Independent and Combined Associations of Blood Manganese, Cadmium and Lead Exposures with the Systemic Immune-Inflammation Index in Adults
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Blood Mn, Cd and Pb Measurements
2.3. Peripheral Blood Cell Count Measurement
2.4. Covariates
2.5. Statistical Analysis
3. Results
3.1. Characteristics of the Study Population
3.2. Correlations between Continuous Variables and Blood Concentrations of Mn, Cd and Pb
3.3. Associations of SII with Mn, Cd and Pb
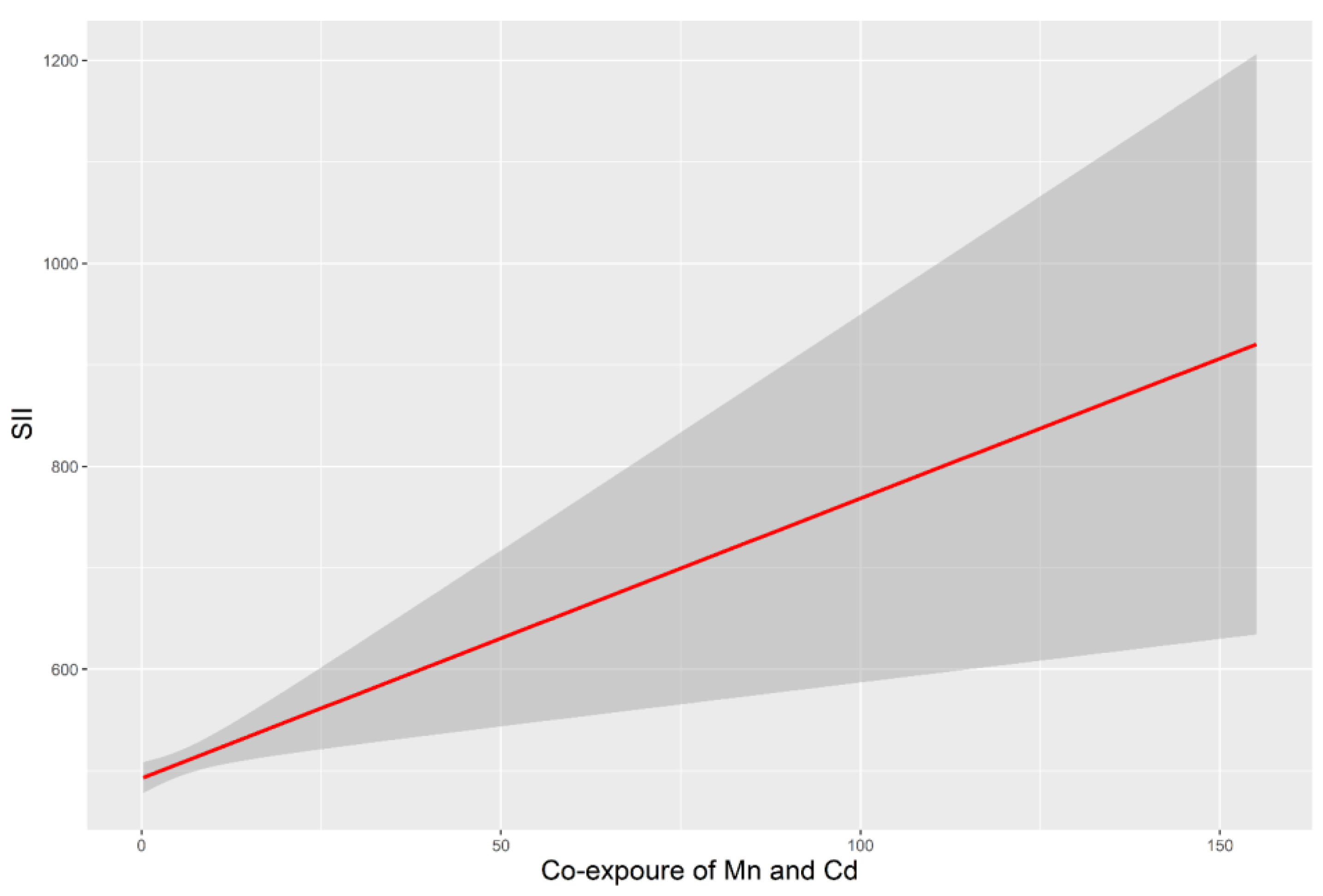
3.4. The Relationship of SII with Co-Exposure to Blood Mn and Cd
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Roth, J.A.; Salvi, R. Ototoxicity of Divalent Metals. Neurotox. Res. 2016, 30, 268–282. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimi, M.; Khalili, N.; Razi, S.; Keshavarz-Fathi, M.; Khalili, N.; Rezaei, N. Effects of lead and cadmium on the immune system and cancer progression. J. Environ. Health Sci. Eng. 2020, 18, 335–343. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Leston, J.; Roma-Torres, J.; Mayan, O.; Schroecksnadel, S.; Fuchs, D.; Moreira, A.O.; Pasaro, E.; Mendez, J.; Teixeira, J.P.; Laffon, B. Assessment of immunotoxicity parameters in individuals occupationally exposed to lead. J. Toxicol. Environ. Health A 2012, 75, 807–818. [Google Scholar] [CrossRef] [PubMed]
- Holaskova, I.; Elliott, M.; Hanson, M.L.; Schafer, R.; Barnett, J.B. Prenatal cadmium exposure produces persistent changes to thymus and spleen cell phenotypic repertoire as well as the acquired immune response. Toxicol. Appl. Pharmacol. 2012, 265, 181–189. [Google Scholar] [CrossRef]
- Zhang, H.; Yan, J.; Niu, J.; Wang, H.; Li, X. Association between lead and cadmium co-exposure and systemic immune inflammation in residents living near a mining and smelting area in NW China. Chemosphere 2022, 287 Pt 3, 132190. [Google Scholar] [CrossRef]
- Huo, X.; Dai, Y.; Yang, T.; Zhang, Y.; Li, M.; Xu, X. Decreased erythrocyte CD44 and CD58 expression link e-waste Pb toxicity to changes in erythrocyte immunity in preschool children. Sci. Total Environ. 2019, 664, 690–697. [Google Scholar] [CrossRef]
- Xu, H.; Mao, Y.; Xu, B.; Hu, Y. Low-level environmental lead and cadmium exposures and dyslipidemia in adults: Findings from the NHANES 2005–2016. J Trace Elem. Med. Biol. 2021, 63, 126651. [Google Scholar] [CrossRef]
- Milton, B.; Krewski, D.; Mattison, D.R.; Karyakina, N.A.; Ramoju, S.; Shilnikova, N.; Birkett, N.; Farrell, P.J.; McGough, D. Modeling U-shaped dose-response curves for manganese using categorical regression. Neurotoxicology 2017, 58, 217–225. [Google Scholar] [CrossRef]
