Antioxidant Potential of Mung Bean (Vigna radiata) Albumin Peptides Produced by Enzymatic Hydrolysis Analyzed by Biochemical and In Silico Methods
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Mung Bean Albumin Extraction
2.3. Gel Electrophoresis
2.3.1. Sodium Dodecyl Sulfate–Polyacrylamide Gel Electrophoresis (SDS-PAGE)
2.3.2. Native Blue–Polyacrylamide Gel Electrophoresis (Native Blue-PAGE)
2.4. In Silico Hydrolysis of Mung Bean Albumin Sequences
2.5. Enzymatic Hydrolysis
2.6. Antioxidant Assays
2.6.1. ABTS Radical Scavenging Assay
2.6.2. Ferrous Ion Chelation Assay
2.6.3. Oxygen Radical Absorbance Capacity (ORAC) Assay
2.7. Peptide Sequencing
2.8. Statistical Analysis
3. Results
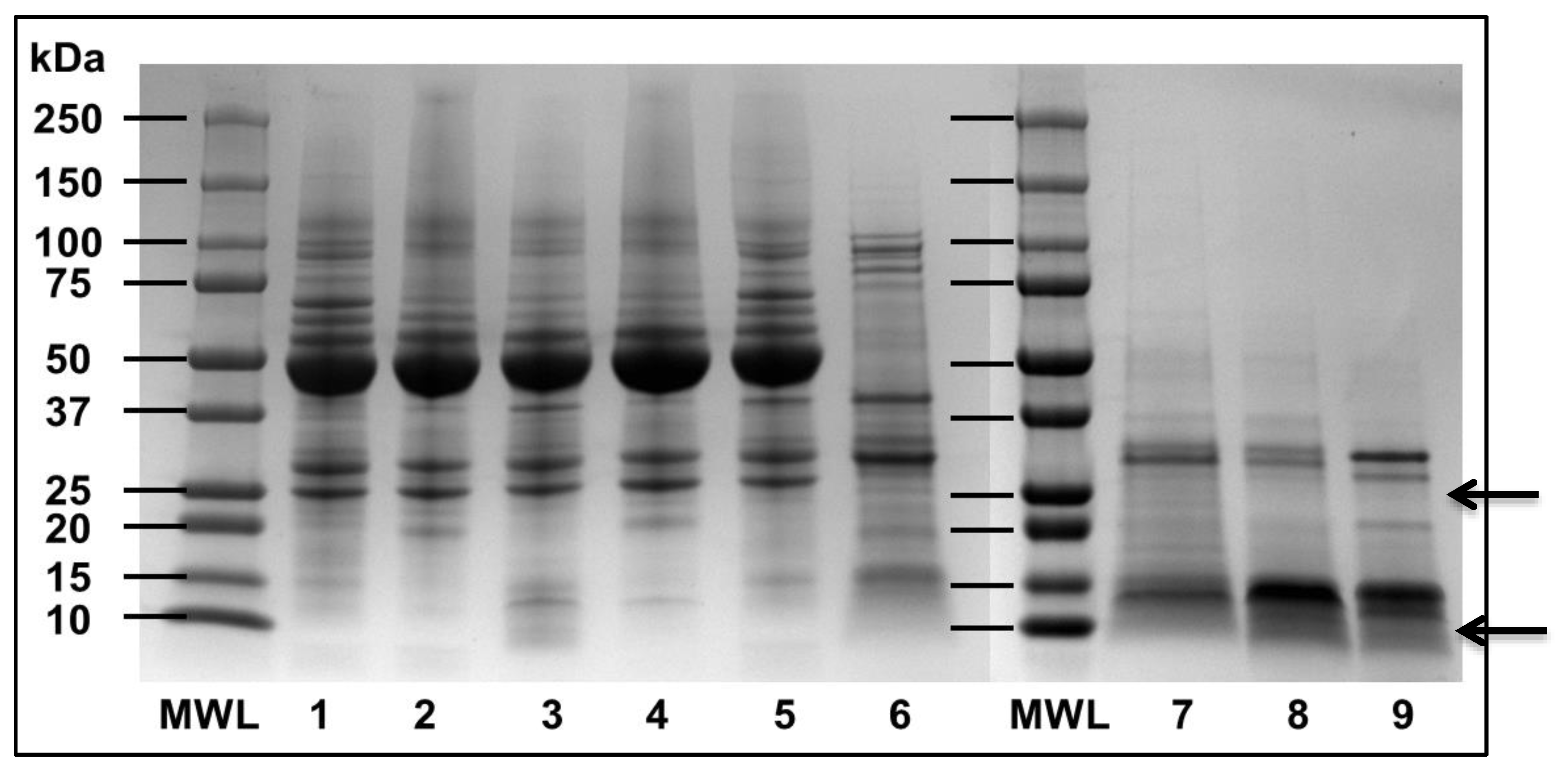
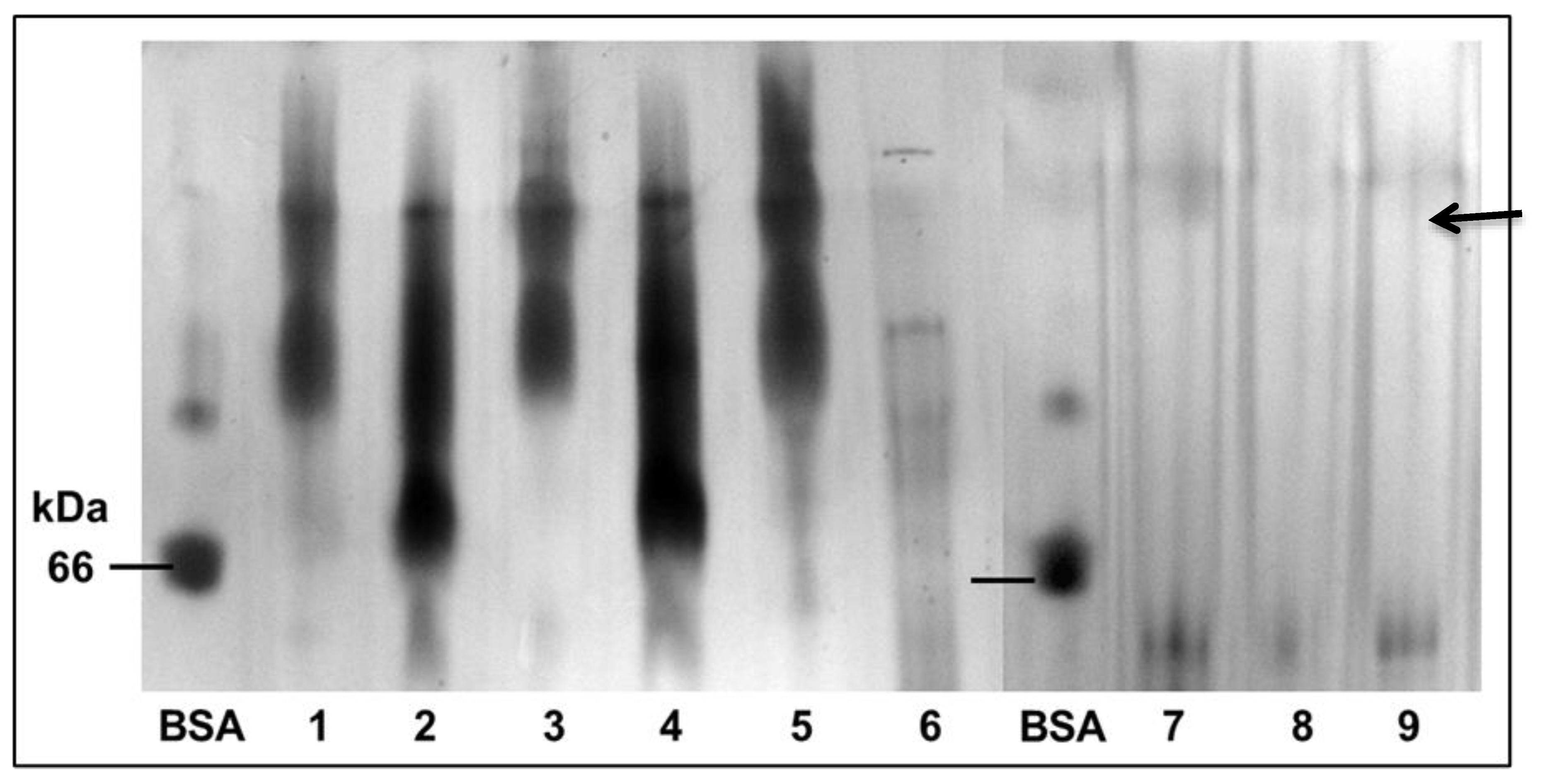
3.1. Mung Bean Albumin Profiles
3.2. Potentially Antioxidant Peptides from Mung Bean Albumins by In Silico Hydrolysis
3.3. Antioxidant Activity of Mung Bean Albumin Hydrolysates and Peptide Sequencing
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Oo, Z.Z.; Ko, L.T.; Than, S.S. Physico-chemical properties of extracted mung bean protein concentrate. Am. J. Food Sci. Technol. 2017, 5, 265–269. [Google Scholar]
- Gunarti, D.R.; Rahmi, H.; Sadikin, M. Isolation and purification of thiamine binding protein from mung bean. HAYATI J. Biosci. 2013, 20, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Tang, D.; Dong, Y.; Ren, H.; Li, L.; He, C. A review of phytochemistry, metabolite changes, and medicinal uses of the common food mung bean and its sprouts (Vigna radiata). Chem. Cent. J. 2014, 8, 4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mendoza, E.M.; Adachi, M.; Bernardo, A.E.; Utsumi, S. Mungbean [Vigna radiata (L.) Wilczek] globulins: Purification and characterization. J. Agric. Food Chem. 2001, 49, 1552–1558. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Jiang, L.; Li, Y.; Liu, Q.; Wang, S.; Sui, X. Optimization of extraction process of protein isolate from mung bean. Procedia Eng. 2011, 15, 5250–5258. [Google Scholar] [CrossRef] [Green Version]
- Brishti, F.H.; Zarei, M.; Muhammad, S.K.S.; Ismail-Fitry, M.R.; Shukri, R.; Saari, N. Evaluation of the functional properties of mung bean protein isolate for development of textured vegetable protein. Int. Food Res. J. 2017, 24, 1595–1605. [Google Scholar]
