Innovative Antimicrobial Chitosan/ZnO/Ag NPs/Citronella Essential Oil Nanocomposite—Potential Coating for Grapes
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of ZnO and Ag Nanoparticles
2.3. Synthesis of Chitosan/ZnO and/or AgNPs Films
2.4. Characterization of Chitosan Composite Films
2.4.1. Microstructural Analysis
2.4.2. Fourier Transform Infrared Spectroscopy
2.4.3. Photoluminescence Spectroscopy
2.4.4. UV-Vis Spectroscopy
2.4.5. Thermal Analysis
2.4.6. Water Vapor Permeability (WVP)
2.5. Antimicrobial Assay
2.6. Statistical Analysis
3. Results and Discussion
3.1. Chitosan Films Characterization
3.2. FTIR Spectroscopy and Microscopy
3.2.1. FTIR Spectroscopy
3.2.2. FTIR Microscopy
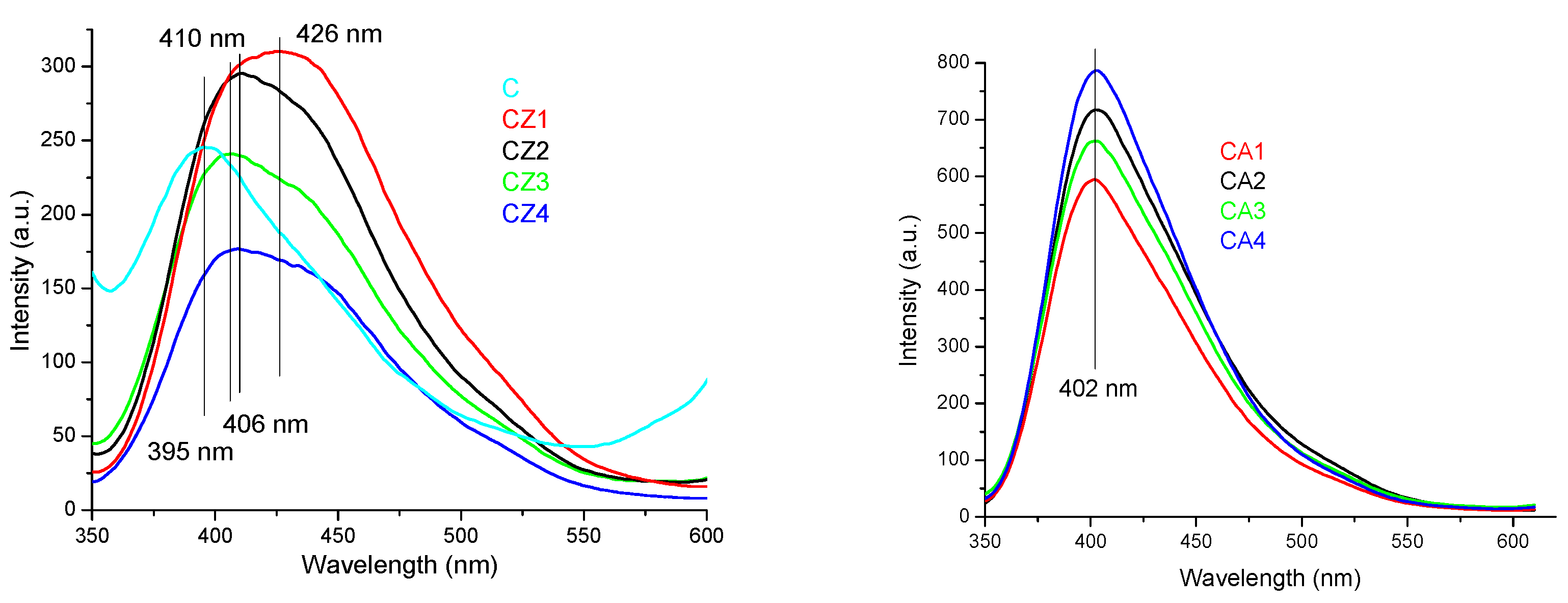
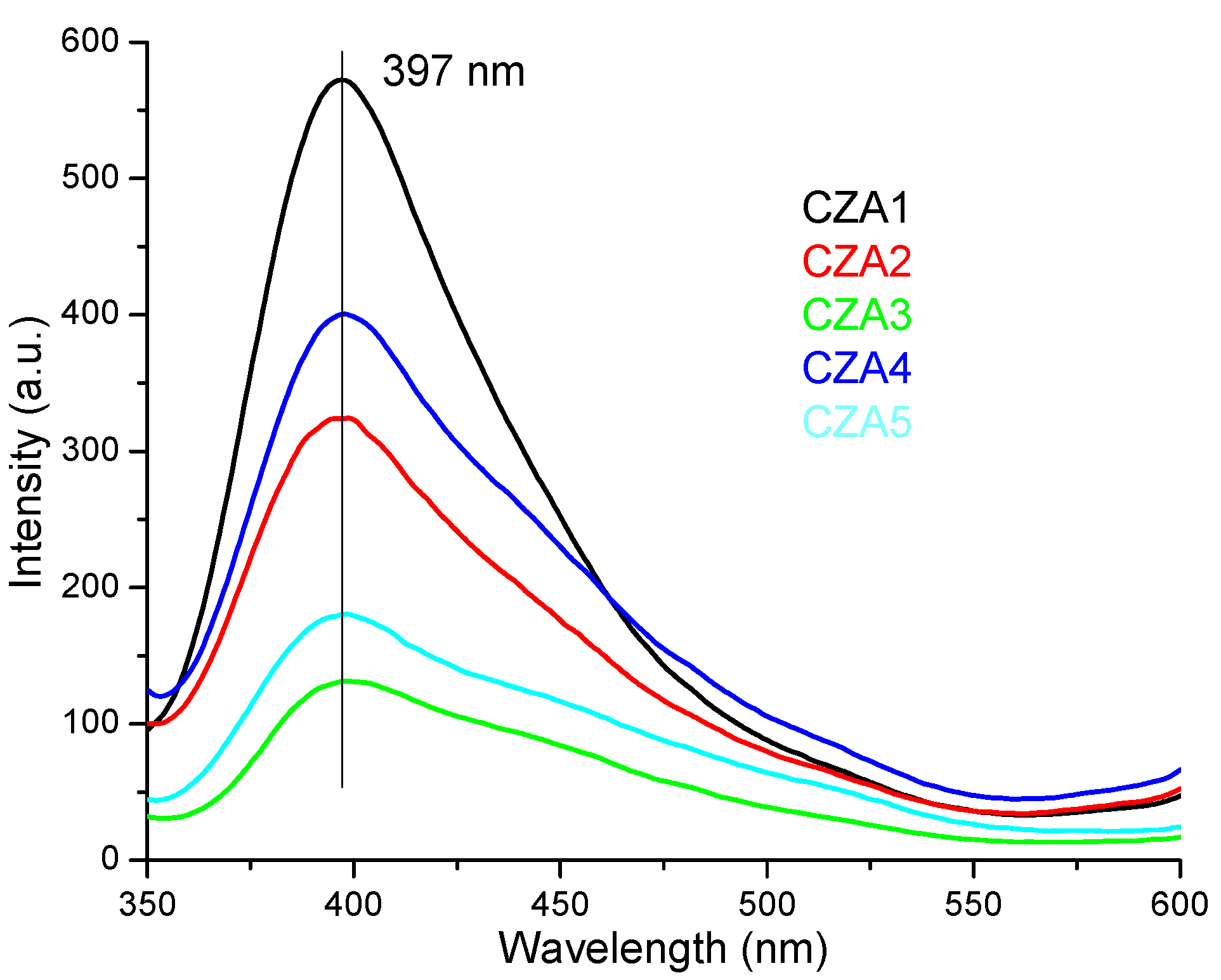
3.3. UV-Vis and PL Spectrometry
3.3.1. UV-Vis Spectrometry
3.3.2. PL Spectrometry
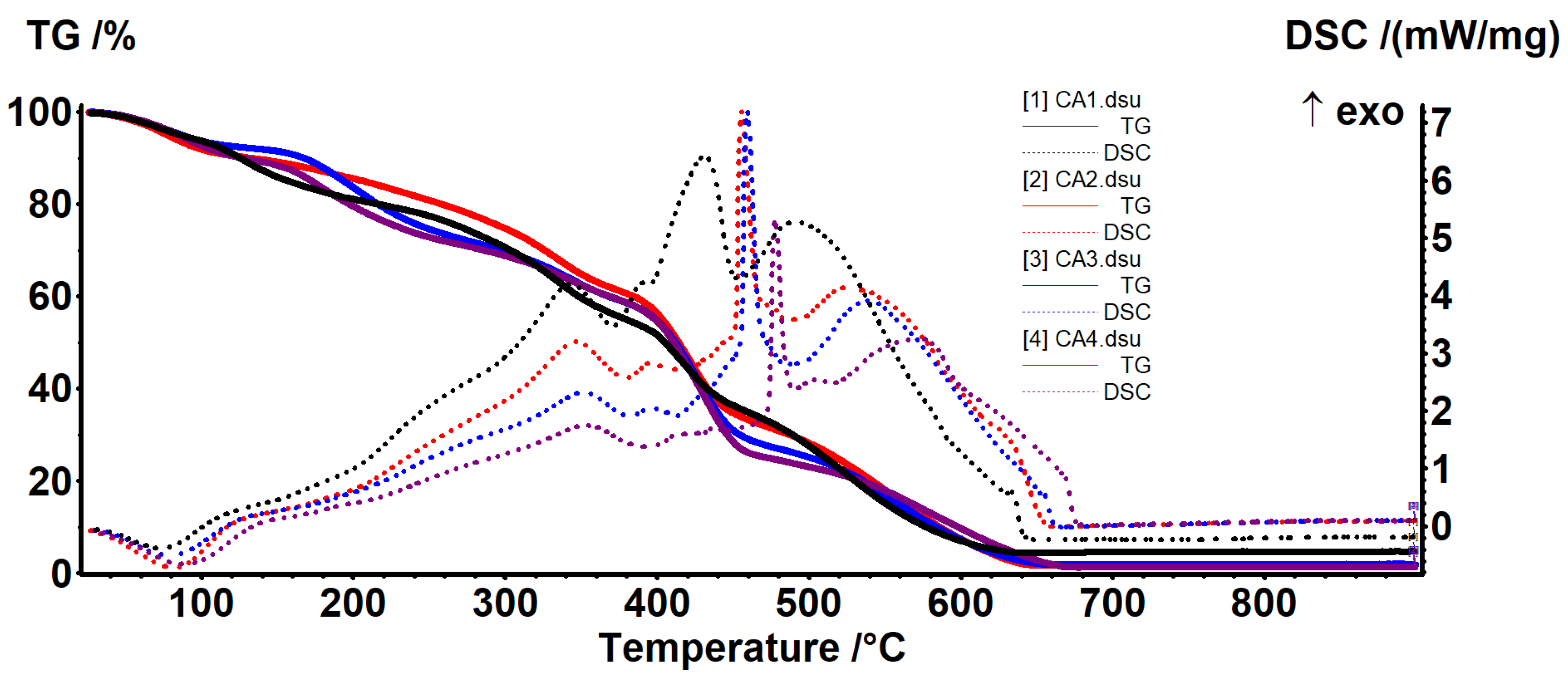
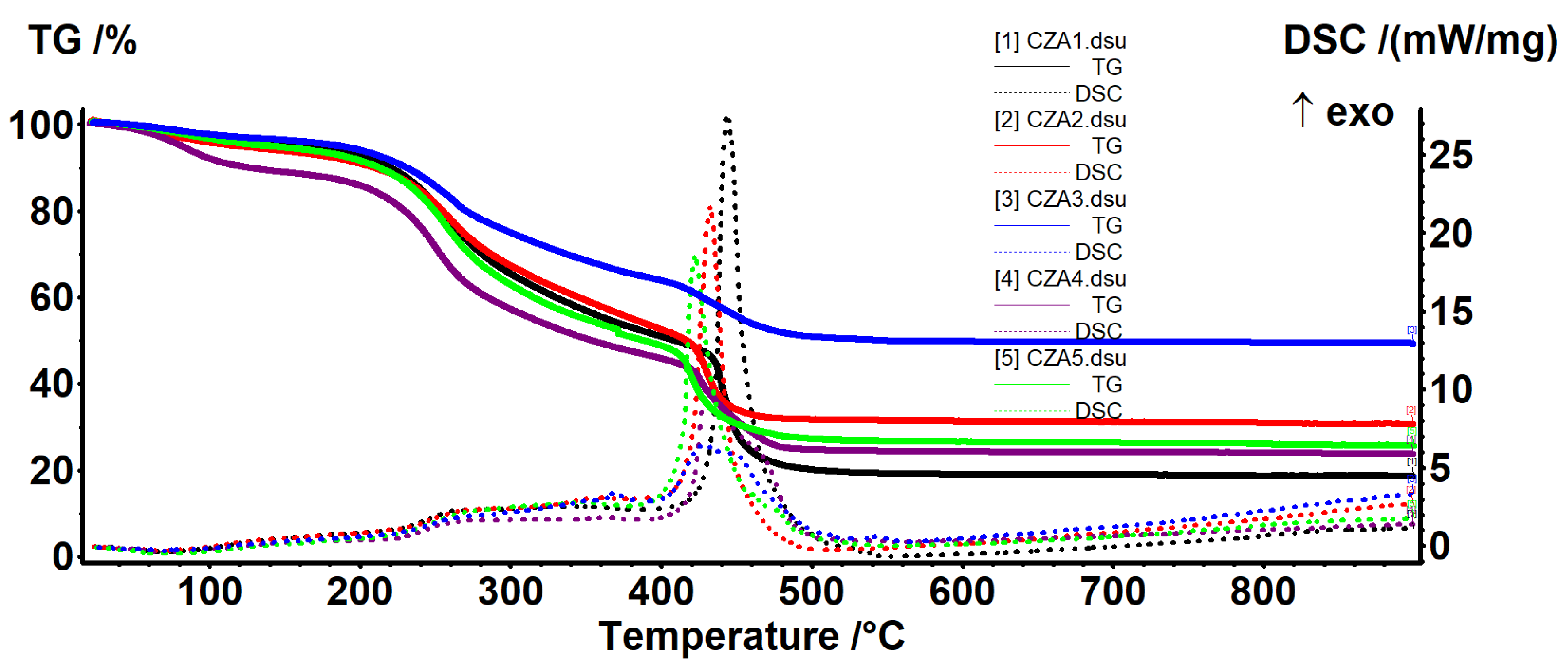
3.4. Thermal Analysis
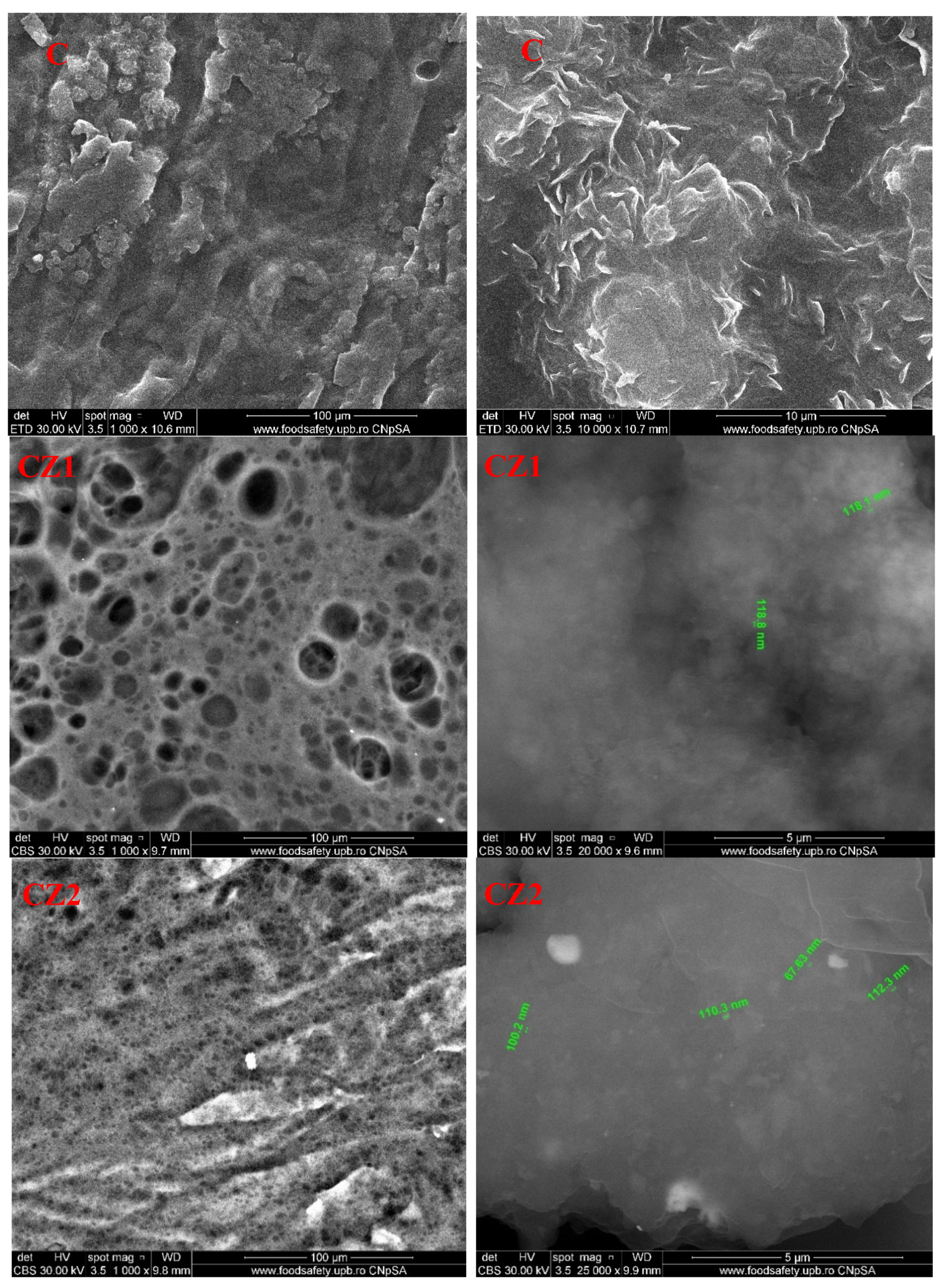