- Wang, J.; Zhou, D.; Dai, Z.; Li, X. Association Between Systemic Immune-Inflammation Index and Diabetic Depression. Clin. Interv. Aging. 2021, 16, 97–105. [Google Scholar] [CrossRef]
- Wu, Q.; Mu, Q.; Xia, Z.; Min, J.; Wang, F. Manganese homeostasis at the host-pathogen interface and in the host immune system. Semin. Cell Dev. Biol. 2021, 115, 45–53. [Google Scholar] [CrossRef]
- Wang, X.; Gao, X.Q.; Wang, X.Y.; Fang, Y.Y.; Xu, L.; Zhao, K.F.; Huang, B.; Liu, B.L. Bioaccumulation of manganese and its effects on oxidative stress and immune response in juvenile groupers (Epinephelus moara female symbol x E. lanceolatus male symbol). Chemosphere 2022, 297, 134235. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Guan, Y.; Lv, M.; Zhang, R.; Guo, Z.; Wei, X.; Du, X.; Yang, J.; Li, T.; Wan, Y.; et al. Manganese Increases the Sensitivity of the cGAS-STING Pathway for Double-Stranded DNA and Is Required for the Host Defense against DNA Viruses. Immunity 2018, 48, 675–687.e7. [Google Scholar] [CrossRef] [PubMed]
- Do, J.W.; Saravanan, M.; Nam, S.E.; Lim, H.J.; Rhee, J.S. Waterborne manganese modulates immunity, biochemical, and antioxidant parameters in the blood of red seabream and black rockfish. Fish Shellfish. Immunol. 2019, 88, 546–555. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.F.; Li, Z.P.; Tie, F.; Liu, N.; Zhang, Z.W.; Xu, S.W. Effects of manganese-toxicity on immune-related organs of cocks. Chemosphere 2013, 90, 2085–2100. [Google Scholar] [CrossRef] [PubMed]
- Martin, K.V.; Edmondson, D.; Cecil, K.M.; Bezi, C.; Vance, M.L.; McBride, D.; Haynes, E.N. Manganese Exposure and Neurologic Outcomes in Adult Populations. Neurol. Clin. 2020, 38, 913–936. [Google Scholar] [CrossRef]
- Nakata, A.; Araki, S.; Park, S.H.; Park, J.T.; Kim, D.S.; Park, H.C. Yokoyama. K, Decreases in CD8+ T, naive (CD4+CD45RA+) T, and B (CD19+) lymphocytes by exposure to manganese fume. Ind. Health 2006, 44, 592–597. [Google Scholar] [CrossRef]
- Velasco, A.; Rodriguez-Revuelta, J.; Olie, E.; Abad, I.; Fernandez-Pelaez, A.; Cazals, A.; Guillaume, S.; de La Fuente-Tomas, L.; Jimenez-Trevino, L.; Gutierrez, L.; et al. Neutrophil-to-lymphocyte ratio: A potential new peripheral biomarker of suicidal behavior. Eur. Psychiatry 2020, 63, e14. [Google Scholar] [CrossRef]
- Sun, H.; Que, J.; Peng, Y.; Ye, H.; Xiang, H.; Han, Y.; Wang, J.; Ji, K. The neutrophil-lymphocyte ratio: A promising predictor of mortality in coronary care unit patients-A cohort study. Int. Immunopharmacol. 2019, 74, 105692. [Google Scholar] [CrossRef]
