Hydroquinone Ecotoxicity: Unveiling Risks in Soil and River Ecosystems with Insights into Microbial Resilience
Abstract
1. Introduction
- (a)
- To evaluate the toxicity of HQ on key indicator organisms in soil and water for which little information exists.
- (b)
- To evaluate for the first time the toxicity of HQ on 16 S rRNA gene-sequenced fluvial and soil microbial communities in order to more realistically assess the impact on these environments.
2. Materials and Methods
2.1. Reagents
2.2. Daphnia Magna Assay
2.3. Aliivibrio Fischeri Assay
2.4. Allium cepa Assay
2.5. Eisenia Fetida Assay
2.6. River and Soil Microorganisms Community Assay
2.6.1. River Samples
2.6.2. Soil Samples
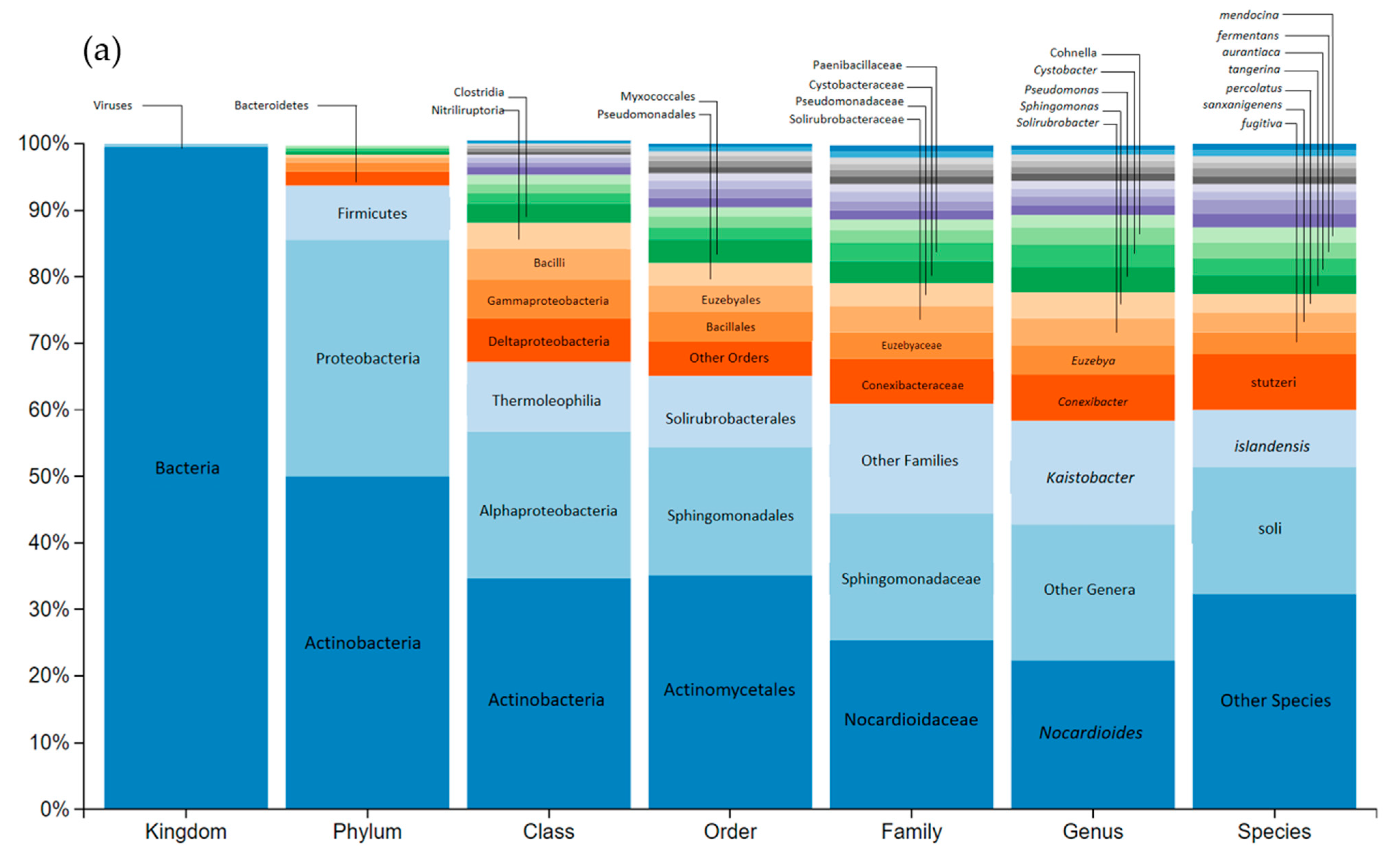
2.6.3. Genetic Sequencing of River and Soil Microorganisms
2.6.4. Community-Level Physiological Profiling (CLPP) of River and Soil Microorganisms
2.7. Statistics and Graphic Representation
3. Results and Discussion
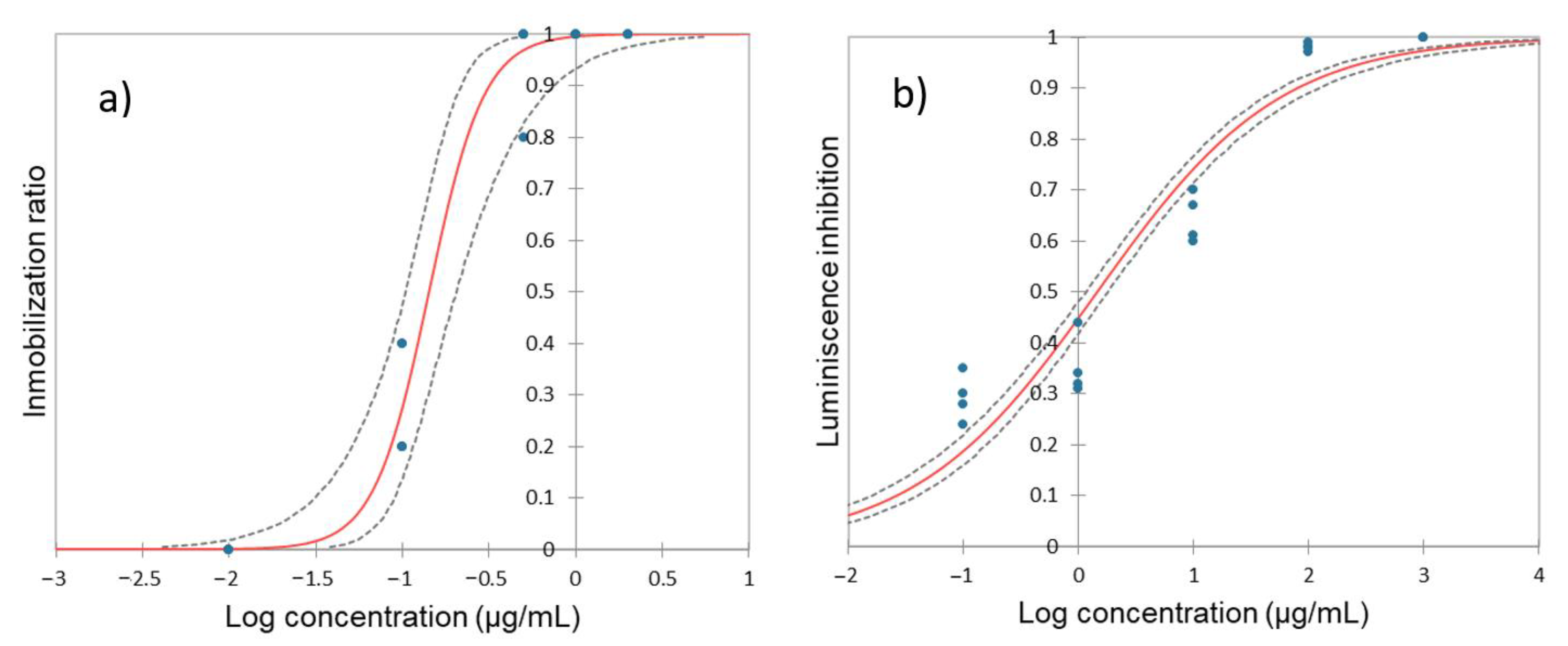
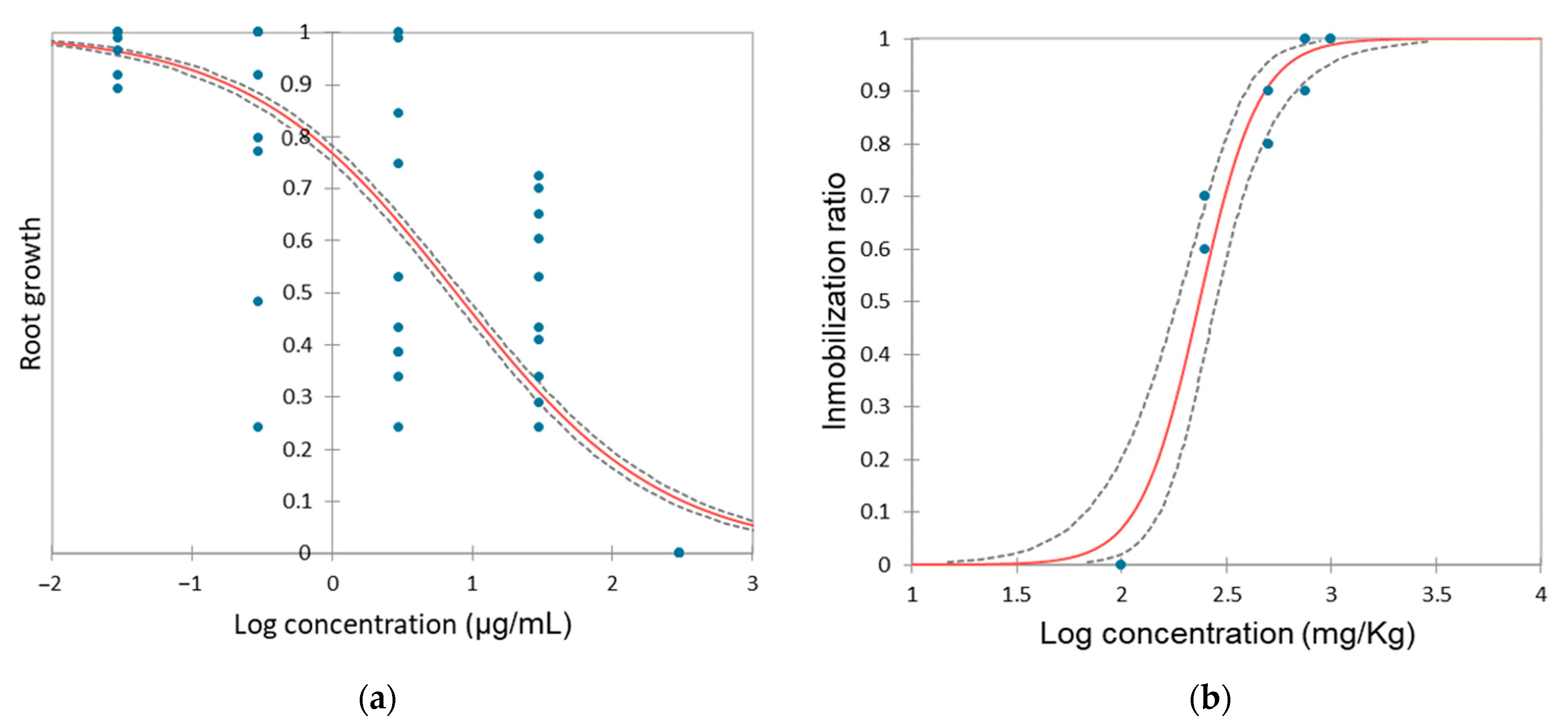
3.1. Impact of Hydroquinone on Daphnia magna
3.2. Impact of Hydroquinone on A. fisheri
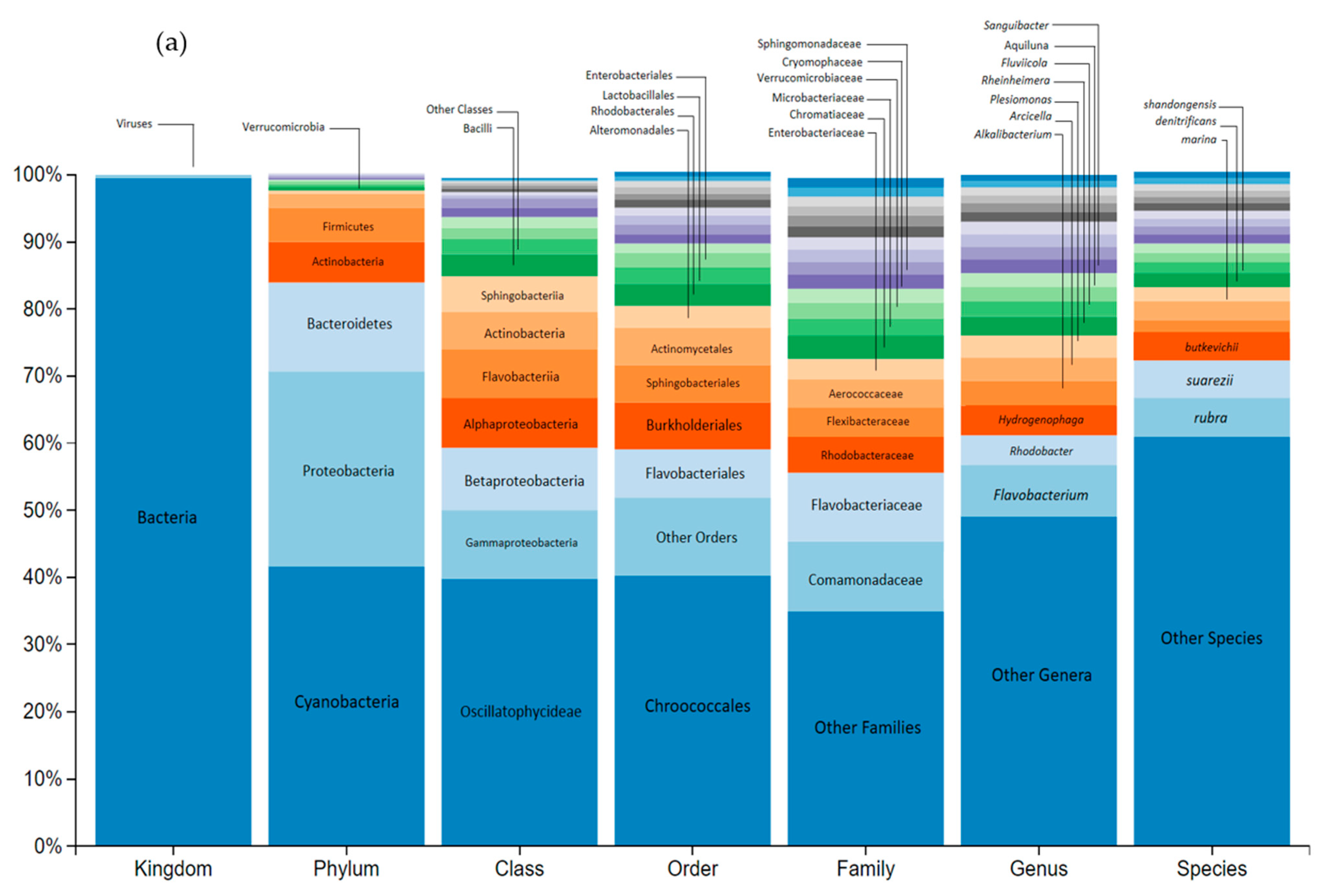
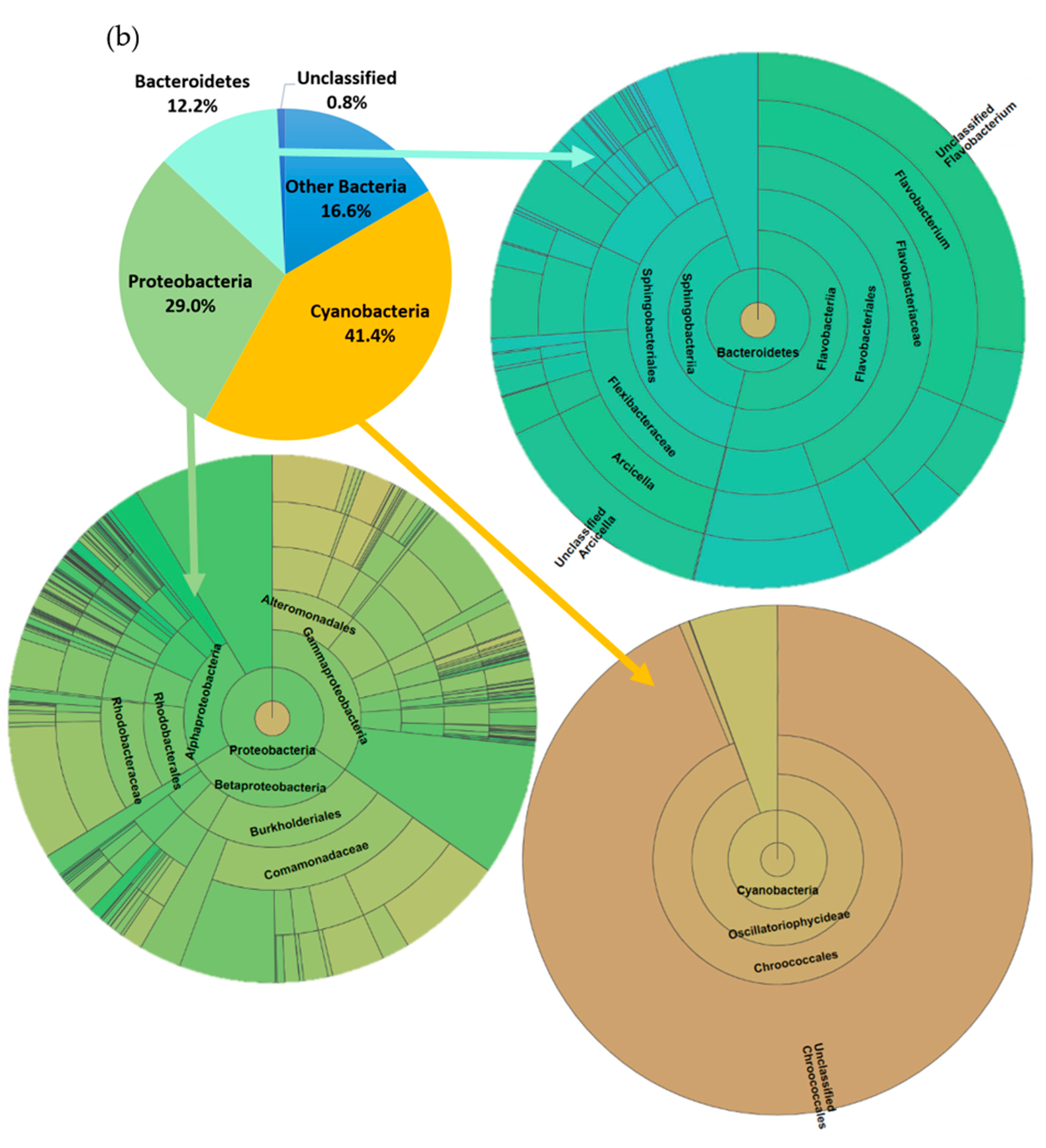
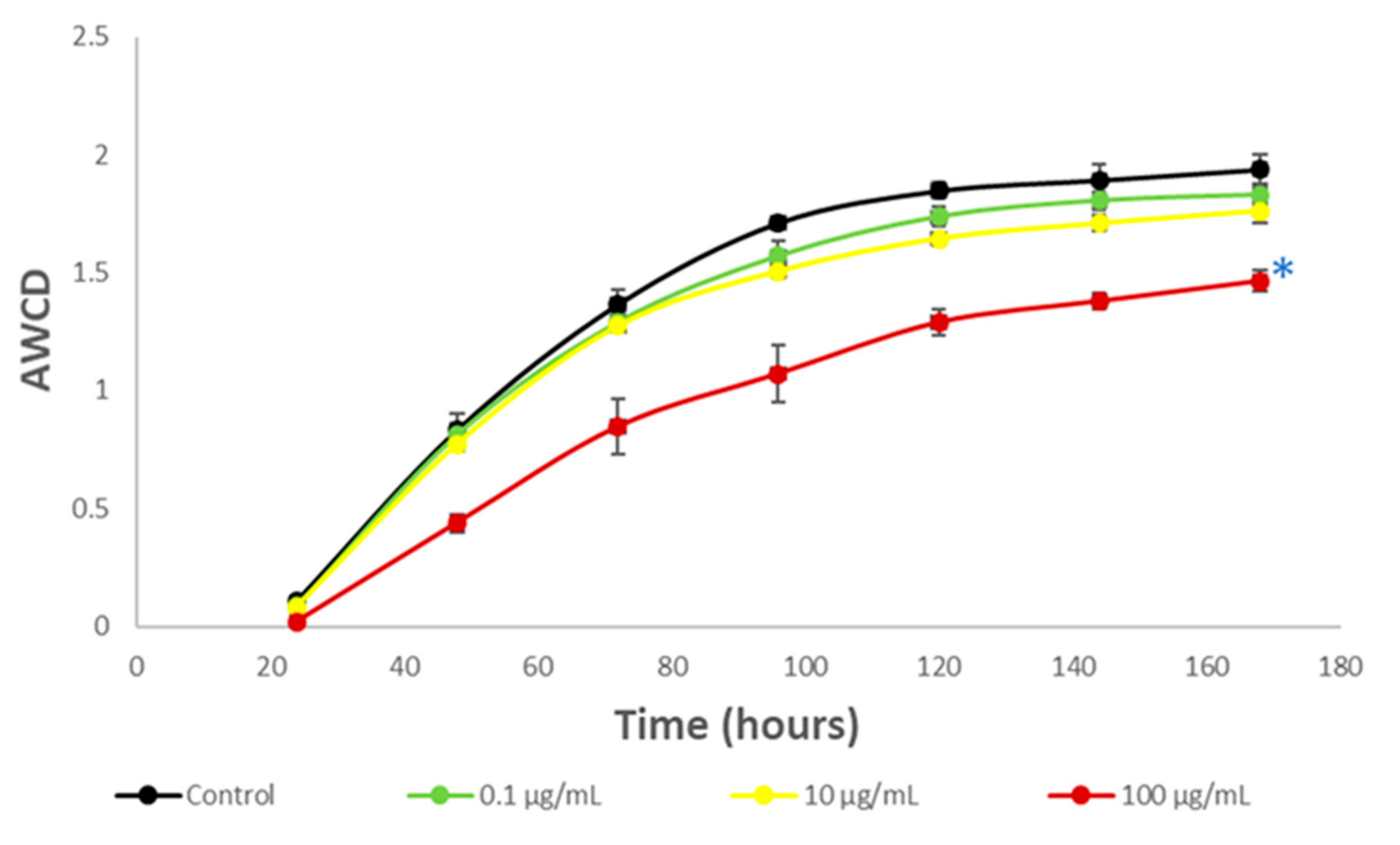
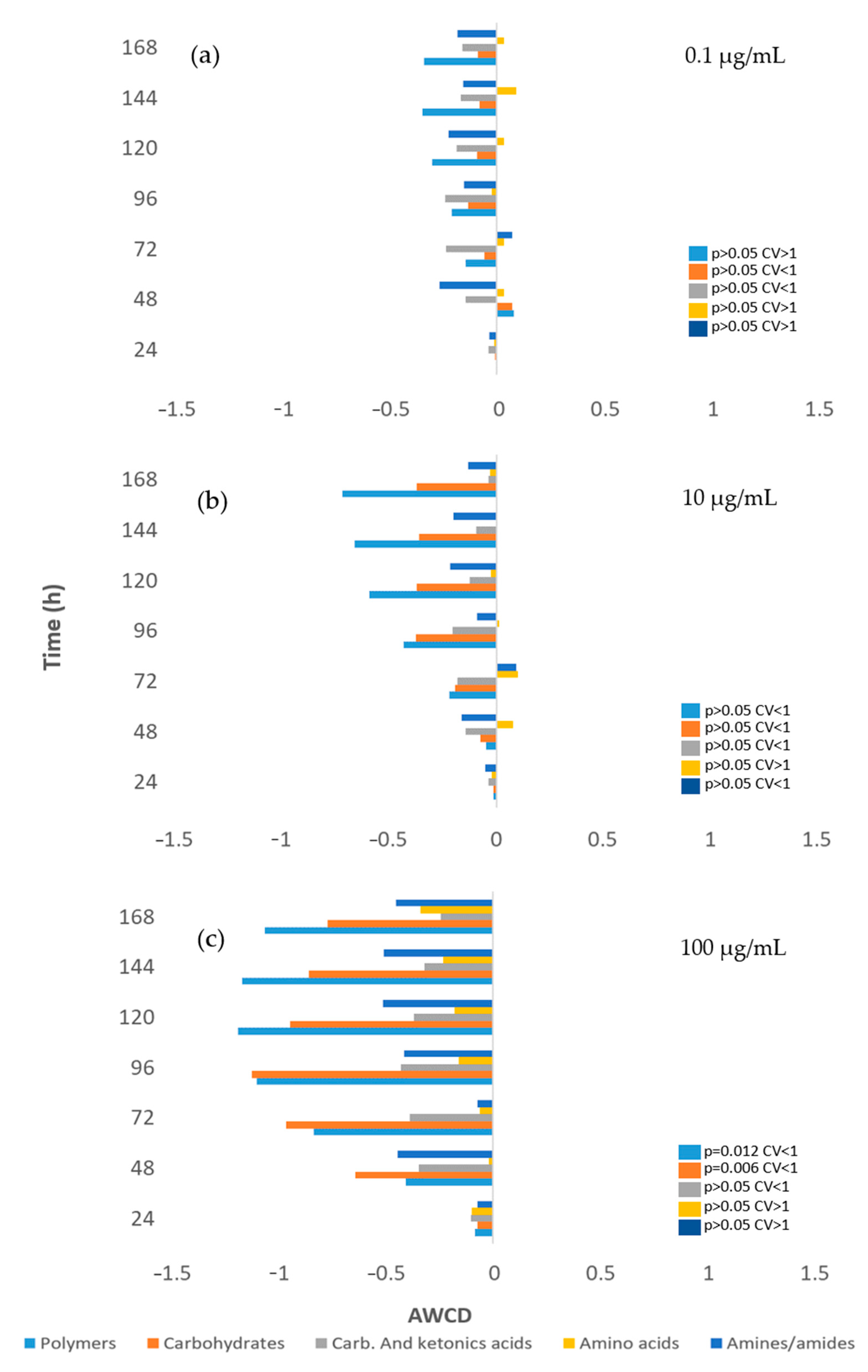
3.3. Impact on River Microbial Communities: Growth and Community-Level Physiological Profiling (CLPP)
3.4. Impact of Hydroquinone on Allium cepa
3.5. Impact of Hydroquinone on Eisenia fetida
3.6. Impact on Soil Microbial Communities: Growth and Community-Level Physiological Profiling (CLPP)
4. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Physical-Chemical Analysis of the River Water Sample | |
| HCO−3 (mg/L) | 313 |
| TDS (mg/L) | 1925 |
| MES (mg/L) | 6 |
| Cl− (mg/L) | 618 ± 93 |
| SO42− (mg/L) | 415 ± 62 |
| NO3− (mg/L) | 17.7 ± 2.7 |
| NO2− (mg/L) | <0.05 |
| F− (mg/L) | 0.071 ± 0.011 |
| PO43− (mg/L) | 0.6 ± 0.09 |
| NH4+(mg/L) | <0.1 |
| O2 (mg/L) | 2.3 |
| DQO (O2) (mg/L) | <25 |
| DBO5 (mg/L) | <5 |
| Ca (mg/L) | 235 ± 80 |
