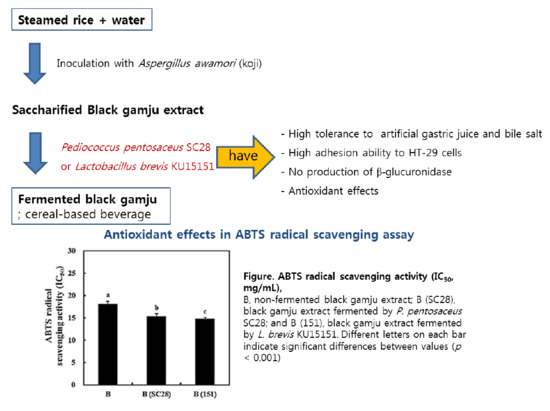
Probiotic Properties and Antioxidant Activities of Pediococcus pentosaceus SC28 and Levilactobacillus brevis KU15151 in Fermented Black Gamju
Abstract
1. Introduction
2. Materials and Methods
2.1. Isolation and Identification of Probiotic Strains
2.2. Tolerance to Artificial Gastric Juice and Bile Salts
2.3. Adhesion Ability to HT-29 Cells
2.4. Enzyme Production
2.5. Antibiotic Susceptibility
2.6. Black Gamju Fermentation and Investigation of Viable Cell Number and pH Value
2.7. Sample Preparation of Bacterial Cells and Extraction of Black Gamju Samples
2.8. Antioxidant Activity of LAB Strains and Black Gamju Extracts
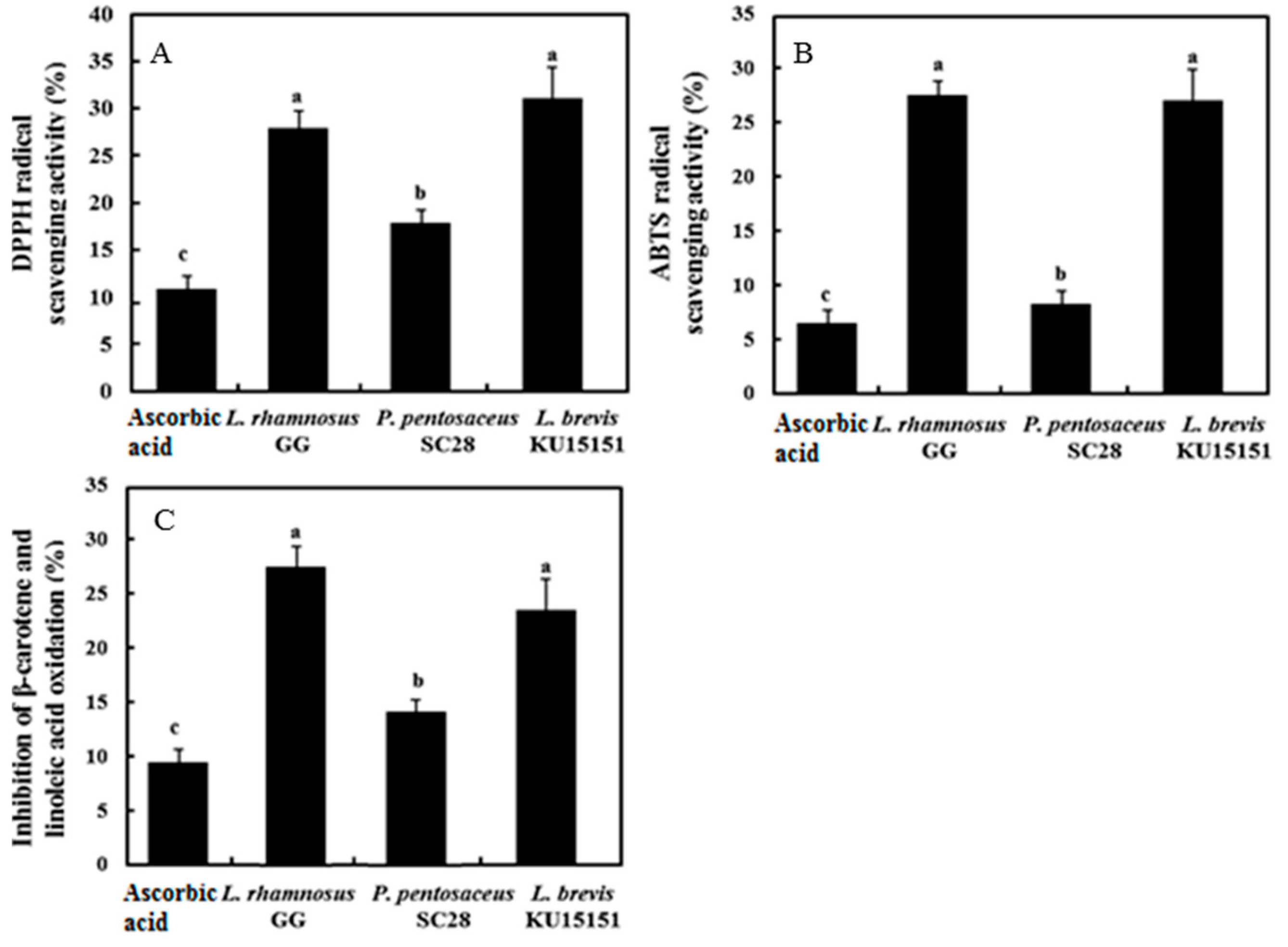
2.8.1. 2,2-Diphenyl-1-picrylhydrazyl (DPPH) Radical Scavenging Activity
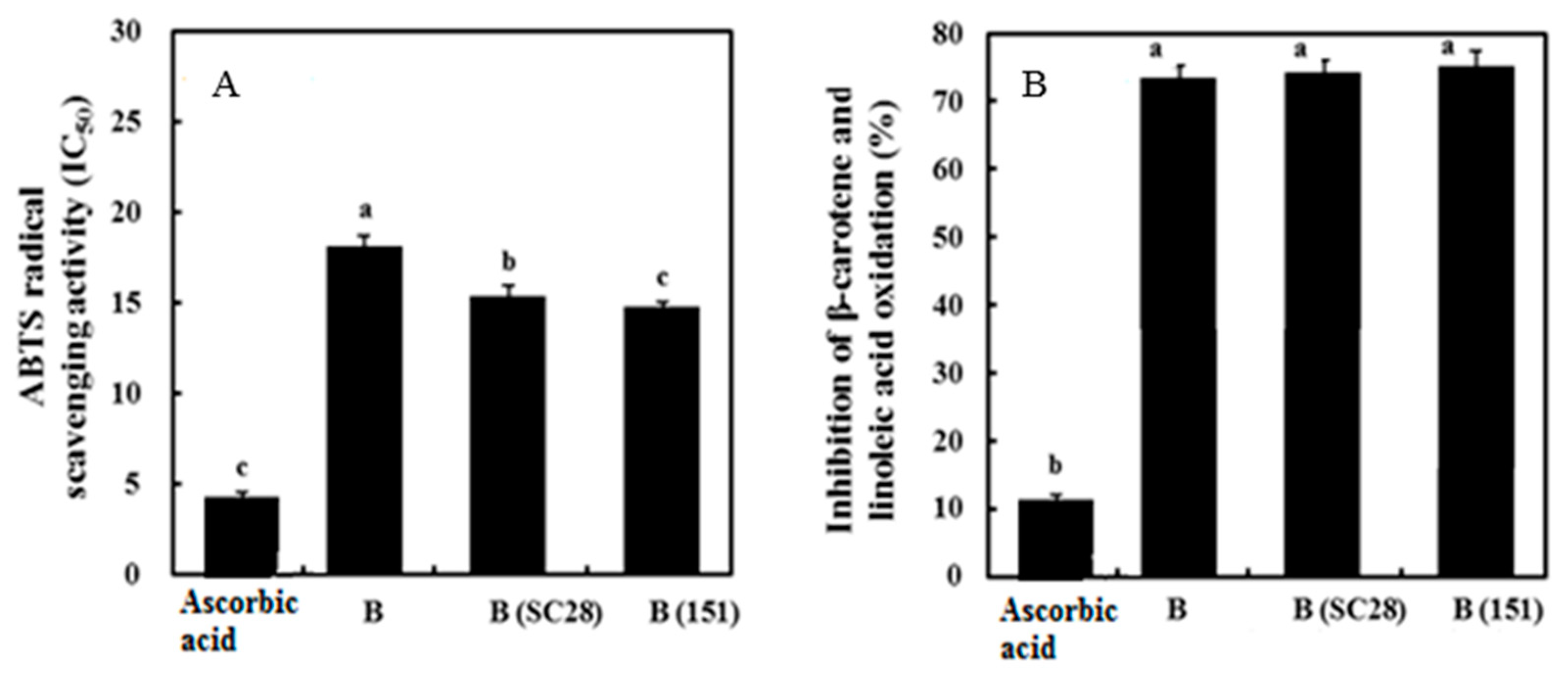
2.8.2. 2,2′-Azinobis(3-ethylbenzothiazoline-6-sulfonate) (ABTS) Radical Scavenging Activity
2.8.3. β-Carotene Bleaching Method
2.9. Statistical Analysis
3. Results
3.1. Tolerance to Artificial Gastric Juice and Bile Salts
3.2. Adhesion Ability of LAB Strains to HT-29 Cells
3.3. Enzyme Production by LAB Strains
3.4. Antibiotic Susceptibility of LAB Strains
3.5. Antioxidant Activity of LAB Strains
3.6. Fermentation of Black Gamju Using LAB Strains
3.7. Antioxidant Activity of Black Gamju Extracts
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Shori, A.B. Influence of food matrix on the viability of probiotic bacteria: A review based on dairy and non-dairy beverages. Food Biosci. 2016, 13, 1–8. [Google Scholar] [CrossRef]
- Choi, H.R.; Chung, Y.H.; Yuk, H.G.; Lee, H.; Jang, H.S.; Kim, Y.; Shin, D. Characterization of Lactobacillus plantarum strains isolated from black raspberry and their effect on BALB/c mice gut microbiota. Food Sci. Biotechnol. 2018, 27, 1747–1754. [Google Scholar] [CrossRef]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. The international scientific association for probiotics and prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastro. Hepat. 2014, 11, 506–514. [Google Scholar] [CrossRef]
- Spyropoulos, B.G.; Misiakos, E.P.; Fotiadis, C.; Stoidis, C.N. Antioxidant properties of probiotics and their protective effects in the pathogenesis of radiation-induced enteritis and colitis. Dig. Dis. Sci. 2011, 56, 285–294. [Google Scholar] [CrossRef]
