Effect of Food Preparations on In Vitro Bioactivities and Chemical Components of Fucus vesiculosus
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Algae Material
2.3. Aqueous Extract Preparation
2.4. SPE Purification
2.5. Chemical Analysis by HPLC-DAD and LC-HRMS/MS
2.6. Total Phenol Content Quantification
2.7. Determination of Antioxidant Activity
2.8. Acetylcholinesterase Activity
2.9. HMG-CoA Reductase Activity
2.10. Permeation Studies
2.11. SEM Observations
2.12. Statistical Analysis
3. Results
3.1. Chromatographic Characterization and Phenolic Content Determination According to Several Origins and Different Extraction Processes
3.1.1. Phenolic Profile Characterization by RP-HPLC-DAD
3.1.2. Separation of Bioactive Metabolites by Solid Phase Extraction (SPE)
3.1.3. Total Phenolic Content
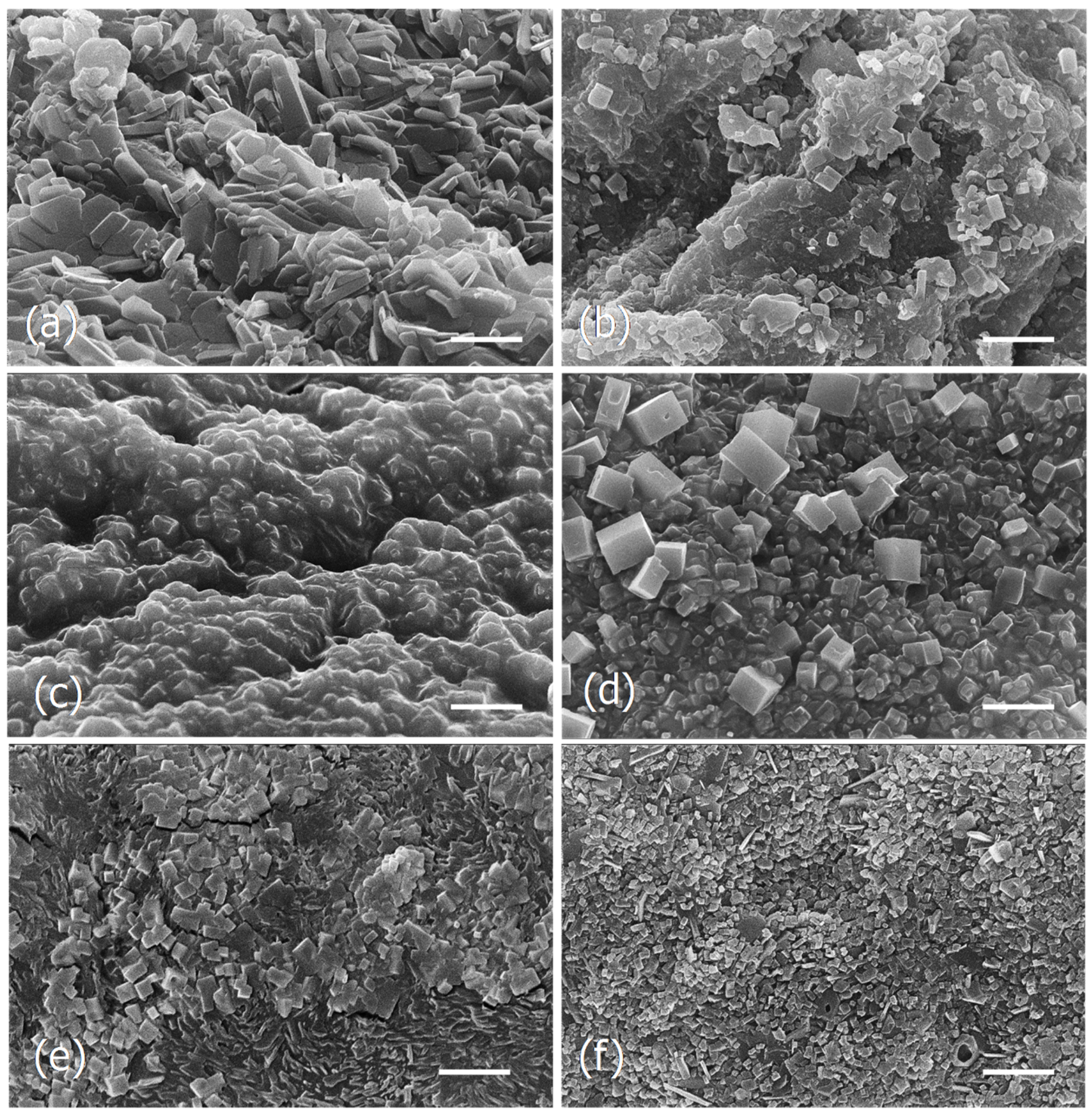
3.1.4. SEM Observations of F. vesiculosus Particles Obtained by Different Drying Methods and Extraction Processes
3.2. Biological Activities
3.2.1. Free Radical Scavenging Capacity
3.2.2. Acetylcholinesterase Activity
3.2.3. HMG-CoA Reductase Inhibitory Activity
3.2.4. Cholesterol Permeation
3.3. Compound Identification by LC-HRMS/MS
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mac Monagail, M.; Cornish, L.; Morrison, L.; Araújo, R.; Critchley, A.T. Sustainable harvesting of wild seaweed resources. Eur. J. Phycol. 2017, 52, 371–390. [Google Scholar] [CrossRef]
- Harnedy, P.A.; Fitzgerald, R.J. Bioactive proteins, peptides, and amino acids from macroalgae. J. Phycol. 2011, 47, 218–232. [Google Scholar] [CrossRef]
- Neto, R.T.; Marçal, C.; Queirós, A.S.; Abreu, H.; Silva, A.M.S.; Cardoso, S.M. Screening of Ulva rigida, Gracilaria sp., Fucus vesiculosus and Saccharina latissima as functional ingredients. Int. J. Mol. Sci. 2018, 19, 2987. [Google Scholar] [CrossRef]
- Ferdouse, F.; Lovstad Holdt, S.; Smith, R.; Murúa, P.; Yang, L. The global status of seaweed production, trade and utilization. FAO Globefish Res. Programme 2017, 124, 1–57. [Google Scholar]
- Mahadevan, K. Seaweeds: A sustainable food source. In Seaweed Sustainability: Food and Non-Food Applications; Elsevier Inc.: Amsterdam, The Netherlands, 2015. [Google Scholar] [CrossRef]
- Parys, S.; Kehraus, S.; Krick, A.; Glombitza, K.W.; Carmeli, S.; Klimo, K.; König, G.M. In vitro chemopreventive potential of fucophlorethols from the brown alga Fucus vesiculosus L. by anti-oxidant activity and inhibition of selected cytochrome P450 enzymes. Phytochemistry 2010, 71, 221–229. [Google Scholar] [CrossRef]
- Bouga, M.; Combet, E. Emergence of Seaweed and Seaweed-Containing Foods in the UK: Focus on Labeling, Iodine Content, Toxicity and Nutrition. Foods 2015, 4, 240–253. [Google Scholar] [CrossRef]
- Laekeman, G. Assessment Report on Fucus Vesiculosus L., Thallus (Vol. 44). 2014. Available online: http://www.ema.europa.eu/docs/en_GB/document_library/Herbal_-_HMPC_assessment_report/2015/07/WC500190398.pdf (accessed on 11 November 2019).
