Toxicity of Per- and Polyfluoroalkyl Substances to Nematodes
Abstract
1. Introduction
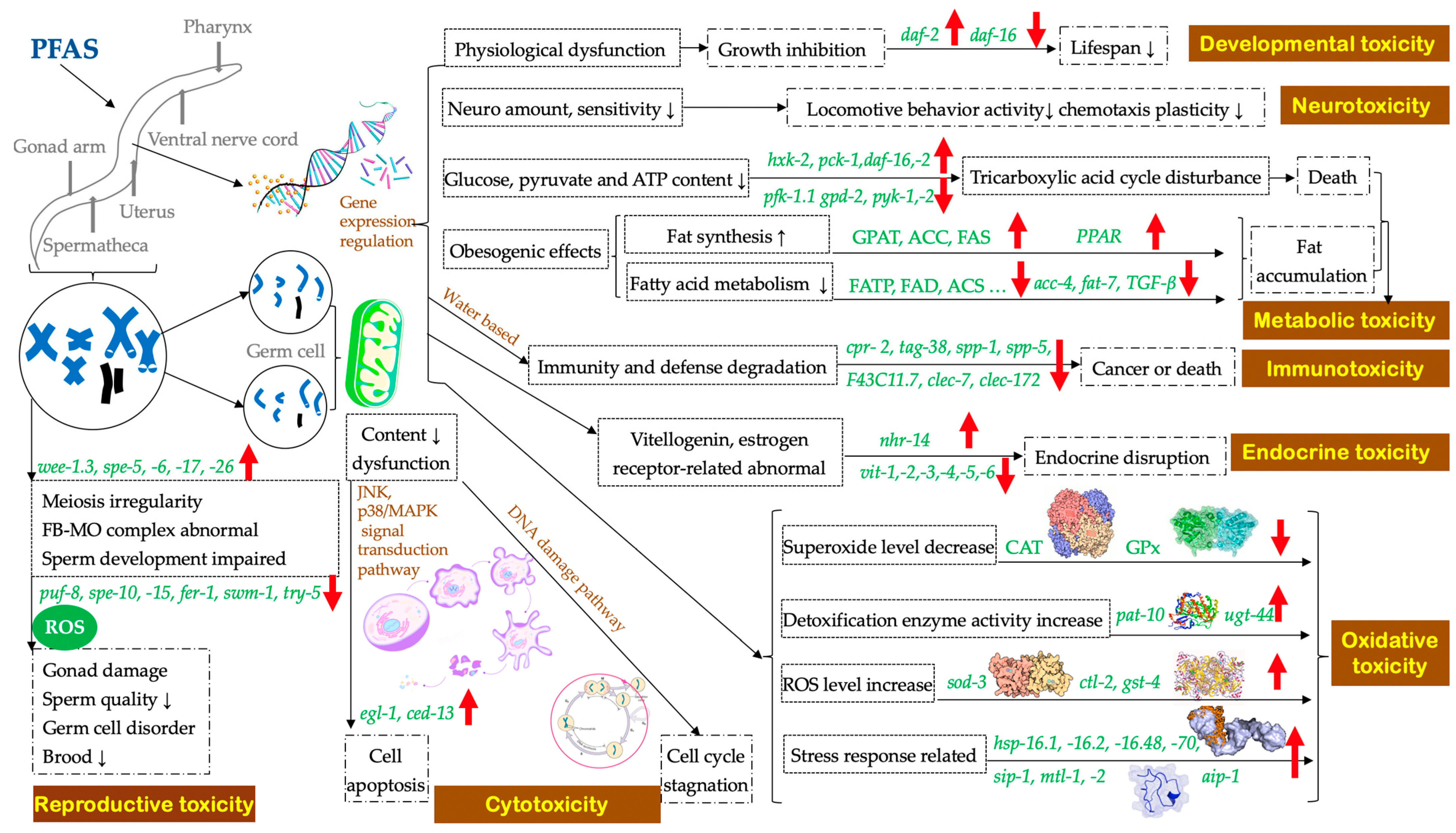
2. Toxicity of Per- and Polyfluoroalkyl Substances to C. elegans
2.1. Accumulation and Lethal Effects
2.2. Reproductive Toxicity
2.3. Cytotoxicity
2.4. Developmental Toxicity
2.5. Neurotoxicity
2.6. Oxidative Toxicity
2.7. Metabolic Toxicity
2.8. Immunotoxicity
2.9. Endocrine Toxicity
3. Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gockener, B.; Fliedner, A.; Rudel, H.; Badry, A.; Koschorreck, J. Long-term trends of per- and polyfluoroalkyl substances (PFAS) in suspended particular matter from German rivers using the direct total oxidizable precursor (dTOP) assay. Environ. Sci. Technol. 2022, 1, 208–217. [Google Scholar] [CrossRef]
- Stahl, T.; Mattern, D.; Brunn, H. Toxicology of perfluorinated compounds. Environ. Sci. Eur. 2011, 23, 38–89. [Google Scholar] [CrossRef]
- Huang, K.; Li, Y.; Bu, D.; Fu, J.; Wang, M.; Zhou, W.; Gu, L.; Fu, Y.; Cong, Z.; Hu, B.; et al. Trophic Magnification of Short-Chain Per- and Polyfluoroalkyl Substances in a Terrestrial Food Chain from the Tibetan Plateau. Environ. Sci. Technol. Lett. 2022, 9, 147–152. [Google Scholar] [CrossRef]
- Kurwadkar, S.; Dane, J.; Kanel, S.R.; Nadagouda, M.N.; Cawdrey, R.W.; Ambade, B.; Struckhoff, G.C.; Wilkin, R. Per- and polyfluoroalkyl substances in water and wastewater: A critical review of their global occurrence and distribution. Sci. Total Environ. 2022, 809, 151003. [Google Scholar] [CrossRef] [PubMed]
- Rashid, F.; Dubinkina, V.; Ahmad, S.; Maslov, S.; Irudayaraj, J.M.K. Gut Microbiome-Host Metabolome Homeostasis upon Exposure to PFOS and GenX in Male Mice. Toxics 2023, 11, 281. [Google Scholar] [CrossRef] [PubMed]
- Ma, T.; Ye, C.; Wang, T.; Li, X.; Luo, Y. Toxicity of Per- and Polyfluoroalkyl Substances to Aquatic Invertebrates, Planktons, and Microorganisms. Int. J. Environ. Res. Public Health 2022, 19, 16729. [Google Scholar] [CrossRef]
- Putten, V.D.; Wim, H.; Bardgett, D.R. Belowground biodiversity and ecosystem functioning. Nature 2014, 515, 505–511. [Google Scholar] [CrossRef]
- Yin, W.Y. Soil Animal in China; China Science Publishing & Media Ltd. (CSPM): Beijing, China, 2000. [Google Scholar]
- Douglas, A.E. Simple animal models for microbiome research. Nat. Rev. Microbiol. 2019, 17, 764–775. [Google Scholar] [CrossRef]
- Van Den Hoogen, J.; Geisen, S.; Wall, D.H.; Wardle, D.A.; Traun-Spurger, W.; De Goede, R.G.M.; Adams, B.J.; Ahmad, W.; Ferris, H.L.; Bardgett, R.D.; et al. A global database of soil nematode abundance and functional group composition. Sci. Data 2020, 7, 103. [Google Scholar] [CrossRef]
- Brazova, T.; Kovacik, P.; Matouskova, M.; Oros, M. Nematodes as soil stress indicators for polycyclic aromatic hydrocarbons: A review. Helminthologia 2022, 59, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Nielsen, U.N.; Yang, X.; Zhang, L.; Zhou, X.; Du, G.; Li, G.; Chen, S.; Xiao, S. Grazing induces direct and indirect shrub effects on soil nematode communities. Soil Biol. Biochem. 2018, 121, 193–201. [Google Scholar] [CrossRef]