- Shi, Z.; Yao, Y.; Zhu, Y.; Ren, G. Nutritional composition and antioxidant activity of twenty mung bean cultivars in China. Crop J. 2016, 4, 398–406. [Google Scholar] [CrossRef] [Green Version]
- Deraz, S.F.; Khalil, A.A. Strategies to improve protein quality and reduce antinutritional factors in mung bean. Food 2008, 2, 25–38. [Google Scholar]
- Anwar, F.; Iqbal, Z.; Sultana, B.; Iqbal, S.; Saari, N. Effects of extraction system on antioxidant attributes of mungbean [Vigna radiata (L.) Wilczek]. Int. J. Food Prop. 2013, 16, 527–535. [Google Scholar] [CrossRef]
- Varma, A.; Mishra, S.P.; Tripathi, A.; Shukla, U.K. Biochemical composition and storage protein profiling of mungbean (Vigna radiata L. wilczek) cultivars. J. Pharmacog. Phytochem. 2018, 7, 708–713. [Google Scholar]
- Gupta, N.; Srivastava, N.; Bhagyawant, S.S. Vicilin—A major storage protein of mungbean exhibits antioxidative potential, antiproliferative effects and ACE inhibitory activity. PLoS ONE 2018, 13, e0191265. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Tse, Y.C.; Hinz, G.; Robinson, D.G.; Jiang, L. Storage globulins pass through the Golgi apparatus and multivesicular bodies in the absence of dense vesicle formation during early stages of cotyledon development in mung bean. J. Exp. Bot. 2012, 63, 1367–1380. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yi-Shen, Z.; Shuai, S.; Fitzgerald, R. Mung bean proteins and peptides: Nutritional, functional and bioactive properties. Food Nutr. Res. 2018, 62, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Cao, Y.; Geng, F. Genome-wide identification and comparative analysis of albumin family in vertebrates. Evol. Bioinform. Online 2017, 13, 1176934317716089. [Google Scholar] [CrossRef]
- Moreno, F.J.; Clemente, A. 2S Albumin storage proteins: What makes them food allergens? Open Biochem. J. 2008, 2, 16–28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Galbas, M.; Porzucek, F.; Woźniak, A.; Slomski, R.; Selwet, M. Isolation of low-molecular albumins of 2S fraction from soybean (Glycine max (L.) Merrill). Acta Biochim. Pol. 2013, 60, 107–110. