Safety of Probiotics: Functional Fruit Beverages and Nutraceuticals
Abstract
1. Introduction
2. Safety and Pathogenicity of Probiotics and Their Assessments
2.1. Probiotics Safety
2.2. Pathogenicity Studies of Probiotics and Clinical Cases
2.3. Drug Interactions of Probiotics
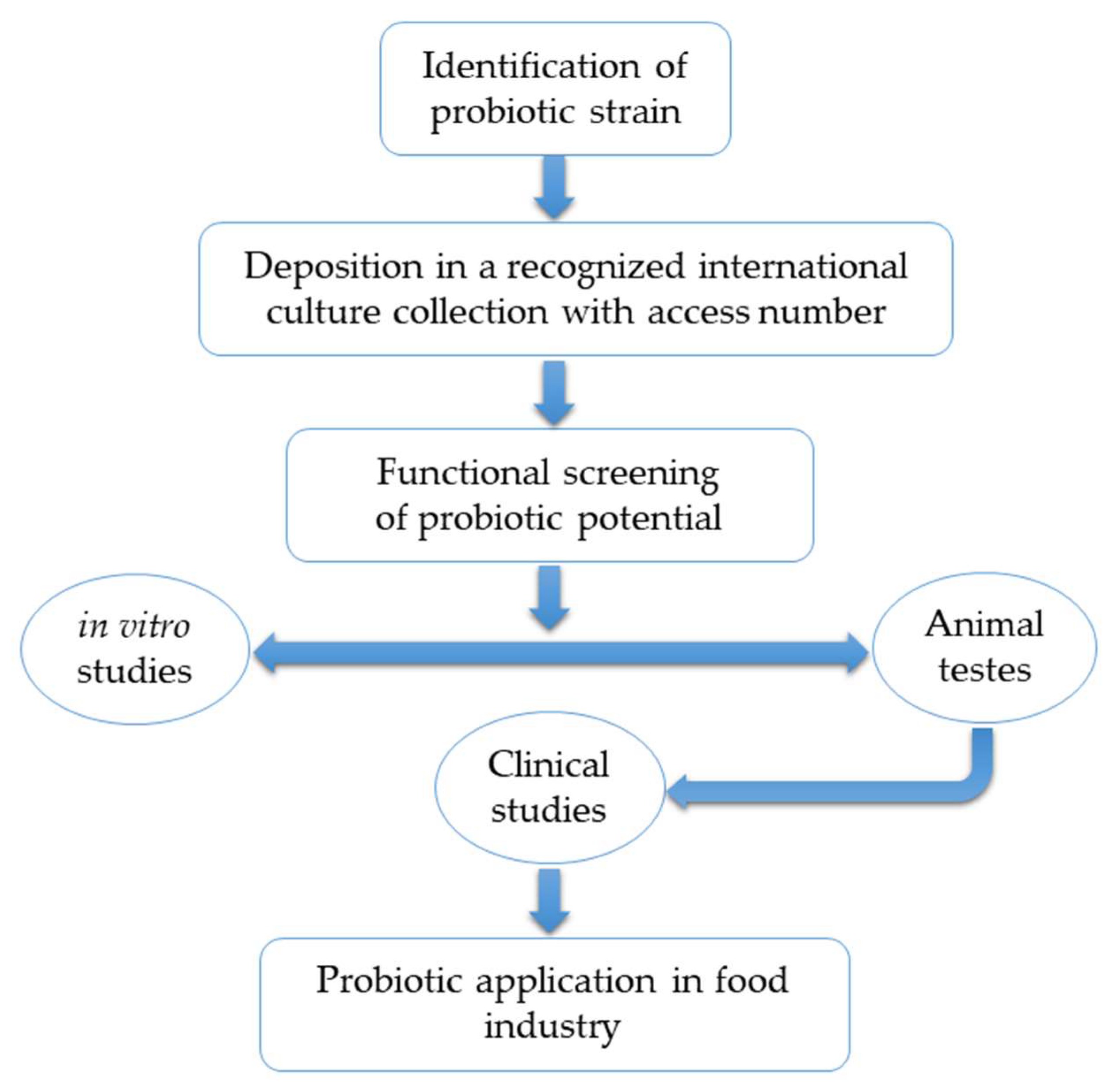
2.4. Assessment of Probiotic Safety
3. Probiotics: Functional Foods
Probiotic Fruit Beverages
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hanson, L.A.; Yolken, R.H.; Services, N.N. Probiotics, Other Nutritional Factors, and Intestinal Microflora. In Nestle Nutrition Workshop Series: Pediatric Program; Lippincott-Raven: La Tour-de-Peilz, Switzerland, 1999; Volume 42. [Google Scholar]
- Mantzourani, I.; Kazakos, S.; Terpou, A.; Alexopoulos, A.; Bezirtzoglou, E.; Bekatorou, A.; Plessas, S. Potential of the probiotic Lactobacillus Plantarum ATCC 14917 strain to produce functional fermented pomegranate juice. Foods 2018, 8, 4. [Google Scholar] [CrossRef] [PubMed]
- Chugh, B.; Kamal-Eldin, A. Bioactive compounds produced by probiotics in food products. Curr. Opin. Food Sci. 2020, 32, 76–82. [Google Scholar] [CrossRef]
- Group, J.F.W.W. Report on Drafting Guidelines for the Evaluation of Probiotics in Food. London, Ontario, Canada. 2002. Available online: https://www.who.int/foodsafety/fs_management/en/probiotic_guidelines.pdf (accessed on 15 June 2020).
