Assessment of Knowledge on Metal Trace Element Concentrations and Metallothionein Biomarkers in Cetaceans
Abstract
1. Introduction
1.1. Metals Are Ubiquitous Pollutants in Cetaceans
1.2. Metallothionein, a Biomarker in Response to Metal Contaminations
1.3. Characterization of Metallothioneins in Cetaceans
2. Material and Methods
2.1. Characterization of Metallothionein Sequences inside Available Transcriptomes and Genomes of Cetaceans
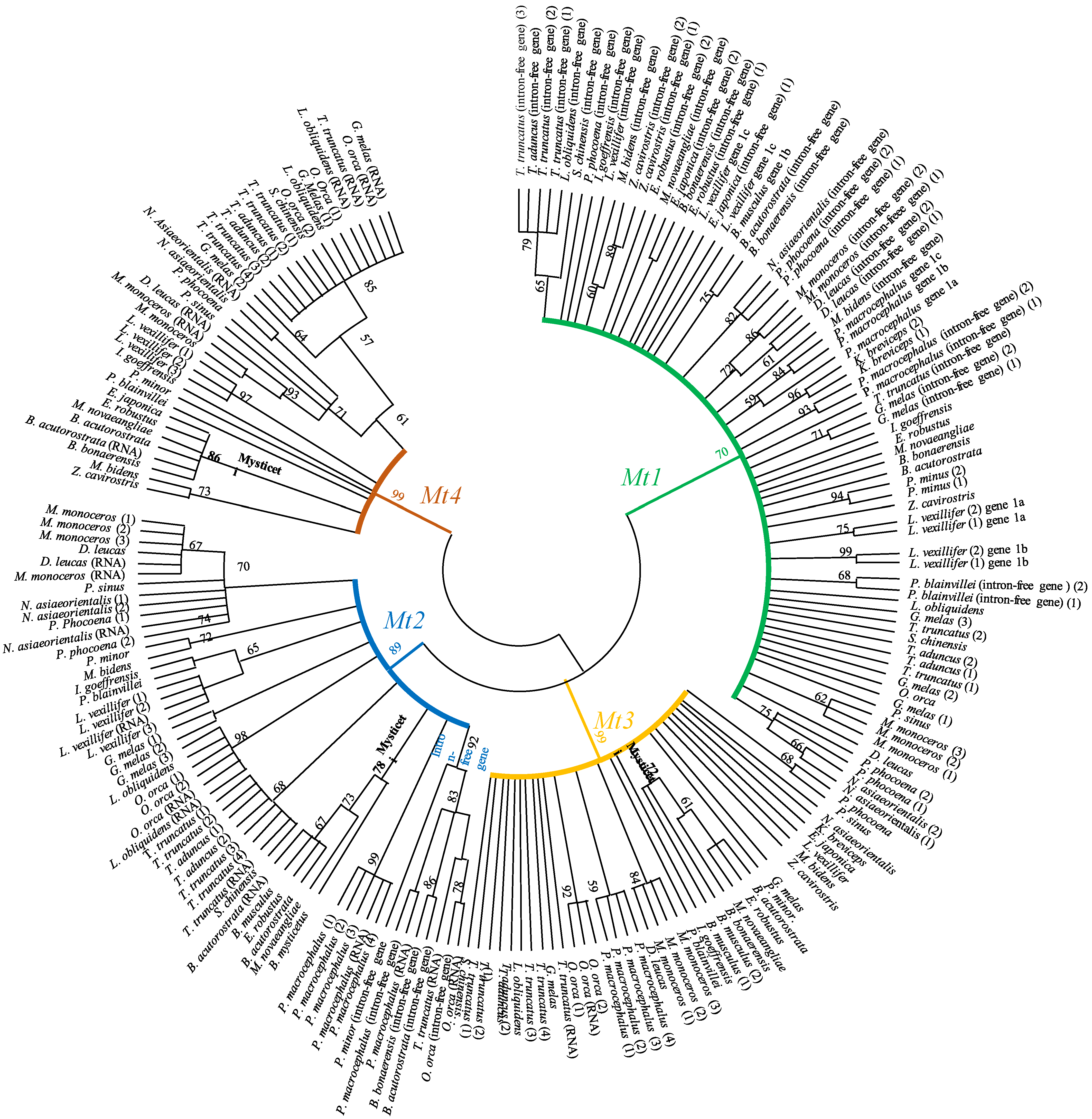
2.2. Phylogenetic Analysis of Metallothioneins in Cetaceans
3. Results
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Page-Karjian, A.; Lo, C.F.; Ritchie, B.; Harms, C.A.; Rotstein, D.S.; Han, S.; Hassan, S.M.; Lehner, A.F.; Buchweitz, J.P.; Thayer, V.G.; et al. Anthropogenic Contaminants and Histopathological Findings in Stranded Cetaceans in the Southeastern United States, 2012–2018. Front. Mar. Sci. 2020, 7, 630. [Google Scholar] [CrossRef]
- Fossi, M.C.; Marsili, L. The use of non-destructive biomarkers in the study of marine mammals. Biomarkers 1997, 2, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Weijs, L.; Zaccaroni, A. Toxicology of marine mammals: News developments on opportunities. Arch. Environ. Contam. Toxicol. 2016, 70, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Baulch, S.; Perry, C. Evaluating the impacts of marine debris on cetaceans. Mar. Pollut. Bull. 2014, 80, 210–221. [Google Scholar] [CrossRef] [PubMed]
- Kehring, H.A.; Hauser-Davis, R.A.; Seixas, T.G.; Pinheiro, A.B.; Paula, A.; Di Beneditto, M. Mercury species, selenium, metallothioneins and glutathione in two dolphins from the southeastern Brazilian coast: Mercury detoxification and physiological differences in diving capacity. Environ. Pollut. 2016, 213, 785–792. [Google Scholar] [CrossRef]
- Kaschner, K.; Tittensor, D.P.; Ready, J.; Gerrodette, T.; Worm, B. Current and future patterns of global marine mammal biodiversity. PLoS ONE 2011, 6, e19653. [Google Scholar] [CrossRef]
- Càceres-Saez, I.; Ribeiro Guevara, S.; Dellabianca, N.A.; Goodall, N.P.; Cappozzo, H.L. Heavy metals and essential elements in Commerson’s dolphins (Cephalorhynchus c. commersonii) from the southwestern South Atlantic Ocean. Environ. Monit. Assess. 2013, 185, 5375–5386. [Google Scholar] [CrossRef]
- Wang, W.-C.; Mao, H.; Ma, D.-D.; Yang, W.-X. Characteristics, functions, and applications of metallothionein in aquatic vertebrates. Front. Mar. Sci. 2014, 1, 34. [Google Scholar] [CrossRef]
- Durante, C.A.; Reis, B.M.M.; Azevedo, A.; Crespo, E.A.; Lailson-Brito, J. Trace elements in trophic webs from South Atlantic: The use of cetaceans as sentinels. Mar. Pollut. Bull. 2020, 150, 110674. [Google Scholar] [CrossRef]
- Dorneles, P.R.; Lailson-Brito, J.; Dos Santos, R.A.; Silva da Costa, P.A.; Malm, O.; Azevedo, A.F.; Machado Torres, J.P. Cephalopods and cetaceans as indicators of offshore bioavailability of cadmium off Central South Brazil Bight. Environ. Pollut. 2007, 148, 352–359. [Google Scholar] [CrossRef]
- Lahaye, V.; Bustamante, P.; Dabin, W.; Van Canneyt, O.; Dhermain, F.; Cesarini, C.; Pierce, G.J.; Caurant, F. New insights from age determination on toxic element accumulation in striped and bottlenose dolphins from Atlantic and Mediterranean waters. Mar. Pollut. Bull. 2006, 52, 1219–1230. [Google Scholar] [CrossRef] [PubMed]
- Seixas, T.G.; Kehrig, H.A.; Fillman, G.; Di Beneditto, A.P.M.; Souza, C.M.; Secchi, E.R.; Moreira, I.; Malm, O. Ecological and biological determinants of trace elements accumulation in liver and kidney of Pontoporia blainvillei. Sci. Total Environ. 2007, 385, 208–220. [Google Scholar] [CrossRef] [PubMed]
