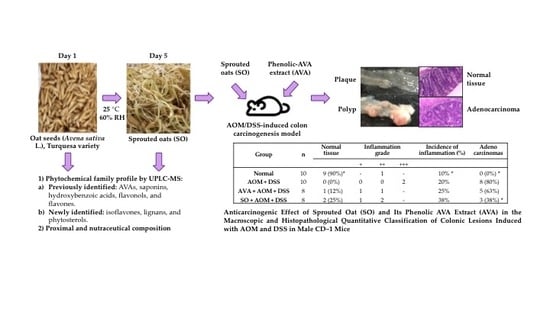
Chemopreventive Effect of the Germinated Oat and Its Phenolic-AVA Extract in Azoxymethane/Dextran Sulfate Sodium (AOM/DSS) Model of Colon Carcinogenesis in Mice
Abstract
1. Introduction
2. Materials and Methods
2.1. Oat Seed Germination
2.2. Chemical Analysis (Proximate Analysis) and Phytic Acid Content
2.3. Methanolic Extraction and Quantification of Total Phenolic Compounds (TPCs)
2.4. Abundance of Phenolic Compounds by Ultra–Performance Liquid Chromatograph (UPLC)–Mass Spectrometre (MS)
2.5. Animals and Treatments
2.6. Assay of β-Glucuronidase Activity (β-GA) and pH in the Cecal, Colonic, and Fecal Contents
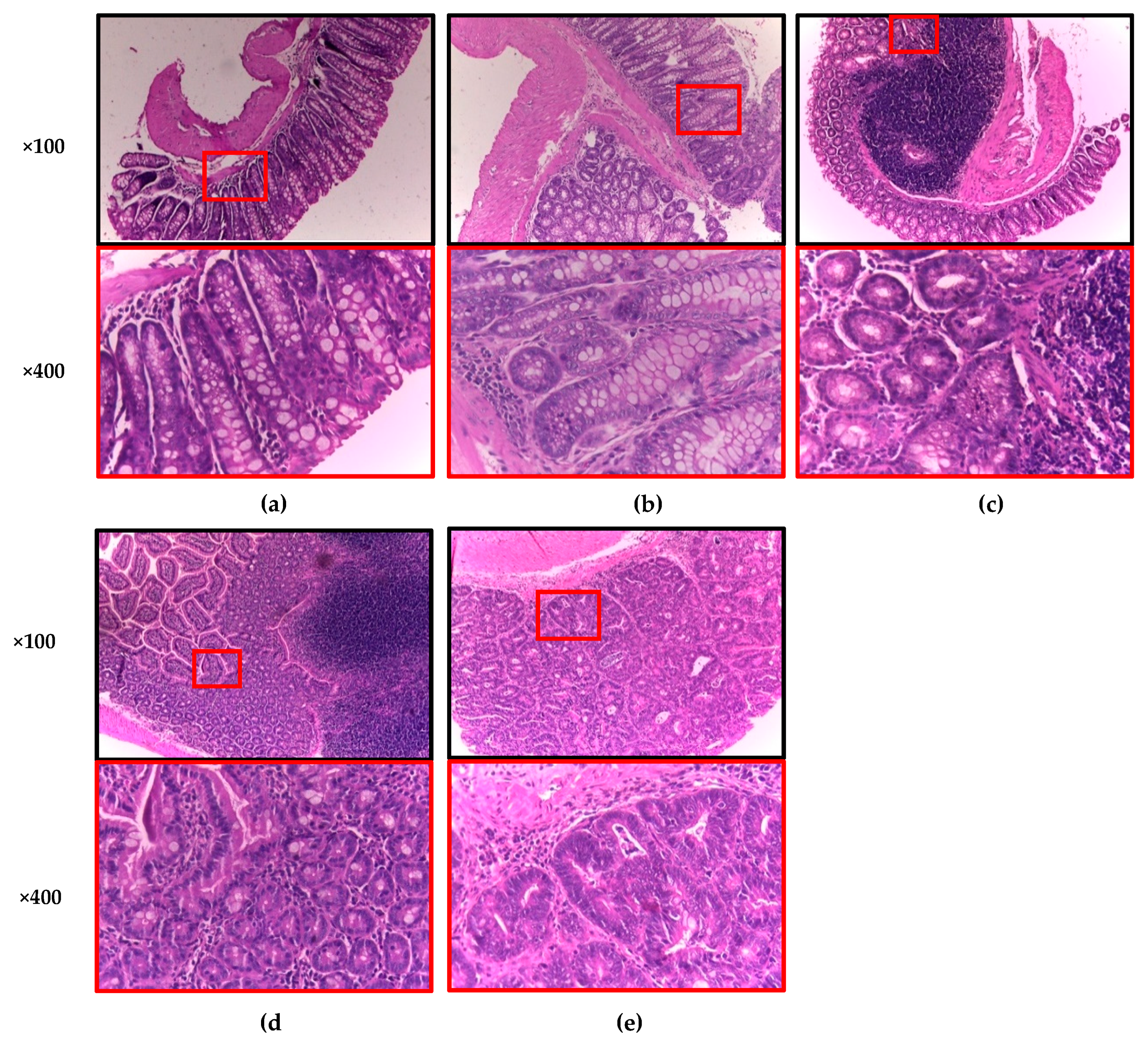
2.7. Macroscopic and Histopathology Analyses
2.8. Erythrocyte-Reduced Glutathione (GSH) Level
2.9. Phase 2 Enzyme Assays
2.10. Statistical Analysis
3. Results
3.1. Characterization of Chemical Composition and Nutraceutical Compounds of Oat Seeds after the Germination Process
3.2. Phytochemical Profile Induced after Germination of Oat Seeds at 25 °C/60% RH
3.3. Effect of Sprouted Oat (SO) and Its Phenolic AVA Extract (AVA) on Body Weight and Intestinal Parameters in CD–1 Mice Induced with AOM and DSS
3.4. Anticarcinogenic Effect of Sprouted Oat (SO) and Its Phenolic AVA Extract (AVA) on the Macroscopic and Histopathological Quantitative Classification of Colonic Lesions Induced with AOM and DSS in Male CD–1 Mice
3.5. Antioxidant Effects of Sprouted Oat (SO) and Its Phenolic AVA Extract (AVA) in the AOM/DSS Model
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ACF | Aberrant crypt foci |
| AOM | Azoxymethane |
| AVA | Avenanthramides |
| β–GA | β–glucuronidase activity |
| CRC | Colorectal cancer |
| DMH | 1,2–Dimethylhydrazine |
| DSS | Dextran sulfate sodium |
| GSH | Glutathione reduced |
| GST | Glutathione S–transferase |
| H&E | Hematoxylin and eosin |
| NQO1 | NADPH:Quinone oxidoreductase 1 |
| RH | Relative humidity |
| SO | Sprouted oat |
| TPC | Total phenolic compounds |
References
- Arnold, M.; Sierra, M.S.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global patterns and trends in colorectal cancer incidence and mortality. Gut 2017, 66, 683–691. [Google Scholar] [CrossRef]
- Dulal, S.; Keku, T.O. Gut microbiome and colorectal adenomas. Cancer J. 2014, 20, 225–231. [Google Scholar] [CrossRef]
- Marshall, J.R. Prevention of colorectal cancer: Diet, chemoprevention, and lifestyle. Gastroenterol. Clin. N. Am. 2008, 37, 73–82. [Google Scholar] [CrossRef]
- Cappellani, A.; Zanghì, A.; Di Vita, M.; Cavallaro, A.; Piccolo, G.; Veroux, P.; Lo Menzo, E.; Cavallaro, V.; de Paoli, P.; Veroux, M.; et al. Strong correlation between diet and development of colorectal cancer. Front. Biosci. 2013, 18, 190–198. [Google Scholar] [CrossRef]
- Kasdagly, M.; Radhakrishnan, S.; Reddivari, L.; Veeramachaneni, D.R.; Vanamala, J. Colon carcinogenesis: Influence of Western diet-induced obesity and targeting stem cells using dietary bioactive compounds. Nutrition 2014, 30, 1242–1256. [Google Scholar] [CrossRef]
- Murphy, J.K.; Marques-Lopes, I.; Sánchez-Tainta, A. Cereals and Legumes. In The Prevention of Cardiovascular Disease through the Mediterranean Diet, 1st ed.; Sánchez-Villegas, A., Sanchez-Taínta, A., Eds.; Academic Press: Waltham, MA, USA, 2018; pp. 111–132. [Google Scholar] [CrossRef]
- Arendt, E.K.; Zannini, E. Oats. In Woodhead Publishing Series in Food Science, Technology and Nutrition, Cereal Grains for the Food and Beverage Industries, 1st ed.; Arendt, E.K., Zannini, E., Eds.; Woodhead Publishing: Cambridge, UK, 2013; pp. 243–283. [Google Scholar] [CrossRef]
- Villaseñor, H.E.; Espitia, R.E.; Huerta, E.J.; Osorio, A.L.; López, H.J. Turquesa, nueva variedad de avena para la producción de grano y forraje en México. Agric. Técnica México 2009, 35, 487–492. [Google Scholar]
- Martínez-Villaluenga, C.; Peñas, E. Health benefits of oat: Current evidence and molecular mechanisms. Curr. Opin. Food Sci. 2017, 14, 26–31. [Google Scholar] [CrossRef]
