Current Knowledge on Interspecific Hybrid Palm Oils as Food and Food Ingredient
Abstract
1. Introduction
2. Chemical Composition of HPO
2.1. Fatty Acids
2.2. Acylglycerols
2.2.1. Triacylglycerols
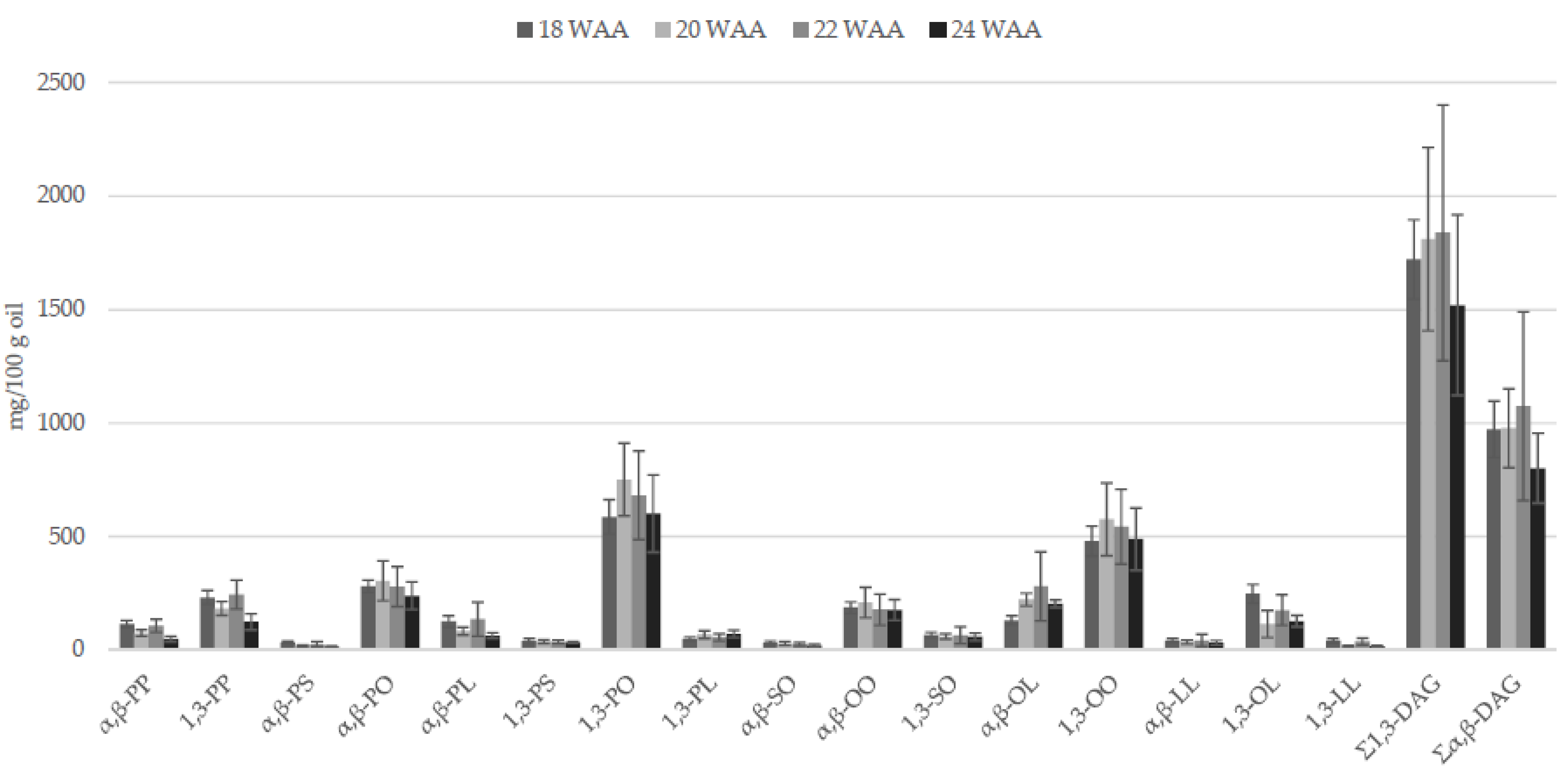
2.2.2. Partial Glycerides
2.3. Unsaponifiable Matter
3. HPO as Food and Food Ingredient
3.1. Quality Parameters
3.2. Food Uses
3.3. Nutritional Properties
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Montoya, C.; Cochard, B.; Flori, A.; Cros, D.; Lopes, R.; Cuellar, T.; Espéout, S.; Syaputra, I.; Villeneuve, P.; Pina, M.; et al. Genetic Architecture of Palm Oil Fatty Acid Composition in Cultivated Oil Palm (Elaeis guineensis Jacq.) Compared to Its Wild Relative E. oleifera (H.B.K) Cortés. PLoS ONE 2014, 9, e95412. [Google Scholar] [CrossRef]
- Astorkia, M.; Hernández, M.; Bocs, S.; Ponce, K.; León, O.; Morales, S.; Quezada, N.; Orellana, F.; Wendra, F.; Sembiring, Z.; et al. Analysis of the allelic variation in the Shell gene homolog of E. oleifera and design of species specific Shell primers. Euphytica 2019, 216, 5. [Google Scholar] [CrossRef]
- Statista—Production Volume of Palm Oil Worldwide from 2012/13 to 2019/20. Available online: https://www.statista.com/statistics/613471/palm-oil-production-volume-worldwide (accessed on 26 March 2020).
