Food and COVID-19: Preventive/Co-therapeutic Strategies Explored by Current Clinical Trials and in Silico Studies
Abstract
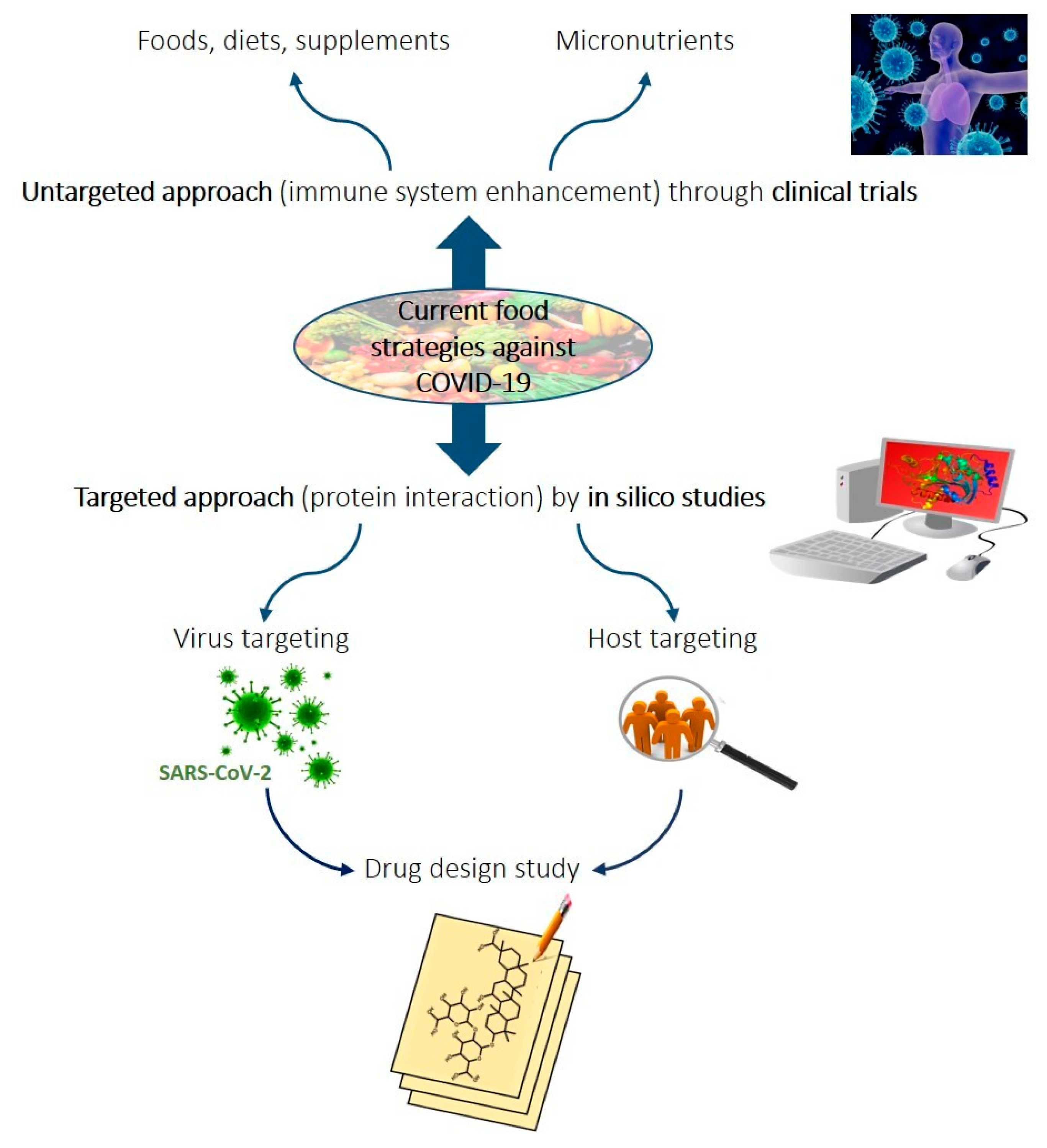
1. Introduction
2. Methodology
3. Food as an Immune System Enhancer in Current Clinical Trials
4. Food Compounds in Preliminary in Silico Studies
5. Conclusions
Supplementary Materials
Funding
Acknowledgments
Conflicts of Interest
References
- Rock, C.L.; Jacob, R.; Bowen, P.E. Update on the biological characteristics of the antioxidant micronutrients: Vitamin C, vitamin E, and the carotenoids. J. Am. Diet. Assoc. 1996, 96, 693–702. [Google Scholar] [CrossRef]
- Hänsch, R.; Mendel, R.R. Physiological functions of mineral micronutrients (Cu, Zn, Mn, Fe, Ni, Mo, B, Cl). Curr. Opin. Plant Biol. 2009, 12, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Shahidi, F.; Ambigaipalan, P. Phenolics and polyphenolics in foods, beverages and spices: Antioxidant activity and health effects—A review. J. Funct. Foods 2015, 18, 820–897. [Google Scholar] [CrossRef]
- Brennen, A.J.S.; Simon, F.M.; Howard, P.N.; Nielsen, R.K. Types, Sources, and Claims of COVID-19 Misinformation; Oxford Univ. Press: Oxford, UK, 2020. [Google Scholar]
- Banerjee, A.; Kulcsar, K.; Misra, V.; Frieman, M.; Mossman, K. Bats and coronaviruses. Viruses 2019, 11, 41. [Google Scholar] [CrossRef] [PubMed]
- Woo, P.C.Y.; Huang, Y.; Lau, S.K.P.; Yuen, K.Y. Coronavirus genomics and bioinformatics analysis. Viruses 2010, 2, 1805–1820. [Google Scholar] [CrossRef] [PubMed]
- Su, S.; Wong, G.; Shi, W.; Liu, J.; Lai, A.C.K.; Zhou, J.; Liu, W.; Bi, Y.; Gao, G.F. Epidemiology, Genetic Recombination, and Pathogenesis of Coronaviruses. Trends Microbiol. 2016, 24, 490–502. [Google Scholar] [CrossRef]
- World Health Organization. Naming the Coronavirus Disease (COVID-19) and the Virus That Causes It. Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/technical-guidance/naming-the-coronavirus-disease-(covid-2019)-and-the-virus-that-causes-it (accessed on 14 April 2020).