- Shen, R.L.; Wang, Z.; Dong, J.L.; Xiang, Q.S.; Liu, Y.Q. Effects of oat soluble and insoluble β-glucan on 1,2-dimethylhydrazine-induced early colon carcinogenesis in mice. Food Agric. Immunol. 2016, 27, 657–666. [Google Scholar] [CrossRef]
- Liu, B.; Lin, Q.; Yang, T.; Zeng, L.; Shi, L.; Chen, Y.; Luo, F. Oat β-glucan ameliorates dextran sulfate sodium (DSS)-induced ulcerative colitis in mice. Food Funct. 2015, 6, 3454–3463. [Google Scholar] [CrossRef]
- Shahidi, F.; de Camargo, A.C. Tocopherols and tocotrienols in common and emerging dietary sources: Occurrence, applications, and health benefits. Int. J. Mol. Sci. 2016, 17, 1745. [Google Scholar] [CrossRef]
- Yang, J.; Ou, B.; Wise, M.L.; Chu, Y. In vitro total antioxidant capacity and anti-inflammatory activity of three common oat-derived avenanthramides. Food Chem. 2014, 160, 338–345. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Wang, L.; Wang, R.; Luo, X.; Li, Y.; Li, J.; Li, Y.; Chen, Z. Phenolic contents, cellular antioxidant activity and antiproliferative capacity of different varieties of oats. Food Chem. 2018, 239, 260–267. [Google Scholar] [CrossRef] [PubMed]
- Fu, R.; Yang, P.; Sajid, A.; Li, Z. Avenanthramide A Induces Cellular Senescence via miR-129-3p/Pirh2/p53 signaling pathway to suppress colon cancer growth. J. Agric. Food Chem. 2019, 67, 4808–4816. [Google Scholar] [CrossRef]
- Aborus, N.E.; Šaponjac, V.T.; Čanadanović-Brunet, J.; Ćetković, G.; Hidalgo, A.; Vulić, J.; Šeregelj, V. Sprouted and freeze-dried wheat and oat seeds–phytochemical profile and in vitro biological activities. Chem. Biodivers. 2018, 15, e1800119. [Google Scholar] [CrossRef]
- Kaukovirta-Norja, A.; Wilhelmson, A.; Poutanen, K. Germination: A means to improve the functionality of oat. Agric. Food Sci. 2004, 13, 100–112. [Google Scholar] [CrossRef]
- Dhakal, H.; Yang, E.J.; Lee, S.; Kim, M.J.; Baek, M.C.; Lee, B.; Park, P.H.; Kwon, T.K.; Khang, D.; Song, K.S.; et al. Avenanthramide C from germinated oats exhibits anti-allergic inflammatory effects in mast cells. Sci. Rep. 2019, 9, 6884. [Google Scholar] [CrossRef]
- Koenig, R.T.; Dickman, J.R.; Wise, M.L.; Ji, L.L. Avenanthramides are bioavailable and accumulate in hepatic, cardiac, and skeletal muscle tissue following oral gavage in rats. J. Agric. Food Chem. 2011, 59, 6438–6443. [Google Scholar] [CrossRef]
- Wang, P.; Chen, H.; Zhu, Y.; McBride, J.; Fu, J.; Sang, S. Oat avenanthramide-C (2c) is biotransformed by mice and the human microbiota into bioactive metabolites. J. Nutr. 2014, 145, 239–245. [Google Scholar] [CrossRef]
- Chen, C.Y.; Milbury, P.E.; Kwak, H.K.; Collins, F.W.; Samuel, P.; Blumberg, J.B. Avenanthramides and phenolic acids from oats are bioavailable and act synergistically with vitamin C to enhance hamster and human LDL resistance to oxidation. J. Nutr. 2004, 134, 1459–1466. [Google Scholar] [CrossRef]
- Chen, C.Y.; Milbury, P.E.; Collins, F.W.; Blumberg, J.B. Avenanthramides are bioavailable and have antioxidant activity in humans after acute consumption of an enriched mixture from oats. J. Nutr. 2007, 137, 1375–1382. [Google Scholar] [CrossRef]
- Fernaández-Ochoa, A.; Borraás-Linares, I.; Peárez-Saánchez, A.; Barrajoán-Catalaán, E.; Gonzaález-Aálvarez, I.; Arraáez-Romaán, D.; Micol, V.; Segura-Carretero, A. Phenolic compounds in rosemary as potential source of bioactive compounds against colorectal cancer: In situ absorption and metabolism study. J. Funct. Foods 2017, 33, 202–210. [Google Scholar] [CrossRef]
- Horwitz, W. AOAC International. In Official Methods of Analysis of AOAC International, 17th ed.; Horwitz, W., AOAC International, Eds.; AOAC International Seventeen: Gaithersburg, MD, USA, 2002. [Google Scholar]
- McCleary, B.V.; Codd, R. Measurement of (1→ 3), (1→ 4)-β-D-glucan in barley and oats: A streamlined enzymic procedure. J. Sci. Food Agric. 1991, 55, 303–312. [Google Scholar] [CrossRef]
- Latta, M.; Eskin, M. A simple and rapid colorimetric method for phytate determination. J. Agric. Food Chem. 1980, 28, 1313–1315. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Rodríguez-González, S.; Pérez-Ramírez, I.F.; Castaño-Tostado, E.; Amaya-Llano, S.; Rodríguez-García, M.E.; Reynoso-Camacho, R. Improvement of physico-chemical properties and phenolic compounds bioavailability by concentrating dietary fiber of peach (Prunus persica) juice by-product. J. Sci. Food Agric. 2018, 98, 3109–3118. [Google Scholar] [CrossRef]
