Distribution and Characterization of Typical Antibiotics in Water Bodies of the Yellow River Estuary and Their Ecological Risks
Abstract
1. Introduction
2. Materials and Methods
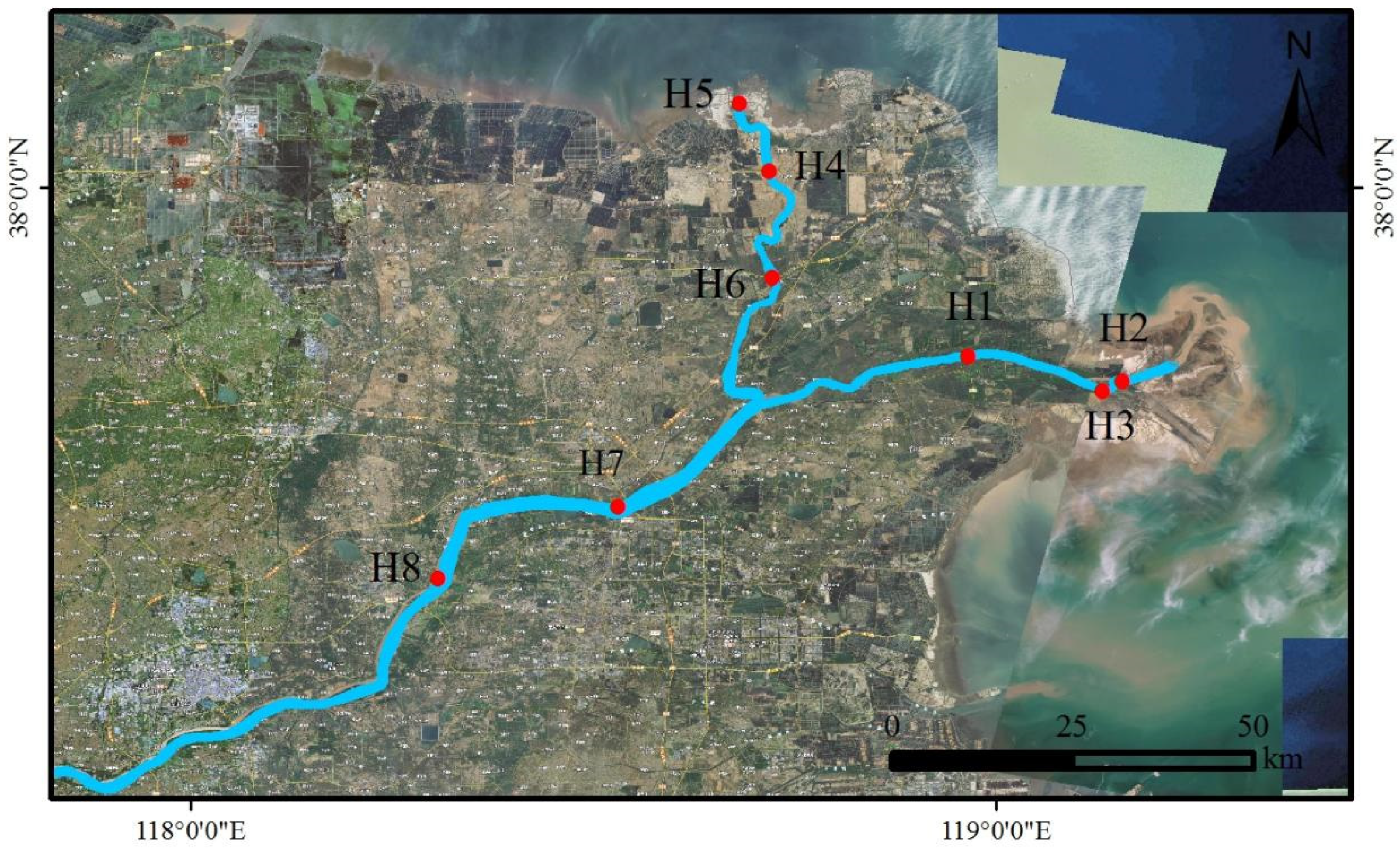
2.1. Sample Collection
2.2. Advanced Analysis Instruments and Reagents
2.3. Sample Treatment
2.4. Instrumental Analysis
2.5. Ecological Risk Assessment
2.6. Data Analysis
3. Results and Discussion
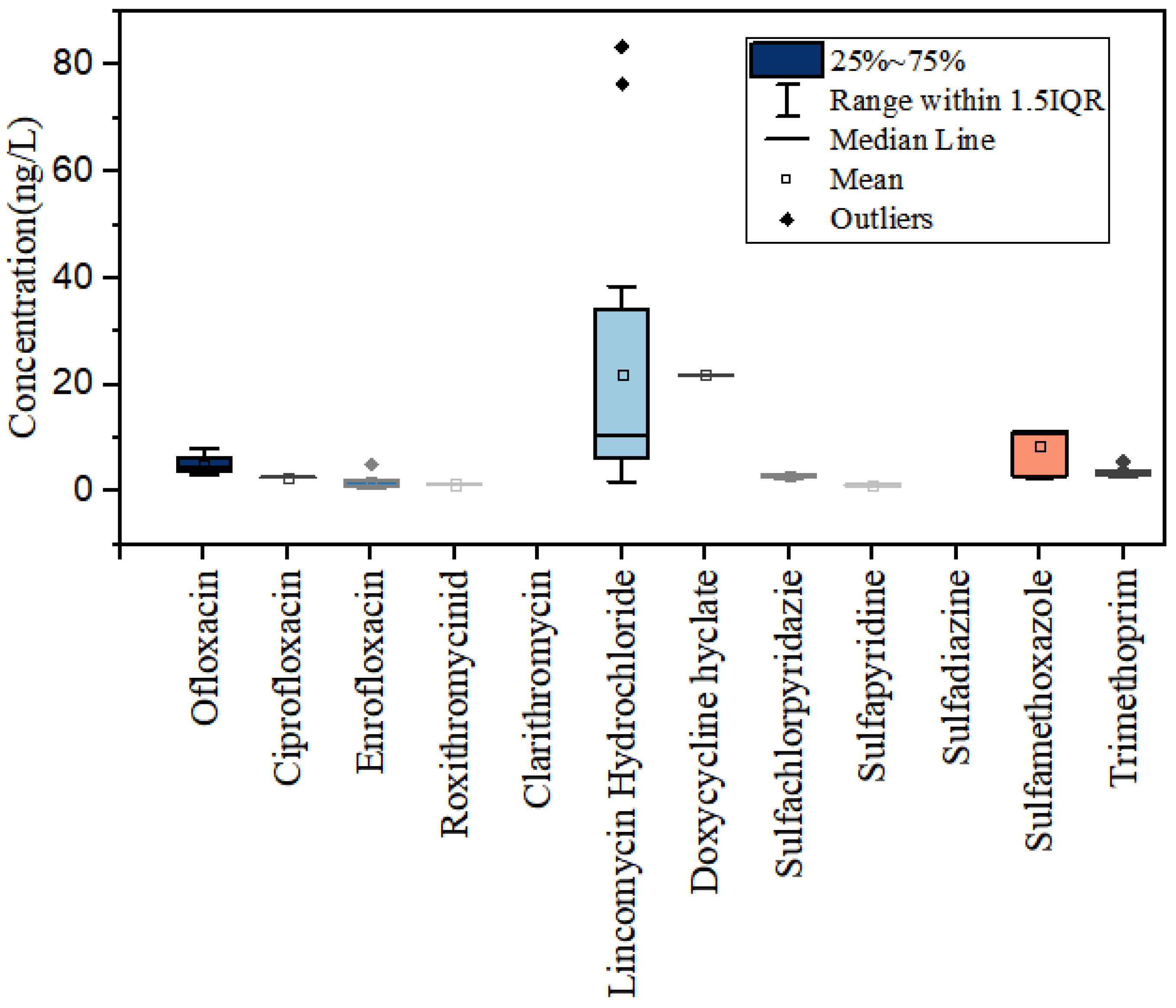
