Biochar Derived from Post-Adsorbent for Immobilizing Cu and Cd in Sediment: The Effect on Heavy Metal Species and the Microbial Community Composition
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of the Post-Adsorbent and BC
2.2. Sediment–BC Contact
2.3. Cu and Cd Contents in the Water and Sediment
2.4. Sequential Extraction Procedure and Toxicity Characteristic Leaching Procedure
2.5. Determination of Enzymes’ Activities and the Microbial Community
2.6. Statistical Analysis
3. Results and Discussion
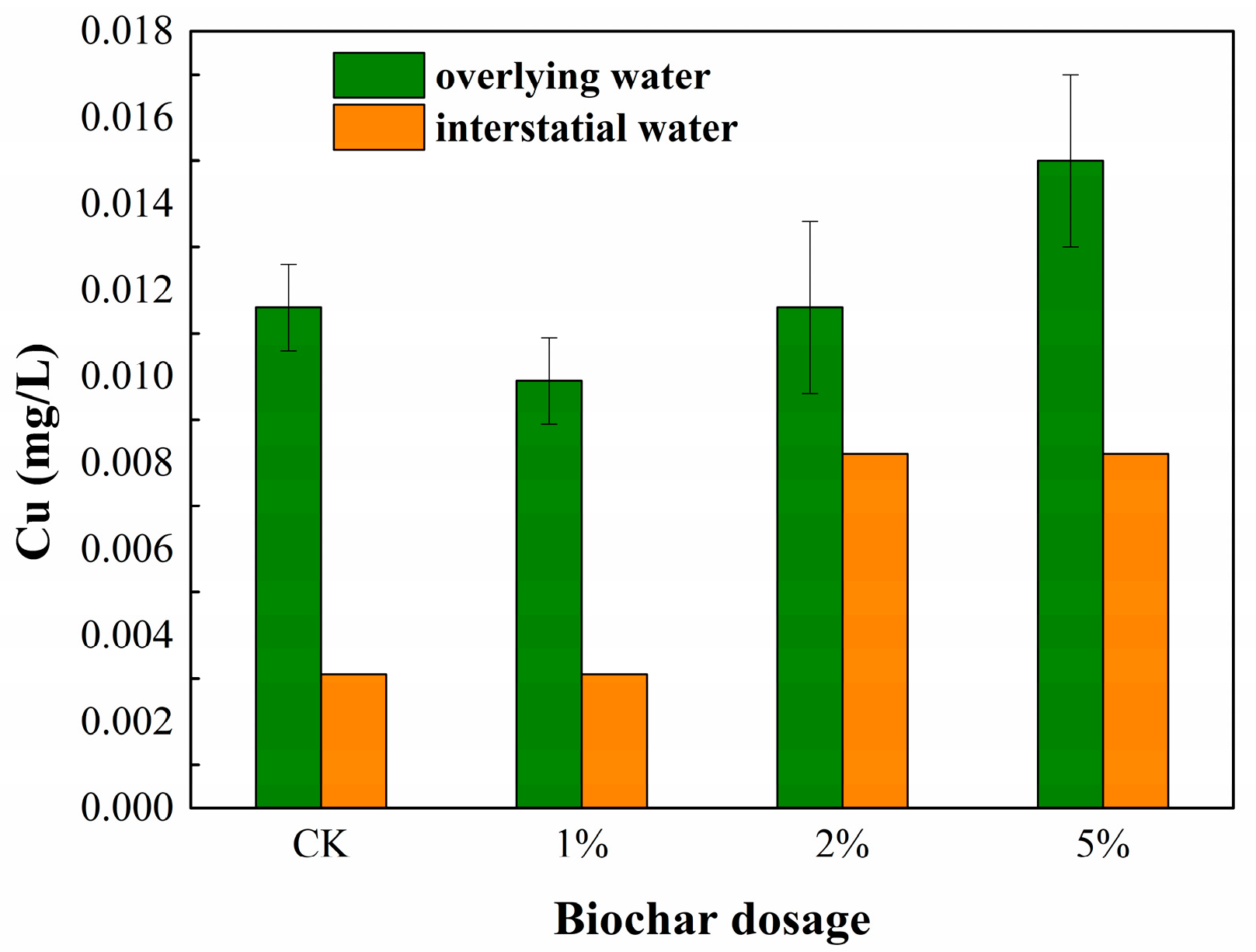
3.1. Cu and Cd Concentration in the Sediment Water Environment
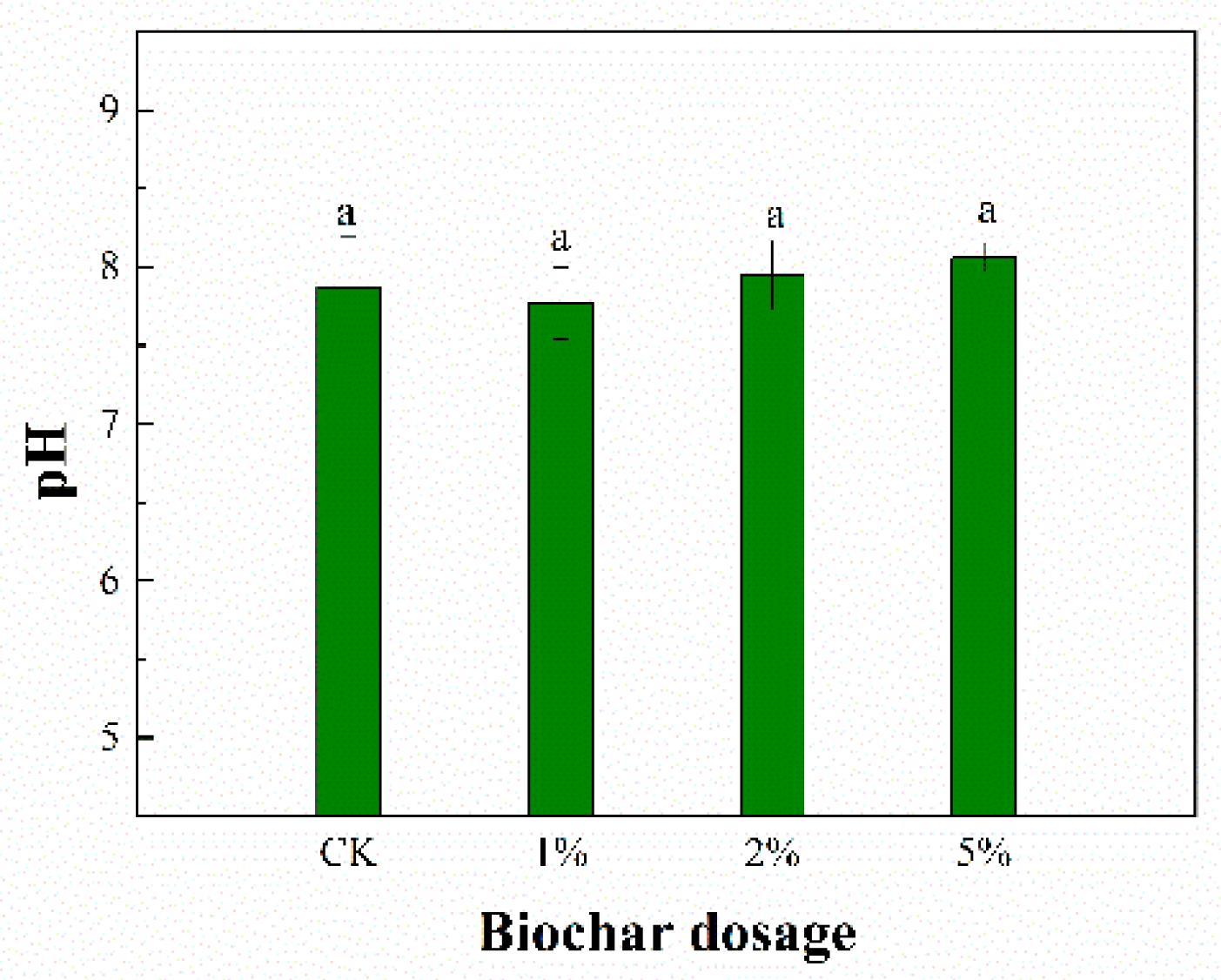
3.2. Sediment pH
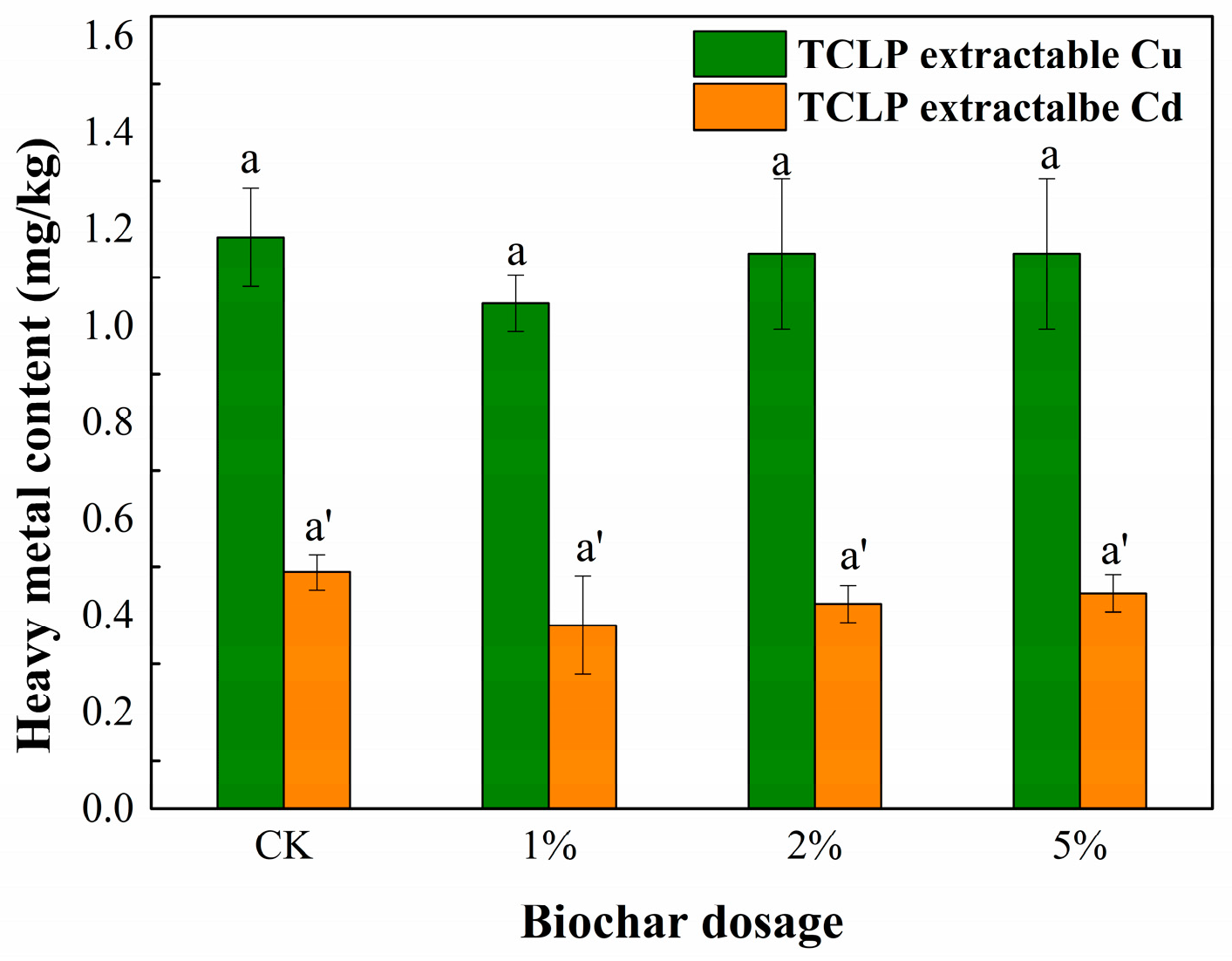
3.3. TCLP Leachability of Cu and Cd in Sediment
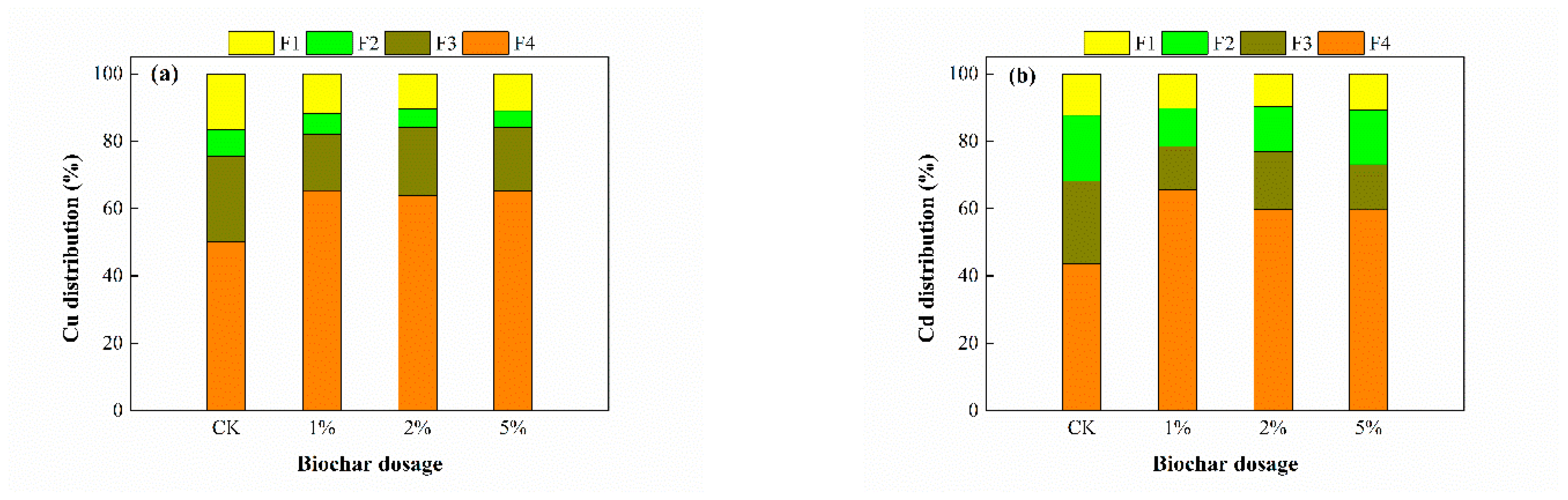
3.4. Fraction of Cu and Cd in Sediment
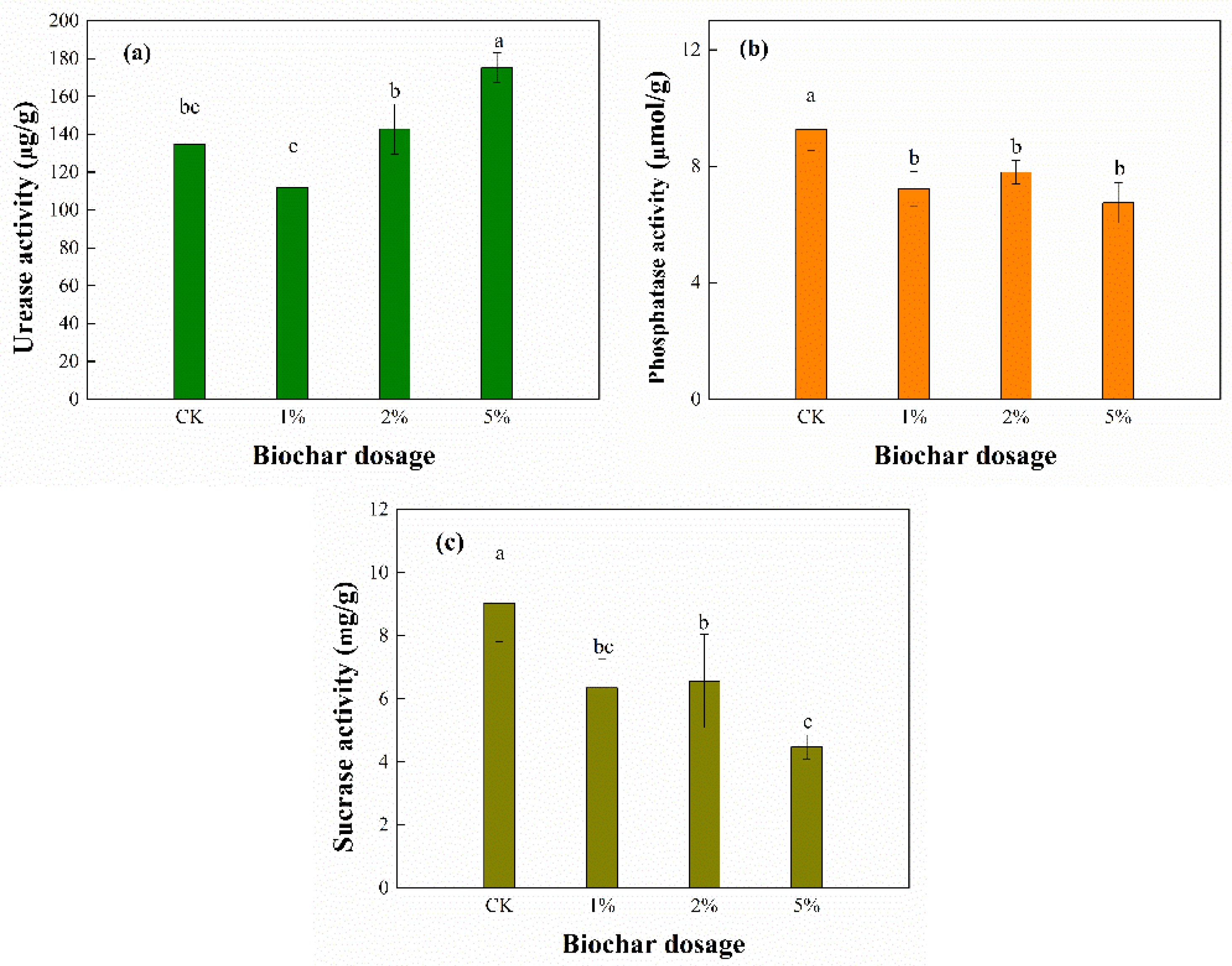
3.5. Enzyme Activities
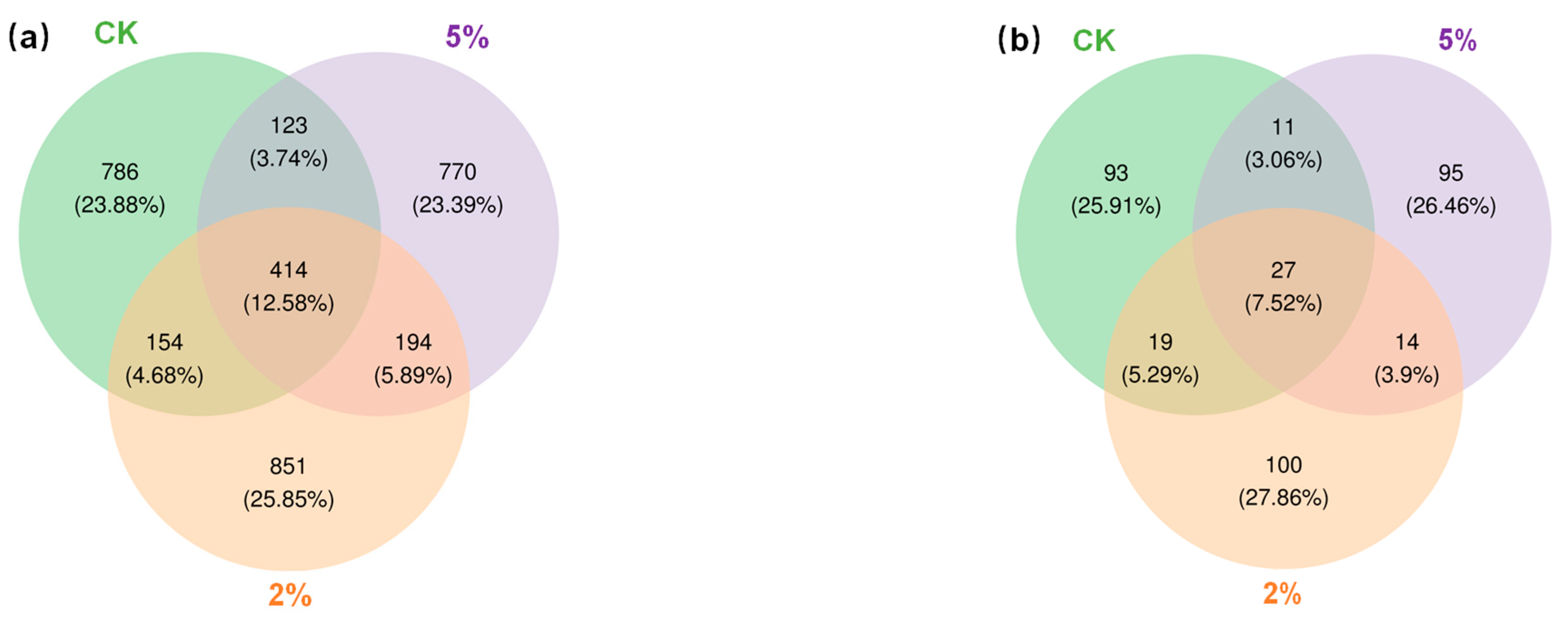
3.6. Bacterial and Fungal Community Structure
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Li, Y.; Gao, L.; Wang, Y.; Cheng, S.; Wu, G.; Yang, X.; Wan, S. Development of an acidized biochar-supported hydrated Fe(III) oxides for highly efficient cadmium and copper sequestration from water. Sci. Total Environ. 2021, 784, 147017. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Zhang, M.; Jin, Z.; Wang, G.; Li, R.; Zhang, X.; Liu, X.; Qu, J.; Wang, H. Characterization of biochars derived from various spent mushroom substrates and evaluation of their adsorption performance of Cu(II) ions from aqueous solution. Environ. Res. 2021, 196, 110323. [Google Scholar] [CrossRef] [PubMed]
