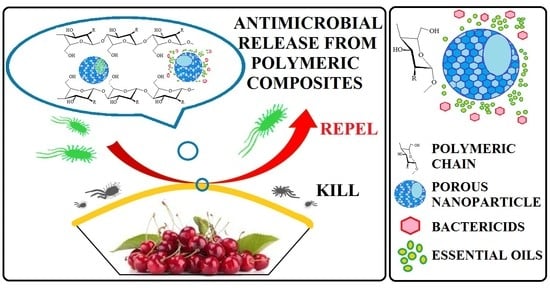
Biodegradable Antimicrobial Food Packaging: Trends and Perspectives
Abstract
1. Introduction
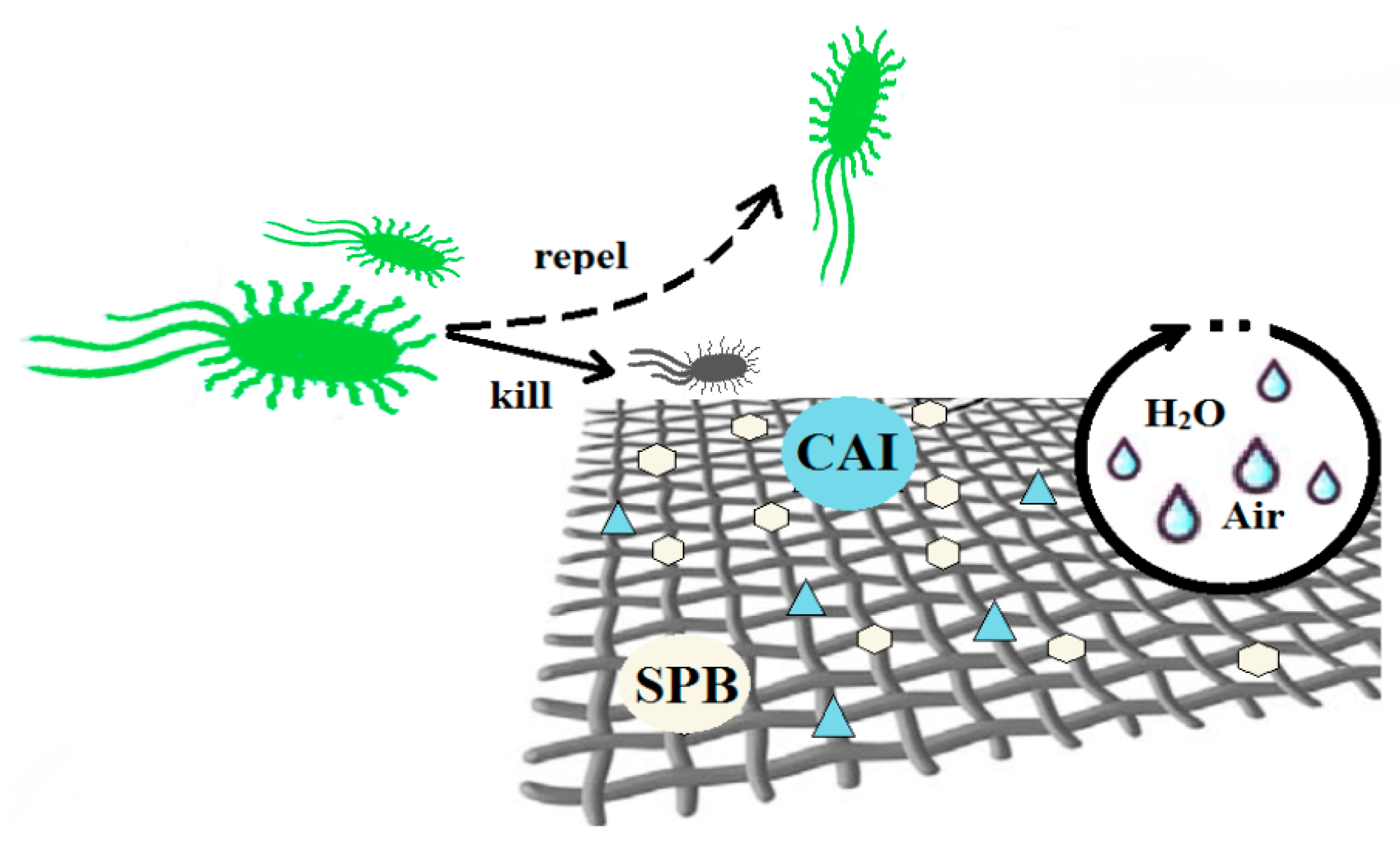
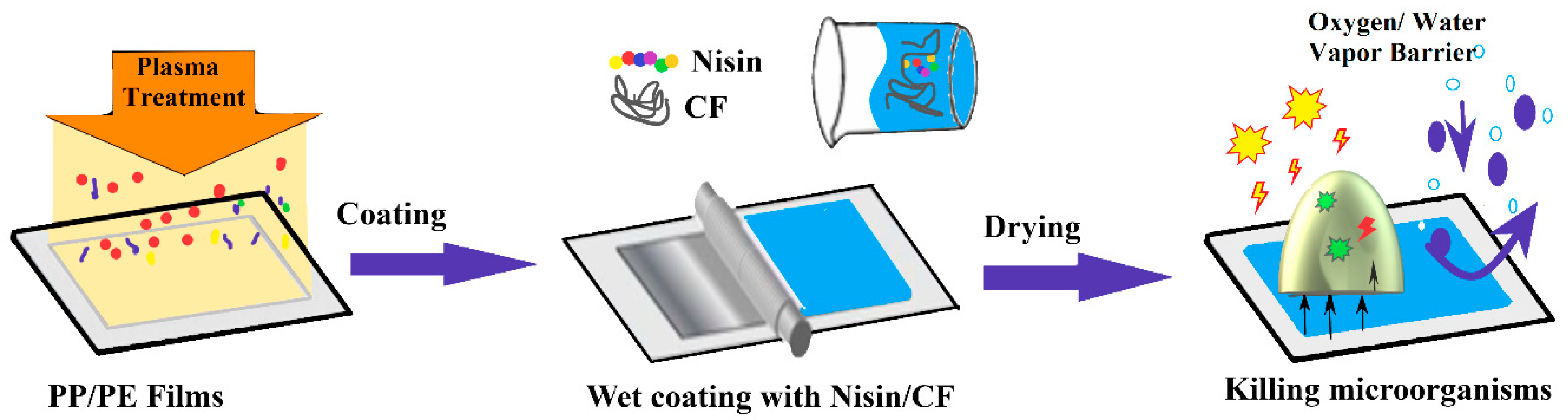
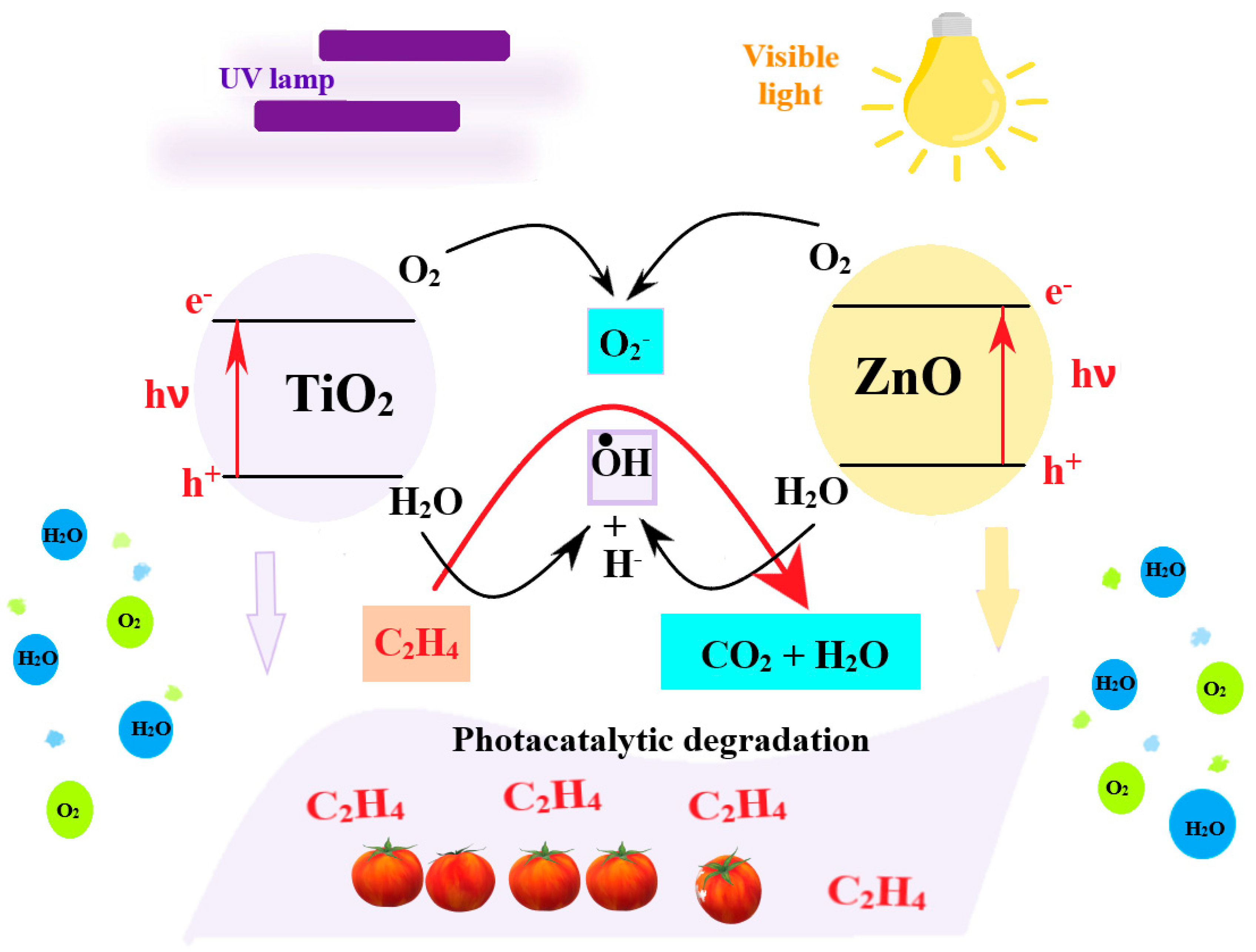