- Ososki, A.L.; Lohr, P.; Reiff, M.; Balick, M.J.; Kronenberg, F.; Fugh-Berman, A.; O’Connor, B. Ethnobotanical literature survey of medicinal plants in the Dominican Republic used for women’s health conditions. J. Ethnopharmacol. 2002, 79, 285–298. [Google Scholar] [CrossRef]
- Romm, A.; Hardy, M.L.; Mills, S.; Bove, M.; Stansbury, J.E.; Romm, A. Chapter 6–Endocrine Disorders and Adrenal Support. In Botanical Medicine for Women’s Health; Churchill Livingstone: London, UK, 2010; pp. 186–210. [Google Scholar] [CrossRef]
- Skibola, C.F. The effect of Fucus vesiculosus, an edible brown seaweed, upon menstrual cycle length and hormonal status in three pre-menopausal women: A case report. BMC Complement. Altern. Med. 2004, 4, 10. [Google Scholar] [CrossRef]
- Díaz-Rubio, M.E.; Pérez-Jiménez, J.; Saura-Calixto, F. Dietary fiber and antioxidant capacity in Fucus vesiculosus products. Int. J. Food Sci. Nutr. 2009, 60, 23–34. [Google Scholar] [CrossRef]
- Rajauria, G.; Foley, B.; Abu-Ghannam, N. Identification and characterization of phenolic antioxidant compounds from brown Irish seaweed Himanthalia elongata using LC-DAD–ESI-MS/MS. Innov. Food Sci. Emerg. Technol. 2016, 37, 261–268. [Google Scholar] [CrossRef]
- Agregán, R.; Munekata, P.E.S.; Franco, D.; Dominguez, R.; Carballo, J.; Lorenzo, J.M. Phenolic compounds from three brown seaweed species using LC-DAD–ESI-MS/MS. Food Res. Int. 2017, 99, 979–985. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, M.T.H.; Bangoura, I.; Kang, J.-Y.; Park, N.-G.; Ahn, D.-H.; Hong, Y.-K. Distribution of Phlorotannins in the Brown Alga Ecklonia cava and Comparison of Pretreatments for Extraction. Fish. Aquat. Sci. 2011, 14, 198–204. [Google Scholar] [CrossRef]
- Wang, T.; Jónsdóttir, R.; Ólafsdóttir, G. Total phenolic compounds, radical scavenging and metal chelation of extracts from Icelandic seaweeds. Food Chem. 2009, 116, 240–248. [Google Scholar] [CrossRef]
- Imbs, T.I.; Zvyagintseva, T.N. Phlorotannins are Polyphenolic Metabolites of Brown Algae. Russian J. Mar. Biol. 2018, 44, 263–273. [Google Scholar] [CrossRef]
- Beaulieu, L. Bioactive Peptides Using Proteomic and Transcriptomic Approaches. Molecules 2019, 24, 1708. [Google Scholar] [CrossRef]
- Choi, E.K.; Park, S.H.; Ha, K.C.; Noh, S.O.; Jung, S.J.; Chae, H.J.; Park, T.S. Clinical trial of the hypolipidemic effects of a brown alga Ecklonia cava extract in patients with hypercholesterolemia. Intl. J. Pharmacol. 2015, 11, 798–805. [Google Scholar] [CrossRef]
- Yeo, A.R.; Lee, J.; Tae, I.H.; Park, S.R.; Cho, Y.H.; Lee, B.H.; Yoo, Y.C. Anti-hyperlipidemic effect of polyphenol extract (SeapolynolTM) and dieckol isolated from ecklonia cava in in vivo and in vitro models. Prev. Nutr. Food Sci. 2012, 17, 1–7. [Google Scholar] [CrossRef]
- Kim, S.K.; Wijesekara, I. Development and biological activities of marine-derived bioactive peptides: A review. J. Funct. Foods 2010, 2, 1–9. [Google Scholar] [CrossRef]
- Benede, S.; Molina, E. Chicken Egg Proteins and Derived Peptides with Antioxidant Properties. Foods 2020, 9, 735. [Google Scholar] [CrossRef] [PubMed]