- Jenab, M.; Thompson, L.U. The influence of flaxseed and lignans on colon carcinogenesis and β-glucuronidase activity. Carcinogenesis 1996, 17, 1343–1348. [Google Scholar] [CrossRef]
- Astler, V.B.; Coller, F.A. The prognostic significance of direct extension of carcinoma of the colon and rectum. Ann. Surg. 1954, 139, 846–852. [Google Scholar] [CrossRef]
- Riddell, R.H.; Goldman, H.; Ransohoff, D.F.; Appelman, H.D.; Fenoglio, C.M.; Haggitt, R.C.; Ahren, C.; Correa, P.; Hamilton, S.R.; Morson, B.C. Dysplasia in inflammatory bowel disease: Standardized classification with provisional clinical applications. Hum. Pathol. 1983, 14, 931–968. [Google Scholar] [CrossRef]
- Klintrup, K.; Mäkinen, J.M.; Kauppila, S.; Väre, P.O.; Melkko, J.; Tuominen, H.; Tuppurainen, K.; Mäkela, J.; Karttunen, T.J.; Mäkinen, M.J. Inflammation and prognosis in colorectal cancer. Eur. J. Cancer 2005, 41, 2645–2654. [Google Scholar] [CrossRef]
- Fleming, M.; Ravula, S.; Tatishchev, S.F.; Wang, H.L. Colorectal carcinoma: Pathologic aspects. J. Gastrointest. Oncol. 2012, 3, 153–173. [Google Scholar] [CrossRef]
- Marchal-Bressenot, A.; Riddell, R.H.; Boulagnon-Rombi, C.; Reinisch, W.; Danese, S.; Schreiber, S.; Peyrin-Biroulet, L. Review article: The histological assessment of disease activity in ulcerative colitis. Aliment. Pharmacol. Ther. 2015, 42, 957–967. [Google Scholar] [CrossRef]
- Ellman, G.L. Tissue sulfhydryl groups. Arch. Biochem. Biophys. 1959, 82, 70–77. [Google Scholar] [CrossRef]
- Habig, W.H.; Pabst, M.J.; Jakoby, W.B. Glutathione S-transferases. The first enzymatic step in mercapturic acid formation. J. Biol. Chem. 1974, 249, 7130–7139. [Google Scholar]
- Prochaska, H.J.; Santamaria, A.B.; Talalay, P. Rapid detection of inducers of enzymes that protect against carcinogens. Proc. Natl. Acad. Sci. USA 1992, 89, 2394–2398. [Google Scholar] [CrossRef]
- Valadez-Bustos, N.; Escamilla-Silva, E.M.; García-Vázquez, F.J.; Gallegos-Corona, M.A.; Amaya-Llano, S.L.; Ramos-Gómez, M. Oral administration of microencapsulated B. longum BAA-999 and lycopene modulates IGF-1/IGF-1R/IGFBP3 protein expressions in a colorectal murine model. Int. J. Mol. Sci. 2019, 20, 4275. [Google Scholar] [CrossRef]
- Tanaka, T.; Kohno, H.; Suzuki, R.; Yamada, Y.; Sugie, S.; Mori, H. A novel inflammation-related mouse colon carcinogenesis model induced by azoxymethane and dextran sodium sulfate. Cancer Sci. 2003, 94, 965–973. [Google Scholar] [CrossRef]
- Tanaka, T. Development of an inflammation-associated colorectal cancer model and its application for research on carcinogenesis and chemoprevention. Int. J. Inflam. 2012, 2012, 658786. [Google Scholar] [CrossRef]
- Neergheen, V.S.; Bahorun, T.; Taylor, E.W.; Jen, L.S.; Aruoma, O.I. Targeting specific cell signaling transduction pathways by dietary and medicinal phytochemicals in cancer chemoprevention. Toxicology 2010, 278, 229–241. [Google Scholar] [CrossRef]
- Aune, D. Plant Foods, antioxidant biomarkers, and the risk of cardiovascular disease, cancer, and mortality: A review of the evidence. Adv. Nutr. 2019, 10, S404–S421. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Morze, J.; Hoffmann, G. Mediterranean diet and health status: Active ingredients and pharmacological mechanisms. Br. J. Pharmacol. 2019, 1–17. [Google Scholar] [CrossRef]
- de Bruijn, W.J.; van Dinteren, S.; Gruppen, H.; Vincken, J.P. Mass spectrometric characterisation of avenanthramides and enhancing their production by germination of oat (Avena sativa). Food Chem. 2019, 277, 682–690. [Google Scholar] [CrossRef]
- Lee, J.H.; Lee, B.K.; Park, H.H.; Lee, B.W.; Woo, K.S.; Kim, H.J.; Han, S.I.; Lee, Y.Y. Oat germination and ultrafiltration process improves the polyphenol and avenanthramide contents with protective effect in oxidative-damaged HepG2 cells. J. Food Biochem. 2019, 43, e12799. [Google Scholar] [CrossRef]
