Evaluation of Polyphenolic Profile and Antioxidant Activity of Pistacia lentiscus L. Leaves and Fruit Extract Obtained by Optimized Microwave-Assisted Extraction
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Chemicals and Reagents
2.3. Microwave-Assisted Extraction
2.4. Conventional Extraction
2.5. Polyphenols Analysis
2.5.1. Determination of Total Phenolic Content
2.5.2. UPLC/ESI-MS2 Analysis
2.5.3. Oxygen Radical Absorbance Capacity (ORAC) Assay
2.6. Experimental Design and Statistical Analysis
3. Results and Discussion
3.1. Microwave-Assisted Extraction Optimization
3.2. Polyphenolic Characterization
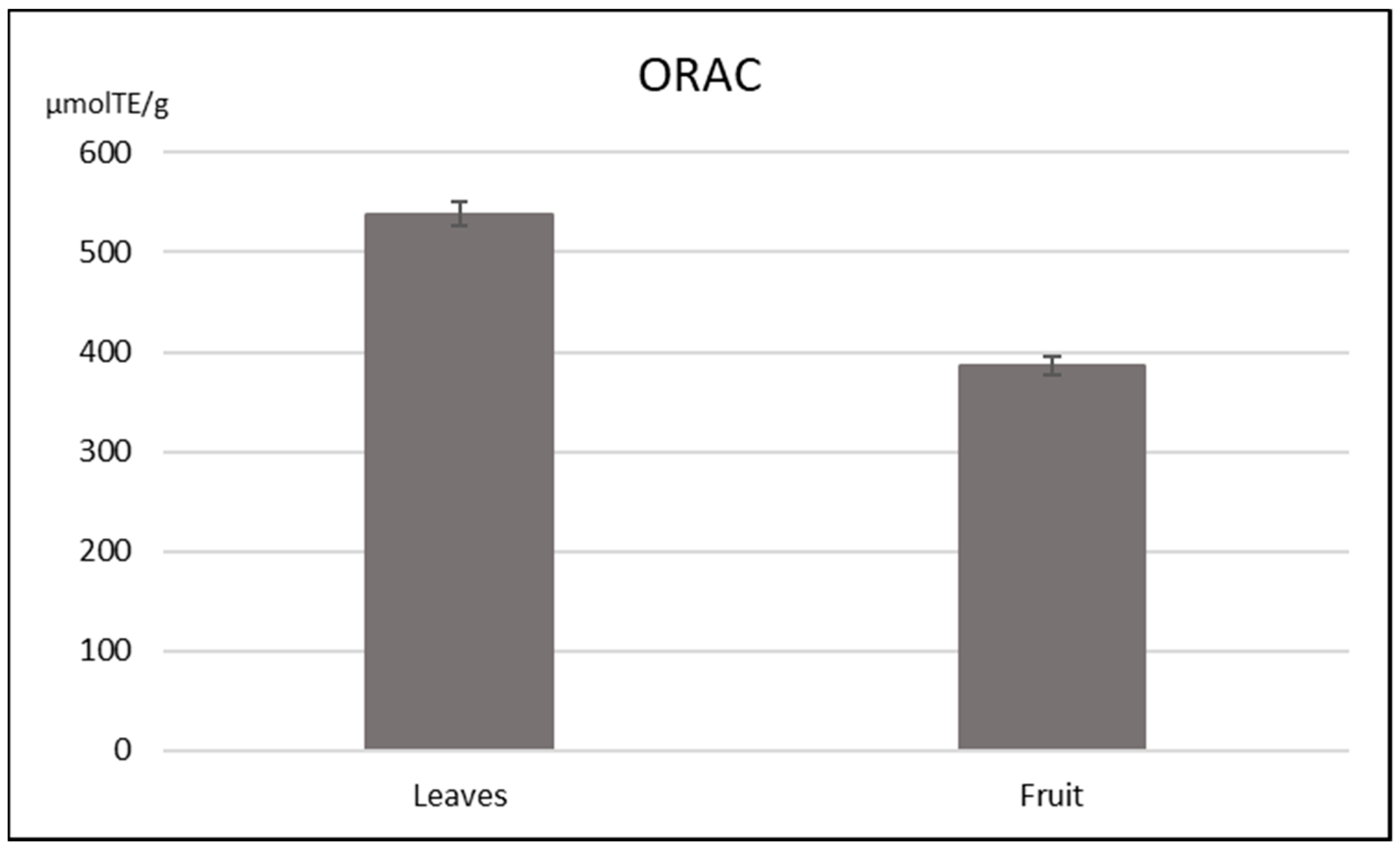
3.3. Antioxidant Capacity
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bampouli, A.; Kyriakopoulou, K.; Papaefstathiou, G.; Louli, V.; Nektarios, A.; Krokida, M.; Magoulas, K. Evaluation of total antioxidant potential of Pistacia lentiscus var. chia leaves extracts using UHPLC-HRMS. J. Food Eng. 2015, 167, 25–31. [Google Scholar] [CrossRef]
- Gardeli, C.; Vassiliki, P.; Athanasios, M.; Kibouris, T.; Komaitis, M. Essential oil composition of Pistacia lentiscus L. and Myrtus communis L.: Evaluation of antioxidant capacity of methanolic extracts. Food Chem. 2008, 107, 1120–1130. [Google Scholar] [CrossRef]
- Dahmoune, F.; Spigno, G.; Moussi, K.; Remini, H.; Cherbal, A.; Madani, K. Pistacia lentiscus leaves as a source of phenolic compounds: Microwave-assisted extraction optimized and compared with ultrasound-assisted and conventional solvent extraction. Ind. Crop. Prod. 2014, 61, 31–40. [Google Scholar] [CrossRef]
- Belhachat, D.; Aidb, F.; Mekimenea, L.; Belhachatc, M. Phytochemical screening and in vitro antioxidant activity of Pistacia lentiscus berries ethanolic extract growing in Algeria. Med. J. Nutr. Metab. 2017, 10, 273–285. [Google Scholar] [CrossRef]
- Dragović, S.; Dragović-Uzelac, V.; Pedisić, S.; Čošić, Z.; Friščoć, M.; Elez Garofulić, I.; Zorić, Z. The Mastic Tree (Pistacia lentiscus L.) Leaves as Source of BACs: Effect of Growing Location, Phenological Stage and Extraction Solvent on Phenolic Content. Food Technol. Biotechnol. in press.
- Piluzza, G.; Bullitta, S. Correlations between phenolic content and antioxidant properties in twenty-four plant species of traditional ethnoveterinary use in the Mediterranean area. Pharm. Biol. 2011, 49, 240–247. [Google Scholar] [CrossRef]