| Mg (mg/L) | 38.1 ± 13.7 |
| Na (mg/L) | 415 ± 95 |
| K (mg/L) | 6.08 ± 1.95 |
| Soil Composition | Surface Soil | 30 cm Deep Soil |
| Clay content (%) | 20.98 | 23.61 |
| Sand content (%) | 16.08 | 13.10 |
| Silt content (%) | 62.94 | 63.29 |
| pH | 7.9 ± 0.5 | 8.1 ± 0.5 |
| K (mg/L) | 238 ± 40 | 208 ± 35 |
| Mg (mg/Kg) | 244 ± 39 | 242 ± 39 |
| P Oslen (mg/Kg) | 13 ± 2 | 10 ± 1.7 |
| EC1:5 (dS/m) | 0.6 ± 0.09 | 0.4 ± 0.06 |
| Organic matter (g/100 g) | 2.46 ± 0.31 | 2.35 ± 0.30 |
References
- Braga, V.C.C.; Pianetti, G.A.; César, I.C. Comparative stability of arbutin in Arctostaphylos uva-ursi by a new comprehensive stability-indicating HPLC method. Phytochem. Anal. 2020, 6, 884–891. [Google Scholar] [CrossRef]
- ECHA; European Chemical Agency. ECHA, Hydroquinone. Available online: https://echa.europa.eu/substance-information/-/substanceinfo/100.004.199 (accessed on 17 January 2024).
- Matsumoto, M.; Todo, H.; Akiyama, T.; Hirata-Koizumi, M.; Sugibayashi, K.; Ikarashi, Y.; Ono, A.; Hirose, A.; Yokoyama, K. Risk assessment of skin lightening cosmetics containing hydroquinone. Regul. Toxicol. Pharmacol. 2016, 16, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Remberger, M.; Hynning, P.A.; Neilson, A.H. 2,5-dichloro-3,6-dihydroxybenzo-1,4-quinone—identification of a new organochlorine compound in kraft mill bleachery effluents. Environ. Sci. Technol. 1991, 25, 1903–1907. [Google Scholar] [CrossRef]
- Neven, L.; Barich, H.; Rutten, R.; De Wael, K. Novel (photo)electrochemical analysis of aqueous industrial samples containing phenols. Microchem. J. 2022, 181, 107778. [Google Scholar] [CrossRef]
- Choi, Y.; Jeon, J.; Kim, S.D. Identification of biotransformation products of organophosphate ester from various aquatic species by suspect and non-target screening approach. Water Res. 2021, 200, 117201. [Google Scholar] [CrossRef] [PubMed]
- Marchlewicz, A.; Guzik, U.; Wojcieszynska, D. Over-the-Counter Monocyclic Non-Steroidal Anti-Inflammatory Drugs in Environment-Sources, Risks, Biodegradation. Water Air Soil. Pollut. 2015, 226, 355. [Google Scholar] [CrossRef] [PubMed]