- Leonova, S.; Grimberg, A.; Marttila, S.; Stymne, S.; Carlsson, A.S. Mobilization of lipid reserves during germination of oat (Avena sativa L.), a cereal rich in endosperm oil. J. Exp. Bot. 2010, 61, 3089–3099. [Google Scholar] [CrossRef]
- Lehtinen, P.; Kaukovirta-Norja, A. Oats: Chemistry and Technology. In Oat Lipids, Enzymes, and Quality; Webster, F.H., Wood, P.J., Eds.; AACC International: Saint Paul, MN, USA, 2011; pp. 143–156. [Google Scholar] [CrossRef]
- Menon, R.; Gonzalez, T.; Ferruzzi, M.; Jackson, E.; Winderl, D.; Watson, J. Oats-from farm to fork. Adv. Food. Nutr. Res. 2016, 77, 1–55. [Google Scholar] [CrossRef]
- Welch, R.W. Nutrient composition and nutritional quality of oats and comparisons with other cereals. In Oats: Chemistry and Technology; Webster, F.H., Wood, P.J., Eds.; AACC International: Saint Paul, MN, USA, 2011; pp. 95–107. [Google Scholar]
- Oksman-Caldentey, K.M.; Kaukovirta-Norja, A.; Heiniö, R.-L.; Kleemola, T.; Mikola, M.; Sontag-Strom, T.; Lehtinen, P.; Pihlava, J.M.; Poutanen, P. Kauran biotekninen prosessointi uusiksi elintarvikkeiksi. In Kau-Ran Biotekninen Prosessointi Uusiksi Elintarvikkeiksi (Biotechnical Processing of Oat for Novel Food Ingredients); Salovaara, H., Sontag-Strom, T., Eds.; EKT-Sarja 1221: Elintarviketeknologian Laitos, Finland, 2001; pp. 85–108. [Google Scholar]
- Silva, E.O.; Bracarense, A.P. Phytic acid: From antinutritional to multiple protection factor of organic systems. J. Food Sci. 2016, 81, R1357–R1362. [Google Scholar] [CrossRef]
- Boz, H. Phenolic amides (avenanthramides) in oats—A review. Czech J. Food Sci. 2015, 33, 399–404. [Google Scholar] [CrossRef]
- Collins, F.W. Oat phenolics: Avenanthramides, novel substituted N-cinnamoylanthranilate alkaloids from oat groats and hulls. J. Agric. Food Chem. 1989, 37, 60–66. [Google Scholar] [CrossRef]
- Emmons, C.L.; Peterson, D.M. Antioxidant activity and phenolic contents of oat groats and hulls. Cereal Chem. 1999, 76, 902–906. [Google Scholar] [CrossRef]
- Emmons, C.L.; Peterson, D.M. Antioxidant activity and phenolic content of oat as affected by cultivar and location. Crop Sci. 2001, 41, 1676–1681. [Google Scholar] [CrossRef]
- Dimberg, L.H.; Gissén, C.; Nilsson, J. Phenolic compounds in oat grains (Avena sativa L.) grown in conventional and organic systems. AMBIO J. Hum. Environ. 2005, 34, 331–337. [Google Scholar] [CrossRef]
- McMurrough, I.; Baert, T. Identification of proanthocyanidins in beer and their direct measurement with a dual electrode electrochemical detector. J. Inst. Brew. 1994, 100, 409–416. [Google Scholar] [CrossRef]
- Pecio, Ł.; Wawrzyniak-Szołkowska, A.; Oleszek, W.; Stochmal, A. Rapid analysis of avenacosides in grain and husks of oats by UPLC-TQ-MS. Food Chem. 2013, 141, 2300–2304. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T. Colorectal carcinogenesis: Review of human and experimental animal studies. J. Carcinog. 2009, 8, 5. [Google Scholar] [CrossRef]
- Perše, M.; Cerar, A. Morphological and molecular alterations in 1,2 dimethylhydrazine and azoxymethane induced colon carcinogenesis in rats. BioMed Res. Int. 2011, 2011, 473964. [Google Scholar] [CrossRef]
- Gkouskou, K.K.; Deligianni, C.; Tsatsanis, C.; Eliopoulos, A.G. The gut microbiota in mouse models of inflammatory bowel disease. Front. Cell. Infect. Microbiol. 2014, 4, 28. [Google Scholar] [CrossRef]
- Grimm, V.; Radulovic, K.; Riedel, C.U. Colonization of C57BL/6 Mice by a potential probiotic Bifidobacterium bifidum strain under germ-free and specific pathogen-free conditions and during experimental colitis. PLoS ONE 2015, 10, e0139935. [Google Scholar] [CrossRef]
- de Robertis, M.; Massi, E.; Poeta, M.L.; Carotti, S.; Morini, S.; Cecchetelli, L.; Signori, E.; Fazio, V.M. The AOM/DSS murine model for the study of colon carcinogenesis: From pathways to diagnosis and therapy studies. J. Carcinog. 2011, 10, 9. [Google Scholar] [CrossRef]
- Doulberis, M.; Angelopoulou, K.; Kaldrymidou, E.; Tsingotjidou, A.; Abas, Z.; Erdman, S.E.; Poutahidis, T. Cholera-toxin suppresses carcinogenesis in a mouse model of inflammation-driven sporadic colon cancer. Carcinogenesis 2015, 36, 280–290. [Google Scholar] [CrossRef]