- Stylianou, M.; Björnsdotter, M.K.; Olsson, P.-E.; Jogsten, I.E.; Jass, J. Distinct transcriptional response of Caenorhabditis elegans to different exposure routes of perfluorooctane sulfonic acid. Environ. Res. 2019, 168, 406–413. [Google Scholar] [CrossRef] [PubMed]
- Yue, Y.; Li, S.; Shen, P.; Park, Y. Caenorhabditis elegans as a model for obesity research. Curr. Res. Food Sci. 2021, 4, 692–697. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.J. Toxicity of Perfluorobutane Sulfonate as a Substitute for Perfluorooctane Sulfonate on Caenorhabditis elegans. Master’s Thesis, Huazhong Agricultural University, Wuhan, China, June 2017. [Google Scholar]
- Lakowski, B.; Hekimi, S. Determination of life-span in Caenorhabditis elegans by four clock genes. Science 1996, 272, 1010–1013. [Google Scholar] [CrossRef] [PubMed]
- Gubert, P.; Gubert, G.; Oliveira, R.C.D.; Fernandes, I.C.O.; Bezerra, I.C.; de Ramos, B.; de Lima, M.F.; Rodrigues, D.T.; de Cruz, A.F.N.; Pereira, E.C.; et al. Caenorhabditis elegans as a Prediction Platform for Nanotechnology-Based Strategies: Insights on Analytical Challenges. Toxics 2023, 11, 239. [Google Scholar] [CrossRef]
- Hitchcock, D.R.; Black, M.C.; Williams, P.L. Investigations into using the nematode Caenorhabditis elegans for municipal and industrial wastewater toxicity testing. Arch. Environ. Contam. Toxicol. 1997, 33, 252–260. [Google Scholar] [CrossRef]
- Dietrich, N.; Tan, C.-H.; Cubillas, C.; Earley, B.J.; Kornfeld, K. Insights into zinc and cadmium biology in the nematode Caenorhabditis elegans. Arch. Biochem. Biophys. 2016, 611, 120–133. [Google Scholar] [CrossRef]
- Meyer, D.; Williams, P.L. Toxicity Testing of Neurotoxic Pesticides in Caenorhabditis elegans. J. Toxicol. Environ. Health Part B 2014, 17, 284–306. [Google Scholar] [CrossRef]
- Zhao, Y.L.; Chen, J.Y.; Wang, R.; Pu, X.X.; Wang, D.Y. A review of transgenerational and multigenerational toxicology in the in vivo model animal Caenorhabditis elegans. J. Appl. Toxicol. 2023, 43, 122–145. [Google Scholar] [CrossRef]
- Haimbaugh, A.; Wu, C.-C.; Akemann, C.; Meyer, D.N.; Connell, M.; Abdi, M.; Khalaf, A.; Johnson, D.; Baker, T.R. Multi- and Transgenerational Effects of Developmental Exposure to Environmental Levels of PFAS and PFAS Mixture in Zebrafish (Danio rerio). Toxics 2022, 10, 334. [Google Scholar] [CrossRef]
- Wang, Z.; Cousins, I.T.; Scheringer, M.; Hungerbühler, K. Fluorinated alternatives to long-chain perfluoroalkyl carboxylic acids (PFCAs), perfluoroalkane sulfonic acids (PFSAs) and their potential precursors. Environ. Int. 2013, 60, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Li, Z. Multi-Generational Effects of Perfluorinated Compounds (PFCs) on Lipid Metabolism of C. elegans and Its Potential Mechanism. Master’s Thesis, Tongji University, Shanghai, China, April 2020. [Google Scholar]