- Pereira, L. Seaweeds as Source of Bioactive Substances and Skin Care Therapy—Cosmeceuticals, Algotheraphy, and Thalassotherapy. Cosmetics 2018, 5, 68. [Google Scholar] [CrossRef]
- Angerhofer, C.K.; Maes, D.; Giacomoni, P.U. Skin Aging Handbook—An Integrated Approach to Biochemistry and Product Development; Andrew, W., Ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2009; pp. 205–263. [Google Scholar]
- Ressaissi, A.; Attia, N.; Falé, P.L.V.; Pacheco, R.; Teixeira, V.H.; Machuqueiro, M.; Borges, C.; Serralheiro, M.L.M. Aqueous Extracts from Nopal (Opuntia Ficus-Indica): Antiacetylcholinesterase and Antioxidant Activity from Phenolic Bioactive Compounds. Int. J. Green Herb. Chem. 2016, 5, 337–348. [Google Scholar]
- Yoon, N.Y.; Chung, H.Y.; Kim, H.R.; Choi, J.S. Acetyl- and butyrylcholinesterase inhibitory activities of sterols and phlorotannins from Ecklonia stolonifera. Fish. Sci. 2008, 74, 200–207. [Google Scholar] [CrossRef]
- Burmaoglu, S.; Yilmaz, A.O.; Taslimi, P.; Algul, O.; Kilic, D.; Gulcin, I. Synthesis and biological evaluation of phloroglucinol derivatives possessing α-glycosidase, acetylcholinesterase, butyrylcholinesterase, carbonic anhydrase inhibitory activity. Arch. Pharm. 2018, 351. [Google Scholar] [CrossRef] [PubMed]
- Wilkins, E.; Wilson, I.; Wickramasinghe, K.; Bhatnagar, P.; Leal, J.; Luengo-Fernandez, R.; Townsend, N. European Cardiovascular Disease Statistics, 2017 ed.; European Heart Network: Bruxelles, Belgium, 2017; Volume 192. [Google Scholar]
- Lopes, G.; Barbosa, M.; Vallejo, F.; Gil-Izquierdo, Á.; Andrade, P.B.; Valentão, P.; Ferreres, F. Profiling phlorotannins from Fucus spp. of the Northern Portuguese coastline: Chemical approach by HPLC-DAD-ESI/MSn and UPLC-ESI-QTOF/MS. Algal Res. 2018, 29, 113–120. [Google Scholar] [CrossRef]
- Falé, P.L.; Ferreira, C.; Rodrigues, A.M.; Cleto, P.; Madeira, P.J.A.; Florêncio, M.H.; Serralheiro, M.L.M. Antioxidant and anti-acetylcholinesterase activity of commercially available medicinal infusions after in vitro gastrointestinal digestion. J. Med. Plants Res. 2013, 7, 1370–1378. [Google Scholar] [CrossRef]
- André, R.; Catarro, J.; Freitas, D.; Pacheco, R.; Oliveira, M.C.; Serralheiro, M.L.; Falé, P.L. Action of euptox A from Ageratina adenophora juice on human cell lines: A top-down study using FTIR spectroscopy and protein profiling. Toxicol. In Vitro 2019, 57. [Google Scholar] [CrossRef]
- Arantes, A.A.; Falé, P.L.; Costa, L.C.B.; Pacheco, R.; Ascensão, L.; Serralheiro, M.L. Inhibition of HMG-CoA reductase activity and cholesterol permeation through Caco-2 cells by caffeoylquinic acids from Vernonia condensata leaves. Braz. J. Pharmacogn. 2016, 26, 738–743. [Google Scholar] [CrossRef]
- Falé, P.L.; Ferreira, C.; Maruzzella, F.; Helena Florêncio, M.; Frazão, F.N.; Serralheiro, M.L.M. Evaluation of cholesterol absorption and biosynthesis by decoctions of Annona cherimola leaves. J. Ethnopharmacol. 2013, 150, 718–723. [Google Scholar] [CrossRef]
- Henriques, J.; Ribeiro, M.J.; Falé, P.L.; Pacheco, R.; Ascensão, L.; Florêncio, M.H.; Serralheiro, M.L.M. Valorization of kiwifruit production: Leaves of the pruning branches of Actinidia deliciosa as a promising source of polyphenols. Eur. Food Res. Technol. 2017, 243, 1343–1353. [Google Scholar] [CrossRef]
