Quality Properties of Chicken Emulsion-Type Sausages Formulated with Chicken Fatty Byproducts
Abstract
1. Introduction
2. Materials and Methods
2.1. Sausages Preparation
2.2. Proximate Composition
2.3. Color Evaluation
2.4. Texture Profile Analysis
2.5. Microbiological Analysis
2.6. Sensory Evaluation
2.7. Statistical Analysis
3. Results & Discussion
3.1. Proximate Composition
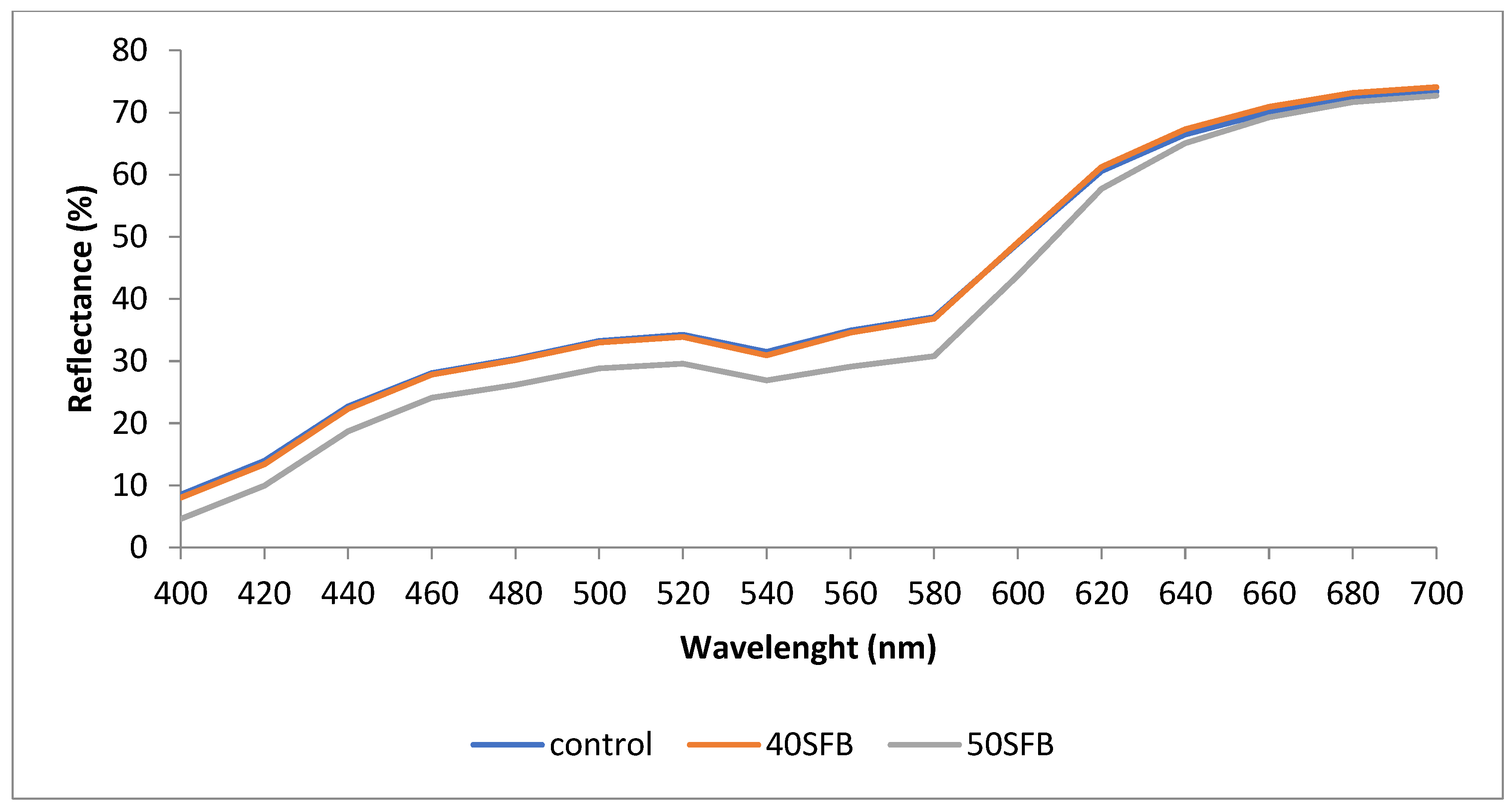