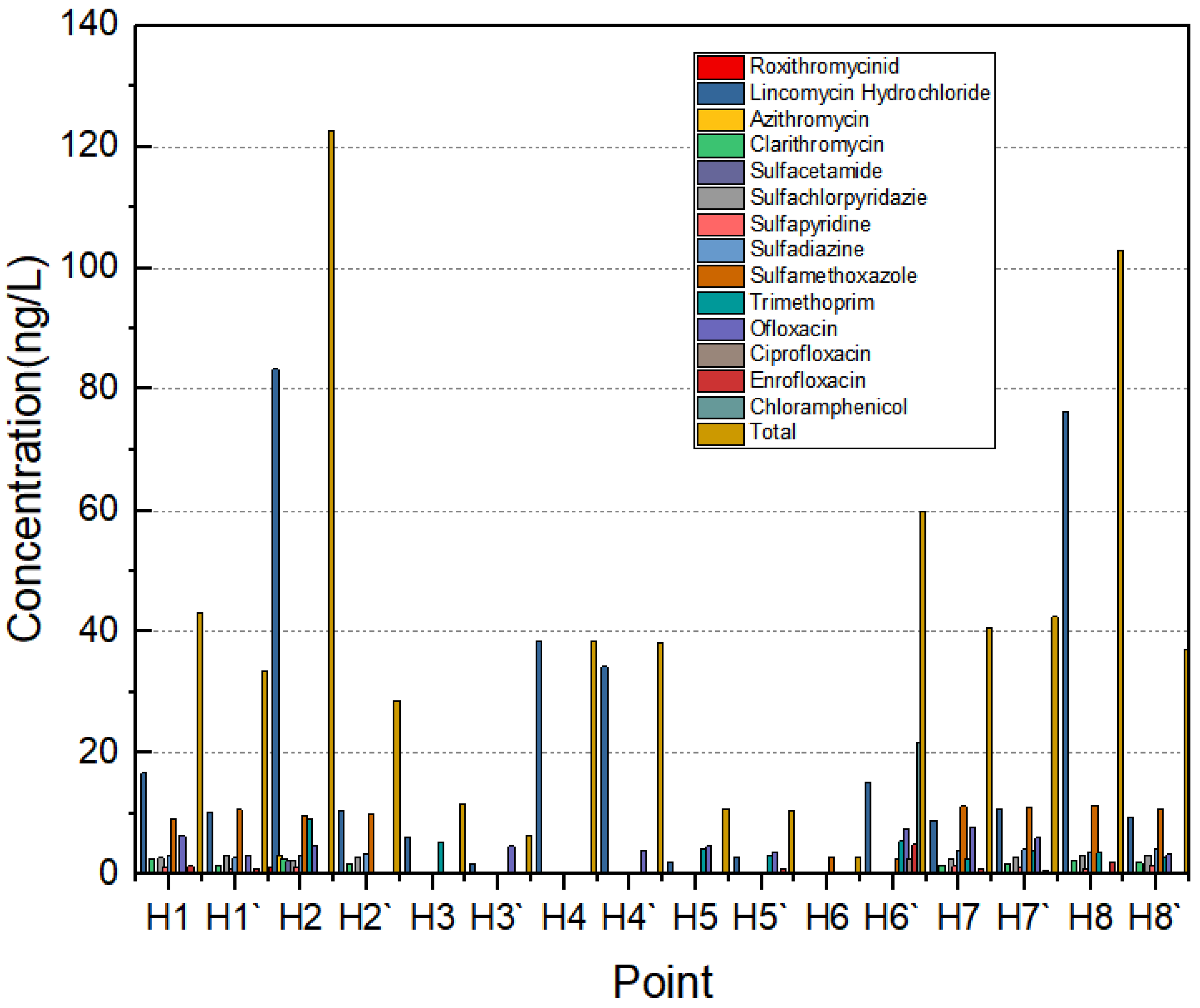
3.1. Concentration Levels of Antibiotics in the Water Bodies of the Yellow River Estuary
3.2. Spatial Distribution of Antibiotics
3.3. Ecological Risk Assessment
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Xi, J.P. Speech at the symposium on ecological protection and high: Quality development of the Yellow River Basin. China Water Resour. 2019, 20, 1–3. (In Chinese) [Google Scholar]
- Li, J.; Peng, K.; Hao, G. Research progress on quantification and control of non-point source pollution load in the Yellow River Basin. Water Resour. Prot. 2021, 37, 90–102. (In Chinese) [Google Scholar]
- Ministry of Ecology and Environment. The 14th Five-Year Plan for Ecological and Environmental Monitoring. Available online: https://www.mee.gov.cn/xxgk2018/xxgk/xxgk03/202201/t20220121_967927.html (accessed on 28 December 2021).