- Sun, B.; Liu, M.; Tang, L.; Hu, C.; Chen, L. Probiotic supplementation mitigates the developmental toxicity of perfluorobutanesulfonate in zebrafish larvae. Sci. Total Environ. 2021, 799, 149458. [Google Scholar] [CrossRef] [PubMed]
- Ma, T.; Wu, P.; Ding, Z.; Wang, T.; Luo, Y. Pet cats, the better sentinels for indoor organic pollutants. Front. Environ. Sci. 2022, 10, 1023818. [Google Scholar] [CrossRef]
- Feng, Z.; McLamb, F.; Vu, J.P.; Gong, S.; Gersberg, R.M.; Bozinovic, G. Physiological and transcriptomic effects of hexafluoropropylene oxide dimer acid in Caenorhabditis elegans during development. Ecotoxicol. Environ. Saf. 2022, 244, 114047. [Google Scholar] [CrossRef]
- Chowdhury, M.I.; Sana, T.; Panneerselvan, L.; Sivarama, A.K.; Megharaj, M. Perfluorooctane sulfonate (PFOS) induces several behavioural defects in Caenorhabditis elegans that can also be transferred to the next generations. Chemosphere 2022, 291, 132896. [Google Scholar] [CrossRef] [PubMed]
- Sana, T.; Chowdhury, M.I.; Logeshwaran, P.; Dharmarajan, R.; Megharaj, M. Perfluorooctanoic acid (PFOA) induces behavioural, reproductive and developmental toxicological impacts in Caenorhabditis elegans at concentrations relevant to the contaminated areas. Environ. Adv. 2021, 4, 100053. [Google Scholar] [CrossRef]
- Chowdhury, M.I.; Sana, T.; Panneerselvan, L.; Dharmarajan, R.; Megharaj, M. Acute Toxicity and Transgenerational Effects of Perfluorobutane Sulfonate on Caenorhabditis elegans. Environ. Toxicol. Chem. 2021, 40, 1971–1980. [Google Scholar] [CrossRef]
- Sana, T.; Chowdhury, M.I.; Logeshwaran, P.; Megharaj, M. Behavioural, developmental and reproductive toxicological impacts of perfluorobutanoic acid (PFBA) in Caenorhabditis elegans. Environ. Chall. 2023, 10, 100662. [Google Scholar] [CrossRef]
- Chen, N. Neurotoxicity of Chronic Exposure to Perfluorooctane Sulfonate and Its Mechanisms, In Vitro and In Vivo. Master’s Thesis, East China Normal University, Shanghai, China, May 2014. [Google Scholar]
- Guo, X.Y. The Toxicity of Growth and Germ Line Development Induced by Perfluorooctane Sulfonates in Caenorhabditis elegans. Doctor’s Thesis, Hefei Institutes of Physical Science, Chinese Academy of Sciences, Hefei, China, May 2014. [Google Scholar]
- Jian, Z.H. The Male Reproductive Toxicity of Perfluorooctane Sulfonates and Perfluorooctanoic Acid on C. elegans. Master’s Thesis, Southeast University, Nanjing, China, June 2017. [Google Scholar]
- Yue, Y.; Li, S.; Qian, Z.; Pereira, R.F.; Lee, J.; Doherty, J.J.; Zhang, Z.; Peng, Y.; Clark, J.M.; Timme-Laragy, A.R.; et al. Perfluorooctanesulfonic acid (PFOS) and perfluorobutanesulfonic acid (PFBS) impaired reproduction and altered offspring physiological functions in Caenorhabditis elegans. Food Chem. Toxicol. 2020, 145, 111695. [Google Scholar] [CrossRef]
