Growth Dynamics and Diversity of Yeasts during Spontaneous Plum Mash Fermentation of Different Varieties
Abstract
1. Introduction
2. Materials and Methods
2.1. Spontaneous Plum Mash Fermentation
2.2. Yeast Enumeration and Isolation
2.3. DNA Extraction and RAPD-PCR Analysis
2.4. Amplification and Sequencing the D1/D2 Domains of the 26S rRNA Gene Region
2.5. Production of Volatile Components by Identified Yeast Strains (SPME-GC-TOFMS)
2.6. Statistical Analysis
3. Results
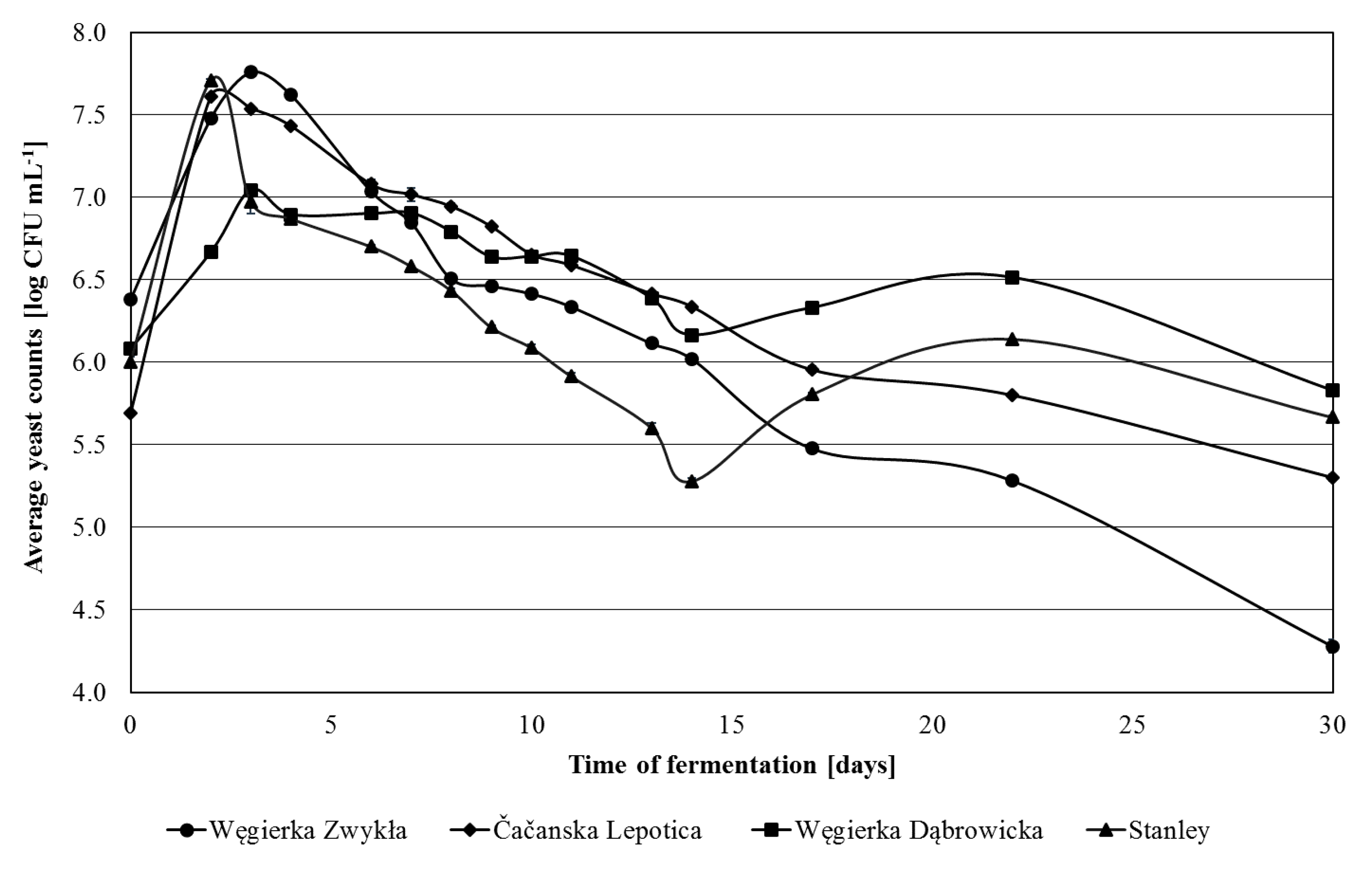
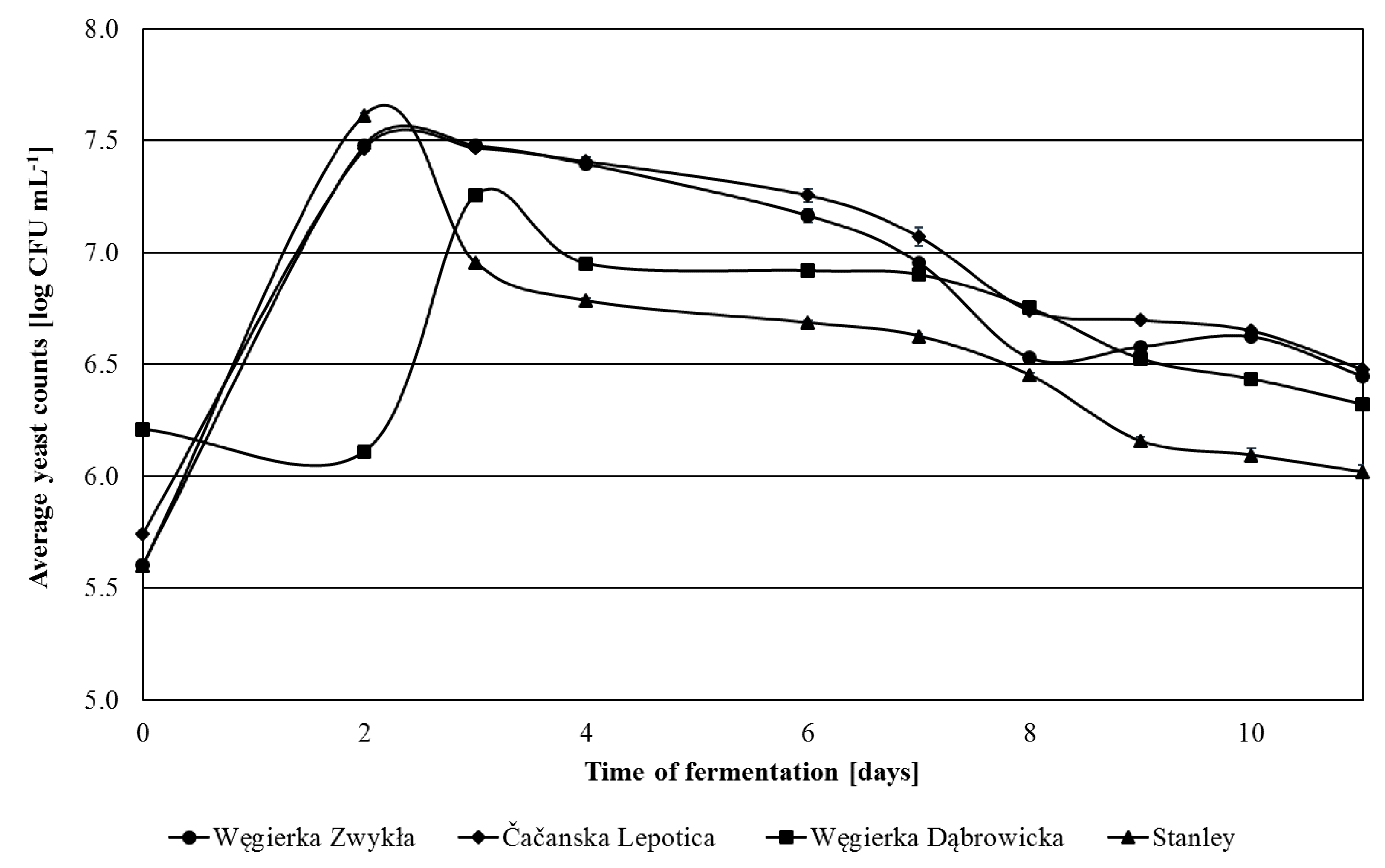
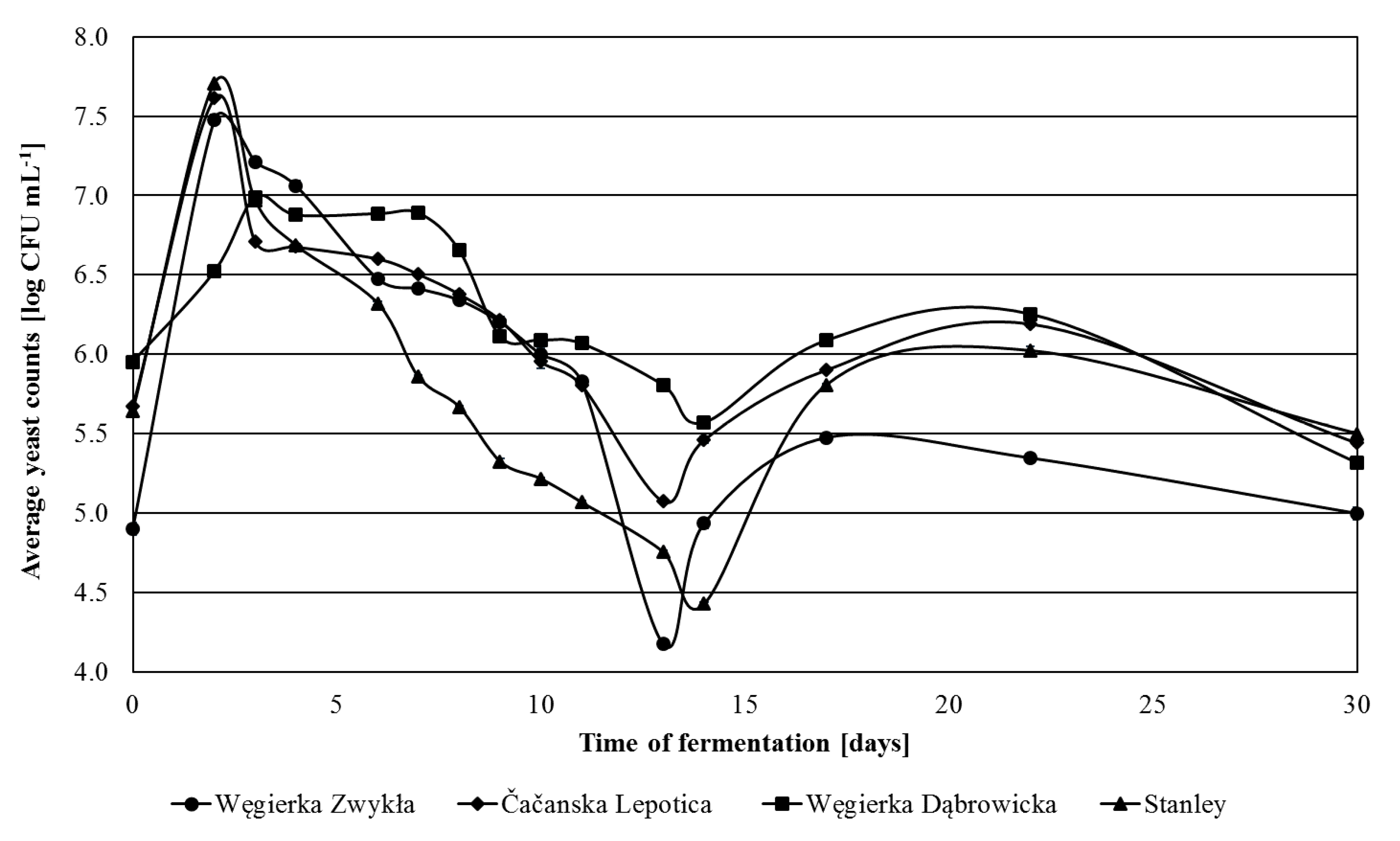
3.1. Yeast Population Changes during Spontaneous Fermentation
3.2. Yeast Identification
3.3. Biodiversity of Yeasts during Fermentation
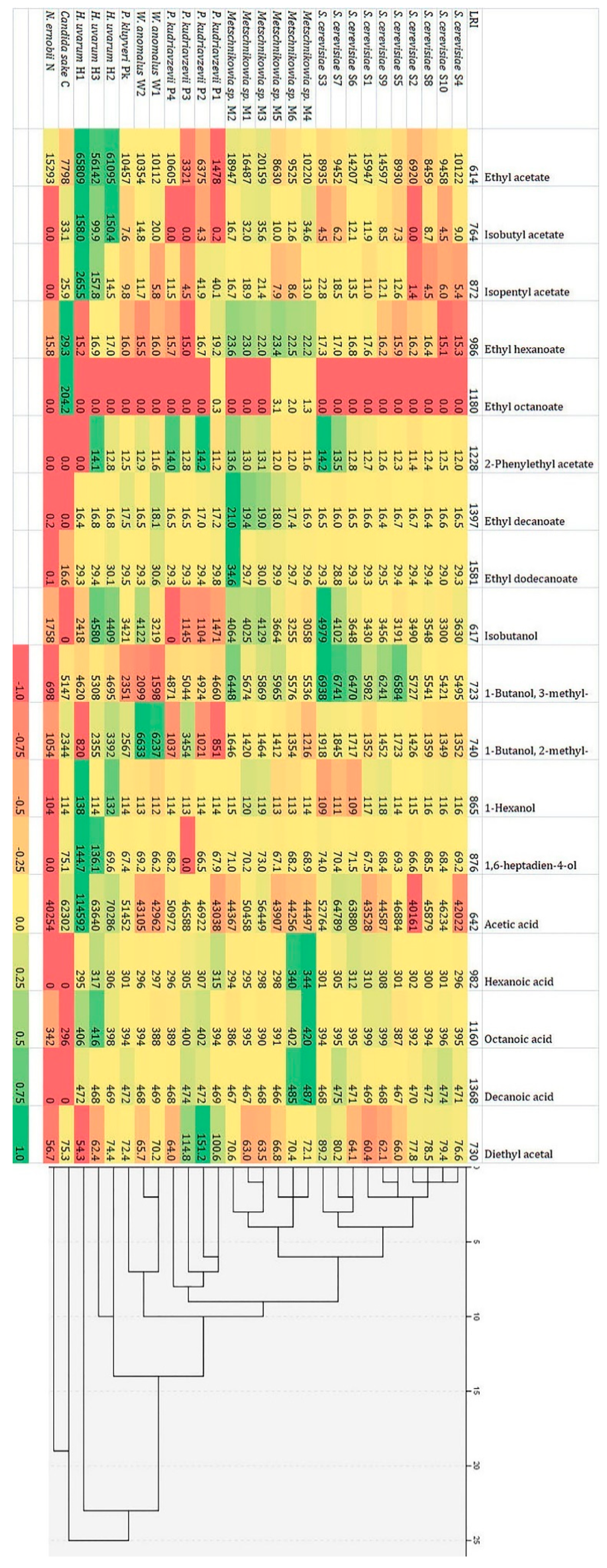
3.4. Production of Volatile Components by Identified Yeast Strains
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Śliwińska, M.; Wiśniewska, P.; Dymerski, T.; Wardencki, W.; Namieśnik, J. The flavour of fruit spirits and fruit liqueurs: A review. Flavour Fragr. J. 2015, 30, 197–207. [Google Scholar] [CrossRef]
- Sosna, I. Ocena wartości produkcyjnej kilkudziesięciu odmian śliwy na podkładce z Ałyczy w rejonie Wrocławia. Acta Sci. Pol. Hortic. Cult. 2004, 3, 47–54. [Google Scholar]
- Moreno-Arribas, M.V.; Polo, M.C. Wine Chemistry and Biochemistry; Springer: New York, NY, USA, 2009. [Google Scholar]
- Schehl, B.; Lachenmeier, D.; Senn, T.; Heinisch, J.J. Effect of the stone content on the quality of plum and cherry spirits produced from mash fermentations with commercial and laboratory yeast Strains. J. Agric. Food Chem. 2005, 53, 8230–8238. [Google Scholar] [CrossRef] [PubMed]
