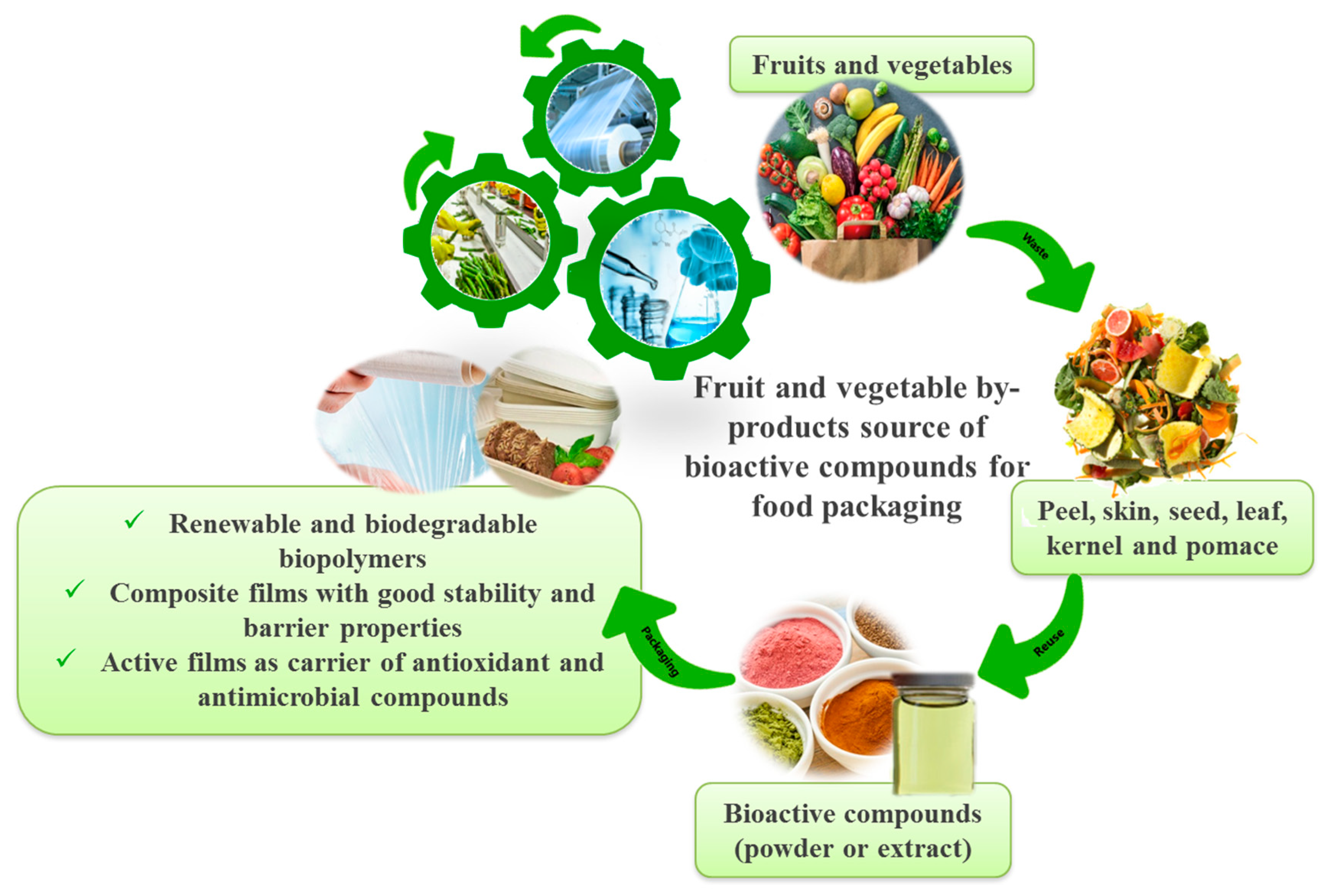
Sustainable Use of Fruit and Vegetable By-Products to Enhance Food Packaging Performance
Abstract
1. Introduction
2. Materials and Methods
3. Fruit and Vegetable By-Products to Enhance Antioxidant Properties of Polymeric Film
3.1. Fruit By-Products
3.2. Olive-Oil Industrial By-Products
4. Fruit and Vegetable By-Products to Enhance Antimicrobial Properties of Polymeric Film
Pomegranate, Grape and Grapefruit By-Products
5. Fruit and Vegetable By-Products to Improve Physical and Mechanical Properties of Films
5.1. Fruit By-Products
5.2. Vegetable By-Products
6. Final Considerations
Author Contributions
Funding
Conflicts of Interest
References
- FAO. Global Food Losses and Waste. Extent, Causes and Prevention; FAO: Rome, Italy, 2011. [Google Scholar]
- Tsang, Y.F.; Kumar, V.; Samadar, P.; Yang, Y.; Lee, J.; Sik Ok, Y.; Song, H.; Kim, K.-H.; Kwon, E.E.; Jeon, Y.J. Production of bioplastic through food waste valorization. Environ. Int. 2019, 127, 625–644. [Google Scholar] [CrossRef] [PubMed]
- FAO. Food Wastage Footprint. Impacts on Natural Resources; FAO: Rome, Italy, 2013. [Google Scholar]
- Yeo, J.; Oh, J.-I.; Cheung, H.H.L.; Lee, P.K.H.; Kyoungjin, A. Smart Food Waste Recycling Bin (S-FRB) to turn food waste into green energy resources. J. Environ. Manag. 2019, 234, 290–296. [Google Scholar] [CrossRef] [PubMed]
- RedCorn, R.; Fatemi, S.; Engelberth, A.S. Comparing end-use potential for industrial food waste sources. Eng. 2018, 4, 371–380. [Google Scholar] [CrossRef]
- Andrade, R.M.S.; Ferreira, M.S.L.; Gonçalves, È.C.B.A. Development and characterization of edible films based on fruit and vegetable residues. J. Food Sci. 2016, 81, E412–E418. [Google Scholar] [CrossRef]
- Majerska, J.; Michalska, A.; Figiel, A. A review of new directions in managing fruit and vegetable processing by-products. Trends Food Sci. Technol. 2019, 88, 207–219. [Google Scholar] [CrossRef]
- Rao, P.; Rathod, V. Valorization of Food and Agricultural Waste: A Step towards Greener Future. Chem. Rec. 2019, 19, 1858–1871. [Google Scholar] [CrossRef] [PubMed]
- Schieber, A. Side streams of plant food processing as a source of valuable compounds: Selected examples. Annu. Rev. Food Sci. Technol. 2017, 8, 97–112. [Google Scholar] [CrossRef]
- Dahiya, S.; Kumar, A.N.; Sravan, J.S.; Chatterjee, S.; Sarkar, O.; Mohan, S.V. Food waste biorefinery: Sustainable strategy for circular bioeconomy. Bioresour. Technol. 2018, 248, 2–12. [Google Scholar] [CrossRef]
- Banerjee, J.; Singh, R.; Vijayaraghavan, R.; MacFarlane, D.; Patti, A.F.; Arora, A. Bioactives from fruit processing wastes: Green approaches to valuable chemicals. Food Chem. 2017, 225, 10–22. [Google Scholar] [CrossRef]
- Demichelis, F.; Pleissner, D.; Fiore, S.; Mariano, S.; Gutiérrez, I.M.N.; Schneider, R.; Venus, J. Investigation of food waste valorization through sequential lactic acid fermentative production and anaerobic digestion of fermentation residues. Bioresour. Technol. 2017, 241, 508–516. [Google Scholar] [CrossRef]
- Kowalska, H.; Czajkowska, K.; Cichowska, J.; Lenart, A. What’s new in biopotential of fruit and vegetable by-products applied in the food processing industry. Trends Food Sci. Technol. 2017, 67, 150–159. [Google Scholar] [CrossRef]
- Bonilla-Hermosa, V.A.; Duarte, W.F.; Schwan, R.F. Utilization of coffee by-products obtained from semi-washed process for production of value-added compounds. Bioresour. Technol. 2014, 166, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Panda, S.K.; Mishra, S.S.; Kayitesi, E.; Ray, R.C. Microbial-processing of fruit and vegetable wastes for production of vital enzymes and organic acids: Biotechnology and scopes. Enviro. Res. 2016, 146, 161–172. [Google Scholar] [CrossRef] [PubMed]
- Ferrentino, G.; Asaduzzaman, M.D.; Scampicchio, M.M. Current technologies and new insights for the recovery of high valuable compounds from fruits by-products. Crit. Rev. Food Sci. Nutr. 2018, 3, 386–404. [Google Scholar] [CrossRef] [PubMed]
- Gull, A.; Prasad, K.; Kumar, P. Effect of millet flours and carrot pomace on cooking qualities, color and texture of developed pasta. LWT Food Sci. Technol. 2015, 63, 470–474. [Google Scholar] [CrossRef]
- Padalino, L.; D’Antuono, I.; Durante, M.; Conte, A.; Cardinali, A.; Linsalata, V.; Mita, G.; Logrieco, A.F.; Del Nobile, M.A. Use of Olive Oil Industrial By-Product for Pasta Enrichment. Antioxidants 2018, 7, 59. [Google Scholar] [CrossRef]