- Zubair, M.; Adnan Ramzani, P.M.; Rasool, B.; Khan, M.A.; ur-Rahman, M.; Akhtar, I.; Turan, V.; Tauqeer, H.M.; Farhad, M.; Khan, S.A.; et al. Efficacy of chitosan-coated textile waste biochar applied to Cd-polluted soil for reducing Cd mobility in soil and its distribution in moringa (Moringa oleifera L.). J. Environ. Manag. 2021, 284, 112047. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Castañeda, M.E.; Medina, D.I. Use of surfactant-modified zeolites and clays for the removal of heavy metals from water. Water 2017, 9, 235. [Google Scholar] [CrossRef]
- Rajhans, A.; Gore, P.M.; Siddique, S.K.; Kandasubramanian, B. Ion-imprinted nanofibers of PVDF/1-butyl-3-methylimidazolium tetrafluoroborate for dynamic recovery of europium (III) ions from mimicked effluent. J. Environ. Chem. Eng. 2019, 7, 103068. [Google Scholar] [CrossRef]
- Inyang, M.I.; Gao, B.; Yao, Y.; Xue, Y.; Zimmerman, A.; Mosa, A.; Pullammanappallil, P.; Ok, Y.S.; Cao, X. A review of biochar as a low-cost adsorbent for aqueous heavy metal removal. Crit. Rev. Environ. Sci. Technol. 2016, 46, 406–433. [Google Scholar] [CrossRef]
- Gogda, A.A.; Patidar, R.; Rebary, B. An adsorption study of Sr2+ from saline sources by coconut shell charcoal. J. Dispers. Sci. Technol. 2017, 38, 1162–1167. [Google Scholar] [CrossRef]
- Popaliya, M.; Mishra, A. Modified zeolite as an adsorbent for dyes, drugs, and heavy metal removal: A review. Int. J. Environ. Sci. Technol. 2022. [Google Scholar] [CrossRef]
- Ko, D.; Lee, J.S.; Patel, H.A.; Jakobsen, M.H.; Hwang, Y.; Yavuz, C.T.; Hansen, H.C.B.; Andersen, H.R. Selective removal of heavy metal ions by disulfide linked polymer networks. J. Hazard. Mater. 2017, 332, 140–148. [Google Scholar] [CrossRef]
- Harikishore Kumar Reddy, D.; Vijayaraghavan, K.; Kim, J.A.; Yun, Y.-S. Valorisation of post-sorption materials: Opportunities, strategies, and challenges. Adv. Colloid Interface Sci. 2017, 242, 35–58. [Google Scholar] [CrossRef]