- Cananzi, F.C.M.; Minerva, E.M.; Sama, L.; Ruspi, L.; Sicoli, F.; Conti, L.; Fumagalli Romario, U.; Quagliuolo, V.L. Preoperative monocyte-to-lymphocyte ratio predicts recurrence in gastrointestinal stromal tumors. J. Surg. Oncol. 2019, 119, 12–20. [Google Scholar] [CrossRef]
- Gasparyan, A.Y.; Ayvazyan, L.; Mukanova, U.; Yessirkepov, M.; Kitas, G.D. The Platelet-to-Lymphocyte Ratio as an Inflammatory Marker in Rheumatic Diseases. Ann. Lab. Med. 2019, 39, 345–357. [Google Scholar] [CrossRef]
- Kumarasamy, C.; Tiwary, V.; Sunil, K.; Suresh, D.; Shetty, S.; Muthukaliannan, G.K.; Baxi, S.; Jayaraj, R. Prognostic Utility of Platelet-Lymphocyte Ratio, Neutrophil-Lymphocyte Ratio and Monocyte-Lymphocyte Ratio in Head and Neck Cancers: A Detailed PRISMA Compliant Systematic Review and Meta-Analysis. Cancers 2021, 13, 4166. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.; Zhu, X.; Lin, Z.; Zeng, M.; Shi, P.; Cao, Y.; Chen, F. Compare the Diagnostic and Prognostic Value of MLR, NLR and PLR in CRC Patients. Clin. Lab. 2021, 67, 78–89. [Google Scholar] [CrossRef]
- Mandaliya, H.; Jones, M.; Oldmeadow, C.; Nordman, I.I. Prognostic biomarkers in stage IV non-small cell lung cancer (NSCLC): Neutrophil to lymphocyte ratio (NLR), lymphocyte to monocyte ratio (LMR), platelet to lymphocyte ratio (PLR) and advanced lung cancer inflammation index (ALI). Transl. Lung Cancer Res. 2019, 8, 886–894. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.L.; Wu, C.H.; Hsu, P.F.; Chen, S.C.; Huang, S.S.; Chan, W.L.; Lin, S.J.; Chou, C.Y.; Chen, J.W.; Pan, J.P.; et al. Systemic immune-inflammation index (SII) predicted clinical outcome in patients with coronary artery disease. Eur. J. Clin. Investig. 2020, 50, e13230. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.H.; Zhai, E.T.; Yuan, Y.J.; Wu, K.M.; Xu, J.B.; Peng, J.J.; Chen, C.Q.; He, Y.L.; Cai, S.R. Systemic immune-inflammation index for predicting prognosis of colorectal cancer. World J. Gastroenterol. 2017, 23, 6261–6272. [Google Scholar] [CrossRef]
- Huang, H.; Liu, Q.; Zhu, L.; Zhang, Y.; Lu, X.; Wu, Y.; Liu, L. Prognostic Value of Preoperative Systemic Immune-Inflammation Index in Patients with Cervical Cancer. Sci. Rep. 2019, 9, 3284. [Google Scholar] [CrossRef] [PubMed]
- Bittoni, A.; Pecci, F.; Mentrasti, G.; Crocetti, S.; Lupi, A.; Lanese, A.; Pellei, C.; Ciotti, C.; Cantini, L.; Giampieri, R.; et al. Systemic immune-inflammation index: A prognostic tiebreaker among all in advanced pancreatic cancer. Ann. Transl. Med. 2021, 9, 251. [Google Scholar] [CrossRef]
- Hua, X.; Long, Z.Q.; Zhang, Y.L.; Wen, W.; Guo, L.; Xia, W.; Zhang, W.W.; Lin, H.X. Prognostic Value of Preoperative Systemic Immune-Inflammation Index in Breast Cancer: A Propensity Score-Matching Study. Front. Oncol. 2020, 10, 580. [Google Scholar] [CrossRef]