- Jihen, E.H.; Imed, M.; Fatima, H.; Abdelhamid, K. Protective effects of selenium (Se) and zinc (Zn) on cadmium (Cd) toxicity in the liver and kidney of the rat: Histology and Cd accumulation. Food Chem. Toxicol. 2008, 46, 3522–3527. [Google Scholar] [CrossRef] [PubMed]
- Elgaml, S.A.; Khalil, R.; Hashis, E.A.; El-Murr, A. Protective effects of selenium and alpha-tocopherol against lead-induced hepatic and renal toxicity in Oreochromis niloticus. J. Aquac. Res. Dev. 2015, 6. [Google Scholar] [CrossRef]
- Botté, A.; Seguin, C.; Nahrgang, J.; Zaidi, M.; Guery, J.; Leignel, V. Lead in the marine environment: Concentrations and effects on invertebrates. Ecotoxicology 2022, 31, 194–207. [Google Scholar] [CrossRef] [PubMed]
- Rogalska, J.; Pilat-Marcikiewicz, B.; Brozka, M.M. Protective effective of zinc against cadmium hepatotoxicity depends on this bioelement intake and level of cadmium exposure: A study in a rat model. Chem. Biol. Interact. 2011, 193, 191–203. [Google Scholar] [CrossRef]
- Monteiro, F.; Lemos, L.S.; Fulgêncio de Moura, J.; Chàvez Rocha, R.C.; Moreira, I.; Di Beneditto, A.P.; Kehrig, H.A.; Bordon, I.C.A.C.; Siciliano, S.; Saint’Pierre, T.D.; et al. Subcellular metal distribution and metallothionein associations in rough-toothed dolphins (Steno bredanesis) from Southeastern Brazil. Mar. Pollut. Bull. 2019, 14, 263–273. [Google Scholar] [CrossRef]
- Méndez-Fernandez, P.; Spitz, J.; Dars, C.; Dabin, W.; Mahfouz, C.; André, J.-M.; Chouvelon, T.; Authier, M.; Caurant, F. Two cetacean species reveal different long-term trends for toxic trace elements in European Atlantic French waters. Chemosphere 2022, 294, 133676. [Google Scholar] [CrossRef]
- Law, R.J.; Fleman, C.F.; Hopkins, A.D.; Baker, J.R.; Harwood, J.; Jackson, D.B.; Kennedy, S.; Maritn, A.R.; Morris, R.J. Concentrations of trace metals in the livers of marine mammals (seals, porpoises and dolphins) from waters around the British Isles. Mar. Pollut. Bull. 1991, 22, 183–191. [Google Scholar] [CrossRef]
- Romero, M.B.; Polizzi, P.; Chiodi, L.; Robles, A.; Gerpe, M. Metals as chemical tracers to discriminate ecological populations of threatened Franciscana dolphins (Pontoporia blainvillei) from Argentina. Environ. Sci. Pollut. Res. 2017, 24, 3940–3950. [Google Scholar] [CrossRef]
- Lailson-Brito, J.; Cruz, R.; Domeles, P.R.; Andrade, L.; Azevedo, A.d.F.; Fragoso, A.B.; Vidal, L.G.; Costa, M.B.; Bisi, T.L.; Almeida, R.; et al. Mercury-Selenium relationship in liver of Guiana dolphin: The possible role of kupffer cells in the detoxification process by tiemannite formation. PLoS ONE 2012, 7, e42162. [Google Scholar] [CrossRef] [PubMed]
- Roditi-Elasar, M.; Kerem, D.; Hornung, H.; Kress, N.; Shoham-Frider, E.; Goffman, O.; Spanier, E. Heavy metal levels in bottlenose and striped dolphins off the Mediterranean coast of Israel. Mar. Pollut. Bull. 2003, 46, 491–521. [Google Scholar] [CrossRef] [PubMed]
- Decataldo, A.; Di Leo, A.; Giandomenico, S.; Cardellicchio, N. Association of metals (mercury, cadmium and zinc) with metallothionein-like proteins in storage organs of stranded dolphins from the Mediterranean Sea (Southern Italy). J. Environ. Monit. 2004, 6, 361–367. [Google Scholar] [CrossRef]
- Hansen, A.M.K.; Bryan, C.E.; West, K.; Jensen, B.A. Trace Element Concentrations in Liver of 16 Species of Cetaceans Stranded on Pacific Islands from 1997 through 2013. Arch. Environ. Contam. Toxicol. 2016, 70, 75–95. [Google Scholar] [CrossRef] [PubMed]