- Suchecka, D.; Harasym, J.; Wilczak, J.; Gromadzka-Ostrowska, J. Hepato- and gastro-protective activity of purified oat 1-3, 1-4-β-d-glucans of different molecular weight. Int. J. Biol. Macromol. 2016, 91, 1177–1185. [Google Scholar] [CrossRef]
- Yang, J.; Wang, P.; Wu, W.; Zhao, Y.; Idehen, E.; Sang, S. Steroidal Saponins in oat bran. J. Agric. Food Chem. 2016, 64, 1549–1556. [Google Scholar] [CrossRef]
- Hazafa, A.; Rehman, K.U.; Jahan, N.; Jabeen, Z. The role of polyphenol (Flavonoids) compounds in the treatment of cancer cells. Nutr. Cancer 2019, 9, 1–12. [Google Scholar] [CrossRef]
- Turrini, E.; Maffei, F.; Milelli, A.; Calcabrini, C.; Fimognari, C. Overview of the anticancer profile of avenanthramides from oat. Int. J. Mol. Sci. 2019, 20, 4536. [Google Scholar] [CrossRef]
- Wang, H.C.; Hung, C.H.; Hsu, J.D.; Yang, M.Y.; Wang, S.J.; Wang, C.J. Inhibitory effect of whole oat on aberrant crypt foci formation and colon tumor growth in ICR and BALB/c mice. J. Cereal Sci. 2011, 53, 73–77. [Google Scholar] [CrossRef]
- Sankaranarayanan, R.; Valiveti, C.K.; Kumar, D.R.; van slambrouck, S.; Kesharwani, S.S.; Seefeldt, T.; Scaria, J.; Tummala, H.; Bhat, G.J. The flavonoid metabolite 2,4,6-trihydroxybenzoic acid is a CDK inhibitor and an anti-proliferative agent: A potential role in cancer prevention. Cancers 2019, 11, 427. [Google Scholar] [CrossRef]
- Cuellar-Nuñez, M.L.; Luzardo-Ocampo, I.; Campos-Vega, R.; Gallegos-Corona, M.A.; de Mejía, E.G.; Loarca-Piña, G. Physicochemical and nutraceutical properties of moringa (Moringa oleifera) leaves and their effects in an in vivo AOM/DSS-induced colorectal carcinogenesis model. Food Res. Int. 2018, 105, 159–168. [Google Scholar] [CrossRef]
- Cheng, C.M.; Chen, F.M.; Lu, Y.L.; Tzou, S.C.; Wang, J.Y.; Kao, C.H.; Liao, K.W.; Cheng, T.C.; Chuang, C.H.; Chen, B.M.; et al. Expression of β-glucuronidase on the surface of bacteria enhances activation of glucuronide prodrugs. Cancer Gene Ther. 2013, 20, 276–281. [Google Scholar] [CrossRef]
- Mandal, P. Potential biomarkers associated with oxidative stress for risk assessment of colorectal cancer. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2017, 390, 557–565. [Google Scholar] [CrossRef]
- Yamamoto, M.; Kensler, T.W.; Motohashi, H. The KEAP1-NRF2 system: A thiol-based sensor-effector apparatus for maintaining redox homeostasis. Physiol. Rev. 2018, 98, 1169–1203. [Google Scholar] [CrossRef]
- Pool-Zobel, B.; Veeriah, S.; Böhmer, F.D. Modulation of xenobiotic metabolising enzymes by anticarcinogens—Focus on glutathione S-transferases and their role as targets of dietary chemoprevention in colorectal carcinogenesis. Mutat. Res.-Fundam. Mol. Mech. Mutagenesis 2005, 591, 74–92. [Google Scholar] [CrossRef]
- Strzelczyk, J.K.; Wielkoszyński, T.; Krakowczyk, Ł.; Adamek, B.; Zalewska-Ziob, M.; Gawron, K.; Kasperczyk, J.; Wiczkowski, A. The activity of antioxidant enzymes in colorectal adenocarcinoma and corresponding normal mucosa. Acta Biochim. Pol. 2012, 59, 549–556. [Google Scholar] [CrossRef]
- Lippmann, D.; Lehmann, C.; Florian, S.; Barknowitz, G.; Haack, M.; Mewis, I.; Wiesner, M.; Schreiner, M.; Glatt, H.; Brigelius-Floh´e, R.; et al. Glucosinolates from pak choi and broccoli induce enzymes and inhibit inflammation and colon cancer differently. Food Funct. 2014, 5, 1073–1081. [Google Scholar] [CrossRef]
- Bobek, P.; Galbavý, Š.; Mariassyova, M. The effect of red beet (Beta vulgaris var. rubra) fiber on alimentary hypercholesterolemia and chemically induced colon carcinogenesis in rats. Nahrung 2000, 44, 184–187. [Google Scholar] [CrossRef]
- Aquilano, K.; Baldelli, S.; Ciriolo, M.R. Glutathione: New roles in redox signaling for an old antioxidant. Front. Pharmacol. 2014, 5, 196. [Google Scholar] [CrossRef]
- Matić, M.M.; Paunović, M.G.; Milošević, M.D.; Ognjanović, B.I.; Saičić, Z.S. Hematoprotective effects and antioxidant properties of β-glucan and vitamin C against acetaminophen-induced toxicity: An experimental study in rats. Drug Chem. Toxicol. 2019, 1–8. [Google Scholar] [CrossRef]