- General Office of the Ministry of Ecology and Environment. Public Consultation on the List of Key Control New Pollutants (2022 Version) (Draft for Comments). Available online: https://www.mee.gov.cn/xxgk2018/xxgk/xxgk06/202209/t20220927_995054.html (accessed on 27 September 2022).
- Kim, K.-R.; Owens, G.; Kwon, S.-I.; So, K.-H.; Lee, D.-B.; Ok, Y.S. Occurrence and Environmental Fate of Veterinary Antibiotics in the Terrestrial Environment. Water Air Soil Pollut. 2011, 214, 163–174. [Google Scholar] [CrossRef]
- Zhang, J.; Ge, H.; Shi, J.; Tao, H.; Li, B.; Yu, X.; Zhang, M.; Xu, Z.; Xiao, R.; Li, X. A tiered probabilistic approach to assess antibiotic ecological and resistance development risks in the fresh surface waters of China. Ecotoxicol. Environ. Saf. 2022, 243, 114018. [Google Scholar] [CrossRef]
- Zhang, Q.; Xin, Q.; Zhu, J. Progress of research on the status of antibiotic pollution in major waters of China and its ecological effects. Environ. Chem. 2014, 33, 1075–1083. [Google Scholar]
- Peng, Q.; Wang, W.Z.; Xu, W.H. Ecological Risk Assessment of Tetracycline Antibiotics in Livestock Manure and Vegetable Soil of Chongqing. Huan Jing Ke Xue 2020, 41, 4757–4766. [Google Scholar] [PubMed]
- Wang, T.; Zhang, W.; Li, L.; Zhang, M.; Liao, G.; Wang, D. Distribution characteristics and risk assessment of antibiotics and polycyclic aromatic hydrocarbons in sediments from Baiyangdian dredging demonstration area. Environ. Sci. 2021, 42, 5303–5311. [Google Scholar] [CrossRef]
- Zhang, Q.Q.; Ying, G.G.; Pan, C.G.; Liu, Y.S.; Zhao, J.L. Comprehensive evaluation of antibiotics emission and fate in the river basins of China: Source analysis, multimedia modeling, and linkage to bacterial resistance. Environ. Sci. Technol. 2015, 49, 6772–6782. [Google Scholar] [CrossRef]
- Lei, K.; Zhu, Y.; Chen, W.; Pan, H.Y.; Cao, Y.X.; Zhang, X.; Guo, B.B.; Sweetman, A.; Lin, C.Y.; Ouyang, W.; et al. Spatial and seasonal variations of antibiotics in river waters in the Haihe River Catchment in China and ecotoxicological risk assessment. Environ. Int. 2019, 130, 104919. [Google Scholar] [CrossRef]
- Huang, F.; An, Z.; Moran, M.J.; Liu, F. Recognition of typical antibiotic residues in environmental media related to groundwater in China (2009−2019). J. Hazard. Mater. 2020, 399, 122813. [Google Scholar] [CrossRef]
- Ting, R.U.A.N.; Guibin, J.I.A.N.G. Basic theory and methods for the discovery of novel environmental organic pollutants. Proc. Chin. Acad. Sci. 2020, 35, 1328–1336. [Google Scholar] [CrossRef]
- Li, X.; Zhang, R.; Tian, T.; Shang, X.; Du, X.; He, Y.; Matsuura, N.; Luo, T.; Wang, Y.; Chen, J.; et al. Screening and ecological risk of 1200 organic micropollutants in Yangtze Estuary water. Water Res. 2021, 201, 117341. [Google Scholar] [CrossRef]