- Bimakr, M.; Rahman, R.A.; Taip, F.S.; Ganjloo, A.; Salleh, L.M.; Selamat, J.; Hamid, A.; Zaidul, I.S.M. Comparison of different extraction methods for the extraction of major bioactive flavonoid compounds from spearmint (Mentha spicata L.) leaves. Food Bioprod. Process. 2011, 89, 67–72. [Google Scholar] [CrossRef]
- Elez Garofulić, I.; Dragović-Uzelac, V.; Režek Jambrak, A.; Jukić, M. The effect of microwave assisted extraction on the isolation of anthocyanins and phenolic acids from sour cherry Marasca (Prunus cerasus var. Marasca). J. Food Eng. 2013, 117, 437–442. [Google Scholar] [CrossRef]
- Dobrinčić, A.; Repajić, M.; Elez Garofulić, I.; Tuđen, L.; Dragović-Uzelac, V.; Levaj, B. Comparison of Different Extraction Methods for the Recovery of Olive Leaves Polyphenols. Processes 2020, 8, 1008. [Google Scholar] [CrossRef]
- Vazquez-Gonzalez, Y.; Ragazzo-Sanchez, J.A.; Calderon-Santoyo, M. Characterization and antifungal activity of jackfruit (Artocarpus heterophyllus Lam.) leaf extract obtained using conventional and emerging technologies. Food Chem. 2020, 330, 127211. [Google Scholar] [CrossRef]
- Pavlić, B.; Kaplan, M.; Bera, O. Microwave-assisted extraction of peppermint polyphenols—Artificial neural networks approach. Food Bioprod. Process. 2019, 118, 258–269. [Google Scholar] [CrossRef]
- Anticona, M.; Blesa, J.; Frigola, A.; Esteve, M.J. High Biological Value Compounds Extraction from Citrus Waste with Non-Conventional Methods. Foods 2020, 9, 811. [Google Scholar] [CrossRef] [PubMed]
- Dragović-Uzelac, V.; Elez Garofulić, I.; Jukić, M.; Penić, M.; Dent, M. The influence of microwave-assisted extraction on the isolation of sage (Salvia officinalis L.) polyphenols. Food Technol. Biotechnol. 2012, 50, 377–383. [Google Scholar]
- Ignat, I.; Volf, I.; Popa, V.I. A critical review of methods for characterisation of polyphenolic compounds in fruits and vegetables. Food Chem. 2011, 126, 1821–1835. [Google Scholar] [CrossRef] [PubMed]
- Prior, R.L.; Wu, X.; Schaich, K. Standardized Methods for the Determination of Antioxidant Capacity and Phenolics in Foods and Dietary Supplements. J. Agric. Food Chem. 2005, 53, 4290–4302. [Google Scholar] [CrossRef]
- Prior, R.L. Oxygen radical absorbance capacity (ORAC): New horizons in relating dietary antioxidants/bioactives and health benefits. J. Funct. Foods 2015, 18, 797–810. [Google Scholar] [CrossRef]
- Huang, D.; Ou, B.; Prior, R.L. The chemistry behind antioxidant capacity assays. J. Agric. Food Chem. 2005, 53, 1841–1856. [Google Scholar] [CrossRef]