- Mba, O.I.; Dumont, M.-J.; Ngadi, M. Palm oil: Processing, characterization and utilization in the food industry—A review. Food Biosci. 2015, 10, 26–41. [Google Scholar] [CrossRef]
- Rudzińska, M.; Hassanein, M.M.M.; Abdel-Razek, A.G.; Kmiecik, D.; Siger, A.; Ratusz, K. Influence of composition on degradation during repeated deep-fat frying of binary and ternary blends of palm, sunflower and soybean oils with health-optimised saturated-to-unsaturated fatty acid ratios. Int. J. Food Sci. Technol. 2017, 53, 1021–1029. [Google Scholar] [CrossRef]
- Mozzon, M.; Pacetti, D.; Lucci, P.; Balzano, M.; Frega, N.G. Crude palm oil from interspecific hybrid Elaeis oleifera × Elaeis guineensis: Fatty acid regiodistribution and molecular species of glycerides. Food Chem. 2013, 141, 245–252. [Google Scholar] [CrossRef]
- Mancini, A.; Imperlini, E.; Nigro, E.; Montagnese, C.; Daniele, A.; Orrù, S.; Buono, P. Biological and Nutritional Properties of Palm Oil and Palmitic Acid: Effects on Health. Molecules 2015, 20, 17339–17361. [Google Scholar] [CrossRef]
- Lieb, V.M.; Kerfers, M.R.; Kronmüller, A.; Esquivel, P.; Alvarado, A.; Jiménez, V.M.; Schmarr, H.-G.; Carle, R.; Schweiggert, R.; Steingass, C.B. Characterization of Mesocarp and Kernel Lipids from Elaeis guineensis Jacq., Elaeis oleifera [Kunth] Cortés, and Their Interspecific Hybrids. J. Agric. Food Chem. 2017, 65, 3617–3626. [Google Scholar] [CrossRef]
- Tan, B.K.; Ong, S.H.; Rajanaidu, N.; Rao, V. Biological modification of oil composition. J. Am. Oil Chem. Soc. 1985, 62, 230–236. [Google Scholar] [CrossRef]
- Ting, N.; Yaakub, Z.; Kamaruddin, K.; Mayes, S.; Massawe, F.; Sambanthamurthi, R.; Jansen, J.; Low, E.-T.L.; Ithnin, M.; Kushairi, A.; et al. Fine-mapping and cross-validation of QTLs linked to fatty acid composition in multiple independent interspecific crosses of oil palm. BMC Genom. 2016, 17, 289. [Google Scholar] [CrossRef]
- Tan, K.; Lee, K.T.; Mohamed, A.R.; Bhatia, S. Palm oil: Addressing issues and towards sustainable development. Renew. Sustain. Energy Rev. 2009, 13, 420–427. [Google Scholar] [CrossRef]
- Fitzherbert, E.; Struebig, M.J.; Morel, A.; Danielsen, F.; Brühl, C.A.; Donald, P.; Phalan, B. How will oil palm expansion affect biodiversity? Trends Ecol. Evol. 2008, 23, 538–545. [Google Scholar] [CrossRef] [PubMed]
- Arias, D.; González, M.; Prada, F.; Ayala-Diaz, I.; Montoya, C.; Daza, E.; Romero, H.M. Genetic and phenotypic diversity of natural American oil palm (Elaeis oleifera (H.B.K.) Cortés) accessions. Tree Genet. Genomes 2015, 11, 22. [Google Scholar] [CrossRef]
- Hardon, J.J. Interspecific hybrids in the genus Elaeis II. vegetative growth and yield of F1 hybrids E. guineensis × E. oleifera. Euphytica 1969, 18, 380–388. [Google Scholar] [CrossRef]
- Camillo, J.; Leão, A.P.; Alves, A.A.; Formighieri, E.F.; Azevedo, A.L.; Nunes, J.D.; De Capdeville, G.; De A Mattos, J.K.; Souza, M.T. Reassessment of the Genome Size in Elaeis guineensis and Elaeis oleifera, and Its Interspecific Hybrid. Genom. Insights 2014, 7, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Macfarlane, N.; Swetman, T.; Cornelius, J.A. Analysis of mesocarp and kernel oils from the American oil palm and F1 hybrids with the West African oil palm. J. Sci. Food Agric. 1975, 26, 1293–1298. [Google Scholar] [CrossRef]
- Rincón, S.M.; Hormaza, P.A.; Moreno, L.P.; Prada, F.; Portillo, D.J.; García, J.A.; Romero, H.M. Use of phenological stages of the fruits and physicochemical characteristics of the oil to determine the optimal harvest time of oil palm interspecific O × G hybrid fruits. Ind. Crop. Prod. 2013, 49, 204–210. [Google Scholar] [CrossRef]
- Ávila-Méndez, K.; Avila-Diazgranados, R.; Pardo, A.; Herrera, M.; Sarria, G.; Romero, H.M. Response of in vitro obtained oil palm and interspecific OxG hybrids to inoculation with Phytophthora palmivora. For. Pathol. 2019, 49, e12486. [Google Scholar] [CrossRef]
- Hardon, J.J.; Tan, G.Y. Interspecific hybrids in the genus Elaeis I. crossability, cytogenetics and fertility of F1 hybrids of E. guineensis x E. oleifera. Euphytica 1969, 18, 372–379. [Google Scholar] [CrossRef]
- Astorkia, M.; Hernandez, M.; Bocs, S.; Ponce, K.; León, O.; Morales, S.; Quezada, N.; Orellana, F.; Wendra, F.; Sembiring, Z.; et al. Detection of significant SNP associated with production and oil quality traits in interspecific oil palm hybrids using RARSeq. Plant Sci. 2019, 291, 110366. [Google Scholar] [CrossRef]
- Osorio-Guarín, J.A.; Garzón-Martínez, G.A.; Delgadillo-Duran, P.; Bastidas, S.; Moreno, L.P.; Enciso-Rodríguez, F.E.; Cornejo, O.E.; Barrero, L.S. Genome-wide association study (GWAS) for morphological and yield-related traits in an oil palm hybrid (Elaeis oleifera × Elaeis guineensis) population. BMC Plant Boil. 2019, 19, 533–611. [Google Scholar] [CrossRef]
- Astorkia, M.; Hernandez, M.; Bocs, A.; Herran, A.; Ponce, K.; León, O.; Morales, S.; Quezada, N.; Orellana, F. Association Mapping Between Candidate Gene SNP and Production and Oil Quality Traits in Interspecific Oil Palm Hybrids. Plants 2019, 8, 377. [Google Scholar] [CrossRef]
- Zulkifli, Y.; Maizura, I.; Rajinder, S.; Marhalil, M.; Mohd Din, A.; Ong-Abdullah, M. An RFLP marker, CB75A, predicts for palmitic acid content across selected oil palm germplasm and interspecific-hybrids. J. Oil Palm Res. 2019, 31, 315–320. [Google Scholar] [CrossRef]
- Junior, R.A.G.; Lopes, R.; Da Cunha, R.N.V.; Pina, A.J.D.A.; Quaresma, C.E.; Santos, R.R.; De Resende, M.D.V. Bunch yield of interspecific hybrids of American oil palm with oil palm in the juvenile phase. Crop. Breed. Appl. Biotechnol. 2016, 16, 86–94. [Google Scholar] [CrossRef]
