Dietary Exposure Estimation to Chemicals Transferred from Milk and Dairy Products Packaging Materials in Spanish Child and Adolescent Population
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents and Analytical Standards
2.2. Samples
2.3. Equipments and Analytical Conditions
2.3.1. Fourier-Transform Infrared Spectroscopy (FTIR)
2.3.2. Confocal Raman Microscopy
2.3.3. Gas Chromatography Coupled to Mass Spectrometry (GC-MS)
2.3.4. Preparation of Food Samples and Exposure Estimation
2.3.5. Extraction Procedure-Foodstuffs
2.3.6. Quality Assurance/Quality Control (QA/QC)
3. Results and Discussion
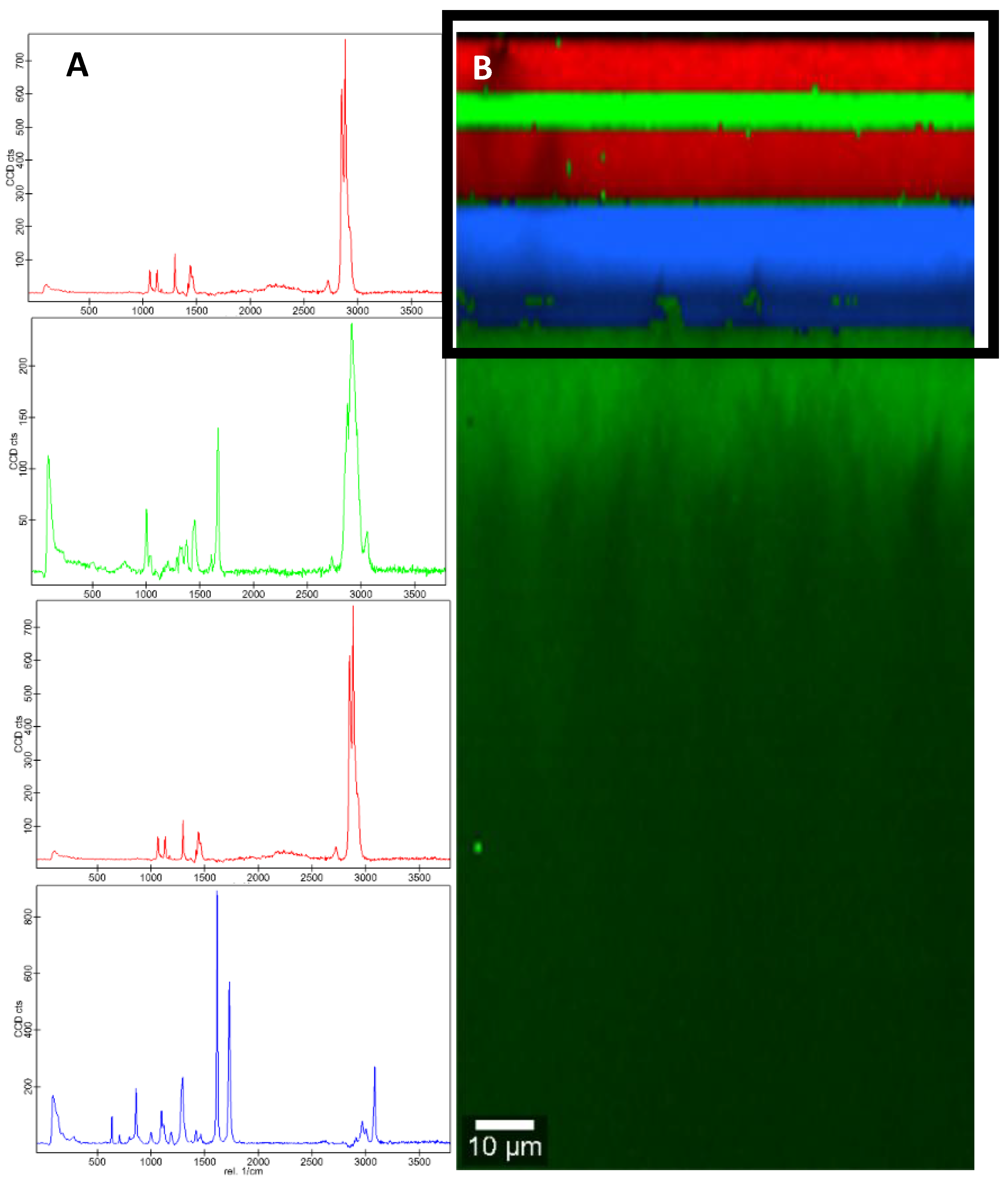
3.1. Characterization of Packaging Materials by FTIR and Confocal Raman Microscopy
3.2. Identification of Potential Migrants in Packaging Materials
3.3. Analytical Parameters
3.4. Migrants Concentration in Food and Dietary Exposure Estimation
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Joint Guidance of EFSA. FAO and WHO—Towards a harmonized Total Diet Study approach: A guidance document. EFSA J. 2011, 9, 2450–2516. Available online: https://apps.who.int/iris/handle/10665/75330 (accessed on 4 June 2020). [CrossRef]
- Sakhi, A.K.; Lillegaard, I.T.L.; Voorspoels, S.; Carlsen, M.H.; Løken, E.B.; Brantsæter, A.L.; Haugen, M.; Meltzer, H.M.; Thomsen, C. Concentrations of phthalates and bisphenol A in Norwegian foods and beverages and estimated dietary exposure in adults. Environ. Int. 2014, 73, 259–269. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.-L. Phthalate Esters in Foods: Sources, Occurrence, and Analytical Methods. Compr. Rev. Food Sci. Food Saf. 2010, 9, 21–43. [Google Scholar] [CrossRef]
- Bradley, E.; Burden, R.A.; Leon, I.; Mortimer, D.N.; Speck, D.R.; Castle, L. Determination of phthalate diesters in foods. Food Addit. Contam. Part A 2013, 30, 722–734. [Google Scholar] [CrossRef] [PubMed]
- Materials, E.A.P.A.; Cep, E.P.O.F.C.; Silano, V.; Baviera, J.M.B.; Bolognesi, C.; Chesson, A.; Cocconcelli, P.S.; Crebelli, R.; Gott, D.M.; Grob, K.; et al. Review and priority setting for substances that are listed without a specific migration limit in Table 1 of Annex 1 of Regulation 10/2011 on plastic materials and articles intended to come into contact with food. EFSA J. 2020, 18, e06124. [Google Scholar] [CrossRef]
- European Food Safety Authority. Toxicological evaluation of benzophenone. EFSA J. 2009, 7, 1104. [Google Scholar] [CrossRef]
- Fierens, T.; Standaert, A.; Cornelis, C.; Sioen, I.; De Henauw, S.; Willems, H.; Bellemans, M.; De Maeyer, M.; Van Holderbeke, M. A semi-probabilistic modelling approach for the estimation of dietary exposure to phthalates in the Belgian adult population. Environ. Int. 2014, 73, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Bradley, E.; Burden, R.A.; Bentayeb, K.; Driffield, M.; Harmer, N.; Mortimer, D.N.; Speck, D.R.; Ticha, J.; Castle, L. Exposure to phthalic acid, phthalate diesters and phthalate monoesters from foodstuffs: UK total diet study results. Food Addit. Contam. Part A 2013, 30, 735–742. [Google Scholar] [CrossRef] [PubMed]