3.2. Color Properties
3.3. Texture Properties
3.4. Microbiological Analysis
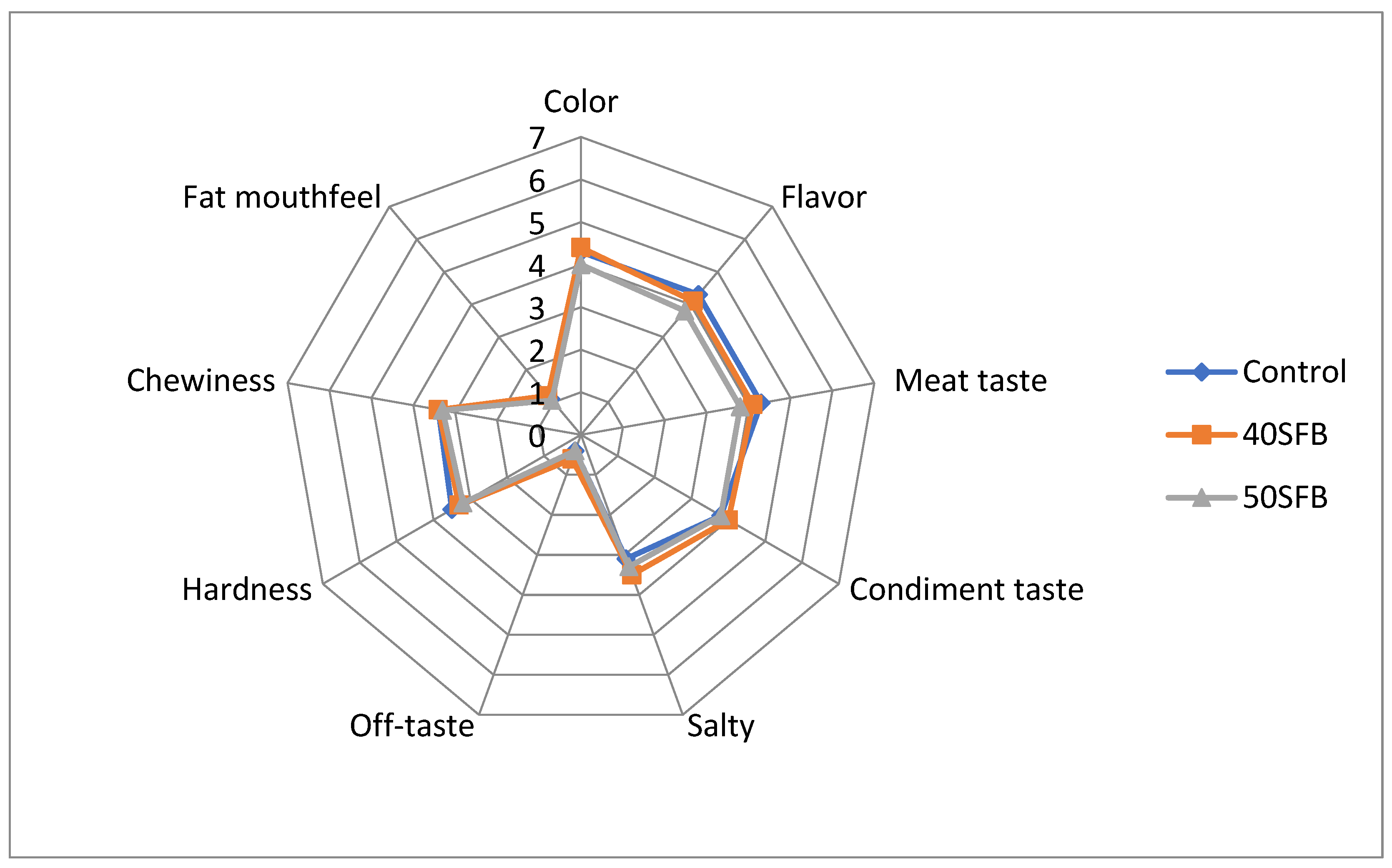
3.5. Sensory Evaluation
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Marques, H.L. Avesui Latin America. 2019. Available online: https://en.avesui.com/noticias/in-latin-america-poultry-production-will-grow-nearly-double-world-average/20191002-091938-K505 (accessed on 10 March 2020).