- Gomes, R.A., Jr.; Gurgel, F.D.L.; Peixoto, L.D.A.; Bhering, L.L.; Da Cunha, R.N.V.; Lopes, R.; Pina, A.J.D.A.; Veiga, A.S. Evaluation of interspecific hybrids of palm oil reveals great genetic variability and potential selection gain. Ind. Crop. Prod. 2014, 52, 512–518. [Google Scholar] [CrossRef]
- Azni, I.N.A.M.; Sundram, S.; Ramachandran, V.; Abu Seman, I. Anin vitro investigation of Malaysian Phytophthora palmivoraisolates and pathogenicity study on oil palm. J. Phytopathol. 2017, 165, 800–812. [Google Scholar] [CrossRef]
- Alvarado, A.; Escobar, H.J. The hybrid O×G Amazon: An alternative for regions affected by bud rot in oil palm. Palmas 2013, 34, 305–314. [Google Scholar]
- De Franqueville, H. Oil palm bud rot in Latin America. Exp. Agric. 2003, 39, 225–240. [Google Scholar] [CrossRef]
- Amblard, P.; Noiret, J.M.; Kouame, B.; Potier, F.; Adon, B. Comparative performances of interspecific hybrids and the commercial material. OCL-Ol. Corps Gras Li. 1995, 2, 335–340. [Google Scholar]
- Lucci, P.; Pacetti, D.; Frega, N.G.; Mozzon, M. Phytonutrient concentration and unsaturation of glycerides predict optimal harvest time for Elaeis oleifera × E. guineensis palm oil hybrids. Eur. J. Lipid Sci. Technol. 2015, 117, 1027–1036. [Google Scholar] [CrossRef]
- Socha, J.; Cayón-Salinas, D.G.; Ligarreto, G.; Chaves, G. Effect of pollen doses on fruit formation and oil production in two hybrid palm genotypes (Elaeis oleifera H.B.K. Cortes × Elaeis guineensis Jacq.). Agronomía Colombiana 2019, 37, 12–17. [Google Scholar] [CrossRef]
- Escobar, H.C.; Chauza, J.J.D. Germinability and pollen viability of four improved cultivars of palm oil under laboratory conditions. Revista Facultad Nacional de Agronomía Medellín 2018, 71, 8395–8405. [Google Scholar] [CrossRef]
- Filho, E.G.A.; Brito, R.S.; Rodrigues, T.H.S.; Silva, L.M.A.; De Brito, E.S.; Canuto, K.M.; Krug, C.; Zocolo, G.J. Association of Pollinators of Different Species of Oil Palm with the Metabolic Profiling of Volatile Organic Compounds. Chem. Biodivers. 2019, 16, e1900050. [Google Scholar] [CrossRef]
- Almeida, R.F.; Santos, I.R.; Meira, F.S.; Grynberg, P.; Lopes, R.; Da Cunha, R.N.V.; Franco, O.L.; Scherwinski-Pereira, J.E.; Mehta, A. Differential protein profiles in interspecific hybrids between Elaeis oleifera and E. guineensis with contrasting responses to somatic embryogenesis competence acquisition. Plant Cell Tissue Organ Cult. (PCTOC) 2019, 137, 11–21. [Google Scholar] [CrossRef]
- Gomes, H.T.; Bartos, P.M.C.; Scherwinski-Pereira, J.E. Dynamics of morphological and anatomical changes in leaf tissues of an interspecific hybrid of oil palm during acquisition and development of somatic embryogenesis. Plant Cell Tissue Organ Cult. (PCTOC) 2017, 131, 269–282. [Google Scholar] [CrossRef]
- De Lima, W.A.A.; Lopes, R.; Green, M.; Cunha, R.N.V.; Abreu, S.C.; Cysne, A.Q. Heat treatment and germination of seeds of interspecific hybrid between American oil palm (Elaeis oleifera (H.B.K) Cortes) and African oil palm (Elaeis guineensis Jacq.). J. Seed Sci. 2014, 36, 451–457. [Google Scholar] [CrossRef]
- Angelo, P.C.D.S.; Moraes, L.A.C.; Lopes, R.; Sousa, N.R.; Da Cunha, R.N.V.; Quisen, R.C. In vitro rescue of interspecific embryos from Elaeis guineensis × E. oleifera (Arecaceae). Revista de Biología Tropical 2011, 59, 1081–1088. [Google Scholar] [CrossRef][Green Version]
- Peixoto, L.D.A.; Bhering, L.L.; Gurgel, F.D.L.; Junior, R.A.G. Parental selection for the formation of interspecific hybrid populations of oil palm. Acta Sci. Agron. 2015, 37, 155. [Google Scholar] [CrossRef]
- Hormaza, P.; Fuquen, E.M.; Romero, H.M. Phenology of the oil palm interspecific hybrid Elaeis oleifera × Elaeis guineensis. Sci. Agric. 2012, 69, 275–280. [Google Scholar] [CrossRef]
- Chia, G.S.; Lopes, M.T.G.; Lopes, R.; Da Cunha, R.N.V.; Da Rocha, R.N.C. Repetibilidade da produção de cachos de híbridos interespecíficos entre o caiaué e o dendezeiro. Acta Amaz. 2009, 39, 249–254. [Google Scholar] [CrossRef]
- Oboh, B.O.; Fakorede, M.A.B. A factor analysis of vegetative and yield traits in backcross progenies of an interspecific hybrid of oil palm. J. Genet. Breed. 1997, 51, 257–262. [Google Scholar]
- Thomas, R.L.; Ng, S.C.; Chan, K.W. Phyllotaxis in the Oil Palm: Applications in Selection of Interspecific Hybrids. Ann. Bot. 1970, 34, 1025–1035. [Google Scholar] [CrossRef]
- Ooi, S.C.; Da Silva, E.B.; Muller, A.A.; Nascimento, J.C. Oil palm genetic resources − native Elaeis oleifera populations in Brazil offer promising sources. Pesqui. Agropecu. Bras. 1981, 16, 385–395. [Google Scholar]
- Rodriguez, C.J.B.; Romero, H.M. Physiological and agronomic behavior of commercial cultivars of oil palm (Elaeis guineensis) and OxG hybrids (Elaeis oleifera × Elaeis guineensis) at rainy and dry seasons. Aust. J. Crop. Sci. 2019, 13, 424–432. [Google Scholar] [CrossRef]
- De Matos, G.S.B.; Fernandes, A.R.; Wadt, P.G.S.; Pina, A.J.D.A.; Franzini, V.I.; Ramos, H.M.N. The Use of DRIS for Nutritional Diagnosis in Oil Palm in the State of Pará. Revista Brasileira de Ciência do Solo 2017, 41, 41. [Google Scholar] [CrossRef]
- Rivera-Méndez, Y.D.; Chacón, A.L.M.; Romero, H.M. Response of the roots of oil palm O×G interspecific hybrids (Elaeis oleifera × Elaeis guineensis) to aluminum (Al3+) toxicity. Austral. J. Crop Sci. 2014, 8, 1526–1533. [Google Scholar]
- Méndez, Y.D.R.; Chacón, L.M.; Bayona, C.J.; Romero, H.M. Physiological response of oil palm interspecific hybrids (Elaeis oleifera H.B.K. Cortes versus Elaeis guineensis Jacq.) to water deficit. Braz. J. Plant Physiol. 2012, 24, 273–280. [Google Scholar] [CrossRef]
- Fooyontphanich, K.; Morcillo, F.; Amblard, P.; Collin, M.; Jantasuriyarat, C.; Verdeil, J.-L.; Tranbarger, T.J. A phenotypic test for delay of abscission and non-abscission oil palm fruit and validation by abscission marker gene expression analysis. Acta Hortic. 2016, 97–104. [Google Scholar] [CrossRef]