- Chen, X.; Zhu, G.; Wang, Z.; Zhou, H.; He, P.; Liu, Y.; Jin, T. The association between lead and cadmium co-exposure and renal dysfunction. Ecotoxicol. Environ. Saf. 2019, 173, 429–435. [Google Scholar] [CrossRef]
- Bartrem, C.; Tirima, S.; von Lindern, I.; von Braun, M.; Worrell, M.C.; Mohammad Anka, S.; Abdullahi, A.; Moller, G. Unknown risk: Co-exposure to lead and other heavy metals among children living in small-scale mining communities in Zamfara State, Nigeria. Int. J. Environ. Health Res. 2014, 24, 304–319. [Google Scholar] [CrossRef]
- Pan, Z.; Guo, Y.; Xiang, H.; Hui, Y.; Ju, H.; Xu, S.; Li, L. Effects of Lead, Mercury, and Cadmium Co-exposure on Children’s Pulmonary Function. Biol. Trace Elem. Res. 2020, 194, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Martin, K.V.; Sucharew, H.; Dietrich, K.N.; Parsons, P.J.; Palmer, C.D.; Wright, R.; Amarasiriwardena, C.; Smith, D.R.; Haynes, E.N. Co-exposure to manganese and lead and pediatric neurocognition in East Liverpool, Ohio. Environ. Res. 2021, 202, 111644. [Google Scholar] [CrossRef] [PubMed]
- Guan, R.; Wang, T.; Dong, X.; Du, K.; Li, J.; Zhao, F.; Xu, J.; Li, B.; Zheng, G.; Shen, X.; et al. Effects of co-exposure to lead and manganese on learning and memory deficits. J. Environ. Sci. 2022, 121, 65–76. [Google Scholar] [CrossRef] [PubMed]
- Oladipo, O.O.; Ayo, J.O.; Ambali, S.F.; Mohammed, B.; Aluwong, T. Dyslipdemia induced by chronic low dose co-exposure to lead, cadmium and manganese in rats: The role of oxidative stress. Environ. Toxicol. Pharmacol. 2017, 53, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Q.; Sun, W.; Qin, Y.; Xu, H. Association of Dietary α-Carotene and β-Carotene Intake with Low Cognitive Performance in Older Adults: A Cross-Sectional Study from the National Health and Nutrition Examination Survey. Nutrients 2023, 15, 239. [Google Scholar] [CrossRef]
- National Health and Nutrition Examination Survey. Cadmium, Lead, Manganese, Mercury, and Selenium Lab Procedure Manual. Available online: https://wwwn.cdc.gov/nchs/data/nhanes/2015-2016/labmethods/PBCD_I_met.pdf (accessed on 18 June 2023).
- Gao, W.; Tong, L.; Zhao, S.; Sun, M.; Fang, J.; Xu, Y.; Ma, Y.; Jin, L. Exposure to Cadmium, Lead, Mercury, and Arsenic and Uric Acid Levels: Results from NHANES 2007–2016. Biol. Trace Elem. Res. 2023, 201, 1659–1669. [Google Scholar] [CrossRef]
- Agency for Toxic Substances and Disease Registry. Case Studies in Environmental Medicine (CSEM) Lead Toxicity. 2010. Available online: https://www.atsdr.cdc.gov/csem/lead/docs/lead.pdf (accessed on 22 July 2023).
