Influences of Electron Beam Irradiation on the Physical and Chemical Properties of Zearalenone- and Ochratoxin A-Contaminated Corn and In Vivo Toxicity Assessment
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
2.2. Main Instruments and Equipment
2.3. EBI Treatment of Contaminated Corn
2.4. Determination of OTA and ZEN in Corn and Fodders
2.5. Test of Physical and Chemical Properties of Corn
2.5.1. Amylose Content Test
2.5.2. Starch Crystallinity Test
2.5.3. Observation of Starch Particles
2.5.4. Test of Amino Acids in Corn
2.6. Mouse Experiment
2.6.1. Animal Grouping and Culture
2.6.2. Serum and Routine Blood Biochemical Examination
2.6.3. Organ Index and Histopathological Examination
2.7. Data Processing
3. Results and Discussion
3.1. Effects of EBI on the Physical and Chemical Properties of Corn
3.1.1. Effects of EBI on Corn Amylose Content
3.1.2. Effects of EBI on Starch Crystallinity of Corn
3.1.3. Effects of EBI on the Appearance of Corn Starch Particles
3.1.4. Effects of EBI on Amino Acid Content
3.2. In vivo Toxicity Experiment of Mice
3.2.1. Weight Changes of Mice
3.2.2. Changes of the Organ Weights and Organ Indexes of Mice
3.2.3. Changes of Blood Indexes
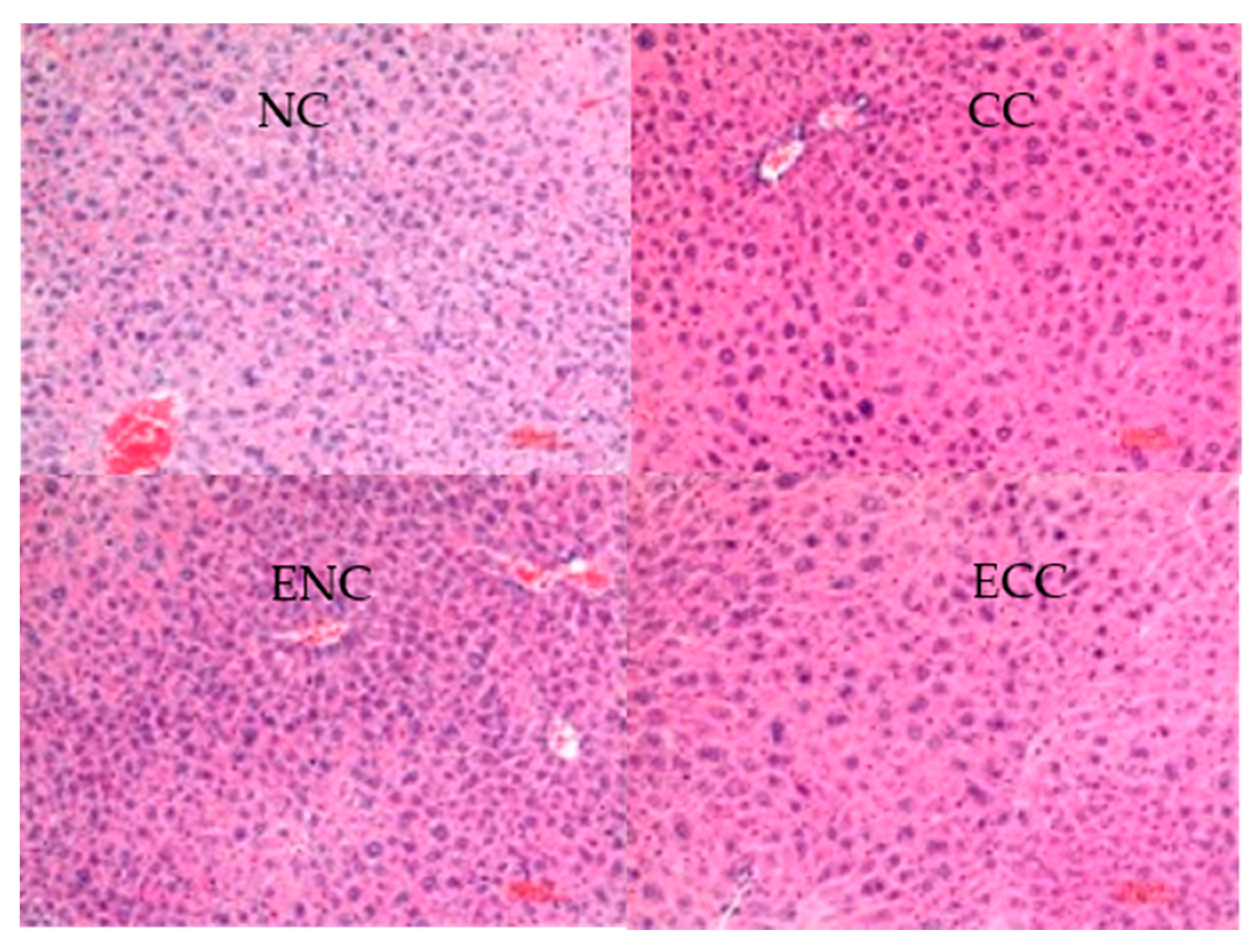
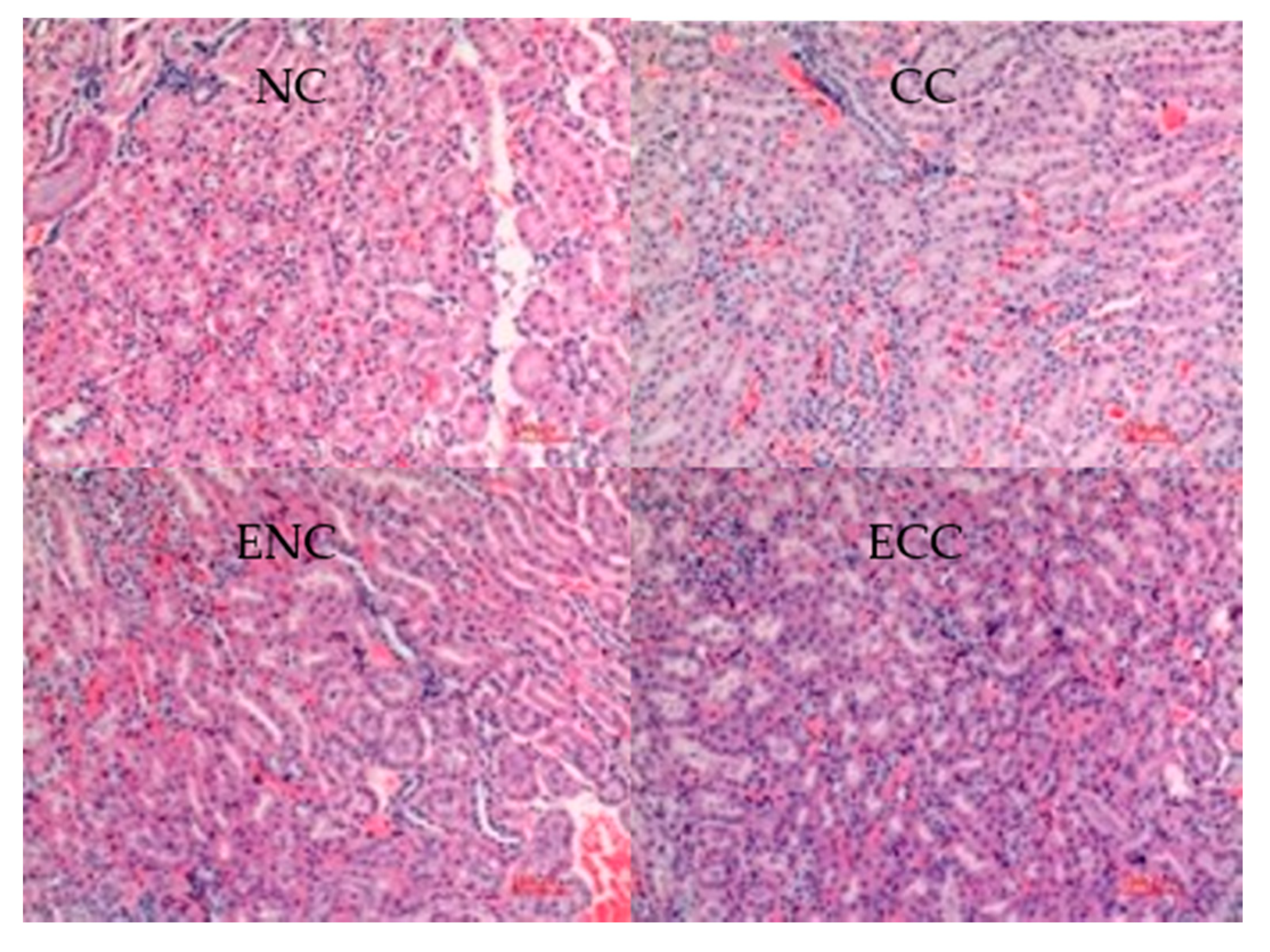
3.2.4. Liver and Kidney Histopathological Tissue Sections of Mice
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hussein, H.S.; Brasel, J.M. Toxicity, metabolism, and impact of mycotoxins on humans and animals. Toxicology 2001, 167, 101–134. [Google Scholar] [CrossRef]