- Gawlik-Dziki, U.; Kaszuba, K.; Piwowarczyk, K.; Świeca, M.; Dziki, D.; Czyż, J. Onion skin Raw material for the production of supplement that enhances the health-beneficial properties of wheat bread. Food Res. Int. 2015, 73, 97–106. [Google Scholar] [CrossRef]
- Spinelli, S.; Conte, A.; Del Nobile, M.A. Microencapsulation of extracted bioactive compounds from brewer’s spent grain to enrich fish-burgers. Food Bioprod. Process. 2016, 100, 450–456. [Google Scholar] [CrossRef]
- Cedola, A.; Cardinali, A.; Del Nobile, M.A.; Conte, A. Fish burger enriched by olive oil industrial by-product. Food Sci. Nutr. 2017, 5, 837–844. [Google Scholar] [CrossRef]
- Bey, N.; Debbebi, H.; Abidi, F.; Marzouki, M.N.; Salah, A.B. The non-edible parts of fennel (Fœniculum vulgare) as a new milk-clotting protease source. Ind. Crop. Prod. 2018, 112, 181–187. [Google Scholar] [CrossRef]
- Lucera, A.; Costa, C.; Marinelli, V.; Saccotelli, M.A.; Del Nobile, M.A.; Conte, A. Fruit and vegetable by-products to fortify spreadable cheese. Antioxidants 2018, 7, 61. [Google Scholar] [CrossRef] [PubMed]
- Mir, S.A.; Don Bosco, S.J.; Shah, M.A.; Santhalakshmy, S.; Mir, M.M. Effect of apple pomace on quality characteristics of brown rice-based cracker. J. Saudi Soc. Agric. Sci. 2017, 16, 25–32. [Google Scholar] [CrossRef]
- Alongi, M.; Melchior, S.; Anese, M. Reducing the glycemic index of short dough biscuits by using apple pomace as a functional ingredient. LWT Food Sci. Technol. 2019, 100, 300–305. [Google Scholar] [CrossRef]
- Soto-Maldonado, C.; Concha-Olmos, J.; Cáceres-Escobar, G.; Meneses-Gόmez, P. Sensory evaluation and glycaemic index of a food developed with flour from whole (pulp and peel) overripe banana (Musa cavendishii) discards. LWT Food Sci. Technol. 2018, 92, 560–575. [Google Scholar] [CrossRef]
- Marinelli, V.; Padalino, L.; Nardiello, D.; Del Nobile, M.A.; Conte, A. New approach to enrich pasta with polyphenols from grape marc. J. Chem. 2015, 2015, 734578. [Google Scholar] [CrossRef]
- Smith, I.N.; Yu, J. Nutritional and sensory quality of bread containing different quantities of grape pomace from different grape cultivars. EC Nutr. 2015, 2, 291–301. [Google Scholar]
- Kaderides, K.; Mourtzinos, I.; Goula, A.M. Stability of pomegranate peel polyphenols encapsulated in orange juice industry by-product and their incorporation in cookies. Food Chem. 2020, 310, 125849. [Google Scholar] [CrossRef]
- Wang, X.; Kristo, E.; LaPointe, G. The effect of apple pomace on the texture, rheology and microstructure of set type yogurt. Food Hydrocoll. 2019, 91, 83–91. [Google Scholar] [CrossRef]
- Chan, C.-L.; Gan, R.-Y.; Shah, N.P.; Corke, H. Enhancing antioxidant capacity of Lactobacillus acidophilus-fermented milk fortified with pomegranate peel extracts. Food Biosci. 2018, 26, 185–192. [Google Scholar] [CrossRef]
- Otoni, C.G.; Avena-Bustillos, R.J.; Azeredo, H.M.C.; Lorevice, M.V.; Moura, M.R.; Mattoso, L.H.C.; McHugh, T.H. Recent advances on edible films based on fruits and vegetables—A review. Compr. Rev. Food Sci. Food Saf. 2017, 16, 1151–1169. [Google Scholar] [CrossRef]
- Deng, Q.; Zhao, Y. Physicochemical, nutritional, and antimicrobial properties of wine grape (cv. Merlot) pomace extract-based films. J. Food Sci. 2011, 76, 309–317. [Google Scholar] [CrossRef]
- Xu, Y.; Scales, A.; Jordan, K.; Kim, C.; Sismour, E. Starch nanocomposite films incorporating grape pomace extract and cellulose nanocrystal. J. Appl. Polym. Sci. 2017, 134, 44438. [Google Scholar] [CrossRef]
- Etxabide, A.; Uranga, J.; Guerrero, P.; de la Caba, K. Development of active gelatin films by means of valorization of food processing waste: A review. Food Hydrocoll. 2017, 68, 192–198. [Google Scholar] [CrossRef]
- Luchese, C.L.; Uranga, J.; Spada, J.C.; Tessaro, I.C.; de la Caba, K. Valorization of blueberry waste and use of compression to manufacture sustainable starch films with enhanced properties. Int. J. Biol. Macromol. 2018, 115, 955–960. [Google Scholar] [CrossRef] [PubMed]
- Del Nobile, M.A.; Conte, A.; Buonocore, G.G.; Incoronato, A.L.; Massaro, A.; Panza, O. Active packaging by extrusion processing of recyclable and biodegradable polymers. J. Food Eng. 2009, 93, 1–6. [Google Scholar] [CrossRef]