- Bielen, A.; Šimatović, A.; Kosić-Vukšić, J.; Senta, I.; Ahel, M.; Babić, S.; Jurina, T.; Plaza, J.J.G.; Milaković, M.; Udiković-Kolić, N. Negative environmental impacts of antibiotic-contaminated effluents from pharmaceutical industries. Water Res. 2017, 126, 79–87. [Google Scholar] [CrossRef]
- Klaus, K. Antibiotics in the aquatic environment—A review—Part I. Chemosphere 2008, 75, 417–434. [Google Scholar]
- Zhao, K.; Wang, Q.; Qian, S.; Li, F. Spatial and temporal distribution characteristics of antibiotics and heavy metals in the Yitong River basin and ecological risk assessment. Sci. Rep. 2023, 13, 4202. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Wang, Z.; Wang, X.L.; Li, Z.; Yang, C.; Li, E.H.; Wei, H.M. Evaluation of antibiotic pollution in surface water environment and its ecological risk in a typical region of China. Environ. Sci. 2019, 40, 2094–2100. [Google Scholar] [CrossRef]
- Zhao, S.; Liu, X.; Cheng, D.; Liu, G.; Liang, B.; Cui, B.; Bai, J. Temporal–spatial variation and partitioning prediction of antibiotics in surface water and sediments from the intertidal zones of the Yellow River Delta, China. Sci. Total Environ. 2016, 569–570, 1350–1358. [Google Scholar] [CrossRef] [PubMed]
- Lü, D.Y.; Yu, C.; Zhuo, Z.J.; Meng, S.R.; Liu, S.B. The distribution and ecological risks of antibiotics in surface water in key cities along the lower reaches of the Yellow River: A case study of Kaifeng City, China. China Geol. 2022, 5, 411–420. (In Chinese) [Google Scholar] [CrossRef]
- Shen, L.; Zhang, L.; Qin, S.; Yao, B.; Cui, J.S. Characteristics of quinolone antibiotic contamination in Baiyangdian and its correlation with environmental factors. J. Environ. Sci. 2019, 39, 3888–3897. [Google Scholar] [CrossRef]
- Li, J.; Wang, Y.; Dong, Y.; Wang, M.; Zhao, Q.; Zhou, Y.; Ding, H.; Yin, L.; Cao, Y.; Xie, J.; et al. Distribution characteristics and ecological risk assessment of typical antibiotics in Yuan River water bodies in Poyang Lake basin. J. Ecotoxicol. 2022, 17, 563–574. (In Chinese) [Google Scholar]
- Wu, W.; Qi, M.; Zhang, Z.; Lan, Q.; Hu, H. Study on the current situation and detection methods of sulfonamide antibiotics contamination. Environ. Sci. Manag. 2022, 47, 121–126. (In Chinese) [Google Scholar]
- Duong, H.A.; Pham, N.H.; Nguyen, H.T.; Hoang, T.T.; Pham, H.V.; Pham, V.C.; Berg, M.; Giger, W.; Alder, A.C. Occurrence, fate and antibiotic resistance of fluoroquinolone antibacterials in hospital wastewaters in Hanoi, Vietnam. Chemosphere 2008, 72, 968–973. [Google Scholar] [CrossRef] [PubMed]
- Figueroa, R.A.; MacKay, A.A. Sorption of oxytetracycline to iron oxides and iron oxide-rich soils. Environ. Sci. Technol. 2005, 39, 6664–6671. [Google Scholar] [CrossRef]