- Urošević, I.; Nikićević, N.; Stanković, L.; Anđelković, B.; Urošević, T.; Krstić, G.; Tešević, V. Influence of yeast and nutrients on the quality of apricot brandy. J. Serbian Chem. Soc. 2014, 79, 1223–1234. [Google Scholar] [CrossRef]
- Popović, B.; Nikićević, N.; Gavrilović-Damnjanović, J.; Mitrović, O.; Srećković, M.; Ogašanović, D. The yield of plum brandy as influenced by different plum cultivars. Voćarstvo 2008, 42, 111–118. [Google Scholar]
- Crowell, E.A.; Guymon, J.F. Aroma constituents of plum brandy. Am. J. Enol. Vitic. 1973, 24, 159–165. [Google Scholar]
- Velíšek, J.; Pudil, F.; Davídek, J.; Kubelka, V. The neutral volatile components of Czechoslovak plum brandy. Zeitschrift Leb. Forsch. 1982, 174, 463–466. [Google Scholar] [CrossRef]
- Tešević, V.; Nikićević, N.; Jovanović, A.; Djoković, D.; Vujisić, L.; Vučković, I.; Bonić, M. Volatile components from old plum brandies. Food Technol. Biotechnol. 2005, 43, 367–372. [Google Scholar]
- Miličević, B.; Lukić, I.; Babić, J. Aroma and sensory characteristics of Slavonian plum brandy. Technol. Acta 2012, 5, 1–7. [Google Scholar]
- Zhang, H.; Woodams, E.E.; Hang, Y.D. Factors affecting the methanol content and yield of plum brandy. J. Food Sci. 2012, 77, T79–T82. [Google Scholar] [CrossRef]
- Pielech-Przybylska, K.; Balcerek, M.; Nowak, A.; Patelski, P.; Dziekońska-Kubczak, U. Influence of yeast on the yield of fermentation and volatile profile of ‘Węgierka Zwykła’ plum distillates. J. Inst. Brew. 2016, 122, 612–623. [Google Scholar]