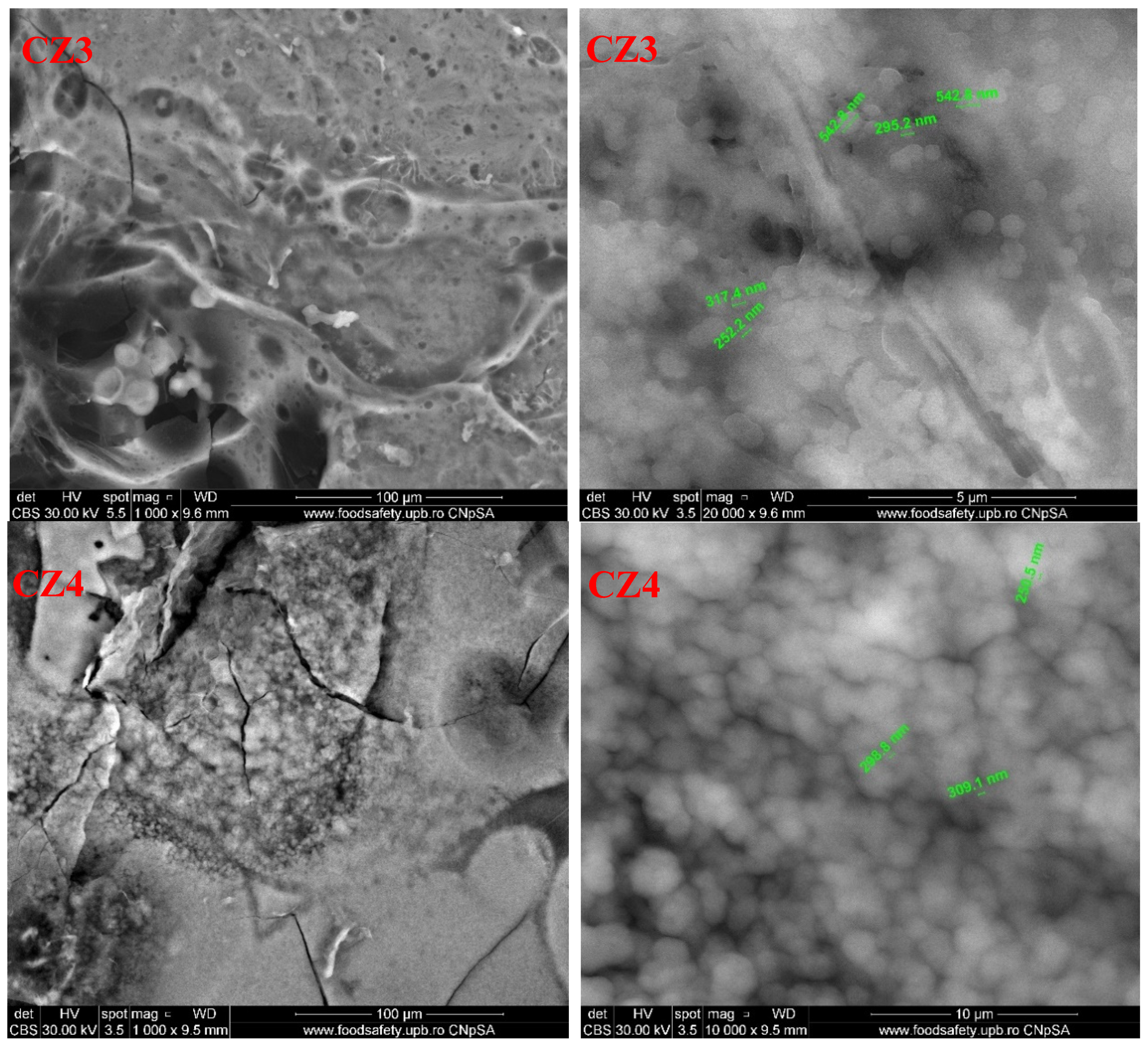
3.5. Scanning Electron Microscopy (SEM)
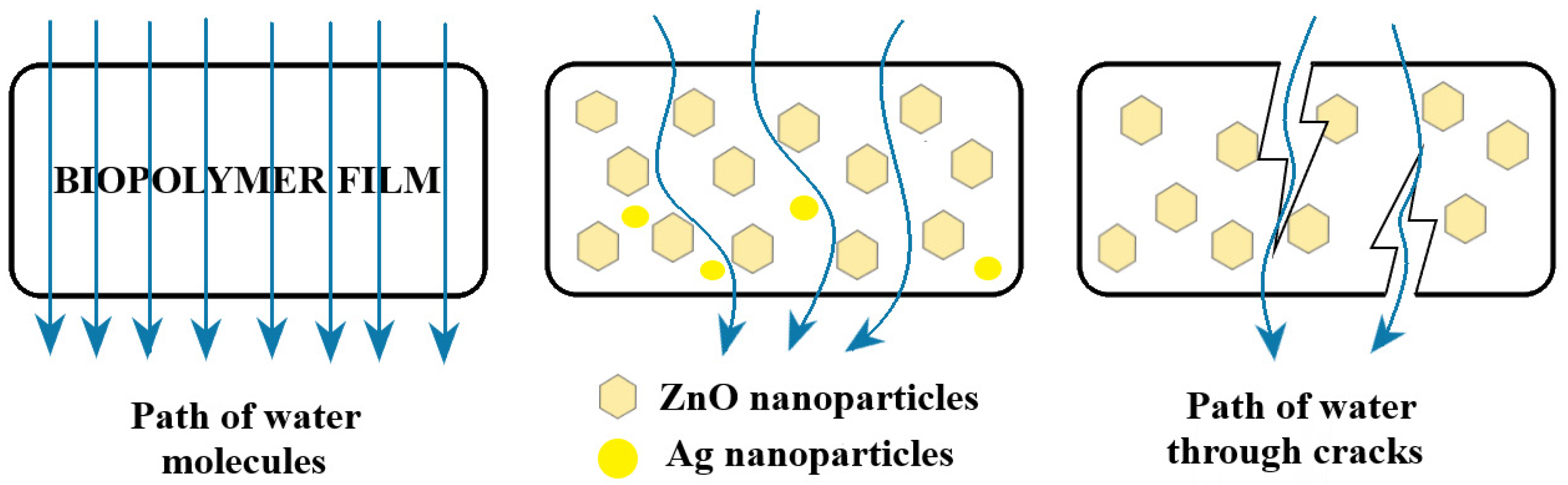
3.6. Water Vapor Permeability (WVP)
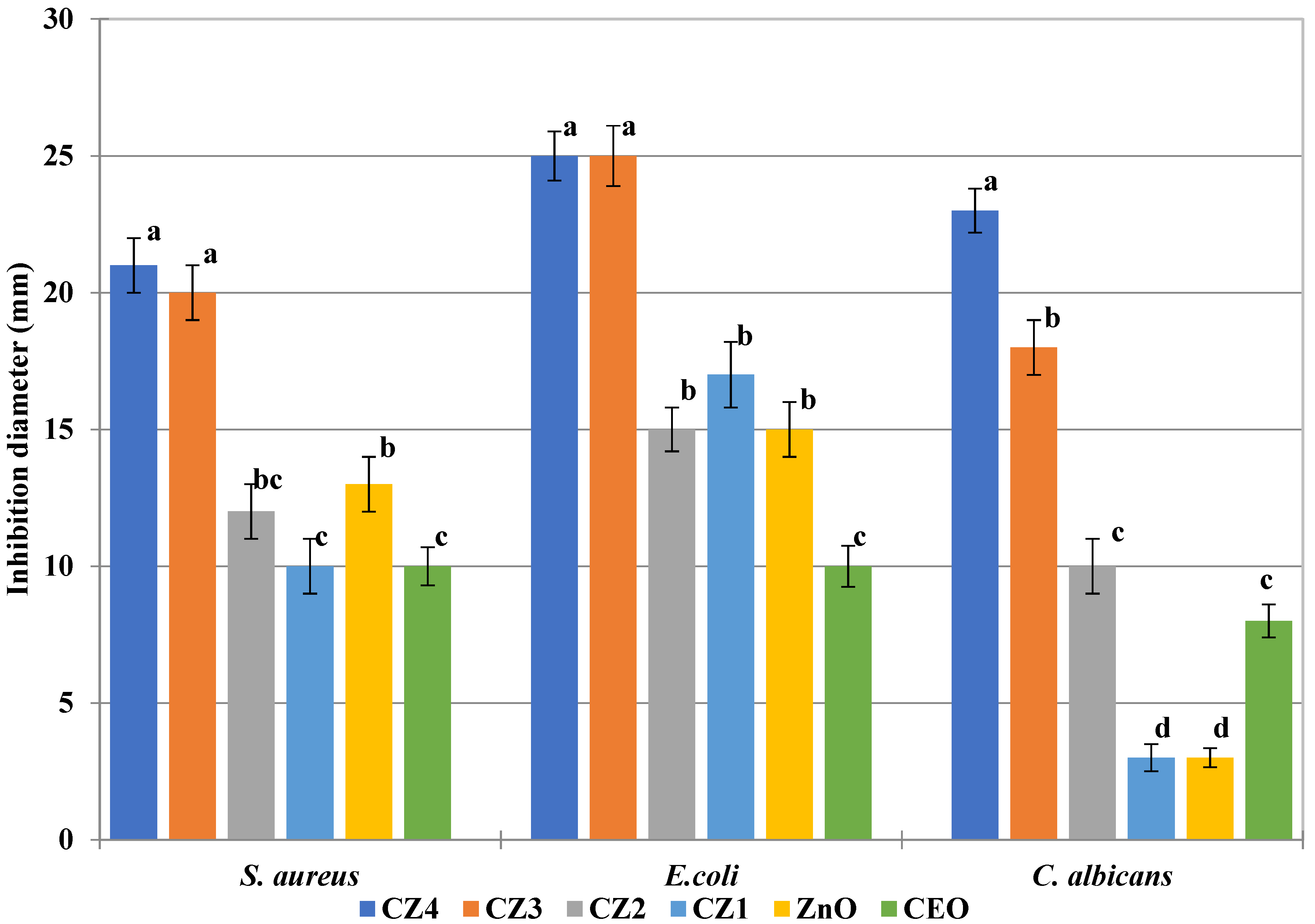
3.7. Antimicrobial Activity
3.8. Preliminary Evaluation of CZA1–CZA5 Films as Coatings for Grapes
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Technical Platform on the Measurement and Reduction of Food Loss and Waste. Available online: http://www.fao.org/platform-food-loss-waste/en/ (accessed on 25 October 2020).