- Zur, J.; Wojcieszynska, D.; Hupert-Kocurek, K.; Marchlewicz, A.; Guzik, U. Paracetamol—toxicity and microbial utilization. Pseudomonas moorei KB4 as a case study for exploring degradation pathway. Chemosphere 2018, 206, 192–202. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Yu, H.; Qin, H.; Long, Y.; Ye, J.; Qu, Y. Bisphenol A degradation pathway and associated metabolic networks in Escherichia coli harboring the gene encoding CYP450. J. Hazard. Mater. 2020, 388, 121737. [Google Scholar] [CrossRef] [PubMed]
- Bolobajev, J.; Oncu, N.B.; Viisimaa, M.; Trapido, M.; Balcioglu, I.; Goi, A. Column experiment on activation aids and biosurfactant application to the persulphate treatment of chlorophene-contaminated soil. Environ. Technol. 2015, 36, 348–357. [Google Scholar] [CrossRef]
- Hoang Nhat Phong, V.; Le, G.K.; Thi Minh Hong, N.; Xuan-Thanh, B.; Khanh Hoang, N.; Rene, E.R.; Thi Dieu Hien, V.; Ngoc-Dan Thanh, C.; Mohan, R. Acetaminophen micropollutant: Historical and current occurrences, toxicity, removal strategies and transformation pathways in different environments. Chemosphere 2019, 236, 124391. [Google Scholar] [CrossRef]
- Wang, Y.; Li, X.; Sun, X. The transformation mechanism and eco-toxicity evaluation of butylated hydroxyanisole in environment. Ecotoxicol. Environ. Saf. 2022, 231, 113179. [Google Scholar] [CrossRef]
- Tentscher, P.R.; Escher, B.I.; Schlichting, R.; König, M.; Bramaz, N.; Schirmer, K.; von Gunten, U. Toxic effects of substituted p-benzoquinones and hydroquinones in in vitro bioassays are altered by reactions with the cell assay medium. Water Res. 2021, 202, 117415. [Google Scholar] [CrossRef] [PubMed]
- Di Marzio, W.D.; Saenz, M.; Alberdi, J.; Tortorelli, M.; Silvana, G. Risk assessment of domestic and industrial effluents unloaded into a freshwater environment. Ecotoxicol. Environ. Saf. 2005, 61, 380–391. [Google Scholar] [CrossRef]