- Amodio, E.; Vitale, F.; Cimino, L.; Casuccio, A.; Tramuto, F. Outbreak of Novel Coronavirus (SARS-CoV-2): First Evidences From International Scientific Literature and Pending Questions. Healthcare 2020, 8, 51. [Google Scholar] [CrossRef]
- World Health Organization. Middle East respiratory Syndrome Coronavirus (MERS-CoV). Available online: https://www.who.int/emergencies/mers-cov/en/ (accessed on 23 April 2020).
- World Health Organization. Consensus Document on the Epidemiology of Severe Acute Respiratory Syndrome (SARS). Available online: https://apps.who.int/iris/handle/10665/70863 (accessed on 23 April 2020).
- World Health Organization. Coronavirus Disease (COVID-2019) Situation Reports. Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/situation-reports (accessed on 23 April 2020).
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef]
- Lupia, T.; Scabini, S.; Mornese Pinna, S.; Di Perri, G.; De Rosa, F.G.; Corcione, S. 2019 novel coronavirus (2019-nCoV) outbreak: A new challenge. J. Glob. Antimicrob. Resist. 2020, 21, 22–27. [Google Scholar] [CrossRef]
- World Health Organization. The Top 10 Causes of Death. Available online: https://www.who.int/en/news-room/fact-sheets/detail/the-top-10-causes-of-death (accessed on 18 April 2020).
- Zhang, L.; Liu, Y. Potential interventions for novel coronavirus in China: A systematic review. J. Med. Virol. 2020, 92, 479–490. [Google Scholar] [CrossRef]
- Battistini, R.; Rossini, I.; Ercolini, C.; Goria, M.; Callipo, M.R.; Maurella, C.; Pavoni, E.; Serracca, L. Antiviral Activity of Essential Oils Against Hepatitis A Virus in Soft Fruits. Food Environ. Virol. 2019, 11, 90–95. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Xu, Y.; Liu, J.; Yang, B.; Yang, C.; Zhang, M.; Dong, X. Drug resistance evolution in patients with human immunodeficiency virus-1 under long-term antiretroviral treatment-failure in Yunnan Province, China. Virol. J. 2019, 16, 5. [Google Scholar] [CrossRef] [PubMed]
- Pawlotsky, J.M. Hepatitis C Virus Resistance to Direct-Acting Antiviral Drugs in Interferon-Free Regimens. Gastroenterology 2016, 151, 70–86. [Google Scholar] [CrossRef]
- Wu, R.; Wang, L.; Kuo, H.C.D.; Shannar, A.; Peter, R.; Chou, P.J.; Li, S.; Hudlikar, R.; Liu, X.; Liu, Z.; et al. An Update on Current Therapeutic Drugs Treating COVID-19. Curr. Pharmacol. Rep. 2020, 6, 56–70. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.T.; Sarkar, C.; El-Kersh, D.M.; Jamaddar, S.; Uddin, S.J.; Shilpi, J.A.; Mubarak, M.S. Natural products and their derivatives against coronavirus: A review of the non-clinical and pre-clinical data. Phyther. Res. 2020. [Google Scholar] [CrossRef] [PubMed]
- Orhan, I.E.; Senol Deniz, F.S. Natural Products as Potential Leads Against Coronaviruses: Could They be Encouraging Structural Models Against SARS-CoV-2? Nat. Prod. Bioprospect. 2020. [Google Scholar] [CrossRef]
- Stahlmann, R.; Lode, H. Medication for COVID-19-an Overview of Approaches Currently Under Study. Dtsch. Arztebl. Int. 2020, 117, 213–219. [Google Scholar] [CrossRef]
- Franquet, T. Respiratory infection in the AIDS and immunocompromised patient. Eur. Radiol. Suppl. 2004, 14, 21–33. [Google Scholar] [CrossRef]
- Simon, A.K.; Hollander, G.A.; McMichael, A. Evolution of the immune system in humans from infancy to old age. Proc. R. Soc. B Biol. Sci. 2015, 282, 20143085. [Google Scholar] [CrossRef]
- Zhang, L.; Lin, D.; Sun, X.; Curth, U.; Drosten, C.; Sauerhering, L.; Becker, S.; Rox, K.; Hilgenfeld, R. Crystal structure of SARS-CoV-2 main protease provides a basis for design of improved α-ketoamide inhibitors. Science 2020, eabb3405. [Google Scholar] [CrossRef]
- Vellingiri, B.; Jayaramayya, K.; Iyer, M.; Narayanasamy, A.; Govindasamy, V.; Giridharan, B.; Ganesan, S.; Venugopal, A.; Venkatesan, D.; Ganesan, H.; et al. COVID-19: A promising cure for the global panic. Sci. Total Environ. 2020, 725, 138277. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Liu, Y.; Yang, Y.; Zhang, P.; Zhong, W.; Wang, Y.; Wang, Q.; Xu, Y.; Li, M.; Li, X.; et al. Analysis of therapeutic targets for SARS-CoV-2 and discovery of potential drugs by computational methods. Acta Pharm. Sin. B 2020. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.T.; Hsu, W.C.; Lin, C.C. Antiviral natural products and herbal medicines. J. Tradit. Complement. Med. 2014, 4, 24–35. [Google Scholar] [CrossRef] [PubMed]
- Naithani, R.; Huma, L.; Holland, L.; Shukla, D.; McCormick, D.; Mehta, R.; Moriarty, R. Antiviral Activity of Phytochemicals: A Comprehensive Review. Mini-Rev. Med. Chem. 2008, 8, 1106–1133. [Google Scholar] [CrossRef]
- Falcó, I.; Randazzo, W.; Rodríguez-Díaz, J.; Gozalbo-Rovira, R.; Luque, D.; Aznar, R.; Sánchez, G. Antiviral activity of aged green tea extract in model food systems and under gastric conditions. Int. J. Food Microbiol. 2019, 292, 101–106. [Google Scholar] [CrossRef]
- U.S. National Library of Medicine. ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ct2/home (accessed on 24 May 2020).