- Sammi, S.R.; Foguth, R.M.; Nieves, C.S.; De Perre, C.; Wipf, P.; McMurray, C.T.; Lee, L.S.; Cannon, J.R. Perfluorooctane Sulfonate (PFOS) Produces Dopaminergic Neuropathology in Caenorhabditis elegans. Toxicol. Sci. 2019, 172, 417–434. [Google Scholar] [CrossRef]
- Wei, C.Y. Effects of PFOS on Lipid and Glucose Metabolism in Caenorhabditis elegans. Master’s Thesis, Huazhong Agricultural University, Wuhan, China, June 2018. [Google Scholar]
- Wei, C.; Zhou, Z.; Wang, L.; Huang, Z.; Liang, Y.; Zhang, J. Perfluorooctane sulfonate (PFOS) disturbs fatty acid metabolism in Caenorhabditis elegans: Evidence from chemical analysis and molecular mechanism exploration. Chemosphere 2021, 277, 130359. [Google Scholar] [CrossRef]
- Li, Z.; Yu, Z.; Gao, P.; Yin, D. Multigenerational effects of perfluorooctanoic acid on lipid metabolism of Caenorhabditis elegans and its potential mechanism. Sci. Total Environ. 2020, 703, 134762. [Google Scholar] [CrossRef]
- Mangu, J.C.K.; Stylianou, M.; Olsson, P.-E.; Jass, J. Per- and polyfluoroalkyl substances enhance Staphylococcus aureus pathogenicity and impair host immune response. Environ. Pollut. 2022, 314, 120294. [Google Scholar] [CrossRef]
- Hanssen, L.; Röllin, H.; Odland, J.Ø.; Moe, M.K.; Sandanger, T.M. Perfluorinated compounds in maternal serum and cord blood from selected areas of South Africa: Results of a pilot study. J. Environ. Monit. 2010, 12, 1355–1361. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Ge, P.; Lu, Z.; Yang, R.; Liu, Z.; Zhao, F.; Chen, M. Reproductive toxicity and underlying mechanisms of fine particulate matter (PM2.5) on Caenorhabditis elegans in different seasons. Ecotoxicol. Environ. Saf. 2022, 248, 114281. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Cao, Z.; Xu, A.; Du, H. Evaluation of toxicity and mutagenicity of oxaliplatin on germ cells in an alternative in vivo model Caenorhabditis elegans. Food Chem. Toxicol. 2023, 178, 113902. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.A.; Huang, C.W.; Wei, C.C. Early-life perfluorooctanoic acid (PFOA) and perfluorooctane sulfonic acid (PFOS) exposure cause obesity by disrupting fatty acids metabolism and enhancing triglyceride synthesis in Caenorhabditis elegans. Aqua. Toxicol. 2022, 251, 106274. [Google Scholar] [CrossRef]
- Haerkens, F.; Kikken, C.; Kirkels, L.; van Amstel, M.; Wouters, W.; van Doornmalen, E.; Francke, C.; Hughes, S. A new use for old drugs: Identifying compounds with an anti-obesity effect using a high through-put semi-automated Caenorhabditis elegans screening platform. Heliyon 2022, 8, e10108. [Google Scholar] [CrossRef]
- Lee, Y.; Choi, S.; Kim, K.W. Dithianon exposure induces dopaminergic neurotoxicity in Caenorhabditis elegans. Ecotoxicol. Environ. Saf. 2023, 255, 114752. [Google Scholar] [CrossRef]