- Lee, N.K.; Kim, W.K.; Paik, H.D. Bacillus strains as human probiotics: Characterization, safety, microbiome, and probiotic carrier. Food Sci. Biotechnol. 2019, 28, 1297–1305. [Google Scholar] [CrossRef]
- Kandylis, P.; Pissaridi, K.; Bekatorou, A.; Kanellaki, M.; Koutinas, A.A. Dairy and non-dairy probiotic beverages. Curr. Opin. Food Sci. 2016, 7, 58–63. [Google Scholar] [CrossRef]
- Son, S.H.; Yang, S.J.; Jeon, H.L.; Yu, H.S.; Lee, N.K.; Park, Y.S.; Paik, H.D. Antioxidant and immunostimulatory effect of potential probiotic Lactobacillus paraplantarum SC61 isolated from Korean traditional fermented food, jangajji. Microb. Pathog. 2018, 125, 486–492. [Google Scholar] [CrossRef]
- Kim, J.Y.; Park, Y.J.; Lee, H.J.; Park, M.Y.; Kwon, O. Effect of Lactobacillus gasseri BNR17 on irritable bowel syndrome: A randomized, double-blind, placebo-controlled, dose-finding trial. Food Sci. Biotechnol. 2018, 27, 853–857. [Google Scholar] [CrossRef]
- Reale, A.; Di Renzo, T.; Rossi, F.; Zotta, T.; Iacumin, L.; Preziuso, M.; Parente, E.; Sorrentino, E.; Coppola, R. Tolerance of Lactobacillus casei, Lactobacillus paracasei and Lactobacillus rhamnosus strains to stress factors encountered in food processing and in the gastro-intestinal tract. LWT-Food Sci. Technol. 2015, 60, 721e728. [Google Scholar]
- Chavan, M.; Gat, Y.; Harmalkar, M.; Waghmare, R. Development of non-dairy fermented probiotic drink based on germinated and ungerminated cereals and legume. LWT-Food Sci. Technol. 2018, 91, 339–344. [Google Scholar] [CrossRef]
- Peyer, L.C.; Zannini, E.; Arendt, E.K. Lactic acid bacteria as sensory biomodulators for fermented cereal-based beverages. Trends Food Sci. Technol. 2016, 54, 17–25. [Google Scholar] [CrossRef]
- Ogunremi, O.R.; Agrawal, R.; Sanni, A.I. Development of cereal-based functional food using cereal-mix substrate fermented with probiotic strain—Pichia kudriavzevii OG32. Food Sci. Nutr. 2015, 3, 486–494. [Google Scholar] [CrossRef]
- Ghosh, K.; Ray, M.; Adak, A.; Dey, P.; Halder, S.K.; Das, A.; Jana, A.; Parua, S.; Das Mohapatra, P.K.; Pati, B.R.; et al. Microbial, saccharifying and antioxidant properties of an Indian rice based fermented beverage. Food Chem. 2015, 168, 196–202. [Google Scholar] [CrossRef]