- Shephard, G.S.; Berthiller, F.; Burdaspal, P.A.; Crews, C.; Jonker, M.A.; Krska, R.; Lattanzio, V.M.T.; MacDonald, S.; Malone, R.J.; Maragos, C.; et al. Developments in mycotoxin analysis: An update for 2011–2012. World Mycotoxin J. 2013, 6, 3–30. [Google Scholar] [CrossRef]
- Janati, S.S.F.; Beheshti, H.R.; Asadi, M.; Mihanparast, S.; Feizy, J. Preliminary survey of aflatoxins and ochratoxin A in dried fruits from Iran. Bull. Environ. Contam. Toxicol. 2012, 88, 391–395. [Google Scholar] [CrossRef] [PubMed]
- Toffa, D.D.; Mahnine, N.; Ouaffak, L.; El Abidi, A.; El Alaoui Faris, F.Z.; Zinedine, A. First survey on the presence of ochratoxin A and fungi in raw cereals and peanut available in the Republic of Niger. Food Control 2013, 32, 558–562. [Google Scholar] [CrossRef]
- Zhang, X.X.; Ou, X.Q.; Zhou, Z.Y.; Ma, L.Y. Ochratoxin A in Chinese dried jujube: Method development and survey. Food Addit. Contam. Part A 2015, 32, 512–517. [Google Scholar] [CrossRef]
- Petzinger, E.; Ziegler, K. Ochratoxin A from a toxicological perspective. J. Vet. Pharmacol. Ther. 2000, 23, 91–98. [Google Scholar] [CrossRef]
- Althali, N.J.; Hassan, A.M.; Abdel-Wahhab, M.A. Effect of grape seed extract on maternal toxicity and in utero development in mice treated with zearalenone. Environ. Sci. Pollut. Res. 2019, 26, 5990–5999. [Google Scholar] [CrossRef]
- Placinta, C.M.; D’Mello, J.P.F.; Macdonald, A.M.C. A review of worldwide contamination of cereal grains and animal feed with Fusarium mycotoxins. Anim. Feed Sci. Technol. 1999, 78, 21–37. [Google Scholar] [CrossRef]