- How, C.M.; Li, S.W.; Liao, V.H.C. Chronic exposure to triadimenol at environmentally relevant concentration adversely affects aging biomarkers in Caenorhabditis elegans associated with insulin/IGF-1 signaling pathway. Sci. Total Environ. 2018, 640–641, 485–492. [Google Scholar] [CrossRef]
- Mukherjee, S.; Russell, J.C.; Carr, D.T.; Burgess, J.D.; Younkin, M.A.; Serie, D.; Boehme, K.L.; Kauwe, J.S.K.; Naj, A.C.; Fardo, D.W.; et al. Systems biology approach to late-onset Alzheimer’s disease genome-wide association study identifies novel candidate genes validated using brain expression data and Caenorhabditis elegans experiments. Alzheimer’s Dement. 2017, 13, 1133–1142. [Google Scholar] [CrossRef]
- Yan, N.; Chai, J.; Lee, E.S.; Gu, L.; Liu, Q.; He, J.; Wu, J.-W.; Kokel, D.; Li, H.; Hao, Q.; et al. Structure of the CED-4–CED-9 complex provides insights into programmed cell death in Caenorhabditis elegans. Nature 2005, 437, 831–837. [Google Scholar] [CrossRef]
- Conradt, B.; Horvitz, A.H.R. The Protein EGL-1 Is Required for Programmed Cell Death and Interacts with the Bcl-2–like Protein CED-9. Cell 1998, 93, 519–529. [Google Scholar] [CrossRef]
- Schumacher, B.; Schertel, C.; Wittenburg, N.; Tuck, S.; Mitani, S.; Gartner, A.; Conradt, B.; Shaham, S. C. elegans ced-13 can promote apoptosis and is induced in response to DNA damage. Cell Death Differ. 2005, 12, 153–161. [Google Scholar] [CrossRef]
- Srinivasan, S. Neuroendocrine control of lipid metabolism: Lessons from C. elegans. J. Neurogenet. 2020, 34, 482–488. [Google Scholar] [CrossRef] [PubMed]
- Crawford, N.; Martell, M.; Nielsen, T.; Khalil, B.; Imtiaz, F.; Nguidjo, E.; Newell-Caito, J.L.; Bornhorst, J.; Schwerdtle, T.; Caito, S.W. Methylmercury-Induced Metabolic Alterations in Caenorhabditis elegans Are Diet-Dependent. Toxics 2021, 9, 287. [Google Scholar] [CrossRef]
- Berger, J.P.; Akiyama, T.E.; Meinke, P.T. PPARs: Therapeutic targets for metabolic disease. Trends Pharmacol. Sci. 2005, 26, 244–251. [Google Scholar] [CrossRef]
- Maradonna, F.; Carnevali, O. Lipid Metabolism Alteration by Endocrine Disruptors in Animal Models: An Overview. Front. Endocrinol. 2018, 9, 654. [Google Scholar] [CrossRef]
- Dewitt, J.C. Toxicological Effects of Perfluoroalkyl and Polyfluoroalkyl Substances; Humana: Cham, Switzerland, 2015; pp. 1–495. [Google Scholar]
- Sze, J.Y.; Victor, M.; Loer, C.; Shi, Y.; Ruvkun, G. Food and metabolic signalling defects in a Caenorhabditis elegans serotonin-synthesis mutant. Nature 2000, 403, 560–564. [Google Scholar] [CrossRef] [PubMed]
- Slobodian, M.R.; Petahtegoose, J.D.; Wallis, A.L.; Levesque, D.C.; Merritt, T.J.S. The Effects of Essential and Non-Essential Metal Toxicity in the Drosophila melanogaster Insect Model: A Review. Toxics 2021, 9, 269. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhang, L.; Wang, X. Molecular toxicity and defense mechanisms induced by silver nanoparticles in Drosophila melanogaster. J. Environ. Sci. 2023, 125, 616–629. [Google Scholar] [CrossRef]