- Gelbke, H.-P.; Banton, M.; Block, C.; Dawkins, G.; Eisert, R.; Leibold, E.; Pemberton, M.; Puijk, I.M.; Sakoda, A.; Yasukawa, A. Risk assessment for migration of styrene oligomers into food from polystyrene food containers. Food Chem. Toxicol. 2019, 124, 151–167. [Google Scholar] [CrossRef]
- Suarez, V.M.; Mañas, J.R.; Fernández, R.S.; De Dios, T.R. Spanish National dietary survey on children and adolescents. EFSA Support. Publ. 2015, 12. [Google Scholar] [CrossRef]
- Application Note. Correlative Raman Imaging of Polymeric Materials. Witec Focus Innovations. Available online: https://www.witec.de/assets/Literature/Files/WITec-AppNote-PolymersCorrelativeRamanMicroscopy-WebVersion.pdf (accessed on 10 June 2020).
- Ibarra, V.A.G.; Sendón, R.; Bustos, J.; Losada, P.P.; De Quirós, A.R.-B. Estimates of dietary exposure of Spanish population to packaging contaminants from cereal based foods contained in plastic materials. Food Chem. Toxicol. 2019, 128, 180–192. [Google Scholar] [CrossRef] [PubMed]
- GEMS-Food Euro. Second workshop on reliable evaluation of low-level contamination of food. In Proceedings of the Workshop in the Frame of GEMS-Food Euro, Kulmbach, Germany, 26–27 May 1995. EUR/HFA Target 22. [Google Scholar]
- ACS, American Chemical Society. Subcommittee of environmental analytical chemistry. Anal. Chem. 1980, 52, 2242–2280. [Google Scholar]
- Castle, L.; Offen, C.P.; Baxter, M.J.; Gilbert, J. Migration studies from paper and board food packaging materials. 1. Compositional analysis. Food Addit. Contam. 1997, 14, 35–44. [Google Scholar] [CrossRef]
- Félix, J.S.; Isella, F.; Bosetti, O.; Nerín, C. Analytical tools for identification of non-intentionally added substances (NIAS) coming from polyurethane adhesives in multilayer packaging materials and their migration into food simulants. Anal. Bioanal. Chem. 2012, 403, 2869–2882. [Google Scholar] [CrossRef]
- Aznar, M.; Vera, P.; Canellas, E.; Nerín, C.; Mercea, P.; Störmer, A. Composition of the adhesives used in food packaging multilayer materials and migration studies from packaging to food. J. Mater. Chem. 2011, 21, 4358. [Google Scholar] [CrossRef]
- Fabbri, D.; Trombini, C.; Vassura, I. Analysis of Polystyrene in Polluted Sediments by Pyrolysis--Gas Chromatography--Mass Spectrometry. J. Chromatogr. Sci. 1998, 36, 600–604. [Google Scholar] [CrossRef]
- Choi, J.O.; Jitsunari, F.; Asakawa, F.; Lee, D.S. Migration of styrene monomer, dimers and trimers from polystyrene to food simulants. Food Addit. Contam. 2005, 22, 693–699. [Google Scholar] [CrossRef]
- Tang, W.; Hemm, I.; Eisenbrand, G. Estimation of human exposure to styrene and ethylbenzene. Toxicology 2000, 144, 39–50. [Google Scholar] [CrossRef]
- Dupáková, Z.; Dobiáš, J.; Votavová, L.; Klaudisová, K.; Voldrich, M. Occurrence of extractable ink residuals in packaging materials used in the Czech Republic. Food Addit. Contam. Part A 2010, 27, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Bach, C.; Dauchy, X.; Séverin, I.; Munoz, J.-F.; Etienne, S.; Chagnon, M.-C. Effect of temperature on the release of intentionally and non-intentionally added substances from polyethylene terephthalate (PET) bottles into water: Chemical analysis and potential toxicity. Food Chem. 2013, 139, 672–680. [Google Scholar] [CrossRef]