- Zhu, T.; Zhou, M.; Yang, S.K.; Wang, Z.Z.; Wang, R.Z.; Wang, W.K.; Zhao, Y.Q. Characteristics of antibiotic distribution and ecological risk assessment in the Shaanxi section of the Weihe River. People’s Yellow River 2018, 40, 85–91. (In Chinese) [Google Scholar]
- Backhaus, T.; Scholze, M.; Grimme, L.H. The single substance and mixture toxicity of quinolones to the bioluminescent bacterium Vibrio fischeri. Aquat. Toxicol. 2000, 49, 49–61. [Google Scholar] [CrossRef]
- Huang, F.; Zou, S.; Deng, D.; Lang, H.; Liu, F. Antibiotics in a typical karst river system in China: Spatiotemporal variation and environmental risks. Sci. Total Environ. 2019, 650 Pt 1, 1348–1355. [Google Scholar] [CrossRef]
- Goyne, K.W.; Chorover, J.; Kubicki, J.D.; Zimmerman, A.R.; Brantley, S.L. Sorption of the antibiotic ofloxacin to mesoporous and nonporous alumina and silica. J. Colloid Interface Sci. 2005, 283, 160–170. [Google Scholar] [CrossRef]
- Brain, R.A.; Johnson, D.J.; Richards, S.M.; Sanderson, H.; Sibley, P.K.; Solomon, K.R. Effects of 25 pharmaceutical compounds to Lemna gibba using a seven-day static-renewal test. Environ. Toxicol. Chem. 2004, 23, 371–382. [Google Scholar] [CrossRef]
- Wu, T.Y.; Li, J.; Yang, A.J.; Li, Y.C.; Chen, Y.; He, Q.; Ma, K.; Hu, X.; Wang, B.; Ai, J.; et al. Characteristics and Risk Assessment of Antibiotic Contamination in Chishui River Basin, Guizhou Province, China. Huan Jing Ke Xue 2022, 43, 210–219. [Google Scholar]
- Robinson, A.A.; Belden, J.B.; Lydy, M.J. Toxicity of fluoroquinolone antibiotics to aquatic organisms. Environ. Toxicol. Chem. 2005, 24, 423–430. [Google Scholar] [CrossRef]
- Xu, L.; Ye, X.; Hao, G. Characterization and ecological risk evaluation of typical antibiotic pollution in surface waters of Campsis River. Mod. Agric. Sci. Technol. 2020, 765, 180–183+187. (In Chinese) [Google Scholar]
- Ferrari, B.; Mons, R.; Vollat, B.; Fraysse, B.; Paxēaus, N.; Giudice, R.L.; Pollio, A.; Garric, J. Environmental risk assessment of six human pharmaceuticals: Are the current environmental risk assessment procedures sufficient for the protection of the aquatic environment? Environ. Toxicol. Chem. 2004, 23, 1344–1354. [Google Scholar] [CrossRef] [PubMed]
- Lützhøft, H.C.H.; Halling-Sørensen, B.; Jørgensen, S.E. Algal toxicity of antibacterial agents applied in Danish fish farming. Arch. Environ. Contam. Toxicol. 1999, 36, 1–6. [Google Scholar] [CrossRef]
- Zhao, T.; Chen, Y.; Han, W.; He, Y. The contamination characteristics and ecological risk assessment of typical antibiotics in the upper reaches of the Dongjiang River. Ecol. Environ. Sci. 2016, 25, 1707–1713. [Google Scholar]
- Zhao, F.; Gao, H.; Zhang, K. Fugacity and risk assessment of antibiotics in typical river waters in China. Environ. Pollut. Prev. 2021, 43, 94–102. [Google Scholar] [CrossRef]