- White, J.; Hekmat, S. Development of probiotic fruit juices using Lactobacillus rhamnosus GR-1 fortified with short chain and long chain inulin fiber. Fermentation 2018, 4, 27. [Google Scholar] [CrossRef]
- Ouwehand, A.C.; Invernici, M.M.; Furlaneto, F.A.C.; Messora, M.R. Effectiveness of Multistrain Versus Single-strain Probiotics: Current Status and Recommendations for the Future. J. Clin. Gastroenterol. 2018, 52, S35–S40. [Google Scholar] [CrossRef] [PubMed]
- Markowiak, P.; Slizewska, K. Effects of Probiotics, Prebiotics, and Synbiotics on Human Health. Nutrients 2017, 9, 1021. [Google Scholar] [CrossRef]
- Sanders, M.E.; Merenstein, D.; Merrifield, C.A.; Hutkins, R. Probiotics for human use. Nutr. Bull. 2018, 43, 212–225. [Google Scholar] [CrossRef]
- The European Parliament and the Council of the European Union. Regulation (EU) 2015/2283 of the European Parliament and of the Council of 25 November 2015 on novel foods, amending Regulation (EU) No 1169/2011. European Parliament and of the Council and repealing Regulation (EC) No 258/97 of the European Parliament and of the Council and Commission Regulation (EC) No 1852/2001, Ed. Off. J. Eur. Union 2015, 327, 1–22. [Google Scholar]
- Forestier, C.; Mangalat, N.; Liu, Y.; Fatheree, N.Y.; Ferris, M.J.; Van Arsdall, M.R.; Chen, Z.; Rahbar, M.H.; Gleason, W.A.; Norori, J.; et al. Safety and tolerability of Lactobacillus reuteri DSM 17938 and effects on biomarkers in healthy adults: Results from a randomized masked trial. PLoS ONE 2012, 7, e43910. [Google Scholar] [CrossRef]
- Allen, S.J.; Jordan, S.; Storey, M.; Thornton, C.A.; Gravenor, M.; Garaiova, I.; Plummer, S.F.; Wang, D.; Morgan, G. Dietary supplementation with Lactobacilli and Bifidobacteria is well tolerated and not associated with adverse events during late pregnancy and early infancy. J. Nutr. 2010, 140, 483–488. [Google Scholar] [CrossRef]
- Dekker, J.W.; Wickens, K.; Black, P.N.; Stanley, T.V.; Mitchell, E.A.; Fitzharris, P.; Tannock, G.W.; Purdie, G.; Crane, J. Safety aspects of probiotic bacterial strains Lactobacillus rhamnosus HN001 and Bifidobacterium animalis subsp. lactis HN019 in human infants aged 0–2 years. Int. Dairy J. 2009, 19, 149–154. [Google Scholar] [CrossRef]
- Olivares, M.; Castillejo, G.; Varea, V.; Sanz, Y. Double-blind, randomised, placebo-controlled intervention trial to evaluate the effects of Bifidobacterium longum CECT 7347 in children with newly diagnosed coeliac disease. Br. J. Nutr. 2014, 112, 30–40. [Google Scholar] [CrossRef] [PubMed]
- Manley, K.J.; Fraenkel, M.B.; Mayall, B.C.; Power, D.A. Probiotic treatment of vancomycin-resistant enterococci: A randomised controlled trial. Med. J. Aust. 2007, 186, 454–457. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, R.; Meyer, R.; Padmanabhan, R.; Britto, J. Clinical safety of Lactobacillus casei Shirota as a probiotic in critically ill children. J. Pediatr. Gastroenterol. Nutr. 2006, 42, 171–173. [Google Scholar] [CrossRef]
- Kunk, D. Probiotics: Elixir or empty promise? Lancet Gastroenterol. Hepatol. 2019, 4, 81. [Google Scholar] [CrossRef]
- Schnadower, D.; Tarr, P.I.; Casper, T.C.; Gorelick, M.H.; Dean, J.M.; O’Connell, K.J.; Mahajan, P.; Levine, A.C.; Bhatt, S.R.; Roskind, C.G.; et al. Lactobacillus rhamnosus GG versus placebo for acute gastroenteritis in children. N. Engl. J. Med. 2018, 379, 2002–2014. [Google Scholar] [CrossRef] [PubMed]
- Freedman, S.B.; Williamson-Urquhart, S.; Farion, K.J.; Gouin, S.; Willan, A.R.; Poonai, N.; Hurley, K.; Sherman, P.M.; Finkelstein, Y.; Lee, B.E.; et al. Multicenter trial of a combination probiotic for children with gastroenteritis. N. Engl. J. Med. 2018, 379, 2015–2026. [Google Scholar] [CrossRef]
- Suez, J.; Zmora, N.; Segal, E.; Elinav, E. The pros, cons, and many unknowns of probiotics. Nat. Med. 2019, 25, 716–729. [Google Scholar] [CrossRef]
- Goldenberg, J.Z.; Yap, C.; Lytvyn, L.; Lo, C.K.-F.; Beardsley, J.; Mertz, D.; Johnston, B.C. Probiotics for the prevention of Clostridium difficile-associated diarrhea in adults and children. Cochrane Database Syst. Rev. 2017. [Google Scholar] [CrossRef]
- Parker, E.A.; Roy, T.; D’Adamo, C.R.; Wieland, L.S. Probiotics and gastrointestinal conditions: An overview of evidence from the Cochrane Collaboration. Nutrition 2018, 45, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Day, R.L.J.; Harper, A.J.; Woods, R.M.; Davies, O.G.; Heaney, L.M. Probiotics: Current landscape and future horizons. Future Sci. OA 2019, 5, FSO391. [Google Scholar] [CrossRef]
- Lerner, A.; Shoenfeld, Y.; Matthias, T. Probiotics: If it does not help it does not do any harm. Really? Microorganisms 2019, 7, 104. [Google Scholar] [CrossRef] [PubMed]
- Van den Nieuwboer, M.; Brummer, R.J.; Guarner, F.; Morelli, L.; Cabana, M.; Claassen, E. The administration of probiotics and synbiotics in immune compromised adults: Is it safe? Benefic. Microbes 2015, 6, 3–17. [Google Scholar] [CrossRef] [PubMed]
- Doron, S.; Snydman, D.R. Risk and Safety of Probiotics. Clin. Infect. Dis. 2015, 60, S129–S134. [Google Scholar] [CrossRef] [PubMed]
- Van der Geest, A.M.; Flach, J.; Claassen, E.; Sijlmans, A.W.; Van de Burgwal, L.H.M.; Larsen, O.F.A. European General Practitioners perceptions on probiotics: Results of a multinational survey. PharmaNutrition 2020, 11. [Google Scholar] [CrossRef]
- Kokosky, G. Pharmacists Play a Key Role in Counseling Patients about Probiotics. Available online: https://www.pharmacytimes.com/publications/issue/2018/September2018/pharmacists-play-a-key-role-in-counseling-patients-who-may-need-probiotics (accessed on 22 June 2020).