- Jirillo, E.; Caccavo, D.; Magrone, T.; Piccigallo, E.; Amati, L.; Lembo, A.; Kalis, C.; Gumenscheimer, M. The role of the liver in the response to LPS: Experimental and clinical findings. J. Endotoxin. Res. 2002, 8, 319–327. [Google Scholar] [CrossRef]
- Zhou, J.L.; Salvador, S.M.; Liu, Y.P.; Sequeira, M. Heavy metals in the tissues of common dolphins (Delphinus delphis) stranded on the Portuguese coast. Sci. Total Environ. 2001, 273, 61–76. [Google Scholar] [CrossRef] [PubMed]
- Machovsky-Capuska, G.E.; von Haeften, G.; Romero, M.A.; Rodriguez, D.H.; Gerpe, M.S. Linking cadmium and mercury accumulation to nutritional intake in common dolphins (Delphinus delphis) from Patagonia, Argentina. Environ. Pollut. 2020, 263, 114480. [Google Scholar] [CrossRef]
- Lischka, A.; Betty, E.L.; Braid, H.E.; Pook, C.J.; Gaw, S.; Bolstad, K.S.R. Trace element concentrations, including Cd and Hg, in long-finned pilot whales (Globicephala melas edwardii) mass stranded on the New Zealand coast. Mar. Pollut. Bull. 2021, 165, 112084. [Google Scholar] [CrossRef]
- Holsbeek, L.; Joiris, C.R.; Debacker, V.; Ali, I.B.; Roose, P.; Nellissen, J.-P.; Gobert, S.; Bouqueneau, J.-M.; Bossicart, M. Heavy metals, organochlorines and polycyclic aromatic hydrocarbons in sperm whales stranded in the southern North Sea during the 1994/1995 winter. Mar. Pollut. Bull. 1999, 38, 304–313. [Google Scholar] [CrossRef]
- Roesijadi, G. Metallothionein induction as a measure of response to metal exposure in aquatic animals. Environ. Health Perspect. 1994, 102, 91–95. [Google Scholar] [CrossRef]
- Roesijadi, G. Metallothioneins in metal regulation and toxicity in aquatic animals. Aquat. Toxicol. 1992, 22, 81–114. [Google Scholar] [CrossRef]
- Palmiter, R.D. The elusive function of metallothioneins. Proc. Natl. Acad. Sci. USA 1998, 95, 8428–8430. [Google Scholar] [CrossRef] [PubMed]
- Kägi, J.H.R.; Vasàk, M.; Lerch, K.; Gilg, D.E.O.; Hunziker, P.; Werner, R.; Good, B.; Good, M. Structure of mammalian metallothionein. Environ. Health Perspect. 1984, 54, 93–103. [Google Scholar] [PubMed]
- Kägi, J.; Vallee, B.L. Metallothionein: A cadmium- and zinc-containing protein from equine renal cortex. J. Biol. Chem. 1960, 235, 3460–3465. [Google Scholar] [CrossRef]
- Nielson, K.B.; Atkin, C.L.; Winge, D.R. Distinct metal-binding configurations in metallothionein. J. Biol. Chem. 1985, 260, 5342–5350. [Google Scholar] [CrossRef]
- Hamer, D.H. Metallothionein. Annu. Rev. Biochem. 1986, 55, 913–951. [Google Scholar] [CrossRef]
- Moffatt, P.; Denizeau, F. Metallothionein in physiological and physiopathological processes. Drug Metab. Rev. 1997, 29, 261–307. [Google Scholar] [CrossRef]
- Sakulsak, N. Metallothionein: An overview on its metal homeostatic regulation in mammals. Int. J. Morphol. 2012, 30, 1007–1012. [Google Scholar] [CrossRef]
- Ruttkay-Nedesky, B.; Nejdl, L.; Gumulec, J.; Zitka, O.; Masarik, M.; Eckschlager, T.; Stiborova, M.; Adam, V.; Kizek, R. The role of metallothionein in oxidative stress. Int. J. Mol. Sci. 2013, 1, 6044–6066. [Google Scholar] [CrossRef]
- Serén, N.; Glaberman, S.; Carretero, M.A.; Chiari, Y. Molecular evolution and functional divergence of the metallothionein gene family in vertebrates. J. Mol. Evol. 2014, 78, 217–233. [Google Scholar] [CrossRef]
- Coyle, P.; Philcox, J.C.; Carey, L.C.; Rofe, A.M. Metallothionein: The multipurpose protein. Cell. Mol. Life. Sci. 2002, 59, 627–647. [Google Scholar] [CrossRef] [PubMed]