- Renzo, T.D.; Reale, A.; Boscaino, F.; Messia, M.C. Flavoring production in Kamut®, quinoa and wheat doughs fermented by Lactobacillus paracasei, Lactobacillus plantarum, and Lactobacillus brevis: A SPME-GC/MS Study. Front. Microbiol. 2018, 9, 429. [Google Scholar] [CrossRef]
- Tsafrakidou, P.; Michaelidou, A.M.; Biliaderis, C.G. Fermented cereal-based products: Nutritional aspects, possible impact on gut microbiota and health implications. Foods 2020, 9, 734. [Google Scholar] [CrossRef]
- Kim, H.; Kim, H.; Bang, J.; Kim, Y.; Beuchat, L.R.; Ryu, J.H. Reduction of Bacillus cereus spores in sikhye, a traditional Korean rice beverage, by modified tyndallization processes with and without carbon dioxide injection. Lett. Appl. Microbiol. 2012, 55, 218–223. [Google Scholar] [CrossRef]
- Jeong, K.Y.; Lee, E.J.; Kim, M.L. Storage properties and sensory characteristics of sikhe added Ulmus pumila L. extract. Korean J. Food Preserv. 2012, 19, 12–18. [Google Scholar] [CrossRef]
- An, Y.H.; Lee, I.S.; Kim, H.S. Quality characteristics of sikhye with varied levels of sweet pumpkin during storage. Korean J. Food Cookery Sci. 2011, 27, 803–814. [Google Scholar] [CrossRef][Green Version]
- Matsubara, T.; Ammar, Y.B.; Anindyawati, T.; Yamamoto, S.; Ito, K.; Iizuka, M.; Minamiura, N. Degradation of raw starch granules by α-amylase purified from culture of Aspergillus awamori KT-11. J. Biochem. Mol. Biol. 2004, 37, 422–428. [Google Scholar] [CrossRef]
- Guo, C.F.; Zhang, L.W.; Han, X.; Yi, H.X.; Li, J.Y.; Tuo, Y.F.; Zhang, Y.C.; Ming, D. Screening for cholesterol-lowering probiotic based on deoxycholic acid removal pathway and studying its functional mechanisms in vitro. Anaerobe 2012, 18, 516–522. [Google Scholar] [CrossRef]
- Yang, S.J.; Lee, J.E.; Lim, S.M.; Kim, Y.J.; Lee, N.K.; Paik, H.D. Antioxidant and immune-enhancing effects of probiotic Lactobacillus plantarum 200655 isolated from kimchi. Food Sci. Biotechnol. 2019, 28, 491–499. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, K.; Ray, M.; Adak, A.; Halder, S.K.; Das, A.; Jana, A.; Parua, S.; Vágvölgyi, C.; Das Mohapatra, P.K.; Pati, B.R.; et al. Role of probiotic Lactobacillus fermentum KKL1 in the preparation of a rice based fermented beverage. Bioresour. Technol. 2015, 188, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Das, D.; Goyal, A. Antioxidant activity and γ-aminobutyric acid (GABA) producing ability of probiotic Lactobacillus plantarum DM5 isolated from Marcha of Sikkim. LWT-Food Sci. Technol. 2015, 61, 263–268. [Google Scholar] [CrossRef]