- Otitoju, O.B.; O Alfred, M.; Olorunnisola, C.G.; Aderinola, F.T.; O Ogunlaja, O.; Olukanni, O.D.; Ogunlaja, A.; O Omorogie, M.; I Unuabonah, E. Distribution and toxicity of dihydroxybenzenes in drinking water sources in Nigeria. RSC Adv. 2024, 14, 982–994. [Google Scholar] [CrossRef]
- Karami-Kolmoti, P.; Beitollahi, H.; Modiri, S. Electrochemical Sensor for Simple and Sensitive Determination of Hydroquinone in Water Samples Using Modified Glassy Carbon Electrode. Biomedicines 2023, 11, 1869. [Google Scholar] [CrossRef]
- Yang, L.; Zhao, H.; Fan, S.; Li, B.; Li, C.-P. A highly sensitive electrochemical sensor for simultaneous determination of hydroquinone and bisphenol A based on the ultrafine Pd nanoparticle@TiO2 functionalized SiC. Anal. Chim. Acta 2014, 852, 28–36. [Google Scholar] [CrossRef]
- Hernandez, S.R.; Kergaravat, S.V.; Isabel Pividori, M. Enzymatic electrochemical detection coupled to multivariate calibration for the determination of phenolic compounds in environmental samples. Talanta 2013, 106, 399–407. [Google Scholar] [CrossRef]
- Varela, A.; Martins, C.; Nunez, O.; Martins, I.; Houbraken, J.A.M.P.; Martins, T.M.; Leitao, M.C.; McLellan, I.; Vetter, W.; Galceran, M.T.; et al. Understanding fungal functional biodiversity during the mitigation of environmentally dispersed pentachlorophenol in cork oak forest soils. Environ. Microbiol. 2015, 17, 2922–2934. [Google Scholar] [CrossRef]
- Lin, J.; Chen, J.; Wang, Y.; Cai, X.; Wei, X.; Qiao, X. More toxic and photoresistant products from photodegradation of fenoxaprop-p-ethyl. J. Agric. Food Chem. 2008, 56, 8226–8230. [Google Scholar] [CrossRef] [PubMed]
- Candido, N.R.; Modolo, L.V.; Pasa, V.M.D.; de Fatima, A. Pyroligneous Acids of Coconut Shell, Black Wattle and Eucalyptus: Physical-Chemical Characterization and in vitro Evaluation as Potential Urease Inhibitors. Quim. Nova 2023, 46, 961–971. [Google Scholar] [CrossRef]
- REACH, Authorization, and Restriction of Chemicals: 01/12/2023. Available online: http://data.europa.eu/eli/reg/2006/1907/oj (accessed on 17 January 2024).