- Motelica, L.; Ficai, D.; Oprea, O.C.; Ficai, A.; Andronescu, E. Smart Food Packaging Designed by Nanotechnological and Drug Delivery Approaches. Coatings 2020, 10, 806. [Google Scholar] [CrossRef]
- Lemnaru, G.-M.; Trusca, R.D.; Ilie, C.-I.; Tiplea, R.D.; Ficai, D.; Oprea, O.; Stoica-Guzun, A.; Ficai, A.; Ditu, L.-M. Antibacterial Activity of Bacterial Cellulose Loaded with Bacitracin and Amoxicillin: In Vitro Studies. Molecules 2020, 25, 4069. [Google Scholar] [CrossRef]
- Makaremi, M.; Yousefi, H.; Cavallaro, G.; Lazzara, G.; Goh, C.B.S.; Lee, S.M.; Solouk, A.; Pasbakhsh, P. Safely Dissolvable and Healable Active Packaging Films Based on Alginate and Pectin. Polymers 2019, 11, 1594. [Google Scholar] [CrossRef]
- Radulescu, M.; Ficai, D.; Oprea, O.; Ficai, A.; Andronescu, E.; Holban, A.M. Antimicrobial Chitosan based Formulations with Impact on Different Biomedical Applications. Curr. Pharm. Biotechnol. 2015, 16, 128–136. [Google Scholar] [CrossRef]
- Gingasu, D.; Mindru, I.; Patron, L.; Ianculescu, A.; Vasile, E.; Marinescu, G.; Preda, S.; Diamandescu, L.; Oprea, O.; Popa, M.; et al. Synthesis and Characterization of Chitosan-Coated Cobalt Ferrite Nanoparticles and Their Antimicrobial Activity. J. Inorg. Organomet. Polym. Mater. 2018, 28, 1932–1941. [Google Scholar] [CrossRef]
- Maluin, F.N.; Hussein, M.Z. Chitosan-Based Agronanochemicals as a Sustainable Alternative in Crop Protection. Molecules 2020, 25, 1611. [Google Scholar] [CrossRef]
- Anaya-Esparza, L.M.; Ruvalcaba-Gomez, J.M.; Maytorena-Verdugo, C.I.; Gonzalez-Silva, N.; Romero-Toledo, R.; Aguilera-Aguirre, S.; Perez-Larios, A.; Montalvo-Gonzalez, E. Chitosan-TiO2: A Versatile Hybrid Composite. Materials 2020, 13, 811. [Google Scholar] [CrossRef]
- Devlieghere, F.; Vermeulen, A.; Debevere, J. Chitosan: Antimicrobial activity, interactions with food components and applicability as a coating on fruit and vegetables. Food Microbiol. 2004, 21, 703–714. [Google Scholar] [CrossRef]
- Dutta, A.S.P.K. Extraction of Chitin-Glucan Complex from Agaricus bisporus: Characterization and Antibacterial Activity. J. Polym. Mater. 2017, 34, 1–9. [Google Scholar]
- Motelica, L.; Ficai, D.; Ficai, A.; Oprea, O.C.; Kaya, D.A.; Andronescu, E. Biodegradable Antimicrobial Food Packaging: Trends and Perspectives. Foods 2020, 9, 1438. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, R.; Lu, R.; Xu, J.; Hu, K.; Liu, Y.W. Preparation of Chitosan/Corn Starch/Cinnamaldehyde Films for Strawberry Preservation. Foods 2019, 8, 423. [Google Scholar] [CrossRef] [PubMed]
- Avramescu, S.M.; Butean, C.; Popa, C.V.; Ortan, A.; Moraru, I.; Temocico, G. Edible and Functionalized Films/Coatings-Performances and Perspectives. Coatings 2020, 10, 687. [Google Scholar] [CrossRef]
- Vaja, F.; Comanescu, C.; Oprea, O.; Ficai, D.; Guran, C. Effects of ZnO Nanoparticles on the Wet Scrub Resistance and Photocatalytic Properties of Acrylic Coatings. Rev. Chim. 2012, 63, 722–726. [Google Scholar]
- Kumar, S.; Mudai, A.; Roy, B.; Basumatary, I.B.; Mukherjee, A.; Dutta, J. Biodegradable Hybrid Nanocomposite of Chitosan/Gelatin and Green Synthesized Zinc Oxide Nanoparticles for Food Packaging. Foods 2020, 9, 1143. [Google Scholar] [CrossRef] [PubMed]
- Nedelcu, I.A.; Ficai, A.; Sonmez, M.; Ficai, D.; Oprea, O.; Andronescu, E. Silver Based Materials for Biomedical Applications. Curr. Org. Chem. 2014, 18, 173–184. [Google Scholar] [CrossRef]
- Istrati, D.; Lacatusu, I.; Bordei, N.; Badea, G.; Oprea, O.; Stefan, L.M.; Stan, R.; Badea, N.; Meghea, A. Phyto-mediated nanostructured carriers based on dual vegetable actives involved in the prevention of cellular damage. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 64, 249–259. [Google Scholar] [CrossRef]
- Lacatusu, I.; Badea, N.; Niculae, G.; Bordei, N.; Stan, R.; Meghea, A. Lipid nanocarriers based on natural compounds: An evolving role in plant extract delivery. Eur. J. Lipid Sci. Technol. 2014, 116, 1708–1717. [Google Scholar] [CrossRef]
- Caputo, L.; Cornara, L.; Bazzicalupo, M.; De Francesco, C.; De Feo, V.; Trombetta, D.; Smeriglio, A. Chemical Composition and Biological Activities of Essential Oils from Peels of Three Citrus Species. Molecules 2020, 25, 1890. [Google Scholar] [CrossRef]
- Zhang, H.J.; Wang, J.Q. Constituents of the Essential Oils of Garlic and Citronella and Their Vapor-phase Inhibition Mechanism against S.aureus. Food Sci. Technol. Res. 2019, 25, 65–74. [Google Scholar] [CrossRef]