- FENAVI. Consumo per Capita Mundial de Carne de Pollo. Federación Nacional de Avicultores de Colombia. 2019. Available online: https://fenavi.org/estadisticas/consumo-per-capita-mundo-pollo/ (accessed on 12 February 2020).
- Le Guerhier, F. Processing of Poultry Fat. 2012. Available online: http://www.fao.org/catalog (accessed on 14 January 2020).
- Savadkoohi, S.; Hoogenkamp, H.; Shamsi, K.; Farahnaky, A. Color, sensory and textural attributes of beef frankfurter, beef ham and meat-free sausage containing tomato pomace. Meat Sci. 2014, 97, 410–418. [Google Scholar] [CrossRef] [PubMed]
- Jayawardana, B.C.; Liyanage, R.; Lalantha, N.; Iddamalgoda, S.; Weththasinghe, P. Antioxidant and antimicrobial activity of drumstick (Moringa oleifera) leaves in herbal chicken sausages. LWT Food Sci. Technol. 2015, 64, 1204–1208. [Google Scholar] [CrossRef]
- Choe, J.H.; Kim, H.Y.; Lee, J.M.; Kim, Y.J.; Kim, C.J. Quality of frankfurter-type sausages with added pig skin and wheat fiber mixture as fat replacers. Meat Sci. 2013, 93, 849–854. [Google Scholar] [CrossRef] [PubMed]
- Giese, J. Fats, oils, and fat replacers. Food Technol. 1996, 50, 48–83. [Google Scholar]
- Ospina, -E.J.C.; Cruz, -S.A.; Pérez-Alvarez, J.A.; Fernández-López, J. Development of combinations of chemically modified vegetable oils as pork backfat substitutes in sausages formulation. Meat Sci. 2010, 84, 491–497. [Google Scholar] [CrossRef] [PubMed]
- Ospina, -E.J.C.; Sierra, -C.A.; Ochoa, O.; Pérez-Alvarez, J.A.; Fernández-López, J. Substitution of saturated fat in processed meat products: A review. Crit. Rev. Food Sci. Nutr. 2012, 52, 113–122. [Google Scholar] [CrossRef]
- Araujo, I.B.S.; Lima, D.A.S.; Pereira, S.F.; Madruga, M.S. Quality of low-fat chicken sausages with added chicken feet collagen. Poult. Sci. 2019, 98, 1064–1074. [Google Scholar] [CrossRef]
- Glorieux, S.; Steen, L.; Van de Walle, D.; Dewettinck, K.; Foubert, I.; Fraeye, I. Effect of meat type, animal fat type, and cooking temperature on microstructural and macroscopic properties of cooked sausages. Food Bioprocess. Tech. 2019, 12, 16–26. [Google Scholar] [CrossRef]
- Utama, D.T.; Jeong, H.S.; Kim, J.; Barido, F.H.; Lee, S.K. Fatty acid composition and quality properties of chicken sausage formulated with pre-emulsified perilla-canola oil as an animal fat replacer. Poult. Sci. 2019, 98, 3059–3063. [Google Scholar] [CrossRef]
- Lafarga, T.; Hayes, M. Bioactive peptides from meat muscle and byproducts: Generation, functionality and application as functional ingredients. Meat Sci. 2014, 98, 227–239. [Google Scholar] [CrossRef]
- Herrera, M. Taking Advantage of By-Products or Waste in the Poultry Industry for the Production of Animal Meal. Virtual Pro, Bogotá, Colombia. 2008. Available online: https://www.virtualpro.co/biblioteca/aprovechamiento-de-los-subproductos-o-residuos-en-la-industria-avicola-para-la-produccion-de-harinas-de-origen-animal (accessed on 12 December 2019).