- Madon, M.; Arulandoo, X.; Sritharan, K.; Nordiana, H.; Muhammad Azwan, Z.; Mohd Zaki, N. Short communication: Genomic constitution of oil palm interspecific hybrid crosses monitored by genomic in situ hybridisation (gish). J. Oil Palm Res. 2018, 30, 338–344. [Google Scholar] [CrossRef]
- Madon, M.; Phoon, L.Q.; Clyde, M.M.; Mohd, D.A. Application of flow cytometry for estimation of nuclear DNA content in Elaeis. J. Oil Palm Res. 2008, 20, 447–452. [Google Scholar]
- Galindo-Castañeda, T.; Romero, H.M. Mycorrhization in oil palm (Elaeis guineensis and E. oleifera × E. guineensis) in the pre-nursery stage. Agronom. Colomb. 2013, 31, 95–102. [Google Scholar]
- Rivera Méndez, Y.D.; Moreno Chacón, A.L.; Romero, H.M. Biochemical and physiological characterization of oil palm interspecific hybrids (Elaeis oleifera × Elaeis guineensis) grown in hydroponics. Acta Biol. Colomb. 2013, 18, 465–472. [Google Scholar]
- Chaves, G.; Moreno, G.A.L.; Cayón-Salinas, D.G. Physicochemical characterization of bunches from American oil palm (Elaeis oleifera H.B.K. Cortes) and their hybrids with African oil palm (Elaeis guineensis Jacq.). Acta Agronómica 2018, 67, 170–178. [Google Scholar] [CrossRef]
- Choo, Y.M.; Yap, S. Carotenes, vitamin E and sterols in oils from Elaeis guineensis, Elaeis oleifera and their hybrids. Palm 1998, 19, 79–85. [Google Scholar]
- Ong, S.H.; Chuah, C.C.; Sow, H.P. The co-dominance theory: Genetic interpretations of analyses of mesocarp oils fromelaeis guineensis, Elaeis oleiferaand their hybrids. J. Am. Oil Chem. Soc. 1981, 58, 1032–1038. [Google Scholar] [CrossRef]
- Opute, F.I.; Obasola, C.O. Breeding for Short-stemmed Oil Palm in Nigeria: Fatty Acids, their Significance and Characteristics. Ann. Bot. 1979, 43, 677–681. [Google Scholar] [CrossRef]
- Harwood, J.L. Fatty acid biosynthesis. In Plant Lipids: Biology, Utilisation and Manipulation; Murphy, D.J., Ed.; Blackwell Publishing: Oxford, UK, 2005; pp. 27–66. [Google Scholar]
- Zulkifli, Y.; Rajinder, S.; Mohd, D.A.; Ting, N.C.; Rajanaidu, N.; Kushairi, A.; Musa, B.; Mohamad, O.; Ismanizan, I. Inheritance of SSR and SNP loci in an oil palm interspecific hybrid backcross (BC2) population. J. Oil Palm Res. 2014, 26, 203–213. [Google Scholar]
- Nordiana, H.M.N.; Ngoot-Chin, T.; Singh, R.; Clyde, M.M.; Madon, M. Evaluation of inter-simple sequence repeat (ISSR) markers for genetic mapping of an oil palm interspecific hybrid mapping population. J. Oil Palm Res. 2014, 26, 214–225. [Google Scholar]
- Singh, R.; Tan, S.; Panandam, J.M.; Rahman, R.A.; Ooi, L.C.-L.; Low, E.-T.L.; Sharma, M.; Jansen, J.; Cheah, S.-C. Mapping quantitative trait loci (QTLs) for fatty acid composition in an interspecific cross of oil palm. BMC Plant Boil. 2009, 9, 114. [Google Scholar] [CrossRef]
- Tan, B.K.; Berger, K.G. Characteristics of kernel oils fromElaeis oleifera, F1 hybrids and back-cross with Elaeis guineensis. J. Sci. Food Agric. 1982, 33, 204–208. [Google Scholar] [CrossRef]
- Mu, H. The digestion of dietary triacylglycerols. Prog. Lipid Res. 2004, 43, 105–133. [Google Scholar] [CrossRef]
- Haddad, I.; Mozzon, M.; Strabbioli, R.; Frega, N.G. Electrospray ionization tandem mass spectrometry analysis of triacylglycerols molecular species in camel milk (Camelus dromedarius). Int. Dairy J. 2011, 21, 119–127. [Google Scholar] [CrossRef]
- Haddad, I.; Mozzon, M.; Strabbioli, R.; Frega, N.G. A comparative study of the composition of triacylglycerol molecular species in equine and human milks. Dairy Sci. Technol. 2011, 92, 37–56. [Google Scholar] [CrossRef]
- Haddad, I.; Mozzon, M.; Strabbioli, R.; Frega, N.G. Stereospecific analysis of triacylglycerols in camel (Camelus dromedarius) milk fat. Int. Dairy J. 2010, 20, 863–867. [Google Scholar] [CrossRef]
- Haddad, I.; Mozzon, M.; Strabbioli, R.; Frega, N.G. Fatty acid composition and regiodistribution in mare’s milk triacylglycerols at different lactation stages. Dairy Sci. Technol. 2011, 91, 397–412. [Google Scholar] [CrossRef][Green Version]
- Frega, N.; Bocci, F.; Lercker, G. High-resolution gas-chromatographic determination of diacylglycerols in common vegetable oils. J. Am. Oil Chem. Soc. 1993, 70, 175–177. [Google Scholar] [CrossRef]
- Irías-Mata, A.; Stuetz, W.; Sus, N.; Hammann, S.; Gralla, K.; Cordero-Solano, A.; Vetter, W.; Frank, J. Tocopherols, Tocomonoenols, and Tocotrienols in Oils of Costa Rican Palm Fruits: A Comparison between Six Varieties and Chemical versus Mechanical Extraction. J. Agric. Food Chem. 2017, 65, 7476–7482. [Google Scholar] [CrossRef]
- Mozzon, M.; Pacetti, D.; Frega, N.G.; Lucci, P. Crude Palm Oil from Interspecific Hybrid Elaeis oleifera × E. guineensis: Alcoholic Constituents of Unsaponifiable Matter. J. Am. Oil Chem. Soc. 2015, 92, 717–724. [Google Scholar] [CrossRef]
- Choo, Y.-M.; Yap, S.-C.; Ooi, C.-K.; Ma, A.-N.; Goh, S.-H.; Ong, A.S.-H. Recovered oil from palm-pressed fiber: A good source of natural carotenoids, vitamin E, and sterols. J. Am. Oil Chem. Soc. 1996, 73, 599–602. [Google Scholar] [CrossRef]
- Lubrano, C.; Robin, J.R.; Khaiat, A. Fatty acid, sterol and tocopherol composition of oil from the fruit mesocarp of six palm species in French Guiana. Oleagineux 1994, 49, 59–65. [Google Scholar]
- Cadena, T.; Prada, F.; Perea, A.; Romero, H.M. Lipase activity, mesocarp oil content, and iodine value in oil palm fruits of Elaeis guineensis, Elaeis oleifera, and the interspecific hybrid O×G (E. oleifera × E. guineensis). J. Sci. Food Agric. 2012, 93, 674–680. [Google Scholar] [CrossRef]
- Joint FAO/WHO Food Standards Programme. In Proceedings of the 25th Session of the Codex Committee on Fats and Oils, Kuala Lumpur, Malaysia, 27 February–3 March 2017.
