Van Hummelen, P.; Kirsch-Volders, M. In vitro expression of hard metal dust (WC-Co)-responsive genes in human peripheral blood mononucleated cells. Toxicol. Appl. Pharmacol. 2008, 227, 299–312. [Google Scholar] [CrossRef] [PubMed]
- Harris, R.M.; Williams, T.D.; Hodges, N.J.; Waring, R.H. Reactive oxygen species and oxidative DNA damage mediate the cytotoxicity of tungsten-nickel-cobalt alloys in vitro. Toxicol. Appl. Pharmacol. 2011, 250, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Schuster, B.E.; Roszell, L.E.; Murr, L.E.; Ramirez, D.A.; Demaree, J.D.; Klotz, B.R.; Rosencrance, A.B.; Dennis, W.E.; Bao, W.; Perkins, E.J.; et al. In vivo corrosion, tumor outcome, and microarray gene expression for two types of muscle-implanted tungsten alloys. Toxicol. Appl. Pharmacol. 2012, 265, 128–138. [Google Scholar] [CrossRef] [PubMed]
- Harris, R.M.; Williams, T.D.; Waring, R.H.; Hodges, N.J. Molecular basis of carcinogenicity of tungsten alloy particles. Toxicol. Appl. Pharmacol. 2015, 283, 223–233. [Google Scholar] [CrossRef] [PubMed]
- Roedel, E.Q.; Cafasso, D.E.; Lee, K.W.; Pierce, L.M. Pulmonary toxicity after exposure to military-relevant heavy metal tungsten alloy particles. Toxicol. Appl. Pharmacol. 2012, 259, 74–86. [Google Scholar] [CrossRef] [PubMed]
- Emond, C.A.; Vergara, V.B.; Lombardini, E.D.; Mog, S.R.; Kalinich, J.F. The Role of the Component Metals in the Toxicity of Military-Grade Tungsten Alloy. Toxics 2015, 3, 499–514. [Google Scholar] [CrossRef] [PubMed]