- Happel, A.-U.; Jaumdally, S.Z.; Pidwell, T.; Cornelius, T.; Jaspan, H.B.; Froissart, R.; Barnabas, S.L.; Passmore, J.-A.S. Probiotics for vaginal health in South Africa: What is on retailers’ shelves? BMC Women Health 2017, 17, 1–10. [Google Scholar] [CrossRef]
- Tachedjian, G.; Aldunate, M.; Bradshaw, C.S.; Cone, R.A. The role of lactic acid production by probiotic Lactobacillus species in vaginal health. Res. Microbiol. 2017, 168, 782–792. [Google Scholar] [CrossRef]
- Venugopalan, V.; Shriner, K.A.; Wong-Beringer, A. Regulatory oversight and safety of probiotic use. Emerg. Infect. Dis. 2010, 16, 1661–1665. [Google Scholar] [CrossRef]
- United Kingdom National Health Service. Probiotics. Available online: https://www.nhs.uk/conditions/probiotics (accessed on 4 July 2020).
- De Simone, C. The unregulated probiotic market. Clin. Gastroenterol. Hepatol. 2019, 17, 809–817. [Google Scholar] [CrossRef]
- Gargari, G.; Taverniti, V.; Koirala, R.; Gardana, C.; Guglielmetti, S. Impact of a multistrain probiotic formulation with high bifidobacterial content on the fecal bacterial community and short-chain fatty acid levels of healthy adults. Microorganisms 2020, 8, 492. [Google Scholar] [CrossRef]
- Binnendijk, K.H.; Rijkers, G.T. What is a health benefit? An evaluation of EFSA opinions on health benefits with reference to probiotics. Benef. Microbes 2013, 4, 223–230. [Google Scholar] [CrossRef]
- Betta, P. Not all probiotics are the same: Gut microbiota modulation with a multistrain probiotics. N. Am. J. Med. Sci. 2014, 6, 58. [Google Scholar] [CrossRef] [PubMed]
- Vinderola, G.; Binetti, A.; Burns, P.; Reinheimer, J. Cell viability and functionality of probiotic bacteria in dairy products. Front. Microbiol. 2011, 2, 70. [Google Scholar] [CrossRef] [PubMed]
- California, B.U.o. Probiotics Pros and Cons by Berkeley Wellness. Available online: https://www.berkeleywellness.com/supplements/other-supplements/article/probiotics-pros-and-cons (accessed on 4 July 2020).
- Marteau, P. Safety aspects of probiotic products. Näringsforskning 2016, 45, 22–24. [Google Scholar] [CrossRef][Green Version]
- Anadón, A.; Martínez-Larrañaga, M.R.; Ares, I.; Martínez, M.A. Probiotics: Safety and Toxicity Considerations. In Nutraceuticals; Gupta, R.C., Ed.; Academic Press: Cambridge, MA, USA, 2016; pp. 777–798. [Google Scholar]
- Liong, M.-T. Safety of probiotics: Translocation and infection. Nutr. Rev. 2008, 66, 192–202. [Google Scholar] [CrossRef] [PubMed]
- Sanders, M.E.; Merenstein, D.J.; Ouwehand, A.C.; Reid, G.; Salminen, S.; Cabana, M.D.; Paraskevakos, G.; Leyer, G. Probiotic use in at-risk populations. J. Am. Pharm. Assoc. 2016, 56, 680–686. [Google Scholar] [CrossRef] [PubMed]
- Kothari, D.; Patel, S.; Kim, S.-K. Probiotic supplements might not be universally-effective and safe: A review. Biomed. Pharmacother. 2019, 111, 537–547. [Google Scholar] [CrossRef] [PubMed]
- Redman, M.G.; Ward, E.J.; Phillips, R.S. The efficacy and safety of probiotics in people with cancer: A systematic review. Ann. Oncol. 2014, 25, 1919–1929. [Google Scholar] [CrossRef]
- Nielsen, D.E.; El-Sohemy, A. Applying genomics to nutrition and lifestyle modification. Personal. Med. 2012, 9, 739–749. [Google Scholar] [CrossRef]
- Anukam, K.C. Probiotic toxicity, any evidence? J. Pharmacol. Toxicol. 2007, 2, 590–598. [Google Scholar] [CrossRef][Green Version]
- Ishibashi, N.; Yamazaki, S. Probiotics and safety. Am. J. Clin. Nutr. 2001, 73, 465–470. [Google Scholar] [CrossRef]
- Enright, E.F.; Gahan, C.G.; Joyce, S.A.; Griffin, B.T. The impact of the gut microbiota on drug metabolism and clinical outcome. Yale J. Biol. Med. 2016, 89, 375–382. [Google Scholar] [PubMed]
- Abdollahi, M.; Abdolghaffari, A.H.; Gooshe, M.; Ghasemi-Niri, F. Safety of Probiotic Bacteria. In Probiotics, Prebiotics, and Synbiotics: Bioactive Foods in Health Promotion; Watson, R.R., Victor, R., Preedy, V.R., Eds.; Academic Press: Cambridge, MA, USA, 2016; pp. 227–237. [Google Scholar] [CrossRef]
- LeDoux, D.; LaBombardi, V.J.; Karter, D. Lactobacillus acidophilus bacteraemia after use of a probiotic in a patient with AIDS and Hodgkin’s disease. Int. J. STD AIDS 2016, 17, 280–282. [Google Scholar] [CrossRef] [PubMed]
- Haghighat, L.; Crum-Cianflone, N.F. The potential risks of probiotics among HIV-infected persons: Bacteraemia due to Lactobacillus acidophilus and review of the literature. Int. J. STD AIDS 2016, 27, 1223–1230. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Hicks, C.; Li, Y.; Su, J.; Shiloach, J.; Kaufman, J.B.; Fitz, Y.; Eichacker, P.Q.; Cui, X. Purified cell wall from the probiotic bacterium Lactobacillus gasseri activates systemic inflammation and, at higher doses, produces lethality in a rat model. Crit. Care 2014, 18, R140. [Google Scholar] [CrossRef]
- Venema, K.; Do Carmo, A.P. Probiotics and Prebiotics, Current Research and Future Trends; Caister Academic Press: Norfolk, UK, 2015. [Google Scholar]