- Manju, V.; Balasubramaniyan, V.; Nalini, N. Rat colonic lipid peroxidation and antioxidant status: The effects of dietary luteolin on 1, 2-dimethylhydrazine challenge. Cell. Mol. Biol. Lett. 2005, 10, 535–551. [Google Scholar]
- Begleiter, A.; Sivananthan, K.; Lefas, G.M.; Maksymiuk, A.W.; Bird, R.P. Inhibition of colon carcinogenesis by post-initiation induction of NQO1 in Sprague-Dawley rats. Oncol. Rep. 2009, 21, 1559–1565. [Google Scholar] [CrossRef]
- Wu, Z.; Ming, J.; Gao, R.; Wang, Y.; Liang, Q.; Yu, H.; Zhao, G. Characterization and antioxidant activity of the complex of tea polyphenols and oat β-glucan. J. Agric. Food Chem. 2011, 59, 10737–10746. [Google Scholar] [CrossRef]
- Ramos-Gomez, M.; Kwak, M.K.; Dolan, P.M.; Itoh, K.; Yamamoto, M.; Talalay, P.; Kensler, T.W. Sensitivity to carcinogenesis is increased and chemoprotective efficacy of enzyme inducers is lost in nrf2 transcription factor-deficient mice. Proc. Natl. Acad. Sci. USA 2001, 98, 3410–3415. [Google Scholar] [CrossRef]
- Murota, K.; Nakamura, Y.; Uehara, M. Flavonoid metabolism: The interaction of metabolites and gut microbiota. Biosci. Biotechnol. Biochem. 2018, 82, 600–610. [Google Scholar] [CrossRef]
- Wang, Y.; Huo, Y.; Zhao, L.; Lu, F.; Wang, O.; Yang, X.; Ji, B.; Zhou, F. Cyanidin-3-glucoside and its phenolic acid metabolites attenuate visible light-induced retinal degeneration in vivo via activation of Nrf2/HO-1 pathway and NF-κB suppression. Mol. Nutr. Food Res. 2016, 60, 1564–1577. [Google Scholar] [CrossRef]
- Yang, S.Y.; Pyo, M.C.; Nam, M.H.; Lee, K.W. ERK/Nrf2 pathway activation by caffeic acid in HepG2 cells alleviates its hepatocellular damage caused by t-butylhydroperoxide-induced oxidative stress. BMC Complement. Altern. Med. 2019, 19, 139. [Google Scholar] [CrossRef] [PubMed]
- Latifah, S.Y.; Armania, N.; Tze, T.H.; Azhar, Y.; Nordiana, A.H.; Norazalina, S.; Hairuszah, I.; Saidi, M.; Maznah, I. Germinated brown rice (GBR) reduces the incidence of aberrant crypt foci with the involvement of beta-catenin and COX-2 in azoxymethane-induced colon cancer in rats. Nutr. J. 2010, 26, 9–16. [Google Scholar] [CrossRef]
- Saki, E.; Saiful-Yazan, L.; Mohd-Ali, R.; Ahmad, Z. Chemopreventive effects of germinated rough rice crude extract in inhibiting azoxymethane-induced aberrant crypt foci formation in Sprague-Dawley rats. BioMed Res. Int. 2017, 2017, 9517287. [Google Scholar] [CrossRef]
- Kanauchi, O.; Mitsuyama, K.; Andoh, A.; Iwanaga, T. Modulation of intestinal environment by prebiotic germinated barley foodstuff prevents chemo-induced colonic carcinogenesis in rats. Oncol. Rep. 2008, 20, 793–801. [Google Scholar] [CrossRef][Green Version]
- Komiyama, Y.; Mitsuyama, K.; Masuda, J.; Yamasaki, H.; Takedatsu, H.; Andoh, A.; Tsuruta, O.; Fukuda, M.; Kanauchi, O. Prebiotic treatment in experimental colitis reduces the risk of colitic cancer. J. Gastroenterol. Hepatol. 2011, 26, 1298–1308. [Google Scholar] [CrossRef]
- Mollah, M.L.; Park, D.K.; Park, H.J. Cordyceps militaris grown on germinated soybean induces G2/M cell cycle arrest through downregulation of cyclin B1 and Cdc25c in human colon cancer HT-29 Cells. Evid. Based Complement. Altern. Med. 2012, 2012, 249217. [Google Scholar] [CrossRef]
- González-Montoya, M.; Hernández-Ledesma, B.; Silván, J.M.; Mora-Escobedo, R.; Martínez-Villaluenga, C. Peptides derived from in vitro gastrointestinal digestion of germinated soybean proteins inhibit human colon cancer cells proliferation and inflammation. Food Chem. 2018, 1, 75–82. [Google Scholar] [CrossRef]
 ) Lymphocyte infiltration; (
) Lymphocyte infiltration; ( ) eosinophils; (
) eosinophils; ( ) goblet cells; (E) epithelial ridges; (M) defined muscularis; (m) mitosis; (N) necrosis; (0,1,2) inflammation grade; and (Ad) adenocarcinoma.
) goblet cells; (E) epithelial ridges; (M) defined muscularis; (m) mitosis; (N) necrosis; (0,1,2) inflammation grade; and (Ad) adenocarcinoma.
 ) Lymphocyte infiltration; (
) Lymphocyte infiltration; ( ) eosinophils; (
) eosinophils; ( ) goblet cells; (E) epithelial ridges; (M) defined muscularis; (m) mitosis; (N) necrosis; (0,1,2) inflammation grade; and (Ad) adenocarcinoma.