- Singh, S.; Kumar, V.; Datta, S.; Dhanjal, D.S.; Sharma, K.; Samuel, J.; Singh, J. Current advancement and future prospect of biosorbents for bioremediation. Sci. Total Environ. 2020, 709, 135895. [Google Scholar] [CrossRef]
- Sun, Q.; Lin, S.; Liu, G.; Xuan, Y.; Gao, L.; Li, P. Functionalized moso bamboo powder adsorbent for Cd(II) complexes with citric acid/tartrate acid: Characterization, adsorptive performance, and mechanism. Environ. Eng. Res. 2022, 27, 210321. [Google Scholar] [CrossRef]
- Dastyar, W.; Zhao, M.; Yuan, W.; Li, H.; Ting, Z.J.; Ghaedi, H.; Yuan, H.; Li, X.; Wang, W. Effective pretreatment of heavy metal-contaminated biomass using a low-cost ionic liquid (triethylammonium hydrogen sulfate): Optimization by response surface methodology–box behnken design. ACS Sustain. Chem. Eng. 2019, 7, 11571–11581. [Google Scholar] [CrossRef]
- Bashir, A.; Malik, L.A.; Ahad, S.; Manzoor, T.; Bhat, M.A.; Dar, G.N.; Pandith, A.H. Removal of heavy metal ions from aqueous system by ion-exchange and biosorption methods. Environ. Chem. Lett. 2019, 17, 729–754. [Google Scholar] [CrossRef]
- Božić, D.; Stanković, V.; Gorgievski, M.; Bogdanović, G.; Kovačević, R. Adsorption of heavy metal ions by sawdust of deciduous trees. J. Hazard. Mater. 2009, 171, 684–692. [Google Scholar] [CrossRef]
- Gómez-Aguilar, D.L.; Rodríguez-Miranda, J.P.; Salcedo-Parra, O.J. Fruit peels as a sustainable waste for the biosorption of heavy metals in wastewater: A review. Molecules 2022, 27, 2124. [Google Scholar] [CrossRef]
- de Sousa, D.A.; de Oliveira, E.; da Costa Nogueira, M.; Espósito, B.P. Development of a heavy metal sorption system through the PS functionalization of coconut (Cocos nucifera) fibers. Bioresour. Technol. 2010, 101, 138–143. [Google Scholar] [CrossRef]