- Bulka, C.M.; Persky, V.W.; Daviglus, M.L.; Durazo-Arvizu, R.A.; Argos, M. Multiple metal exposures and metabolic syndrome: A cross-sectional analysis of the National Health and Nutrition Examination Survey 2011–2014. Environ. Res. 2019, 168, 397–405. [Google Scholar] [CrossRef]
- Xu, H.; Bo, Y. Associations between pyrethroid exposure and serum sex steroid hormones in adults: Findings from a nationally representative sample. Chemosphere 2022, 300, 134591. [Google Scholar] [CrossRef]
- Rapp, J.L.; Alpert, N.; Flores, R.M.; Taioli, E. Serum cotinine levels and nicotine addiction potential of e-cigarettes: An NHANES analysis. Carcinogenesis 2020, 41, 1454–1459. [Google Scholar] [CrossRef]
- Patel, M.A.; Mener, D.J.; Garcia-Esquinas, E.; Navas-Acien, A.; Agrawal, Y.; Lin, S.Y. Tobacco Smoke Exposure and Eustachian Tube Disorders in US Children and Adolescents. PLoS ONE 2016, 11, e0163926. [Google Scholar] [CrossRef]
- Menezes-Filho, J.A.; Carvalho, C.F.; Rodrigues, J.L.G.; Araújo, C.F.S.; Dos Santos, N.R.; Lima, C.S.; Bandeira, M.J.; Marques, B.L.d.S.; Anjos, A.L.S.; Bah, H.A.F.; et al. Environmental Co-Exposure to Lead and Manganese and Intellectual Deficit in School-Aged Children. Int. J. Environ. Res. Public Health 2018, 15, 2418. [Google Scholar] [CrossRef] [PubMed]
- Bo, Y.; Xu, H.; Zhang, H.; Zhang, J.; Wan, Z.; Zhao, X.; Yu, Z. Intakes of Folate, Vitamin B6, and Vitamin B12 in Relation to All-Cause and Cause-Specific Mortality: A National Population-Based Cohort. Nutrients 2022, 14, 2253. [Google Scholar] [CrossRef] [PubMed]
- Sanders, A.P.; Claus Henn, B.; Wright, R.O. Perinatal and childhood exposure to cadmium, manganese, and metal mixtures and effects on cognition and behavior: A review of recent literature. Curr. Environ. Health Rep. 2015, 2, 284–294. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.; Wu, X.; Bai, Y.; Feng, W.; Wang, S.; Chen, Z.; Fu, W.; Li, G.; Chen, W.; Wang, G.; et al. Effect of Thallium Exposure and Its Interaction with Smoking on Lung Function Decline: A Prospective Cohort Study. Environ. Int. 2019, 127, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Huo, X.; Xu, L.; Wei, X.; Wu, W.; Wu, X.; Xu, X. Hearing loss in children with e-waste lead and cadmium exposure. Sci. Total Environ. 2018, 624, 621–627. [Google Scholar] [CrossRef]
- Palzes, V.A.; Sagiv, S.K.; Baker, J.M.; Rojas-Valverde, D.; Gutierrez-Vargas, R.; Winkler, M.S.; Fuhrimann, S.; Staudacher, P.; Menezes-Filho, J.A.; Reiss, A.L.; et al. Manganese exposure and working memory-related brain activity in smallholder farmworkers in Costa Rica: Results from a pilot study. Environ. Res. 2019, 173, 539–548. [Google Scholar] [CrossRef]
- Li, D.; Ge, X.; Liu, Z.; Huang, L.; Zhou, Y.; Liu, P.; Qin, L.; Lin, S.; Liu, C.; Hou, Q.; et al. Association between long-term occupational manganese exposure and bone quality among retired workers. Environ. Sci. Pollut. Res. Int. 2020, 27, 482–489. [Google Scholar] [CrossRef]
- Finley, J.W. Does Environmental Exposure to Manganese Pose a Health Risk to Healthy Adults? Nutr. Rev. 2004, 62, 148–153. [Google Scholar] [CrossRef][Green Version]