- Li, Y.; Fang, J.; Yuan, X.; Chen, Y.; Yang, H.; Fei, X. Distribution characteristics and ecological risk assessment of tetracyclines pollution in the Weihe river, China. Int. J. Environ. Res. Public Health 2018, 15, 1803. [Google Scholar] [CrossRef]
- Wu, X.Y.; Zou, H.; Zhu, R.; Jingguo, W. Characterization and ecological risk assessment of antibiotic pollution in the waters of Lake Taiko Gonghu Bay. Environ. Sci. 2016, 37, 4596–4604. (In Chinese) [Google Scholar] [CrossRef]
- Wang, T.; Yang, Z.F.; Chen, Y.H.; Yaoyao, Z.; Ranran, S.; Ying, X.; Mengting, Z. Ecological risk assessment of sulfonamide antibiotics in surface water. J. Ecol. Environ. 2016, 25, 1508–1514. [Google Scholar] [CrossRef]

| Lakes (Year of Survey) | Antibiotic Concentration | Average Concentration |
|---|---|---|
| Yellow River Estuary (2022) | ND~415.53 | 25.97 |
| Jinan section of the lower Yellow River (2022) | ND~13,462 | 373.94 |
| Yellow River Delta Section (2019) | ND~256.6 | 15.09 |
| Yellow River Delta intertidal zone (2016) | ND~82.94 | 10.37 |
| Canal in Kaifeng, Henan, a key city on the lower reaches of the Yellow River (2022) | ND~12,224.99 | 815.00 |
| Weihe River (2018) | ND~573.26 | 13.98 |
| Antibiotics | PNEC | References |
|---|---|---|
| Ofloxacin | 21~17,400 | [27,28,29] |
| Ciprofloxacin | 2~30,000 | [30,31] |
| Enrofloxacin | 28.8~49 | [32,33] |
| Roxithromycinid | 4.3~10,000 | [34] |
| Clarithromycin | 2 | [22,35] |
| Lincomycin Hydrochloride | 50~50,000 | [28,29,36] |
| Doxycycline hyclate | 131 | [37] |
| Sulfachlorpyridazie | 2330~1,720,000 | [38,39] |
| Sulfapyridine | 460~5280 | [38,39] |
| Sulfadiazine | 107.394~135 | [28,29,38,40] |
| Sulfamethoxazole | 27~4674 | [28,29,38,40] |
| Trimethoprim | 29~255.516 | [28,29,38,40] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, J.; Yan, Z.; Qiao, Y.; Liu, D.; Feng, C.; Bai, Y. Distribution and Characterization of Typical Antibiotics in Water Bodies of the Yellow River Estuary and Their Ecological Risks. Toxics 2023, 11, 400. https://doi.org/10.3390/toxics11050400
Wang J, Yan Z, Qiao Y, Liu D, Feng C, Bai Y. Distribution and Characterization of Typical Antibiotics in Water Bodies of the Yellow River Estuary and Their Ecological Risks. Toxics. 2023; 11(5):400. https://doi.org/10.3390/toxics11050400
Chicago/Turabian StyleWang, Jindong, Zhenfei Yan, Yu Qiao, Daqing Liu, Chenglian Feng, and Yingchen Bai. 2023. "Distribution and Characterization of Typical Antibiotics in Water Bodies of the Yellow River Estuary and Their Ecological Risks" Toxics 11, no. 5: 400. https://doi.org/10.3390/toxics11050400
APA StyleWang, J., Yan, Z., Qiao, Y., Liu, D., Feng, C., & Bai, Y. (2023). Distribution and Characterization of Typical Antibiotics in Water Bodies of the Yellow River Estuary and Their Ecological Risks. Toxics, 11(5), 400. https://doi.org/10.3390/toxics11050400