- Longo, L.; Scardino, A.; Vasapollo, G. Identification and quantification of anthocyanins in the berries of Pistacia lentiscus L., Phillyrea latifolia L. and Rubia peregrina L. Innov. Food Sci. Emerg. 2007, 8, 360–364. [Google Scholar] [CrossRef]
- Mehenni, C.; Atmani-Kilani, D.; Dumarcay, S.; Perrin, D.; Gerardin, P.; Atmani, D. Hepatoprotective and antidiabetic effects of Pistacia lentiscus leaf and fruit extracts. J. Food Drug Anal. 2006, 24, 653–669. [Google Scholar] [CrossRef]
- Trabelsi, H.; Lajnef, H.B.; Arfa, K.B.; Boukhchina, S. Phenolic Compounds Characterization from Pistacia lentiscus (lentisc) Fruit. J. Chem. Pharm. Res. 2016, 8, 1–8. [Google Scholar]
- Elez Garofulić, I.; Zorić, Z.; Pedisić, S.; Brnčić, M.; Dragović-Uzelac, V. UPLC-MS2 profiling of blackthorn flower polyphenols isolated by ultrasound-assisted extraction. J. Food Sci. 2018, 83, 2782–2789. [Google Scholar] [CrossRef]
- Altıok, E.; Bayçın, D.; Bayraktar, O.; Ülkü, S. Isolation of polyphenols from the extracts of olive leaves (Olea europaea L.) by adsorption on silk fibroin. Sep. Purif. Technol. 2008, 62, 342–348. [Google Scholar] [CrossRef]
- Shortle, E.; O’Grady, M.N.; Gilroy, D.; Furey, A.; Quinn, N.; Kerry, J.P. Influence of extraction technique on the anti-oxidative potential of hawthorn (Crataegus monogyna) extracts in bovine muscle homogenates. Meat Sci. 2014, 98, 828–834. [Google Scholar] [CrossRef] [PubMed]
- Bender, C.; Graziano, S.; Zimmerman, B.F.; Weidlich, H.H. Antioxidant potential of aqueous plant extracts assessed by the cellular antioxidant activity assay. Am. J. Biol. Life Sci. 2014, 2, 72–79. [Google Scholar]
- Khuri, A.I.; Mukhopadhyay, S. Response surface methodology. Advanced Review. Wires Comput. Mol. Sci. 2010, 2, 128–149. [Google Scholar] [CrossRef]
- Yemmen, M.; Landolsi, A.; Ben Hamida, J.; Mégraud, F.; Trabelsi Ayadi, M. Antioxidant activities, anticancer activity and polyphenolics profile, of leaf, fruit and stem extracts of Pistacia lentiscus from Tunisia. Cell. Mol. Biol. 2017, 63, 87–95. [Google Scholar] [CrossRef]
- Rafiee, Z.; Jafari, S.M.; Alami, M.; Khomeiri, M. Microwave-assisted extraction of phenolic compounds from olive leaves; a comparison with maceration. J. Anim. Plant Sci. 2011, 21, 738–745. [Google Scholar]
- Alara, O.R.; Abdurahman, N.H.; Olalere, O.A. Optimization of microwave-assisted extraction of flavonoids and antioxidants from Vernonia amygdalina leaf using response surface methodology. Food Bioprod. Process. 2018, 107, 36–48. [Google Scholar] [CrossRef]