- Jain, R.B. Concentrations of cadmium, lead, and mercury in blood among US cigarettes, cigars, electronic cigarettes, and dual cigarette-e-cigarette users. Environ. Pollut. 2019, 251, 970–974. [Google Scholar] [CrossRef]
- Bakulski, K.M.; Seo, Y.A.; Hickman, R.C.; Brandt, D.; Vadari, H.S.; Hu, H.; Park, S.K. Heavy Metals Exposure and Alzheimer’s Disease and Related Dementias. J. Alzheimer’s Dis. 2020, 76, 1215–1242. [Google Scholar] [CrossRef]
- Seth, P.; Husain, M.M.; Gupta, P.; Schoneboom, B.A.; Grieder, F.B.; Mani, H.; Maheshwari, R.K. Early onset of virus infection and up-regulation of cytokines in mice treated with cadmium and manganese. BioMetals 2003, 16, 359–368. [Google Scholar] [CrossRef] [PubMed]
- Brucker, N.; Moro, A.; Charao, M.; Bubols, G.; Nascimento, S.; Goethel, G.; Barth, A.; Prohmann, A.C.; Rocha, R.; Moresco, R.; et al. Relationship between blood metals and inflammation in taxi drivers. Clin. Chim. Acta 2015, 444, 176–181. [Google Scholar] [CrossRef] [PubMed]
- Talukdar, S.; Oh, D.Y.; Bandyopadhyay, G.; Li, D.; Xu, J.; McNelis, J.; Lu, M.; Li, P.; Yan, Q.; Zhu, Y.; et al. Neutrophils mediate insulin resistance in mice fed a high-fat diet through secreted elastase. Nat. Med. 2012, 18, 1407–1412. [Google Scholar] [CrossRef] [PubMed]
- Gladkevich, A.; Nelemans, S.A.; Kauffman, H.F.; Korf, J. Microarray profiling of lymphocytes in internal diseases with an altered immune response: Potential and methodology. Mediat. Inflamm. 2005, 2005, 317–330. [Google Scholar] [CrossRef] [PubMed]
- Semple, J.W.; Italiano, J.E., Jr.; Freedman, J. Platelets and the immune continuum. Nat. Rev. Immunol. 2011, 11, 264–274. [Google Scholar] [CrossRef] [PubMed]
- Lolli, C.; Caffo, O.; Scarpi, E.; Aieta, M.; Conteduca, V.; Maines, F.; Bianchi, E.; Massari, F.; Veccia, A.; Chiuri, V.E.; et al. Systemic Immune-Inflammation Index Predicts the Clinical Outcome in Patients with mCRPC Treated with Abiraterone. Front. Pharmacol. 2016, 7, 376. [Google Scholar] [CrossRef]
- Geng, Y.; Shao, Y.; Zhu, D.; Zheng, X.; Zhou, Q.; Zhou, W.; Ni, X.; Wu, C.; Jiang, J. Systemic Immune-Inflammation Index Predicts Prognosis of Patients with Esophageal Squamous Cell Carcinoma: A Propensity Score-matched Analysis. Sci. Rep. 2016, 6, 39482. [Google Scholar] [CrossRef]
- Charkiewicz, A.E.; Backstrand, J.R. Lead Toxicity and Pollution in Poland. Int. J. Environ. Res. Public. Health 2020, 17, 4385. [Google Scholar] [CrossRef]
- Mishra, K.P. Lead exposure and its impact on immune system: A review. Toxicol. Vitr. 2009, 23, 969–972. [Google Scholar] [CrossRef]
- Dou, J.; Zhou, L.; Zhao, Y.; Jin, W.; Shen, H.; Zhang, F. Effects of long-term high-level lead exposure on the immune function of workers. Arch. Environ. Occup. Health 2022, 77, 301–308. [Google Scholar] [CrossRef]
- Zheng, K.; Zeng, Z.; Tian, Q.; Huang, J.; Zhong, Q.; Huo, X. Epidemiological evidence for the effect of environmental heavy metal exposure on the immune system in children. Sci. Total Environ. 2023, 868, 161691. [Google Scholar] [CrossRef] [PubMed]

| Characters | Participants [n (%)] | Blood Mn (μg/L) | p Value | Blood Cd (μg/L) | p Value | Blood Pb (μg/dL) | p Value |