- Ruedas, R.J.F.; Dibildox-Alvarado, E.; Martínez, J.D.P.; Hernández, N.I.M. Enzymatically interesterified hybrid palm stearin as an alternative to conventional palm stearin. CYTA J. Food 2019, 18, 1–10. [Google Scholar] [CrossRef]
- Mayamol, P.N.; Balachandran, C.; Samuel, T.; Sundaresan, A.; Arumughan, C. Process Technology for the Production of Micronutrient Rich Red Palm Olein. J. Am. Oil Chem. Soc. 2007, 84, 587–596. [Google Scholar] [CrossRef]
- Vidoca, L.; Almeida, E.; Cardoso, M.; Otavio, L.; Valadares, L.; Monteiro, S. Extraction of carotene from crude hybrid palm oil using polymeric resin. J. Food Eng. 2020, 278. [Google Scholar] [CrossRef]
- Almeida, E.S.; Carvalho, A.C.B.; Soares, I.O.D.S.; Valadares, L.F.; Mendonça, A.R.V.; Monteiro, S.; Junior, I.J.S. Elucidating how two different types of bleaching earths widely used in vegetable oils industry remove carotenes from palm oil: Equilibrium, kinetics and thermodynamic parameters. Food Res. Int. 2019, 121, 785–797. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, J.A.; Almeida, E.S.; Neto, B.A.D.; Abdelnur, P.V.; Monteiro, S. Identification of carotenoid isomers in crude and bleached palm oils by mass spectrometry. LWT Food Sci. Technol. 2018, 89, 631–637. [Google Scholar] [CrossRef]
- Pedersen, J.; Kirkhus, B. Health Aspects of Saturated Fatty Acids. In Reducing Saturated Fats in Foods; Talbott, G., Ed.; Elsevier BV: Amsterdam, The Netherlands, 2011; pp. 77–97. [Google Scholar]
- Puah, C.W.; May, C.Y.; Ma, A.N.; Chuah, C.H. The Effect of Physical Refining on Palm Vitamin E (Tocopherol, Tocotrienol and Tocomonoenol). Am. J. Appl. Sci. 2007, 4, 374–377. [Google Scholar] [CrossRef]
- EFSA Panel on Contaminants in the Food Chain. Risks for human health related to the presence of 3- and 2-monochloropropanediol (MCPD), and their fatty acid esters, and glycidyl fatty acid esters in food. EFSA J. 2016, 14, 4426. [Google Scholar] [CrossRef]
- EFSA Panel on Contaminants in the Food Chain. Update of the risk assessment on 3-monochloropropane diol and its fatty acid esters. EFSA J. 2018, 16, 5083. [Google Scholar] [CrossRef]
- Caramia, G.; Gori, A.; Valli, E.; Cerretani, L. Virgin olive oil in preventive medicine: From legend to epigenetics. Eur. J. Lipid Sci. Technol. 2012, 114, 375–388. [Google Scholar] [CrossRef]
- Tomé-Carneiro, J.; Crespo, M.C.; Hazas, M.C.L.D.L.; Visioli, F.; Dávalos, A. Olive oil consumption and its repercussions on lipid metabolism. Nutr. Rev. 2020, 014. [Google Scholar] [CrossRef]
- Rodríguez, J.C.; Gómez, D.; Pacetti, D.; Núñez, O.; Gagliardi, R.; Frega, N.G.; Ojeda, M.; Loizzo, M.R.; Tundis, R.; Lucci, P. Effects of the Fruit Ripening Stage on Antioxidant Capacity, Total Phenolics, and Polyphenolic Composition of Crude Palm Oil from Interspecific Hybrid Elaeis oleifera × Elaeis guineensis. J. Agric. Food Chem. 2016, 64, 852–859. [Google Scholar] [CrossRef] [PubMed]