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scarafoni, A.; Gualtieri, E.; Barbiroli, A.; Carpen, A.; Negri, A.; Duranti, M. Biochemical and functional characterization of an albumin protein belonging to the hemopexin superfamily from Lens culinaris seeds. J. Agric. Food Chem. 2011, 59, 9637–9644. [Google Scholar] [CrossRef]
- Kortt, A.A. Isolation and characterisation of a major seed albumin. Int. J. Peptide Prot. Res. 1986, 28, 613–619. [Google Scholar] [CrossRef]
- Kang, Y.J.; Kim, S.K.; Kim, M.Y.; Lestari, P.; Kim, K.H.; Ha, B.K.; Jun, T.H.; Hwang, W.J.; Lee, T.; Lee, J.; et al. Genome sequence of mungbean and insights into evolution within Vigna species. Nat. Commun. 2014, 5, 5443. [Google Scholar] [CrossRef] [Green Version]
- Yamazaki, T.; Takaoka, M.; Katoh, E.; Hanada, K.; Sakita, M.; Sakata, K.; Nishiuchi, Y.; Hirano, H. A possible physiological function and the tertiary structure of a 4-kDa peptide in legumes. Eur. J. Biochem. 2003, 270, 1269–1276. [Google Scholar] [CrossRef]
- Kou, X.; Gao, J.; Zhang, Z.; Wang, H.; Wang, X. Purification and identification of antioxidant peptides from chickpea (Cicer arietinum L.) albumin hydrolysates. LWT Food Sci. Technol. 2013, 50, 591–598. [Google Scholar] [CrossRef]
- Xue, Z.; Wen, H.; Zhai, L.; Yu, Y.; Li, Y.; Yu, W.; Cheng, A.; Wang, C.; Kou, X. Antioxidant activity and anti-proliferative effect of a bioactive peptide from chickpea (Cicer arietinum L.). Food Res. Int. 2015, 77, 75–81. [Google Scholar] [CrossRef]
- Benedé, S.; Molina, E. Chicken egg proteins and derived peptides with antioxidant properties. Foods 2020, 9, 735. [Google Scholar] [CrossRef] [PubMed]
- Lule, V.K.; Garg, S.; Pophaly, S.D.; Tomar, S.K. Potential health benefits of lunasin: A multifaceted soy-derived bioactive peptide. J. Food Sci. 2015, 80, R485–R494. [Google Scholar] [CrossRef]
- Hernández-Ledesma, B.; Hsieh, C.; De Lumen, B.O. Biochemical and biophysical research communications antioxidant and anti-inflammatory properties of cancer preventive peptide lunasin in RAW 264.7 macrophages. Biochem. Biophys. Res. Commun. 2009, 390, 803–808. [Google Scholar] [CrossRef]