) goblet cells; (E) epithelial ridges; (M) defined muscularis; (m) mitosis; (N) necrosis; (0,1,2) inflammation grade; and (Ad) adenocarcinoma.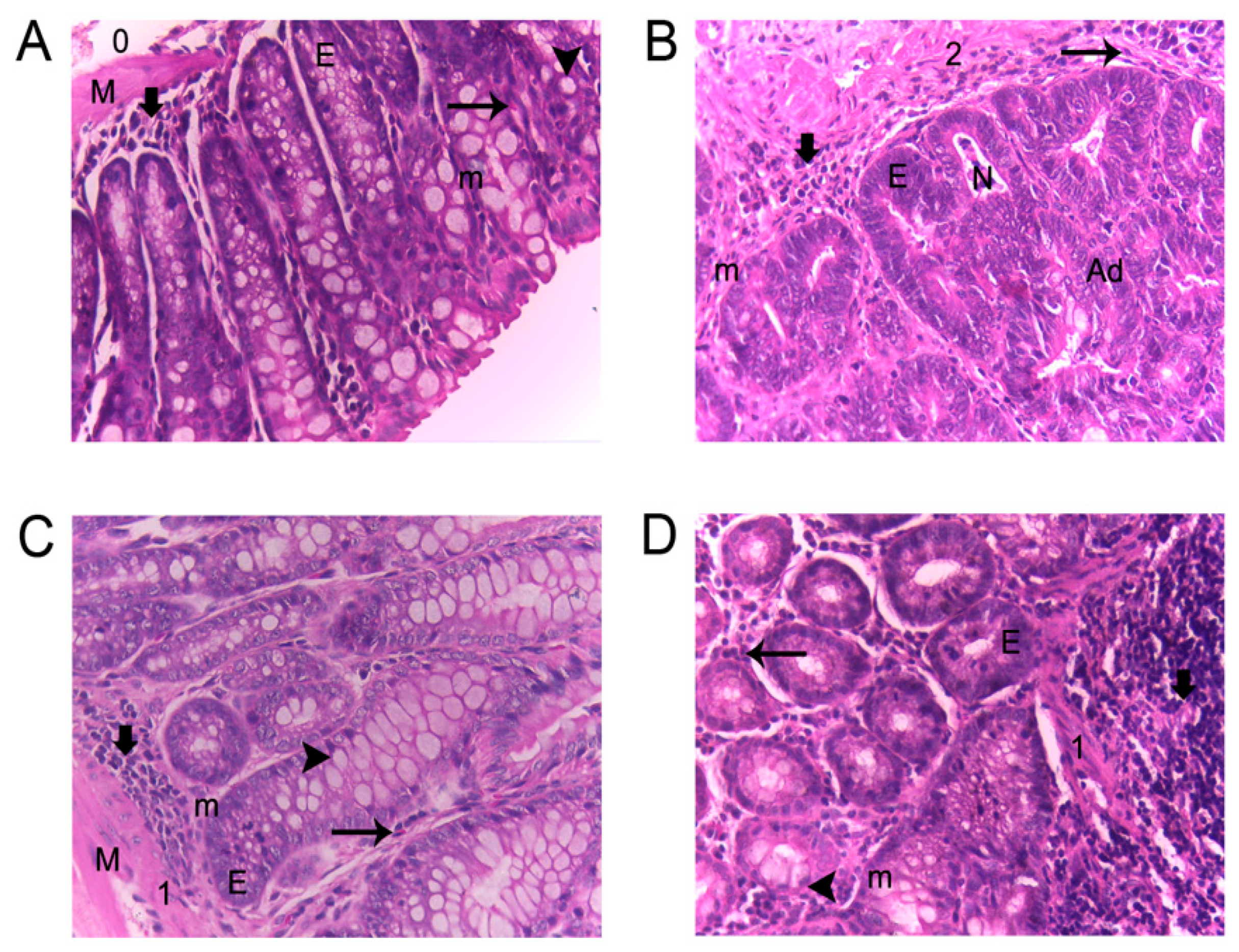
| Sample | Protein 1 | Carbohydrates 2 | Lipids 1 | Ash 1 | Moisture 1 |
|---|---|---|---|---|---|
| Oat seed | 8.83 ± 0.23 a | 73.37 ± 1.1 a | 4.41 ± 0.20 a | 3.99 ± 0.30 a | 9.4 ± 0.7 a |
| SO | 10.05 ± 0.30 b | 71.08 ± 0.7 b | 5.55 ± 0.01 b | 3.62 ± 0.10 b | 9.7 ± 0.2 a |
| Insoluble Fiber 1 | Soluble Fiber 1 | Total Fiber 1 | Phytic Acid 1 | TPC 3 | |
| Oat seed | 55.04 ± 1.89 a | 18.49 ± 0.74 a | 73.53 ± 2.51 a | 0.44 ± 0.0 a | 0.64 ± 0.01 a |
| SO | 30.87 ± 4.23 b | 2.28 ± 0.52 b | 33.15 ± 4.65 b | 0.04 ± 0.0 b | 2.79 ± 0.06 b |
| Body Weight | Cecum | Colon | Feces | |||||
|---|---|---|---|---|---|---|---|---|
| Group | Initial | Final | pH | β-GA 1 | pH | β-GA 1 | pH | β-GA 1 |
| Normal | 30.6 ± 1.01 a | 39.8 ± 1.01 a | 7.12 ± 0.04 b | 3.67 ± 0.13 c | 7.22 ± 0.07 a | 3.99 ± 0.09 d | 7.22 ± 0.13 b | 4.34 ± 0.17 b |
| AOM + DSS control | 30.0 ± 0.76 a | 41.8 ± 0.79 a | 7.29 ± 0.04 a,b | 5.28 ± 0.26 a | 7.33 ± 0.04 a | 7.02 ± 0.15 a | 7.77 ± 0.12 a | 5.70 ± 0.15 a |
| AVA + AOM + DSS | 28.8 ± 1.03 a | 40.1 ± 1.10 a | 7.46 ± 0.05 a | 3.93 ± 0.08 b,c | 7.31 ± 0.10 a | 6.17 ± 0.06 b | 7.67 ± 0.11 a | 5.87 ± 0.27 a |
| SO + AOM + DSS | 28.9 ± 0.88 a | 38.9 ± 1.03 a | 7.13 ± 0.07 b | 4.11 ± 0.19 b | 7.09 ± 0.12 a | 4.50 ± 0.06 c | 7.62 ± 0.22 a | 2.51 ± 0.20 c |
| Early Lesions (Flat-Type Lesions) 1 | Tumors (Protuberant-Type Lesions) 2 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Group | n | Incidence (%) | Mean Number 3 | Colon Distribution 4 | Incidence (%) | Mean Number 3 | Colon Distribution 4 | Classification 5 | |||||
| Proximal | Distal | Proximal | Distal | P | S | EX | EN | ||||||
| Normal | 10 | 10 * | 0.10 ± 0.10 b | 1 (100%) | 0 (0%) | 0 * | 0.00 ± 0.00 b | 0 (0%) | 0 (0%) | 0 | 0 | 0 | 0 |
| AOM + DSS | 10 | 60 | 1.00 ± 0.30 a,b | 0 (0%) | 10 (100%) | 80 | 4.20 ± 1.01 a | 0 (0%) | 42 (100%) | 14 | 16 | 2 | 10 |
| AVA + AOM + DSS | 8 | 100 | 2.13 ± 0.30 a | 0 (0%) | 17 (100%) | 50 | 0.63 ± 0.26 b | 1 (20%) | 4 (80%) | 4 | 1 | 0 | 0 |
| SO + AOM + DSS | 8 | 100 | 2.13 ± 0.61 a | 1 (6%) | 16 (94%) | 38 | 0.38 ± 0.18 b | 0 (0%) | 3 (100%) | 3 | 0 | 0 | 0 |
| Group | n | Incidence (%) | Lymphocyte Infiltration 1 | Eosinophils | Calceiform Cells 2 | Epithelial Ridges | Defined Muscularis | Necrosis | Mitosis | Inflammation Grade | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Normal Tissue | Inflammation | Adeno Carcinomas | L | M | H | Yes | No | F | M | I | Yes | No | Yes | No | Yes | No | + | ++ | +++ | |||