- Guedes, L.; Pedro, B.; Reis, P.S.; Machuqueiro, M.; Ressaissi, A.; Pacheco, R.; Serralheiro, M.L. Bioactivities of Centaurium erythraea (Gentianaceae) Decoctions: Antioxidant Activity, Enzyme Inhibition and Docking Studies. Molecules 2019, 24, 3795. [Google Scholar] [CrossRef]
- Mata, A.T.; Proença, C.; Ferreira, A.R.; Serralheiro, M.L.M.; Nogueira, J.N.F.; Araújo, M.E.M. Antioxidant and antiacetylcholinesterase activities of five plants used as Portuguese food spices. Food Chem. 2007, 103, 778–786. [Google Scholar] [CrossRef]
- Barbosa Filho, J.M.; Medeiros, K.C.P.; Diniz, M.d.F.F.M.; Batista, L.M.; Athayde-Filho, P.F.; Silva, M.S.; Quintans-Júnior, L.J. Natural products inhibitors of the enzyme acetylcholinesterase. Rev. Bras. Farmacogn. 2006, 16, 258–285. [Google Scholar] [CrossRef]
- Ferruzza, S.; Rossi, C.; Scarino, M.L.; Sambuy, Y. A protocol for differentiation of human intestinal Caco-2 cells in asymmetric serum-containing medium. Toxicol. In Vitro 2012, 26, 1252–1255. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Fu, X.; Duan, D.; Liu, X.; Xu, J.; Gao, X. Extraction and Identification of Phlorotannins from the Brown Alga, Sargassum fusiforme (Harvey) Setchell. Mar. Drugs 2017, 15, 49. [Google Scholar] [CrossRef] [PubMed]
- Lafarga, T.; Acién-Fernández, F.G.; Garcia-Vaquero, M. Bioactive peptides and carbohydrates from seaweed for food applications: Natural occurrence, isolation, purification, and identification. Algal Res. 2020, 48, 101909. [Google Scholar] [CrossRef]
- Catarino, M.D.; Silva, A.M.S.; Mateus, N.; Cardoso, S.M. Optimization of phlorotannins extraction from Fucus vesiculosus and evaluation of their potential to prevent metabolic disorders. Mar. Drugs 2019, 17, 162. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Cao, Y.L.; Jiang, J.G.; Lin, Q.S.; Chen, J.; Zhu, L. Response surface optimization of ultrasound-assisted flavonoids extraction from the flower of Citrus aurantium L. var. amara Engl. J. Sep. Sci. 2010, 33, 1349–1355. [Google Scholar] [CrossRef]
- Falé, P.L.; Amaral, F.; Amorim Madeira, P.J.; Sousa Silva, M.; Florêncio, M.H.; Frazão, F.N.; Serralheiro, M.L.M. Acetylcholinesterase inhibition, antioxidant activity and toxicity of Peumus boldus water extracts on HeLa and Caco-2 cell lines. Food Chem. Toxicol. 2012, 50, 2656–2662. [Google Scholar] [CrossRef]
- Orhan, I.E.; Jedrejek, D.; Senol, F.S.; Salmas, R.E.; Durdagi, S.; Kowalska, I.; Oleszek, W. Molecular modeling and in vitro approaches towards cholinesterase inhibitory effect of some natural xanthohumol, naringenin, and acyl phloroglucinol derivatives. Phytomedicine 2018, 42, 25–33. [Google Scholar] [CrossRef]
- Prasasty, V.; Radifar, M.; Istyastono, E. Natural peptides in drug discovery targeting acetylcholinesterase. Molecules 2018, 23, 2344. [Google Scholar] [CrossRef]
- Shen, W.; Matsui, T. Intestinal absorption of small peptides: A review. Int. J. Food Sci. Technol. 2019, 54, 1942–1948. [Google Scholar] [CrossRef]
- Park, Y.; Carr, T.P. Unsaturated fatty acids and phytosterols regulate cholesterol transporter genes in Caco-2 and HepG2 cell lines. Nutr. Res. 2013, 33, 154–161. [Google Scholar] [CrossRef]