- Zheng, J.J.; Zhang, Y.; Xu, W.T.; Luo, Y.B.; Mao, J.R.; Shen, X.L.; Yang, X.; Li, X.H.; Huang, K.L. Zinc protects HepG2 cells against the oxidative damage and DNA damage induced by ochratoxin A. Toxicol. Appl. Pharmacol. 2013, 268, 123–131. [Google Scholar] [CrossRef]
- Scudamore, K.A.; Patel, S. Survey for aflatoxins, ochratoxin A, zearalenone and fumonisins in maize imported into the United Kingdom. Food Addit. Contam. 2000, 17, 407–416. [Google Scholar] [CrossRef]
- Rodrigues, I.; Naehrer, K. A three-year survey on the worldwide occurrence of mycotoxins in feedstuffs and feed. Toxins 2012, 4, 663–675. [Google Scholar] [CrossRef]
- Misihairabgwi, J.M.; Ezekiel, C.N.; Sulyok, M.; Shephard, G.S.; Krska, R. Mycotoxin contamination of foods in Southern Africa: A 10-year review (2007–2016). Crit. Rev. Food Sci. Nutr. 2019, 59, 43–58. [Google Scholar] [CrossRef]
- Smith, M.-C.; Madec, S.; Coton, E.; Hymery, N. Natural co-occurrence of mycotoxins in foods and feeds and their in vitro combined toxicological effects. Toxins 2016, 8, 94. [Google Scholar] [CrossRef] [PubMed]
- Rivas-San Vicente, M.; Larios-Zarate, G.; Plasencia, J. Disruption of sphingolipid biosynthesis in Nicotiana benthamiana activates salicylic acid-dependent responses and compromises resistance to Alternaria alternata f. sp lycopersici. Planta 2013, 237, 121–136. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhu, J.J. Review of development of electron accelerators on foods. Food Mach. 2013, 29, 236–239+258. [Google Scholar]