- Maggini, S.; Pierre, A.; Calder, P.C. Immune function and micronutrient requirements change over the life course. Nutrients 2018, 10, 1531. [Google Scholar] [CrossRef]
- Gombart, A.F.; Pierre, A.; Maggini, S. A review of micronutrients and the immune system–working in harmony to reduce the risk of infection. Nutrients 2020, 12, 236. [Google Scholar] [CrossRef]
- Maggini, S.; Wintergerst, E.S.; Beveridge, S.; Hornig, D.H. Selected vitamins and trace elements support immune function by strengthening epithelial barriers and cellular and humoral immune responses. Proc. Br. J. Nutr. 2007, 98, 29–35. [Google Scholar] [CrossRef]
- Martineau, A.R.; Jolliffe, D.A.; Hooper, R.L.; Greenberg, L.; Aloia, J.F.; Bergman, P.; Dubnov-Raz, G.; Esposito, S.; Ganmaa, D.; Ginde, A.A.; et al. Vitamin D supplementation to prevent acute respiratory tract infections: Systematic review and meta-analysis of individual participant data. BMJ 2017, 356. [Google Scholar] [CrossRef]
- Taylor, C.E.; Camargo, C.A. Impact of micronutrients on respiratory infections. Nutr. Rev. 2011, 69, 259–269. [Google Scholar] [CrossRef]
- Hemilä, H.; Douglas, R.M. Vitamin C and acute respiratory infections. Int. J. Tuberc. Lung Dis. 1999, 3, 756–761. [Google Scholar] [PubMed]
- Hemilä, H. Vitamin C Supplementation and Respiratory Infections: A Systematic Review. Mil. Med. 2004, 169, 920–925. [Google Scholar] [CrossRef] [PubMed]
- Fowler, A.A., III; Kim, C.; Lepler, L.; Malhotra, R.; Debesa, O.; Natarajan, R.; Fisher, B.J.; Syed, A.; DeWilde, C.; Priday, A.; et al. Intravenous vitamin C as adjunctive therapy for enterovirus/rhinovirus induced acute respiratory distress syndrome. World J. Crit. Care Med. 2017, 6, 85. [Google Scholar] [CrossRef] [PubMed]
- Urashima, M.; Segawa, T.; Okazaki, M.; Kurihara, M.; Wada, Y.; Ida, H. Randomized trial of vitamin D supplementation to prevent seasonal influenza A in schoolchildren. Am. J. Clin. Nutr. 2010, 91, 1255–1260. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Zinc Supplementation to Improve Treatment Outcomes Among Children Diagnosed with Respiratory Infections. Available online: https://www.who.int/elena/titles/bbc/zinc_pneumonia_children/en/ (accessed on 3 July 2020).
- Sazawal, S.; Black, R.E.; Jalla, S.; Mazumdar, S.; Sinha, A.; Bhan, M.K. Zinc supplementation reduces the incidence of acute lower respiratory infections in infants and preschool children: A double-blind, controlled trial. Pediatrics 1998, 102, 1–5. [Google Scholar] [CrossRef]
- Al-Saady, N.M.; Blackmore, C.M.; Bennett, E.D. High fat, low carbohydrate, enteral feeding lowers PaCO2 and reduces the period of ventilation in artificially ventilated patients. Intensive Care Med. 1989, 15, 290–295. [Google Scholar] [CrossRef]
- Yamanashi, T.; Iwata, M.; Kamiya, N.; Tsunetomi, K.; Kajitani, N.; Wada, N.; Iitsuka, T.; Yamauchi, T.; Miura, A.; Pu, S.; et al. Beta-hydroxybutyrate, an endogenic NLRP3 inflammasome inhibitor, attenuates stress-induced behavioral and inflammatory responses. Sci. Rep. 2017, 7, 1–11. [Google Scholar]
- Chen, H.; Zhuo, Q.; Yuan, W.; Wang, J.; Wu, T. Vitamin A for preventing acute lower respiratory tract infections in children up to seven years of age. Cochrane Database Syst. Rev. 2008, 1–46. [Google Scholar] [CrossRef]
- Sempértegui, F.; Estrella, B.; Camaniero, V.; Betancourt, V.; Izurieta, R.; Ortiz, W.; Fiallo, E.; Troya, S.; Rodríguez, A.; Griffiths, J.K. The beneficial effects of weekly low-dose vitamin A supplementation on acute lower respiratory infections and diarrhea in Ecuadorian children. Pediatrics 1999, 104. [Google Scholar] [CrossRef]
- Griffiths, J.K. The vitamin A paradox. J. Pediatr. 2000, 137, 604–607. [Google Scholar] [CrossRef]
- Gay, R.; Meydani, S.N. The Effects of Vitamin E, Vitamin B6, and Vitamin B12 on Immune Function. Nutr. Clin. Care 2001, 4, 188–198. [Google Scholar] [CrossRef]
- Strand, T.A.; Taneja, S.; Bhandari, N.; Refsum, H.; Ueland, P.M.; Gjessing, H.K.; Bahl, R.; Schneede, J.; Bhan, M.K.; Sommerfelt, H. Folate, but not vitamin B-12 status, predicts respiratory morbidity in north Indian children. Am. J. Clin. Nutr. 2007, 86, 139–144. [Google Scholar] [CrossRef] [PubMed]