- Lucci, P.; Borrero, M.; Ruiz, A.J.; Pacetti, D.; Frega, N.G.; Díez, O.; Ojeda, M.; Gagliardi, R.; Parra, L.; Angel, M. Palm oil and cardiovascular disease: A randomized trial of the effects of hybrid palm oil supplementation on human plasma lipid patterns. Food Funct. 2016, 7, 347–354. [Google Scholar] [CrossRef] [PubMed]
- Ojeda, M.; Borrero, M.; Sequeda-Castañeda, L.G.; Diez, O.; Castro, V.; García, Á.; Ruiz, A.J.; Pacetti, D.; Frega, N.; Gagliardi, R.; et al. Hybrid palm oil (Elaeis oleifera × Elaeis guineensis) supplementation improves plasma antioxidant capacity in humans. Eur. J. Lipid Sci. Technol. 2016, 119, 1600070. [Google Scholar] [CrossRef]
- Spreafico, F.; Sales, R.C.; Gil-Zamorano, J.; Medeiros, P.; Latasa, M.-J.; Lima, M.R.; De Souza, S.A.L.; Martin-Hernandez, R.; Gómez-Coronado, D.; Iglesias-Gutiérrez, E.; et al. Dietary supplementation with hybrid palm oil alters liver function in the common Marmoset. Sci. Rep. 2018, 8, 2765. [Google Scholar] [CrossRef]
- Sales, R.C.; Medeiros, P.; Spreafico, F.; De Velasco, P.C.; Gonçalves, F.K.A.; Martin-Hernandez, R.; Mantilla-Escalante, D.C.; Gil-Zamorano, J.; Peres, W.A.F.; Souza, S.A.L.; et al. Olive Oil, Palm Oil, and Hybrid Palm Oil Distinctly Modulate Liver Transcriptome and Induce NAFLD in Mice Fed a High-Fat Diet. Int. J. Mol. Sci. 2018, 20, 8. [Google Scholar] [CrossRef]
- Gesteiro, E.; Galera-Gordo, J.; González-Gross, M. Palm oil and cardiovascular health: Considerations to evaluate the literature critically. Nutr. Hosp. 2018, 35, 1229–1242. [Google Scholar] [CrossRef]
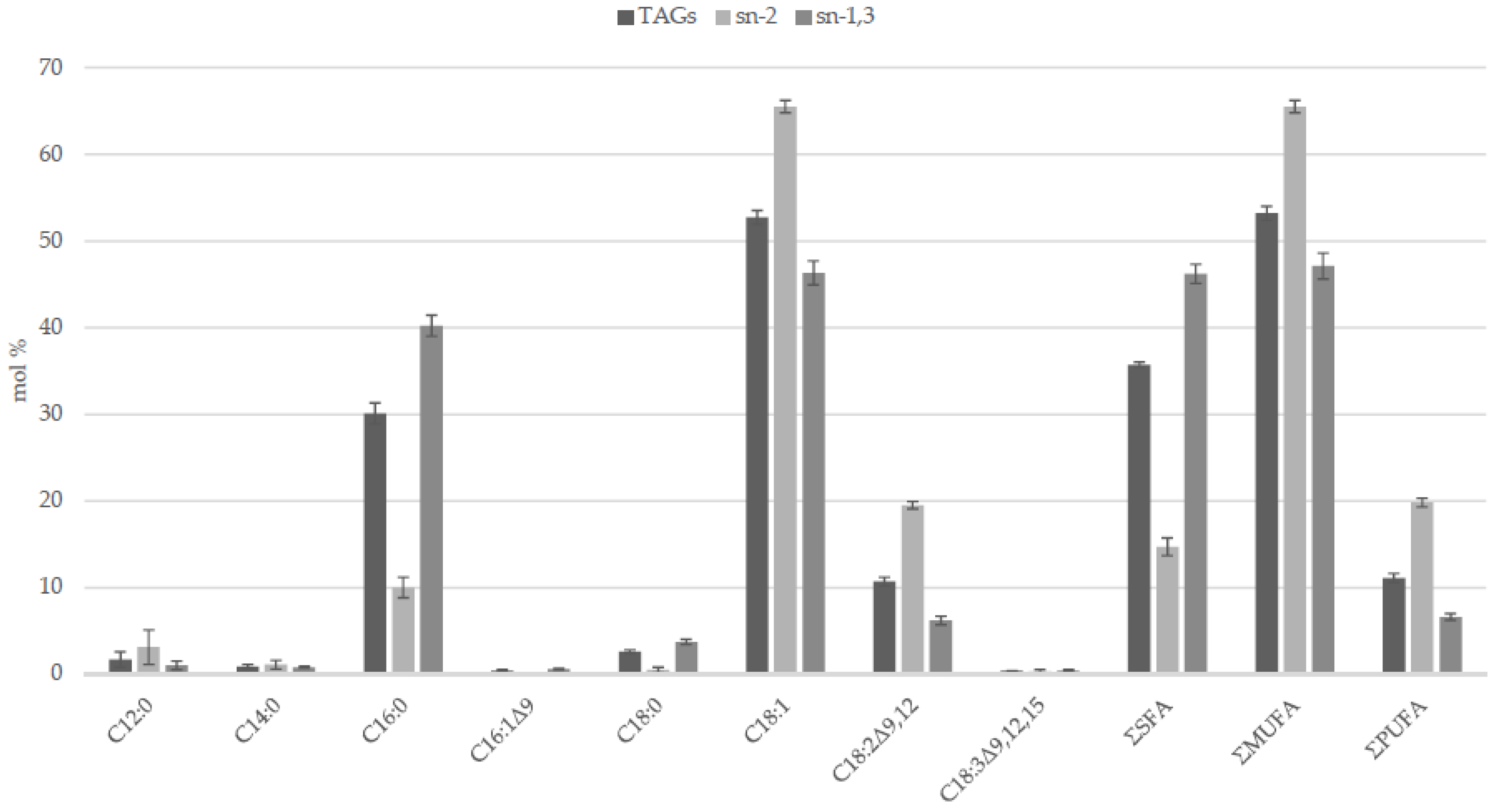
| C12:0 1 | C14:0 | C16:0 | C16:1 Δ9 | C18:0 | C18:1 2 | C18:2 Δ9,12 | C18:3 Δ9,12,15 | Reference 3 |
|---|---|---|---|---|---|---|---|---|
| 0.5–1.7 | 0.5–0.9 | 27.7–29.5 | 0.3–0.4 | 2.6–3.1 | 53.5–55.2 | 10.7–11.5 | 0.4–0.4 | [6] |
| 0.9–0.9 | 37.0–43.5 | 0.2–0.2 | 4.0–4.3 | 38.7–43.4 | 10.7–12.7 | 0.3–0.4 | [8] | |
| 0.1–0.5 | 22.4–44.7 | 1.4–4.9 | 36.9–60.1 | 8.2–16.8 | [9] | |||
| 0.1–0.6 | 22.3–34.3 | 0.2–0.8 | 1.5–3.1 | 48.2–61.4 | 10.5–15.1 | 0.4–0.7 | [10] | |
| 0.0–0.1 | 0.5–0.9 | 27.3–32.5 | 3.4–6.1 | 48.0–52.5 | 11.3–11.8 | 0.4–1.3 | [14] | |
| tr. | 0.4–0.9 | 29.3–35.5 | 3.0–4.6 | 50.2–53.4 | 10.3–13.9 | [16] | ||
| 28.1–31.3 | 2.3–2.7 | 51.8–56.4 | 9.4–10.4 | [17] | ||||
| 0.4–0.5 | 0.4–0.4 | 32.2–40.3 | 0.3–0.5 | 2.7–3.8 | 49.7–57.0 | 4.1–5.6 | 0.1–0.2 | [30] |
| 0.2–0.3 | 26.2–32.5 | 0.1–1.4 | 1.5–4.8 | 48.4–58.2 | 9.9–13.0 | 0.3–0.5 | [53] | |
| 0.5–1,6 | 32.2–43.1 | 3.2–4.1 | 36.9–60.1 | 8.2–16.8 | [54] | |||
| 0.4–0.8 | 36.2–41.4 | tr. | 0.4–1.5 | 48.2–53.3 | 6.5–9.3 | 0.1–0.7 | [55] 4 | |
| 0.3–0.9 | 28.9–38.6 | 3.3–5.9 | 44.9–56.0 | 9.3–11.5 | [56] |
| C6:0 1 | C8:0 | C10:0 | C12:0 | C14:0 | C16:0 | C18:0 | C18:1 2 | C18:2 Δ9,12 | Reference 3 |
|---|---|---|---|---|---|---|---|---|---|
| 1.2–2.3 | 1.1–2.2 | 35.0–42.3 | 19.6–24.7 | 9.1–10.2 | 2.4–3.5 | 17.2–19.1 | 4.4.–4.7 | [8] | |
| 0.2–0.2 | 3.2–3.4 | 2.7–2.9 | 44.4–46.8 | 18.1–18.6 | 7.9–8.8 | 2.1–2.2 | 14.8–16.3 | 3.2–3.4 | [9,61] |
| 0.2 | 4.0 | 3.5 | 50.0 | 16.5 | 7.8 | 2.2 | 13.1 | 2.4 | [9,61] |