- Betters, J.L.; Yu, L. NPC1L1 and cholesterol transport. FEBS Lett. 2010, 584, 2740–2747. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.; Cheung, B.M.Y.; Tomlinson, B. Safety of statins: An update. Ther. Adv. Drug Saf. 2012, 3, 133–144. [Google Scholar] [CrossRef]
- Ressaissi, A.; Attia, N.; Pacheco, R.; Falé, P.; Serralheiro, M.L.M. Cholesterol transporter proteins in HepG2 cells can be modulated by phenolic compounds present in Opuntia ficus-indica aqueous solutions. J. Funct. Foods 2020, 64, 103674. [Google Scholar] [CrossRef]
- Ressaissi, A.; Attia, N.; Falé, P.L.V.; Pacheco, R.; Victor, B.; Machuqueiro, M.; Serralheiro, M.L.M. Isorhamnetin derivatives and piscidic acid for hypercholesterolemia: Cholesterol permeability, HMG-CoA reductase inhibition, and docking studies. Arch. Pharm. Res. 2017, 40, 1278–1286. [Google Scholar] [CrossRef] [PubMed]
- Falé, P.V.L.; Ferreira, C.; Rodrigues, A.M.; Frazão, M.F.; Serralheiro, M.L.M. Studies on the molecular mechanism of cholesterol reduction by Fraxinus angustifolia, Peumus boldus, Cynara cardunculus and Pterospartum tridentatum infusions. J. Med. Plants Res. 2014, 8, 9–17. [Google Scholar] [CrossRef]
- Nekohashi, M.; Ogawa, M.; Ogihara, T.; Nakazawa, K.; Kato, H.; Misaka, T.; Kobayashi, S. Luteolin and quercetin affect the cholesterol absorption mediated by epithelial cholesterol transporter Niemann-Pick C1-Like 1 in Caco-2 cells and rats. PLoS ONE 2014, 9, e97901. [Google Scholar] [CrossRef]
 ) Ocean samples; (
) Ocean samples; ( ) Tagus samples; (
) Tagus samples; ( ) capsules; (
) capsules; ( ) Tagus samples oven-dried. (a): Ocean, Tagus, capsules, and Tagus oven-dried extracted at 100 °C, 30 min; (b): ocean, Tagus, capsules, and Tagus oven-dried extracted at 25 °C, 24 h; (c): ocean and Tagus samples extracted at 100 °C and purified by SPE.
) Tagus samples oven-dried. (a): Ocean, Tagus, capsules, and Tagus oven-dried extracted at 100 °C, 30 min; (b): ocean, Tagus, capsules, and Tagus oven-dried extracted at 25 °C, 24 h; (c): ocean and Tagus samples extracted at 100 °C and purified by SPE.
 ) Ocean samples; (
) Ocean samples; ( ) Tagus samples; (
) Tagus samples; ( ) capsules; (
) capsules; ( ) Tagus samples oven-dried. (a): Ocean, Tagus, capsules, and Tagus oven-dried extracted at 100 °C, 30 min; (b): ocean, Tagus, capsules, and Tagus oven-dried extracted at 25 °C, 24 h; (c): ocean and Tagus samples extracted at 100 °C and purified by SPE.
) Tagus samples oven-dried. (a): Ocean, Tagus, capsules, and Tagus oven-dried extracted at 100 °C, 30 min; (b): ocean, Tagus, capsules, and Tagus oven-dried extracted at 25 °C, 24 h; (c): ocean and Tagus samples extracted at 100 °C and purified by SPE.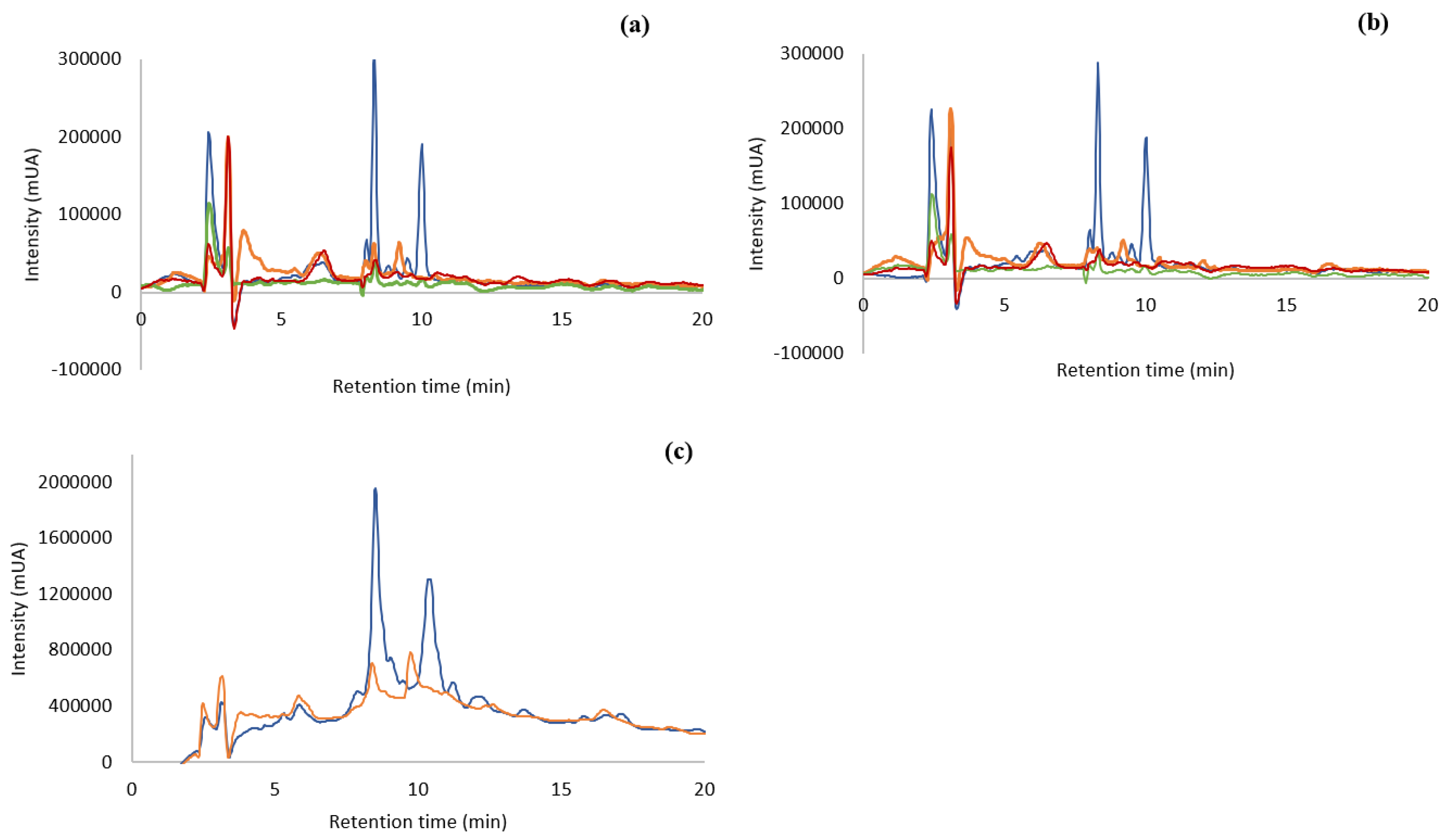
 ) H2O 25 °C; (
) H2O 25 °C; ( ) H2O 100 °C; (
) H2O 100 °C; ( ) SPE purification (H2O 100 °C).
) SPE purification (H2O 100 °C).
 ) H2O 25 °C; (
) H2O 25 °C; ( ) H2O 100 °C; (
) H2O 100 °C; ( ) SPE purification (H2O 100 °C).
) SPE purification (H2O 100 °C).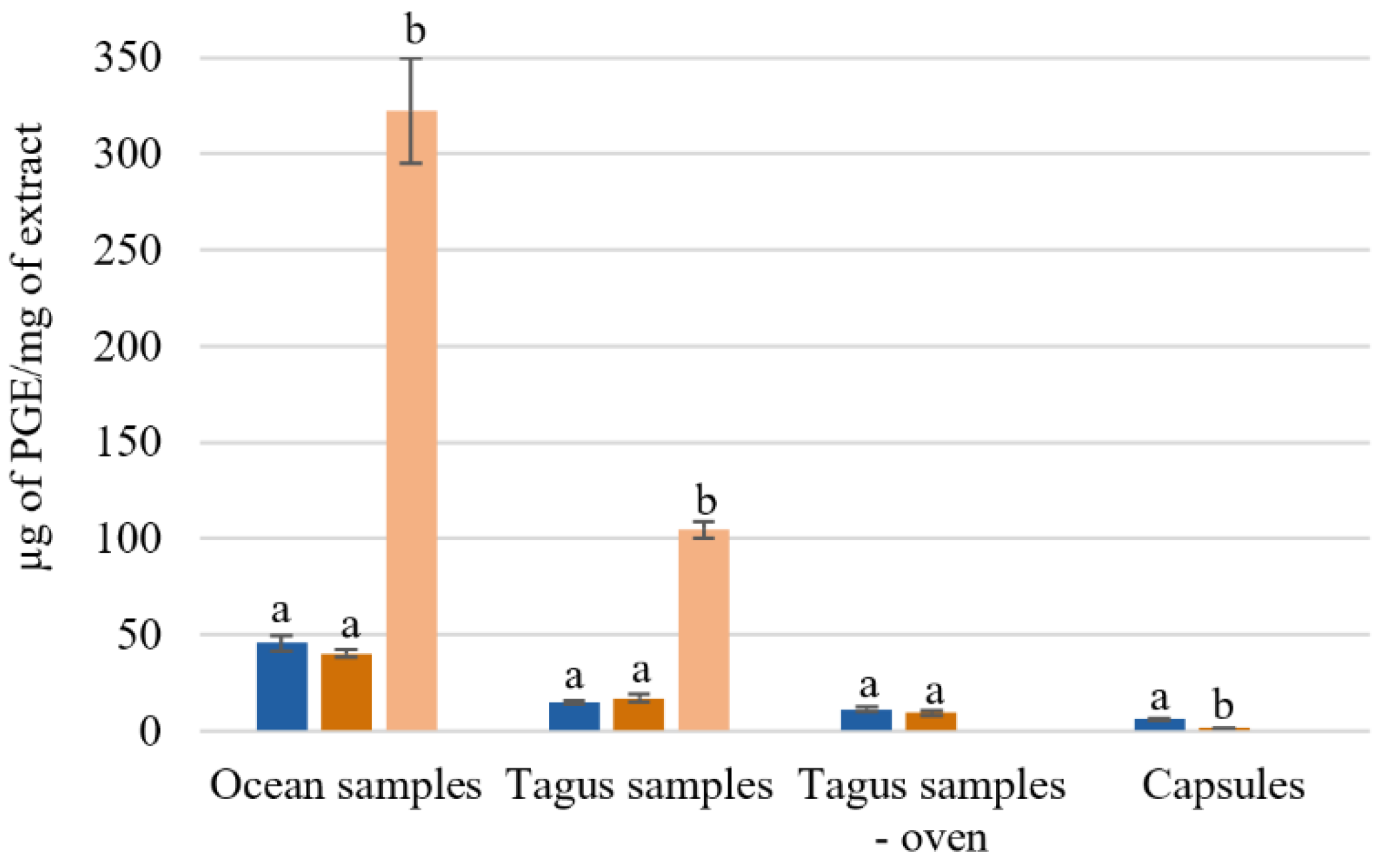

 ) H2O 25 °C; (
) H2O 25 °C; ( ) H2O 100 °C; (
) H2O 100 °C; ( ) SPE purification (H2O 100 °C).
) SPE purification (H2O 100 °C).