- Xue, P.Y.; Zhao, Y.; Wen, C.R.; Cheng, S.; Lin, S.Y. Effects of electron beam irradiation on physicochemical properties of corn flour and improvement of the gelatinization inhibition. Food Chem. 2017, 233, 467–475. [Google Scholar] [CrossRef]
- Abdellah, Z.; Soriano, J.M.; Molto, J.C.; Manes, J. Review on the toxicity, occurrence, metabolism, detoxification, regulations and intake of zearalenone: An oestrogenic mycotoxin. Food Chem. Toxicol. 2007, 45, 1–18. [Google Scholar] [CrossRef]
- Escriva, L.; Font, G.; Manyes, L. In vivo toxicity studies of fusarium mycotoxins in the last decade: A review. Food Chem. Toxicol. 2015, 78, 185–206. [Google Scholar] [CrossRef]
- Luo, X.H.; Qi, L.J.; Liu, Y.T.; Wang, R.; Yang, D.; Li, K.; Wang, L.; Li, Y.N.; Zhang, Y.W.; Chen, Z.X. Effects of electron beam irradiation on zearalenone and ochratoxin A in naturally contaminated corn and corn quality parameters. Toxins 2017, 9, 84. [Google Scholar] [CrossRef]
- National Health and Family Planning Commission of the people’s Republic of China (NHFPCRC). Test of Zearalenone in Foods; GB 5009.209–2016 [S]; NHFPCRC: Beijing, China, 2016.
- National Health and Family Planning Commission of the people’s Republic of China (NHFPCRC). Test of Ochratoxin A in Foods; GB 5009.96–2016 [S]; NHFPCRC: Beijing, China, 2016.
- General Administration of Quality Supervision (GAQS). Test of Amylose Content in Rice; GB/T 15683–2008 [S]; GAQS: Beijing, China, 2008.
- National Health and Family Planning Commission of the people’s Republic of China (NHFPCRC). Test of Starch in Foods; GB/T 5009.9–2016 [S]; NHFPCRC: Beijing, China, 2016.