| tr. | 1.3–3.2 | 1.8–3.2 | 40.6–49.0 | 17.4–22.1 | 8.0–9.5 | 1.5–2.5 | 14.1–18.5 | 1.0–4.5 | [16] |
| Mesocarp Oil [Reference] 3 | Kernel Oil [Reference] 3 | |||||||
|---|---|---|---|---|---|---|---|---|
| TAG m:n 1 | TAG ABC 2 | [6] | [9] | [15] | [17] | [30] | [9] | [61] |
| ΣC28 | 0.1 | 0.1–0.2 | ||||||
| ΣC30 | 0.7 | 0.4–0.7 | ||||||
| ΣC32 | 4.7 | 2.9–3.9 | ||||||
| ΣC34 | 7.1 | 4.8–6.0 | ||||||
| ΣC36 | 24.5 | 17.9–19.6 | ||||||
| ΣC38 | 18.9 | 17.2–18.0 | ||||||
| ΣC40 | 10.3 | 10.9–11.3 | ||||||
| ΣC42 | 9.1 | 9.9–10.6 | ||||||
| ΣC44 | 6.7 | 8.0–8.8 | ||||||
| 46:0 | MPP | 0.1 ± 0.0 | 0.1–0.5 | |||||
| 46:1 | MMO + LaPO | 0.3 ± 0.2 | tr–0.3 | |||||
| ΣC46 | 0.0–1.1 | 5.1 | 6.3–7.2 | |||||
| 48:0 | PPP | 1.3 ± 1.3 | 0.0–0.6 | 1.5–2.8 | ||||
| 48:1 | MOP | 0.8 ± 0.0 | 0.4–0.6 | |||||
| 48:2 | MLP | 0.2 ± 0.1 | 0.4 ± 0.0 | 0.0 – 0.7 | tr–0.1 | |||
| ΣC48 | 0.9–8.9 | 6.0 | 7.1–8.0 | |||||
| 50:0 | PPS | 0.4 ± 0.3 | 0.0–0.2 | 0.2–0.6 | ||||
| 50:1 | PPO | 20.4 ± 0.2 | 17.0 ± 2.4 | 10.4–15.3 | 20.3–21.1 | |||
| 50:2 | PPL | 5.5 ± 0.2 | 9.4 ± 0.9 | 5.6–9.4 | 2.5–3.2 | |||
| 50:2 | MOO | 0.5 ± 0.1 | 0.3–0.5 | |||||
| ΣC50 | 11.1–25.5 | 2.3 | 3.3–3.9 | |||||
| 52:0 | PSS | tr | tr | |||||
| 52:1 | POS | 3.3 ± 0.2 | 2.8 ± 0.5 | 1.5–1.8 | 2.8–3.8 | |||
| 52:2 | PLS | 1.6 ± 0.3 | 1.7–2.0 | |||||
| 52:2 | POO | 32.6 ± 2.4 | 23.4 ± 0.7 | 21.9–24.8 | 33.1–35.8 | |||
| 52:3 | PLO | 11.2 ± 0.2 | 17.7 ± 0.8 | 17.8–20.2 | 7.4–8.9 | |||
| 52:4 | PLL + POLn | 2.0 ± 0.0 | 6.7 ± 0.6 | 7.4–9.4 | 1.1–1.9 | |||
| ΣC52 | 43.5–50.5 | 1.9 | 2.9–3.3 | |||||
| 54:1 | SSO | 0.3 ± 0.0 | 0.2–0.4 | 0.3–0.4 | ||||
| 54:2 | SOO | 2.6 ± 0.1 | 1.8 ± 0.3 | 1.1–2.5 | 2.3–3.5 | |||
| 54:3 | SLO | 0.7 ± 0.7 | 1.4–1.5 | |||||
| 54:3 | OOO | 10.7 ± 0.2 | 7.6 ± 1.0 | 8.5–12.8 | 12.2–14.2 | |||
| 54:4 | OOL | 4.7 ± 0.1 | 7.6 ± 0.6 | 8.5–11.3 | 4.6–5.2 | |||
| 54:5 | OLL | 0.2 ± 0.1 | 3.2 ± 0.3 | 3.9–5.0 | tr–0.3 | |||
| ΣC54 | 21.8–44.7 | 2.6 | 3.0–3.5 | |||||
| 56:1 | SOA | 1.1 ± 0.1 | 0.9–1.1 | |||||
| 56:2 | AOO | 0.1 ± 0.0 | 0.1–0.1 | |||||
| ΣC56 | 0.0–0.6 | |||||||
| Reference | [14] | [17] | [30] | [53] | [54] | [68] | [69] |
|---|---|---|---|---|---|---|---|
| Carotenoids | 1070–1800 | 514–1375 | 1172.1–1449.6 | 800–2400 | 10,389.3 ± 1004.9 | ||
| α-carotene | 447.9–577.7 | 32.8–36.4 | |||||
| β-carotene | 724.2–911.8 | 51.6%–60.5% | |||||
| Squalene | 20.3–83.1 | 247.4 ± 3.3 | |||||
| 4-desmethylsterols | |||||||
| Cholesterol | 7.8–10.2 3.5%–5.4% | 3%–5% | 10.0 ± 2.6 1.8 ± 0.4% | ||||
| Campesterol | 18.8–47.6 11.8%–16.3% | 20%–22% | 93.1 ± 23.4 19.3 ± 1.2% | ||||
| Ergosterol | 11.0 ± 3.4 1.9 ± 0.3% | ||||||
| Stigmasterol | 25.8–45.2 15.3%–16.3% | 13%–19% | 62.8 ± 10.8 13.1 ± 0.6% | ||||
| Δ7-campesterol | 2.3–3.6 0.8%–1.9% | 1.7 ± 0.7 0.5 ± 0.3% | |||||
| β-sitosterol | 98.2–180.9 61.5%–62.4% | 58%–61% | 275.6 ± 57.4 59.3 ± 1.0% | ||||
| Δ5-avenasterol | 1.6–3.9 0.9%–1.4% | 8.8 ± 1.3 1.9 ± 0.2% | |||||
| Δ5,24-stigmastadienol | 2.3–3.6 1.1%–1.9% | 2.1 ± 1.4 0.5 ± 0.2% | |||||
| Fucosterol | 5.6 ± 2.9 1.1 ± 0.6% | ||||||