- Budseekoad, S.; Yupanqui, C.T.; Sirinupong, N.; Alashi, A.M.; Aluko, R.E.; Youravong, W. Structural and functional characterization of calcium and iron-binding peptides from mung bean protein hydrolysate. J. Funct. Foods 2018, 49, 333–341. [Google Scholar] [CrossRef]
- Lapsongphon, N.; Yongsawatdigul, J. Production and purification of antioxidant peptides from a mungbean meal hydrolysate by Virgibacillus sp. SK37 proteinase. Food Chem. 2013, 141, 992–999. [Google Scholar] [CrossRef]
- Du, M.; Xie, J.; Gong, B.; Xu, X.; Tang, W.; Li, X.; Li, C.; Xie, M. Extraction, physicochemical characteristics and functional properties of Mung bean protein. Food Hydrocoll. 2018, 76, 131–140. [Google Scholar] [CrossRef]
- Singh, D.K.; Rao, A.S.; Singh, R.; Jambunathan, R. Amino acid composition of storage proteins of a promising chickpea (Cicer arietinum L) cultivar. J. Sci. Food Agric. 1988, 43, 373–379. [Google Scholar] [CrossRef] [Green Version]
- De Souza Rocha, T.; Hernandez, L.M.R.; Chang, Y.K.; de Mejía, E.G. Impact of germination and enzymatic hydrolysis of cowpea bean (Vigna unguiculata) on the generation of peptides capable of inhibiting dipeptidyl peptidase IV. Food Res. Int. 2014, 64, 799–809. [Google Scholar] [CrossRef]
- Daniel, H. Molecular and integrative physiology of intestinal peptide transport. Annu. Rev. Physiol. 2004, 66, 361–384. [Google Scholar] [CrossRef] [PubMed]
- Mojica, L.; de Mejía, E.G. Optimization of enzymatic production of anti-diabetic peptides from black bean (Phaseolus vulgaris L.) proteins, their characterization and biological potential. Food Funct. 2016, 7, 713–727. [Google Scholar] [CrossRef] [PubMed]
- Held, P.; Brescia, P.J. Determining Antioxidant Potential Using an Oxygen Radical Absorbance Capacity (ORAC) Assay with SynergyTM H4. Available online: https://www.labmanager.com/products-in-action/determining-antioxidant-potential-using-an-oxygen-radical-absorbance-capacity-orac-assay-10182 (accessed on 1 June 2020).
- Grancieri, M.; Martino, H.S.D.; Gonzalez de Mejia, E. Digested total protein and protein fractions from chia seed (Salvia hispanica L.) had high scavenging capacity and inhibited 5-LOX, COX-1-2, and iNOS enzymes. Food Chem. 2019, 289, 204–214. [Google Scholar] [CrossRef] [PubMed]