- Satora, P.; Tuszyński, T. Biodiversity of yeasts during plum Wegierka Zwykla spontaneous fermentation. Food Technol. Biotechnol. 2005, 43, 277–282. [Google Scholar]
- Kurtzman, C.P.; Robnett, C.J. Identification and phylogeny of ascomycetous yeasts from analysis of nuclear large subunit (26S) ribosomal DNA partial sequences. Anton. Leeuw. 1998, 73, 331–371. [Google Scholar] [CrossRef] [PubMed]
- Zdaniewicz, M.; Satora, P.; Pater, A.; Bogacz, S. Low lactic acid-producing strain of Lachancea thermotolerans as a new starter for beer production. Biomolecules 2020, 10, 256. [Google Scholar] [CrossRef]
- Fleet, G.H.; Herd, G.M. Yeasts—Growth during fermentation. In Wine Microbiology and Biotechnology; Fleet, G.H., Ed.; Harwood Academic Publishers: Chur, Switzerland, 1993; pp. 27–55. [Google Scholar]
- Boulton, R.B.; Singleton, V.L.; Bisson, L.F.; Kunkee, R.E. Principles and Practices of Winemaking; Chapman & Hall: New York, NY, USA, 1996. [Google Scholar]
- Ribereau-Gayon, P.; Dubourdieu, D.; Doneche, B.; Lonvaud, A. Handbook of Enology: The Microbiology of Wine and Vinifications; John Wiley & Sons, Ltd.: New Jersey, NJ, USA, 2006. [Google Scholar]
- Jolly, N.P.; Augustyn, O.P.H.; Pretorius, I.S. The role and use of non-Saccharomyces yeasts in wine production. S. Afr. J. Enol. Vitic. 2006, 27, 15–38. [Google Scholar] [CrossRef]