- FAO. Fats and fatty acids in human nutrition. Report of an expert consultation. FAO Food Nutr. Pap. 2010, 91, 1–166. [Google Scholar]
- Barbut, S. Waste Treatment and By-Products. In The Science of Poultry and Meat Processing; Creative Commons: Mountain View, CA, USA, 2015; pp. 1–27. [Google Scholar]
- Vikman, M.; Siipola, V.; Kanerva, H.; Slizyte, R.; Wikberg, H. Poultry by-products as potential source of nutrients. Adv. Recycl. Waste Mang. 2017, 2, 1–5. [Google Scholar]
- AOAC. Official Methods of Analysis of AOAC International, 18th ed.; Association of Official Analytical Chemistry: Rockville, MD, USA, 2005. [Google Scholar]
- AMSA. Meat Color Measurement Guidelines; American Meat Science Association: Savoy, IL, USA, 2012. [Google Scholar]
- Claus, J.R. Methods for the objective measurement of meat product texture. Recipr. Meat Conf. Proc. 1995, 48, 96–100. [Google Scholar]
- ICMSF. International Commission on Microbiological Specifications for Foods. Microorganismos de Los Alimentos: Técnicas de Análisis Microbiológico, 2nd ed.; Acribia: Zaragoza, Spain, 1983. [Google Scholar]
- ISO 6579. Microbiology of Food and Animal Feeding Stuffs-Horizontal Method for the Detection of Salmonella spp.; International Organization for Standardization: Geneva, Switzerland, 2003. [Google Scholar]
- ISO 11290-2. Microbiology of Food and Animal Feeding Stuffs-Horizontal Method for the Detection and Enumeration of Listeria Monocytogenes; International Organization for Standardization: Geneva, Switzerland, 2000. [Google Scholar]
- ISO 7937. Microbiology of Food and Animal Feeding Stuffs—Horizontal Method for the Enumeration of Clostridium Perfringens—Colony-Count Technique; ISO: Geneva, Switzerland, 2004. [Google Scholar]
- ISO 4832. Microbiology of Food and Animal Feeding Stuffs—Horizontal Method for the Enumeration of Coliforms—Colony-Count Technique; ISO: Geneva, Switzerland, 2006. [Google Scholar]
- ISO 6888. Microbiology of Food and Animal Feeding Stuffs—Horizontal Method for the Enumeration of Coagulase-Positive Staphylococci (Staphylococcus aureus and Other Species); ISO: Geneva, Switzerland, 2018. [Google Scholar]
- Bennett, R.W.; Lancette, G.A. Staphylococcus aureus. In Bacteriological Analytical Manual, 8th ed.; Food and Drug Administration: Silver Spring, MD, USA, 2001. [Google Scholar]
- ISO 4121. Sensory Analysis-Guidelines for the Use of Quantitative Response Scales. International Organization for Standardization; ISO: Geneva, Switzerland, 2003. [Google Scholar]
- Feddern, V.; Kupski, L.; Cipolatti, E.P.; Giacobbo, G.; Mendes, G.L.; Badiale-Furlong, E.; de Souza-Soares, L.A. Physico-chemical composition, fractionated glycerides and fatty acid profile of chicken skin fat. Eur. J. Lipid. Sci. Technol. 2010, 112, 1277–1284. [Google Scholar] [CrossRef]
- Peña-Saldarriaga, L.M.; Fernández-López, J.; Sayas-Barberá, E.; Pérez-Alvarez, J.A. Broiler chickens: Characterization of abdominal and gizzard fat. In Proceedings of the 1st World Congress of Food Safety and Security, Leiden, The Netherlands, 24–28 March 2019; p. 76. [Google Scholar]
- Troutt, E.S.; Hunt, M.C.; Johnson, D.E.; Claus, J.R.; Kastner, C.L.; Kroff, D.H.; Stroda, S. Chemical, physical and sensory characterization of ground beef containing 5 to 30 percent fat. J. Food Sci. 1992, 57, 25–29. [Google Scholar] [CrossRef]
- Biswas, S.; Chakraborty, A.; Sarkar, S.; Barpuzari, R.N.; Barpuzari, T. Effect of incorporation of chicken fat and skin on the quality of chicken sausages. J. Poult. Sci. 2007, 44, 111–115. [Google Scholar] [CrossRef][Green Version]
- NTC 1325. Norma Técnica Colombiana Para La Industria Alimentaria: Productos Cárnicos Procesados no Enlatados; ICONTEC: Bogota, Colombia, 2008.