- Vergara, V.B.; Emond, C.A.; Kalinich, J.F. Tissue distribution patterns of solubilized metals from internalized tungsten alloy in the F344 rat. AIMS Environ. Sci. 2016, 3, 290–304. [Google Scholar] [CrossRef]
- Adams, V.H.; Dennis, W.E.; Bannon, D.I. Toxic and transcriptional responses of PC12 cells to soluble tungsten alloy surrogates. Toxicol. Rep. 2015, 2, 1437–1444. [Google Scholar] [CrossRef] [PubMed]
- Verma, R.; Xu, X.; Jaiswal, M.K.; Olsen, C.; Mears, D.; Caretti, G.; Galdzicki, Z.J.T. In vitro profiling of epigenetic modifications underlying heavy metal toxicity of tungsten-alloy and its components. Toxicol. Appl. Pharmacol. 2011, 253, 178–187. [Google Scholar] [CrossRef] [PubMed]
- Rakusan, K.; Cicutti, N.; Kolar, F. Cardiac function, microvascular structure, and capillary hematocrit in hearts of polycythemic rats. Am. J. Physiol. Heart Circ. Physiol. 2001, 281, H2425–H2431. [Google Scholar] [CrossRef] [PubMed]
- Salnikow, K.; Donald, S.P.; Bruick, R.K.; Zhitkovich, A.; Phang, J.M.; Kasprzak, K.S. Depletion of intracellular ascorbate by the carcinogenic metals nickel and cobalt results in the induction of hypoxic stress. J. Biol. Chem. 2004, 279, 40337–40344. [Google Scholar] [CrossRef] [PubMed]
- Moriyama, H.; Kobayashi, M.; Takada, T.; Shimizu, T.; Terada, M.; Narita, J.-I.; Maruyama, M.; Watanabe, K.; Suzuki, E.; Gejyo, F. Two-dimensional analysis of elements and mononuclear cells in hard metal lung disease. Am. J. Respir. Crit. Care Med. 2007, 176, 70–77. [Google Scholar] [CrossRef] [PubMed]