- Xu, D.P.; Li, Y.; Meng, X.; Zhou, T.; Zhou, Y.; Zheng, J.; Zhang, J.J.; Li, H.B. Natural antioxidants in foods and medicinal plants: Extraction, assessment and resources. Int. J. Mol. Sci. 2017, 18, 96. [Google Scholar] [CrossRef]
- Alberto Casazza, A.; Pettinato, M.; Perego, P. Polyphenols from apple skins: A study on microwave-assisted extraction optimization and exhausted solid characterization. Sep. Purif. Technol. 2020, 240, 116640. [Google Scholar] [CrossRef]
- Alonso-Carrillo, N.; Aguilar-Santamaría, M.D.L.Á.; Vernon-Carter, E.J.; Jiménez-Alvarado, R.; Cruz-Sosa, F.; Román-Guerrero, A. Extraction of phenolic compounds from Satureja macrostema using microwave-ultrasound assisted and reflux methods and evaluation of their antioxidant activity and cytotoxicity. Ind. Crop. Prod. 2017, 103, 213–221. [Google Scholar] [CrossRef]
- Guo, Z.; Zhao, B.; Li, H.; Miao, S.; Zheng, B. Optimization of ultrasound-microwave synergistic extraction of prebiotic oligosaccharides from sweet potatoes (Ipomoea batatas L.). Innov. Food Sci. Emerg. Technol. 2019, 54, 51–63. [Google Scholar] [CrossRef]
- Gao, M.; Song, B.Z.; Liu, C.Z. Dynamic microwave-assisted extraction of flavonoids from Saussurea medusa Maxim cultured cells. Biochem. Eng. J. 2006, 32, 79–83. [Google Scholar] [CrossRef]
- Kala, H.K.; Mehta, R.; Sen, K.K.; Tandey, R.; Mandal, V. Critical analysis of research trends and issues in microwave assisted extraction of phenolics: Have we really done enough. Trends Anal. Chem. 2016, 85, 140–152. [Google Scholar] [CrossRef]
- Sik, B.; Lakatos Hanczné, E.; Kapcsándi, V.; Ajtony, Z. Conventional and nonconventional extraction techniques for optimal extraction processes of rosmarinic acid from six Lamiaceae plants as determined by HPLC-DAD measurement. J. Pharm. Biomed. Anal. 2020, 184, 113–173. [Google Scholar] [CrossRef]
- Bozorgi, M.; Memariani, Z.; Mobli, M.; Salehi Surmaghi, M.H.; Shams-Ardekani, M.R.; Rahimi, R. Five Pistacia species (P. vera, P. atlantica, P. terebinthus, P. khinjuk, and P. lentiscus): A Review of Their Traditional Uses, Phytochemistry, and Pharmacology. Sci. World J. 2013, 2013, 1–33. [Google Scholar] [CrossRef]
- Wyrepkowski, C.C.; Maldonado Gomes da Costa, D.L.; Sinhorin, A.P.; Vilegas, W.; De Grandis, R.A.; Resende, F.A.; Varanda, E.A.; Campaner dos Santos, L. Characterization and Quantification of the Compounds of the Ethanolic Extract from Caesalpinia ferrea Stem Bark and Evaluation of Their Mutagenic Activity. Molecules 2014, 19, 16039–16057. [Google Scholar] [CrossRef]