- Enache-Angoulvant, A.; Hennequin, C. Invasive Saccharomyces infection: A comprehensive review. Clin. Infect. Dis. 2005, 41, 1559–1568. [Google Scholar] [CrossRef]
- Munoz, P.; Bouza, E.; Cuenca-Estrella, M.; Eiros, J.M.; Perez, M.J.; Sanchez-Somolinos, M.; Rincon, C.; Hortal, J.; Pelaez, T. Saccharomyces cerevisiae fungemia: An emerging infectious disease. Clin. Infect. Dis. 2005, 40, 1625–1634. [Google Scholar] [CrossRef]
- Gao, X.; Liu, J.; Li, L.; Liu, W.; Sun, M. A brief review of nutraceutical ingredients in gastrointestinal disorders: Evidence and suggestions. Int. J. Mol. Sci. 2020, 21, 1822. [Google Scholar] [CrossRef] [PubMed]
- Fadhel, M.; Patel, S.; Liu, E.; Levitt, M.; Asif, A. Saccharomyces cerevisiae fungemia in a critically ill patient with acute cholangitis and long term probiotic use. Med. Mycol. Case Rep. 2019, 23, 23–25. [Google Scholar] [CrossRef]
- Sherid, M.; Samo, S.; Sulaiman, S.; Husein, H.; Sifuentes, H.; Sridhar, S. Liver abscess and bacteremia caused by lactobacillus: Role of probiotics? Case report and review of the literature. BMC Gastroenterol. 2016, 16. [Google Scholar] [CrossRef]
- Sullivan, Å.; Erik Nord, C. Probiotic lactobacilli and bacteraemia in Stockholm. Scand. J. Infect. Dis. 2009, 38, 327–331. [Google Scholar] [CrossRef]
- Cannon, J.P.; Lee, T.A.; Bolanos, J.T.; Danziger, L.H. Pathogenic relevance of Lactobacillus: A retrospective review of over 200 cases. Eur. J. Clin. Microbiol. Infect. Dis. 2004, 24, 31–40. [Google Scholar] [CrossRef]
- Botros, M.; Mukundan, D. Lactobacillus endocarditis with prosthetic material: A case report on non-surgical management with corresponding literature review. Infect. Dis. Rep. 2014, 6. [Google Scholar] [CrossRef] [PubMed]
- Salvana, E.M.T.; Frank, M. Lactobacillus endocarditis: Case report and review of cases reported since 1992. J. Infect. 2006, 53, e5–e10. [Google Scholar] [CrossRef]
- Ze-Ze, L. Case of aortic endocarditis caused by Lactobacillus casei. J. Med. Microbiol. 2004, 53, 451–453. [Google Scholar] [CrossRef] [PubMed]
- Franko, B.; Vaillant, M.; Recule, C.; Vautrin, E.; Brion, J.P.; Pavese, P. Lactobacillus paracasei endocarditis in a consumer of probiotics. Méd. Et Mal. Infect. 2013, 43, 171–173. [Google Scholar] [CrossRef] [PubMed]
- Besselink, M.G.H.; van Santvoort, H.C.; Buskens, E.; Boermeester, M.A.; van Goor, H.; Timmerman, H.M.; Nieuwenhuijs, V.B.; Bollen, T.L.; van Ramshorst, B.; Witteman, B.J.M.; et al. Probiotic prophylaxis in predicted severe acute pancreatitis: A randomised, double-blind, placebo-controlled trial. Lancet 2008, 371, 651–659. [Google Scholar] [CrossRef]
- Taylor, A.L.; Dunstan, J.A.; Prescott, S.L. Probiotic supplementation for the first 6 months of life fails to reduce the risk of atopic dermatitis and increases the risk of allergen sensitization in high-risk children: A randomized controlled trial. J. Allergy Clin. Immunol. 2007, 119, 184–191. [Google Scholar] [CrossRef]
- Pelucchi, C.; Chatenoud, L.; Turati, F.; Galeone, C.; Moja, L.; Bach, J.-F.; La Vecchia, C. Probiotics supplementation during pregnancy or infancy for the prevention of atopic dermatitis. Epidemiology 2012, 23, 402–414. [Google Scholar] [CrossRef]
- Garcia-Larsen, V.; Ierodiakonou, D.; Jarrold, K.; Cunha, S.; Chivinge, J.; Robinson, Z.; Geoghegan, N.; Ruparelia, A.; Devani, P.; Trivella, M.; et al. Diet during pregnancy and infancy and risk of allergic or autoimmune disease: A systematic review and meta-analysis. PLoS Med. 2018, 15. [Google Scholar] [CrossRef]
- Olier, M.; Marcq, I.; Salvador-Cartier, C.; Secher, T.; Dobrindt, U.; Boury, M.; Bacquié, V.; Penary, M.; Gaultier, E.; Nougayrède, J.-P.; et al. Genotoxicity of Escherichia coli Nissle 1917 strain cannot be dissociated from its probiotic activity. Gut Microb. 2014, 3, 501–509. [Google Scholar] [CrossRef]
- Imperial, I.C.V.J.; Ibana, J.A. Addressing the antibiotic resistance problem with probiotics: Reducing the risk of its double-edged sword effect. Front. Microbiol. 2016, 07, 1983. [Google Scholar] [CrossRef]
- Ashraf, R.; Shah, N.P. Antibiotic resistance of probiotic organisms and safety of probiotic dairy products. Int. Food Res. J. 2011, 18, 837–853. [Google Scholar]
- Gueimonde, M.; Sánchez, B.G.; De los Reyes-Gavilán, C.; Margolles, A. Antibiotic resistance in probiotic bacteria. Front. Microbiol. 2013, 4, 202. [Google Scholar] [CrossRef]
- Connolly, E.; Abrahamsson, T.; Björkstén, B. Safety of D(-)-lactic acid producing bacteria in the human infant. J. Pediatr. Gastroenterol. Nutr. 2005, 41, 489–492. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.S.C.; Rehman, A.; Yu, S.; de Andino, N.M. Brain fogginess, gas and bloating: A link between SIBO, probiotics and metabolic acidosis. Clin. Transl. Gastroenterol. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Munakata, S.; Arakawa, C.; Kohira, R.; Fujita, Y.; Fuchigami, T.; Mugishima, H. A case of D-lactic acid encephalopathy associated with use of probiotics. Brain Dev. 2010, 32, 691–694. [Google Scholar] [CrossRef]