- Roesijadi, G. Metallothionein and its role in toxic metal regulation. Comp. Biochem. Physiol. 1996, 113, 117–123. [Google Scholar] [CrossRef]
- Arizono, K.; Peterson, K.L.; Brady, F.O. Inhibitors of Ca2+ channels, calmodulin and protein kinases prevent A23187 and other inductions of metallothionein mRNA in EC, rat hepatoma cells. Life Sci. 1993, 53, 1031–1037. [Google Scholar] [CrossRef] [PubMed]
- Ridlington, J.W.; Chapman, D.C.; Goeger, D.E.; Whanger, P.D. Metallothionein and Cu-chelatin: Characterization of metal-binding proteins from tissues of four marine animals. Comp. Biochem. Physiol. Part B Comp. Biochem. 1981, 70, 93–104. [Google Scholar] [CrossRef]
- Kwohn, Y.-T.; Yamazaki, S.; Okubo, A.; Yoshimura, E.; Tatsukawa, R.; Toda, S. Isolation and characterization of metallothionein from kidney of striped dolphins Stenella coeruleoalba. Agric. Biol. Chem. 1986, 50, 2881–2885. [Google Scholar]
- Das, K.; Jacob, V.; Debacker, V.; Millerioux, G.; Biondo, R.; Gerardin, N.; Bouquegneau, J.-M. Characterization and potential role of the metallothioneins in marine mammals. Comp. Biochem. Physiol. Part A 2000, 126, S1–S163. [Google Scholar] [CrossRef]
- Das, K.; Jacob, V.; Bouquegneau, J.M. White-sided dolphin metallothioneins: Purification, characterization and potential role. Comp. Biochem. Physiol. Part C 2002, 131, 245–251. [Google Scholar] [CrossRef]
- Mehra, P.K.; Brenmer, I. Studies on the metabolism of rat liver copper-metabolism. Biochem. J. 1985, 227, 903–908. [Google Scholar] [CrossRef]
- Caurant, F.; Navarro, M.; Amiard, J.C. Mercury in pilot whales: Possible limits to the detoxification process. Sci. Total Environ. 1996, 186, 95–104. [Google Scholar] [CrossRef]
- Ikemoto, T.; Kunito, T.; Anan, Y.; Tanaka, H.; Baba, N.; Miyazari, N.; Tanabe, S. Association of heavy metals with metallothionein and other proteins in hepatic cytosol of marine mammals and seabirds. Environ. Toxicol. Chem. 2004, 23, 2008–2016. [Google Scholar] [CrossRef]
- Das, K.; De Groof, A.; Jauniaux, T.; Bouquegneau, J.-M. Zn, Cu, Cd and Hg binding to metallothioneins in harbor porpoises Phocoena phocoena from the Southern North Sea. BMC Ecol. 2006, 6, 2. [Google Scholar] [CrossRef] [PubMed]
- Pedrero, Z.; Ouerdane, L.; Mounicou, S.; Lobinski, R.; Monperrrus, M.; Amouroux, D. Identification of mercury and other metals complexes with Metallothioneins in Dolphin liver by hydrophilic interaction liquid chromatography with the parallel detection by ICP MS and electrospray hybrid linear/orbital trap MS/MS. Metallomics 2012, 4, 473–479. [Google Scholar] [CrossRef] [PubMed]
- Pollizzi, P.S.; Romero, M.B.; Chiodi Boudet, L.N.; Das, K.; DEnuncio, P.E.; Rodriguez, D.H.; Gerpe, M.S. Metallothioneins pattern during ontogeny of coastal dolphin, Pontoporia blainvillei, from Argentina. Mar. Pollut. Bull. 2014, 80, 275–281. [Google Scholar] [CrossRef]
- Càceres-Saez, I.; Polizzi, P.; Romero, B.; Dellabianca, N.A.; Guevara, S.R.; Goodall, R.N.P.; Cappozzo, H.L.; Gerpe, M. Hepatic and renal metallothionein concentrations in Comerson’s dolphins (Cephalorhynchus commersonii) from Tierra del Fuego, South Atlantic Ocean. Mar. Pollut. Bull. 2016, 108, 263–267. [Google Scholar] [CrossRef] [PubMed]
- Ellman, G.L. Tissue sulfhydryl groups. Arch. Biochem. Biophys. 1959, 82, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Viarengo, A.; Ponzano, E.; Dondero, F.; Fabbri, R. A simple spectrophotometric method for metallothionein evaluation in marine organisms: An application to Mediterranean and Antarctic molluscs. Mar. Environ. Res. 1997, 44, 69–84. [Google Scholar] [CrossRef]