- Carrillo-Fernández, L.; Dalmau-Serra, J.; Martínez-Alvarez, R.; Solá-Alberich, R.; Pérez-Jiménez, F. Dietary fats and cardiovascular health. Clin. Investig. Arterioscler. 2011, 23, 1–36. [Google Scholar]
- Chiu, M.C.; Gioielli, L.A.; Grimaldi, R. Structured lipids from chicken fat, its stearin and medium chain triacylglycerol blends. I-Fatty acid and triacylglycerol compositions. Quim. Nova 2008, 31, 232–237. [Google Scholar] [CrossRef][Green Version]
- Kamani, M.H.; Safari, O.; Mortazavi, S.A.; Atash, M.M.S.; Azghadi, N.M. Using an image processing based technique and predictive models for assessing lipid oxidation in rainbow trout fillet. Food Biosci. 2017, 19, 42–48. [Google Scholar] [CrossRef]
- Hughes, E.; Cofrades, S.; Troy, D.J. Effects of fat level, oat fibre and carrageenan on frankfurters formulated with 5, 12 y 30% fat. Meat Sci. 1997, 45, 273–281. [Google Scholar] [CrossRef]
- Jiménez-Colmenero, F.; Reig, M.; Toldrá, F. New approaches for the development of functional meat products. In Advanced Technologies for Meat Processing; Nollet, L.M.L., Toldrá, Y.F., Eds.; CRC Press: Boca Raton, FL, USA, 2006; Chapter 11; pp. 275–308. [Google Scholar]
- Choe, J.H.; Kim, H.Y. Quality characteristics of reduced fat emulsion-type chicken sausages using chicken skin and wheat fiber mixture as fat replacer. Poult. Sci. 2019, 98, 2662–2669. [Google Scholar] [CrossRef]
- Herrero, A.M.; Ordóñez, J.A.; de Avila, R.; Herranz, B.; de la Hoz, L.; Cambero, M.I. Breaking strength of dry fermented sausages and their correlation with texture profile analysis (TPA) and physico-chemical characteristics. Meat Sci. 2007, 36, 331–338. [Google Scholar] [CrossRef] [PubMed]
- Bolger, Z.; Brunton, N.P.; Monaham, F.J. Impact of inclusión of flaxeed oil (pre-emulsified or encapasulated) on the physical characteristics of chicken sausages. J. Food Eng. 2018, 230, 39–48. [Google Scholar] [CrossRef]
- Kamani, M.H.; Meera, M.S.; Bhaskar, N.; Modi, V.K. Partial and total replacement of meat by plant-based proteins in chicken sausage: Evaluation of mechanical, physico-chemical and sensory characteristics. J. Food Sci. Technol. 2019, 56, 2660–2669. [Google Scholar] [CrossRef] [PubMed]
| Ingredient | Mixing Time (min) | Control | 40SFB | 50SFB |
|---|---|---|---|---|
| kg | kg | kg | ||
| Chicken breast | 10 min | 43 | 43 | 43 |
| MDM 1(chicken) | 18 | 18 | 18 | |
| Sodium lactate | 2.3 | 2.3 | 2.3 | |
| Water | 3 | 3 | 3 | |
| Salt | 0.25 | 0.25 | 0.25 | |
| Cured salt (12% potassium nitrate) | 0.2 | 0.2 | 0.2 | |
| Accord phosphates | 0.54 | 0.54 | 0.54 | |