- De la Fuente, M.; Hernanz, A.; Guayerbas, N.; Victor, V.M.; Arnalich, F. Vitamin E ingestion improves several immune functions in elderly men and women. Free Radic. Res. 2008, 42, 272–280. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.H.; Lee, S.J.; Lee, M.K.; Lee, W.Y.; Yong, S.J.; Kim, S.H. Serum selenium levels in patients with respiratory diseases: A prospective observational study. J. Thorac. Dis. 2016, 8, 2068–2078. [Google Scholar] [CrossRef]
- De Silva, A.; Atukorala, S.; Weerasinghe, I.; Ahluwalia, N. Iron supplementation improves iron status and reduces morbidity in children with or without upper respiratory tract infections: A randomized controlled study in Colombo, Sri Lanka. Am. J. Clin. Nutr. 2003, 77, 234–241. [Google Scholar] [CrossRef]
- Parada, J.; Aguilera, J.M. Food microstructure affects the bioavailability of several nutrients. J. Food Sci. 2007, 72, 21–32. [Google Scholar] [CrossRef]
- Gayoso, L.; Claerbout, A.S.; Calvo, M.I.; Cavero, R.Y.; Astiasarán, I.; Ansorena, D. Bioaccessibility of rutin, caffeic acid and rosmarinic acid: Influence of the in vitro gastrointestinal digestion models. J. Funct. Foods 2016, 26, 428–438. [Google Scholar] [CrossRef]
- Pressman, P.; Clemens, R.A.; Hayes, A.W. Bioavailability of micronutrients obtained from supplements and food. Toxicol. Res. Appl. 2017, 1, 239784731769636. [Google Scholar] [CrossRef]
- Stanger, M.J.; Thompson, L.A.; Young, A.J.; Lieberman, H.R. Anticoagulant activity of select dietary supplements. Nutr. Rev. 2012, 70, 107–117. [Google Scholar] [CrossRef]
- Olas, B. Anti-Aggregatory Potential of Selected Vegetables—Promising Dietary Components for the Prevention and Treatment of Cardiovascular Disease. Adv. Nutr. 2019, 10, 280–290. [Google Scholar] [CrossRef]
- Tang, N.; Bai, H.; Chen, X.; Gong, J.; Li, D.; Sun, Z. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J. Thromb. Haemost. 2020, 18, 1094–1099. [Google Scholar] [CrossRef] [PubMed]
- Driggin, E.; Madhavan, M.V.; Bikdeli, B.; Chuich, T.; Laracy, J.; Biondi-Zoccai, G.; Brown, T.S.; Der Nigoghossian, C.; Zidar, D.A.; Haythe, J.; et al. Cardiovascular Considerations for Patients, Health Care Workers, and Health Systems During the COVID-19 Pandemic. J. Am. Coll. Cardiol. 2020, 75, 2352–2371. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Pandemic Influenza Vaccine Manufacturing Process and Timeline. Available online: https://www.who.int/csr/disease/swineflu/notes/h1n1_vaccine_20090806/en/ (accessed on 3 July 2020).
- Ostaszewski, M.; Mazein, A.; Gillespie, M.E.; Kuperstein, I.; Niarakis, A.; Hermjakob, H.; Pico, A.R.; Willighagen, E.L.; Evelo, C.T.; Hasenauer, J.; et al. COVID-19 Disease Map, building a computational repository of SARS-CoV-2 virus-host interaction mechanisms. Sci. Data 2020, 7, 8–11. [Google Scholar] [CrossRef]
- Lu, R.; Zhao, X.; Li, J.; Niu, P.; Yang, B.; Wu, H.; Wang, W.; Song, H.; Huang, B.; Zhu, N.; et al. Genomic characterisation and epidemiology of 2019 novel coronavirus: Implications for virus origins and receptor binding. Lancet 2020, 395, 565–574. [Google Scholar] [CrossRef]
- Fani, M.; Teimoori, A.; Ghafari, S. Comparison of the COVID-2019 (SARS-CoV-2) pathogenesis with SARS-CoV and MERS-CoV infections. Future Virol. 2020, 10, 280–290. [Google Scholar] [CrossRef]
- Hoffmann, M.; Kleine-Weber, H.; Schroeder, S.; Krüger, N.; Herrler, T.; Erichsen, S.; Schiergens, T.S.; Herrler, G.; Wu, N.H.; Nitsche, A.; et al. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell 2020, 181, 271–280. [Google Scholar] [CrossRef]
- Milanetti, E.; Miotto, M.; Di Rienzo, L.; Monti, M.; Gosti, G.; Ruocco, G. In-Silico evidence for two receptors based strategy of SARS-CoV-2. BioRxiv 2020, 11107, under review. [Google Scholar] [CrossRef]
- Huynh, T.; Wang, H.; Luan, B. In Silico Exploration of Molecular Mechanism of Clinically Oriented Drugs for Possibly Inhibiting SARS-CoV-2’s Main Protease. J. Phys. Chem. Lett. 2020. [Google Scholar] [CrossRef]
- U.S. Food and Drog Administration. The Drug Development Process. Available online: https://www.fda.gov/patients/learn-about-drug-and-device-approvals/drug-development-process (accessed on 3 July 2020).