- Alvarez, M.A.; Moreno-Arribas, M.V. The problem of biogenic amines in fermented foods and the use of potential biogenic amine-degrading microorganisms as a solution. Trends Food Sci. Technol. 2014, 39, 146–155. [Google Scholar] [CrossRef]
- Kuley, E.; Balıkcı, E.; Özoğul, İ.; Gökdogan, S.; Özoğul, F. Stimulation of cadaverine production by foodborne pathogens in the presence of Lactobacillus, Lactococcus, and Streptococcus spp. J. Food Sci. 2012, 77, M650–M658. [Google Scholar] [CrossRef]
- Jansen, S.C.; van Dusseldorp, M.; Bottema, K.C.; Dubois, A.E.J. Intolerance to dietary biogenic amines: A review. Ann. Allergy Asthma Immunol. 2003, 91, 233–241. [Google Scholar] [CrossRef]
- Linares, D.M.; Martín, M.; Ladero, V.; Alvarez, M.A.; Fernández, M. Biogenic amines in dairy products. Crit. Rev. Food Sci. Nutr. 2011, 51, 691–703. [Google Scholar] [CrossRef]
- Terpou, A.; Papadaki, A.; Lappa, I.K.; Kachrimanidou, V.; Bosnea, L.A.; Kopsahelis, N. Probiotics in food systems: Significance and emerging strategies towards improved viability and delivery of enhanced beneficial value. Nutrients 2019, 11, 1591. [Google Scholar] [CrossRef] [PubMed]
- Hungin, A.P.S.; Mitchell, C.R.; Whorwell, P.; Mulligan, C.; Cole, O.; Agréus, L.; Fracasso, P.; Lionis, C.; Mendive, J.; Philippart de Foy, J.M.; et al. Systematic review: Probiotics in the management of lower gastrointestinal symptoms-An updated evidence-based international consensus. Aliment. Pharmacol. Ther. 2018, 47, 1054–1070. [Google Scholar] [CrossRef]
- Williams, N.T. Probiotics. Am. J. Health Syst. Pharm. 2010, 67, 449–458. [Google Scholar] [CrossRef] [PubMed]
- Pakdaman, M.N.; Udani, J.K.; Molina, J.P.; Shahani, M. The effects of the DDS-1 strain of lactobacillus on symptomatic relief for lactose intolerance-a randomized, double-blind, placebo-controlled, crossover clinical trial. Nutr. J. 2016, 15, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Fernandes Pereira, A.L. Turning Fruit Juice into Probiotic Beverages. In Fruit Juices Extraction, Composition, Qquality and Analysis; Rajauria, G., Tiwari, B., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 279–285. [Google Scholar]
- Koziolek, M.; Alcaro, S.; Augustijns, P.; Basit, A.W.; Grimm, M.; Hens, B.; Hoad, C.L.; Jedamzik, P.; Madla, C.M.; Maliepaard, M.; et al. The mechanisms of pharmacokinetic food-drug interactions – A perspective from the UNGAP group. Eur. J. Pharm. Sci. 2019, 134, 31–59. [Google Scholar] [CrossRef]
- Noh, K.; Kang, Y.R.; Nepal, M.R.; Shakya, R.; Kang, M.J.; Kang, W.; Lee, S.; Jeong, H.G.; Jeong, T.C. Impact of gut microbiota on drug metabolism: An update for safe and effective use of drugs. Arch. Pharm. Res. 2017, 40, 1345–1355. [Google Scholar] [CrossRef]
- Wilson, I.D.; Nicholson, J.K. Gut microbiome interactions with drug metabolism, efficacy, and toxicity. Transl. Res. 2017, 179, 204–222. [Google Scholar] [CrossRef]
- El Aidy, S.; van den Bogert, B.; Kleerebezem, M. The small intestine microbiota, nutritional modulation and relevance for health. Curr. Opin. Biotechnol. 2015, 32, 14–20. [Google Scholar] [CrossRef]
- Deli, M.A.; Matuskova, Z.; Anzenbacherova, E.; Vecera, R.; Tlaskalova-Hogenova, H.; Kolar, M.; Anzenbacher, P. Administration of a probiotic can change drug pharmacokinetics: Effect of E. coli Nissle 1917 on amidarone absorption in rats. PLoS ONE 2014, 9, e87150. [Google Scholar] [CrossRef]
- Mikov, M.; Đanić, M.; Pavlović, N.; Stanimirov, B.; Goločorbin-Kon, S.; Stankov, K.; Al-Salami, H. Potential applications of gliclazide in treating Type 1 Diabetes Mellitus: Formulation with bile acids and probiotics. Eur. J. Drug Metab. Pharmacokinet. 2017, 43, 269–280. [Google Scholar] [CrossRef]
- Saputri, F.A.; Kang, D.; Kusuma, A.S.W.; Rusdiana, T.; Hasanah, A.N.; Mutakin; Surono, I.S.; Koyama, H.; Abdulah, R. Lactobacillus plantarum IS-10506 probiotic administration increases amlodipine absorption in a rabbit model. J. Int. Med. Res. 2018, 46, 5004–5010. [Google Scholar] [CrossRef] [PubMed]
- Al-Salami, H.; Butt, G.; Tucker, I.; Skrbic, R.; Golocorbin-Kon, S.; Mikov, M. Probiotic pre-treatment reduces gliclazide permeation (ex vivo) in healthy rats but increases it in diabetic rats to the level seen in untreated healthy rats. Arch. Drug Inf. 2008, 1, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Stepkowski, S.; Lee, J.R.; Muthukumar, T.; Dadhania, D.; Taur, Y.; Jenq, R.R.; Toussaint, N.C.; Ling, L.; Pamer, E.; Suthanthiran, M. Gut microbiota and tacrolimus dosing in kidney transplantation. PLoS ONE 2015, 10, e0122399. [Google Scholar] [CrossRef]
- Holm, R.; Müllertz, A.; Mu, H. Bile salts and their importance for drug absorption. Int. J. Pharm. 2013, 453, 44–55. [Google Scholar] [CrossRef] [PubMed]