- Galuszka, A.; Konieczka, P.; Migaszewski, Z.M.; Namiesnik, J. Analytical Eco-Scale for assessing the greenness of analytical procedures. TrAC Trends Anal. Chem. 2012, 37, 61–72. [Google Scholar] [CrossRef]
- Plotka-Wasylka, J. A new tool for the evaluation of the analytical procedure: Green analytical procedure index. Talanta 2018, 181, 204–209. [Google Scholar] [CrossRef] [PubMed]
- Zeng, S.X.; Chen, B.Y.; Zeng, H.L.; Guo, Z.B.; Lu, X.; Zhang, Y.; Zheng, B.D. Effect of microwave irradiation on the physicochemical and digestive properties of lotus seed starch. J. Agric. Food Chem. 2016, 64, 2442–2449. [Google Scholar] [CrossRef] [PubMed]
- Nemtanu, M.R.; Brasoveanu, M. Degradation of amylose by ionizing radiation processing. Starch-Starke 2017, 69. [Google Scholar] [CrossRef]
- Oneh Abu, J.; Gyebi Duodu, K.; Minnaar, A. Effect of gamma-irradiation on some physicochemical and thermal properties of cowpea (Vigna unguiculata L. Walp) starch. Food Chem. 2006, 95, 386–393. [Google Scholar] [CrossRef]
- Chung, H.J.; Liu, Q. Molecular structure and physicochemical properties of potato and bean starches as affected by gamma-irradiation. Int. J. Biol. Macromol. 2010, 47, 214–222. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.J.; Huang, M.; Yin, S.W.; Zi, M.; Wen, Q.B. Effects of electron-beam irradiation on physicochemical properties of starches separated from stored wheat. Starch-Starke 2011, 63, 121–127. [Google Scholar] [CrossRef]
- Sokhey, A.S.; Chinnaswamy, R. Physicochemical properties of irradiation modified starch extrudates. Food Struct. 1992, 11, 361–371. [Google Scholar]
- Chung, K.-H.; Othman, Z.; Lee, J.-S. Gamma irradiation of corn starches with different amylose-to-amylopectin ratio. J. Food Sci. Technol. Mysore 2015, 52, 6218–6229. [Google Scholar] [CrossRef][Green Version]
- Li, C.; Yang, W.G.; Xu, D.L.; Ou, C.R.; Shi, H.D. Effect of electron beam irradiation on nutritional ingredient of Tegilarca granosa meat. Acta Agric. Nucleatae Sin. 2011, 25, 959–964. [Google Scholar]
- Feng, X.; Lee, E.J.; Nam, K.; Jo, C.; Ko, K.; Ahn, D.U. Mechanisms of volatile production from amino acid esters by irradiation. Food Res. Int. 2016, 81, 100–107. [Google Scholar] [CrossRef]
- Hooshmand, H.; Klopfenstein, C.F. Effects of gamma-irradiation on mycotoxin disappearance and amino-acid contents of corn, wheat, and soybeans with different moisture contents. Plant Foods Hum. Nutr. 1995, 47, 227–238. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.; Zhang, Y.; Yin, S.; Jia, Z.; Shan, A. Biochemical changes and oxidative stress induced by zearalenone in the liver of pregnant rats. Hum. Exp. Toxicol. 2015, 34, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Conkova, E.; Laciakova, A.; Pastorova, B.; Seidel, H.; Kovac, G. The effect of zearalenone on some enzymatic parameters in rabbits. Toxicol. Lett. 2001, 121, 145–149. [Google Scholar] [CrossRef]