- Francikowski, J.; Baran, B.; Cup, M.; Janiec, J.; Krzyzowski, M. Commercially Available Essential Oil Formulas as Repellents Against the Stored-Product Pest Alphitobius diaperinus. Insects 2019, 10, 96. [Google Scholar] [CrossRef]
- Go, E.J.; Song, K.B. Effect of java citronella essential oil addition on the physicochemical properties of Gelidium corneum-chitosan composite films. Food Sci. Biotechnol. 2020, 29, 909–915. [Google Scholar] [CrossRef] [PubMed]
- Munteanu, S.B.; Vasile, C. Vegetable Additives in Food Packaging Polymeric Materials. Polymers 2020, 12, 28. [Google Scholar] [CrossRef] [PubMed]
- Valdes, A.; Garcia-Serna, E.; Martinez-Abad, A.; Vilaplana, F.; Jimenez, A.; Garrigos, M.C. Gelatin-Based Antimicrobial Films Incorporating Pomegranate (Punica granatum L.) Seed Juice by-Product. Molecules 2020, 25, 166. [Google Scholar] [CrossRef]
- Becerril, R.; Nerin, C.; Silva, F. Encapsulation Systems for Antimicrobial Food Packaging Components: An Update. Molecules 2020, 25, 1134. [Google Scholar] [CrossRef] [PubMed]
- Oprea, M.; Panaitescu, D.M. Nanocellulose Hybrids with Metal Oxides Nanoparticles for Biomedical Applications. Molecules 2020, 25, 4045. [Google Scholar] [CrossRef]
- Espitia, P.J.P.; Otoni, C.G.; Soares, N.F.F. Zinc Oxide Nanoparticles for Food Packaging Applications. Antimicrob. Food Packag. 2016, 425–431. [Google Scholar] [CrossRef]
- Kumar, S.; Boro, J.C.; Ray, D.; Mukherjee, A.; Dutta, J. Bionanocomposite films of agar incorporated with ZnO nanoparticles as an active packaging material for shelf life extension of green grape. Heliyon 2019, 5, e01867. [Google Scholar] [CrossRef]
- Al-Naamani, L.; Dutta, J.; Dobretsov, S. Nanocomposite Zinc Oxide-Chitosan Coatings on Polyethylene Films for Extending Storage Life of Okra (Abelmoschus esculentus). Nanomaterials 2018, 8, 479. [Google Scholar] [CrossRef]
- Zorraquin-Pena, I.; Cueva, C.; Bartolome, B.; Moreno-Arribas, M.V. Silver Nanoparticles against Foodborne Bacteria. Effects at Intestinal Level and Health Limitations. Microorganisms 2020, 8, 132. [Google Scholar] [CrossRef]
- Souza, V.G.L.; Rodrigues, C.; Valente, S.; Pimenta, C.; Pires, J.R.A.; Alves, M.M.; Santos, C.F.; Coelhoso, I.M.; Fernando, A.L. Eco-Friendly ZnO/Chitosan Bionanocomposites Films for Packaging of Fresh Poultry Meat. Coatings 2020, 10, 110. [Google Scholar] [CrossRef]
- Giubilato, E.; Cazzagon, V.; Amorim, M.J.B.; Blosi, M.; Bouillard, J.; Bouwmeester, H.; Costa, A.L.; Fadeel, B.; Fernandes, T.F.; Fito, C.; et al. Risk Management Framework for Nano-Biomaterials Used in Medical Devices and Advanced Therapy Medicinal Products. Materials 2020, 13, 4532. [Google Scholar] [CrossRef] [PubMed]
- Chaudhary, R.G.; Bhusari, G.S.; Tiple, A.D.; Rai, A.R.; Somkuvar, S.R.; Potbhare, A.K.; Lambat, T.L.; Ingle, P.P.; Abdala, A.A. Metal/Metal Oxide Nanoparticles: Toxicity, Applications, and Future Prospects. Curr. Pharm. Des. 2019, 25, 4013–4029. [Google Scholar] [CrossRef] [PubMed]
- Ferdous, Z.; Nemmar, A. Health Impact of Silver Nanoparticles: A Review of the Biodistribution and Toxicity Following Various Routes of Exposure. Int. J. Mol. Sci. 2020, 21, 2375. [Google Scholar] [CrossRef] [PubMed]
- Abou Baker, D.H.; Al-Moghazy, M.; ElSayed, A.A.A. The in vitro cytotoxicity, antioxidant and antibacterial potential of Satureja hortensis L. essential oil cultivated in Egypt. Bioorg. Chem. 2020, 95, 103559. [Google Scholar]
- Sinha, S.; Jothiramajayam, M.; Ghosh, M.; Mukherjee, A. Evaluation of toxicity of essential oils palmarosa, citronella, lemongrass and vetiver in human lymphocytes. Food Chem. Toxicol. 2014, 68, 71–77. [Google Scholar] [CrossRef]
- Sonseca, A.; Madani, S.; Rodriguez, G.; Hevilla, V.; Echeverria, C.; Fernandez-Garcia, M.; Munoz-Bonilla, A.; Charef, N.; Lopez, D. Multifunctional PLA Blends Containing Chitosan Mediated Silver Nanoparticles: Thermal, Mechanical, Antibacterial, and Degradation Properties. Nanomaterials 2020, 10, 22. [Google Scholar] [CrossRef]
- Fontecha-Umana, F.; Rios-Castillo, A.G.; Ripolles-Avila, C.; Rodriguez-Jerez, J.J. Antimicrobial Activity and Prevention of Bacterial Biofilm Formation of Silver and Zinc Oxide Nanoparticle-Containing Polyester Surfaces at Various Concentrations for Use. Foods 2020, 9, 442. [Google Scholar] [CrossRef]
- Naskar, A.; Lee, S.; Lee, Y.; Kim, S.; Kim, K.S. A New Nano-Platform of Erythromycin Combined with Ag Nano-Particle ZnO Nano-Structure against Methicillin-Resistant Staphylococcus aureus. Pharmaceutics 2020, 12, 841. [Google Scholar] [CrossRef]