- Liu, J.; Chen, B.; Jefferson, T.A.; Wang, H.; Yang, G. Trace element concentrations, risks and their correlation with metallothionein genes polymorphism: A case study of narrow-ridged finless porpoises (Neophocaena asiaeorientalis) in the East China Sea. Sci. Total Environ. 2017, 575, 628–638. [Google Scholar] [CrossRef] [PubMed]
- Vasàk, M.; Meloni, G. Chemistry and biology of mammalian metallothioneins. J. Biol. Inorg. Chem. 2011, 16, 1067–1078. [Google Scholar] [CrossRef]
- Hidalgo, J.; Chung, R.; Penkowa, M.; Vasàk, M. Structure and function of vertebrate metallothioneins. Met. Ions Life Sci. 2009, 5, 279–317. [Google Scholar]
- Nei, M.; Kumar, S. Molecular Evolution and Phylogenetics; Oxford University Press: New York, NY, USA, 2000. [Google Scholar]
- Tamura, K.; Stecher, G.; Kumar, S. MEGA11: Molecular Evolutionary Genetics Analysis version 11. Mol. Biol. Evol. 2021, 38, 3022–3027. [Google Scholar] [CrossRef]

| Species | Family | Mitochondrion | A Cluster of Mt Genes (Contig) | Free-Intron Mt Gene | Mt Gene with Intron | Mt mRNA |
|---|---|---|---|---|---|---|
| Balaena mysticetus | Balaenidae | NC_005268 | Mt1: SRR17645797, Mt2: AF022117 | |||
| Balaenoptera acutorostrata | Balaemopteridae | NC_005271 | Mt3-Mt2: ATDI01082660-661 | Mt1: ATDI01132427, ATDI01131740, ATDI01081017, Mt2: ATDI01041776 | Mt1: ATDI01082662 Mt4: ATDI01082657 | Mt1: XM_007167588, XR_003622960, XR_003622964, XR_003622677, XM_007193604, XM_007193603, XM_007193605, XM_007193602, XM_007167587, XM_007167582, XM_007198029, XM_007178847, XM_007167583, XM_007164196, XM_007175023 Mt2: XM_007167584 Mt4: XM_028162920 |
| Balaenoptera bonaerensis | Balaenopteridae | NC_006926 | Mt3-Mt2: BAUQ01093692 | Mt1: BAUQ01195848, BAUQ01613210 Mt2: BAUQ01307512 | Mt1: BAUQ01180559 Mt4: BAUQ01234274 | |
| Balaenoptera musculus | Balaenopteridae | MF409242 | Mt4-Mt3: VNFD01000017 | Mt1: VNFD01005770 | Mt1: VNFD01001442 Mt2: VNFC01000015 Mt3: VNFC01000015 | |
| Delphinapterus leucas | Monodontidae | NC_034236 | Mt3-Mt2-Mt1: NQVZ01021645 | Mt1: NQVZ01003203 | Mt1: XM_022555910, XR_002642604 Mt2: XM_022555911 Mt3: GGBT01018098 Mt4: XM_022555909 | |
| Eschrichtius robustus | Eschrichtiidae | NC_005270 | Mt4-Mt3-Mt2-Mt1: RJWN010001895 | Mt1: RJWN010023894, RJWN010012658, NIPP01004159, NIPP01000460, RJWN010001847, NIPP01050325, RJWN010001516, NIPP01000414 | ||
| Eubalaena japonica | Balaenidae | NC_006931 | Mt4-Mt3-Mt2: RJWP010002310 | Mt1: RJWP010029515, RJWP010014455, RJWP011049311, RJWP010044123, RJWP010003859, RJWP010517844, RJWP010045258 | ||
| Globicephala melas | Delphinidae | NC_019441 | Mt3-Mt2: SWEB01012070 Mt1-Mt1: SWEB01015162 | Mt1: personal data | Mt1: personal data Mt2: personal data Mt3: personal data Mt4: personal data | Mt4: XM_030847937 |
| Inia geoffrensis | Iniidae | NC_005276 | Mt4-Mt3-Mt2-Mt1: RJWO010009779 | Mt1: RJWO010024955, RJWO010005555 | ||
| Kogia breviceps | Kogiidae | NC_005272 | Mt3-Mt2: RJWL010036575 | Mt1: RJWL010001100 | Mt1: RJWL010167408 | |
| Lagenorhynchus obliquidens | Delphinidae | NC_035426 | Mt3-Mt2-Mt1: RCWK01007239 | Mt1: RCWK01003005 | Mt4: RCWK01007238 | Mt1: XM_027119875, XM_027092179, XR_003432150 XR_003433512, XR_003429008 Mt2: XM_027119873 Mt4: XM_027119872 |
| Lipotes vexillifer | Lipotidae | NC_007629 | Mt4-Mt3: AUPI01085919, Mt4-Mt3-Mt2-Mt1-Mt1: NW_006786802, Mt2-Mt1-Mt1-Mt1: AUPI01085920 | Mt1: AUPI01016291, AUPI01032811 | Mt2: AUPI01085920 | Mt1: XM_007450307, XM_007459446, XM_007459445, XM_007459251 Mt2: XM_007459444 Mt4: XM_007459443 |