- PepDraw. Available online: https://www.tulane.edu/~biochem/WW/PepDraw/index.html (accessed on 30 July 2019).
- Segura-Campos, M.; Ruiz-Ruiz, J.; Chel-guerrero, L.; Betancur-Ancona, D. Antioxidant activity of Vigna unguiculata L. walp and hard-to-cook Phaseolus vulgaris L. protein hydrolysates. CyTA J. Food 2013, 11, 208–215. [Google Scholar] [CrossRef]
- García-Nebot, M.J.; Recio, I.; Hernández-ledesma, B. Antioxidant activity and protective effects of peptide lunasin against oxidative stress in intestinal Caco-2 cells. Food Chem. Tox. 2014, 65, 155–161. [Google Scholar] [CrossRef] [Green Version]
- Arise, A.K.; Alashi, A.M.; Nwachukwu, I.D. Function (Vigna subterranea) protein hydrolysates and. Food Funct. 2016, 7, 2431–2437. [Google Scholar] [CrossRef]
- Carrasco-Castilla, J.; Hernández-Alvarez, A.J.; Jiménez-Martínez, C.; Jacinto-Hernández, C.; Alaiz, M.; Girón-Calle, J.; Vioque, J.; Dávila-Ortiz, G. Antioxidant and metal chelating activities of peptide fractions from phaseolin and bean protein hydrolysates. Food Chem. 2012, 135, 1789–1795. [Google Scholar] [CrossRef]
- Pownall, T.L.; Udenigwe, C.C.; Aluko, R.E. Amino acid composition and antioxidant properties of pea seed (Pisum sativum L.) enzymatic protein hydrolysate fractions. J. Agric. Food Chem. 2010, 58, 4712–4718. [Google Scholar] [CrossRef]
- Tang, X.; He, Z.; Dai, Y.; Xiong, Y.L.; Xie, M.; Chen, J. Peptide fractionation and free radical scavenging activity of zein hydrolysate. J. Agric. Food Chem. 2010, 58, 587–593. [Google Scholar] [CrossRef]
- Sonklin, C.; Laohakunjit, N.; Kerdchoechuen, O. Assessment of antioxidant properties of membrane ultrafiltration peptides from mungbean meal protein hydrolysates. PeerJ 2018, 6, e5337. [Google Scholar] [CrossRef]
- Kong, B.; Xiong, Y.L. Antioxidant activity of zein hydrolysates in a liposome system and the possible mode of action. J. Agric. Food Chem. 2006, 54, 6059–6068. [Google Scholar] [CrossRef] [PubMed]