- Verón, H.E.; Di Risio, H.D.; Isla, M.I.; Torres, S. Isolation and selection of potential probiotic lactic acid bacteria from Opuntia ficus-indica fruits that grow in Northwest Argentina. LWT-Food Sci. Technol. 2017, 84, 231–240. [Google Scholar]
- Kassim, N.K.; Lim, P.C.; Ismail, A.; Awang, K. Isolation of antioxidative compounds from Micromelum minutum guided by preparative thin layer chromatography-2,2-diphenyl-1-picrylhydrazyl (PTLC-DPPH) bioautography method. Food Chem. 2019, 272, 185–191. [Google Scholar] [CrossRef]
- Vidhyasagar, V.; Jeevaratnam, K.; Osmanagaoglu, O.; Kiran, F.; Ataoglu, H. Evaluation of Pediococcus pentosaceus strains isolated from Idly batter for probiotic properties in vitro. J. Funct. Foods 2013, 5, 235–243. [Google Scholar] [CrossRef]
- Bujnakova, D.; Strakova, E.; Kmet, V. In vitro evaluation of the safety and probiotic properties of Lactobacilli isolated from chicken and calves. Anaerobe 2014, 29, 118–127. [Google Scholar] [CrossRef]
- Cho, K.M.; Ahn, B.Y.; Seo, W.T. Lactic acid fermentation of gamju manufactured using medicinal herb decoction. Korean J. Food Sci. Technol. 2008, 40, 6–12. [Google Scholar]
- Guo, Z.; Wang, J.; Yan, L.; Chen, W.; Liu, X.M.; Zhang, H.P. In vitro comparison of probiotic properties of Lactobacillus casei Zhang, a potential new probiotic, with selected probiotic strains. LWT-Food Sci. Technol. 2009, 42, 1640–1646. [Google Scholar] [CrossRef]
- Jeon, E.B.; Son, S.H.; Jeewanthi, R.K.C.; Lee, N.K.; Paik, H.D. Characterization of Lactobacillus plantarum Lb41, an isolate from kimchi and its application as a probiotic in cottage cheese. Food Sci. Biotechnol. 2016, 25, 1129–1133. [Google Scholar] [CrossRef]
- Osmanagaoglu, O.; Kiran, F.; Ataoglu, H. Evaluation of in vitro probiotic potential of Pediococcus pentosaceus OZF isolated from human breast milk. Probiotics Antimicrob. Proteins 2010, 2, 162–174. [Google Scholar] [CrossRef]
- Han, Q.; Kong, B.; Chen, Q.; Sun, F.; Zhang, H. In vitro comparison of probiotic properties of lactic acid bacteria isolated from Harbin dry sausages and selected probiotics. J. Func. Foods 2017, 32, 391–400. [Google Scholar] [CrossRef]