- Mastromatteo, M.; Chillo, S.; Buonocore, G.G.; Massaro, A.; Conte, A.; Del Nobile, M.A. Effects of spelt and wheat bran on the performances of wheat gluten films. J. Food Eng. 2008, 88, 202–212. [Google Scholar] [CrossRef]
- Urbina, L.; Eceiza, A.; Gabilondo, N.; Corcuera, M.A.; Retegi, A. Valorization of apple waste for active packaging: Multicomponent T polyhydroxyalkanoate coated nanopapers with improved hydrophobicity and antioxidant capacity. Food Packag. Shelf Life 2019, 21, 100356. [Google Scholar] [CrossRef]
- Ferrentino, G.; Morozova, K.; Mosibo, O.K.; Ramezani, M.; Scampicchio, M. Biorecovery of antioxidants from apple pomace by supercritical fluid extraction. J. Clean. Prod. 2018, 186, 253–261. [Google Scholar] [CrossRef]
- Li, W.; Yang, R.; Ying, D.; Yu, J.; Sanguansri, L.; Augustin, M.A. Analysis of polyphenols in apple pomace: A comparative study of different extraction and hydrolysis procedures. Ind. Crop. Prod. 2020, 147, 112250. [Google Scholar] [CrossRef]
- Jiménez-Moreno, N.; Cimminelli, M.J.; Volpe, F.; Ansó, R.; Esparza, I.; Mármol, I.; Rodríguez-Yoldi, M.J.; Ancín-Azpilicueta, C. Phenolic composition of artichoke waste and its antioxidant capacity on differentiated caco-2 cells. Nutrients 2019, 11, 1723. [Google Scholar] [CrossRef]
- Sihem, D.; Samia, D.; Pandino, G.; Lombardo, S.; Mauromicale, G.; Chahdoura, H.; Gasco, L.; Noureddine, H.A. In vitro antioxidant activities and phenolic content in crop residues of Tunisian globe artichoke. Sci. Hortic. 2015, 190, 128–136. [Google Scholar] [CrossRef]
- Cruz-Tirado, J.P.; Vejarano, R.; Tapia-Blácido, D.R.; Angelats-Silva, L.M.; Siche, R. The addition of sugarcane bagasse and asparagus peel enhances the properties of sweet potato starch foams. Packag. Technol. Sci. 2019, 32, 227–237. [Google Scholar] [CrossRef]
- Chen, X.H.; Ma, L.H.; Dong, Y.W.; Song, H.; Pu, Y.; Zhou, Q.Y. Evaluation of the differences in phenolic compounds and antioxidant activities of five green asparagus (Asparagus officinalis L.) cultivars. Qual. Assur. Saf. Crop. Food 2017, 9, 479–487. [Google Scholar] [CrossRef]
- de Moraes Crizel, T.; Hermes, V.S.; de Oliveira Rios, A.; Flôres, S.H. Evaluation of bioactive compounds, chemical and technological properties of fruits by-products powder. J. Food Sci. Technol. 2016, 53, 4067–4075. [Google Scholar] [CrossRef]
- Avram, A.M.; Morin, P.; Brownmiller, C.; Howard, L.R.; Sengupta, A.; Wickramasinghe, S.R. Concentrations of polyphenols from blueberry pomace extract using nanofiltration. Food Bioprod. Process. 2017, 106, 91–101. [Google Scholar] [CrossRef]
- M’hiri, N.; Ioannou, I.; Ghoul, M.; Boudhrioua, N.M. Phytochemical characteristics of Citrus peel and effect of conventional and nonconventional processing on phenolic compounds: A review. Food Rev. Int. 2016, 33, 587–619. [Google Scholar] [CrossRef]
- Singh, B.; Singh, J.P.; Kaur, A.; Singh, N. Phenolic composition, antioxidant potential and health benefits of citrus peel. Food Res. Int. 2020, 132, 109114. [Google Scholar] [CrossRef] [PubMed]
- Kurek, M.; Hlupić, L.; Garofulić, I.E.; Descours, E.; Ščetar, M.; Galić, K. Comparison of protective supports and antioxidative capacity of two biobased films with revalorised fruit pomaces extracted from blueberry and red grape skin. Food Packag. Shelf Life 2019, 20, 100315. [Google Scholar] [CrossRef]
- Shahbazi, Y. The properties of chitosan and gelatin films incorporated with ethanolic red grape seed extract and Ziziphora clinopodioides essential oil as biodegradable materials for active food packaging. Int. J. Biol. Macromol. 2017, 99, 746–753. [Google Scholar] [CrossRef]
- Ferreira, A.S.; Nunes, C.; Castro, A.; Ferreira, P.; Coimbra, M.A. Influence of grape pomace extract incorporation on chitosan films properties. Carbohydr. Polym. 2014, 113, 490–499. [Google Scholar] [CrossRef]
- Peixoto, C.M.; Dias, M.I.; Alves, M.J.; Calhelha, R.C.; Barros, L.; Pinho, S.P.; Ferreira, I.C.F.R. Grape pomace as a source of phenolic compounds and diverse bioactive properties. Food Chem. 2018, 253, 132–138. [Google Scholar] [CrossRef]
- Pasini, F.; Chinnici, F.; Caboni, M.F.; Verardo, V. Recovery of oligomeric proanthocyanidins and other phenolic compounds with established bioactivity from grape seed by-products. Molecules 2019, 24, 677. [Google Scholar] [CrossRef] [PubMed]
- Melo, P.E.F.; Silva, A.P.M.; Marques, F.P.; Ribeiro, P.R.V.; Souza Filho, M.M.; Brito, E.S.; Lima, J.R.; Azeredo, H.M.C. Antioxidant films from mango kernel components. Food Hydrocoll. 2019, 95, 487–495. [Google Scholar] [CrossRef]