 ) H2O 25 °C; (
) H2O 25 °C; ( ) H2O 100 °C; (
) H2O 100 °C; ( ) SPE purification (H2O 100 °C).
) SPE purification (H2O 100 °C).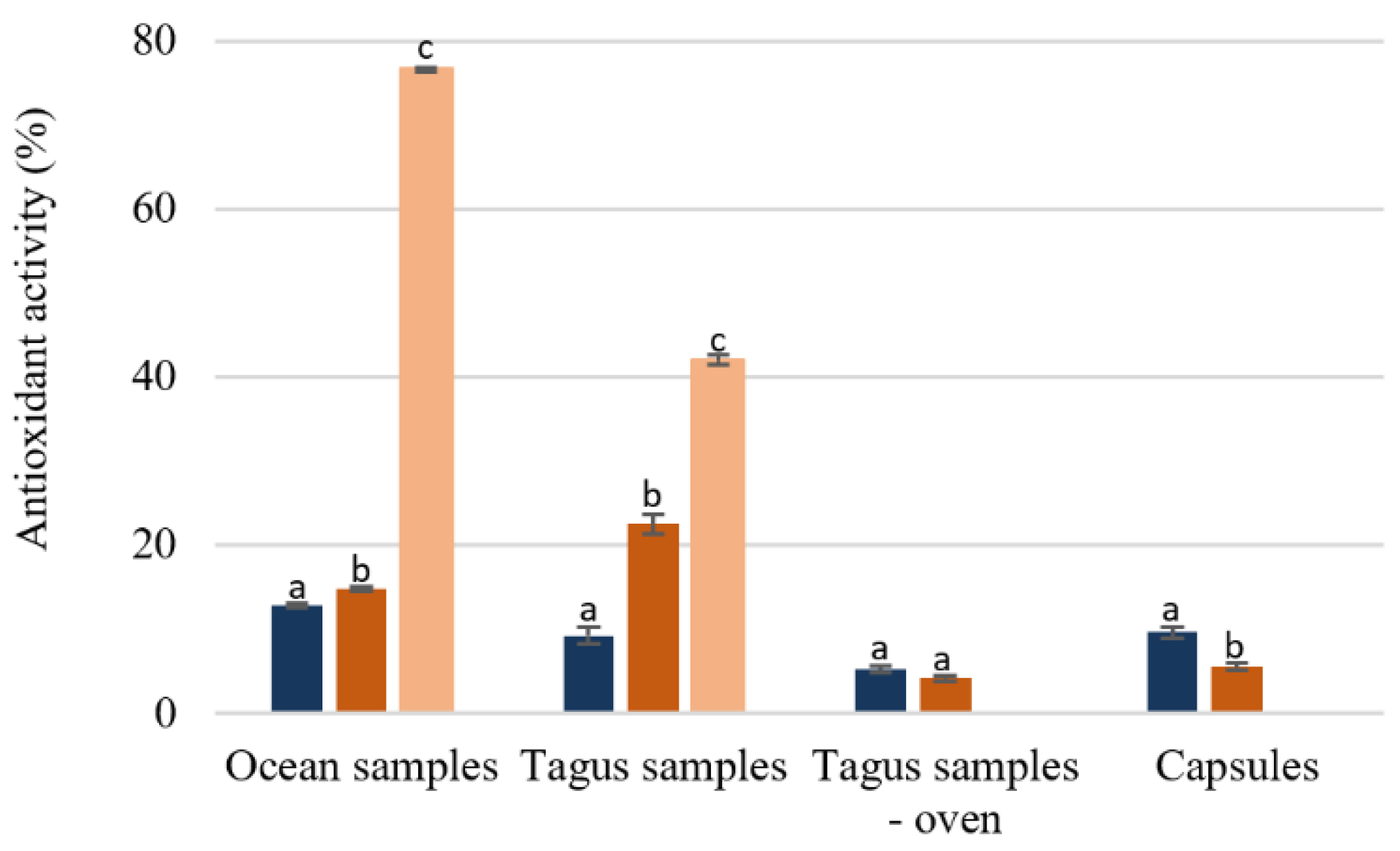
| Extract | Acetylcholinesterase Inhibition | HMGR * Inhibition | Inhibition of Cholesterol Permeation (%) | ||
|---|---|---|---|---|---|
| 1000 µg/mL | IC50 (µg/mL) | 10 µg/mL | IC50 (µg/mL) | ||
| Ocean samples | 89.96% ± 0.54 a | 210.86 ± 22.11 a | 27.74% ± 1.60 a | - | - |
| Ocean samples–SPE fraction | - | 14.97 ± 0.13 b | 86.41% ± 3.20 b | 4.16 ± 0.11 | 45.3 ± 4.4 |
| Tagus samples | 4.199% ± 0.36 b | - | 1.07% ± 0.65 c | - | - |
| Tagus samples–SPE fraction | 60.44% ± 1.21c | 849.80 ± 10.24 c | 52.95% ± 3.17 d | - | - |
| Mass [M − H]− | Samples Origin | Mass [M − H]− | Samples Origin | Mass [M − H]− | Samples Origin | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| No | m/z | Ocean | Tagus | No | m/z | Ocean | Tagus | No | m/z | Ocean | Tagus |
| 1.0–1.3 min | 1.9–2.0 min | 268.9734 | |||||||||
| 102.9572 | 191.0198 | 516.9721 | |||||||||
| 1 | 181.0722 | 117.0194 | 268.9734 | ||||||||
| 146.0460 | 269.0874 | 494.9909 | |||||||||
| 165.0406 | 203.9732 | 531.0673 | |||||||||
| 227.0773 | 223.0820 | 536.1138 | |||||||||
| 240.0080 | 230.9893 | 699.1822 | |||||||||
| 2 | 191.0201 | 7 | 276.0184 | 3.2–3.4 min | |||||||
| 217.0484 | 384.1527 | 164.0715 | |||||||||
| 219.0457 | 8 | 455.1898 | 185.0454 | ||||||||
| 126.9051 | 713.2366 | 232.0592 | |||||||||
| 111.0091 | 9 | 497.0728 | 16 | 277.0932 | |||||||
| 1.5–1.6 min | 10 | 875.2911 | 299.0754 | ||||||||
| 117.0193 | 2.0–2.1 min | 17 | 351.1334 | ||||||||
| 140.0115 | 11 | 327.1299 | 375.0613 | ||||||||
| 149.9627 | 425.0990 | 397.0428 | |||||||||
| 181.0715 | 2.1–2.3 min | 18 | 410.1695 | ||||||||
| 219.9681 | 12 | 237.0078 | 19 | 555.1934 | |||||||
| 3 | 256.0940 | 475.0231 | 855.2730 | ||||||||
| 300.9335 | 265.9819 | 3.5–3.6 min | |||||||||
| 319.9066 | 13 | 125.0247 | 20 | 355.1642 | |||||||
| 322.9155 | 174.9560 | 21 | 497.0748 | ||||||||
| 338.8896 | 2.4–2.6 min | 387.0309 | |||||||||
| 370.1380 | 14 | 246.9916 | 4.5–4.7 min | ||||||||
| 4 | 403.0557 | 328.9980 | 22 | 355.1622 | |||||||
| 5 | 445.0962 | 15 | 373.0572 | 227.1037 | |||||||
| 541.1551 | 680.2201 | 453.1301 | |||||||||
| 6 | 551.1836 | 536.1125 | 23 | 711.3323 | |||||||
| 248.9894 | |||||||||||
| 4.7–4.8 min | 6.1–6.2 min | 12.7–12.8 min | |||||||||
| 24 | 203.0824 | 131.0714 | 174.9558 | ||||||||
| 25 | 291.0988 | 186.1133 | 43 | 527.2535 | |||||||
| 369.1787 | 785.3471 | ||||||||||
| 26 | 419.1573 | 6.7–6.8 min | |||||||||
| 27 | 621.0891 | 147.0452 | |||||||||
| 5.0–5.1 min | 40 | 165.0557 | |||||||||
| 138.0562 | 206.0822 | ||||||||||
| 28 | 175.0611 | 119.0504 | |||||||||
| 187.9041 | 331.1190 | ||||||||||
| 375.8086 | 353.1007 | ||||||||||
| 29 | 745.1056 | 619.2774 | |||||||||
| 5.4–5.7 min | 41 | 930.1220 | |||||||||
| 30 | 324.1576 | 1054.6409 | |||||||||
| 31 | 369.1787 | 7.8 min | |||||||||
| 405.1553 | 42 | 361.1503 | |||||||||
| 427.9317 | 383.1323 | ||||||||||
| 32 | 437.1652 | 587.2592 | |||||||||
| 33 | 467.1465 | 433.1622 | |||||||||
| 34 | 739.3642 | 9.8–10 | |||||||||
| 241.1192 | 394.9648 | ||||||||||
| 35 | 531.2215 | 395.9679 | |||||||||
| 36 | 682.0892 | 10.9–11.1 min | |||||||||