- Remila, S.; Atmani-Kilani, D.; Delemasure, S.; Connat, J.L.; Azib, L.; Richard, T.; Atmani, D. Antioxidant, cytoprotective, anti-inflammatory and anticancer activities of Pistacia lentiscus (Anacardiaceae) leaf and fruit extracts. Eur. J. Integr. Med. 2015, 7, 274–286. [Google Scholar] [CrossRef]
- Baratto, M.C.; Tattini, M.; Galardi, C.; Pinelli, P.; Romani, A.; Visioli, F.; Basosi, R.; Pogni, R. Antioxidant Activity of Galloyl Quinic Derivatives Isolated from P. lentiscus Leaves. Free Radic. Res. 2003, 37, 405–412. [Google Scholar] [CrossRef]
- Romani, A.; Pinelli, P.; Galardi, C.; Mulinacci, N.; Tattini, M. Identification and Quantification of Galloyl Derivatives, Flavonoid Glycosides and Anthocyanins in Leaves of Pistacia lentiscus L. Phytochem. Anal. 2002, 13, 79–86. [Google Scholar] [CrossRef]
- Clifford, M.N.; Johnston, K.L.; Knight, S.; Kuhnert, N. Hierarchical Scheme for LC-MSn Identification of Chlorogenic Acids. J. Agric. Food Chem. 2003, 51, 2900–2911. [Google Scholar] [CrossRef] [PubMed]
- Osterc, G.; Cunja, V.; Mikulic-Petkovseka, M.; Schmitzer, V.; Stampar, F.; Bavcon, J. Foliage identification of different autochtonous common cyclamen genotypes (Cyclamen purpurascens Mill.) using various biochemical parameters. Sci. Hortic. 2014, 173, 37–44. [Google Scholar] [CrossRef]
- Carocho, M.; Barros, L.; Bento, A.; Santos-Buelga, C.; Morales, P.; Ferreira, I.C.F.R. Castanea sativa Mill. Flowers amongst the Most Powerful Antioxidant Matrices: A Phytochemical Approach in Decoctions and Infusions. Biomed. Res. Int. 2014, 2014, 232956. [Google Scholar] [CrossRef] [PubMed]
- Kawashty, S.A.; Mosharrafa, S.A.M.; El-Gibali, M.; Saleh, N.A.M. The favonoids of four Pistacia species in Egypt. Biochem. Syst. Ecol. 2000, 28, 915–917. [Google Scholar] [CrossRef]
- Barros, L.; Tiago Alves, C.; Duenas, M.; Silvac, S.; Oliveirac, R.; Carvalhoa, A.M.; Henriques, M.; Santos-Buelga, C.; Ferreira, I.C.F.R. Characterization of phenolic compounds in wild medicinal flowers from Portugal by HPLC–DAD–ESI/MS and evaluation of antifungal properties. Ind. Crop. Prod. 2013, 44, 104–110. [Google Scholar] [CrossRef]
- Pinheiro, P.F.; Justino, G.C. Structural Analysis of Flavonoids and Related Compounds—A Review of Spectroscopic Applications. In Phytochemicals—A Global Perspective of Their Role in Nutrition and Health; Rao, V., Ed.; InTech: Rijeka, Croatia, 2012; pp. 33–56. [Google Scholar]