- Bengoa, A.A.; Zavala, L.; Carasi, P.; Trejo, S.A.; Bronsoms, S.; Serradell, M.Á.; Garrote, G.L.; Abraham, A.G. Simulated gastrointestinal conditions increase adhesion ability of Lactobacillus paracasei strains isolated from kefir to Caco-2 cells and mucin. Food Res. Int. 2018, 103, 462–467. [Google Scholar] [CrossRef]
- Hernández-Alcántara, A.M.; Wacher, C.; Llamas, M.G.; López, P.; Pérez-Chabela, M.L. Probiotic properties and stress response of thermotolerant lactic acid bacteria isolated from cooked meat products. LWT-Food Sci. Technol. 2018, 91, 249–257. [Google Scholar]
- Jeon, H.L.; Lee, N.K.; Yang, S.J.; Kim, W.S.; Paik, H.D. Probiotic characterization of Bacillus subtilis P223 isolated from kimchi. Food Sci. Biotechnol. 2017, 26, 1641–1648. [Google Scholar] [CrossRef]
- Lee, K.W.; Shim, J.M.; Park, S.K.; Heo, H.J.; Kim, H.J.; Ham, K.S.; Kim, J.H. Isolation of lactic acid bacteria with probiotic potentials from kimchi, traditional Korean fermented vegetable. LWT-Food Sci. Technol. 2016, 71, 130–137. [Google Scholar] [CrossRef]
- Son, S.H.; Jeon, H.L.; Yang, S.J.; Lee, N.K.; Paik, H.D. In vitro characterization of Lactobacillus brevis KU15006, an isolate from kimchi, reveals anti-adhesion activity against foodborne pathogens and antidiabetic properties. Microb. Pathog. 2017, 112, 135–141. [Google Scholar] [CrossRef]
- Ljungh, A.; Wadstrom, T. Lactic acid bacteria as probiotics. Curr. Issues Intest. Microbiol. 2006, 7, 73–90. [Google Scholar]
- Wang, J.; Wei, X.; Fan, M. Assessment of antibiotic susceptibility within lactic acid bacteria and coagulase-negative staphylococci isolated from hunan smoked pork, a naturally fermented meat product in China. J. Food Sci. 2018, 83, 1707–1715. [Google Scholar] [CrossRef]
- Tang, W.; Xing, Z.; Li, C.; Wang, J.; Wang, Y. Molecular mechanisms and in vitro antioxidant effects of Lactobacillus plantarum MA2. Food Chem. 2017, 221, 1642–1649. [Google Scholar] [CrossRef]
- Li, S.; Zhao, Y.; Zhang, L.; Huang, L.; Li, D.; Niu, C.; Yang, Z.; Wang, Q. Antioxidant activity of Lactobacillus plantarum strains isolated from traditional Chinese fermented foods. Food Chem. 2012, 135, 1914–1919. [Google Scholar] [CrossRef] [PubMed]
| LAB Strains | Survival Rate (%) | Adhesion Ability (%) | |
|---|---|---|---|
| Gastric Acid Tolerance (0.3% Pepsin, pH 2.5) | Bile Salt Tolerance (0.3% Oxgall) | ||
| L. rhamnosus GG | 96.69 ± 1.04 | 101.83 ± 0.87 a | 6.30 ± 0.51 a |