- Luzardo, O.P.; del Mar Bernal-Suarez, M.; Camacho, M.; Alberto Henriquez-Hernandez, L.; Boada, L.D.; Rial-Berriel, C.; Almeida-Gonzalez, M.; Zumbado, M.; Diaz-Diaz, R. Estimated exposure to EU regulated mycotoxins and risk characterization of aflatoxin-induced hepatic toxicity through the consumption of the toasted cereal flour called “gofio”, a traditional food of the Canary Islands (Spain). Food Chem. Toxicol. 2016, 93, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Xiao, Z.H.; Liu, M.; Zhang, N.Y.; Mohamed Khalil, M.; Gu, C.Q.; Qi, D.S.; Sun, L.H. Dietary silymarin supplementation alleviates zearalenone-induced hepatotoxicity and reproductive toxicity in rats. J. Nutr. 2018, 148, 1209–1216. [Google Scholar] [CrossRef]
- Gan, F.; Zhou, Y.J.; Qian, G.; Huang, D.; Hou, L.L.; Liu, D.D.; Chen, X.X.; Wang, T.; Jiang, P.; Lei, X.G.; et al. PCV2 infection aggravates ochratoxin A-induced nephrotoxicity via autophagy involving p38 signaling pathway in vivo and in vitro. Environ. Pollut. 2018, 238, 656–662. [Google Scholar] [CrossRef]
- Stoev, S.D.; Stoeva, J.K.; Anguelov, G.; Hald, B.; Creppy, E.E.; Radic, B. Haematological, biochemical and toxicological investigations in spontaneous cases with different frequency of porcine nephropathy in Bulgaria. J. Vet. Med. Ser. Physiol. Pathol. Clin. Med. 1998, 45, 229–236. [Google Scholar] [CrossRef]
- Liang, Z.S.; Ma, Y.J.; Liu, C.Y.; Deng, X.B.; Fan, X.L.; Yan, H.K.; Hu, Q.X. In vivotoxicity of zearalenone on liver and kidney in mice. Chin. J. Vet. Sci. 2010, 30, 673–676. [Google Scholar]

| Groups | Treatment Conditions | ZEN Content in Corn (μg/kg) | OTA Content in Corn (μg/kg) | ZEN Content in Fodders (μg/kg) | OTA Content in Fodders (μg/kg) |
|---|---|---|---|---|---|
| NC | Non-contaminated corn | - | - | - | - |
| CC | Contaminated corn | 2875.79 ± 78.12 | 65.04 ± 8.39 | 1366.90 ± 39.06 | 30.89 ± 4.19 |
| ENC | EBI treatment of non-contaminated corn | - | - | - | - |
| ECC | 50 kGy EBI treatment of contaminated corn | 815.62 ± 38.40 | 19.44 ± 4.65 | 346.64 ± 17.34 | 8.26 ± 0.35 |
| Doses (kGy) | 5 | 10 | 20 | 30 | 50 |
|---|---|---|---|---|---|
| Amylose (%) | 22.28 ± 0.16 a | 23.28 ± 0.32 a | 16.86 ± 0.88 b | 14.11 ± 0.16 bc | 10.19 ± 0.88 d |
| Sample | Control | 10 kGy | 30 kGy | 50 kGy |
|---|---|---|---|---|
| Crystallinity (%) | 10.9 ± 1.4 a | 10.0 ± 1.2 a | 8.2 ± 1.1 b | 7.6 ± 0.8 b |
| Amino Acid (g/100 g) | Control | 5 kGy | 10 kGy | 20 kGy | 30 kGy | 50 kGy |
|---|---|---|---|---|---|---|