- Öztürk, H.; Özgür, A.; Ozkirimli, E. DeepDTA: Deep drug-target binding affinity prediction. Proc. Bioinf. 2018, 34, 821–829. [Google Scholar] [CrossRef]
- Joshi, R.S.; Jagdale, S.S.; Bansode, S.B.; Shankar, S.S.; Tellis, M.B.; Pandya, V.K.; Chugh, A.; Giri, A.P.; Kulkarni, M.J. Discovery of Potential Multi-Target-Directed Ligands by Targeting Host-specific SARS-CoV-2 Structurally Conserved Main Protease. J. Biomol. Struct. Dyn. 2020, 1–16. [Google Scholar] [CrossRef]
- Chen, H.; Du, Q. Potential natural compounds for preventing SARS-CoV-2 (2019-nCoV) infection. Preprints 2020, 2020010358, under review. [Google Scholar] [CrossRef]
- Cinatl, J.; Morgenstern, B.; Bauer, G.; Chandra, P.; Rabenau, H.; Doerr, H.W. Glycyrrhizin, an active component of liquorice roots, and replication of SARS-associated coronavirus. Lancet 2003, 361, 2045–2046. [Google Scholar] [CrossRef]
- Islam, R.; Parves, M.R.; Paul, A.S.; Uddin, N.; Rahman, M.S.; Al Mamun, A.; Hossain, M.N.; Ali, M.A.; Halim, M.A. A molecular modeling approach to identify effective antiviral phytochemicals against the main protease of SARS-CoV-2. J. Biomol. Struct. Dyn. 2020. [Google Scholar] [CrossRef] [PubMed]
- Hashem, H.E. IN Silico Approach of Some Selected Honey Constituents as SARS-CoV-2 Main Protease (COVID-19) Inhibitors. Eurasian J. Med. Oncol. 2020. [Google Scholar] [CrossRef]
- Weng, J.R.; Lin, C.S.W.S.W.; Lai, H.C.; Lin, Y.P.; Wang, C.Y.; Tsai, Y.C.; Wu, K.C.; Huang, S.H.; Lin, C.S.W.S.W. Antiviral activity of Sambucus FormosanaNakai ethanol extract and related phenolic acid constituents against human coronavirus NL63. Virus Res. 2019, 273, 197767. [Google Scholar] [CrossRef] [PubMed]
- Wahedi, H.M.; Ahmad, S.; Abbasi, S.W. Stilbene-based natural compounds as promising drug candidates against COVID-19. J. Biomol. Struct. Dyn. 2020, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.Q.; Li, Z.L.; Zhao, W.J.; Wen, R.X.; Meng, Q.W.; Zeng, Y. Synthesis of stilbene derivatives with inhibition of SARS coronavirus replication. Eur. J. Med. Chem. 2006, 41, 1084–1089. [Google Scholar] [CrossRef]
- Khaerunnisa, S.; Kurniawan, H.; Awaluddin, R.; Suhartati, S. Potential Inhibitor of COVID-19 Main Protease (M pro) from Several Medicinal Plant Compounds by Molecular Docking Study. Preprints 2020, 1–14, under review. [Google Scholar] [CrossRef]
- Schwarz, S.; Sauter, D.; Wang, K.; Zhang, R.; Sun, B.; Karioti, A.; Bilia, A.R.; Efferth, T.; Schwarz, W. Kaempferol derivatives as antiviral drugs against the 3a channel protein of coronavirus. Planta Med. 2014, 80, 177–182. [Google Scholar] [CrossRef]
- Jena, A.B. Catechin and Curcumin interact with corona (2019-nCoV/SARS-CoV2 ) viral S protein and ACE2 of human cell membrane. Insights Comput. Study Implic. Interv. 2020, 1–19, under review. [Google Scholar] [CrossRef]
- Park, J.Y.; Jeong, H.J.; Kim, J.H.; Kim, Y.M.; Park, S.J.; Kim, D.; Park, K.H.; Lee, W.S.; Ryu, Y.B. Diarylheptanoids from Alnus japonica inhibit papain-like protease of severe acute respiratory syndrome coronavirus. Biol. Pharm. Bull. 2012, 35, 2036–2042. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Lee, J.M.; Lee, N.R.; Kim, D.E.; Jeong, Y.J.; Chong, Y. Investigation of the pharmacophore space of Severe Acute Respiratory Syndrome coronavirus (SARS-CoV) NTPase/helicase by dihydroxychromone derivatives. Bioorganic Med. Chem. Lett. 2009, 19, 4538–4541. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.K.; Lo, S.C.; Wang, Y.S.; Hou, M.H. Recent insights into the development of therapeutics against coronavirus diseases by targeting N protein. Drug Discov. Today 2016, 21, 562–572. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.W.; Tsai, F.J.; Tsai, C.H.; Lai, C.C.; Wan, L.; Ho, T.Y.; Hsieh, C.C.; Chao, P.D.L. Anti-SARS coronavirus 3C-like protease effects of Isatis indigotica root and plant-derived phenolic compounds. Antivir. Res. 2005, 68, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Meneguzzo, F.; Ciriminna, R.; Zabini, F.; Pagliaro, M. Review of evidence available on hesperidin-rich products as potential tools against COVID-19 and hydrodynamic cavitation-based extraction as a method of increasing their production. Processes 2020, 8, 549. [Google Scholar] [CrossRef]
- Thuy, B.T.P.; My, T.T.A.; Hai, N.T.T.; Hieu, L.T.; Hoa, T.T.; Thi Phuong Loan, H.; Triet, N.T.; Van Anh, T.T.; Quy, P.T.; Van Tat, P.; et al. Investigation into SARS-CoV-2 Resistance of Compounds in Garlic Essential Oil. ACS Omega 2020, 5, 8312–8320. [Google Scholar] [CrossRef]
- Prasanth, D.S.N.B.K.; Murahari, M.; Chandramohan, V.; Panda, P.; Atmakuri, L.R.; Guntupalli, C. In silico identification of potential inhibitors from Cinnamon against main protease and spike glycoprotein of SARS CoV-2. J. Biomol. Struct. Dyn. 2020, 1–15. [Google Scholar] [CrossRef]