- Nieva-Echevarría, B.; Manzanos, M.J.; Goicoechea, E.; Guillén, M.D. 2,6-Di-Tert-Butyl-Hydroxytoluene and Its Metabolites in Foods. Compr. Rev. Food Sci. Food Saf. 2014, 14, 67–80. [Google Scholar] [CrossRef]
- Lago, M.A.; Ackerman, L.K. Identification of print-related contaminants in food packaging. Food Addit. Contam. Part A 2016, 33, 1–12. [Google Scholar] [CrossRef]
- Bentayeb, K.; Ackerman, L.K.; Lord, T.; Begley, T. Non-visible print set-off of photoinitiators in food packaging: Detection by ambient ionisation mass spectrometry. Food Addit. Contam. Part A 2013, 30, 750–759. [Google Scholar] [CrossRef]
- Fierens, T.; Servaes, K.; Van Holderbeke, M.; Geerts, L.; De Henauw, S.; Sioen, I.; Vanermen, G. Analysis of phthalates in food products and packaging materials sold on the Belgian market. Food Chem. Toxicol. 2012, 50, 2575–2583. [Google Scholar] [CrossRef]
- Goulas, A.; Anifantaki, K.; Kolioulis, D.; Kontominas, M. Migration of di-(2-ethylhexylexyl)Adipate Plasticizer from Food-Grade Polyvinyl Chloride Film into Hard and Soft Cheeses. J. Dairy Sci. 2000, 83, 1712–1718. [Google Scholar] [CrossRef]
- Quintana, R.; Persenaire, O.; Lemmouchi, Y.; Sampson, J.; Martin, S.; Bonnaud, L.; Dubois, P. Enhancement of cellulose acetate degradation under accelerated weathering by plasticization with eco-friendly plasticizers. Polym. Degrad. Stab. 2013, 98, 1556–1562. [Google Scholar] [CrossRef]
- Turco, V.L.; Di Bella, G.; Potortì, A.G.; Fede, M.R.; Dugo, G. Determination of plasticizer residues in tea by solid phase extraction–gas chromatography–mass spectrometry. Eur. Food Res. Technol. 2014, 240, 451–458. [Google Scholar] [CrossRef]
- Cao, X.-L.; Zhao, W.; Dabeka, R. Di-(2-ethylhexyl) adipate and 20 phthalates in composite food samples from the 2013 Canadian Total Diet Study. Food Addit. Contam. Part A 2015, 32, 1893–1901. [Google Scholar] [CrossRef] [PubMed]
- Jia, W.; Chu, X.; Ling, Y.; Huang, J.; Chang, J. Analysis of phthalates in milk and milk products by liquid chromatography coupled to quadrupole Orbitrap high-resolution mass spectrometry. J. Chromatogr. A 2014, 1362, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Van Holderbeke, M.; Geerts, L.; Vanermen, G.; Servaes, K.; Sioen, I.; De Henauw, S.; Fierens, T. Determination of contamination pathways of phthalates in food products sold on the Belgian market. Environ. Res. 2014, 134, 345–352. [Google Scholar] [CrossRef]
- Cariou, R.; Larvor, F.; Monteau, F.; Marchand, P.; Bichon, E.; Dervilly-Pinel, G.; Antignac, J.-P.; Le Bizec, B. Measurement of phthalates diesters in food using gas chromatography–tandem mass spectrometry. Food Chem. 2016, 196, 211–219. [Google Scholar] [CrossRef] [PubMed]
- Sirot, V.; Hommet, F.; Tard, A.; Leblanc, J.-C. Dietary acrylamide exposure of the French population: Results of the second French Total Diet Study. Food Chem. Toxicol. 2012, 50, 889–894. [Google Scholar] [CrossRef]
- World Health Organization. Diethyl Phthalate-Concise International Chemical Assessment Document 52; World Health Organisation: Geneva, Switzerland, 2003; ISBN 92-4-153052-9. [Google Scholar]