- Baehrs, H.; Putschew, A.; Steinberg, C.E.W. Toxicity of hydroquinone to different freshwater phototrophs is influenced by time of exposure and pH. Environ. Sci. Pollut. Res. 2013, 20, 146–154. [Google Scholar] [CrossRef] [PubMed]
- Pandey, D.K.; Mishra, N.; Singh, P. Relative phytotoxicity of hydroquinone on rice (Oryza sativa L.) and associated aquatic weed green musk chary (Chary zeylanica Willd.). Pestic. Biochem. Physiol. 2005, 83, 82–96. [Google Scholar] [CrossRef]
- Briggs, G.G.; Henderson, I.F. Some Factors Affecting the Toxicity of Poisons to the Slug Deroceras-reticulatum (Muller) (Pulmonata, Limacidae). Crop Prot. 1987, 6, 341–346. [Google Scholar] [CrossRef]
- Lahnsteiner, F. The sensitivity and reproducibility of the zebrafish (Danio rerio) embryo test for the screening of waste water quality and for testing the toxicity of chemicals. Atla-Altern. Lab. Anim. 2008, 36, 299–311. [Google Scholar] [CrossRef] [PubMed]
- Abugazleh, M.K.; Ali, H.M.; Chester, J.A.; Al-Fa’ouri, A.M.; Bouldin, J.L. Aquatic toxicity of hydroquinone and catechol following metal oxide treatment to Ceriodaphnia dubia and Pimephales promelas. Ecotoxicology 2023, 32, 656–665. [Google Scholar] [CrossRef]
- Pereira, P.; Enguita, F.J.; Ferreira, J.; Leitão, A.L. DNA damage induced by hydroquinone can be prevented by fungal detoxification. Toxicol. Rep. 2014, 1, 1096–1105. [Google Scholar] [CrossRef]
- Stadnichenko, A.P.; Pogorelova, N.S.; Rudenko, S.A. The Effect of Different Concentrations of Hydroquinone on Horn Snails (Gastropoda, Pulmonata, Bulinidae) Infected with Parthenitae of Tylodelphys-Excavata (Trematoda, Diplostomatidae). Parazitologiya 1991, 25, 462–467. [Google Scholar]
- Devillers, J.; Boule, P.; Vasseur, P.; Prevot, P.; Steiman, R.; Seiglemurandi, F.; Benoitguyod, J.L.; Nendza, M.; Grioni, C.; Dive, D.; et al. Environmental and Health Risks of Hydroquinone. Ecotoxicol. Environ. Saf. 1990, 19, 327–354. [Google Scholar] [CrossRef]
- Wang, X.D.; Yu, J.Z.; Wang, Y.; Wang, L.S. Mechanism-based quantitative structure-activity relationships for the inhibition of substituted phenols on germination rate of Cucumis sativus. Chemosphere 2002, 46, 241–250. [Google Scholar] [CrossRef]
- Zhang, X.; Linghu, S.; Chen, Z.; Gu, H.; Chen, X.; Wei, X.; Hu, X.; Yang, Y.; Gao, Y. Bacterial diversity evolution process based on physicochemical characteristics of sludge treating hydroquinone during acclimation. Environ. Sci. Pollut. Res. 2022, 29, 31686–31699. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Hu, X.; Lu, Q.; Yang, Y.; Linghu, S.; Zhang, X. Study on the differences in sludge toxicity and microbial community structure caused by catechol, resorcinol and hydroquinone with metagenomic analysis. J. Environ. Manag. 2022, 302, 114027. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Wu, B.; Pan, S.; Yang, K.; Hu, Z.; Yuan, S. Performance robustness of the UASB reactors treating saline phenolic wastewater and analysis of microbial community structure. J. Hazard. Mater. 2017, 331, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Tian, H.; Xu, X.; Qu, J.; Li, H.; Hu, Y.; Huang, L.; He, W.; Li, B. Biodegradation of phenolic compounds in high saline wastewater by biofilms adhering on aerated membranes. J. Hazard. Mater. 2020, 392, 122463. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Xiao, Q.; Hu, C.; Liu, B.; Sun, R. A comparison of the efficiency of different urease inhibitors and their effects on soil prokaryotic community in a short-term incubation experiment. Geoderma 2019, 354, 113877. [Google Scholar] [CrossRef]
- Dong, D.; Kou, Y.; Yang, W.; Chen, G.; Xu, H. Effects of urease and nitrification inhibitors on nitrous oxide emissions and nitrifying/denitrifying microbial communities in a rainfed maize soil: A 6-year field observation. Soil. Tillage Res. 2018, 180, 82–90. [Google Scholar] [CrossRef]
- Weaver, J.H.; Frederikse, H.P.R. CRC Handbook of Chemistry and Physics; CRC Press: Boca Raton, FL, USA, 1977. [Google Scholar]
- ECD; SIDS. “Hydroquinone,” CAS 123-31-9; UNEP Publications: Helsinki, Finland, 2012. [Google Scholar]
- Suresh, S.; Srivastava, V.C.; Mishra, I.M. Adsorption of catechol, resorcinol, hydroquinone, and their derivatives: A review. Int. J. Energy Environ. Eng. 2012, 3, 32. [Google Scholar] [CrossRef]
- Organisation for Economic Co-operation and Development (OECD). Test No. 202: Daphnia sp. Acute Immobilisation Test. In OECD Guidelines for the Testing of Chemicals, Section 2; OECD: Paris, France, 2004. [Google Scholar]
- UNE-EN ISO 6341; Water Quality—Determination of the Inhibition of the Mobility of Daphnia magna Straus (Cladocera Crustacea)—Acute Toxicity Test. AENOR: Madrid, Spain, 2012.
- ISO 11348; Water Quality—Determination of the Inhibitory Effect of Water Samples on the Light Emission of Aliivibrio fischeri (Luminescent Bacteria Test). International Organization for Standardization: Geneva, Switzerland, 2007.
- Fiskesjö, G. The allium test in wastewater monitoring. Environ. Toxicol. Water Qual. 1993, 8, 291–298. [Google Scholar] [CrossRef]
- Organisation for Economic Co-operation and Development (OECD). Test No. 207: Earthworm, Acute Toxicity Tests. In OECD Guidelines for the Testing of Chemicals, Section 2; OECD: Paris, France, 1984. [Google Scholar]
- Pino, M.R.; Val, J.; Mainar, A.M.; Zuriaga, E.; Español, C.; Langa, E. Acute toxicological effects on the earthworm Eisenia fetida of 18 common pharmaceuticals in artificial soil. Sci. Total Environ. 2015, 518–519, 225–237. [Google Scholar] [CrossRef]
- ISO 19458:2006; Water Quality—Sampling for Microbiological Analysis. AENOR: Madrid, Spain, 2007.