- Zhuang, M.; Jiang, H.; Suzuki, Y.; Li, X.; Xiao, P.; Tanaka, T.; Ling, H.; Yang, B.; Saitoh, H.; Zhang, L.; et al. Procyanidins and butanol extract of Cinnamomi Cortex inhibit SARS-CoV infection. Antivir. Res. 2009, 82, 73–81. [Google Scholar] [CrossRef]
- Yu, M.S.; Lee, J.; Lee, J.M.; Kim, Y.; Chin, Y.W.; Jee, J.G.; Keum, Y.S.; Jeong, Y.J. Identification of myricetin and scutellarein as novel chemical inhibitors of the SARS coronavirus helicase, nsP13. Bioorganic Med. Chem. Lett. 2012, 22, 4049–4054. [Google Scholar] [CrossRef]
- Keum, Y.S.; Jeong, Y.J. Development of chemical inhibitors of the SARS coronavirus: Viral helicase as a potential target. Biochem. Pharmacol. 2012, 84, 1351–1358. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, H.; Chen, L.; Shan, L.; Fan, G.; Gao, X. Liquorice, a unique “guide drug” of traditional Chinese medicine: A review of its role in drug interactions. J. Ethnopharmacol. 2013, 150, 781–790. [Google Scholar] [CrossRef]
- Fiore, C.; Eisenhut, M.; Krausse, R.; Ragazzi, E.; Pellati, D.; Armanini, D.; Bielenberg, J. Antiviral effects of Glycyrrhiza species. Phyther. Res. 2008, 22, 141–148. [Google Scholar] [CrossRef]
- Wang, L.; Yang, R.; Yuan, B.; Liu, Y.; Liu, C. The antiviral and antimicrobial activities of licorice, a widely-used Chinese herb. Acta Pharm. Sin. B 2015, 5, 310–315. [Google Scholar] [CrossRef] [PubMed]
- Carino, A.; Moraca, F.; Fiorillo, B.; Marcanó, S.; Sepe, V.; Biagioli, M.; Finamore, C.; Bozza, S.; Francisci, D.; Distrutti, E.; et al. Hijacking SARS-Cov-2/ACE2 receptor interaction by natural and semi-synthetic steroidal agents acting on functional pockets on receptor binding region. BioRxiv 2020. [Google Scholar] [CrossRef]
- Koech, R.K.; Wanyoko, J.; Wachira, F. Antioxidant, antimicrobial and synergistic activities of tea polyphenols. Int. J. Infect. Dis. 2014, 21, 98. [Google Scholar] [CrossRef]
- Meinhart, A.D.; Damin, F.M.; Caldeirão, L.; de Jesus Filho, M.; da Silva, L.C.; da Silva Constant, L.; Filho, J.T.; Wagner, R.; Godoy, H.T. Chlorogenic and caffeic acids in 64 fruits consumed in Brazil. Food Chem. 2019, 286, 51–63. [Google Scholar] [CrossRef]
- Francioso, A.; Mastromarino, P.; Masci, A.; d’Erme, M.; Mosca, L. Chemistry, Stability and Bioavailability of Resveratrol. Med. Chem. 2014, 10, 237–245. [Google Scholar] [CrossRef]
- Tian, B.; Liu, J. Resveratrol: A review of plant sources, synthesis, stability, modification and food application. J. Sci. Food Agric. 2020, 100, 1392–1404. [Google Scholar] [CrossRef]
- Filardo, S.; Di Pietro, M.; Mastromarino, P.; Sessa, R. Therapeutic potential of resveratrol against emerging respiratory viral infections. Pharmacol. Ther. 2020, 214, 107613. [Google Scholar] [CrossRef]
- Pezet, R.; Perret, C.; Jean-Denis, J.B.; Tabacchi, R.; Gindro, K.; Viret, O. δ-viniferin, a resveratrol dehydrodimer: One of the major stilbenes synthesized by stressed grapevine leaves. J. Agric. Food Chem. 2003. [Google Scholar] [CrossRef]
- Lee, S.; Mailar, K.; Il Kim, M.; Park, M.; Kim, J.; Min, D.H.; Heo, T.H.; Bae, S.K.; Choi, W.; Lee, C. Plant-derived purification, chemical synthesis, and in vitro/in vivo evaluation of a resveratrol dimer, viniferin, as an HCV Replication inhibitor. Viruses 2019, 11, 890. [Google Scholar] [CrossRef] [PubMed]
- Hossen, M.J.; Uddin, M.B.; Ahmed, S.S.U.; Yu, Z.L.; Cho, J.Y. Kaempferol: Review on natural sources and bioavailability. In Kaempferol: Biosynthesis, Food Sources and Therapeutic Uses; Nova Science Publishers: New York, NY, USA, 2016; pp. 101–150. ISBN 9781634858588. [Google Scholar]
- Dixit, P. Medicinal Properties of Garlic: A Review; Anusandhaan-Vigyaan Shodh Patrika, B.S.N.V. Post Graduate College: Lucknow, India, 2018; Volume 6. [Google Scholar]
- Swamy, M.K.; Akhtar, M.S.; Sinniah, U.R. Antimicrobial properties of plant essential oils against human pathogens and their mode of action: An updated review. Evidence-based Complement. Altern. Med. 2016, 2016, 1–21. [Google Scholar]
- Bayan, L.; Koulivand, P.H.; Gorji, A. Garlic: A review of potential therapeutic effects. Avicenna J. Phytomed. 2014, 4, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Kumari, R.; Mishra, S. Pharmacological properties and their medicinal uses of Cinnamomum: A review. J. Pharm. Pharmacol. 2019, 71, 1735–1761. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations (FAO). Maintaining a Healthy Diet during the COVID-19 Pandemic. Available online: http://www.fao.org/documents/card/en/c/ca8380en/ (accessed on 2 July 2020).