- Romano, P.; Suzzi, G.; Comi, G.; Zironi, R.; Maifreni, M. Glycerol and other fermentation products of apiculate wine yeasts. J. Appl. Microbiol. 1997, 82, 615–618. [Google Scholar] [CrossRef]
- Di Maro, E.; Ercolini, D.; Coppola, S. Yeast dynamics during spontaneous wine fermentation of the Catalanesca grape. Int. J. Food Microbiol. 2007, 117, 201–210. [Google Scholar] [CrossRef]
- Pretorius, I.S. Tailoring wine yeast for the new millennium: Novel approaches to the ancient art of winemaking. Yeast 2000, 16, 675–729. [Google Scholar] [CrossRef]
- Fleet, G.H.; Prakitchaiwattana, P.; Beh, A.L.; Heard, G. The yeast ecology of wine grapes. In Biodiversity and Biotechnology of Wine Yeasts; Ciani, M., Ed.; Research Signpost: Kerala, India, 2002; pp. 1–17. [Google Scholar]
- Satora, P.; Kostrz, M.; Sroka, P.; Tarko, T. Chemical profile of spirits obtained by spontaneous fermentation of different varieties of plum fruits. Eur. Food Res. Technol. 2017, 243, 489–499. [Google Scholar] [CrossRef]
- Cioch-Skoneczny, M.; Satora, P.; Skotniczny, M.; Skoneczny, S. Quantitative and qualitative composition of yeast microbiota in spontaneously fermented grape musts obtained from cool climate grape varieties ‘Rondo’and ‘Regent’. FEMS Yeast Res. 2018, 18, foy089. [Google Scholar] [CrossRef]
- Combina, M.; Mercado, L.; Borgo, P.; Elia, A.; Jofré, V.; Ganga, A.; Martinez, C.; Catania, C. Yeasts associated to Malbec grape berries from Mendoza, Argentina. J. Appl. Microbiol. 2005, 98, 1055–1061. [Google Scholar] [CrossRef] [PubMed]
- Statistical Yearbook of Agriculture; Statistics Poland: Warszawa, Poland, 2014.