- Pino-Otín, M.R.; Muñiz, S.; Val, J.; Navarro, E. Effects of 18 pharmaceuticals on the physiological diversity of edaphic microorganisms. Sci. Total Environ. 2017, 595, 441–450. [Google Scholar] [CrossRef] [PubMed]
- Pohland, B.; Owen, B. TAS technical bulletin. Biolog 2009, 1, 1–3. [Google Scholar]
- Garland, J.L.; Mills, A.L. Classification and characterization of heterotrophic microbial communities on the basis of patterns of community-level sole-carbon-source utilization. Appl. Environ. Microbiol. 1991, 57, 2351–2359. [Google Scholar] [CrossRef]
- Pino-Otin, M.R.; Langa, E.; Val, J.; Mainar, A.M.; Ballestero, D. Impact of citronellol on river and soil environments using non-target model organisms and natural populations. J. Environ. Manag. 2021, 287, 112303. [Google Scholar] [CrossRef]
- Pino-Otín, M.R.; Ballestero, D.; Navarro, E.; González-Coloma, A.; Val, J.; Mainar, A.M. Ecotoxicity of a novel biopesticide from Artemisia absinthium on non-target aquatic organisms. Chemosphere 2019, 216, 131–146. [Google Scholar] [CrossRef] [PubMed]
- Lu, N.; Lu, Y.; Liu, F.; Zhao, K.; Yuan, X.; Zhao, Y.; Li, Y.; Qin, H.; Zhu, J. H3PW12O40/TiO2 catalyst-induced photodegradation of bisphenol A (BPA): Kinetics, toxicity and degradation pathways. Chemosphere 2013, 91, 1266–1272. [Google Scholar] [CrossRef] [PubMed]
- Tissot, A.; Boule, P.; Lemaire, J.; Lambert, S.; Palla, J.C. Photochemistry and Environment 10: Evaluation of the Toxicity of Phototransformation Products of Hydroquinone and Chlorophenols in Aqueous-Media. Chemosphere 1985, 14, 1221–1230. [Google Scholar] [CrossRef]
- Crisinel, A.; Delaunay, L.; Rossel, D.; Tarradellas, J.; Meyer, H.; Saiah, H.; Vogel, P.; Delisle, C.; Blaise, C. Cyst-Based Ecotoxicological Tests Using Anostracans—Comparison of 2 Species of Streptocephalus. Environ. Toxicol. Water Qual. 1994, 9, 317–326. [Google Scholar] [CrossRef]
- Kuznetsova, T.V.; Sladkova, G.V.; Kholodkevich, S.V. Evaluation of functional state of crayfish Pontastacus leptodactylus in normal and toxic environment by characteristics of their cardiac activity and hemolymph biochemical parameters. J. Evol. Biochem. Physiol. 2010, 46, 241–250. [Google Scholar] [CrossRef]
- Prosser, L. Temperature. In Sravnitel ‘naya fiziologiya zhivotnykh (Comparative Animal Physiology); Wiley: Hoboken, NJ, USA, 1977; Volume 2, pp. 84–209. ISBN 978-0-471-85767-9. [Google Scholar]
- IPCS. Hydroquinone. Environ. Health Criteria 1994, 157. Available online: https://www.inchem.org/documents/ehc/ehc/ehc157.htm (accessed on 17 January 2024).
- Sladkova, S.V.; Kholodkevich, S.V. Total protein in hemolymph of crawfish Pontastacus leptodactylus as a parameter of the functional state of animals and a biomarker of quality of habitat. J. Evol. Biochem. Physiol. 2011, 47, 160–167. [Google Scholar] [CrossRef]
- Mondrala, S.; Eastmond, D.A. Topoisomerase II inhibition by the bioactivated benzene metabolite hydroquinone involves multiple mechanisms. Chem. Biol. Interact. 2010, 184, 259–268. [Google Scholar] [CrossRef]
- El Najjar, N.H.; Touffet, A.; Deborde, M.; Journel, R.; Leitner, N.K.V. Kinetics of paracetamol oxidation by ozone and hydroxyl radicals, formation of transformation products and toxicity. Sep. Purif. Technol. 2014, 136, 137–143. [Google Scholar] [CrossRef]
- Muneer, M.; Singh, H.K.; Bahnemann, D. Semiconductor-mediated photocatalysed degradation of two selected priority organic pollutants, benzidine and 1,2-diphenylhydrazine, in aqueous suspension. Chemosphere 2002, 49, 193–203. [Google Scholar] [CrossRef] [PubMed]
- Turkay, O.; Barisci, S.; Ozturk, H.; Ozturk, B.; Seker, M.G. Toxicological Profile of 1,4-Benzoquinone and Its Degradation Byproducts during Electro-Fenton, Electrocoagulation, and Electrosynthesized Fe(VI) Oxidation. J. Environ. Eng. 2018, 144, 04018124. [Google Scholar] [CrossRef]
- Guo, Q.; Zhou, Y.; Pang, S.-Y.; Gao, Y.; Duan, J.; Li, J.; Jiang, J. Transformation and detoxification of sulfamethoxazole by permanganate (Mn(VII)) in the presence of phenolic humic constituents. Chem. Eng. J. 2021, 413, 127534. [Google Scholar] [CrossRef]
- El-Ghenymy, A.; Maria Rodriguez, R.; Brillas, E.; Oturan, N.; Oturan, M.A. Electro-Fenton degradation of the antibiotic sulfanilamide with Pt/carbon-felt and BDD/carbon-felt cells. Kinetics, reaction intermediates, and toxicity assessment. Environ. Sci. Pollut. Res. 2014, 21, 8368–8378. [Google Scholar] [CrossRef] [PubMed]
- Rosal, R.; Gonzalo, M.S.; Boltes, K.; Leton, P.; Vaquero, J.J.; Garcia-Calvo, E. Identification of intermediates and assessment of ecotoxicity in the oxidation products generated during the ozonation of clofibric acid. J. Hazard. Mater. 2009, 172, 1061–1068. [Google Scholar] [CrossRef] [PubMed]
- Calza, R.; Massolino, C.; Pelizzetti, E. Photo-induced transformation of hexaconazole and dimethomorph over TiO2 suspension. J. Photochem. Photobiol. A-Chem. 2008, 200, 356–363. [Google Scholar] [CrossRef]
- Santos, A.; Yustos, P.; Quintanilla, A.; Garcia-Ochoa, F.; Casas, J.A.; Rodriguez, J.J. Evolution of toxicity upon wet catalytic oxidation of phenol. Environ. Sci. Technol. 2004, 38, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Jeyanthi, V.; Anbu, P.; Vairamani, M.; Velusamy, P. Isolation of hydroquinone (benzene-1,4-diol) metabolite from halotolerant Bacillus methylotrophicus MHC10 and its inhibitory activity towards bacterial pathogens. Bioprocess Biosyst. Eng. 2016, 39, 429–439. [Google Scholar] [CrossRef]
- Jurica, K.; Gobin, I.; Kremer, D.; Cepo, D.V.; Grubesic, R.J.; Karaconji, I.B.; Kosalec, I. Arbutin and its metabolite hydroquinone as the main factors in the antimicrobial effect of strawberry tree (Arbutus unedo L.) leaves. J. Herb. Med. 2017, 8, 17–23. [Google Scholar] [CrossRef]
- Bikowska, B.Z.; Franiczek, R.; Sowa, A.; Polukord, G.; Krzyzanowska, B.; Sroka, Z. Antimicrobial and Antiradical Activity of Extracts Obtained from Leaves of Five Species of the Genus Bergenia: Identification of Antimicrobial Compounds. Microb. Drug Resist. 2017, 23, 771–780. [Google Scholar] [CrossRef] [PubMed]
- Sathiyamoorthi, E.; Faleye, O.S.; Lee, J.-H.; Lee, J. Hydroquinone derivatives attenuate biofilm formation and virulence factor production in Vibrio spp. Int. J. Food Microbiol. 2023, 384, 127534. [Google Scholar] [CrossRef] [PubMed]
- Mol, V.P.L.; Abdulaziz, A.; Sneha, K.G.; Praveen, P.J.; Raveendran, T.V.; Parameswaran, P.S. Inhibition of pathogenic Vibrio harveyi using calamenene, derived from the Indian gorgonian Subergorgia reticulata, and its synthetic analog. 3 Biotech 2020, 10, 248. [Google Scholar] [CrossRef]
- Genuario, D.B.; Vaz, M.; de Vielo, I.S. Phylogenetic insights into the diversity of homocytous cyanobacteria from Amazonian rivers. Mol. Phylogenetics Evol. 2017, 116, 120–135. [Google Scholar] [CrossRef] [PubMed]
- Sun, F.L.; Wang, Y.S.; Wu, M.L.; Sun, C.C. Cyanobacterial community diversity in the sediments of the Pearl River Estuary in China. Sci. Mar. 2017, 81, 477–485. [Google Scholar] [CrossRef]
- McGregor, G.B.; Fabbro, L.D.; Lobegeiger, J.S. Freshwater planktic Chroococcales (Cyanoprokaryota) from North-Eastern Australia: A morphological evaluation. Nova Hedwig. 2007, 84, 299–331. [Google Scholar] [CrossRef]