- Zitouni, A.; Belyagoubi-Benhammou, N.; Ghembaza, N.; Toul, F.; AtikBekkara, F. Assessment of Phytochemical Composition and Antioxidant Properties of Extracts from the Leaf, Stem, Fruit and Root of Pistacia lentiscus L. Int. J. Pharmacogn. Phytochem. Res. 2016, 8, 627–633. [Google Scholar]
| Sample | Temperature (°C) | Microwave Power (W) | Time (min) | Total Phenolic Content (mg GAE/g) | |
|---|---|---|---|---|---|
| Leaves | Fruit | ||||
| 1 | 50 | 200 | 4 | 94.95 ± 3.43 | 29.54 ± 0.81 |
| 2 | 50 | 200 | 12 | 104.00 ± 4.04 | 33.00 ± 0.20 |
| 3 | 50 | 500 | 4 | 95.14 ± 3.43 | 30.57 ± 1.21 |
| 4 | 50 | 500 | 12 | 99.76 ± 0.30 | 37.71 ± 2.12 |
| 5 | 70 | 200 | 4 | 85.71 ± 0.2 | 35.86 ± 1.31 |
| 6 | 70 | 200 | 12 | 100.90 ± 1.41 | 41.72 ± 2.82 |
| 7 | 70 | 500 | 4 | 85.14 ± 2.63 | 36.78 ± 2.92 |
| 8 | 70 | 500 | 12 | 110.18 ± 2.22 | 38.29 ± 0.81 |
| 9 | 43 | 350 | 8 | 88.63 ± 0.91 | 22.69 ± 1.41 |
| 10 | 77 | 350 | 8 | 110.14 ± 3.64 | 34.36 ± 2.52 |
| 11 | 60 | 98 | 8 | 94.19 ± 2.42 | 38.14 ± 0.20 |
| 12 | 60 | 602 | 8 | 107.93 ± 2.52 | 34.43 ± 2.53 |
| 13 | 60 | 350 | 1 | 67.72 ± 1.51 | 27.00 ± 2.22 |
| 14 | 60 | 350 | 15 | 94.05 ± 5.05 | 40.82 ± 1.01 |
| 15 (C) | 60 | 350 | 8 | 94.67 ± 1.21 | 33.50 ± 0.60 |
| 16 (C) | 60 | 350 | 8 | 92.57 ± 3.64 | 34.25 ± 1.01 |
| Factor | Total Phenolic Content | |||
|---|---|---|---|---|
| Leaves | Fruit | |||
| F-Ratio | p-Value | F-Ratio | p-Value | |
| Temperature (X1) | 19.99 | 0.14 | 448.01 | 0.03 * |
| Microwave power (X2) | 25.58 | 0.12 | 2.35 | 0.37 |
| Time (X3) | 320.93 | 0.04 * | 447.32 | 0.03 * |
| X1X2 | 9.23 | 0.20 | 30.25 | 0.11 |
| X1X3 | 39.99 | 0.10 | 4.64 | 0.28 |
| X2X3 | 1.67 | 0.42 | 0.20 | 0.73 |
| Lack of fit | 27.29 | 0.14 | 34.70 | 0.13 |
| Model | 226.6418−3.9910 X1 + 0.0261 X12 − 0.1614 X2 + 0.0010 X22 + 0.0012 X3 − 0.2254 X32 + 0.0110 X1X2 + 0.0830 X1X3 + 0.0110 X2X3 | −46.7913 + 2.1914 X1 − 0.0131 X12 − 0.0034 X2 + 0.0001 X22 + 0.9019 X3 + 0.0312 X32 − 0.0007 X1X2 − 0.0101 X1X3 − 0.0011 X2X3 | ||
| R2 | 0.83 | 0.87 | ||
| Total Phenolic Content (mg/g) | Optimized Microwave Assisted Ekstraction | Conventional Extraction | |||
|---|---|---|---|---|---|
| Predicted | Experimental | ||||
| Leaves | Temperature, °C | 69 | 112.71 | 108.14 ± 2.12 | 108.71 ± 1.87 |
| Microwave power, W | 512 | ||||
| Time, min | 12 | ||||
| Fruit | Temperature, °C | 75 | 41.85 | 41.14 ± 0.76 | 42.71 ± 0.93 |