- Adilah, Z.A.M.; Jamilah, B.; Nur Hanani, Z.A. Functional and antioxidant properties of protein-based films incorporated with mango kernel extract for active packaging. Food Hydrocoll. 2018, 74, 207–218. [Google Scholar] [CrossRef]
- Adilah, Z.A.M.; Hanani, Z.A.N. Storage stability of soy protein isolate films incorporated with mango kernel extract at different temperature. Food Hydrocoll. 2019, 87, 541–549. [Google Scholar] [CrossRef]
- Fernández-Ponce, M.T.; Casas, L.; Mantell, C.; de la Ossa, E.M. Use of high pressure techniques to produce Mangifera indica L. leaf extracts enriched in potent antioxidant phenolic compounds. Innov. Food Sci. Emerg. Technol. 2015, 29, 94–106. [Google Scholar] [CrossRef]
- Rambabu, K.; Bharath, G.; Banat, F.; Show, P.L.; Cocoletzi, H.H. Mango leaf extract incorporated chitosan antioxidant film for active food packaging. Int. J. Biol. Macromol. 2019, 126, 1234–1243. [Google Scholar] [CrossRef]
- de Moraes Crizel, T.; de Oliveira Rios, A.; Alves, V.D.; Bandarra, N.; Moldão-Martins, M.; Flôres, S.H. Active food packaging prepared with chitosan and olive pomace. Food Hydrocoll. 2018, 74, 139–150. [Google Scholar] [CrossRef]
- Moudache, M.; Colon, M.; Nerín, C.; Zaidi, F. Phenolic content and antioxidant activity of olive by-products and antioxidant film containing olive leaf extract. Food Chem. 2016, 212, 521–527. [Google Scholar] [CrossRef]
- Khalifa, I.; Barakat, H.; El-Mansy, H.A.; Soliman, S.A. Effect of chitosan–olive oil processing residues coatings on keeping quality of cold-storage strawberry (Fragaria ananassa. Var. Festival). J. Food Qual. 2016, 39, 504–515. [Google Scholar] [CrossRef]
- Khalifa, I.; Barakat, H.; El-Mansy, H.A.; Soliman, A. Preserving apple (Malus domestica var. Anna) fruit bioactive substances using olive wastes extract-chitosan film coating. Inf. Process. Agric. 2017, 4, 90–99. [Google Scholar] [CrossRef]
- Licciardello, F.; Wittenauer, J.; Saengerlaub, S.; Reinelt, M.; Stramm, C. Rapid assessment of the effectiveness of antioxidant active packaging—Study with grape pomace and olive leaf extracts. Food Packag. Shelf Life 2015, 6, 1–6. [Google Scholar] [CrossRef]
- Andrade, M.A.; Lima, V.; Sanches Silva, A.; Vilarinho, F.; Castilho, M.C.; Khwaldia, K.; Ramos, F. Pomegranate and grape by-products and their active compounds: Are they a valuable source for food applications? Trends Food Sci. Technol. 2019, 86, 68–84. [Google Scholar] [CrossRef]
- Hanani, Z.A.N.; Husna, A.B.A.; Syahida, S.N.; Nor-Khaizura, M.A.B.; Jamilah, B. Effect of different fruit peels on the functional properties of gelatin/polyethylene bilayer films for active packaging. Food Packag. Shelf Life 2018, 18, 201–211. [Google Scholar] [CrossRef]
- Hanani, Z.A.N.; Yee, F.C.; Nor-Khaizura, M.A.R. Effect of pomegranate (Punica granatum L.) peel powder on the antioxidant and antimicrobial properties of fish gelatin films as active packaging. Food Hydrocoll. 2019, 89, 253–259. [Google Scholar] [CrossRef]
- Singh, B.; Singh, J.P.; Kaur, A.; Singh, N. Phenolic compounds as beneficial phytochemicals in pomegranate (Punica granatum L.) peel: A review. Food Chem. 2018, 261, 75–86. [Google Scholar] [CrossRef] [PubMed]
- Pereira de Abreu, D.A.; Villalba Rodriguez, K.; Cruz, J.M. Extraction, purification and characterization of an antioxidant extract from barley husks and development of an antioxidant active film for food package. Innov. Food Sci. Emerg. Technol. 2012, 13, 134–141. [Google Scholar] [CrossRef]
- Hajji, T.; Mansouri, S.; Vecino-Bello, X.; Cruz-Freire, J.M.; Rezgui, S.; Ferchichi, A. Identification and characterization of phenolic compounds extracted from barley husks by LC-MS and antioxidant activity in vitro. J. Cereal Sci. 2018, 81, 83–90. [Google Scholar] [CrossRef]
- Nile, S.H.; Nile, A.S.; Keum, Y.S.; Sharma, K. Utilization of quercetin and quercetin glycosides from onion (Allium cepa L.) solid waste as an antioxidant, urease and xanthine oxidase inhibitors. Food Chem. 2017, 235, 119–126. [Google Scholar] [CrossRef]
- Campone, L.; Celano, R.; Piccinelli, A.L.; Pagano, I.; Carabetta, S.; Di Sanzo, R.; Russo, M.; Ibaṅez, E.; Cifuentes, A.; Rastrelli, L. Response surface methodology to optimize supercritical carbon dioxide/co-solvent extraction of brown onion skin by-product as source of nutraceutical compounds. Food Chem. 2018, 269, 495–502. [Google Scholar] [CrossRef]