- Elbasiouny, H.; Darwesh, M.; Elbeltagy, H.; Abo-alhamd, F.G.; Amer, A.A.; Elsegaiy, M.A.; Khattab, I.A.; Elsharawy, E.A.; Ebehiry, F.; El-Ramady, H.; et al. Ecofriendly remediation technologies for wastewater contaminated with heavy metals with special focus on using water hyacinth and black tea wastes: A review. Environ. Monit. Assess. 2021, 193, 449. [Google Scholar] [CrossRef]
- Nighojkar, A.; Sangal, V.K.; Dixit, F.; Kandasubramanian, B. Sustainable conversion of saturated adsorbents (SAs) from wastewater into value-added products: Future prospects and challenges with toxic per- and poly-fluoroalkyl substances (PFAS). Environ. Sci. Pollut. Res. 2022, 29, 78207–78227. [Google Scholar] [CrossRef]
- Chen, F.-F.; Liang, Y.; Chen, L.; Liang, X.; Feng, Y.-N.; Wu, J.; Zhu, Y.-J.; Yu, Y. Upcycling of heavy metal adsorbents into sulfide semiconductors for photocatalytic CO2 reduction. Appl. Surf. Sci. 2021, 558, 149647. [Google Scholar] [CrossRef]
- Tuhy, Ł.; Samoraj, M.; Witkowska, Z.; Rusek, P.; Chojnacka, K. Conversion of spent mushroom substrate into micronutrient fertilizer via biosorption in a pilot plant. Ecol. Eng. 2015, 84, 370–374. [Google Scholar] [CrossRef]
- Yao, Y.; Gao, B.; Wu, F.; Zhang, C.; Yang, L. Engineered biochar from biofuel residue: Characterization and its silver removal potential. ACS Appl. Mater. Interfaces 2015, 7, 10634–10640. [Google Scholar] [CrossRef]
- Chen, Q.; Dong, J.; Yi, Q.; Liu, X.; Zhang, J.; Zeng, Z. Proper mode of using rice straw biochar to treat Cd-contaminated irrigation water in mining regions based on a multiyear in situ experiment. ACS Sustain. Chem. Eng. 2019, 7, 9928–9936. [Google Scholar] [CrossRef]
- Liu, S.-J.; Liu, Y.-G.; Tan, X.-F.; Zeng, G.-M.; Zhou, Y.-H.; Liu, S.-B.; Yin, Z.-H.; Jiang, L.-H.; Li, M.-F.; Wen, J. The effect of several activated biochars on Cd immobilization and microbial community composition during in-situ remediation of heavy metal contaminated sediment. Chemosphere 2018, 208, 655–664. [Google Scholar] [CrossRef]