| Microwave power, W | 602 | ||||
| Time, min | 15 | ||||
| Compound | Rt, min | Cone Voltage (V) | Collision Energy (V) | Ionization Mode | Precursor Ion (m/z) | Fragment Ions (m/z) | Tentative Identification | Concentration mg/100 g | |
|---|---|---|---|---|---|---|---|---|---|
| Leaves | Fruit | ||||||||
| 1 | 0.905 | 80 | 5 | - | 367 | 193 | feruloylquinic acid | 2.29 ± 0.09 | 1.38 ± 0.06 |
| 2 | 0.916 | 80 | 10 | - | 337 | 173 | p-coumaroylquinic acid 1 | 0.21 ± 0.01 | 0.22 ± 0.01 |
| 3 | 0.918 | 80 | 10 | - | 353 | 191 | 5-O-caffeoylquinic acid | 0.46 ± 0.02 | 0.48 ± 0.02 |
| 4 | 0.979 | 100 | 10 | - | 331 | 169 | monogalloyl glucose | 18.76 ± 0.26 | 15.08 ± 0.18 |
| 5 | 1.313 | 100 | 10 | - | 169 | 125 | gallic acid * | 4.34 ± 0.10 | 59.27 ± 0.65 |
| 6 | 2.144 | 100 | 10 | - | 343 | 191 | 5-O-galloylquinic acid | 3.08 ± 0.12 | 0.60 ± 0.02 |
| 7 | 2.639 | 80 | 10 | - | 353 | 191 | chlorogenic acid * | 0.27 ± 0.01 | 0.22 ± 0.01 |
| 8 | 3.251 | 100 | 10 | - | 495 | 343 | digalloylquinic acid | 605.88 ± 4.33 | 57.39 ± 0.61 |
| 9 | 3.346 | 120 | 5 | + | 579 | 427 | procyanidin B1 * | 13.58 ± 0.19 | 20.69 ± 0.19 |
| 10 | 3.468 | 120 | 5 | + | 579 | 427 | procyanidin B2 * | 10.72 ± 0.15 | 11.93 ± 0.12 |
| 11 | 3.602 | 80 | 10 | - | 337 | 163 | p-coumaroylquinic acid 2 | 0.33 ± 0.01 | 0.27 ± 0.01 |
| 12 | 3.935 | 100 | 10 | + | 291 | 139 | epicatechin * | 31.66 ± 0.31 | 18.28 ± 0.17 |
| 13 | 3.944 | 100 | 5, 10 | + | 291 | 139 | catechin * | 31.70 ± 0.26 | 18.56 ± 0.16 |
| 14 | 5.171 | 100 | 5, 15 | + | 459 | 139 | epigallocatechin gallate * | 9.16 ± 0.17 | 1.76 ± 0.03 |
| 15 | 5.208 | 100 | 10 | - | 647 | 495 | trigalloylquinic acid | 102.10 ± 2.09 | 13.59 ± 0.11 |
| 16 | 5.836 | 120 | 15 | + | 627 | 319 | myricetin rutinoside | 528.90 ± 4.27 | 172.63 ± 2.98 |
| 17 | 5.94 | 80 | 10 | - | 163 | 119 | p-coumaric acid * | ni | 0.89 ± 0.02 |
| 18 | 5.992 | 120 | 15 | + | 495 | 319 | myricetin glucuronide | 750.00 ± 6.11 | 251.44 ± 2.17 |
| 19 | 5.992 | 140 | 25 | + | 319 | 273 | myricetin * | 304.09 ± 2.98 | 14.64 ± 0.16 |
| 20 | 6.549 | 120 | 5 | + | 611 | 303 | quercetin-3-hexoside | 31.94 ± 0.23 | 23.94 ± 0.19 |
| 21 | 6.665 | 120 | 15 | + | 465 | 319 | myricetin rhamnoside | 1782.39 ± 10.78 | 24.72 ± 0.21 |
| 22 | 6.77 | 100 | 5, 15 | + | 442.9 | 139 | epicatechin gallate * | 1.47 ± 0.06 | 1.26 ± 0.03 |
| 23 | 6.886 | 100 | 5 | + | 465 | 303.1 | quercetin-3-glucoside * | 39.33 ± 0.40 | 156.61 ± 2.77 |