| Flavor mix for sausages 2 | 1.51 | 1.51 | 1.51 | |
| Isolate soya protein | 2 min | 2.5 | 2.5 | 2.5 |
| Binder XT 202 | 4.8 | 4.8 | 4.8 | |
| Water | 4 min | 4 | 4 | 4 |
| Acid violet 051 (added to water for dilution) | 30 mL | 30 mL | 30 mL | |
| Water | 4 min | 4 | 4 | 4 |
| Wheat flour | 4.5 | 4.5 | 4.5 | |
| Sodium ascorbate | 0.40 | 0.40 | 0.40 | |
| Wheat starch | 4 | 4 | 4 | |
| Chicken skin (minced) | 27 | 16.2 | 13.5 | |
| Chicken fat byproducts | 10.8 | 13.5 | ||
| TOTAL | 20 min | 120 | 120 | 120 |
| Moisture (%) | Fat (%) | Protein (%) | Ash (%) | |
|---|---|---|---|---|
| Control | 62.17a | 10.14c | 13.04a | 3.42a |
| 40SFB | 61.63b | 13.08b | 12.54a | 3.52a |
| 50SFB | 61.40b | 14.67a | 12.43a | 3.42a |
| SAMPLE | L* | a* | b* | C* | h* | ΔE* |
|---|---|---|---|---|---|---|
| CONTROL | 66.41 ± 0.21b | 13.33 ± 0.09a | 18.76 ± 0.15b | 23.00 ± 0.15a | 54.60 ± 0.20b | |
| 40SFB | 67.62 ± 0.25b | 13.42 ± 0.11a | 19.70 ± 0.20a | 23.82 ± 0.23a | 56.73 ± 0.25a | 2.2 |
| 50SFB | 69.42 ± 0.26a | 12.51 ± 0.12b | 19.93 ± 0.17a | 23.53 ± 0.19a | 57.87 ± 0.15a | 3.3 |
| Microorganisms | Results (CFU/g) | ||
|---|---|---|---|
| CONTROL | 40SFB | 50SFB | |
| Mesophiles | 1234 ± 130 | 1086 ± 180 | 1160 ± 147 |
| Total coliforms | <10 | <10 | <10 |
| E. coli | <10 | <10 | <10 |
| Coagulase-positive Staphylococcus | <100 | <100 | <100 |
| Salmonella | Absence/25 g | Absence/25 g | Absence/25 g |
| Listeria monocytogenes | Absence/25 g | Absence/25 g | Absence/25 g |
| Spores of sulfite-reducing Clostridium | <10 | <10 | <10 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peña-Saldarriaga, L.M.; Pérez-Alvarez, J.A.; Fernández-López, J. Quality Properties of Chicken Emulsion-Type Sausages Formulated with Chicken Fatty Byproducts. Foods 2020, 9, 507. https://doi.org/10.3390/foods9040507
Peña-Saldarriaga LM, Pérez-Alvarez JA, Fernández-López J. Quality Properties of Chicken Emulsion-Type Sausages Formulated with Chicken Fatty Byproducts. Foods. 2020; 9(4):507. https://doi.org/10.3390/foods9040507
Chicago/Turabian StylePeña-Saldarriaga, Lina María, José Angel Pérez-Alvarez, and Juana Fernández-López. 2020. "Quality Properties of Chicken Emulsion-Type Sausages Formulated with Chicken Fatty Byproducts" Foods 9, no. 4: 507. https://doi.org/10.3390/foods9040507
APA StylePeña-Saldarriaga, L. M., Pérez-Alvarez, J. A., & Fernández-López, J. (2020). Quality Properties of Chicken Emulsion-Type Sausages Formulated with Chicken Fatty Byproducts. Foods, 9(4), 507. https://doi.org/10.3390/foods9040507