- Taha, A.; Ben Aissa, M.; Dana, E. Green Synthesis of an Activated Carbon-Supported Ag and ZnO Nanocomposite for Photocatalytic Degradation and Its Antibacterial Activities. Molecules 2020, 25, 1586. [Google Scholar] [CrossRef]
- Burlibasa, L.; Chifiriuc, M.C.; Lungu, M.V.; Lungulescu, E.M.; Mitrea, S.; Sbarcea, G.; Popa, M.; Marutescu, L.; Constantin, N.; Bleotu, C.; et al. Synthesis, physico-chemical characterization, antimicrobial activity and toxicological features of Ag-ZnO nanoparticles. Arab. J. Chem. 2020, 13, 4180–4197. [Google Scholar] [CrossRef]
- Oprea, O.; Andronescu, E.; Vasile, B.S.; Voicu, G.; Covaliu, C. Synthesis and Characterization of Zno Nanopowder by Non-Basic Route. Dig. J. Nanomater. Biostruct. 2011, 6, 1393–1401. [Google Scholar]
- Popescu, E.L.; Balasoiu, M.; Cristea, O.M.; Stoica, A.E.; Oprea, O.C.; Vasile, B.S.; Grumezescu, A.M.; Bancescu, G.; Busuioc, C.J.; Mogosanu, G.D.; et al. Study of antimicrobial effects of functionalized silver nanoparticles. Rom. J. Morphol. Embryol. 2019, 60, 939–946. [Google Scholar] [PubMed]
- Ipe, D.S.; Kumar, P.T.S.; Love, R.M.; Hamlet, S.M. Silver Nanoparticles at Biocompatible Dosage Synergistically Increases Bacterial Susceptibility to Antibiotics. Front. Microbiol. 2020, 11, 1074. [Google Scholar] [CrossRef] [PubMed]
- Mazur, P.; Skiba-Kurek, I.; Mrowiec, P.; Karczewska, E.; Drozdz, R. Synergistic ROS-Associated Antimicrobial Activity of Silver Nanoparticles and Gentamicin Against Staphylococcus epidermidis. Int. J. Nanomed. 2020, 15, 3551–3562. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Zheng, K.; Chen, H.; Feng, S.; Wang, W.; Qin, C. Influence of Nano Titanium Dioxide and Clove Oil on Chitosan–Starch Film Characteristics. Polymers 2019, 11, 1418. [Google Scholar] [CrossRef]
- Kildeeva, N.; Chalykh, A.; Belokon, M.; Petrova, T.; Matveev, V.; Svidchenko, E.; Surin, N.; Sazhnev, N. Influence of Genipin Crosslinking on the Properties of Chitosan-Based Films. Polymers 2020, 12, 1086. [Google Scholar] [CrossRef]
- Xing, Y.G.; Li, X.L.; Guo, X.L.; Li, W.X.; Chen, J.W.; Liu, Q.; Xu, Q.L.; Wang, Q.; Yang, H.; Shui, Y.R.; et al. Effects of Different TiO(2)Nanoparticles Concentrations on the Physical and Antibacterial Activities of Chitosan-Based Coating Film. Nanomaterials 2020, 10, 1365. [Google Scholar] [CrossRef]
- Prokhorov, E.; Luna-Barcenas, G.; Limon, J.M.Y.; Sanchez, A.G.; Kovalenko, Y. Chitosan-ZnO Nanocomposites Assessed by Dielectric, Mechanical, and Piezoelectric Properties. Polymers 2020, 12, 1991. [Google Scholar] [CrossRef]
- Oghani, F.Z.; Tahvildari, K.; Nozari, M. Novel Antibacterial Food Packaging Based on Chitosan Loaded ZnO Nano Particles Prepared by Green Synthesis from Nettle Leaf Extract. J. Inorg. Organomet. Polym. Mater. 2020. [Google Scholar] [CrossRef]
- Qiu, B.; Xu, X.F.; Deng, R.H.; Xia, G.Q.; Shang, X.F.; Zhou, P.H. Construction of chitosan/ZnO nanocomposite film by in situ precipitation. Int. J. Biol. Macromol. 2019, 122, 82–87. [Google Scholar] [CrossRef]
- Levi-Polyachenko, N.; Jacob, R.; Day, C.; Kuthirummal, N. Chitosan wound dressing with hexagonal silver nanoparticles for hyperthermia and enhanced delivery of small molecules. Colloids Surf. B Biointerfaces 2016, 142, 315–324. [Google Scholar] [CrossRef] [PubMed]
- Haghighi, H.; Biard, S.; Bigi, F.; De Leo, R.; Bedin, E.; Pfeifer, F.; Siesler, H.W.; Licciardello, F.; Pulvirenti, A. Comprehensive characterization of active chitosan-gelatin blend films enriched with different essential oils. Food Hydrocoll. 2019, 95, 33–42. [Google Scholar] [CrossRef]
- Wang, A.W.; Zhu, Q.; Xing, Z.P. Multifunctional quaternized chitosan@surface plasmon resonance Ag/N-TiO2 core-shell microsphere for synergistic adsorption-photothermal catalysis degradation of low-temperature wastewater and bacteriostasis under visible light. Chem. Eng. J. 2020, 393, 124781. [Google Scholar] [CrossRef]
- Wang, Z.; Wang, Q.; Liu, C.X.; He, F.Y.; Song, Z.G.; Qiu, Q.B.; Yan, J.; Zhu, W.W. Interaction of polyphenols and Ag on the surface plasmon resonance absorption and resonance Rayleigh scattering spectra. Vib. Spectrosc. 2020, 107, 103037. [Google Scholar] [CrossRef]
- Mehrdel, B.; Abdul Aziz, A. The Sensitivity of Surface Plasmon Resonance Damping for ColloidalSilver Nanoparticles. J. Phys. Conf. Ser. 2018, 1083, 012042. [Google Scholar] [CrossRef]