- Kim, J.H.; Barbagallo, B.; Annunziato, K.; Farias-Pereira, R.; Doherty, J.J.; Lee, J.; Zina, J.; Tindal, C.; McVey, C.; Aresco, R.; et al. Maternal preconception PFOS exposure of Drosophila melanogaster alters reproductive capacity, development, morphology and nutrient regulation. Food Chem. Toxicol. 2021, 151, 112153. [Google Scholar] [CrossRef]
- Shen, C.; Zuo, Z. Zebrafish (Danio rerio) as an excellent vertebrate model for the development, reproductive, cardiovascular, and neural and ocular development toxicity study of hazardous chemicals. Environ. Sci. Pollut. Res. 2020, 27, 43599–43614. [Google Scholar] [CrossRef] [PubMed]
- Ma, T.; Wu, P.; Wang, L.; Li, Q.; Li, X.; Luo, Y. Toxicity of per- and polyfluoroalkyl substances to aquatic vertebrates. Front. Environ. Sci. 2023, 11, 1101100. [Google Scholar] [CrossRef]
- Fey, M.E.; Goodrum, P.E.; Razavi, N.R.; Whipps, C.M.; Fernando, S.; Anderson, J.K. Is Mixtures’ Additivity Supported by Empirical Data? A Case Study of Developmental Toxicity of PFOS and 6:2 FTS in Wildtype Zebrafish Embryos. Toxics 2022, 10, 418. [Google Scholar] [CrossRef] [PubMed]
- Rericha, Y.; Truong, L.; Leong, C.; Cao, D.; Field, J.A.; Tanguay, R.L. Dietary Perfluorohexanoic Acid (PFHxA) Exposures in Juvenile Zebrafish Produce Subtle Behavioral Effects across Generations. Toxics 2022, 10, 372. [Google Scholar] [CrossRef]
- Spurgeon, D.J.; Morgan, A.J.; Kille, P. Current research in soil invertebrate ecotoxicogenomics. Adv. Exp. Biol. 2008, 2, 133–326. [Google Scholar] [CrossRef]
- Organization for Economic Cooperation and Development. Acute Toxicity Tests. Guideline for Testing Chemicals; No. 207; OECD: Paris, France, 1984. [Google Scholar]
- Melo, T.M.; Schauerte, M.; Bluhm, A.; Slany, M.; Paller, M.; Bolan, N.; Bosch, J.; Fritzsche, A.; Rinklebe, J. Ecotoxicological effects of per- and polyfluoroalkyl substances (PFAS) and of a new PFAS adsorbing organoclay to immobilize PFAS in soils on earthworms and plants. J. Hazard. Mater. 2022, 433, 128771. [Google Scholar] [CrossRef]
- Cai, Y.; Wang, Q.; Zhou, B.; Yuan, R.; Wang, F.; Chen, Z.; Chen, H. A review of responses of terrestrial organisms to perfluorinated compounds. Sci. Total Environ. 2021, 793, 148565. [Google Scholar] [CrossRef]
- Wang, Z.; Shi, Y.; Zhang, Z.; Qi, F.; Xue, W.; Tian, D.; Teng, S. Assessing joint toxic effects of arsenate and perfluorooctane sulfonate on earthworm by combining integrated biomarker response and mixture toxicity indices. Ecolo. Indic. 2022, 144, 109521. [Google Scholar] [CrossRef]
- Zhang, C.; Zhang, X.; Xu, Y.; Qiao, M. Ecotoxicity of Alternatives of Typical Perfluorooctane Sulfonate (PFOS) to Springtails. Asian J. Ecotoxicol. 2012, 7, 501–507. Available online: http://ir.rcees.ac.cn/handle/311016/8247 (accessed on 16 July 2012).