- Wang, T.; Sun, H.; Jiang, C.; Mao, H.; Zhang, Y. Immobilization of Cd in soil and changes of soil microbial community by bioaugmentation of UV-mutated bacillus subtilis 38 assisted by biostimulation. Eur. J. Soil Biol. 2014, 65, 62–69. [Google Scholar] [CrossRef]
- Yang, Y.; Ye, S.; Zhang, C.; Zeng, G.; Tan, X.; Song, B.; Zhang, P.; Yang, H.; Li, M.; Chen, Q. Application of biochar for the remediation of polluted sediments. J. Hazard. Mater. 2021, 404, 124052. [Google Scholar] [CrossRef]
- Zhang, A.; Li, X.; Xing, J.; Xu, G. Adsorption of potentially toxic elements in water by modified biochar: A review. J. Environ. Chem. Eng. 2020, 8, 104196. [Google Scholar] [CrossRef]
- Tan, X.-F.; Liu, S.-B.; Liu, Y.-G.; Gu, Y.-L.; Zeng, G.-M.; Hu, X.-J.; Wang, X.; Liu, S.-H.; Jiang, L.-H. Biochar as potential sustainable precursors for activated carbon production: Multiple applications in environmental protection and energy storage. Bioresour. Technol. 2017, 227, 359–372. [Google Scholar] [CrossRef]
- Han, B.; Song, L.; Li, H.; Song, H. Naked oats biochar-supported nanoscale zero-valent iron composite: Effects on Cd immobilization and enzyme activities in Ulansuhai River sediments of China. J. Soils Sediments 2019, 19, 2650–2662. [Google Scholar] [CrossRef]
- Yue, W.; Hong-Sheng, W.; Chao-Sheng, T.; Kai, G.; Bin, S. Remediation of heavy-metal-contaminated soils by biochar: A review. Environ. Geotech. 2022, 9, 135–148. [Google Scholar] [CrossRef]