| 37 | 806.1051 | 396.9627 | |||||||||
| 38 | 869.1216 | 160.8416 | |||||||||
| 467.1461 | 162.8387 | ||||||||||
| 5.7–5.8 min | 174.9556 | ||||||||||
| 39 | 247.0823 | 447.1349 | |||||||||
| 145.0598 | |||||||||||
| 269.0646 | |||||||||||
| Rt (min) | [M − H]− m/z | Molecular Formula | Error (ppm) | Fragmentation (%) | Name | No |
|---|---|---|---|---|---|---|
| 1.5 | 445.0962 | C18H22O13 | 3.3 | 314 (50%); 221 (100%); 80 (60%) | Phloroglucinol derivative | 5 |
| 1.9 | 455.1898 | C18H28N6O8 | −0.5 | 238.0839 (96.97%); 126.0196 (37.10%); 88.0404 (23.56%) | Peptide derivative ThrGluHisAla | 8 |
| 1.9 | 875.2911 | C49H48O15 | −0.2 | 829 (100%); 125 (7%); 179 (14%); 383 (16%) | Phloroglucinol derivative (7 units) | 10 |
| 2.6 | 373.0572 | C18H14O9 | −1.5 | 355 (37%); 141.0192 (100%); 207.0303 (20%) | Phloroglucinol derivative (3 units) | 15 |
| 3.2 | 410.1695 | C17H25N5O7 | −4.8 | 136 (45%); 109 (45%); 366 (58%); 339 (35%) | Benarthin | 18 |
| 3.5 | 355.1642 | C15H24N4O6 | −4.5 | 311 (74%); 252 (19%); 194 (24%); 96 (46%) | Tetrapeptide ProProGlySer | 20 |
| 3.6 | 497.0748 | C24H18O12 | 0.6 | 125 (45%); 141 (30%); 372 (20%); 356 (20%) | Phloroglucinol derivative (4 units) | 21 |
| 4.5 | 355.1622 | C15H24N4O6 | 0.2 | 311 (12%); 238 (95%); 240 (82%); 183 (3 7%); 116 (100%); 70 (8%) | Tetrapeptide ProSerGlyPro | 22 |
| 4.5 | 711.3323 | C30H48N8O12 | −2.2 | 355 (100%); 129 (2%); 116 (9%); 356 (20%) | Octapeptide of ProProGlySer | 23 |
| 4.7 | 203.0824 | C11H12N2O2 | 1 | 116.0508 (100%); 74.0251 (37,77%) | Tryptophan | 24 |
| 4.7 | 291.0982 | C14H16N2O5 | 1.5 | Didn’t fragment | Tryptophan derivative | 25 |
| 4.7 | 419.1573 | C19H24N4O7 | 0 | Didn’t fragment | Tetrapeptide of (GluSerTrp) | 26 |
| 4.7 | 621.0891 | C30H22O15 | −2.5 | 125 (84%); 141(6%); 165 (6%); 485 (3%); 289 (3%); 478 (50%) | Phloroglucinol derivative (5 units) | 27 |
| 5.0 | 745.1039 | C36H26O18 | −0.5 | 125 (29%); 141 (14%); 497 (100%); 413 (4%) | Phloroglucinol derivative (6 units) | 29 |
| 5.4 | 324.1576 | C15H22N3O5 | −1.9 | 209.0944 (45.86%); 166.0868 (34.90%); 114.0565 (100%); | N-(5-Hydrazinyl-5-oxopentyl)-3,4,5-trimethoxybenzamide | 30 |
| 5.4 | 369.1787 | C16H26N4O6 | −0.1 | 325.1879 (19.73%); 238.0834 (87.76%); 130.0872 (100%) 58.0303 (15.04%); | Tetrapeptide ThrProProGly | 31 |
| 5.4 | 437.1652 | C19H26N4O8 | 3.8 | 392 (23%); 320 (11%); 348 (1%) | Tetrapeptide GlyAspPheThr | 32 |
| 5.4 | 467.1459 | C17H32N4O5S3 | −1.7 | 423; 319 (0.4%) | Tetrapeptide LeuCysCysMet | 33 |
| 5.4 | 739.3642 | C32H52N8O12 | −2.9 | 369 (100%); 143 (1%); 238 (5%) | Peptide derivative | 34 |
| 5.6 | 531.2215 | C24H32N6O8 | 1.0 | Didn’t fragment | Tetrapeptide ValAspTrpAsn | 35 |
| 5.6 | 682.0892 | C27H25NO20 | −4.4 | 125 (44%); 141 (27%); 134 (13%); 495 (43%) | Phloroglucinol derivative | 36 |
| 5.6 | 806.1051 | C33H29NO23 | −0.8 | 369 (95%); 195 (21%); 761(23%); 238 (31%); 567(19%) | Phloroglucinol derivative | 37 |
| 5.6 | 869.1216 | C42H30O21 | -2.4 | 125 (40%); 141 (9%); 137 (9%);194 (6%); 335 (14%); | Phloroglucinol derivative (7 units) | 38 |
| 6.7 | 930.1225 | C39H32NO26 | −0.7 | Didn’t fragment | Phloroglucinol and amino acid derivative | 41 |
| 12.7 | 527.2535 | C32H36N2O5 | 3.1 | 418 (0.2%); 165 (0.8%); 130 (0.3%) | Chaetoglobosin A | 44 |