- Draft Scientific Opinion. Draft update of the risk assessment of di-butylphthalate (DBP), butyl-benzyl-phthalate (BBP), bis(2-2 ethylhexyl)phthalate (DEHP), di-isononylphthalate (DINP) and di-isodecylphthalate (DIDP) for use in food contact materials. EFSA J. 2019, 17, e05838. [Google Scholar] [CrossRef]
- Cirillo, T.; Latini, G.; Castaldi, M.A.; DiPaola, L.; Fasano, E.; Esposito, F.; Scognamiglio, G.; Di Francesco, F.; Cobellis, L. Exposure to Di-2-Ethylhexyl Phthalate, Di-N-Butyl Phthalate and Bisphenol A through Infant Formulas. J. Agric. Food Chem. 2015, 63, 3303–3310. [Google Scholar] [CrossRef]
- Sui, H.-X.; Zhang, L.; Wu, P.-G.; Song, Y.; Yong, L.; Yang, D.-J.; Jiang, D.-G.; Liu, Z. Concentration of di(2-ethylhexyl) phthalate (DEHP) in foods and its dietary exposure in China. Int. J. Hyg. Environ. Health 2014, 217, 695–701. [Google Scholar] [CrossRef] [PubMed]
- Sioen, I.; Fierens, T.; Van Holderbeke, M.; Geerts, L.; Bellemans, M.; De Maeyer, M.; Servaes, K.; Vanermen, G.; Boon, P.E.; De Henauw, S. Phthalates dietary exposure and food sources for Belgian preschool children and adults. Environ. Int. 2012, 48, 102–108. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority. 18th list of substances for food contact materials-Opinion of the Scientific Panel on food additives, flavourings, processing aids and materials in contact with food. EFSA J. 2008, 6, 628. [Google Scholar] [CrossRef]
- European Food Safety Authority. International Frameworks Dealing with Human Risk Assessment of Combined Exposure to Multiple Chemicals. EFSA J. 2013, 11, 3313–3382. [Google Scholar] [CrossRef]

| Code | Type of Sample | Part of the Sample | Type of Material | Thickness (µm) | Fat Content | |
|---|---|---|---|---|---|---|
| Internal Side | External Side | |||||
| LE01 | Whole milk | Packaging (P) | PE | PE | 433 ± 0.6 | 3.6 g/100 mL (Satur. 2.5 g) |
| LE02 | Whole milk | Packaging | PE | PE | 431 ± 0.6 | 3.6 g/100 mL (Satur. 2 g) |
| LE03 | Whole milk | Packaging | PE | PE | 296 ± 1.0 | 3.6 g/100 mL (Satur. 2.5 g) |
| LS01 | Semi-skimmed milk | Packaging | PE | PE | 430 ± 0.6 | 1.6 g/100 mL (Satur. 1.1 g) |
| LS02 | Semi-skimmed milk | Packaging | PE | PE | 454 ± 1.2 | 1.9 g/100 mL (Satur. 1.1 g) |
| YN01 | Natural yogurt | Lid (L) | PES | NC | 59 ± 1.0 | 2.9 g/100 mL (Satur. 1.8 g) |
| Packaging | PS | PS | 252 ± 2.0 | |||
| YS01 | Strawberry yogurt | Lid | PES | NC | 65 ± 0.6 | 1.9 g/100 g (Satur. 1.1 g) |
| Packaging | PS | PS | 245 ± 2.0 | |||
| FN01 | Egg flan with caramel | Lid | SBI | NC | 106 ± 1.2 | 1.8 g/100 g (Satur. 0.6 g) |
| Packaging | PP | PP | 667 ± 1.5 | |||
| FN02 | Egg flan | Lid | MP | PES | 73 ± 0.6 | 3.2 g/100 g (Satur. 1.2 g) |
| Packaging | VP | PR | 186 ± 1.2 | |||
| FN03 | Custard | Lid | PES | NC | 70 ± 0.6 | 3 g/100 g (Satur. 1.8 g) |
| Packaging | PS | PS | 300 ± 1.0 | |||
| QS01 | Semi-cured cheese | Lid | PE | PES | 90 ± 1.0 | 35 g/100 g (Satur. 24 g) |
| Packaging | PE | PET | 272 ± 1.2 | |||
| Intermediate sheet | PS | PS | 56 ± 0.6 | |||
| QL01 | Molten cheese | External packaging (E) | PE | PES | 51 ± 0.6 | 13.5 g/100 g (Satur. 9 g) |
| Internal packaging (I) | PE | PP | 25 ± 0 | |||
| QF01 | Mozzarella | Packaging | PE | Nylon (PA) | 70 ± 0.6 | 18 g/100 g (Satur. 13 g) |
| QF02 | Pasteurized cheese | Lid | PES | PES | 38 ± 1.0 | 14 g/100 g (Satur. 9.6 g) |