| №. | Treatment | Brief Trial Description ⱡ ,$,* | |
|---|---|---|---|
| Micronutrients | |||
| 1 | Vitamin C Zinc gluconate | Aim | Reduce symptom duration |
| Enrolment | 18 years and older, COVID-19-positive, 520 participants | ||
| Intervention |
| ||
| 2 | Vitamin C | Aim | Reduce mortality and secondary symptoms |
| Enrolment | All ages, COVID-19-positive, 500 participants | ||
| Intervention |
| ||
| 3 | Zinc gluconate Vitamin D3 | Aim | Reduce inflammatory reaction, which worsens acute respiratory distress syndrome |
| Enrolment | 60 years and older, COVID-19-positive, 3140 participants ¥ | ||
| Intervention |
| ||
| 4 | Vitamin D | Aim | Improve hard endpoints related to COVID-19 deleterious consequences |
| Enrolment | 40 years to 70 years, COVID-19-positive, 200 participants | ||
| Intervention |
| ||
| 5 | Hydroxychloroquine Vitamin C Vitamin D Zinc | Aim | Determine whether the combined therapy prevents COVID-19 symptoms ⴕ |
| Enrolment | 18 years and older, FM, COVID-19-negative: high-risk individuals, 600 participants | ||
| Intervention |
| ||
| 6 | Hydroxychloroquine Azithromycin Vitamin C Vitamin D Zinc | Aim | Determine whether the combined therapy can treat COVID-19 infection |
| Enrolment | 18 years and older, COVID-19-positive, 600 participants | ||
| Intervention |
| ||
| 7 | Vitamin C | Aim | Increase ventilator-free days, acute-inflammation-free days and organ-failure-free days |
| Enrolment | 18 years and older, FM, COVID-19-positive, 200 participants | ||
| Intervention |
| ||
| 8 | Vitamin D3 | Aim | Improve the prognosis of older patients |
| Enrolment | 70 years and older, FM, COVID-19-positive, 260 participants ¥ | ||
| Intervention |
| ||
| 9 | Vitamin C | Aim | Improve the prognosis of patients |
| Enrolment | 18 years and older, COVID-19-positive, 140 participants | ||
| Intervention |
| ||
| 10 | Vitamin C | Aim | Reduce the risk of respiratory failure requiring mechanical ventilation |
| Enrolment | 18 years to 99 years, FM, COVID-19-positive, 20 participants | ||
| Intervention |
| ||
| 11 | Vitamin C Active comparator treatment (hydroxychloroquine, azithromycin, zinc citrate, vitamin D3 and vitamin B12) | Aim | Prevent COVID-19 progression |
| Enrolment | 18 years and older, COVID-19-positive, 200 participants | ||
| Intervention |
| ||
| 12 | Vitamin D2 Vitamin D3 | Aim | Determine the efficacy of vitamin D in patients |
| Enrolment | 17 years and older, FM, COVID-19-positive, 64 participants | ||
| Intervention |
| ||
| 13 | Calcifediol | Aim | Study the preventive and therapeutic effects of oral calcifediol ⴕ |
| Enrolment | 18 years to 75 years, COVID-19-negative and at high risk of acquiring COVID-19, or at risk for its morbidity and mortality, 1500 participants | ||
| Intervention |
| ||
| 14 | Calcifediol | Aim | Reduce the development of COVID-19 and the worsening of the various syndrome phases |
| Enrolment | 18 years to 90 years, COVID-19-positive, 1008 participants | ||
| Intervention |
| ||
| 15 | Vitamin C | Aim | Evaluate the safety and efficacy of ascorbic acid infusions in COVID-19 treatment |
| Enrolment | 18 years and older, COVID-19-positive, 66 participants | ||
| Intervention |
| ||
| 16 | Aspirin Vitamin D | Aim | Test the hypothesis that treatment with aspirin and vitamin D in COVID-19 can mitigate the prothrombotic state and reduce hospitalization rates |
| Enrolment | 18 years and older, FM, COVID-19-positive, 1080 participants | ||
| Intervention |
| ||
| Foods/diets | |||
| 17 | Natural honey | Aim | Study the efficacy of natural honey in patient treatment |
| Enrolment | 5 years to 75 years, COVID-19-positive, 1000 participants | ||
| Intervention |
| ||
| 18 | Natural honey Black cumin | Aim | Reduce COVID-19 symptoms |
| Enrolment | 18 years and older, COVID-19-positive, 30 participants | ||
| Intervention |
| ||
| 19 | Ketogenic diet | Aim | Improve gas exchange, reduce inflammation and duration of mechanical ventilation |
| Enrolment | 18 years to 80 years, COVID-19-positive, 15 participants | ||
| Intervention |