- Pavlović, N.; Goločorbin-Kon, S.; Ðanić, M.; Stanimirov, B.; Al-Salami, H.; Stankov, K.; Mikov, M. Bile acids and their derivatives as potential modifiers of drug release and pharmacokinetic profiles. Front. Pharmacol. 2018, 9, 1283. [Google Scholar] [CrossRef] [PubMed]
- Moghimipour, E.; Ameri, A.; Handali, S. Absorption-enhancing effects of bile salts. Molecules 2015, 20, 14451–14473. [Google Scholar] [CrossRef]
- Pavlović, N.; Stankov, K.; Mikov, M. Probiotics—Interactions with bile acids and impact on cholesterol metabolism. Appl. Biochem. Biotechnol. 2012, 168, 1880–1895. [Google Scholar] [CrossRef]
- Mikawlrawng, K.; Kumar, S.; Bhatnagar, K. Drug interactions, safety and efficacy of probiotics. Asian J. Med. Health 2016, 1, 1–8. [Google Scholar] [CrossRef]
- Vivarelli, S.; Falzone, L.; Basile, M.; Nicolosi, D.; Genovese, C.; Libra, M.; Salmeri, M. Benefits of using probiotics as adjuvants in anticancer therapy (Review). World Acad. Sci. J. 2019. [Google Scholar] [CrossRef]
- Mego, M.; Chovanec, J.; Vochyanova-Andrezalova, I.; Konkolovsky, P.; Mikulova, M.; Reckova, M.; Miskovska, V.; Bystricky, B.; Beniak, J.; Medvecova, L.; et al. Prevention of irinotecan induced diarrhea by probiotics: A randomized double blind, placebo controlled pilot study. Complement. Ther. Med. 2015, 23, 356–362. [Google Scholar] [CrossRef]
- Saarela, M.; Mogensen, G.; Fondén, R.; Mättö, J.; Mattila-Sandholm, T. Probiotic bacteria: Safety, functional and technological properties. J. Biotechnol. 2000, 84, 197–215. [Google Scholar] [CrossRef]
- Pradhan, D.; Mallappa, R.H.; Grover, S. Comprehensive approaches for assessing the safety of probiotic bacteria. Food Control 2020, 108, 106872. [Google Scholar] [CrossRef]
- Zielińska, D.; Sionek, B.; Kołożyn-Krajewska, D. Safety of Probiotics. In Diet, Microbiome and Health; Mihai Grumezescu, A., Holban, A.M., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 131–161. [Google Scholar] [CrossRef]
- Khedkar, S.; Carraresi, L.; Bröring, S. Food or pharmaceuticals? Consumers’ perception of health-related borderline products. PharmaNutrition 2017, 5, 133–140. [Google Scholar] [CrossRef]
- Granato, D.; Barba, F.J.; Bursać Kovačević, D.; Lorenzo, J.M.; Cruz, A.G.; Putnik, P. Functional Foods: Product Development, Technological Trends, Efficacy Testing, and Safety. Annu. Rev. Food Sci. Technol. 2020, 11, 93–118. [Google Scholar] [CrossRef]
- Zendeboodi, F.; Khorshidian, N.; Mortazavian, A.M.; da Cruz, A.G. Probiotic: Conceptualization from a new approach. Curr. Opin. Food Sci. 2020, 32, 103–123. [Google Scholar] [CrossRef]
- Sarao, L.K.; Arora, M. Probiotics, prebiotics, and microencapsulation: A review. Crit. Rev. Food Sci. Nutr. 2017, 57, 344–371. [Google Scholar] [CrossRef]
- Liu, X.; Champagne, C.P.; Lee, B.H.; Boye, J.I.; Casgrain, M. Thermostability of probiotics and their α-galactosidases and the potential for bean products. Biotechnol. Res. Int. 2014, 2014, 1–21. [Google Scholar] [CrossRef]
- Konuray, G.; Erginkaya, Z. Potential use of Bacillus coagulans in the food industry. Foods 2018, 7, 92. [Google Scholar] [CrossRef]
- Banik, A.; Halder, S.K.; Ghosh, C.; Mondal, K.C. Fungal probiotics: Opportunity, challenge, and prospects. In Recent Advancement in White Biotechnology Through Fungi; Yadav, A., Singh, S., Mishra, S., Gupta, A., Eds.; Springer: Cham, Switzerland, 2019; pp. 101–117. [Google Scholar] [CrossRef]
- Champagne, C.P.; Gomes da Cruz, A.; Daga, M. Strategies to improve the functionality of probiotics in supplements and foods. Curr. Opin. Food Sci. 2018, 22, 160–166. [Google Scholar] [CrossRef]
- Wilkinson, M.G. Flow cytometry as a potential method of measuring bacterial viability in probiotic products: A review. Trends Food Sci. Technol. 2018, 78, 1–10. [Google Scholar] [CrossRef]
- Putta, S.; Yarla, N.S.; Lakkappa, D.B.; Imandi, S.B.; Malla, R.R.; Chaitanya, A.K.; Chari, B.P.V.; Saka, S.; Vechalapu, R.R.; Kamal, M.A.; et al. Probiotics: Supplements, Food, pharmaceutical Industry. In Therapeutic, Probiotic, and Unconventional Foods; Grumezescu, A.M., Holban, A.M., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 15–25. [Google Scholar] [CrossRef]
- Kalima, P.; Masterton, R.G.; Roddie, P.H.; Thomas, A.E. Lactobacillus rhamnosus infection in a child following bone marrow transplant. J. Infect. 1996, 32, 165–167. [Google Scholar] [CrossRef]
- Rautio, M.; Jousimies-Somer, H.; Kauma, H.; Pietarinen, I.; Saxelin, M.; Tynkkynen, S.; Koskela, M. Liver abscess due to a Lactobacillus rhamnosus strain indistinguishable from L. rhamnosus strain GG. Clin. Infect. Dis. 1999, 28, 1159–1160. [Google Scholar] [CrossRef] [PubMed]
- Elsanhoty, R.M.; Salam, S.A.; Ramadan, M.F.; Badr, F.H. Detoxification of aflatoxin M1 in yoghurt using probiotics and lactic acid bacteria. Food Control 2014, 43, 129–134. [Google Scholar] [CrossRef]
- AIJN. AIJF European Fruit Juice Association. Annual Report 2018. Available online: http://viewer.zmags.com/publication/bc62cfea#/bc62cfea/1 (accessed on 12 May 2018).