| Normal | 10 | 9 (90%) * | 1 (10%) * | 0 (0%) * | None | X | X | X | X | X | 5% | 1 | ||||||||||
| AOM + DSS | 10 | 0 (0%) | 2 (20%) | 8 (80%) | X | X | X | X | X | X | 20% | 0 | 0 | 2 | ||||||||
| AVA + AOM + DSS | 8 | 1 (12%) | 2 (25%) | 5 (63%) | X | X | X | X | Focal | X | 15% | 1 | 1 | |||||||||
| SO + AOM + DSS | 8 | 2 (25%) | 3 (38%) | 3 (38%) * | X | X | X | X | X | X | 17% | 1 | 2 | |||||||||
| Erythrocyte | Liver | Colon | ||||
|---|---|---|---|---|---|---|
| Group | n | GSH 1 | GST 2 | NQO1 2 | GST 2 | NQO1 2 |
| Normal | 10 | 1.40 ± 0.10 b | 411.5 ± 7.7 a | 36.2 ± 1.0 a,b | 60.6 ± 3.0 a | 54.6 ± 2.2 ab |
| AOM + DSS | 10 | 2.81 ± 0.43 a | 438.6 ± 12.3 b | 38.6 ± 0.9 a | 99.2 ± 6.6 b | 88.7 ± 2.3 c |
| AVA + AOM + DSS | 8 | 1.39 ± 0.15 b | 417.7 ± 10.0 a | 33.4 ± 0.8 b | 61.9 ± 2.1 a | 47.5 ± 3.3 a |
| SO + AOM + DSS | 8 | 1.34 ± 0.24 b | 506.3 ± 2.7 c | 58.7 ± 1.8 c | 55.8 ± 1.6 a | 58.4 ± 1.7 b |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Damazo-Lima, M.; Rosas-Pérez, G.; Reynoso-Camacho, R.; Pérez-Ramírez, I.F.; Rocha-Guzmán, N.E.; de los Ríos, E.A.; Ramos-Gomez, M. Chemopreventive Effect of the Germinated Oat and Its Phenolic-AVA Extract in Azoxymethane/Dextran Sulfate Sodium (AOM/DSS) Model of Colon Carcinogenesis in Mice. Foods 2020, 9, 169. https://doi.org/10.3390/foods9020169
Damazo-Lima M, Rosas-Pérez G, Reynoso-Camacho R, Pérez-Ramírez IF, Rocha-Guzmán NE, de los Ríos EA, Ramos-Gomez M. Chemopreventive Effect of the Germinated Oat and Its Phenolic-AVA Extract in Azoxymethane/Dextran Sulfate Sodium (AOM/DSS) Model of Colon Carcinogenesis in Mice. Foods. 2020; 9(2):169. https://doi.org/10.3390/foods9020169
Chicago/Turabian StyleDamazo-Lima, Margarita, Guadalupe Rosas-Pérez, Rosalía Reynoso-Camacho, Iza F. Pérez-Ramírez, Nuria Elizabeth Rocha-Guzmán, Ericka A. de los Ríos, and Minerva Ramos-Gomez. 2020. "Chemopreventive Effect of the Germinated Oat and Its Phenolic-AVA Extract in Azoxymethane/Dextran Sulfate Sodium (AOM/DSS) Model of Colon Carcinogenesis in Mice" Foods 9, no. 2: 169. https://doi.org/10.3390/foods9020169
APA StyleDamazo-Lima, M., Rosas-Pérez, G., Reynoso-Camacho, R., Pérez-Ramírez, I. F., Rocha-Guzmán, N. E., de los Ríos, E. A., & Ramos-Gomez, M. (2020). Chemopreventive Effect of the Germinated Oat and Its Phenolic-AVA Extract in Azoxymethane/Dextran Sulfate Sodium (AOM/DSS) Model of Colon Carcinogenesis in Mice. Foods, 9(2), 169. https://doi.org/10.3390/foods9020169