- Huang, D.; Liu, L.; Zeng, G.; Xu, P.; Huang, C.; Deng, L.; Wang, R.; Wan, J. The effects of rice straw biochar on indigenous microbial community and enzymes activity in heavy metal-contaminated sediment. Chemosphere 2017, 174, 545–553. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.; Xue, W.; Zeng, G.; Wan, J.; Chen, G.; Huang, C.; Zhang, C.; Cheng, M.; Xu, P. Immobilization of Cd in river sediments by sodium alginate modified nanoscale zero-valent iron: Impact on enzyme activities and microbial community diversity. Water Res. 2016, 106, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Que, W.; Zhou, Y.-H.; Liu, Y.-G.; Wen, J.; Tan, X.-F.; Liu, S.-J.; Jiang, L.-H. Appraising the effect of in-situ remediation of heavy metal contaminated sediment by biochar and activated carbon on Cu immobilization and microbial community. Ecol. Eng. 2019, 127, 519–526. [Google Scholar] [CrossRef]
- Arunachellan, I.C.; Sypu, V.S.; Kera, N.H.; Pillay, K.; Maity, A. Flower-like structures of carbonaceous nanomaterials obtained from biomass for the treatment of copper ion-containing water and their re-use in organic transformations. J. Environ. Chem. Eng. 2021, 9, 105242. [Google Scholar] [CrossRef]
- Yuan, J.-H.; Xu, R.-K.; Zhang, H. The forms of alkalis in the biochar produced from crop residues at different temperatures. Bioresour. Technol. 2011, 102, 3488–3497. [Google Scholar] [CrossRef] [PubMed]
- Bailey, V.L.; Fansler, S.J.; Smith, J.L.; Bolton, H. Reconciling apparent variability in effects of biochar amendment on soil enzyme activities by assay optimization. Soil Biol. Biochem. 2011, 43, 296–301. [Google Scholar] [CrossRef]
- Dempster, D.N.; Gleeson, D.B.; Solaiman, Z.M.; Jones, D.L.; Murphy, D.V. Decreased soil microbial biomass and nitrogen mineralisation with Eucalyptus biochar addition to a coarse textured soil. Plant Soil 2012, 354, 311–324. [Google Scholar] [CrossRef]
- Elzobair, K.A.; Stromberger, M.E.; Ippolito, J.A. Stabilizing effect of biochar on soil extracellular enzymes after a denaturing stress. Chemosphere 2016, 142, 114–119. [Google Scholar] [CrossRef]