- Clavijo, A.; Calderón, I.L.; Paneque, P. Diversity of Saccharomyces and non-Saccharomyces yeasts in three red grape varieties cultured in the Serranía de Ronda (Spain) vine-growing region. Int. J. Food Microbiol. 2010, 143, 241–245. [Google Scholar] [CrossRef] [PubMed]
- Guzzon, R.; Widmann, G.; Settanni, L.; Malacarne, M.; Francesca, N.; Larcher, R. Evolution of yeast populations during different biodynamic winemaking processes. S. Afr. J. Enol. Vitic. 2011, 32, 242–250. [Google Scholar] [CrossRef]
- Barata, A.; Malfeito-Ferreira, M.; Loureiro, V. The microbial ecology of wine grape berries. Int. J. Food Microbiol. 2012, 153, 243–259. [Google Scholar] [CrossRef]
- Beltran, G.; Torija, M.J.; Novo, M.; Ferrer, N.; Poblet, M.; Guillamón, J.M.; Rozès, N.; Mas, A. Analysis of yeast populations during alcoholic fermentation: A six year follow-up study. Syst. Appl. Microbiol. 2002, 25, 287–293. [Google Scholar] [CrossRef]
- Tuszyński, T.; Satora, P. Microbiological characteristics of the Węgierka Zwykła plum orchard in submontane region. Pol. J. Food Nutr. Sci. 2003, 12, 43–48. [Google Scholar]
- Sipiczki, M.; Pfliegler, W.P.; Holb, I.J. Metschnikowia species share a pool of diverse rRNA genes differing in regions that determine hairpin-loop structures and evolve by reticulation. PLoS ONE 2013, 8, e67384. [Google Scholar] [CrossRef]
- Sipiczki, M.; Horvath, E.; Pfliegler, W.P. Birth-and-death evolution and reticulation of ITS segments of Metschnikowia andauensis and Metschnikowia fructicola rDNA repeats. Front. Microbiol. 2018, 9, 1193. [Google Scholar] [CrossRef]
- Chavan, P.; Mane, S.; Kulkarni, G.; Shaikh, S.; Ghormade, V.; Nerkar, D.P.; Shouche, Y.; Deshpande, M.V. Natural yeast flora of different varieties of grapes used for wine making in India. Food Microbiol. 2009, 26, 801–808. [Google Scholar] [CrossRef]
- Li, S.S.; Cheng, C.; Li, Z.; Chen, J.Y.; Yan, B.; Han, B.Z.; Reeves, M. Yeast species associated with wine grapes in China. Int. J. Food Microbiol. 2010, 138, 85–90. [Google Scholar] [CrossRef]
- Bezerra-Bussoli, C.; Baffi, M.A.; Gomes, E.; Da-Silva, R. Yeast diversity isolated from grape musts during spontaneous fermentation from a Brazilian winery. Curr. Microbiol. 2013, 67, 356–361. [Google Scholar] [CrossRef] [PubMed]
- Garofalo, C.; Tristezza, M.; Grieco, F.; Spano, G.; Capozzi, V. From grape berries to wine: Population dynamics of cultivable yeasts associated to “Nero di Troia” autochthonous grape cultivar. World J. Microbiol. Biotechnol. 2016, 32, 59. [Google Scholar] [CrossRef]
- Tuset, J.J.; Perucho, R. First recognition of Issatchenkia orientalis in mature citrus fruits in Spain. Plant Dis. 2001, 85, 335. [Google Scholar] [CrossRef] [PubMed]
- Zhanlin, B.; Xin, Z.; Peijie, H.; Andong, Z.; Hao, Z. Yeast from Pyrus ussuriensis maxim fruit peel. J. Pure Appl. Microbiol. 2016, 10, 1943–1949. [Google Scholar]
- Batt, C.A.; Robinson, R.K. Encyclopedia of Food Microbiology, 2nd ed.; Academic Press: Cambridge, MA, USA, 2014. [Google Scholar]
- Vadkertiová, R.; Molnárová, J.; Vránová, D.; Sláviková, E. Yeasts and yeast-like organisms associated with fruits and blossoms of different fruit trees. Can. J. Microbiol. 2012, 58, 1344–1352. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.-H.; Hong, Y.A.; Park, H.-D. Co-fermentation of grape must by Issatchenkia orientalis and Saccharomyces cerevisiae reduces the malic acid content in wine. Biotechnol. Lett. 2008, 30, 1633–1638. [Google Scholar] [CrossRef]