- Pimentel, T.C.; Klososki, S.J.; Rosset, M.; Barão, C.E.; Marcolino, V.A. Fruit Juices as Probiotic Foods. In Sports and Energy Drinks; Grumezescu, A., Holban, A.M., Eds.; Academic Press: Cambridge, MA, USA, 2019; pp. 483–513. [Google Scholar] [CrossRef]
- Šárka, H.; Kristýna, R.; Kristina, B.; Iveta, K.; Marcela, S. Fruit juices with probiotics – new type of functional foods. Czech J. Food Sci. 2018, 36, 284–288. [Google Scholar] [CrossRef]
- Nagpal, R.; Kumar, A.; Kumar, M. Fortification and fermentation of fruit juices with probiotic lactobacilli. Ann. Microbiol. 2012, 62, 1573–1578. [Google Scholar] [CrossRef]
- Shah, N.P.; Ding, W.K.; Fallourd, M.J.; Leyer, G. Improving the stability of probiotic bacteria in model fruit juices using vitamins and antioxidants. J. Food Sci. 2010. [Google Scholar] [CrossRef]
- Coman, M.M.; Silvi, S.; Verdenelli, M.C.; Cecchini, C.; Orpianesi, C.; Cresc, A. Fruit and vegetable juices tested as possible probiotic beverage. Agro Food Ind. Hi Tech 2010, 21, 1–4. [Google Scholar]
- Speranza, B.; Petruzzi, L.; Bevilacqua, A.; Gallo, M.; Campaniello, D.; Sinigaglia, M.; Corbo, M.R. Encapsulation of active compounds in fruit and vegetable juice processing: Current state and perspectives. J. Food Sci. 2017, 82, 1291–1301. [Google Scholar] [CrossRef]
- Rodríguez-Roque, M.J.; Rojas-Graü, M.A.; Elez-Martínez, P.; Martín-Belloso, O. In vitro bioaccessibility of health-related compounds as affected by the formulation of fruit juice- and milk-based beverages. Food Res. Int. 2014, 62, 771–778. [Google Scholar] [CrossRef]
- Fenster, K.; Freeburg, B.; Hollard, C.; Wong, C.; Rønhave Laursen, R.; Ouwehand, A. The production and delivery of probiotics: A review of a practical approach. Microorganisms 2019, 7, 83. [Google Scholar] [CrossRef] [PubMed]
- Saarela, M.; Alakomi, H.L.; Mättö, J.; Ahonen, A.M.; Tynkkynen, S. Acid tolerant mutants of Bifidobacterium animalis subsp. lactis with improved stability in fruit juice. LWT Food Sci. Technol. 2011, 44, 1012–1018. [Google Scholar] [CrossRef]
- Champagne, C.P.; Gardner, N.J. Effect of storage in a fruit drink on subsequent survival of probiotic lactobacilli to gastro-intestinal stresses. Food Res. Int. 2008, 41, 539–543. [Google Scholar] [CrossRef]
- Antunes, A.E.C.; Liserre, A.M.; Coelho, A.L.A.; Menezes, C.R.; Moreno, I.; Yotsuyanagi, K.; Azambuja, N.C. Acerola nectar with added microencapsulated probiotic. LWT Food Sci. Technol. 2013, 54, 125–131. [Google Scholar] [CrossRef]
- Martins, E.M.F.; Ramos, A.M.; Vanzela, E.S.L.; Stringheta, P.C.; de Oliveira Pinto, C.L.; Martins, J.M. Products of vegetable origin: A new alternative for the consumption of probiotic bacteria. Food Res. Int. 2013, 51, 764–770. [Google Scholar] [CrossRef]
- Nicolesco, C.L.; Buruleanu, L.C. Correlation of some substrate parameters in growing Lactobacillus acidophilus on vegetable and fruit cocktail juices. Bull. Univ. Agric. Sci. Vet. Med. Cluj Napoca Agric. 2010, 67, 352–359. [Google Scholar]
- Nualkaekul, S.; Charalampopoulos, D. Survival of Lactobacillus plantarum in model solutions and fruit juices. Int. J. Food Microbiol. 2011, 146, 111–117. [Google Scholar] [CrossRef]
- Sheela, T.; Suganya, R.S. Studies on anti-diarrhoeal activity of synbiotic plums juice. Int. J. Sci. Res. Publ. 2012, 2, 1–5. [Google Scholar]
- Priya, N.; Vasudevan, A. Effect of lactic acid bacteria in development of papaya juice using response surface methodology. Int. J. Biotechnol. Biochem. 2016, 12, 27–32. [Google Scholar]
- Da Costa, G.M.; De Carvalho Silva, J.V.; Mingotti, J.D.; Barão, C.E.; Klososki, S.J.; Pimentel, T.C. Effect of ascorbic acid or oligofructose supplementation on L. paracasei viability, physicochemical characteristics and acceptance of probiotic orange juice. LWT Food Sci. Technol. 2017, 75, 195–201. [Google Scholar] [CrossRef]
- Pimentel, T.C.; Madrona, G.S.; Garcia, S.; Prudencio, S.H. Probiotic viability, physicochemical characteristics and acceptability during refrigerated storage of clarified apple juice supplemented with Lactobacillus paracasei ssp. paracasei and oligofructose in different package type. LWT Food Sci. Technol. 2015, 63, 415–422. [Google Scholar] [CrossRef]