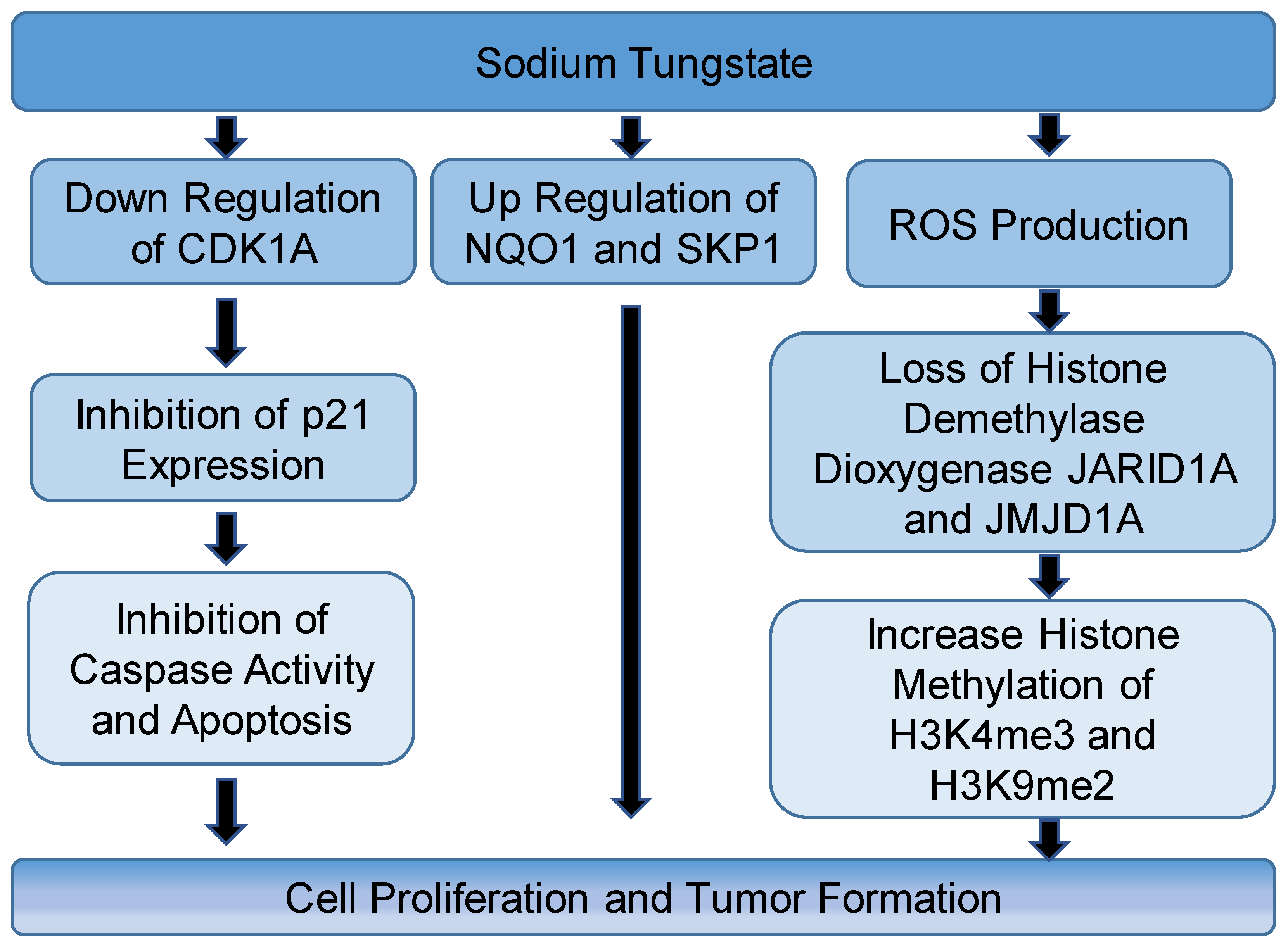
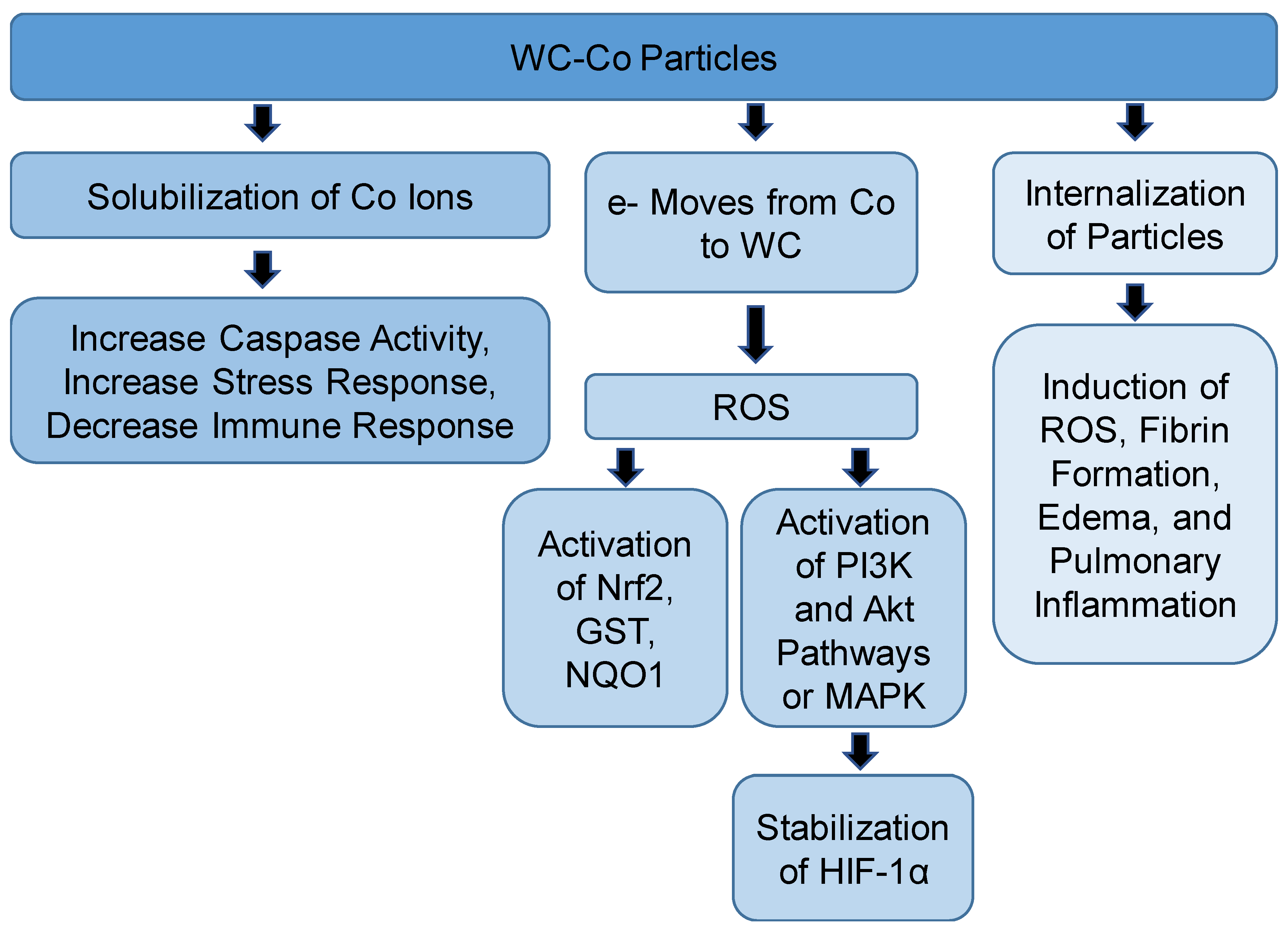
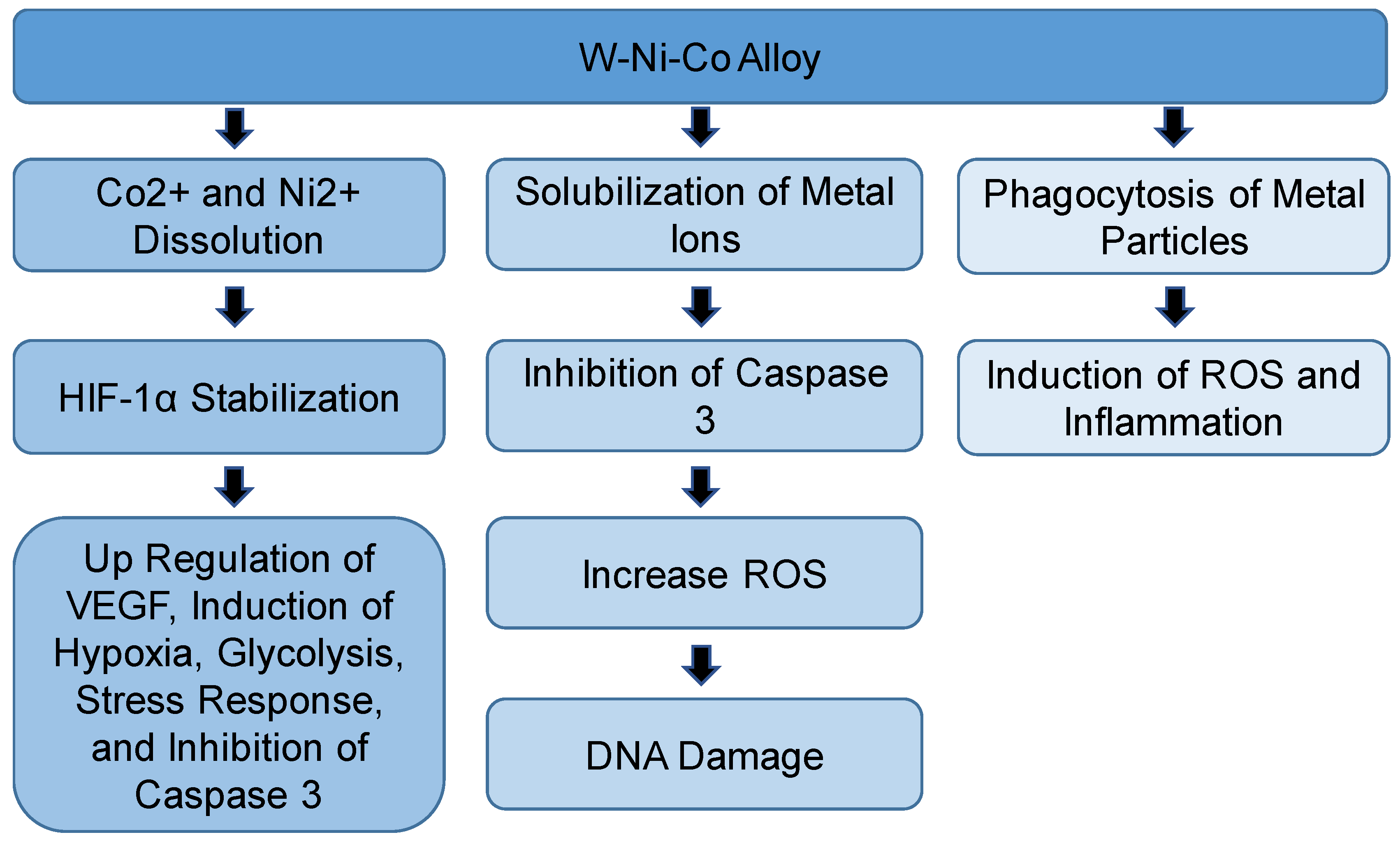
| Compound/s | Model | Route of Exposure | Effects | Outcomes and Proposed mechanisms | Reference |
|---|---|---|---|---|---|
| Sodium tungstate | Immortalized human bronchial epithelial cells (Beas-2B) |
|
| Tumorgenicity Degradation of demethylases or induction of ROS | [11,15] |
| Tungstate-based shield | Breast cancer patients | Tungsten-based shield placed during intraoperative radiotherapy | Tungsten was detected in urine of patients with mastectomy | Mobilization of tungsten | [16] |
| Sodium tungstate | BALB/c mice injected with 66Cl4 cells | Tungsten added to drinking water | Lung metastasis | Activation of fibroblasts, myeloid derived suppressor cells, and matrix proteinases (changes in microenvironment of tumor) | [16] |
| Sodium tungstate | 66Cl4 cells | In vitro | No change in proliferation | Tungsten does not directly induce tumor formation | [16] |
| Tungsten oxide microparticle and nanoparticle | Wistar rats | Oral administration | Induced DNA damage, micronuclei formation reduction of glutathione, and catalase | ROS, genotoxicity | [17] |
| Tungsten carbide nanoparticles | HepG2 and HaCat cells | In vitro | ROS and chromosomal instability | Small size, and presence of carbon black in WC particles | [49] |