- Díaz, C.; Molina, A.M.; Nähring, J.; Fischer, R. Characterization and dynamic behavior of wild yeast during spontaneous wine fermentation in steel tanks and amphorae. BioMed Res. Int. 2013, 2013, 540465. [Google Scholar] [CrossRef]
- Mateo, J.; Maicas, S. Application of non-Saccharomyces yeasts to wine-making process. Fermentation 2016, 2, 14. [Google Scholar] [CrossRef]
- Aponte, M.; Blaiotta, G. Potential role of yeast strains isolated from grapes in the production of taurasi DOCG. Front. Microbiol. 2016, 7, 809. [Google Scholar] [CrossRef]
- Ciani, M.; Fatichenti, F. Selective sugar consumption by apiculate yeasts. Lett. Appl. Microbiol. 1999, 28, 203–206. [Google Scholar] [CrossRef]
- Tristezza, M.; Tufariello, M.; Capozzi, V.; Spano, G.; Mita, G.; Grieco, F. The oenological potential of Hanseniaspora uvarum in simultaneous and sequential co-fermentation with Saccharomyces cerevisiae for industrial wine production. Front. Microbiol. 2016, 7, 670. [Google Scholar] [CrossRef] [PubMed]
- Sadoudi, M.; Tourdot-Maréchal, R.; Rousseaux, S.; Steyer, D.; Gallardo-Chacón, J.J.; Ballester, J.; Vichi, S.; Guérin-Schneider, R.; Caixach, J.; Alexandre, H. Yeast-yeast interactions revealed by aromatic profile analysis of Sauvignon Blanc wine fermented by single or co-culture of non-Saccharomyces and Saccharomyces yeasts. Food Microbiol. 2012, 32, 243–253. [Google Scholar] [CrossRef] [PubMed]
- Hornsey, I. The Chemistry and Biology of Winemaking; The Royal Society of Chemistry: Cambridge, UK, 2007. [Google Scholar]
- Raspor, P.; Milek, D.M.; Polanc, J.; Mozina, S.S.; Cadez, N. Yeasts isolated from three varieties of grapes cultivated in different locations of the Dolenjska vine-growing region, Slovenia. Int. J. Food Microbiol. 2006, 109, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Bokulich, N.A.; Thorngate, J.H.; Richardson, P.M.; Mills, D.A. Microbial biogeography of wine grapes is conditioned by cultivar, vintage, and climate. Proc. Natl. Acad. Sci. USA 2014, 111, E139–E148. [Google Scholar] [CrossRef]
- Torija, M.J.; Rozès, N.; Poblet, M.; Guillamón, J.M.; Mas, A. Yeast population dynamics in spontaneous fermentations: Comparison between two different wine-producing areas over a period of three years. Anton. Leeuw. 2001, 79, 345–352. [Google Scholar] [CrossRef]
- Lopes, C.A.; Van Broock, M.; Querol, A.; Caballero, A.C. Saccharomyces cerevisiae wine yeast populations in a cold region in Argentinean Patagonia. A study at different fermentation scales. J. Appl. Microbiol. 2002, 93, 608–615. [Google Scholar] [CrossRef]
- Schuller, D.; Alves, H.; Dequin, S.; Casal, M. Ecological survey of Saccharomyces cerevisiae strains from vineyards in the Vinho Verde Region of Portugal. FEMS Microbiol. Ecol. 2005, 51, 167–177. [Google Scholar] [CrossRef]

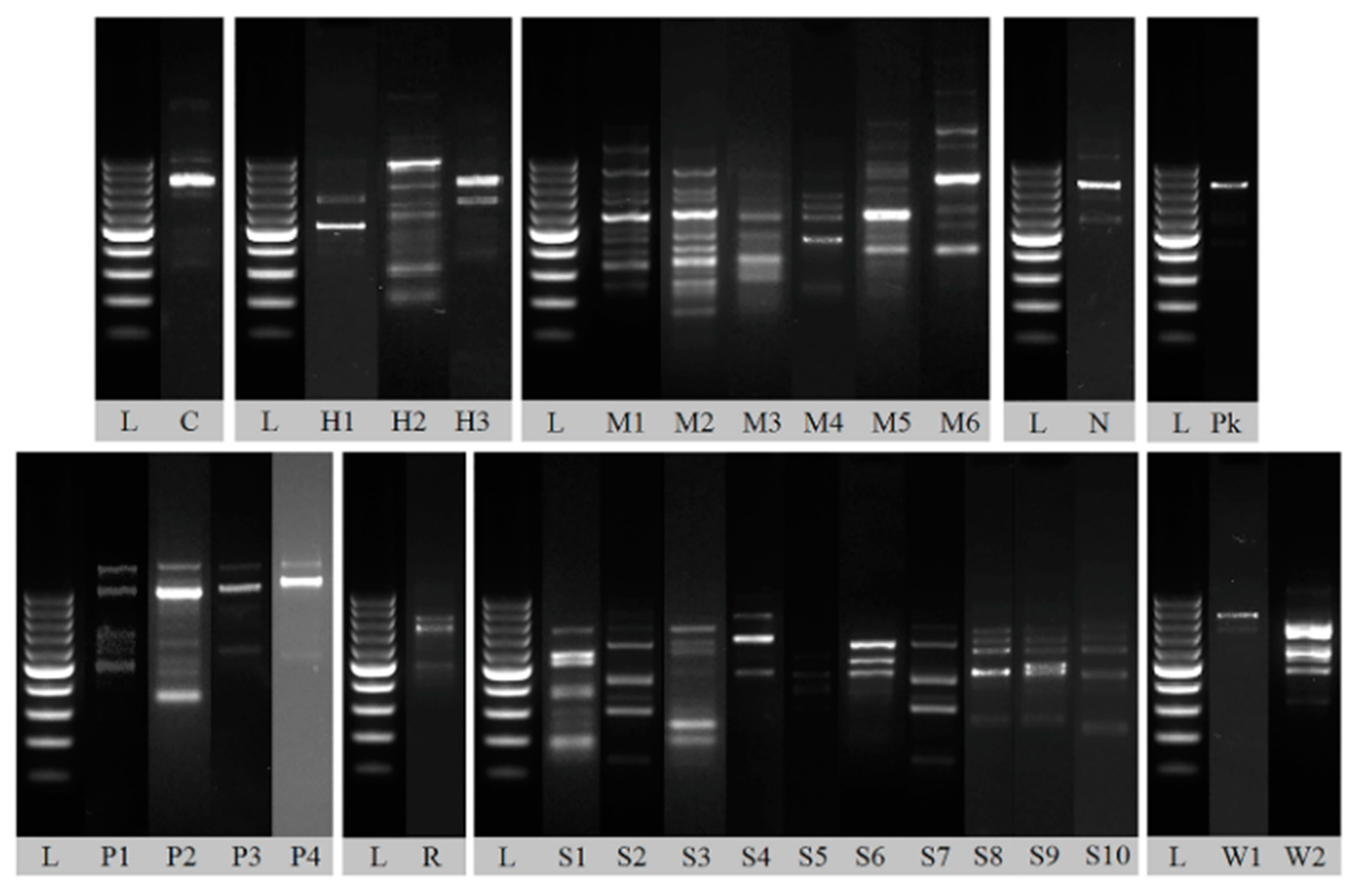
| Strain Symbol | Identification | % Identity | Query Length [bp] | Query Cover | GenBank Accession No. | Number of Isolates |
|---|---|---|---|---|---|---|
| C | Candida sake | 99.82% | 561 | 100% | MN464117 | 2 |
| H1 | Hanseniaspora uvarum | 100.00% | 569 | 100% | MN464118 | 20 |
| H2 | Hanseniaspora uvarum | 100.00% | 566 | 100% | MN464119 | 43 |
| H3 | Hanseniaspora uvarum | 100.00% | 556 | 100% | MN464120 | 16 |
| M1 | Metschnikowia sp. | 99.59% | 490 | 100% | MN464121 | 33 |
| M2 | Metschnikowia sp. | 99.59% | 492 | 100% | MN464122 | 6 |
| M3 | Metschnikowia sp. | 99.39% | 495 | 100% | MN464123 | 3 |
| M4 | Metschnikowia sp. | 99.80% | 507 | 98% | MN464124 | 6 |