| P. pentosaceus SC28 | 96.37 ± 3.02 | 100.05 ± 0.22 b | 4.45 ± 0.19 b |
| L. brevis KU15151 | 94.52 ± 2.87 | 97.96 ± 0.37 c | 6.87 ± 0.35 a |
| Enzymes | Enzyme Activity (1) | |
|---|---|---|
| P. pentosaceus SC28 | L. brevis KU15151 | |
| Control | 0 | 0 |
| N-Acetyl-β-glucosaminidase | 0 | 0 |
| Acid phosphatase | 1 | 1 |
| Alkaline phosphate | 0 | 0 |
| α-Chymotrypsin | 0 | 0 |
| Cystine arylamidase | 0 | 1 |
| Esterase | 0 | 1 |
| Esterase lipase | 0 | 1 |
| α-Fucosidase | 0 | 0 |
| α-Galactosidase | 0 | 1 |
| β-Galactosidase | 1 | 3 |
| α-Glucosidase | 0 | 1 |
| β-Glucosidase | 0 | 3 |
| β-Glucuronidase | 0 | 0 |
| α-Mannosidase | 0 | 0 |
| Naphthol-AS-BI-phosphohydrolase | 1 | 1 |
| Valine arylamidase | 3 | 2 |
| Antibiotics | LAB Strains | |
|---|---|---|
| P. pentosaceus SC28 | L. brevis KU15151 | |
| Ampicillin | S (1) | S |
| Chloramphenicol | S | S |
| Ciprofloxacin | R | R |
| Doxycycline | S | S |
| Gentamicin | R | R |
| Kanamycin | R | R |
| Streptomycin | R | R |
| Tetracycline | S | S |
| Sample (1) | Viable Cell Number (log CFU/mL) | pH |
|---|---|---|
| B | – | 4.37 ± 0.02 a |
| B (SC28) | 8.25 ± 0.08 b (2) | 3.41 ± 0.02 b |
| B (151) | 8.95 ± 0.01 a | 3.33 ± 0.01 c |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, S.J.; Kim, K.-T.; Kim, T.Y.; Paik, H.-D. Probiotic Properties and Antioxidant Activities of Pediococcus pentosaceus SC28 and Levilactobacillus brevis KU15151 in Fermented Black Gamju. Foods 2020, 9, 1154. https://doi.org/10.3390/foods9091154
Yang SJ, Kim K-T, Kim TY, Paik H-D. Probiotic Properties and Antioxidant Activities of Pediococcus pentosaceus SC28 and Levilactobacillus brevis KU15151 in Fermented Black Gamju. Foods. 2020; 9(9):1154. https://doi.org/10.3390/foods9091154
Chicago/Turabian StyleYang, Seo Jin, Kee-Tae Kim, Tae Young Kim, and Hyun-Dong Paik. 2020. "Probiotic Properties and Antioxidant Activities of Pediococcus pentosaceus SC28 and Levilactobacillus brevis KU15151 in Fermented Black Gamju" Foods 9, no. 9: 1154. https://doi.org/10.3390/foods9091154
APA StyleYang, S. J., Kim, K.-T., Kim, T. Y., & Paik, H.-D. (2020). Probiotic Properties and Antioxidant Activities of Pediococcus pentosaceus SC28 and Levilactobacillus brevis KU15151 in Fermented Black Gamju. Foods, 9(9), 1154. https://doi.org/10.3390/foods9091154