|---|---|---|---|---|---|---|---|
| Age | |||||||
| 20–49 | 1065 (49.0) | 9.84 (7.92, 12.53) | <0.001 | 0.25 (0.16, 0.44) | <0.001 | 0.68 (0.47, 1.11) | <0.001 |
| 49–65 | 618 (28.4) | 9.21 (7.55, 11.60) | 0.33 (0.20, 0.65) | 1.18 (0.82, 1.82) | |||
| >65 | 491 (22.6) | 8.85 (7.01, 10.80) | 0.35 (0.24, 0.56) | 1.43 (0.94, 2.14) | |||
| Sex | |||||||
| Male | 1065 (49.0) | 8.77 (7.18, 13.52) | <0.001 | 0.27 (0.17, 0.49) | <0.001 | 1.15 (0.72, 1.84) | <0.001 |
| Female | 1109 (51.0) | 10.03 (8.17, 12.72) | 0.33 (0.21, 0.55) | 0.81 (0.52, 1.33) | |||
| Race/ethnicity | |||||||
| Mexican American | 662 (30.5) | 9.93 (7.92, 12.54) | <0.001 | 0.26 (0.18, 0.42) | <0.001 | 0.89 (0.57, 1.41) | <0.001 |
| Non-Hispanic white | 778 (35.8) | 9.03 (7.51, 11.30) | 0.29 (0.17, 0.54) | 0.96 (0.58, 1.66) | |||
| Non-Hispanic black | 424 (19.5) | 8.36 (6.76, 12.39) | 0.34 (0.20, 0.66) | 1.01 (0.60, 1.68) | |||
| Others | 310 (14.3) | 11.24 (9.00, 14.33) | 0.36 (0.24, 0.62) | 1.11 (0.69, 1.65) | |||
| Education levels | |||||||
| No and elementary school | 253 (11.6) | 9.98 (7.73, 12.27) | 0.136 | 0.30 (0.20, 0.56) | <0.001 | 1.16 (0.74, 1.92) | <0.001 |
| Middle school | 236 (10.9) | 9.66 (7.86, 11.77) | 0.40 (0.24, 0.71) | 1.20 (0.76, 1.88) | |||
| High school | 483 (22.2) | 9.22 (7.34, 11.75) | 0.34 (0.21, 0.66) | 0.34 (0.58, 1.65) | |||
| College or over | 1201 (55.3) | 9.34 (7.63, 11.84) | 0.27 (0.17, 0.44) | 0.27 (0.56, 1.42) | |||
| Annual house income | |||||||
| <20,000 | 2059 (94.7) | 9.41 (7.83, 11.63) | 0.726 | 0.30 (0.18, 0.53) | 0.018 | 0.95 (0.42, 1.56) | 0.183 |
| >20,000 | 115 (5.3) | 9.43 (7.55, 12.91) | 0.26 (0.17, 0.38) | 1.08 (0.63, 1.70) | |||
| BMI | |||||||
| <25 | 578 (26.6) | 9.43 (7.71, 11.97) | 0.024 | 0.35 (0.19, 0.66) | <0.001 | 1.07 (0.64, 1.76) | <0.001 |
| 25–30 | 701 (32.2) | 9.21 (7.30, 11.60) | 0.29 (0.19, 0.50) | 1.09 (0.69, 1.72) | |||
| ≥30 | 895 (41.2) | 9.53 (7.85, 11.07) | 0.27 (0.18, 0.47) | 0.83 (0.54, 1.33) | |||
| Alcohol intake | |||||||
| Yes | 15,264 (70.1) | 9.19 (7.52, 11.64) | <0.001 | 0.30 (0.18, 0.54) | 0.260 | 1.01 (0.62, 1.64) | <0.001 |
| No | 650 (29.9) | 9.96 (7.90, 11.47) | 0.29 (0.18, 0.49) | 0.87 (0.56, 1.45) | |||
| Exposure to smoking | |||||||
| Yes | 1394 (64.1) | 9.31 (7.60, 11.75) | 0.176 | 0.34 (0.20, 0.67) | <0.001 | 1.04 (0.62, 1.69) | <0.001 |
| No | 780 (35.9) | 9.57 (7.71, 12.02) | 0.25 (0.17, 0.37) | 0.87 (0.57, 1.40) | |||
| SII | |||||||
| <357.52 | 724 (33.3) | 9.14 (7.31, 11.58) | <0.001 | 0.29 (0.18, 0.48) | 0.264 | 1.01 (0.62, 1.55) | 0.556 |
| 357.52–554.91 | 726 (33.4) | 9.41 (7.62, 11.63) | 0.29 (0.19, 0.51) | 0.95 (0.59, 1.51) | |||
| >554.91 | 724 (33.3) | 9.71 (7.87, 12.46) | 0.30 (0.19, 0.57) | 0.92 (0.58, 1.62) |
| Variables | Mn | p Value | Cd | p Value | Pb | p Value |
|---|---|---|---|---|---|---|
| SII | 0.075 | 0.001 ** | 0.055 | 0.011 * | 0.090 | 0.629 |
| Age | −0.144 | <0.001 *** | 0.076 | <0.001 *** | 0.286 | <0.001 *** |
| BMI | 0.045 | 0.037 * | −0.093 | <0.001 *** | −0.137 | <0.001 *** |
| Variables | β (95% CI), p Values for SII | ||