| Packaging | PS | SB | 162 ± 1.5 | |||
| TR (min) | CAS No. | Compound Name | TC | Ion | Milk | Cheese | Yogurt and Dairy Desserts |
|---|---|---|---|---|---|---|---|
| 6.25 | 110-43-0 | 2-Heptanone | II | 43, 58, 71 | LE01 | ||
| 6.29 | 100-42-5 | Styrene | I | 104, 78, 51 | LS01, LS02 | QS01_I, QF02_P | YN01_P, YS01_P, FN01_L, FN03_P/L |
| 6.62 | 497-23-4 | 2(5H)-Furanone | III | 55, 84, 39 | YN01_L, YS01_L | ||
| 6.80 | 98-82-8 | Cumene | I | 105, 120, 79 | QF02_P | YN01_P, YS01_P, FN03_P | |
| 7.32 | 103-65-1 | n-Propylbenzene | I | 91, 120, 65 | QF02_P | FN03_P | |
| 7.01 | 6137-06-0 | 4-Methyl-2-heptanone | II | 43, 58, 85 | FN03_P | ||
| 7.50 | 620-02-0 | 5-Methyl-2-furfural | III | 110, 53, 81 | FN02_P | ||
| 7.57 | 100-52-7 | Benzaldehyde | I | 105, 77, 51 | LE01, LE02, LS01, LS02 | QF02_P | YN01_P/L, YS01_P/L, FN03_P/L |
| 7.87 | 98-83-9 | Alpha-methylstyrene | I | 118, 103, 78 | YN01_P, YS01_P, FN01_L, FN03_P | ||
| 8.19 | 111-90-0 | Diethylene glycol monoethyl ether | I | 45, 59, 72 | LE01, LE02, LE03, LS01, LS02 | ||
| 8.35 | 93-53-8 | 2-Phenylpropanal | I | 105, 134, 91 | QF02_P | ||
| 8.38 | 13429-07-7 | 1-(2-Methoxypropoxy)-2-propanol | III | 59, 73, 104 | LE02, LS02 | ||
| 8.58 | 110-98-5 | 1,1′-Oxydi-2-propanol | III | 45, 89, 59 | LE01, LE02, LE03, LS01, LS02 | ||
| 8.67 | 104-76-7 | 2-Ethyl-1-hexanol | I | 57, 43, 83 | FN03_P | ||
| 8.98 | 122-78-1 | Phenylacetaldehyde | I | 91, 120, 65 | QF02_P | FN03_P | |
| 9.23 | 110-03-2 | 2,5-Dimethyl-2,5-hexanediol | III | 43, 59, 70 | FN01_P | ||
| 9.36 | 96-09-3 | Styrene-7,8-oxide | III | 91, 119, 63 | QF02_P | ||
| 9.40 | 98-86-2 | Acetophenone | I | 105, 77, 120 | LE02, LS02 | QF02_P | YN01_P, YS01_P, FN03_P |
| 9.79 | 821-55-6 | 2-Nonanone | II | 58, 43, 71 | LE01 | ||
| 10.75 | 28564-83-2 | 2,3-Dihydro-3,5-dihydroxy-6-methyl-4H-pyran-4-one | III | 144, 101, 43 | FN02_P | ||
| 11.18 | 65-85-0 | Benzoic acid | I | 105, 122, 77 | YN01_P/L, YS01_P/L, FN02_P | ||
| 11.55 | 106-32-1 | Ethyl caprylate | I | 88, 101, 127 | LE01, LS01 | ||
| 12.05 | 103-11-7 | 2-Ethylhexyl acrylate | I | 55, 70, 83 | LE01, LS01 | ||
| 12.06 | 122-99-6 | Phenoxyethanol | II | 94, 138, 77 | YN01_P, YS01_P | ||
| 12.12 | 67-47-0 | 5-Hidroxymethyl-2-furfuraldehyde | III | 95, 126, 69 | FN01_P, FN02_P | ||
| 12.60 | 105-60-2 | Caprolactam * | III | 113, 55, 85 | LE01, LE02, LS01, LS02 | QF01, QF02_L | YN01_P/L, YS01_L/P, FN01_P/L, FN02_P, FN03_P/L |
| 13.00 | 104-55-2 | Cinnamaldehyde | I | 131, 103, 77 | LE01, LE02, LS01, LS02 | FN03_P/L | |
| 13.39 | 93-56-1 | Styrene glycol | I | 107, 79, 51 | FN03_P | ||
| 13.49 | 85-44-9 | Phthalic anhydride | III | 104, 76, 148 | FN01_L, FN03_P | ||
| 13.81 | 102-76-1 | Triacetin * | I | 43, 103, 145 | QL01_E | YN01_L, YS01_L | |
| 14.00 | 91-08-7 | 2,6-Toluene diisocyanate * | III | 174, 146, 118 | QF02_L | YN01_L, YS01_L, FN02_P, FN03_L | |
| 14.10 | 584-84-9 | 2,4-Toluene diisocyanate * | III | 174, 145, 132 | QF02_L | YN01_L, YS01_L | |
| 14.09 | 97-53-0 | Eugenol | I | 164, 149, 103 | LE01, LE02, LE03, LS01, LS02 | ||
| 15.00 | 480-63-7 | Mesitylene carboxylic acid | I | 146, 164, 118 | FN02_P/L | ||