- Zhang, X.; Zhang, C.; Wang, G.; Qiao, M.; Zhu, Y. Ecotoxicity of Perfluorooctane Sulfonate (PFOS) to Springtails in Soils. Asian J. Ecotoxicol. 2012, 7, 525–529. [Google Scholar]
- Groffen, T.; Prinsen, E.; Stoffels, O.A.D.; Maas, L.; Vincke, P.; Lasters, R.; Eens, M.; Bervoets, L. PFAS accumulation in several terrestrial plant and invertebrate species reveals species-specific differences. Environ. Sci. Pollut. Res. 2023, 30, 23820–23835. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.Y.; Miao, Y.L.; Gao, Q.; Cui, Z.K.; Xiong, B. Exposure to perfluorooctane sulfonate in vitro perturbs the quality of porcine oocytes via induction of apoptosis. Environ. Pollut. 2021, 284, 117508. [Google Scholar] [CrossRef] [PubMed]
| Pollutant | Indicator | Value | Reference |
|---|---|---|---|
| PFOS | 48 h | 1.4 μM (0.699 mg/L) | [15] |
| PFBS | 793.6 μM (238.82 mg/L) | ||
| PFOS | 48 h | 3.15 μM (1.575 mg/L) | [28] |
| PFOA | 48 h | 4.42 μM (1.83 mg/L) | [29] |
| PFBS | 48 h | 1.60 mM (481.5 mg/L) | [30] |
| PFBA | 48 h | 2.86 mM (612.15 mg/L) | [31] |
| PFOS | 24 h | 3.484 mM (1738.76 mg/L) | [32] |
| 48 h | 2.028 mM (1012.11 mg/L) | ||
| 72 h | 0.842 mM (420.22 mg/L) |
| Effect | Impairment Detail | Pollutant | Reference |
|---|---|---|---|
| Gonad damage | Germ cell arrangement disorder | PFOS | [33] |
| Cell spacing increase | |||
| Gonad size decrease | |||
| Gonad cell number decrease | |||
| Brood inhibition | Brood amount/size (total egg number) decrease | PFOS, PFBS, PFOA, PFBA | [28,29,30] |
| Progeny number (hatched offspring number) decrease | PFOA, PFOS, PFBS | [15,33,34] | |
| Generation time prolongation | PFOS, PFBA | [31,34] | |
| Growth inhibition | Body length/area decrease | PFOS, PFOA, PFBA, PFBS | [15,28,31,33] |
| Body width decrease | PFOS | [32] | |
| Developmental delay | PFOS, PFBS, GenX | [27,33] | |
| Progeny production delay | GenX | [27] | |
| Life expectancy inhibition | PFOS, PFOA, PFBA, PFBS | [28,29,30,31,33] | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, T.; Pan, X.; Wang, T.; Li, X.; Luo, Y. Toxicity of Per- and Polyfluoroalkyl Substances to Nematodes. Toxics 2023, 11, 593. https://doi.org/10.3390/toxics11070593
Ma T, Pan X, Wang T, Li X, Luo Y. Toxicity of Per- and Polyfluoroalkyl Substances to Nematodes. Toxics. 2023; 11(7):593. https://doi.org/10.3390/toxics11070593
Chicago/Turabian StyleMa, Tingting, Xia Pan, Tiantian Wang, Xiuhua Li, and Yongming Luo. 2023. "Toxicity of Per- and Polyfluoroalkyl Substances to Nematodes" Toxics 11, no. 7: 593. https://doi.org/10.3390/toxics11070593
APA StyleMa, T., Pan, X., Wang, T., Li, X., & Luo, Y. (2023). Toxicity of Per- and Polyfluoroalkyl Substances to Nematodes. Toxics, 11(7), 593. https://doi.org/10.3390/toxics11070593