- Battistuzzi, F.U.; Hedges, S.B. A Major Clade of Prokaryotes with Ancient Adaptations to Life on Land. Mol. Biol. Evol. 2009, 26, 335–343. [Google Scholar] [CrossRef]
- Madigan, T.M.; Martinko, J.M.; Bender, K.S.; Buckley, D.H.; Stahl, D.A.; Aiyer, J.; Martinko, J.M. Brock Biology of Microorganisms, Global Edition, 14th ed.; Pearson Education: Madrid, Spain, 2015. [Google Scholar]
- Xia, N.; Xia, X.H.; Zhu, B.T.; Zheng, S.K.; Zhuang, J. Bacterial diversity and community structure in the sediment of the middle and lower reaches of the Yellow River, the largest turbid river in the world. Aquat. Microb. Ecol. 2013, 71, 43–55. [Google Scholar] [CrossRef]
- Narciso-da-Rocha, C.; Manaia, C.M. Multidrug resistance phenotypes are widespread over different bacterial taxonomic groups thriving in surface water. Sci. Total Environ. 2016, 563, 1–9. [Google Scholar] [CrossRef]
- Zhang, S.; Sun, W.; Xu, L.; Zheng, X.; Chu, X.; Tian, J.; Wu, N.; Fan, Y. Identification of the para-nitrophenol catabolic pathway, and characterization of three enzymes involved in the hydroquinone pathway, in pseudomonas sp. 1-7. BMC Microbiol. 2012, 12, 27. [Google Scholar] [CrossRef]
- Spain, J.C.; Gibson, D.T. Pathway for Biodegradation of Para-Nitrophenol in a Moraxella sp. Appl. Environ. Microbiol. 1991, 57, 812–819. [Google Scholar] [CrossRef]
- Liu, S.; Wang, P.; Wang, C.; Chen, J.; Wang, X.; Hu, B.; Yuan, Q. Ecological insights into the disturbances in bacterioplankton communities due to emerging organic pollutants from different anthropogenic activities along an urban river. Sci. Total Environ. 2021, 796, 148973. [Google Scholar] [CrossRef]
- Zhang, M.; Sun, Q.; Chen, P.; Wei, X.; Wang, B. How microorganisms tell the truth of potentially toxic elements pollution in environment. J. Hazard. Mater. 2022, 431, 128456. [Google Scholar] [CrossRef]
- Wang, W.; Weng, Y.; Luo, T.; Wang, Q.; Yang, G.; Jin, Y. Antimicrobial and the Resistances in the Environment: Ecological and Health Risks, Influencing Factors, and Mitigation Strategies. Toxics 2023, 11, 185. [Google Scholar] [CrossRef]
- Schoffelen, N.J.; Mohr, W.; Ferdelman, T.G.; Duerschlag, J.; Littmann, S.; Ploug, H.; Kuypers, M.M.M. Phosphate availability affects fixed nitrogen transfer from diazotrophs to their epibionts. ISME J. 2019, 13, 2701–2713. [Google Scholar] [CrossRef]
- Wang, Z.; Han, S.; Cai, M.; Du, P.; Zhang, Z.; Li, X. Environmental behavior of methamphetamine and ketamine in aquatic ecosystem: Degradation, bioaccumulation, distribution, and associated shift in toxicity and bacterial community. Water Res. 2020, 174, 115585. [Google Scholar] [CrossRef]
- Zhang, C.; Li, J.; Cheng, F.; Liu, Y. Enhanced phenol removal in an innovative lignite activated coke-assisted biological process. Bioresour. Technol. 2018, 260, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Chen, X.; Bao, L.; Bao, Z.; He, Y.; Zhang, Y.; Li, J. Correlation between microbial diversity and toxicity of sludge treating synthetic wastewater containing 4-chlorophenol in sequencing batch reactors. Chemosphere 2016, 153, 138–145. [Google Scholar] [CrossRef] [PubMed]
- Ansola, G.; Arroyo, P.; Saenz de Miera, L.E. Characterisation of the soil bacterial community structure and composition of natural and constructed wetlands. Sci. Total Environ. 2014, 473, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Rios-Miguel, A.B.; Smith, G.J.; Cremers, G.; van Alen, T.; Jetten, M.S.M.; Camp, H.J.M.O.d.; Welte, C.U. Microbial paracetamol degradation involves a high diversity of novel amidase enzyme candidates. Water Res. X 2022, 16, 100152. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Oh, S. Activated sludge-degrading analgesic drug acetaminophen: Acclimation, microbial community dynamics, degradation characteristics, and bioaugmentation potential. Water Res. 2020, 182, 115957. [Google Scholar] [CrossRef] [PubMed]
- Traversi, D.; Villa, S.; Lorenzi, E.; Degan, R.; Gilli, G. Application of a real-time qPCR method to measure the methanogen concentration during anaerobic digestion as an indicator of biogas production capacity. J. Environ. Manag. 2012, 111, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Villegas, L.G.C.; Mashhadi, N.; Chen, M.; Mukherjee, D.; Taylor, K.E.; Biswas, N. A Short Review of Techniques for Phenol Removal from Wastewater. Curr. Pollut. Rep. 2016, 2, 157–167. [Google Scholar] [CrossRef]
- Commission Implementing Decision (EU). 2018/840 of 5 June 2018 establishing a watch list of substances for Union-wide monitoring in the field of water policy pursuant to Directive 2008/105/EC of the European Parliament and of the Council and repealing Commission Implementing Decision (EU) 2015/495 (notified under document C(2018) 3362). Official Journal of the European Union. L 141/9. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A32018D0840 (accessed on 17 January 2024).
- Westman, C.A. The Effect Hydroquinone on Mitosis in Allium cepa; Fordham University: New York, NY, USA, 1949. [Google Scholar]
- Shettel, N.L.; Balke, N.E. Plant-Growth Response to Several Allelopathic Chemicals. Weed Sci. 1983, 31, 293–298. [Google Scholar] [CrossRef]
- Keller, C.P.; Barkosky, R.R.; Seil, J.E.; Mazurek, S.A.; Grundstad, M.L. The electrical response of Phaseolus vulgaris roots to abrupt exposure to hydroquinone. Plant Signal. Behav. 2008, 3, 633–640. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bouknana, D.; Jodeh, S.; Sbaa, M.; Hammouti, B.; Arabi, M.; Darmous, A.; Slamini, M.; Haboubi, K. A phytotoxic impact of phenolic compounds in olive oil mill wastewater on fenugreek “Trigonella foenum-graecum”. Environ. Monit. Assess. 2019, 191, 405. [Google Scholar] [CrossRef] [PubMed]
- Pino-Otín, M.R.; Lorca, G.; Val, J.; Ferrando, N.; Ballestero, D.; Langa, E. Ecotoxicological Study of Tannic Acid on Soil and Water Non-Target Indicators and Its Impact on Fluvial and Edaphic Communities. Plants 2023, 12, 4041. [Google Scholar] [CrossRef]
- Wang, H.; Cheng, Z.; Djouonkep, L.D.W.; Wang, L.; Cai, S.; Gauthier, M. Synthesis and properties of biodegradable aliphatic-aromatic polyesters derived from 4-hydroxybenzaldehyde. J. Appl. Polym. Sci. 2023, 140, e54063. [Google Scholar] [CrossRef]
- Osman, A.M.; Den Besten, P.J.; van Noort, P.C.M. Menadione enhances oxyradical formation in earthworm extracts: Vulnerability of earthworms to quinone toxicity. Aquat. Toxicol. 2003, 65, 101–109. [Google Scholar] [CrossRef]
- Suthar, S.; Singh, S.; Dhawan, S. Earthworms as bioindicator of metals (Zn, Fe, Mn, Cu, Pb and Cd) in soils: Is metal bioaccumulation affected by their ecological category? Ecol. Eng. 2008, 32, 99–107. [Google Scholar] [CrossRef]
- Saxena, P.N.; Gupta, S.K.; Murthy, R.C. Comparative toxicity of carbaryl, carbofuran, cypermethrin and fenvalerate in Metaphire posthuman and Eisenia fetida—A possible mechanism. Ecotoxicol. Environ. Saf. 2014, 100, 218–225. [Google Scholar] [CrossRef]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Waomar, M. Biological effects of essential oils—A review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef]