| 15.28 | 101-81-5 | Diphenylmethane | III | 167, 153 | YN01_L, YS01_L, FN03_L | ||
| 15.67 | 719-22-2 | 2,6-Di-tert-butyl-p-benzoquinone * | II | 177, 135, 220 | LE03, LS02 | QS01_L, QF02_L/P | YN01_P/L, YS01_P/L, FN01_P/L, FN03_P/L |
| 15.78 | 2607-52-5 | 2,6-Di-tert-butyl-4-methylene-2,5-cyclohexadienone | II | 161, 203, 175 | QS01_L, QL01_I | FN02_L | |
| 15.81 | 112-53-8 | 1-Dodecanol | I | 55, 69, 83 | LE01, LE02, LS01, LS02 | FN03_L | |
| 16.22 | 128-37-0 | Butylated hydroxytoluene * | II | 205, 220, 177 | LE01, LE02, LE03, LS01, LS02 | QS01_L/P/I, QL01_I, QF02_L/P | YN01_L, YS01_L, FN01_L, FN02_P/L, FN03_P/L |
| 16.25 | 96-76-4 | 2,4-Di-tert-butylphenol | I | 191, 206, 57 | LE01, LE03, LS01, LS02 | QL01_E, QF01 | YN01_P/L, YS01_P/L, FN01_P, FN02_L, FN03_L |
| 16.36 | 93-28-7 | Eugenol acetate | I | 164, 149, 131 | FN03_L | ||
| 16.43 | 530-48-3 | 1,1-Diphenylethylene | III | 180, 165, 89 | QF01_P | YN01_P, YS01_P, FN03_P | |
| 16.60 | 105-76-0 | Dibutyl maleate | I | 99, 57, 117 | QF02_L | ||
| 16.92 | 5814-85-7 | 1,2-Diphenylpropane | III | 105, 79, 91 | YN01_P, YS01_P | ||
| 17.37 | 4098-71-9 | Isophorone diisocyanate | III | 110, 123, 81 | QS01_L, QL01_E | ||
| 17.37 | 84-66-2 | Diethyl phthalate * | I | 149, 177, 150 | LE01, LE02, LE03, LS01, LS02 | QS01_-/P/I, QF02_L/P | YN01_P/L, YS01_P/L, FN01_P/L, FN02_L, FN03_P/L |
| 17.80 | 10233-13-3 | Isopropyl laurate | I | 43, 102, 183 | FN02_L | ||
| 18.02 | 119-61-9 | Benzophenone * | III | 105, 77, 182 | LE01, LE02, LE03, LS01, LS02 | QF02_L/P | |
| 18.27 | 1081-75-0 | 1,3-Diphenylpropane * | III | 92, 105, 196 | QS01_I, QF02_P | YN01_P, YS01_P, FN03_P | |
| 18.78 | 20071-09-4 | trans-1,2-Diphenylcyclobutane | III | 104, 78, 178 | QS01_I, QF02_P | FN03_P | |
| 18.98 | 3910-35-8 | 1,1,3-Trimethyl-3-phenylindan | III | 221, 143, 128 | LS01 | QF02_L | YN01_P/L, YS01_P/L, FN03_P |
| 19.00 | 1020-31-1 | 3,5-Di-tert-butylcatechol | I | 207, 222, 179 | FN01_P | ||
| 19.04 | Unknown (naphthalene compound structure) | 197, 155, 212 | LE02, LE03, LS01, LS02 | QF02_L/P | |||
| 19.51 | 1620-98-0 | 3,5-Di-tert-butyl-4-hydroxybenzaldehyde | II | 219, 191, 234 | FN02_L | ||
| 19.73 | 3018-20-0 | 1-Phenyltetralin | III | 130, 180, 208 | QS01_I, QF02_P | YN01_P, YS01_P, FN03_P | |
| 19.92 | 1889-67-4 | 2,3-Dimethyl-2,3-diphenylbutane | III | 119, 91, 77 | LS01 | QF02_L | YN01_P/L, YS01_P/L, FN03_P |
| 20.25 | 110-27-0 | Isopropyl myristate | I | 102, 60, 228 | LE01, LE02, LS01, LS02 | ||
| 20.56 | 1222-05-5 | Galoxolide | III | 243, 213, 258 | LE01, LS01, LS02 | ||
| 20.70 | 84-69-5 | Diisobutyl phthalate (DIBP) * | I | 149, 57, 150 | LE01, LE02, LE03, LS01, LS02 | QF02_P | YN01_P/L, YS01_P/L, FN01_P/L, FN02_P/L, FN03_P/L |
| 20.78 | 605-02-7 | 1-Phenylnaphthalene | III | 204, 101, 88 | QS01_I, QF02_P | ||
| 21.21 | 82304-66-3 | 7,9-Di-tert-butyl-1-oxaspiro[4.5]deca-6,9-diene-2,8-dione | III | 205, 57, 175 | LE01, LE02, LE03, LS01, LS02 | QS01_L, QL01_E, QF01, QF02_L/P | YN01_P/L, YS01_P/L, FN01_L, FN02_P/L, FN03_P/L |
| 21.41 | 112-39-0 | Methyl palmitate * | I | 74, 87, 43 | LE01, LS01, LS02 | ||
| 21.47 | 6386-38-5 | Benzenepropanoic acid | II | 277, 147, 219 | QL01_I | FN01_P | |