| M5 | Metschnikowia sp. | 99.39% | 493 | 100% | MN464125 | 6 |
| M6 | Metschnikowia sp. | 99.39% | 494 | 100% | MN464126 | 3 |
| N | Nakazawaea ernobii | 99.29% | 561 | 99% | MN464127 | 1 |
| Pk | Pichia kluyveri | 100.00% | 567 | 100% | MN464128 | 3 |
| P1 | Pichia kudriavzevii | 99.82% | 562 | 100% | MN464129 | 1 |
| P2 | Pichia kudriavzevii | 100.00% | 556 | 100% | MN464130 | 4 |
| P3 | Pichia kudriavzevii | 100.00% | 470 | 100% | MN464131 | 6 |
| P4 | Pichia kudriavzevii | 100.00% | 559 | 99% | MN464132 | 7 |
| R | Rhodotorula mucilaginosa | 99.82% | 562 | 100% | MN464133 | 2 |
| S1 | Saccharomyces cerevisiae | 100.00% | 569 | 100% | MN464134 | 19 |
| S2 | Saccharomyces cerevisiae | 100.00% | 572 | 99% | MN464135 | 30 |
| S3 | Saccharomyces cerevisiae | 100.00% | 567 | 100% | MN464136 | 9 |
| S4 | Saccharomyces cerevisiae | 100.00% | 570 | 100% | MN464137 | 1 |
| S5 | Saccharomyces cerevisiae | 100.00% | 567 | 100% | MN464138 | 2 |
| S6 | Saccharomyces cerevisiae | 99.63% | 542 | 100% | MN464139 | 4 |
| S7 | Saccharomyces cerevisiae | 99.65% | 565 | 100% | MN464140 | 17 |
| S8 | Saccharomyces cerevisiae | 99.82% | 571 | 100% | MN464141 | 4 |
| S9 | Saccharomyces cerevisiae | 100.00% | 573 | 99% | MN464142 | 5 |
| S10 | Saccharomyces cerevisiae | 99.47% | 565 | 100% | MN464143 | 2 |
| W1 | Wickerhamomyces anomalus | 100.00% | 558 | 100% | MN464144 | 5 |
| W2 | Wickerhamomyces anomalus | 100.00% | 562 | 100% | MN464145 | 2 |
| Strain | 2012 | 2013 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sampling Day | Sampling Day | |||||||||||||
| 0 | 2 | 3 | 6 | 10 | 22 | 30 | 0 | 1 | 2 | 4 | 7 | 14 | 30 | |
| Hanseniaspora uvarum H1 | 21 | 17 | ||||||||||||
| Hanseniaspora uvarum H2 | 57 | 40 | 50 | 29 | 14 | |||||||||
| Hanseniaspora uvarum H3 | 43 | 83 | 40 | 14 | 10 | |||||||||
| Metschnikowia sp. M1 | 32 | 17 | ||||||||||||
| Metschnikowia sp. M5 | 5 | |||||||||||||
| Pichia kudriavzevii P1 | 14 | |||||||||||||
| Pichia kudriavzevii P2 | 80 | |||||||||||||
| Pichia kudriavzevii P3 | 20 | |||||||||||||
| Pichia kudriavzevii P4 | 43 | 17 | 15 | |||||||||||
| Saccharomyces cerevisiae S1 | 20 | 60 | ||||||||||||
| Saccharomyces cerevisiae S2 | 14 | 20 | 16 | 57 | 43 | 20 | 65 | 60 | 51 | |||||
| Saccharomyces cerevisiae S3 | 29 | 33 | 32 | 15 | 20 | |||||||||
| Saccharomyces cerevisiae S5 | 20 | |||||||||||||
| Saccharomyces cerevisiae S7 | 28 | 20 | 20 | 49 | ||||||||||
| Wickerhamomyces anomalus W1 | 47 | |||||||||||||
| Strain | 2012 | 2013 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sampling Day | Sampling Day | |||||||||||||
| 0 | 2 | 3 | 6 | 10 | 22 | 30 | 0 | 1 | 2 | 4 | 7 | 14 | 30 | |
| Candida sake C | 11 | |||||||||||||
| Hanseniaspora uvarum H1 | 39 | 15 | ||||||||||||
| Hanseniaspora uvarum H2 | 50 | 80 | 43 | 25 | 54 | 33 | 38 | 33 | 20 | 17 | ||||
| Hanseniaspora uvarum H3 | 8 | 25 | ||||||||||||
| Metschnikowia sp. M1 | 39 | 20 | 31 | 67 | 54 | 50 | 20 | 16 | 20 | |||||
| Pichia kluyveri Pk | 50 | |||||||||||||
| Pichia kudriavzevii P2 | 6 | |||||||||||||
| Pichia kudriavzevii P3 | 25 | 18 | 8 | 17 | 20 | |||||||||
| Rhodotorula mucilaginosa R | 12 | |||||||||||||
| Saccharomyces cerevisiae S1 | 5 | 42 | 25 | 64 | 40 | 51 | 80 | |||||||
| Saccharomyces cerevisiae S2 | 29 | |||||||||||||
| Saccharomyces cerevisiae S5 | 9 | |||||||||||||
| Saccharomyces cerevisiae S6 | 14 | |||||||||||||
| Saccharomyces cerevisiae S7 | 14 | 17 | ||||||||||||
| Saccharomyces cerevisiae S8 | 8 | |||||||||||||