| Megaptera novaengliae | Balaenopteridae | NC_006927 | Mt4-Mt3-Mt2-Mt1: RYZJ01000704 | Mt1: RYZJ01002277 | ||
| Mesoplodon bidens | Ziphiidae | NC_042218 | Mt3-Mt2-Mt1: PVJJ010038290 | Mt1: PVJJ010001248, PVJJ010035753, PVJJ010000716 | Mt4: PVJJ010048532 | |
| Monodon monoceros | Monodontidae | NC_005279 | Mt4-Mt3-Mt2-Mt1: SIHG01006952, Mt3-Mt2-Mt1: PVJE01024091, PVJF01025398 | Mt1: PVJF01009326, PVJE01004367, SIHG01006957, RWIC01000029 | Mt1: XM_029210741, XM_022566857, XR_003793663, XM_029213332, XM_029213320, XM_029213311, XR_003792376, XM_029243937 Mt2: XM_029210730 Mt4: XM_029207165 | |
| Neophocaena asiaeorientalis | Phocoenidae | NC_026456 | Mt3-Mt2: MKKW01002943 | Mt1: XR_003002470, MKKW01009685, MKKW01050072 | Mt1: MKKW01002942, NW_020172079 Mt2, Mt4: NW_020172079 | Mt1: XM_024751546, XM_024752871 Mt2: XM_024751535 Mt4: XM_024751560 |
| Orcinus orca | Delphinidae | NC_023889 | Mt3-Mt2: ANOL02076608, Mt1-Mt1: ANOL02076611, Mt4-Mt3-Mt2: NW_004438720 | Mt1: ANOL02015359, ANOL02053476, ANOL02005809 Mt2: ANOL02033101 | Mt4: ANOL02076604 | Mt1: XR_001119644, XR_001120057, XM_004286377 Mt2: XM_004272467, XM_004286376 Mt3: XM_004286375 Mt4: XM_004286374 |
| Phocoena phocoena | Phocoenidae | NC_005280 | Mt3-Mt2-Mt1: RJWQ010020171, Mt2-Mt1: PKGA01134162 | Mt1: RJWQ010000213, PKGA01141836, RJWQ010001208, PKGA01000712 | Mt4: RJWQ010008745 | |
| Phocoena sinus | Phocoenidae | MZ772969 | Mt4-Mt3-Mt2-Mt1: VOSU01000010 | |||
| Physeter catodon | Physeteridae | KU891394 | Mt3-Mt2: PGGR02120841, Mt1-Mt1-Mt1: PGGR02120842, Mt3-Mt2-Mt1: AWZP01019177, Mt3-Mt2: UEMC01002060 | Mt1: AWZP01108577, AWZP01061491, UEMC01000019, PGGR02098163, AWZP01094651, AWZP01036149 Mt2: AWZP01013965 | Mt2: personal data Mt3: personal data | Mt1: XM_028487398, XM_028487397, XR_002892573, XM_024124849, XR_002890606, XR_002890985, XM_007111896, XM_007106395 Mt2: XR_002891953, XM_024124902, XR_002891953, XM_007104604 |
| Platanista minor | Platanistidae | NC_005275 | Mt1-Mt1: RJWK010077258 | Mt1: RJWK010030898 RJWK010019970 | Mt1: RJWK010077258 Mt2: RJWK010120550 Mt3: RJWK010071068 Mt4: RJWK010018570 | |
| Pontoporia blainvillei | Pontoporiidae | NC_005277 | Mt2-Mt1: RJWI010022586 | Mt1: RJWI010118407 | Mt1: RJWI010124881 Mt3: RJWI010018702 Mt4: RJWI010009362 | |
| Sousa chinensis | Delphinidae | NC_012057 | Mt4-Mt3-Mt2-Mt1: QWLN02017480 | Mt1: QWLN02012546, QWLN02014060 | ||
| Tursiops aduncus | Delphinidae | KF570360 | Mt4-Mt3-Mt2-Mt1: NCQN01002597 | Mt1: NCQN01000091, NCQN01000487 | ||
| Tursiops truncatus | Delphinidae | EU557093 | Mt4-Mt3-Mt2-Mt1: NW_004202941, QUXD02065780, Mt4-Mt3-Mt2: ABRN02426572 | Mt1: ABRN02374863, ABRN02315981, ABRN02301451, QUXD02004646, QUXD02000953, QMGA01000002, MRVK01000157, QUXD02061382, QUXD02003404, QMGA01000469, MRVK01000730, | Mt2: QUXD02065780, ABRN02426572 | Mt1: XM_019951660, XR_002175011, XR_002178769, XR_002172975, XM_004322331 Mt2: XM_004331916, XM_004322332 Mt3: XM004322333 Mt4: XM004322334 |
| Ziphius cavirostris | Ziphiidae | KC776698 | Mt2-Mt1: RJWS010029178 | Mt1: RJWS010650073, RJWS011128550, RJWS010020051 | Mt3: RJWS010262321 Mt4: RJWS010091044 |
| Species | IGR (Mt4-Mt3) | IGR (Mt3-Mt2) | IGR (Mt2-Mt1a) | IGR (Mt1a-Mt1b) | IGR (Mt1b-Mt1c) |
|---|---|---|---|---|---|
| Balaenoptera acutorostrata | 7129 | ||||
| Balaenoptera bonaerensis | 7137 | ||||
| Balaenoptera musculus | 21933 | ||||