- Serrano-León, J.S.; Bergamaschi, K.B.; Yoshida, C.M.P.; Saldańa, E.; Selani, M.M.; Rios-Mera, J.D.; Alencar, S.M.; Contreras-Castillo, C.J. Chitosan active films containing agro-industrial residue extracts for shelf life extension of chicken restructured product. Food Res. Int. 2018, 108, 93–100. [Google Scholar] [CrossRef]
- Adhikari, B.; Dhungana, S.K.; Ali, M.W.; Adhikari, A.; Kim, I.-D.; Shin, D.-H. Antioxidant activities, polyphenol, flavonoid, and amino acid contents in peanut shell. J. Saudi Soc. Agric. Sci. 2019, 18, 437–442. [Google Scholar] [CrossRef]
- Silva-Beltrán, N.P.; Chaidez-Quiroz, C.; López-Cuevas, O.; Ruiz-Cruz, S.; López-Mata, M.; Del-Toro-Sáncez, C.L.; Marquez-Rios, E.; De Jesús Ornelas-Paz, J. Phenolic compounds of potato peel extracts: Their antioxidant activity and protection against human enteric viruses. J. Microbiol. Biotechnol. 2017, 27, 234–241. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Saldaña, M.D.A. Use of potato by-products and gallic acid for development of bioactive film packaging by subcritical water technology. J. Supercrit. Fluids 2019, 143, 97–106. [Google Scholar] [CrossRef]
- Ramesh, S.; Radhakrishnan, P. Cellulose nanoparticles from agro-industrial waste for the development of active packaging. Appl. Surf. Sci. 2019, 484, 1274–1281. [Google Scholar] [CrossRef]
- Nour, V.; Panaite, T.D.; Ropota, M.; Turcu, R.; Trandafir, I.; Corbu, A.R. Nutritional and bioactive compounds in dried tomato processing waste. CyTA J. Food 2018, 16, 222–229. [Google Scholar] [CrossRef]
- Szabo, K.; Dulf, F.V.; Diaconeasa, Z.; Vodnar, D.C. Antimicrobial and antioxidant properties of tomato processing by-products and their correlation with the biochemical composition. LWT 2019, 116, 108558. [Google Scholar] [CrossRef]
- Riaz, A.; Lei, S.; Akhtar, H.M.S.; Wan, P.; Chen, D.; Jabbar, S.; Abid, M.; Hashim, M.M.; Zeng, X. Preparation and characterization of chitosan-based antimicrobial active food packaging film incorporated with apple peel polyphenols. Int. J. Biol. Macromol. 2018, 114, 547–555. [Google Scholar] [CrossRef] [PubMed]
- Choi, I.; Chang, Y.; Shin, S.-H.; Joo, E. Development of biopolymer composite films using a microfluidization technique for carboxymethylcellulose and apple skin particles. Int. J. Mol. Sci. 2017, 18, 1278. [Google Scholar] [CrossRef]
- Priyadarshi, R.; Sauraj; Kumar, B.; Deeba, F.; Kulshreshtha, A.; Negi, Y.S. Chitosan films incorporated with Apricot (Prunus armeniaca) kernel essential oil as active food packaging material. Food Hydrocoll. 2018, 85, 158–166. [Google Scholar] [CrossRef]
- Yang, G.; Yue, J.; Gong, X.; Qian, B.; Wang, H.; Deng, Y.; Zhao, Y. Blueberry leaf extracts incorporated chitosan coatings for preserving postharvest quality of fresh blueberries. Postharvest Biol. Technol. 2014, 92, 46–53. [Google Scholar] [CrossRef]
- Sogut, E.; Seydim, A.C. The effects of Chitosan and grape seed extract-based edible films on the T quality of vacuum packaged chicken breast fillets. Food Packag. Shelf Life 2018, 18, 13–20. [Google Scholar] [CrossRef]
- Alves, V.L.C.D.; Rico, B.P.M.; Cruz, R.M.S.; Vicente, A.A.; Khmelinskii, I.; Vieira, M.C. Preparation and characterization of a chitosan film with grape seed extract- carvacrol microcapsules and its effect on the shelf-life of refrigerated Salmon (Salmo salar). LWT Food Sci. Technol. 2018, 89, 525–534. [Google Scholar] [CrossRef]
- Shankar, S.; Rhim, J.-W. Antimicrobial wrapping paper coated with a ternary blend of carbohydrates (alginate, carboxymethyl cellulose, carrageenan) and grapefruit seed extract. Carbohydr. Polym. 2018, 196, 92–101. [Google Scholar] [CrossRef] [PubMed]
- Kanmani, P.; Rhim, J.-W. Development and characterization of carrageenan/grapefruit seed extract composite films for active packaging. Int. J. Biol. Macromol. 2014, 68, 258–266. [Google Scholar] [CrossRef]
- Kanmani, P.; Rhim, J.-W. Antimicrobial and physical-mechanical properties of agar-based films incorporated with grapefruit seed extract. Carbohydr. Polym. 2014, 102, 708–716. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-H.; Hong, W.-S.; Oh, S.-W. Effect of layer-by-layer antimicrobial edible coating of alginate and chitosan with grapefruit seed extract for shelf-life extension of shrimp (Litopenaeus vannamei) stored at 4 °C. Int. J. Biol. Macromol. 2018, 120, 1468–1473. [Google Scholar] [CrossRef]