- Ajibola, C.F.; Fashakin, J.B.; Fagbemi, T.N.; Aluko, R.E. Effect of Peptide Size on Antioxidant Properties of African Yam Bean Seed (Sphenostylis stenocarpa) Protein Hydrolysate Fractions. Int. J. Mol. Sci. 2011, 12, 6685–6702. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, L.; Chen, J.; Tang, X.; Xiong, Y.L. Reducing, radical scavenging, and chelation properties of in vitro digests of alcalase-treated zein hydrolysate. J. Agric. Food Chem. 2008, 56, 2714–2721. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.; Hong, H.; Wu, J.; Yan, X. Bioavailability of bioactive peptides derived from food proteins across the intestinal epithelial membrane: A review. Trends Food Sci. Technol. 2019, 86, 399–411. [Google Scholar] [CrossRef]
- Wang, B.; Li, B. Charge and hydrophobicity of casein peptides influence transepithelial transport and bioavailability. Food Chem. 2018, 245, 646–652. [Google Scholar] [CrossRef] [PubMed]



| Protease | P4 Position | P3 Position | P2 Position | P1 Position | P1′ Position | P2′ Position | P3′ Position | P4′ Position |
|---|---|---|---|---|---|---|---|---|
| Alcalase | Gly, Pro, Ala, Val, Leu, Met, Phe, Tyr, Ser, Thr, Asn, Glu, His | Gly, Ala, Val, Leu, Phe, Ser, Thr, Asn, Gln, Asp, Glu, Lys, Arg, His | Gly, Pro, Ala, Val, Leu, Ile, Phe, Tyr, Ser, Thr, Asn, Gln, Glu, Arg, His | Ala, Val, Leu, Met, Phe, Tyr, Ser, Asn, Gln, Glu, Lys, Arg, His | Gly. Ala, Val, Leu, Met, Phe, Tyr, Ser, Thr, Gln, His | Gly, Pro, Ala, Val, Leu, Ile, Phe, Tyr, Ser, Thr, Gln, Glu, Arg, His | Gly, Pro, Ala, Val, Leu, Met, Phe, Tyr, Ser, Thr, Asn, Lys, Arg, His | Gly, Pro, Ala, Val, Leu, Tyr, Ser, Thr, Cys, Asn, Gln, Asp, Glu, Lys, His |
| Stem Bromelain | Pro, Ala, Leu, Ile, Phe, Tyr, Ser, Thr, Cys, Glu, Arg, His | Gly, Pro, Ala, Val, Leu, Phe, Ser, Thr, Lys, Arg, His | Gly, Pro, Val, Leu, Phe, Tyr, Ser, Thr, Asn, Arg, His | Gly, Ala, Val, Leu, Phe, Tyr, Ser, Thr, Asn, Gln, Arg | Gly, Val, Leu, Ile, Phe, Tyr, Ser, Thr, Asn, Gln, Asp, Glu, Lys, His | Pro, Ala, Val, Leu, Tyr, Ser, Thr, Asn, Gln, Asp, Glu, Lys, His | Gly, Pro, Val, Leu, Phe, Ser, Thr, Cys, Asn, Gln, Asp, Glu, Arg, His | Gly, Pro, Ala, Val, Leu, Phe, Tyr, Ser, Thr, Asp, Glu, Lys, Arg, His |
| Ficin | Pro, Leu, Ser, Glu, Lys, Arg, His | Gly, Ala, Val, Leu, Ile, Phe, Thr, Arg, His | Gly. Val, Leu, Phe, Thr, Lys | Gly, Leu, Phe, Tyr, Ser, Lys, Arg, His | Leu, Phe, Tyr, Ser, Thr, Lys, His | Val, Leu, Tyr, Thr, Asn, Lys, His | Pro, Ala, Val, Leu, Ser, Thr, Glu, Lys | Gly, Pro, Val, Asn, Asp, Glu, Lys |