| Delphinapterus leucas | 7173 | 5015 | |||
| Eschrichtius robustus | 21903 | 7521 | 4739 | 3141 | |
| Eubalaena japonica | 22674 | 7171 | |||
| Globicephala melas | 7202 | 3068 | |||
| Inia geoffrensis | 19583 | 7232 | 5310 | 3084 | |
| Kogia breviceps | 7195 | ||||
| Lagenorhynchus obliquidens | 7184 | 5026 | |||
| Lipotes vexillifer | 20328 | 7897 | 5309 | 3214 | 3926 |
| Megaptera novaengliae | ±22163 | 7175 | 4740 | 3083 | |
| Mesoplodon bidens | 7196 | 4994 | |||
| Monodon monoceros | 27526 | 7159 | 5005 | ||
| Neophocaena asiaeorientalis | 7201 | 3092 | |||
| Orcinus orca | ±36109 | 7194 | 3055 | ||
| Phocoena phocoena | 7198 | 5137 | 3091 | ||
| Phocoena sinus | 27958 | 7196 | 5090 | ||
| Physeter catodon | 7206 | 4448 | 2729 | 3088 | |
| Platanista minor | 5083 | 3048 | |||
| Pontoporia blainvillei | 2064 | ||||
| Sousa chinensis | 32782 | 7177 | 5016 | ||
| Tursiops aduncus | ±34939 | 7182 | 5027 | ||
| Tursiops truncatus | 23064 | 7186–7219 | 6269 | 3151 | |
| Ziphius cavirostris | 4955 | ||||
| Average ± SD (bp) | 25913.5 ± 5840.65 | 7236.25 ± 174.37 | 4895.71 ± 818.35 | 3068.72 ± 122.72 | 3507 ± 592.55 |
| Min–Max (bp) | 19,583–36,109 | 7129–7571 | 2064–5310 | 2729–3214 | 3088–3926 |
| Species | Mt1 | Mt2 | Mt3 | Mt4 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Exon I | Exon II | Exon III | Exon I | Exon II | Exon III | Exon I | Exon II | Exon III | Exon I | Exon II | Exon III | |
| Balaenoptera acutorostrata | 28 | 66 | 92 | 28 | 66 | 92 | 31 | 66 | 107 | 31 | 66 | 92 |
| Balaenoptera bonaerensis | 28 | 66 | 92 | 28 | 66 | 31 | 66 | 107 | 31 | 66 | 92 | |
| Balaenoptera musculus | 28 | 66 | 92 | 28 | 66 | 92 | 31 | 66 | 107 | 31 | 66 | 92 |
| Delphinapterus leucas | 28 | 66 | 92 | 28 | 66 | 92 | 31 | 66 | 107 | |||
| Eschrichtius robustus | 28 | 66 | 92 | 28 | 66 | 92 | 31 | 66 | 107 | 31 | 66 | 92 |
| Eubalaena japonica | 28 | 66 | 31 | 66 | 107 | 31 | 66 | 92 | ||||
| Globicephala melas | 28 | 66 | 92 | 28 | 66 | 92 | 31 | 66 | 107 | 31 | 66 | 92 |
| Inia geoffrensis | 28 | 66 | 92 | 28 | 66 | 92 | 31 | 66 | 107 | 31 | 66 | 92 |
| Kogia breviceps | 28 | 66 | 92 | 28 | 81 | 31 | 66 | 107 | ||||
| Lagenorhynchus obliquidens | 28 | 66 | 92 | 28 | 66 | 92 | 31 | 66 | 107 | 31 | 66 | 92 |
| Lipotes vexillifer | 28 | 66 | 92 | 28 | 66 | 92 | 31 | 66 | 107 | 31 | 66 | 92 |
| Megaptera novaengliae | 28 | 66 | 92 | 28 | 66 | 92 | 31 | 66 | 107 | 31 | 66 | 92 |
| Mesoplodon bidens | 28 | 28 | 66 | 92 | 31 | 66 | 104 | 31 | 66 | 92 | ||
| Monodon monoceros | 28 | 66 | 92 | 28 | 66 | 92 | 31 | 66 | 107 | 31 | 66 | 92 |
| Neophocaena asiaeorientalis | 28 | 66 | 92 | 28 | 66 | 92 | 31 | 66 | 107 | 31 | 66 | 92 |
| Orcinus orca | 28 | 66 | 92 | 28 | 66 | 92 | 31 | 66 | 107 | 31 | 66 | 92 |
| Phocoena phocoena | 28 | 66 | 92 | 28 | 66 | 92 | 31 | 66 | 107 | 31 | 66 | 92 |
| Phocoena sinus | 31 | 66 | 92 | 28 | 66 | 92 | 31 | 66 | 107 | 31 | 66 | 92 |
| Physeter catodon | 28 | 66 | 92 | 28 | 66 | 92 | 31 | 66 | 104 | |||
| Platanista minor | 28 | 66 | 92 | 28 | 66 | 92 | 31 | 66 | 107 | 31 | 66 | 92 |
| Pontoporia blainvillei | 28 | 66 | 28 | 66 | 92 | 31 | 66 | 107 | 31 | 66 | 92 | |
| Sousa chinensis | 28 | 66 | 92 | 28 | 66 | 92 | 31 | 66 | 107 | 31 | 66 | 92 |
| Tursiops aduncus | 28 | 66 | 92 | 28 | 66 | 92 | 31 | 66 | 107 | 31 | 66 | 92 |
| Tursiops truncatus | 28 | 66 | 92 | 28 | 66 | 92 | 31 | 66 | 107 | 31 | 66 | 92 |
| Ziphius cavirostris | 28 | 66 | 92 | 28 | 92 | 31 | 66 | 104 | 31 | 66 | 92 | |