| Asp | 0.88 ± 0.13 a,b | 0.81 ± 0.07a,b | 0.90 ± 0.05 a | 0.80 ± 0.03 b | 0.66 ± 0.02 c | 0.67 ± 0.01 c |
| Glu | 1.91 ± 0.16 a | 1.72 ± 0.12 a,b | 1.69 ± 0.15 a,b | 1.72 ± 0.15 a,b | 1.58 ± 0.11 b | 1.69 ± 0.13 a,b |
| Ser | 0.32 ± 0.01 a | 0.26 ± 0.01 b,c | 0.24 ± 0.02 c | 0.21 ± 0.01 c | 0.24 ± 0.02 c | 0.28 ± 0.01 b |
| His | 0.29 ± 0.01 a | 0.29 ± 0.01 a | 0.23 ± 0.02 b | 0.29 ± 0.02 a | 0.20 ± 0.02 b | 0.24 ± 0.01 b |
| Gly | 0.35 ± 0.02 b | 0.33 ± 0.02 b | 0.35 ± 0.04 a,b | 0.35 ± 0.01 b | 0.33 ± 0.01 b | 0.40 ± 0.02 a |
| Thr | 0.31 ± 0.01 a,b | 0.32 ± 0.01 a | 0.27 ± 0.03 b | 0.22 ± 0.01 c | 0.23 ± 0.01 b,c | 0.17 ± 0.02 d |
| Arg | 0.96 ± 0.12 a | 0.94 ± 0.03 a | 0.87 ± 0.04 a | 0.84 ± 0.03 a,b | 0.79 ± 0.02 b | 0.85 ± 0.03 a |
| Ala | 0.62 ± 0.11 a,b | 0.70 ± 0.03 a | 0.60 ± 0.03 b | 0.59 ± 0.02 b | 0.56 ± 0.01 b | 0.54 ± 0.04 b |
| Tyr | 0.37 ± 0.02 a | 0.36 ± 0.03 a,b | 0.34 ± 0.01 a,b | 0.35 ± 0.03 a,b | 0.32 ± 0.01 b | 0.32 ± 0.01 b |
| Cys | 0.16 ± 0.04 a | 0.14 ± 0.03 a | 0.16 ± 0.04 a | 0.10 ± 0.02 a | 0.13 ± 0.02 a | 0.13 ± 0.04 a |
| Val | 0.45 ± 0.02 b | 0.33 ± 0.03 c | 0.32 ± 0.01 c | 0.34 ± 0.01 c | 0.45 ± 0.03 b | 0.51 ± 0.02 a |
| Met | 0.16 ± 0.01 a | 0.11 ± 0.02 b | 0.17 ± 0.01 a | 0.14 ± 0.02 a,b | 0.12 ± 0.02 b | 0.11 ± 0.01 b |
| Phe | 0.41 ± 0.03 a | 0.45 ± 0.03 a | 0.40 ± 0.02 a | 0.33 ± 0.01 b | 0.30 ± 0.02 b,c | 0.29 ± 0.02 c |
| Ile | 0.33 ± 0.02 a | 0.25 ± 0.01 b | 0.24 ± 0.01 b | 0.23 ± 0.02 b | 0.25 ± 0.04 b | 0.30 ± 0.02 a,b |
| Leu | 0.96 ± 0.07 a | 0.72 ± 0.03 b | 0.70 ± 0.02 b | 0.70 ± 0.02 b | 0.63 ± 0.02 c | 0.64 ± 0.05 b,c |
| Lys | 0.32 ± 0.04 a | 0.28 ± 0.01 a | 0.21 ± 0.02 b | 0.22 ± 0.02 b | 0.23 ± 0.02 b | 0.14 ± 0.02 c |
| Pro | 0.53 ± 0.05 a | 0.48 ± 0.01 a | 0.46 ± 0.03 a | 0.47 ± 0.03 a | 0.40 ± 0.02 b | 0.37 ± 0.01 b |
| EAA | 2.92 ± 0.06 a | 2.46 ± 0.09 b | 2.31 ± 0.04 c | 2.19 ± 0.05 d | 2.20 ± 0.05 d | 2.16 ± 0.01 d |
| TAA | 9.31 ± 0.08 a | 8.49 ± 0.07 b | 8.15 ± 0.05 c | 7.89 ± 0.09 d | 7.42 ± 0.02 f | 7.64 ± 0.02 e |
| EAA/TAA | 0.31 ± 0.04 a | 0.29 ± 0.05 a | 0.28 ± 0.07 a | 0.28 ± 0.10 a | 0.30 ± 0.01 a | 0.28 ± 0.08 a |
| Weekly Age | 5 (g) | 6 (g) | 7 (g) | 8 (g) | 9 (g) | 10 (g) |
|---|---|---|---|---|---|---|
| NC | 28.5 ± 2.7 a | 30.4 ± 2.6 a | 32.3 ± 2.7 a | 34.3 ± 2.6 a | 36.2 ± 2.6 a | 38.2 ± 2.5 a |
| CC | 28.4 ± 2.6 a | 30.1 ± 2.5 a | 31.7 ± 2.4 a | 33.2 ± 2.5 a | 34.5 ± 2.2 a | 36.0 ± 2.2 a |
| ENC | 29.4 ± 2.0 a | 31.0 ± 2.0 a | 32.6 ± 1.9 a | 34.1 ± 1.9 a | 35.6 ± 1.9 a | 37.3 ± 1.8 a |
| ECC | 29.5 ± 1.8 a | 31.1 ± 1.8 a | 32.6 ± 1.8 a | 34.2 ± 1.7 a | 35.7 ± 1.7 a | 37.4 ± 1.7 a |
| Groups | Weight of Liver (g) | Weight of Kidney (g) | Weight of Spleen (g) | Weight of Testis (g) |
|---|---|---|---|---|