- Tan, Y.M.; Lim, S.H.; Tay, B.Y.; Lee, M.W.; Thian, E.S. Functional chitosan-based grapefruit seed extract composite films for applications in food packaging technology. Mater. Res. Bull. 2015, 69, 142–146. [Google Scholar] [CrossRef]
- Wang, L.-F.; Rhim, J.-W. Grapefruit seed extract incorporated antimicrobial LDPE and PLA films: Effect of type of polymer matrix. LWT Food Sci. Technol. 2016, 74, 338–345. [Google Scholar] [CrossRef]
- Khalifa, I.; Barakat, H.; El-Mansy, H.A.; Soliman, S.A. Improving the shelf-life stability of apple and strawberry fruits applying chitosan-incorporated olive oil processing residues coating. Food Packag. Shelf Life 2016, 9, 10–19. [Google Scholar] [CrossRef]
- Emam-Djomeh, Z.; Moghaddam, A.; Ardakani, S.A.Y. Antimicrobial activity of pomegranate (Punica granatum L.) peel extract, physical, mechanical, barrier and antimicrobial properties of pomegranate peel extract-incorporated sodium caseinate film and application in packaging for ground beef. Packag. Technol. Sci. 2015, 28, 869–881. [Google Scholar] [CrossRef]
- Yuan, G.; Lv, H.; Tang, W.; Zhang, X.; Sun, H. Effect of chitosan coating combined with pomegranate peel extract on the quality of Pacific white shrimp during iced storage. Food Control 2016, 59, 818–823. [Google Scholar] [CrossRef]
- Licciardello, F.; Kharchoufi, S.; Muratore, G.; Restuccia, C. Effect of edible coating combined with pomegranate peel extract on the quality maintenance of white shrimps (Parapenaeus longirostris) during refrigerated storage. Food Packag. Shelf Life 2018, 17, 14–119. [Google Scholar] [CrossRef]
- Kharchoufi, S.; Parafati, L.; Licciardello, F.; Muratore, G.; Hamdi, M.; Cirvilleri, G.; Restuccia, C. Edible coatings incorporating pomegranate peel extract and biocontrol yeast to reduce Penicillium digitatum postharvest decay of oranges. Food Microbiol. 2018, 74, 107–112. [Google Scholar] [CrossRef]
- Mushtaq, M.; Gani, A.; Gani, A.; Punoo, H.A.; Masoodi, F.A. Use of pomegranate peel extract incorporated zein film with improved properties for prolonged shelf life of fresh Himalayan cheese (Kalari/kradi). Innov. Food Sci. Emerg. Technol. 2018, 48, 25–32. [Google Scholar] [CrossRef]
- Ali, A.; Chen, Y.; Liu, H.; Yu, L.; Baloch, Z.; Khalid, S.; Zhu, J.; Chen, L. Starch-based antimicrobial films functionalized by pomegranate peel. Int. J. Biol. Macromol. 2019, 129, 1120–1126. [Google Scholar] [CrossRef] [PubMed]
- Kasaai, M.R.; Moosavi, A. Treatment of Kraft paper with citrus wastes for food packaging applications: Water and oxygen barrier properties improvement. Food Packag. Shelf Life 2017, 12, 59–65. [Google Scholar] [CrossRef]
- Munir, S.; Hu, Y.; Liu, Y.; Xiong, S. Enhanced properties of silver carp surimi-based edible films incorporated with pomegranate peel and grape seed extracts under acidic condition. Food Packag. Shelf Life 2019, 19, 114–120. [Google Scholar] [CrossRef]
- Torres-León, C.; Vicente, A.A.; Flores-López, M.L.; Rojas, R.; Serna-Cock, L.; Alvarez-Pérez, O.B.; Aguilar, C.N. Edible films and coatings based on mango (var. Ataulfo) by-products to improve gas transfer rate of peach. LWT Food Sci. Technol. 2018, 97, 624–631. [Google Scholar] [CrossRef]
- Nor Adilah, A.; Jamilah, B.; Noranizan, M.A.; Nur Hanani, Z.A. Utilization of mango peel extracts on the biodegradable films for active packaging. Food Packag. Shelf Life 2018, 16, 1–7. [Google Scholar] [CrossRef]
- Nawab, A.; Alam, F.; Haq, M.A.; Lutfi, Z.; Hasnain, A. Mango kernel starch-gum composite films: Physical, mechanical and barrier properties. Int. J. Biol. Macromol. 2017, 98, 869–876. [Google Scholar] [CrossRef]
- Fontes-Candia, C.; Erboz, E.; Martínez-Abad, A.; López-Rubio, A.; Martínez-Sanz, M. Superabsorbent food packaging bioactive cellulose-based aerogels from Arundo donax waste biomass. Food Hydrocoll. 2019, 96, 151–160. [Google Scholar] [CrossRef]
- Benito-González, I.; López-Rubio, A.; Martínez-Sanz, M. High-performance starch biocomposites with celullose from waste biomass: Film properties and retrogradation behavior. Carbohydr. Polym. 2019, 216, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Niu, X.; Yang, J.; Fan, R.; Shi, J.; Ullah, N.; Feng, X.; Chen, L. Active biodegradable films based on the whole potato peel incorporated with bacterial cellulose and curcumin. Int. J. Biol. Macromol. 2020, 150, 480–491. [Google Scholar] [CrossRef] [PubMed]