| Min–Max (bp) | 28–31 | 66 | 92 | 28 | 66–81 | 92 | 31 | 66 | 104–107 | 31 | 66 | 92 |
| Species | Mt1(a)-mt1(b)-mt1(c) | Mt2 | Mt3 | Mt4 | ||||
|---|---|---|---|---|---|---|---|---|
| Intron I | Intron II | Intron I | Intron II | Intron I | Intron II | Intron I | Intron II | |
| Balaenoptera acutorostrata | 611 | 351 | 295 | 175 | 854 | 1642 | 527 | |
| Balaenoptera bonaerensis | 611 | 351 | 295 | 175 | 854 | 1646 | 527 | |
| Balaenoptera musculus | 575(c) | 737(c) | 295 | 209 | 175 | 856 | 1666 | 527 |
| Delphinapterus leucas | 601 | 352 | 295 | 216 | 173 | 854 | ||
| Eschrichtius robustus | 611 | 351 | 295 | 219 | 175 | 857 | 1671 | 527 |
| Eubalaena japonica | 295 | 175 | 855 | 1662 | 527 | |||
| Globicephala melas | 611(a)-578(b) | 350(a)-710(b) | 295 | 216 | 173 | 853 | 1171 | 526 |
| Inia geoffrensis | 611 | 344 | 291 | 215 | 175 | 849 | 1368 | 527 |
| Kogia breviceps | 577 | 351 | 295 | 221 | 181 | 854 | ||
| Lagenorhynchus obliquidens | 611 | 350 | 295 | 216 | 173 | 851 | 1308 | 526 |
| Lipotes vexillifer | 608(a)-721(b)-578(c) | 350(a)-753(b)-708(c) | 295 | 214 | 175 | 853 | 1436 | 527 |
| Megaptera novaengliae | 611 | 351 | 295 | 205 | 175 | 854 | 1608 | 526 |
| Mesoplodon bidens | 294 | 215 | 175 | 861 | 1395 | 526 | ||
| Monodon monoceros | 612 | 355 | 295 | 216 | 173 | 854 | 1395 | 527 |
| Neophocaena asiaeorientalis | 610 | 350 | 295 | 217 | 173 | 850 | 1391 | 527 |
| Orcinus orca | 611 | 350 | 295 | 216 | 173 | 853 | 1309 | 526 |
| Phocoena phocoena | 610 | 350 | 295 | 217 | 173 | 849 | 1393 | 527 |
| Phocoena sinus | 601 | 352 | 295 | 217 | 173 | 849 | 1390 | 527 |
| Physeter macrocephalus | 611(a)-713(b)-716(c) | 353(a)-359(b)-359(c) | 295 | 216 | 176 | 857 | ||
| Platanista minor | 594 | 356 | 297 | 224 | 176 | 803 | 1399 | 529 |
| Pontoporia blainvillei | 611 | 295 | 215 | 175 | 861 | 1357 | 520 | |
| Sousa chinensis | 611 | 351 | 294 | 216 | 173 | 852 | 1308 | 527 |
| Tursiops aduncus | 611 | 350 | 295 | 216 | 173 | 852 | 1309 | 527 |
| Tursiops truncatus | 611(a)-612(b) | 350(a)-714(b) | 295 | 216 | 173 | 1098 | 1309 | 527 |
| Ziphius cavirostris | 608 | 351 | 175 | 861 | 1393 | 526 | ||
| Average ± SD (bp) | 607.30 ± 7.95(a) 682 ± 60.75(b) 611.75 ± 69.51(c) | 351 ± 2.20(a) 608.67 ± 217.1(b) 628.50 ± 180.15(c) | 294.85 ± 0.92 | 218.88 ± 14.13 | 174.38 ± 1.75 | 861.38 ± 49.40 | 1425.98 ± 142.38 | 548.130 ± 105 |
| Min–Max (bp) | 577–612(a) 612–721(b) 575–716(c) | 344–356(a) 359–753(b) 359–737(c) | 291–297 | 205–283 | 173–181 | 803–1098 | 1171–1671 | 520–1041 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leignel, V.; Pillot, L.; Gerpe, M.S.; Caurant, F. Assessment of Knowledge on Metal Trace Element Concentrations and Metallothionein Biomarkers in Cetaceans. Toxics 2023, 11, 454. https://doi.org/10.3390/toxics11050454
Leignel V, Pillot L, Gerpe MS, Caurant F. Assessment of Knowledge on Metal Trace Element Concentrations and Metallothionein Biomarkers in Cetaceans. Toxics. 2023; 11(5):454. https://doi.org/10.3390/toxics11050454
Chicago/Turabian StyleLeignel, Vincent, Louis Pillot, Marcela Silvia Gerpe, and Florence Caurant. 2023. "Assessment of Knowledge on Metal Trace Element Concentrations and Metallothionein Biomarkers in Cetaceans" Toxics 11, no. 5: 454. https://doi.org/10.3390/toxics11050454
APA StyleLeignel, V., Pillot, L., Gerpe, M. S., & Caurant, F. (2023). Assessment of Knowledge on Metal Trace Element Concentrations and Metallothionein Biomarkers in Cetaceans. Toxics, 11(5), 454. https://doi.org/10.3390/toxics11050454