| NC | 1.65 ± 0.18 a | 0.61 ± 0.05 a | 0.10 ± 0.02 a | 0.30 ± 0.03 a |
| CC | 1.73 ± 0.24 a | 0.60 ± 0.06 a | 0.11 ± 0.03 a | 0.26 ± 0.04 a |
| ENC | 1.68 ± 0.19 a | 0.64 ± 0.08 a | 0.12 ± 0.02 a | 0.30 ± 0.04 a |
| ECC | 1.80 ± 0.17 a | 0.67 ± 0.07 a | 0.13 ± 0.02 a | 0.29 ± 0.05 a |
| Groups | Liver Index (%) | Kidney Index (%) | Spleen Index (%) | Testis Index (%) |
|---|---|---|---|---|
| NC | 4.33 ± 0.37 a | 1.60 ± 0.12 a | 0.27 ± 0.04 a | 0.80 ± 0.10 a |
| CC | 4.95 ± 0.66 a | 1.74 ± 0.20 a | 0.33 ± 0.09 a | 0.74 ± 0.09 a |
| ENC | 4.51 ± 0.49 a | 1.72 ± 0.18 a | 0.33 ± 0.06 a | 0.80 ± 0.14 a |
| ECC | 4.83 ± 0.44 a | 1.80 ± 0.19 a | 0.36 ± 0.07 a | 0.77 ± 0.14 a |
| - | Indexes | ||||||
|---|---|---|---|---|---|---|---|
| Groups | ALT | AST | TBIL | TP | ALB | BUN | SCr |
| - | (IU/L) | (IU/L) | (μmol/L) | (g/L) | (g/L) | (mmol/L) | (μmol/L) |
| NC | 22 ± 2 c | 86 ± 5 c | 2.88 ± 0.40 b | 62.6 ± 3.5 a | 38.0 ± 2.8 a | 7.6 ± 0.3 b | 14.4 ± 0.8 b |
| CC | 66 ± 15 a | 172 ± 32 a | 4.76 ± 0.46 a | 46.0 ± 4.8 c | 23.2 ± 6.3 c | 9.4 ± 1.2 a | 20.4 ± 2.5 a |
| ENC | 19 ± 4 c | 110 ± 5 b | 2.56 ± 0.35 b | 56.0 ± 0.8 b | 33.0 ± 1.8 b | 7.1 ± 0.2 b,c | 14.8 ± 0.6 b |
| ECC | 30 ± 3 b | 134 ± 18 a | 2.56 ± 0.26 b | 59.9 ± 2.8 a | 36.1 ± 1.5 a,b | 6.8 ± 0.1 c | 15.7 ± 0.8 b |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luo, X.; Zhai, Y.; Qi, L.; Pan, L.; Wang, J.; Xing, J.; Wang, R.; Wang, L.; Zhang, Q.; Yang, K.; et al. Influences of Electron Beam Irradiation on the Physical and Chemical Properties of Zearalenone- and Ochratoxin A-Contaminated Corn and In Vivo Toxicity Assessment. Foods 2020, 9, 376. https://doi.org/10.3390/foods9030376
Luo X, Zhai Y, Qi L, Pan L, Wang J, Xing J, Wang R, Wang L, Zhang Q, Yang K, et al. Influences of Electron Beam Irradiation on the Physical and Chemical Properties of Zearalenone- and Ochratoxin A-Contaminated Corn and In Vivo Toxicity Assessment. Foods. 2020; 9(3):376. https://doi.org/10.3390/foods9030376
Chicago/Turabian StyleLuo, Xiaohu, Yuheng Zhai, Lijun Qi, Lihong Pan, Jing Wang, Jiali Xing, Ren Wang, Li Wang, Qingchuan Zhang, Kai Yang, and et al. 2020. "Influences of Electron Beam Irradiation on the Physical and Chemical Properties of Zearalenone- and Ochratoxin A-Contaminated Corn and In Vivo Toxicity Assessment" Foods 9, no. 3: 376. https://doi.org/10.3390/foods9030376
APA StyleLuo, X., Zhai, Y., Qi, L., Pan, L., Wang, J., Xing, J., Wang, R., Wang, L., Zhang, Q., Yang, K., & Chen, Z. (2020). Influences of Electron Beam Irradiation on the Physical and Chemical Properties of Zearalenone- and Ochratoxin A-Contaminated Corn and In Vivo Toxicity Assessment. Foods, 9(3), 376. https://doi.org/10.3390/foods9030376