| Tungsten carbide-cobalt | Human peripheral blood mononuclear cells | In vitro | Apoptosis, ROS, oxidative stress, inhibition of immune response | Induction of HIF-1α | [27,30,51] |
| Tungsten carbide-cobalt nanoparticles | HaCaT cells | In vitro | Increase transcription of genes involved in hypoxia pathways | Effects similar to Co ion’s effect | [41] |
| Tungstate carbide- cobalt nanoparticles | Mouse epidermal JB6 cells | In vitro | Activation Nrf2 and NQO1 | ROS | [43] |
| Tungsten carbide-cobalt micro or nano particles | Beas-2B cells | In vitro | Decrease in cell viability, apoptosis, oxidative stress | Internalization of WC-Co inside the cells | [45] |
| Tungsten carbide-cobalt nanoparticles | JB6P + cells | In vitro | Activation of Ap-1, Nf-KB, MAPK, depletion of GSH | Oxidative stress | [47,48] |
| Tungsten alloy (W/Co/Ni) | F344 rats | Intramuscular implantation | Incidence of rhabdomyosarcoma, increase in RBCs, WBCs, neutrophils | [49] | |
| W/Co/Ni or W/Co/Fe particles | L6-c11 | In vitro | DNA damage, ROS | Dissolution of Co and Ni ions and stabilization of HIF-1α | [52] |
| W/Ni/Co or W/Ni/Fe pellets | F344 rats | Intramuscular implantation | Aggressive rhabdomyosarcomas formed around W/Co/Ni pellets only | Mobilization of the metal ions from the pellet | [53] |
| W/Ni/Co | hSKMC cells | In vitro |
| Carcinogenesis | [54] |
| W/Ni/Fe or W/Ni/Co | Rat | Intratracheal instillation | ROS, induction of inflammatory cytokines | Phagocytosis of metal particles by lung macrophages | [55] |