- Masiello, C.A.; Chen, Y.; Gao, X.; Liu, S.; Cheng, H.Y.; Bennett, M.R.; Rudgers, J.A.; Wagner, D.S.; Zygourakis, K.; Silberg, J.J. Biochar and microbial signaling: Production conditions determine effects on microbial communication. Environ. Sci. Technol. 2013, 47, 11496–11503. [Google Scholar] [CrossRef]
- Chintala, R.; Schumacher, T.E.; Kumar, S.; Malo, D.D.; Rice, J.A.; Bleakley, B.; Chilom, G.; Clay, D.E.; Julson, J.L.; Papiernik, S.K.; et al. Molecular characterization of biochars and their influence on microbiological properties of soil. J. Hazard. Mater. 2014, 279, 244–256. [Google Scholar] [CrossRef]
- Zhu, X.; Chen, B.; Zhu, L.; Xing, B. Effects and mechanisms of biochar-microbe interactions in soil improvement and pollution remediation: A review. Environ. Pollut. 2017, 227, 98–115. [Google Scholar] [CrossRef]
- Nielsen, S.; Minchin, T.; Kimber, S.; van Zwieten, L.; Gilbert, J.; Munroe, P.; Joseph, S.; Thomas, T. Comparative analysis of the microbial communities in agricultural soil amended with enhanced biochars or traditional fertilisers. Agric. Ecosyst. Environ. 2014, 191, 73–82. [Google Scholar] [CrossRef]

| Properties | Value |
|---|---|
| pH | 8.3 ± 0.1 |
| Yield (%) | 20.2 |
| Specific surface area (m2/g) | 0.01 |
| Elemental composition (%) | |
| C | 74.85 |
| H | 1.24 |
| N | 1.25 |
| Total Cu (mg/g) | 2.76 |
| pH | Cd (mg/kg) | Cu (mg/kg) | Organic Content (%) | Total Nitrogen (g/kg) | Total Phosphorus (g/kg) |
|---|---|---|---|---|---|
| 7.5 ± 0.1 | 3.98 ± 0.33 | 87.51 ± 0.5 | 5.1 ± 0.3 | 4.91 ± 0.13 | 0.39 ± 0.06 |
| Cu Fraction | 1% Treatment | 2% Treatment | 5% Treatment | |||
|---|---|---|---|---|---|---|
| Before | After | Before | After | Before | After | |