| Papain | Gly, Pro, Ala, Val, Leu, Ile, Phe, Tyr, Ser, Thr, Cys, Asn, Asp, Glu, Arg, His | Gly, Pro, Ala, Val, Leu, Ile, Met, Phe, Tyr, Ser, Gln, Asp, Glu, Lys, Arg, His | All 20 Canonical AA | All 20 Canonical AA | Gly, Ala, Val, Leu, Ile, Met, Phe, Tyr, Ser, Thr, Asn, Gln, Asp, Glu, Lys, His | Gly, Pro, Ala, Val, Leu, Ile, Phe, Tyr, Ser, Thr, Cys, Asn, Gln, Glu, Arg, His | Gly, Pro, Ala, Val, Leu, Phe, Tyr, Ser, Cys, Glu, Lys, Arg, His | Gly, Pro, Ala, Val, Leu, Ile, Phe, Tyr, Ser, Thr, Asn, Asp, Glu, Lys, Arg, His |
| Thermolysin | All 20 Canonical AA | All 20 Canonical AA | All 20 Canonical AA | All 20 Canonical AA | Gly, Pro, Ala, Val, Leu, Ile, Met, Phe, Tyr, Trp, Ser, Thr, Asn, Gln, Asp, Glu, Lys, His | All 20 Canonical AA | All 20 Canonical AA | All 20 Canonical AA |
| Protease | Protein Sequence | Potentially Antioxidant Di-/Tri-Peptides Expected to be Produced 1 | Number of Potentially Broken Antioxidant Fragments |
|---|---|---|---|
| Papain | Q9FRT8 | GFC | 0 |
| Q43680 | EVY, VY, VYF, EAY, AY, AYV, VLK, KPS | 6 | |
| Thermolysin | Q9FRT8 | GFC, FC, FCI, LK, LKK, NFC | 1 |
| Q43680 | EVY, VY, VYF, EAY, AY, AYV, AYI, VYL, SIR, IR, IRN, AYL, VLK, LK, LKA, AFC, FC, EL, ELN, NKP, KP, KPS, NHL, HL, HLS | 9 |
| Enzyme Scheme | Time (min) | ABTS Radical Scavenging Activity (Ascorbic Acid Equivalent, μM) | Iron Chelating Activity (EDTA Equivalent, μM) | ORAC (Trolox Equivalent, μM) |
|---|---|---|---|---|
| Thermolysin + MBA | 0 | 685.0 ± 333.8 | 3034.8 ± 216.4 | 127 ± 19 |
| 5 | 632.7± 288.6 | 5334.9 ± 45.3 | 125 ± 21 | |
| 10 | 683.2 ± 296.1 | 5337.8 ± 38.3 | 115 ± 17 | |
| 25 | 449.7 ± 301.4 | 5328.1 ± 17.4 | 98 ± 39 | |
| 45 | 511.0 ± 255.7 | 5329.2 ± 35.4 | 116 ± 17 | |
| 60 | 493.9 ± 297.7 | 5336.1 ± 26.2 | 127 ± 17 | |
| 240 | 646.2 ± 324.7 | 5161.7 ± 280.2 | 127 ± 17 | |
| 0 | 513.7 ± 230.4 | 3005.5 ± 248.0 | 122 ± 17 | |
| 15 | 533.5 ± 332.0 | 472.4 ± 227.4 | 126 ± 8 | |
| Pepsin + MBA | 25 | 535.3 ± 328.0 | 529.4 ± 234.1 | 96 ± 20 |
| 45 | 540.8 ± 307.3 | 500.2 ± 173.3 | 99 ± 35 | |
| 60 | 477.7 ± 316.6 | 490.7 ± 300.4 | 122 ± 12 | |
| 90 | 638.1 ± 340.2 | 594.3 ± 108.6 | 107 ± 42 | |