- Sarojini, K.S.; Indumathi, M.P.; Rajarajeswari, G.R. Mahua oil-based polyurethane/chitosan/nano ZnO composite films for biodegradable food packaging applications. Int. J. Biol. Macromol. 2019, 124, 163–174. [Google Scholar]
- Krishnan, R.A.; Mhatre, O.; Sheth, J.; Prabhu, S.; Jain, R.; Dandekar, P. Synthesis of zinc oxide nanostructures using orange peel oil for fabricating chitosan-zinc oxide composite films and their antibacterial activity. J. Polym. Res. 2020, 27, 206. [Google Scholar] [CrossRef]
- Vasile, B.S.; Oprea, O.; Voicu, G.; Ficai, A.; Andronescu, E.; Teodorescu, A.; Holban, A. Synthesis and characterization of a novel controlled release zinc oxide/gentamicin-chitosan composite with potential applications in wounds care. Int. J. Pharm. 2014, 463, 161–169. [Google Scholar] [CrossRef]
- Su, H.X.; Huang, C.X.; Liu, Y.; Kong, S.; Wang, J.; Huang, H.H.; Zhang, B.B. Preparation and Characterization of Cinnamomum Essential Oil-Chitosan Nanocomposites: Physical, Structural, and Antioxidant Activities. Processes 2020, 8, 834. [Google Scholar] [CrossRef]
- Shafiq, M.; Yasin, T.; Rafiq, M.A. Shaista, Structural, Thermal, and Antibacterial Properties of Chitosan/ZnO Composites. Polym. Compos. 2014, 35, 79–85. [Google Scholar] [CrossRef]
- de Araujo, L.O.; Anaya, K.; Pergher, S.B.C. Synthesis of Antimicrobial Films Based on Low-Density Polyethylene (LDPE) and Zeolite A Containing Silver. Coatings 2019, 9, 786. [Google Scholar] [CrossRef]
- Cobos, M.; De-La-Pinta, I.; Quindos, G.; Fernandez, M.J.; Fernandez, M.D. Graphene Oxide-Silver Nanoparticle Nanohybrids: Synthesis, Characterization, and Antimicrobial Properties. Nanomaterials 2020, 10, 376. [Google Scholar] [CrossRef] [PubMed]
- Rezaei, M.; Pirsa, S.; Chavoshizadeh, S. Photocatalytic/Antimicrobial Active Film Based on Wheat Gluten/ZnO Nanoparticles. J. Inorg. Organomet. Polym. Mater. 2020, 30, 2654–2665. [Google Scholar] [CrossRef]
- Taghizadeh, S.M.; Lal, N.; Ebrahiminezhad, A.; Moeini, F.; Seifan, M.; Ghasemi, Y.; Berenjian, A. Green and Economic Fabrication of Zinc Oxide (ZnO) Nanorods as a Broadband UV Blocker and Antimicrobial Agent. Nanomaterials 2020, 10, 530. [Google Scholar] [CrossRef]
- Salmas, C.; Giannakas, A.; Katapodis, P.; Leontiou, A.; Moschovas, D.; Karydis-Messinis, A. Development of ZnO/Na-Montmorillonite Hybrid Nanostructures Used for PVOH/ZnO/Na-Montmorillonite Active Packaging Films Preparation via a Melt-Extrusion Process. Nanomaterials 2020, 10, 1079. [Google Scholar] [CrossRef]
- Mocanu, A.; Isopencu, G.; Busuioc, C.; Popa, O.M.; Dietrich, P.; Socaciu-Siebert, L. Bacterial cellulose films with ZnO nanoparticles and propolis extracts: Synergistic antimicrobial effect. Sci. Rep. 2019, 9, 17687. [Google Scholar] [CrossRef]
- Pillai, A.M.; Sivasankarapillai, V.S.; Rahdar, A.; Joseph, J.; Sadeghfar, F.; Anuf, A.R.; Rajesh, K.; Kyzas, G.Z. Green synthesis and characterization of zinc oxide nanoparticles with antibacterial and antifungal activity. J. Mol. Struct. 2020, 1211, 128107. [Google Scholar] [CrossRef]
- Oprea, O.; Andronescu, E.; Ficai, D.; Ficai, A.; Oktar, F.N.; Yetmez, M. ZnO Applications and Challenges. Curr. Org. Chem. 2014, 18, 192–203. [Google Scholar] [CrossRef]
- Motelica, L.; Marinof, L.; Holban, A.; Vasile, B.S.; Ficai, A. Optical, Photocatalytic and Antibacterial Properties of Zinc Oxide Nanoparticles Obtained by a Solvothermal Method. UPB Sci. Bull. Ser. B 2020, 82, 59–70. [Google Scholar]
- Boura-Theodoridou, O.; Giannakas, A.; Katapodis, P.; Stamatis, H.; Ladavos, A.; Barkoula, N.M. Performance of ZnO/chitosan nanocomposite films for antimicrobial packaging applications as a function of NaOH treatment and glycerol/PVOH blending. Food Packag. Shelf Life 2020, 23, 100456. [Google Scholar] [CrossRef]
- Kaur, M.; Kalia, A.; Thakur, A. Effect of biodegradable chitosan-rice-starch nanocomposite films on post-harvest quality of stored peach fruit. Starch Starke 2017, 69, 1600208. [Google Scholar] [CrossRef]
- Kumar, S.; Shukla, A.; Baul, P.P.; Mitra, A.; Halder, D. Biodegradable hybrid nanocomposites of chitosan/gelatin and silver nanoparticles for active food packaging applications. Food Packag. Shelf Life 2018, 16, 178–184. [Google Scholar] [CrossRef]
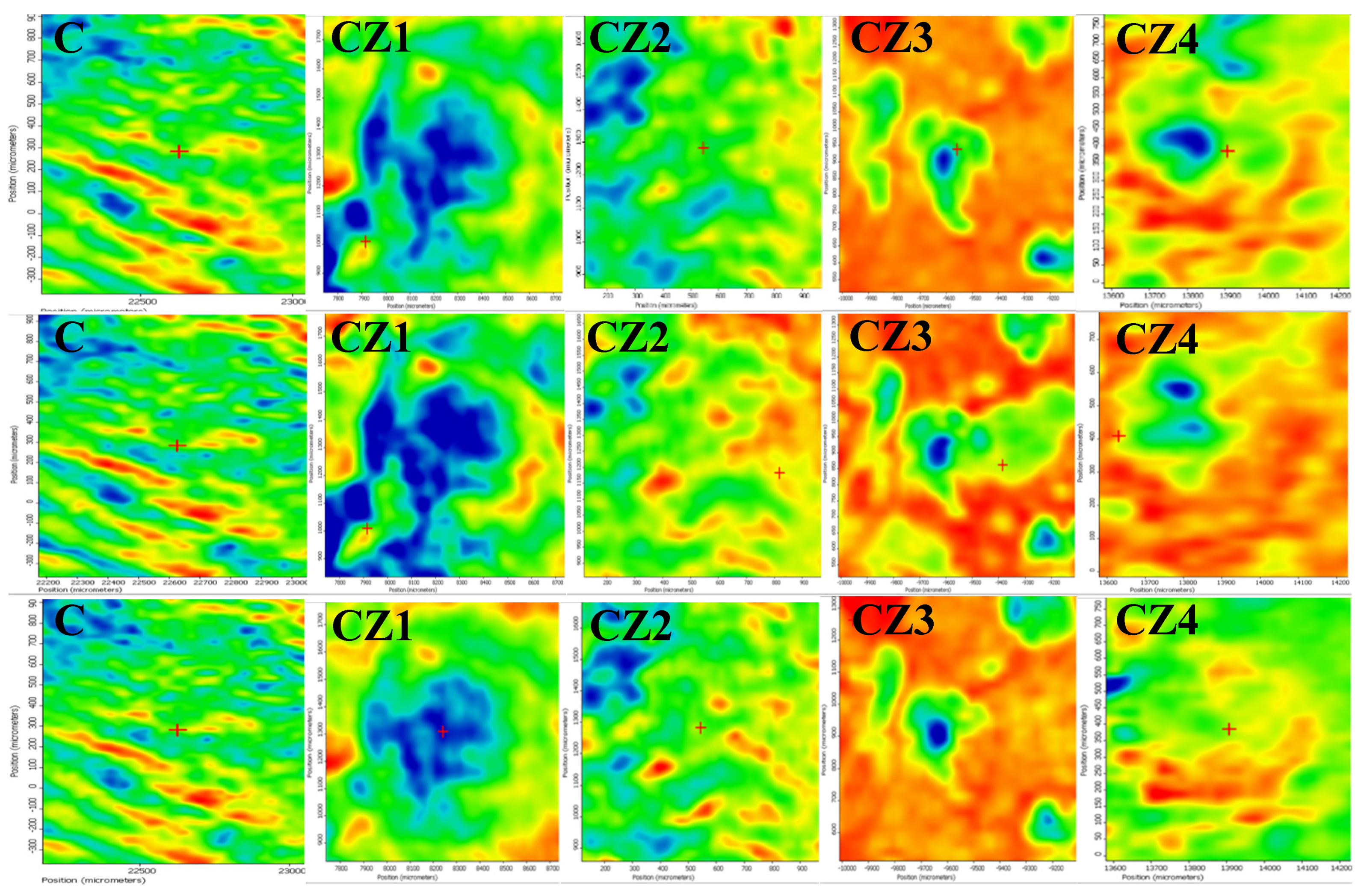

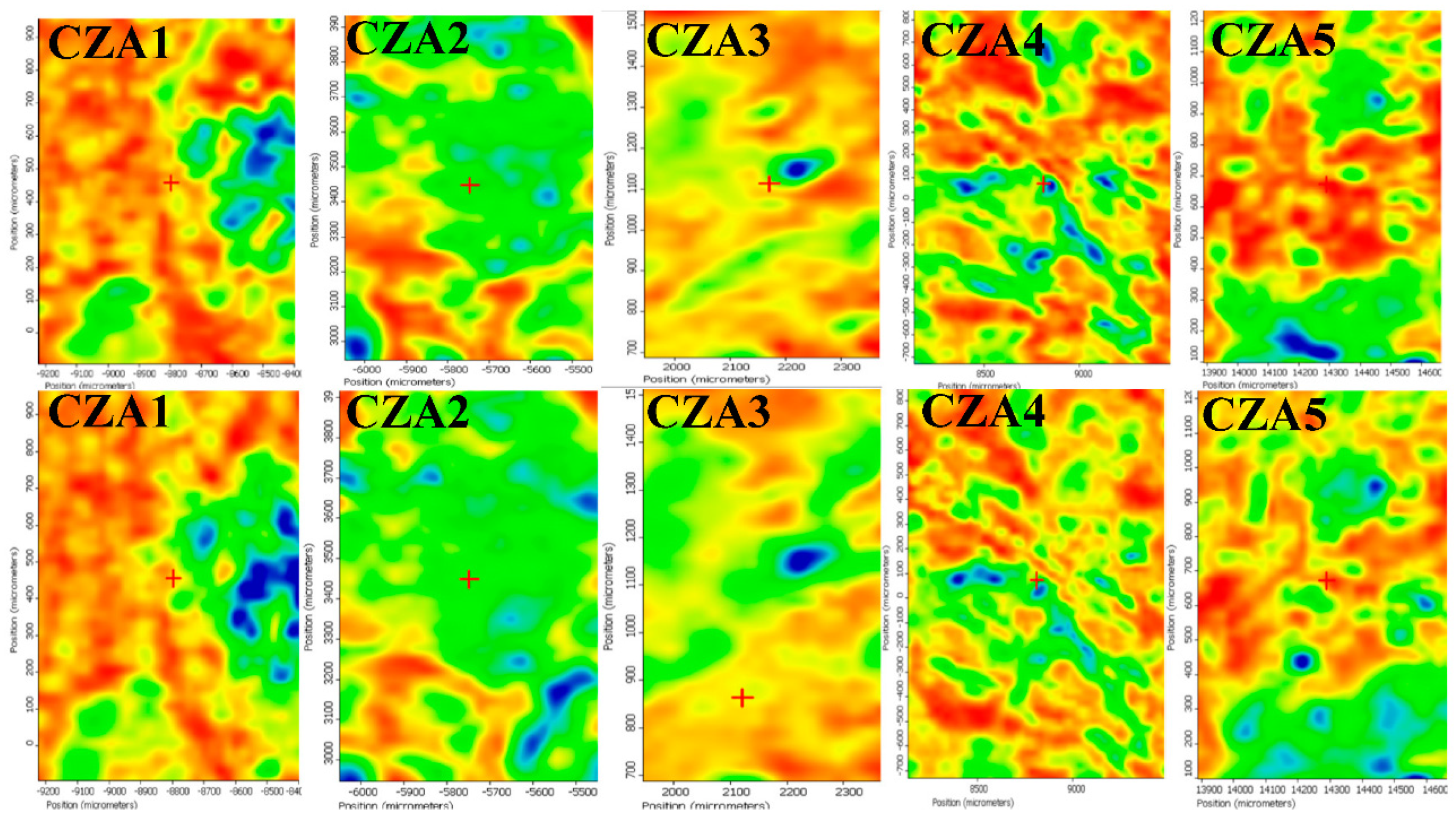
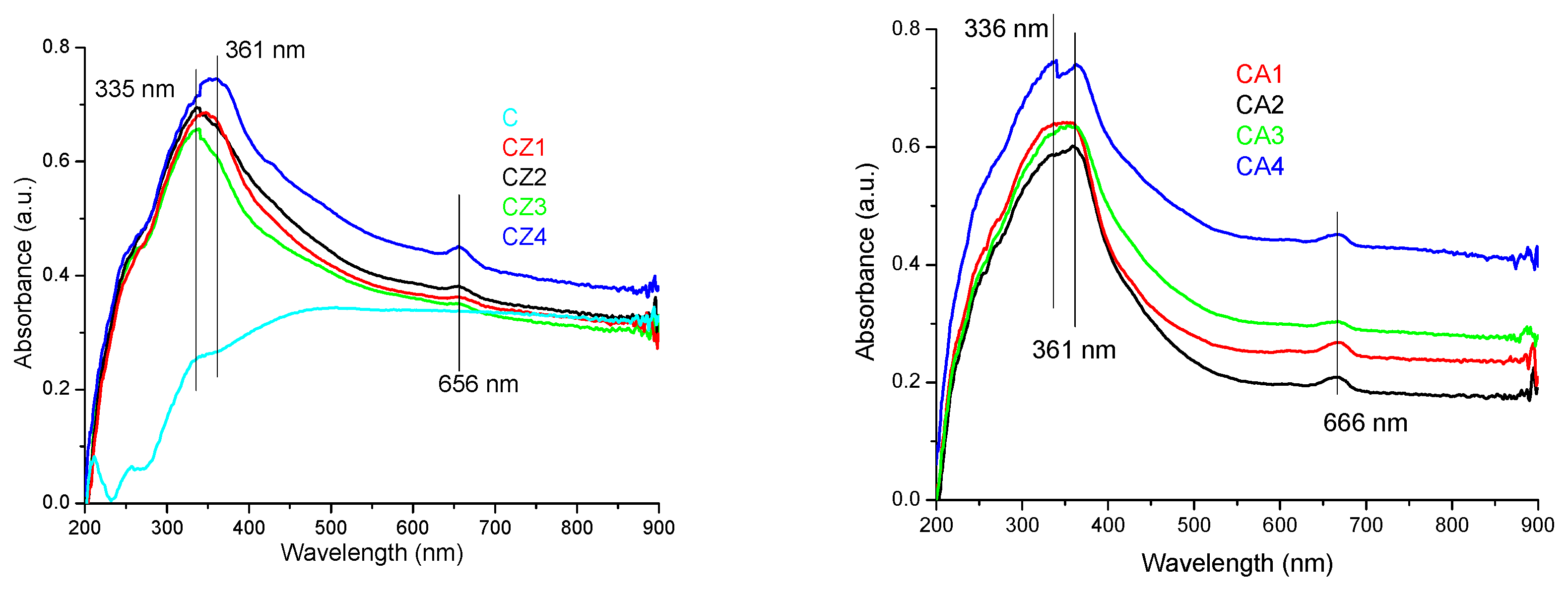
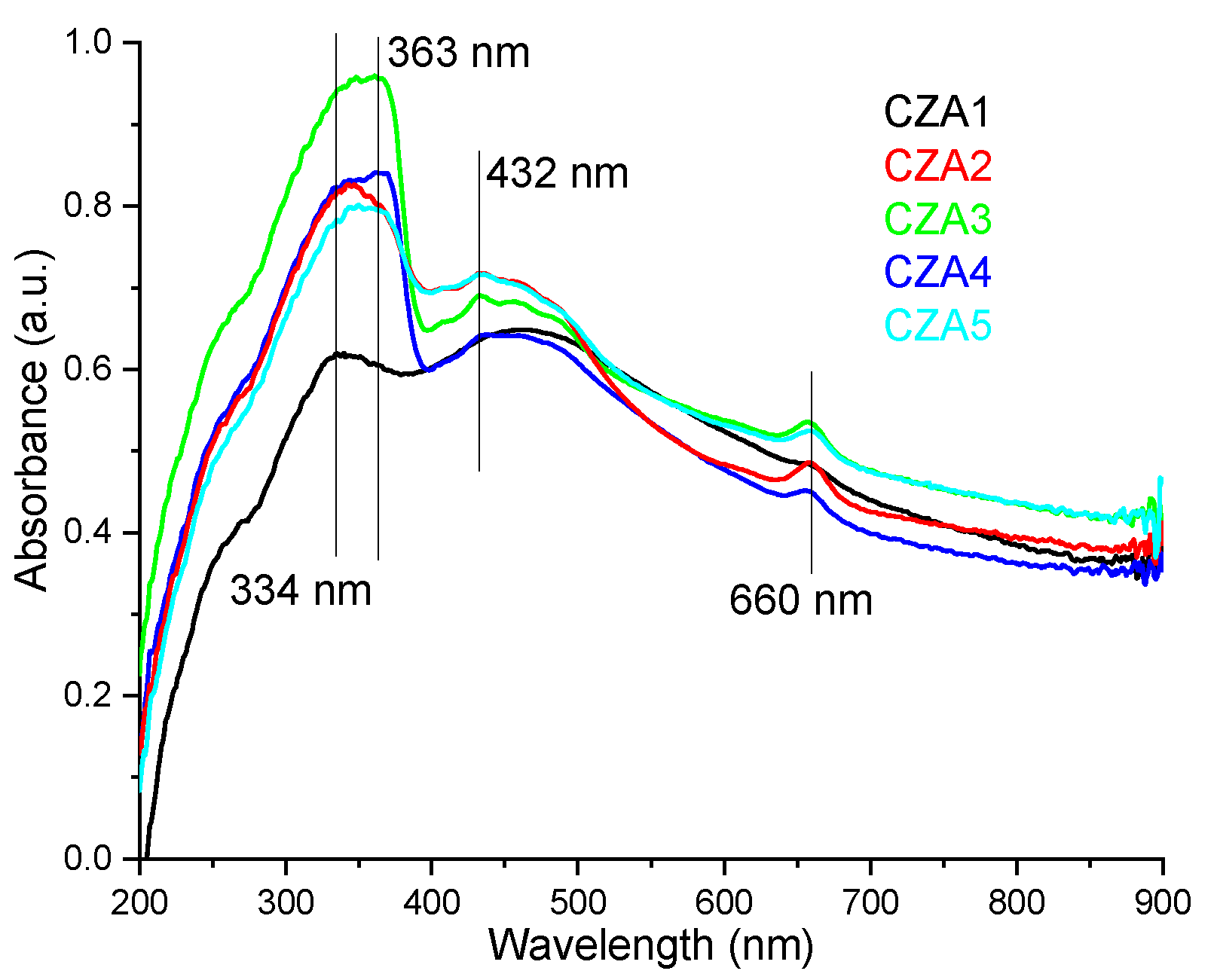