| Acid-soluble fraction (F1) | 19.86 | 11.68 | 16.92 | 10.50 | 12.41 | 10.96 |
| Reducible fraction (F2) | 9.51 | 6.41 | 7.78 | 5.47 | 5.12 | 4.97 |
| Oxidizable fraction (F3) | 26.78 | 16.55 | 23.02 | 20.25 | 17.26 | 18.77 |
| Residual fraction (F4) | 43.83 | 65.36 | 52.28 | 63.78 | 65.22 | 65.3 |
| Microbe | Treatment | OTU | Chao1 | Simpson | Shannon | Coverage |
|---|---|---|---|---|---|---|
| Bacteria | CK | 1477 | 1588.51 | 0.9610 | 7.6148 | 0.995 |
| 2% | 1613 | 1698.35 | 0.9814 | 8.3428 | 0.996 | |
| 5% | 1501 | 1605.29 | 0.9314 | 7.4963 | 0.995 | |
| Fungi | CK | 150 | 156.05 | 0.5210 | 2.5250 | 0.999 |
| 2% | 160 | 161.57 | 0.9391 | 5.1545 | 0.999 | |
| 5% | 147 | 147.07 | 0.8225 | 4.1710 | 0.999 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, Q.; Lin, S.; Liu, G.; Li, P. Biochar Derived from Post-Adsorbent for Immobilizing Cu and Cd in Sediment: The Effect on Heavy Metal Species and the Microbial Community Composition. Toxics 2023, 11, 666. https://doi.org/10.3390/toxics11080666
Sun Q, Lin S, Liu G, Li P. Biochar Derived from Post-Adsorbent for Immobilizing Cu and Cd in Sediment: The Effect on Heavy Metal Species and the Microbial Community Composition. Toxics. 2023; 11(8):666. https://doi.org/10.3390/toxics11080666
Chicago/Turabian StyleSun, Qinju, Shaohua Lin, Guohua Liu, and Pingping Li. 2023. "Biochar Derived from Post-Adsorbent for Immobilizing Cu and Cd in Sediment: The Effect on Heavy Metal Species and the Microbial Community Composition" Toxics 11, no. 8: 666. https://doi.org/10.3390/toxics11080666
APA StyleSun, Q., Lin, S., Liu, G., & Li, P. (2023). Biochar Derived from Post-Adsorbent for Immobilizing Cu and Cd in Sediment: The Effect on Heavy Metal Species and the Microbial Community Composition. Toxics, 11(8), 666. https://doi.org/10.3390/toxics11080666