| 24 | 6.946 | 100 | 5 | + | 449 | 287 | kaempferol-3-hexoside | 19.44 ± 0.29 | 1.75 ± 0.04 |
| 25 | 7.309 | 120 | 15 | + | 595 | 287 | kaempferol rhamnosyl hexoside | 2.44 ± 0.07 | 1.91 ± 0.04 |
| 26 | 7.312 | 120 | 15 | + | 595 | 287 | kaempferol-3-rutinoside * | 2.41 ± 0.09 | 1.93 ± 0.05 |
| 27 | 7.342 | 100 | 5 | + | 435 | 303 | quercetin pentoside | 10.64 ± 0.15 | 1.07 ± 0.03 |
| 28 | 7.452 | 120 | 15 | + | 581 | 287 | kaempferol pentosyl hexoside | 0.21 ± 0.01 | 0.17 ± 0.00 |
| 29 | 7.702 | 100 | 5 | + | 449 | 303 | quercetin rhamnoside | 130.92 ± 2.23 | 1.90 ± 0.03 |
| 30 | 8.373 | 120 | 5 | + | 419 | 287 | kaempferol pentoside | 2.13 ± 0.09 | 1.60 ± 0.03 |
| 31 | 8.585 | 120 | 5 | + | 433 | 287 | kaempferol rhamnoside | 11.29 ± 0.21 | 2.17 ± 0.04 |
| 32 | 9.989 | 140 | 35 | + | 287 | 153 | luteolin * | 26.54 ± 0.34 | 1.54 ± 0.03 |
| 33 | 11.162 | 120 | 15 | + | 637 | 287 | kaempferol acetyl rutinoside | 0.20 ± 0.01 | 0.21 ± 0.01 |
| 34 | 11.185 | 80 | 30 | + | 271 | 153 | apigenin * | 2.02 ± 0.08 | 0.15 ± 0.00 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elez Garofulić, I.; Kruk, V.; Martić, A.; Martić, I.; Zorić, Z.; Pedisić, S.; Dragović, S.; Dragović-Uzelac, V. Evaluation of Polyphenolic Profile and Antioxidant Activity of Pistacia lentiscus L. Leaves and Fruit Extract Obtained by Optimized Microwave-Assisted Extraction. Foods 2020, 9, 1556. https://doi.org/10.3390/foods9111556
Elez Garofulić I, Kruk V, Martić A, Martić I, Zorić Z, Pedisić S, Dragović S, Dragović-Uzelac V. Evaluation of Polyphenolic Profile and Antioxidant Activity of Pistacia lentiscus L. Leaves and Fruit Extract Obtained by Optimized Microwave-Assisted Extraction. Foods. 2020; 9(11):1556. https://doi.org/10.3390/foods9111556
Chicago/Turabian StyleElez Garofulić, Ivona, Valentina Kruk, Ana Martić, Ivan Martić, Zoran Zorić, Sandra Pedisić, Sanja Dragović, and Verica Dragović-Uzelac. 2020. "Evaluation of Polyphenolic Profile and Antioxidant Activity of Pistacia lentiscus L. Leaves and Fruit Extract Obtained by Optimized Microwave-Assisted Extraction" Foods 9, no. 11: 1556. https://doi.org/10.3390/foods9111556
APA StyleElez Garofulić, I., Kruk, V., Martić, A., Martić, I., Zorić, Z., Pedisić, S., Dragović, S., & Dragović-Uzelac, V. (2020). Evaluation of Polyphenolic Profile and Antioxidant Activity of Pistacia lentiscus L. Leaves and Fruit Extract Obtained by Optimized Microwave-Assisted Extraction. Foods, 9(11), 1556. https://doi.org/10.3390/foods9111556