|---|---|---|---|
| Crude Model | Mode I a | Model Ⅱ b | |
| Blood Mn (μg/L) | |||
| Continuous c | 5.80 (2.53, 9.06) 0.001 | 5.65 (2.33, 8.97) 0.001 | 5.96 (2.62, 9.29) <0.001 |
| T1 (<8.21) | 0 [Reference] | 0 [Reference] | 0 [Reference] |
| T2 (8.21–10.94) | −7.45 (−37.85, 22.96) 0.631 | −11.42 (−41.85, 19.00) 0.462 | −10.13 (−40.56, 20.31) 0.514 |
| T3 (>10.94) | 36.57 (5.95, 67.18) 0.019 | 32.67 (1.64, 63.70) 0.039 | 33.49 (4.31, 66.66) 0.026 |
| p value for trend | 0.021 | 0.040 | 0.027 |
| Blood Cd (μg/L) | |||
| Continuous c | 30.06 (7.00, 53.12) 0.011 | 39.88 (16.74,63.03) 0.001 | 42.10 (18.05, 66.14) 0.001 |
| T1 (<0.22) | 0 [Reference] | 0 [Reference] | 0 [Reference] |
| T2 (0.22–0.42) | 14.20 (−16.49, 44.90) 0.364 | 14.32 (−16.73, 45.34) 0.336 | 15.20 (−16.00, 46.40) 0.340 |
| T3 (>0.42) | 30.80 (0.57,61.03) 0.046 | 45.16 (13.78,76.55) 0.005 | 48.91 (16.20,81.64) 0.003 |
| p value for trend | 0.006 | 0.005 | 0.004 |
| Blood Pb (μg/dL) | |||
| Continuous c | 2.02 (−7.99, 12.03) 0.692 | 6.42 (−74.20, 17.04) 0.236 | 6.63 (−4.13, 17.40) 0.227 |
| T1 (<0.71) | 0 [Reference] | 0 [Reference] | 0 [Reference] |
| T2 (0.71–1.33) | −20.93 (−51.47, 9.61) 0.179 | −16.64 (−48.91, 15.63) 0.312 | −17.93 (−50.93, 14.52) 0.279 |
| T3 (>1.33) | −13.76 (−44.46, 16.94) 0.380 | 0.62 (−34.94, 36.17) 0.973 | −1.31 (−37.63, 35.00) 0.943 |
| p value for trend | 0.378 | 0.936 | 0.979 |
| Variables | β (95% CI), p Values for SII | ||
|---|---|---|---|
| Crude Model | Model I a | Model Ⅱ b | |
| Mn*Cd | |||
| Continuous c | 2.76 (0.86, 4.66) 0.004 | 3.59 (1.58, 5.39) <0.001 | 3.66 (1.69, 5.60) <0.001 |
| T1 (<2.00) | 0 [Reference] | 0 [Reference] | 0 [Reference] |
| T2 (2.00–4.24) | 21.70 (−8.88, 52.28) 0.164 | 20.77 (−9.83, 51.37) 0.183 | 22.22 (−8.58, 53.02) 0.157 |
| T3 (>4.24) | 47.85 (17.29, 78.41) 0.002 | 53.34 (27.08, 89.60) <0.001 | 62.57 (30.31, 94.82) <0.001 |
| p value for trend | 0.002 | <0.001 | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhong, Q.; Zhou, W.; Lin, J.; Sun, W.; Qin, Y.; Li, X.; Xu, H. Independent and Combined Associations of Blood Manganese, Cadmium and Lead Exposures with the Systemic Immune-Inflammation Index in Adults. Toxics 2023, 11, 659. https://doi.org/10.3390/toxics11080659
Zhong Q, Zhou W, Lin J, Sun W, Qin Y, Li X, Xu H. Independent and Combined Associations of Blood Manganese, Cadmium and Lead Exposures with the Systemic Immune-Inflammation Index in Adults. Toxics. 2023; 11(8):659. https://doi.org/10.3390/toxics11080659
Chicago/Turabian StyleZhong, Qiya, Wenxin Zhou, Jiaqi Lin, Wen Sun, Yao Qin, Xiang Li, and Huadong Xu. 2023. "Independent and Combined Associations of Blood Manganese, Cadmium and Lead Exposures with the Systemic Immune-Inflammation Index in Adults" Toxics 11, no. 8: 659. https://doi.org/10.3390/toxics11080659
APA StyleZhong, Q., Zhou, W., Lin, J., Sun, W., Qin, Y., Li, X., & Xu, H. (2023). Independent and Combined Associations of Blood Manganese, Cadmium and Lead Exposures with the Systemic Immune-Inflammation Index in Adults. Toxics, 11(8), 659. https://doi.org/10.3390/toxics11080659