| 21.75 | 84-74-2 | Dibutyl phthalate (DBP) * | I | 149, 150, 104 | LE01, LE02, LE03, LS01, LS02 | YN01_L, YS01_L | |
| 22.06 | 110-36-1 | Butyl myristate | I | 56, 229, 129 | FN01_L | ||
| 22.47 | 142-91-6 | Isopropyl palmitate | I | 43, 102, 256 | LE02 | ||
| 22.98 | 3524-68-3 | Pentaerythritol triacrylate | I | 55, 81, 126 | FN02_L | ||
| 23.27 | 112-62-9 | Methyl oleate | I | 55, 43, 69 | QF02_L/P | FN02_P | |
| 23.35 | 101-68-8 | 4,4′-Diphenylmethane diisocyanate | III | 250, 208, 221 | QL01_E | ||
| 23.37 | 103-41-3 | Benzyl cinnamate | I | 131, 91, 192 | FN03_L | ||
| 23.82 | 7568-58-3 | Tributyl trans-aconitate | I | 112, 157, 139 | QL01_E | ||
| 23.98 | Unknown (phthalate compound structure) | 149, 150, 86 | FN01_L | ||||
| 24.07 | 629-54-9 | Hexadecanamide * | III | 59, 72, 43 | QS01_P, QL01_E | YN01_L, YS01_L, FN01_P, FN02_L, FN03_L | |
| 24.14 | 111-06-8 | Butyl palmitate | I | 56, 257, 129 | YN01_P/L, YS01_P/L, FN01_L, FN02_L, FN03_P | ||
| 24.24 | 959-26-2 | Bis(2-hydroxyethyl) terephthalate | I | 149, 193, 211 | QS01_P | ||
| 24.47 | 65745-83-7 | 3,6,9,12,15-Oxabicyclo(15,3)heneicosa-1(21),17,19-triene-2,16-dione | III | 149, 193, 104 | FN01_L | ||
| 24.59 | 483-65-8 | Retene | III | 219, 234, 204 | LE01 | ||
| 24.72 | 77-90-7 | Acetyltributyl citrate (ATBC) * | I | 185, 129, 259 | QS01_L, QL01_E/I, QF01, QF02_L | YN01_P/L, YS01_P/L, FN01_L, FN02_P/L, FN03_L | |
| 24.72 | 21956-56-9 | 3,5-Dimethoxystilbene | III | 240, 165, 152 | LE02, LE03, LS02 | ||
| 24.90 | 5776-79-4 | 1,8-Diazacyclotetradecane-2,9-dione | III | 114, 198, 55 | QF01 | ||
| 25.09 | 13601-88-2 | Dehydroabietal | II | 159, 173, 129 | LE01, LE02, LE03, LS01, LS02 | ||
| 25.70 | 1235-74-1 | Methyl dehydroabietate | II | 239, 141, 129 | LE01, LE02, LE03, LS01, LS02 | YN01_L, YS01_L, FN02_P | |
| 25.76 | 112-63-0 | Methyl linoleate | I | 67, 81, 95 | FN03_L | ||
| 26.16 | 103-23-1 | Diethylhexyl adipate (DEHA) * | I | 129, 70, 111 | LE01, LE03, LS01, LS02 | YN01_L, YS01_L, FN03_P/L | |
| 26.86-28.39 | Unknown (phthalate compound structure) | 149, 57, 71 | LE01, LS02 | ||||
| 26.68 | 1740-19-8 | Dehydroabietic acid | II | 239, 285, 197 | YN01_L, YS01_L, FN03_L | ||
| 27.09 | 514-10-3 | Abietic acid | III | 136, 105, 302 | YN01_L, YS01_L, FN03_L | ||
| 27.35 | 117-81-7 | Bis(2-ethylhexyl)phthalate (DEHP) * | I | 149, 167, 57 | LE03, LS01 | QS01_P, QL01_E, QF01, QF02_L | YN01_L, YS01_L, FN01_P/L, FN02_P/L, FN03_L |
| 27.48 | 56728-02-0 | (1-Methyl-2,2-diphenylcyclopropyl)sulfanylbenzene | III | 129, 91, 207 | QS01_I, QF02_P | ||
| 28.38 | 6197-30-4 | Octocrylene * | III | 204, 232, 250 | LE02, LS01, LS02 | FN01_P, FN02_L | |
| 28.48 | 1633-22-3 | Di-p-xylylene | III | 104, 208, 78 | QS01_I, QF02_P | YN01_P, YS01_P, FN03_P | |
| 29.11 | 6422-86-2 | Bis(2-ethylhexyl) terephthalate (DEHT) * | I | 70, 149, 112 | LE02, LS01, LS02 | QF01, QF02_L | FN02_L |
| 29.42 | 112-84-5 | Erucamide * | III | 59, 72, 43 | QS01_L/P, QL01_E/I, QF01 | FN01_P, FN02_P/L, FN03_P/L | |
| 29.67 | 111-02-4 | Squalene * | I | 69, 81, 95 | LE01, LE02, LS01, LS02 | QS01_P/I, QL01_I, QF01, QF02_L/P | YN01_P/L, YS01_P/L, FN01_P/L, FN02_P/L, FN03_P/L |
| 29.71 | Unknown (phthalate compound structure) | 149.0232 | LE03, LS01 | FN01_L | |||