- Patel, A.R. Probiotic fruit and vegetable juices-recent advances and future perspectiv. Int. Food Res. J. 2017, 25, 1850–1857. [Google Scholar]
- Perricone, M.; Bevilacqua, A.; Altieri, C.; Sinigaglia, M.; Corbo, M. Challenges for the production of probiotic fruit juices. Beverages 2015, 1, 95–103. [Google Scholar] [CrossRef]
- Ding, W.K.; Shah, N.P. Survival of free and microencapsulated probiotic bacteria in orange and apple juice. Int. Food Res. J. 2008, 15, 219–232. [Google Scholar]
- Rakin, M.; Vukasinovic, M.; Siler-Marinkovic, S.; Maksimovic, M. Contribution of lactic acid fermentation to improved nutritive quality vegetable juices enriched with brewer’s yeast autolysate. Food Chem. 2007, 100, 599–602. [Google Scholar] [CrossRef]
- Gaudreau, H.; Champagne, C.P.; Remondetto, G.E.; Bazinet, L.; Subirade, M. Effect of catechins on the growth of oxygen-sensitive probiotic bacteria. Food Res. Int. 2013, 53, 751–757. [Google Scholar] [CrossRef]
- Granato, D.; Putnik, P.; Bursać Kovačević, D.; Santos, J.S.; Calado, V.; Rocha, R.S.; Cruz, A.G.D.; Jarvis, B.; Rodionova, O.Y.; Pomerantsev, A. Trends in chemometrics: Food authentication, microbiology, and effects of processing. Compr. Rev. Food Sci. Food Saf. 2018, 17, 663–677. [Google Scholar] [CrossRef]
- Rinke, P.; Jamin, E. Fruit Juices. In Food Integrity Handbook; 2018; pp. 243–264. Available online: https://secure.fera.defra.gov.uk/foodintegrity/index.cfm?sectionid=83 (accessed on 4 July 2020).
- Vallabhaneni, S.; Walker, T.A.; Lockhart, S.R.; Ng, D.; Chiller, T.; Melchreit, R.; Brandt, M.E.; Smith, R.M. Notes from the field: Fatal gastrointestinal mucormycosis in a premature infant associated with a contaminated dietary supplement-Connecticut, 2014. MMWR Morb. Mortal. Wkly. Rep. 2015, 64, 155–156. [Google Scholar]
- Santeramo, F.G.; Carlucci, D.; De Devitiis, B.; Seccia, A.; Stasi, A.; Viscecchia, R.; Nardone, G. Emerging trends in European food, diets and food industry. Food Res. Int. 2018, 104, 39–47. [Google Scholar] [CrossRef]
- Mantzourani, I.; Nouska, C.; Terpou, A.; Alexopoulos, A.; Bezirtzoglou, E.; Panayiotidis, M.; Galanis, A.; Plessas, S. Production of a novel functional fruit beverage consisting of cornelian cherry juice and probiotic bacteria. Antioxidants 2018, 7, 163. [Google Scholar] [CrossRef]
- Putnik, P.; Pavlić, B.; Šojić, B.; Zavadlav, S.; Žuntar, I.; Kao, L.; Kitonić, D.; Bursać Kovačević, D. Innovative hurdle technologies for the preservation of functional fruit juices. Foods 2020, 9, 699. [Google Scholar] [CrossRef]
| Lactobacillus Species | Bifidobacterium Species | Others |
|---|---|---|
| L. acidophilus | B. adolescentis B. animalis B. breve B. bifidum B. infantis B. lactis B. longum | Bacillus coagulans |
| L. amylovorus | Bacillus cereus | |
| L. brevis | Clostridium botyricum | |
| L. casei | Enterococcus faecalis | |
| L. rhamnosus | Enterococcus faecium | |
| L. crispatus | Escherichia coli | |
| L. delbrueckii subsp. Bulgaricus | Lactococcus lactis subsp. Cremoris | |
| L. fermentum | Lactococcus lactis subsp. Lactis | |
| L. gasseri | Leuconostoc mesenteroides subsp. Dextranicum | |
| L. helveticus | Pediococcus acidilactici | |
| L. johnsonii | Propionibacterium freudenreichii | |
| L. lactis | Saccharomyces boulardii | |
| L. paracasei | Streptococcus salivarius subsp. Thermophilus | |
| L. plantarum | Sporolactobacillus inulinus | |
| L. reuteri | ||
| L. salivarius | ||
| L. gallinarum |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Žuntar, I.; Petric, Z.; Bursać Kovačević, D.; Putnik, P. Safety of Probiotics: Functional Fruit Beverages and Nutraceuticals. Foods 2020, 9, 947. https://doi.org/10.3390/foods9070947
Žuntar I, Petric Z, Bursać Kovačević D, Putnik P. Safety of Probiotics: Functional Fruit Beverages and Nutraceuticals. Foods. 2020; 9(7):947. https://doi.org/10.3390/foods9070947
Chicago/Turabian StyleŽuntar, Irena, Zvonimir Petric, Danijela Bursać Kovačević, and Predrag Putnik. 2020. "Safety of Probiotics: Functional Fruit Beverages and Nutraceuticals" Foods 9, no. 7: 947. https://doi.org/10.3390/foods9070947
APA StyleŽuntar, I., Petric, Z., Bursać Kovačević, D., & Putnik, P. (2020). Safety of Probiotics: Functional Fruit Beverages and Nutraceuticals. Foods, 9(7), 947. https://doi.org/10.3390/foods9070947