| Saccharomyces cerevisiae S9 | 9 | |||||||||||||
| Wickerhamomyces anomalus W2 | 13 | 16 | ||||||||||||
| Strain | 2012 | 2013 | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sampling Day | Sampling Day | ||||||||||||||
| 0 | 2 | 3 | 4 | 7 | 11 | 23 | 30 | 0 | 1 | 2 | 4 | 7 | 14 | 30 | |
| Hanseniaspora uvarum H1 | 52 | 40 | 16 | ||||||||||||
| Hanseniaspora uvarum H2 | 5 | 75 | 25 | 50 | 49 | 30 | 32 | 16 | |||||||
| Hanseniaspora uvarum H3 | 100 | 40 | 11 | 25 | 34 | ||||||||||
| Metschnikowia sp. M1 | 39 | 18 | |||||||||||||
| Metschnikowia sp. M3 | 16 | ||||||||||||||
| Metschnikowia sp. M6 | 16 | 20 | |||||||||||||
| Pichia kudriavzevii P2 | 4 | 29 | 10 | ||||||||||||
| Pichia kudriavzevii P3 | 17 | 50 | |||||||||||||
| Saccharomyces cerevisiae S1 | 30 | 16 | 17 | 14 | |||||||||||
| Saccharomyces cerevisiae S2 | 33 | 75 | 33 | 17 | 17 | 28 | 51 | 67 | |||||||
| Saccharomyces cerevisiae S3 | 34 | ||||||||||||||
| Saccharomyces cerevisiae S6 | 25 | 17 | 14 | ||||||||||||
| Saccharomyces cerevisiae S7 | 44 | 25 | 17 | 33 | 14 | 17 | 17 | ||||||||
| Saccharomyces cerevisiae S9 | 11 | 33 | |||||||||||||
| Saccharomyces cerevisiae S10 | 20 | ||||||||||||||
| Strain | 2012 | 2013 | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sampling Day | Sampling Day | ||||||||||||||
| 0 | 2 | 3 | 4 | 7 | 11 | 23 | 30 | 0 | 1 | 2 | 4 | 7 | 14 | 30 | |
| Hanseniaspora uvarum H1 | 20 | 20 | 20 | ||||||||||||
| Hanseniaspora uvarum H2 | 50 | 40 | 37 | 20 | 40 | 45 | 50 | 34 | 28 | 32 | |||||
| Hanseniaspora uvarum H3 | 20 | 20 | 10 | ||||||||||||
| Metschnikowia sp. M1 | 25 | 38 | 40 | 45 | 50 | 16 | 14 | ||||||||
| Metschnikowia sp. M2 | 20 | 25 | 10 | ||||||||||||
| Metschnikowia sp. M3 | 5 | 20 | |||||||||||||
| Metschnikowia sp. M4 | 5 | 40 | 12 | ||||||||||||
| Metschnikowia sp. M5 | 20 | 10 | |||||||||||||
| Nakazawaea ernobii N | 10 | ||||||||||||||
| Rhodothorula mucilaginosa R1 | 5 | ||||||||||||||
| Saccharomyces cerevisiae S1 | 10 | 17 | 14 | 34 | |||||||||||
| Saccharomyces cerevisiae S2 | 100 | 33 | 42 | ||||||||||||
| Saccharomyces cerevisiae S3 | 34 | ||||||||||||||
| Saccharomyces cerevisiae S4 | 13 | ||||||||||||||
| Saccharomyces cerevisiae S7 | 30 | 24 | |||||||||||||
| Saccharomyces cerevisiae S8 | 25 | 80 | 100 | ||||||||||||
| Saccharomyces cerevisiae S9 | 20 | ||||||||||||||
| Saccharomyces cerevisiae S10 | 13 | ||||||||||||||
| Wickerhamomyces anomalus W1 | 13 | ||||||||||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Skotniczny, M.; Satora, P.; Pańczyszyn, K.; Cioch-Skoneczny, M. Growth Dynamics and Diversity of Yeasts during Spontaneous Plum Mash Fermentation of Different Varieties. Foods 2020, 9, 1054. https://doi.org/10.3390/foods9081054
Skotniczny M, Satora P, Pańczyszyn K, Cioch-Skoneczny M. Growth Dynamics and Diversity of Yeasts during Spontaneous Plum Mash Fermentation of Different Varieties. Foods. 2020; 9(8):1054. https://doi.org/10.3390/foods9081054
Chicago/Turabian StyleSkotniczny, Magdalena, Paweł Satora, Katarzyna Pańczyszyn, and Monika Cioch-Skoneczny. 2020. "Growth Dynamics and Diversity of Yeasts during Spontaneous Plum Mash Fermentation of Different Varieties" Foods 9, no. 8: 1054. https://doi.org/10.3390/foods9081054
APA StyleSkotniczny, M., Satora, P., Pańczyszyn, K., & Cioch-Skoneczny, M. (2020). Growth Dynamics and Diversity of Yeasts during Spontaneous Plum Mash Fermentation of Different Varieties. Foods, 9(8), 1054. https://doi.org/10.3390/foods9081054