| 120 | 495.7 ± 342.3 | 434.3 ± 230.1 | 101 ± 46 | |
| 0 | 487.6 ± 338.4 | 448.5 ± 240.1 | 145 ± 38 | |
| 15 | 450.2 ± 283.0 | 4924.6 ± 119.4 | 105 ± 5 | |
| 25 | 474.1 ± 245.5 | 4901.0 ± 95.0 | 97 ± 31 | |
| Pepsin-Pancreatin + MBA | 45 | 559.7 ± 197.0 | 4886.9 ± 182.8 | 121 ± 17 |
| 60 | 456.9 ± 260.3 | 4884.4 ± 105.6 | 125 ± 16 | |
| 90 | 552.9 ± 256.4 | 4794.9 ± 16.5 | 124 ± 16 | |
| 120 | 581.3 ± 242.9 | 4843.5 ± 155.5 | 127 ± 15 |
| Peptide Sequence | Molecular Mass (Da) | Hydrophobicity (Kcal/mol) | Isoelectric Point | Charge |
|---|---|---|---|---|
| MD | 264 | 10.87 | 2.95 | –1 |
| QSA | 304 | 9.63 | 5.49 | 0 |
| EW | 333 | 9.44 | 3.27 | –1 |
| LGW | 374 | 5.71 | 5.69 | 0 |
| KK | 274 | 13.5 | 10.57 | 2 |
| SVP | 301 | 8.04 | 5.18 | 0 |
| DVAF | 450 | 9.87 | 3.05 | –1 |
| Peptide Sequence | Molecular Mass (Da) | Hydrophobicity (Kcal/mol) | Isoelectric Point | Charge |
|---|---|---|---|---|
| KK | 274 | 14.50 | 10.57 | 2 |
| DM | 264 | 10.87 | 3.02 | –1 |
| SY | 268 | 7.65 | 5.38 | 0 |
| W | 204 | 5.81 | 5.64 | 0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kusumah, J.; Real Hernandez, L.M.; Gonzalez de Mejia, E. Antioxidant Potential of Mung Bean (Vigna radiata) Albumin Peptides Produced by Enzymatic Hydrolysis Analyzed by Biochemical and In Silico Methods. Foods 2020, 9, 1241. https://doi.org/10.3390/foods9091241
Kusumah J, Real Hernandez LM, Gonzalez de Mejia E. Antioxidant Potential of Mung Bean (Vigna radiata) Albumin Peptides Produced by Enzymatic Hydrolysis Analyzed by Biochemical and In Silico Methods. Foods. 2020; 9(9):1241. https://doi.org/10.3390/foods9091241
Chicago/Turabian StyleKusumah, Jennifer, Luis M. Real Hernandez, and Elvira Gonzalez de Mejia. 2020. "Antioxidant Potential of Mung Bean (Vigna radiata) Albumin Peptides Produced by Enzymatic Hydrolysis Analyzed by Biochemical and In Silico Methods" Foods 9, no. 9: 1241. https://doi.org/10.3390/foods9091241
APA StyleKusumah, J., Real Hernandez, L. M., & Gonzalez de Mejia, E. (2020). Antioxidant Potential of Mung Bean (Vigna radiata) Albumin Peptides Produced by Enzymatic Hydrolysis Analyzed by Biochemical and In Silico Methods. Foods, 9(9), 1241. https://doi.org/10.3390/foods9091241