| Rt (min) | Accurate [M − H]− m/z | Molecular Formula (Error, ppm) | Fragmentation (%) | Proposed Compound | No |
|---|---|---|---|---|---|
| 1.1 | 181.0722 | C6H14O6 (2.3) | - | Mannitol * | 1 |
| 1.2 | 191.0201 | C6H8O7 (2.0) | - | Citric acid * | 2 |
| 1.5 | 256.0941 | C10H16N3O5 (5.4) | 212.10(6); 194.09(93); 166.06(7); 141.06(9); 82.03(32) | Cytidine derivative | 3 |
| 1.5 | 403.0555 | C12H20O13 (−3.7) | 384.98(2); 370.13(4); 231.45(1) 219.96(21); 149.96(100); 96.96(2); 79.95(46) | Galactose-sulfate derivative | 4 |
| 1.6 | 551.1836 | C19H36O18 (−5.3) | 506.12(10); 341.10(15); 325.11(6); 179.05(100); 161.04(17) | Glycidyl compound | 6 |
| 1.9 | 276.0187 | C9H12NO7S (−3.0) | 230.01(9); 196.06(100); 179.05(45);135.04(92) | Tyrosine sulfate | 7 |
| 1.9 | 497.0727 | C24H18O12 (−2.6) | 479.05(10); 383.17(26); 331.04(27);287.05(6); 165.01(100); 137.02(26) | Phloroglucinol derivative | 9 |
| 2.0 | 327.1299 | C12H24O10 (−1.3) | 238.08(26); 101.07(8); 88.04(100) | Glycosidic derivative | 11 |
| 2.1 | 237.0078 | C10H6O7 (−4.4) | 153.05(12); 136.73(1); 123.04(13); 96.96(93)M 79.95(100) | 3. ,5,7-Trihydroxy-4-oxochromene-2-carboxylic acid | 12 |
| 2.2 | 125.0247 | C6H6O3 (−2.6) | - | Phloroglucinol * | 13 |
| 2.4 | 246.9916 | C8H8O7S (−0.4) | 164.93(2); 121.02(23); 108.02(100); 80.96(76) | Vanillic acid sulfate | 14 |
| 3.2 | 277.0930 | C11H18O8 (0.7) | 141.05(5); 97.06(100); 91.03(10) | Phloroglucinol glycosidic derivative | 16 |
| 3.2 | 351.1334 | C15H20N4O6 (6) | 238.12(5); 181.04(3); 164.07(100) | Tetrapeptide Gly Tyr | 17 |
| 3.2 | 555.1930 | C22H36O19 (0.1) | 277.09(100); 185.04(97); 141.05(23) | Phloroglucinol glycosidic derivative | 19 |
| 5.0 | 175.0611 | C7H12O5 (0.0) | 115.04(45); 85.06(100); 59.01(13) | Butanedioic derivate | 28 |
| 5.7 | 247.0823 | C10H16O7 (0.1) | 145.05(40); 127.04(5); 101.06(100);83.05(97) | Glycosidic derivative | 39 |
| 6.7 | 165.0557 | C9H10O3 (0.1) | 147.04(18); 119.05(60); 91.05(5); 73.99(100) | Chroman-2,5-diol | 40 |
| 7.8 | 361.1503 | C16H26O9 (−3.3) | 233.10(13); 145.05(16); 127.04(11); 88.05(100) | Gibboside isomer | 42 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
André, R.; Guedes, L.; Melo, R.; Ascensão, L.; Pacheco, R.; Vaz, P.D.; Serralheiro, M.L. Effect of Food Preparations on In Vitro Bioactivities and Chemical Components of Fucus vesiculosus. Foods 2020, 9, 955. https://doi.org/10.3390/foods9070955
André R, Guedes L, Melo R, Ascensão L, Pacheco R, Vaz PD, Serralheiro ML. Effect of Food Preparations on In Vitro Bioactivities and Chemical Components of Fucus vesiculosus. Foods. 2020; 9(7):955. https://doi.org/10.3390/foods9070955
Chicago/Turabian StyleAndré, Rebeca, Laura Guedes, Ricardo Melo, Lia Ascensão, Rita Pacheco, Pedro D. Vaz, and Maria Luísa Serralheiro. 2020. "Effect of Food Preparations on In Vitro Bioactivities and Chemical Components of Fucus vesiculosus" Foods 9, no. 7: 955. https://doi.org/10.3390/foods9070955
APA StyleAndré, R., Guedes, L., Melo, R., Ascensão, L., Pacheco, R., Vaz, P. D., & Serralheiro, M. L. (2020). Effect of Food Preparations on In Vitro Bioactivities and Chemical Components of Fucus vesiculosus. Foods, 9(7), 955. https://doi.org/10.3390/foods9070955