| Other unidentified sterols | 2.1 ± 1.4 0.5 ± 0.2% | ||||||
| Total 4-desmethylsterols | 469–1417 | 158.7–293.8 | 700–1400 | 472.7 ± 102.8 | |||
| Isoprenoid alcohols | |||||||
| Phytol | 127.5–175.0 | 120.7 ± 26.1 | |||||
| 3,7,11,15-tetramethyl-2,6-hexadien-1-ol | 11.3 ± 2.1 | ||||||
| 3,7,11,15-tetramethyl-2,6,10-hexatrien-1-ol | 7.7 ± 1.5 | ||||||
| Geranylgeraniol | 31.3–76.3 | 129.0 ± 31.7 | |||||
| Total Isoprenoid alcohols | 160.7–251.3 | 269.3 ± 60.0 | |||||
| n-Alkanols | |||||||
| n-octadecanol | 5.3 ± 2.1 | ||||||
| n-docosanol | 0.5–1.4 | 1.8 ± 1.3 | |||||
| n-tetracosanol | 0.4–1.2 | 1.2 ± 0.5 | |||||
| n-hexacosanol | 0.4–2.5 | 2.7 ± 0.2 | |||||
| n-octacosanol | 3.0–5.2 | 7.3 ± 0.8 | |||||
| n-nonacosanol | tr | ||||||
| n-triacontanol | 7.2–12.9 | 15.6 ± 1.6 | |||||
| n-hentriacontanol | 0.7 ± 1.2 | ||||||
| n-dotriacontanol | 6.9–13.1 | 18.1 ± 6.5 | |||||
| n-tritriacontanol | 0.7 ± 1.2 | ||||||
| n-tetratriacontanol | 2.2–37.4 | 8.2 ± 7.6 | |||||
| Total n-Alkanols | 24.9–37.4 | 61.7 ± 17.0 | |||||
| 4-methylsterols | |||||||
| gramisterol | |||||||
| obtusifoliol | 2.7–5.2 | ||||||
| citrostadienol | 4.2–9.8 | ||||||
| Total 4-methylsterols | 6.9–14.9 | 12.7 ± 1.5 | |||||
| 4,4-dimethylsterols | |||||||
| Cycloartenol | 14.6–24.9 | ||||||
| 24-methylene-cycloartanol | 2.0–3.4 | ||||||
| Isoarborinol | 2.0–3.9 | ||||||
| 9,19-cyclopropanesterol | 0.8–1.6 | ||||||
| Total 4,4-dimethylsterols | 20.0–33.7 | 74.0 ± 12.3 | |||||
| Tocols | |||||||
| α-tocopherol | 1.5–7.4 | 26.8–142.8 | 11–24% | 4.2–5.1 | 27.1 ± 7.4 10.0 ± 0.2% | ||
| β- tocopherol | 0–0.15 | tr. 0.3 ± 0.3% | |||||
| γ- tocopherol | 0–0.27 | tr. 0.3 ± 0.4% | |||||
| δ- tocopherol | 0–0.17 | ||||||
| α-tocotrienol | 199.3–383.9 | 22%–31% | 59.4–101.9 | 44.7 ± 13.7 15.0 ± 1.9% | |||
| β- tocotrienol | 1.1–3.0 | 3.7 ± 1.2 1.4 ± 0.4% | |||||
| γ- tocotrienol | 9.4–18.9 | 666.0–998.42 | 42%–51% | 146.0–343.3 | 148.1 ± 23.3 59.7 ± 1.1% | ||
| δ- tocotrienol | 41.2–45.6 | 5%–9% | 9.4–17.9 | 31.8 ± 4.2 11.7 ± 0.8% | |||
| α-tocomonoenol | 0.59–1.4 | 4.0 ± 1.7 1.6 ± 0.3% | |||||
| Total Tocols (Vitamin E) | 452–2189 | 10.9–26.2 | 937.6–1549.6 | 600–1000 | 222.0–471.9 | 259.3 ± 48.4 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mozzon, M.; Foligni, R.; Mannozzi, C. Current Knowledge on Interspecific Hybrid Palm Oils as Food and Food Ingredient. Foods 2020, 9, 631. https://doi.org/10.3390/foods9050631
Mozzon M, Foligni R, Mannozzi C. Current Knowledge on Interspecific Hybrid Palm Oils as Food and Food Ingredient. Foods. 2020; 9(5):631. https://doi.org/10.3390/foods9050631
Chicago/Turabian StyleMozzon, Massimo, Roberta Foligni, and Cinzia Mannozzi. 2020. "Current Knowledge on Interspecific Hybrid Palm Oils as Food and Food Ingredient" Foods 9, no. 5: 631. https://doi.org/10.3390/foods9050631
APA StyleMozzon, M., Foligni, R., & Mannozzi, C. (2020). Current Knowledge on Interspecific Hybrid Palm Oils as Food and Food Ingredient. Foods, 9(5), 631. https://doi.org/10.3390/foods9050631