- Ahmed, N.; Al-Mutairi, K.A. Earthworms Effect on Microbial Population and Soil Fertility as Well as Their Interaction with Agriculture Practices. Sustainability 2022, 14, 7803. [Google Scholar] [CrossRef]
- Medina-Sauza, R.M.; Álvarez-Jiménez, M.; Delhal, A.; Reverchon, F.; Blouin, M.; Guerrero-Analco, J.A.; Cerdán, C.R.; Guevara, R.; Villain, L.; Barois, I. Earthworms Building Up Soil Microbiota, a Review. Front. Environ. Sci. 2019, 7, 81. [Google Scholar] [CrossRef]
- Janssen, P.H. Identifying the dominant soil bacterial taxa in libraries of 16S rRNA and 16S rRNA genes. Appl. Environ. Microbiol. 2006, 72, 1719–1728. [Google Scholar] [CrossRef]
- Spain, A.M.; Krumholz, L.R.; Elshahed, M.S. Abundance, composition, diversity and novelty of soil Proteobacteria. Isme J. 2009, 3, 992–1000. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Xu, Z.H. Assessing bacterial diversity in soil. J. Soils Sediments 2008, 8, 379–388. [Google Scholar] [CrossRef]
- Hugenholtz, P.; Goebel, B.M.; Pace, N.R. Impact of culture-independent studies on the emerging phylogenetic view of bacterial diversity. J. Bacteriol. 1998, 180, 6793. [Google Scholar] [CrossRef]
- Wang, Y.J.; Liu, L.; Yang, J.F.; Duan, Y.M.; Luo, Y.; Taherzadeh, M.J.; Li, Y.F.; Li, H.K.; Awasthi, M.K.; Zhao, Z.Y. The diversity of microbial community and function varied in response to different agricultural residues composting. Sci. Total Environ. 2020, 715, 136983. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.Y.; Fan, C.; Wang, Y.X.; Xia, Y.S.; Xiao, W.; Cui, X.L. Salt-tolerant and plant-growth-promoting bacteria isolated from high-yield paddy soil. Can. J. Microbiol. 2018, 64, 968–978. [Google Scholar] [CrossRef] [PubMed]
- Blain, N.P.; Helgason, B.L.; Germida, J.J. Endophytic root bacteria associated with the natural vegetation growing at the hydrocarbon-contaminated Bitumount Provincial Historic site. Can. J. Microbiol. 2017, 63, 502–515. [Google Scholar] [CrossRef]
- Zhang, W.; Chen, L.; Zhang, R.; Lin, K. High throughput sequencing analysis of the joint effects of BDE209-Pb on soil bacterial community structure. J. Hazard. Mater. 2016, 301, 1–7. [Google Scholar] [CrossRef]
- Feng, G.; Xie, T.; Wang, X.; Bai, J.; Tang, L.; Zhao, H.; Wei, W.; Wang, M.; Zhao, Y. Metagenomic analysis of microbial community and function involved in cd-contaminated soil. BMC Microbiol. 2018, 18, 11. [Google Scholar] [CrossRef]
- Pino-Otin, M.R.; Gan, C.; Terrado, E.; Sanz, M.A.; Ballestero, D.; Langa, E. Antibiotic properties of Satureja montana L. hydrolate in bacteria and fungus of clinical interest and its impact in non-target environmental microorganisms. Sci. Rep. 2022, 12, 18460. [Google Scholar] [CrossRef]
- Chen, H.; Yao, J.; Wang, F.; Choi, M.M.F.; Bramanti, E.; Zaray, G. Study on the toxic effects of diphenol compounds on soil microbial activity by a combination of methods. J. Hazard. Mater. 2009, 167, 846–851. [Google Scholar] [CrossRef]
- Chien, S.H.; Prochnow, L.I.; Cantarella, H. Chapter 8 Recent Developments of Fertilizer Production and Use to Improve Nutrient Efficiency and Minimize Environmental Impacts. In Advances in Agronomy; Academic Press: Cambridge, MA, USA, 2009; Volume 102, pp. 267–322. [Google Scholar]
- Bremner, J.M.; Chai, H.S. Effects of Phosphoroamides on Ammonia Volatilization and Nitrite Accumulation in Soils Treated with Urea. Biol. Fertil. Soils 1989, 8, 227–230. [Google Scholar] [CrossRef]
- Zaman, M.; Nguyen, M.L.; Blennerhassett, J.D.; Quin, B.F. Reducing NH3, N2O and NO3−N losses from a pasture soil with urease or nitrification inhibitors and elemental S-amended nitrogenous fertilizers. Biol. Fertil. Soils 2008, 44, 693–705. [Google Scholar] [CrossRef]
- Luisa Castrejon-Godinez, M.; Tovar-Sanchez, E.; Ortiz-Hernandez, M.L.; Encarnacion-Guevara, S.; Gabriel Martinez-Batallar, A.; Hernandez-Ortiz, M.; Sanchez-Salinas, E.; Rodriguez, A.; Mussali-Galante, P. Proteomic analysis of Burkholderia zhejiangensis CEIB S4-3 during the methyl parathion degradation process. Pestic. Biochem. Physiol. 2022, 187, 105197. [Google Scholar] [CrossRef]
- Jia, Y.H. Diversity of utilizing substrates of strain Dyella sp. la-4 and its application in soi remediation. 2009; Dissertation/Thesis. PQDT:67377505. [Google Scholar]
- Flood, J.J.; Copley, S.D. Genome-Wide Analysis of Transcriptional Changes and Genes That Contribute to Fitness during Degradation of the Anthropogenic Pollutant Pentachlorophenol by Sphingobium chlorophenolicum. Msystems 2018, 3, e00275–e00318. [Google Scholar] [CrossRef] [PubMed]
- Jain, R.K.; Dreisbach, J.H.; Spain, J.C. Biodegradation of P-Nitrophenol via 1,2,4-Benzenetriol by an Arthrobacter sp. Appl. Environ. Microbiol. 1994, 60, 3030–3032. [Google Scholar] [CrossRef]
- Cejková, A.; Masák, J.; Jirku, V.; Vesely, M.; Pátek, M.; Nesvera, J. Potential of Rhodococcus erythropolis as a bioremediation organism. World J. Microbiol. Biotechnol. 2005, 21, 317–321. [Google Scholar] [CrossRef]
- EC. Directive 2008/105/EC of the European Parliament and of the Council of 16 December 2008 Amending Directives 2000/60/EC as regards priority substances in the field of water policy. 16 December 2008 L 348, 24.12.2008. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:02008L0105-20130913 (accessed on 17 January 2024).
- EC. Directive 2013/39/EU of the European Parliament and of the Council of 12 August 2013 amending Directives 2000/60/EC and 2008/105/EC as regards priority substances in the field of water policy. 12 August 2013L 226 1 24.8.2013. Available online: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2013:226:0001:0017:en:PDF (accessed on 17 January 2024).



| Hydroquinone Properties | |
|---|---|
| Molecular weight | 110.11 g/mol [38] |
| Water solubility | 73 g/L at 25 °C [38] |
| Melting point | 170–172 °C [38] |
| Boiling point | 287 °C [38] |
| Dipole moment | 1.4–2.4 D [38] |
| Density | 1341 kg/m [39] |
| Vapour pressure | 2.34 × 10−3 Pa at 25 °C [39] |
| pH stability | 4.0–7.0 [39] |
| Partition coefficient (log pow) | 0.59 [40] |
| pKa | pK1 = 9.9 pK2 = 11.6 [40] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valenzuela, A.; Ballestero, D.; Gan, C.; Lorca, G.; Langa, E.; Pino-Otín, M.R. Hydroquinone Ecotoxicity: Unveiling Risks in Soil and River Ecosystems with Insights into Microbial Resilience. Toxics 2024, 12, 115. https://doi.org/10.3390/toxics12020115
Valenzuela A, Ballestero D, Gan C, Lorca G, Langa E, Pino-Otín MR. Hydroquinone Ecotoxicity: Unveiling Risks in Soil and River Ecosystems with Insights into Microbial Resilience. Toxics. 2024; 12(2):115. https://doi.org/10.3390/toxics12020115
Chicago/Turabian StyleValenzuela, Antonio, Diego Ballestero, Cristina Gan, Guillermo Lorca, Elisa Langa, and María Rosa Pino-Otín. 2024. "Hydroquinone Ecotoxicity: Unveiling Risks in Soil and River Ecosystems with Insights into Microbial Resilience" Toxics 12, no. 2: 115. https://doi.org/10.3390/toxics12020115
APA StyleValenzuela, A., Ballestero, D., Gan, C., Lorca, G., Langa, E., & Pino-Otín, M. R. (2024). Hydroquinone Ecotoxicity: Unveiling Risks in Soil and River Ecosystems with Insights into Microbial Resilience. Toxics, 12(2), 115. https://doi.org/10.3390/toxics12020115