- Sugumaran, V.; Kapur, G.S.; Narula, A.K. Sustainable potato peel powder–LLDPE biocomposite preparation and effect of maleic anhydride-grafted polyolefins on their properties. Polym. Bull. 2018, 75, 5513–5533. [Google Scholar] [CrossRef]
- Borah, P.P.; Das, P.; Badwaik, L.S. Ultrasound treated potato peel and sweet lime pomace based biopolymer film development. Ultrason. Sonochem. 2017, 36, 11–19. [Google Scholar] [CrossRef]
- González, A.; Gastelú, G.; Barrera, G.N.; Ribotta, P.D.; Igarzabal, C.I.Á. Preparation and characterization of soy protein films reinforced with cellulose nanofibers obtained from soybean by-products. Food Hydrocoll. 2019, 89, 758–764. [Google Scholar] [CrossRef]


| Products and By-Products Percentage | Main Phenolic Compounds | References | |
|---|---|---|---|
| Apple (25–30% solid waste) | Pomace | Hydroxycinnamates, phloretin glycosides, quercetin glycosides, catechins, procyanidins, epicatechin, chlorogenic acid, cryptochlorogenic acid. | [39,40,41] |
| Artichoke (66% solid waste) | Bracts, leaves and stems | Chlorogenic acid, luteolin-7-O-rutinoside, luteolin-7-O-glucoside and apigenin. | [42,43] |
| Asparagus (40–50% solid waste) | Peel | Dietary fiber, rutin, peroxidases, phenols, flavonoids, hydroxycinnamic acids and saponins. | [44,45] |
| Blueberry (20–30% solid waste) | Pomace | Anthocyanins, cinnamic acid derivatives and flavonol-glycosides. | [46,47] |
| Citrus (50% solid waste) | Peel | Eriocitrin, hesperidin, naringin. | [48,49] |
| Grape (20% solid waste) | Skin | Catechins, epicatechins, epigallocatechin, picatechin gallate. | [50,51,52,53,54] |
| Pomace | Catechins, anthocyanins, stilbenes, flavonol glycosides. | ||
| Seeds | Procyanidins. | ||
| Mango (45% solid waste) | Kernel and leaves | Gallates, gallotannins, gallic acid, ellagic acid, glucosides. | [55,56,57,58,59] |
| Olive (30–50% liquid waste) | Pomace, peel and vegetation water | Myricetin, ferulic, sinapic caffeic, gallic, ellagic, oleuropein and hydroxytyrosol derivatives. | [60,61,62,63,64] |
| Pomegranate (49% solid waste) | Peel and pomace | Penduncalin, punicalagin, caffeic acid, chlorogenic acid, ellagic acid, apigenin, quercitin and gallic acid. | [65,66,67,68] |
| Husk (15–20% solid waste) | Barley husk | Vanillin, gallic acid, p-coumaric acid, p-hydroxybenzaldehyde, ferulic acid, syringic acid, p-hydroxibenzoic acid, vanillic acid and acetovanillon. | [69,70] |
| Onion (17% solid waste) | Skin | Quercetin 3,40-O-diglucoside and quercetin 40-O-monoglucoside and isorhamnetin-3-glucoside. | [71,72] |
| Peanut (35–40% solid waste) | Shell | Proanthocyanidins and procyanidins. | [73,74] |
| Potato (15% solid waste) | Peel | Chlorogenic acid, ferulic, gallic, protocatechuic and caffeic acid. | [75,76,77] |
| Tomato (20% solid waste) | Skin and seeds | Lycopene; Caffeic acid-glucoside, isomer I and isomer II, 3-caffeoylquinic acid, 5-caffeoylquinic acid, quercetin-triglucoside, quercetin-3-rutinoside, 3,4-di-O-caffeoylquinic acid, 3,4,5-tri-caffeoylquinic acid, naringenin chalcone and naringenin. | [78,79] |
| By-Products | Active Packaging | Target Microorganisms | References |
|---|---|---|---|
| Apple skin | Chitosan edible film | E. coli, S. enterica and L. monocytogenes | [80] |
| Apple skin (powder, extract) | Composite films ASP/CMC | L. monocytogenes, S. aureus, S. enterica and S. flexner | [81] |
| Apricot kernel extract | Chitosan film | E. coli and Bacillus subtilis | [82] |
| Blueberry leaf extract | Chitosan coating | S. aureus, L. monocytogenes, S. typhimurium, E. coli and fungi | [83] |
| Grape seed extract | Chitosan film | E. coli, L. monocytogenes, S. aureus and P. aeruginosa | [84] |
| Chitosan film and carvacrol | Mesophilic and psychrophilic bacteria and Pseudomonas spp | [85] | |
| Grapefruit seed extract | Coated wrapping paper | L. monocytogenes and E. coli | [86] |
| Biopolymer carrageenan | Gram-positive and Gram-negative food-borne pathogens | [87,88] | |
| Layer-by-layer coating with alginate, chitosan | Mesophilic and psychrotrophic bacteria | [89] | |
| Chitosan films | Fungi | [90] | |
| LDPE and PLA | E. coli and L. monocytogenes | [91] | |
| Olive oil leaves and olive pomace | Chitosan film | R. stolonifer, P. expansum | [62,92] |
| Pomegranate peel extract | Casein-based film | S. aureus, E. coli | [93] |
| Chitosan coating | Total aerobic bacteria, Pseudomonas spp., P. digitatum | [94,95,96] | |