| W/Ni/Co or W/Ni/Fe alloy | B6C3FI mouse | Implantation in leg muscles | Rhabdomyosarcoma formation around the W/Ni/Co pellet | [2] | |
| W/Ta, Co/Ta, W/Ni/Co or W/Ni/Ta | B6C3FI mouse | Implantation in hind limb |
|
| [56] |
| W/Ni/Co | F344 rats | Implantation | Systemic distribution of the metals. Liver, kidney and spleen are most affected organs | [57] | |
| W, Co, Ni soluble single or paired metal salts | PCl2 cells | In vitro | Ni and Co in absence or presence of W led to changes in gene expression | W has minimal effect on the observed effects | [58] |
| W, Co, Ni | Hippocampal primary neuronal culture, mouse myoblast (C2C12) | In vitro | W/Ni/Co induced epigenetic alteration | W/Ni/Co synergistically caused the effect. | [59] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wasel, O.; Freeman, J.L. Comparative Assessment of Tungsten Toxicity in the Absence or Presence of Other Metals. Toxics 2018, 6, 66. https://doi.org/10.3390/toxics6040066
Wasel O, Freeman JL. Comparative Assessment of Tungsten Toxicity in the Absence or Presence of Other Metals. Toxics. 2018; 6(4):66. https://doi.org/10.3390/toxics6040066
Chicago/Turabian StyleWasel, Ola, and Jennifer L. Freeman. 2018. "Comparative Assessment of Tungsten Toxicity in the Absence or Presence of Other Metals" Toxics 6, no. 4: 66. https://doi.org/10.3390/toxics6040066
APA StyleWasel, O., & Freeman, J. L. (2018). Comparative Assessment of Tungsten Toxicity in the Absence or Presence of Other Metals. Toxics, 6(4), 66. https://doi.org/10.3390/toxics6040066