| ||
| Other supplements | |||
| 20 | Nutritional supplement enriched in eicosapentaenoic acid, gamma linolenic acid, and antioxidants | Aim | Reduce COVID-19 severity with more preservation of the nutritional status |
| Enrolment | 18 years to 65 years, COVID-19-positive, 30 participants | ||
| Intervention |
| ||
| 21 | Resistant potato starch Nonresistant corn starch | Aim | Determine the efficacy of resistant potato starch in reducing the need for hospitalization |
| Enrolment | 19 years and older, COVID-19-positive, 1300 participants | ||
| Intervention |
| ||
| 22 | Gum arabic Pectin | Aim | Study the efficacy of gum arabic as an immunomodulator and anti-inflammatory agent |
| Enrolment | 5 years to 90 years, COVID-19-positive, 110 participants | ||
| Intervention |
| ||
| 23 | Quercetin | Aim | Evaluate the possible role of quercetin on prophylaxis and treatment of COVID-19 ⴕ |
| Enrolment | 18 years and older, COVID-19-negative and -positive, 50 participants | ||
| Intervention |
| ||
| Compound | Chemical Class | Food Source | In Silico Anti-COVID-19 Target | Previous in Vitro Activity on Other HCoVs |
|---|---|---|---|---|
| Glycyrrhizin | Triterpene Saponin | Glycyrrhiza glabra | ACE2 [71] | SARS-CoV [72] |
| Glabridin | Isoflavane | Glycyrrhiza glabra | Spike S protein [73] | |
| Caffeic acid | Phenolic acid | Fruits, vegetables, coffee, propolis | Mpro [74] | NL63 [75] |
| Caffeic acid phenylethyl ester | Phenolic ester | Honey, propolis | Mpro [74] | |
| Chrysin | Flavone | Honey, propolis | Mpro [74] | |
| Galangin | Flavonol | Honey, propolis | Mpro [74] | |
| Resveratrol | Flavonol | Grapes, peanut, blueberry, bilberry, Cranberry | ACE2 [76] | SARS-CoV [77] |
| Kaempferol | Stilbenoid | Tea, spinach, broccoli, Cabbage | Mpro [78] | SARS-CoV [79] |
| Curcumin | Diarylheptanoid | Turmeric | Mpro [78] ACE2 [80] Spike S protein [80] | SARS-CoV [81] |
| Demethoxycurcumin | Diarylheptanoid | Turmeric | Mpro [78] | |
| Quercetin | Flavonol | Fruits, vegetables | Mpro [78] | SARS-CoV [82] |
| Catechin | Flavanol | Tea | Mpro [78] ACE2 [80] Spike S protein [80] | SARS-CoV [83] |
| Epicatechigallate | Flavanol | Tea | Mpro [78] | |
| Hesperetin | Flavanone | Citrus peel and albedo | ACE2 [71] | SARS-CoV [84] |
| Hesperidin | Flavanone | Citrus peel and albedo | Mpro [85] ACE2 [85] Spike S protein [85] | SARS-CoV [85] |
| diallyl tetrasulfide, trisulfide 2-propenyl propyl | Organosulfur | Garlic | Mpro [86] ACE2 [86] | |
| Tenufolin | Triterpene saponin | Cinnamomun verum | Mpro [87] | SARS-CoV [88] |
| Pavettanin C1 | Lignin | Cinnamomun verum | Spike S protein [87] | SARS-CoV [88] |
| δ-Viniferin | Flavonol | Red wine | Mpro [89] ACE2 [89] RNA-dependent RNA polymerase [89] | |
| Myricitrin | Flavone | Myrica esculenta | Mpro [89] ACE2 [89] | SARS-CoV [78,90] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Matteo, G.; Spano, M.; Grosso, M.; Salvo, A.; Ingallina, C.; Russo, M.; Ritieni, A.; Mannina, L. Food and COVID-19: Preventive/Co-therapeutic Strategies Explored by Current Clinical Trials and in Silico Studies. Foods 2020, 9, 1036. https://doi.org/10.3390/foods9081036
Di Matteo G, Spano M, Grosso M, Salvo A, Ingallina C, Russo M, Ritieni A, Mannina L. Food and COVID-19: Preventive/Co-therapeutic Strategies Explored by Current Clinical Trials and in Silico Studies. Foods. 2020; 9(8):1036. https://doi.org/10.3390/foods9081036
Chicago/Turabian StyleDi Matteo, Giacomo, Mattia Spano, Michela Grosso, Andrea Salvo, Cinzia Ingallina, Mariateresa Russo, Alberto Ritieni, and Luisa Mannina. 2020. "Food and COVID-19: Preventive/Co-therapeutic Strategies Explored by Current Clinical Trials and in Silico Studies" Foods 9, no. 8: 1036. https://doi.org/10.3390/foods9081036
APA StyleDi Matteo, G., Spano, M., Grosso, M., Salvo, A., Ingallina, C., Russo, M., Ritieni, A., & Mannina, L. (2020). Food and COVID-19: Preventive/Co-therapeutic Strategies Explored by Current Clinical Trials and in Silico Studies. Foods, 9(8), 1036. https://doi.org/10.3390/foods9081036