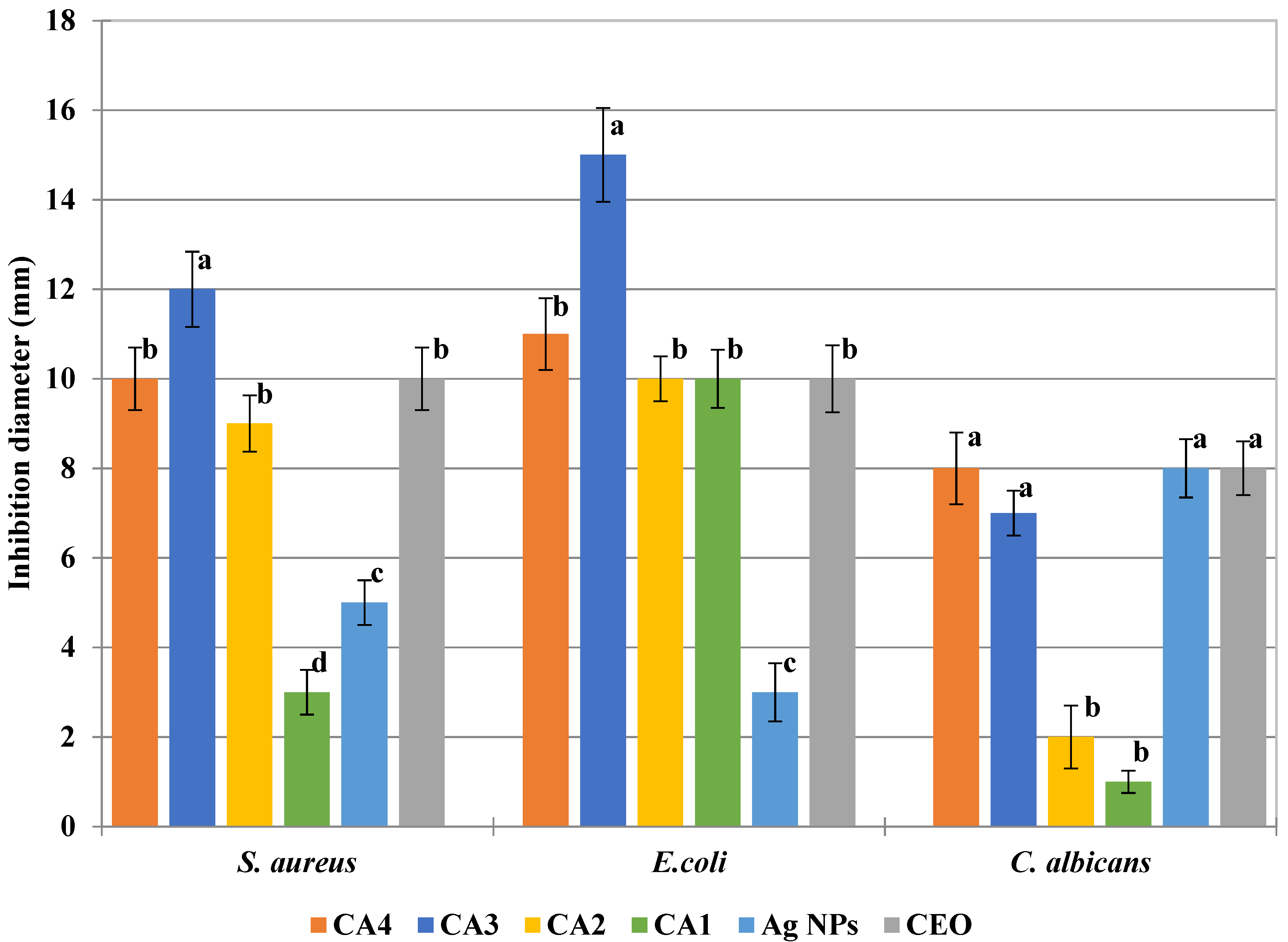


| Sample Code | Chitosan (g in 100 mL of 1% (v/v) Acetic Acid) | ZnO NPs (g in 15 mL Water) | Ag NPs (mL Solution) | CEO (mL) |
|---|---|---|---|---|
| CZ1 | 0.66 | 0.165 | - | 1 |
| CZ2 | 0.66 | 0.33 | - | 1 |
| CZ3 | 0.66 | 0.66 | - | 1 |
| CZ4 | 0.66 | 0.99 | - | 1 |
| CA1 | 0.66 | - | 10 | 1 |
| CA2 | 0.66 | - | 15 | 1 |
| CA3 | 0.66 | - | 20 | 1 |
| CA4 | 0.66 | - | 30 | 1 |
| CZA1 | 0.66 | 0.33 | 15 | 1 |
| CZA2 | 0.66 | 0.66 | 15 | 1 |
| CZA3 | 0.66 | 0.99 | 15 | 1 |
| CZA4 | 0.66 | 0.66 | 10 | 1 |
| CZA5 | 0.66 | 0.66 | 30 | 1 |
| Sample | υZn-O | As. C-O-C | Amide III Band (υC-N; δN-H) | Amide II Band (υC-N; δN-H) | Amide I Band (υC-N; υC=O) | C=O Group of CEO [22] | υC-H (sat) | υO-H υN-H |
|---|---|---|---|---|---|---|---|---|
| C | - | 1152 | 1318 | 1553 | 1651 | - | 2921 | 3277 |
| CZ1 | 426 473 | 1152 | 1338 | 1558 | 1643 | 1745 | 2918 | 3255 |
| CZ2 | 427 474 | 1152 | 1338 | 1559 | 1643 | 1747 | 2920 | 3263 |
| CZ3 | 428 467 | 1152 | 1338 | 1557 | 1643 | 1744 | 2919 | 3295 |
| CZ4 | 428 467 | 1152 | 1338 | 1558 | 1643 | 1747 | 2922 | 3305 |
| CA1 | - | 1152 | 1317 | 1558 | 1647 | 1746 | 2921 | 3368 |
| CA2 | - | 1154 | 1317 | 1563 | 1647 | 1746 | 2922 | 3392 |
| CA3 | - | 1153 | 1317 | 1560 | 1645 | 1745 | 2920 | 3393 |
| CA4 | - | 1155 | 1317 | 1560 | 1644 | 1744 | 2920 | 3394 |
| CZA1 | 419 475 | 1152 | 1339 | 1559 | 1643 | 1744 | 2918 | 3254 |
| CZA2 | 421 472 | 1153 | 1329 | 1559 | 1643 | 1745 | 2921 | 3302 |
| CZA3 | 426 469 | 1152 | 1338 | 1555 | 1643 | 1744 | 2924 | 3297 |
| CZA4 | 419 468 | 1153 | 1339 | 1559 | 1643 | 1746 | 2922 | 3304 |
| CZA5 | 429 470 | 1153 | 1340 | 1559 | 1647 | 1747 | 2918 | 3292 |
| Sample | T10 | T20 | T30 | T40 | T50 | Residual Mass |
|---|---|---|---|---|---|---|
| CZ1 | 192 °C | 229 °C | 252 °C | 273 °C | 306 °C | 12.23% |
| CZ2 | 159 °C | 216 °C | 239 °C | 260 °C | 293 °C | 14.86% |
| CZ3 | 192 °C | 227 °C | 244 °C | 260 °C | 295 °C | 18.91% |
| CZ4 | 200 °C | 244 °C | 270 °C | 330 °C | 412 °C | 25.33% |
| Sample | T10 | T20 | T30 | T40 | T50 | Residual Mass |
|---|---|---|---|---|---|---|
| CA1 | 125 °C | 215 °C | 303 °C | 348 °C | 405 °C | 4.65% |
| CA2 | 130 °C | 258 °C | 326 °C | 384 °C | 414 °C | 1.89% |
| CA3 | 167 °C | 216 °C | 297 °C | 368 °C | 412 °C | 1.87% |
| CA4 | 126 °C | 197 °C | 286 °C | 369 °C | 411 °C | 1.24% |
| Sample | T10 | T20 | T30 | T40 | T50 | Residual Mass |
|---|---|---|---|---|---|---|
| CZA1 | 220 °C | 254 °C | 281 °C | 330 °C | 408 °C | 18.47% |
| CZA2 | 210 °C | 255 °C | 287 °C | 346 °C | 417 °C | 30.64% |
| CZA3 | 231 °C | 270 °C | 337 °C | 427 °C | 542 °C | 49.27% |
| CZA4 | 126 °C | 230 °C | 254 °C | 285 °C | 354 °C | 23.72% |
| CZA5 | 213 °C | 250 °C | 273 °C | 316 °C | 389 °C | 25.67% |
| Film Code | WVP (10−10 g/Pa∙m∙s) |
|---|---|
| C (chitosan control) | 1.411 ± 0.011 a |
| CZ1 | 1.214 ± 0.018 a,b,c,d |
| CZ2 | 1.165 ± 0.023 b,c,d |
| CZ3 | 1.298 ± 0.056 a,b |
| CZ4 | 1.315 ± 0.124 a,b |
| CA1 | 1.326 ± 0.078 a,b |
| CA2 | 1.301 ± 0.081 a,b |
| CA3 | 1.278 ± 0.056 a,b,c |
| CA4 | 1.262 ± 0.035 a,b,c |
| CZA1 | 1.143 ± 0.091 b,c,d |
| CZA2 | 1.055 ± 0.078 c,d |
| CZA3 | 1.281 ± 0.137 a,b,c |
| CZA4 | 1.117 ± 0.069 b,c,d |
| CZA5 | 1.026 ± 0.086 d |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Motelica, L.; Ficai, D.; Ficai, A.; Truşcă, R.-D.; Ilie, C.-I.; Oprea, O.-C.; Andronescu, E. Innovative Antimicrobial Chitosan/ZnO/Ag NPs/Citronella Essential Oil Nanocomposite—Potential Coating for Grapes. Foods 2020, 9, 1801. https://doi.org/10.3390/foods9121801
Motelica L, Ficai D, Ficai A, Truşcă R-D, Ilie C-I, Oprea O-C, Andronescu E. Innovative Antimicrobial Chitosan/ZnO/Ag NPs/Citronella Essential Oil Nanocomposite—Potential Coating for Grapes. Foods. 2020; 9(12):1801. https://doi.org/10.3390/foods9121801
Chicago/Turabian StyleMotelica, Ludmila, Denisa Ficai, Anton Ficai, Roxana-Doina Truşcă, Cornelia-Ioana Ilie, Ovidiu-Cristian Oprea, and Ecaterina Andronescu. 2020. "Innovative Antimicrobial Chitosan/ZnO/Ag NPs/Citronella Essential Oil Nanocomposite—Potential Coating for Grapes" Foods 9, no. 12: 1801. https://doi.org/10.3390/foods9121801
APA StyleMotelica, L., Ficai, D., Ficai, A., Truşcă, R.-D., Ilie, C.-I., Oprea, O.-C., & Andronescu, E. (2020). Innovative Antimicrobial Chitosan/ZnO/Ag NPs/Citronella Essential Oil Nanocomposite—Potential Coating for Grapes. Foods, 9(12), 1801. https://doi.org/10.3390/foods9121801