| Zein-based film | E. coli, P. perfringens, M. luteus, E. faecalis, S. aureus, P. vulgaris and S. typhii | [97] | |
| Pomegranate peel powder | Fish Gelatin film | S. aureus, L. monocytogenes and E. coli | [67] |
| Starch-based film | S. aureus, Salmonella | [98] |
| By-Products | Packaging System | Physical and Mechanical Properties | References |
|---|---|---|---|
| Apricot kernel oil (AKo) | Chitosan film with AKo (1:0, 1:0.125, 1:0.25, 1:0.5 and 1:1 w/v). | Essential oil improved TS* and WVB**, and reduced film solubility (from 18.42 to 4.76%). | [82] |
| Blueberry waste (BW) | Cassava starch film with BW powder (4, 8 and 12 wt%). | BW decreased SI** (pH 2.5, 7.0 and 10.0) and promoted UV protection. | [36] |
| Citrus peel and leaves | Kraft paper + peel:leaf extract (2:0, 2:1, 3:0). | Peel:leaf extract (2:1) increased WVB** and O2B**. | [99] |
| Grape seed (GSE) | Chitosan and gelatin films with GSE (1% v/w) and Ziziphora clinopodioides essential oil (ZEO). | 1% GSE + 1% ZEO decreased TS*, PF*, PD* and SI**; increased WVB**. | [51] |
| Grape seed (GSE) + Pomegranate peel (PPE) | Surimi edible films with GSE + PPE (0%, 2%, 4% and 6%). | 6% PPE improved TS*; 6% GSE increased WVB** and both reduced light transmission. | [100] |
| Mango peel and kernel (MKE) | Edible mango peel coating with MKE (0.078 g/L). | MKE reduced WVB** and film solubility (from 60.24 to 52.56%). | [101] |
| Mango peel extract (MPE) | Fish gelatin film with MPE (1%, 3% and 5%). | MPE improved TS* (from 7.65 to 15.78 MPa) and reduced solubility from 40% to 20%. | [102] |
| Mango kernel starch | Composite film (kernel starch and guar/xanthan gum 10%, 20% and 30%). | The different % of gums increased TS and O2B**, but decreased the film solubility and WVB*. | [103] |
| Pomegranate peel extract (PPE) | Zein film with PPE (0, 25, 50, and 75 mg/mL of film forming solution). | PPE improved TS* and WVB**, increased film solubility from 6.166% (control) to 18.29% (75 mg PPE). | [97] |
| By-Products | Packaging System | Physical and Mechanical Properties | References |
|---|---|---|---|
| Arundo donax | Cellulose-based aerogels | Porous aerogel has great adsorption capacity. | [104] |
| Posidonia oceanica | Corn starch films with cellulose fillers | The stronger interaction between starch and cellulose improved TS* and WVB**. | [105] |
| Potato peel (PP) | PP film + Bacterial cellulose (BC) + curcumin | BC-10% improved TS*, WVB** and O2B**, while BC-15% reduced O2B** and WVB**. | [106] |
| Cellulose nanoparticles in chitosan and/or PVA | Nanoparticles improved the flexibility, elasticity and O2B** of films. | [77] | |
| PP as filler (0–40%) in LLDPE | PP from 10% to 40% increased the water absorption and reduced TS* compared to the control. | [107] | |
| Potato peel (PP) and cull | PP:cull ratio (g/g) (0:1, 0.5:1, 1:1, 1.3:1) | The ratio 1.3:1 improved film TS* and WVB** due to high fiber content of PP compared to cull. | [76] |
| Potato peel (PP) and sweet lime pomace (SLP) | Biopolymer films with PP and SLP | US-treatment improved WVB both in PP and in SLP films and reduced solubility. | [108] |
| Soybean hulls and pods | Cellulose nanofibers in soy protein films | Nano-fibers improved TS* and SI** of films. | [109] |
| Sugarcane bagasse (SB) and asparagus peel (AP) | Trays based on potato starch and fibers from SB or AP | Fibers improved thermal stability; SB make the trays less porous with higher TS* and lower SI** compared to control trays. | [44] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dilucia, F.; Lacivita, V.; Conte, A.; Del Nobile, M.A. Sustainable Use of Fruit and Vegetable By-Products to Enhance Food Packaging Performance. Foods 2020, 9, 857. https://doi.org/10.3390/foods9070857
Dilucia F, Lacivita V, Conte A, Del Nobile MA. Sustainable Use of Fruit and Vegetable By-Products to Enhance Food Packaging Performance. Foods. 2020; 9(7):857. https://doi.org/10.3390/foods9070857
Chicago/Turabian StyleDilucia, Flavia, Valentina Lacivita, Amalia Conte, and Matteo A. Del Nobile. 2020. "Sustainable Use of Fruit and Vegetable By-Products to Enhance Food Packaging Performance" Foods 9, no. 7: 857. https://doi.org/10.3390/foods9070857
APA StyleDilucia, F., Lacivita, V., Conte, A., & Del Nobile, M. A. (2020). Sustainable Use of Fruit and Vegetable By-Products to Enhance Food Packaging Performance. Foods, 9(7), 857. https://doi.org/10.3390/foods9070857