| 30.58 | 538-23-8 | Glyceryl tricaprylate * | I | 127, 57, 201 | QS01_P |
| Compound | Equation | r | LOD (µg/g) | LOQ (µg/g) | Recovery (%) (n = 6) | Intermediate Precision (RSD %) (n = 6) | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| 0.25 µg/g | 0.5 µg/g | 1 µg/g | 0.25 µg/g | 0.5 µg/g | 1 µg/g | |||||
| DEP | Y = 1.6592x + 0.1137 | 0.9940 | 0.01 | 0.025 | 112 | 114 | 102 | 16.1 | 8.36 | 2.50 |
| BP | Y = 0.5995x + 0.0401 | 0.9941 | 0.05 | 0.1 | 107 | 104 | 102 | 14.9 | 7.67 | 2.03 |
| 1,3-DPP | Y = 1.4391x + 0.0164 | 0.9932 | 0.025 | 0.05 | 88.8 | 80 | 78.4 | 5.03 | 1.73 | 7.01 |
| DIBP | Y = 2.3345x + 0.0726 | 0.9981 | < 0.005 | 0.005 | 112 | 123 | 124 | 5.40 | 2.80 | 1.61 |
| DBP | Y = 2.323x − 0.0116 | 0.9984 | < 0.005 | 0.005 | 120 | 124 | 123 | 8.52 | 0.98 | 4.26 |
| DEHP | Y = 0.9686x − 0.0019 | 0.9900 | < 0.005 | 0.005 | 123 | 124 | 116 | 1.62 | 1.04 | 7.51 |
| DEHT | Y = 0.3052x − 0.009 | 0.9909 | 0.025 | 0.05 | 96.2 | 89.6 | 98.9 | 16.0 | 15.9 | 18.2 |
| Compound | Concentration (µg/g) | Dietary Exposure µg/kg bw per Day | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Mean | P95 | ||||||||
| 12–35 Months | 3–9 Years | 10–17 Years | 12–35 Months | 3–9 Years | 10–17 Years | 12–35 Months | 3–9 Years | 12–35 Months | |
| DEP | 0.03 ± 0.005 | 0.03 ± 0.005 | 0.03 ± 0.004 | 1.19 | 0.82 | 0.47 | 6.24 | 3.63 | 2.29 |
| BP | ˂LOQ | ˂LOQ | ˂ LOQ | 2.25 | 1.60 | 0.74 | 11.80 | 7.06 | 3.61 |
| 1,3-DPP | ˂LOQ | ˂LOQ | ˂ LOQ | 1.13 | 0.80 | 0.37 | 5.88 | 3.53 | 1.81 |
| DIBP | 0.08 ± 0.01 | 0.05 ± 0.004 | 0.10 ± 0.01 | 3.49 | 1.61 | 1.55 | 18.20 | 7.10 | 7.51 |
| DBP | 0.10 ± 0.003 | 0.09 ± 0.002 | 0.16 ± 0.04 | 4.40 | 2.95 | 2.42 | 23.00 | 13.00 | 11.80 |
| DEHP | 0.09 ± 0.03 | 0.07 ± 0.03 | 0.09 ± 0.02 | 4.07 | 2.13 | 1.35 | 21.30 | 9.45 | 6.54 |
| DEHT | ˂LOQ | ˂LOQ | ˂LOQ | 1.13 | 0.80 | 0.37 | 5.88 | 3.53 | 1.81 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lestido-Cardama, A.; Sendón, R.; Bustos, J.; Lomo, M.L.; Losada, P.P.; de Quirós, A.R.B. Dietary Exposure Estimation to Chemicals Transferred from Milk and Dairy Products Packaging Materials in Spanish Child and Adolescent Population. Foods 2020, 9, 1554. https://doi.org/10.3390/foods9111554
Lestido-Cardama A, Sendón R, Bustos J, Lomo ML, Losada PP, de Quirós ARB. Dietary Exposure Estimation to Chemicals Transferred from Milk and Dairy Products Packaging Materials in Spanish Child and Adolescent Population. Foods. 2020; 9(11):1554. https://doi.org/10.3390/foods9111554
Chicago/Turabian StyleLestido-Cardama, Antía, Raquel Sendón, Juana Bustos, Mª Luisa Lomo, Perfecto Paseiro Losada, and Ana Rodríguez Bernaldo de Quirós. 2020. "Dietary Exposure Estimation to Chemicals Transferred from Milk and Dairy Products Packaging Materials in Spanish Child and Adolescent Population" Foods 9, no. 11: 1554. https://doi.org/10.3390/foods9111554
APA StyleLestido-Cardama, A., Sendón, R., Bustos, J., Lomo, M. L., Losada, P. P., & de Quirós, A. R. B. (2020). Dietary Exposure Estimation to Chemicals Transferred from Milk and Dairy Products Packaging Materials in Spanish Child and Adolescent Population. Foods, 9(11), 1554. https://doi.org/10.3390/foods9111554