2. Antimicrobial Packaging Obtained by Modification of Current Materials
3. Biodegradable Polymeric Antimicrobial Packaging
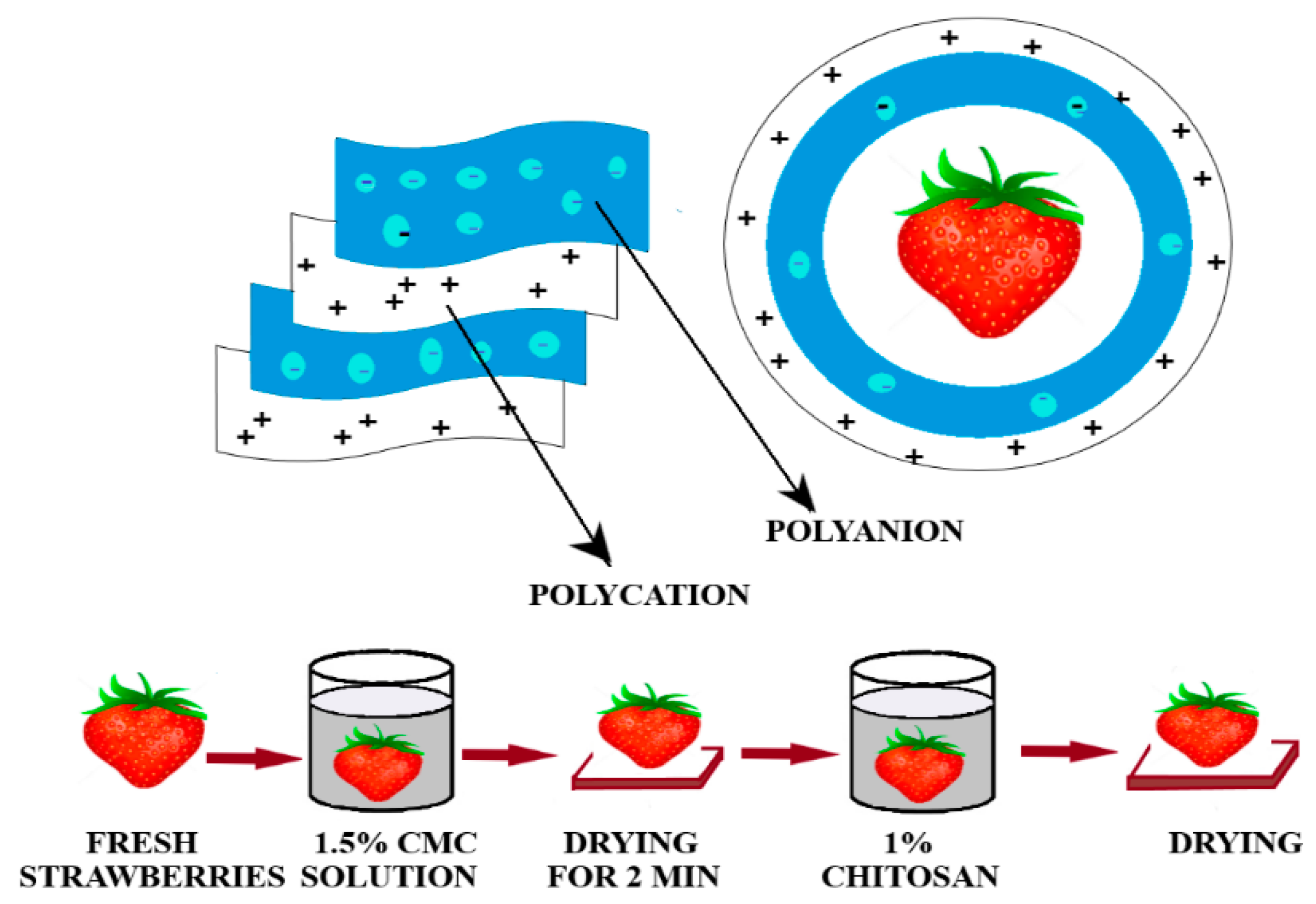
3.1. Edible Films
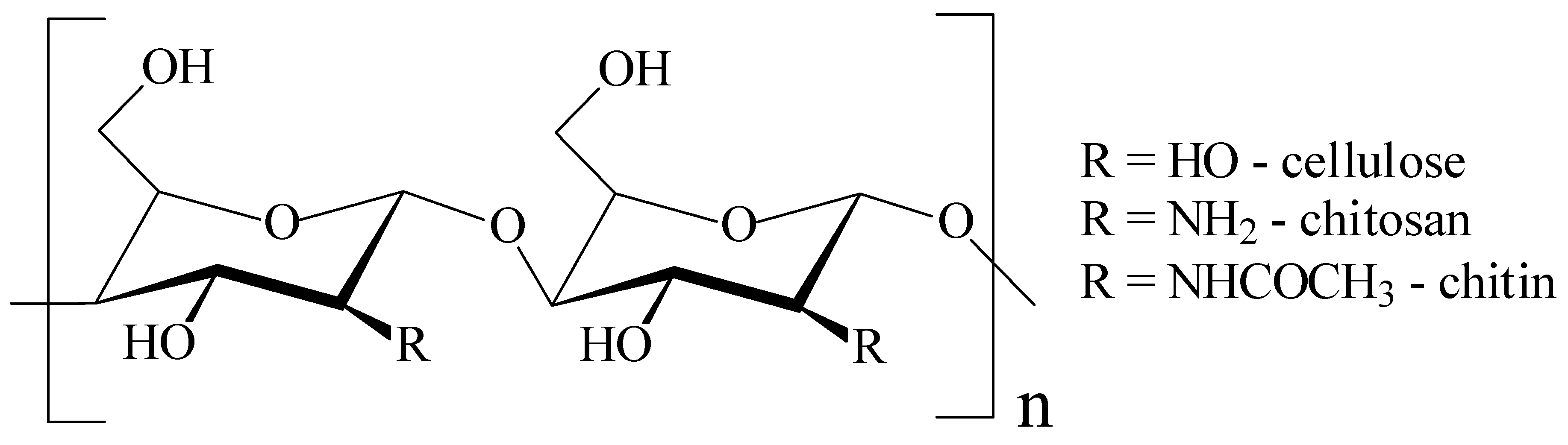
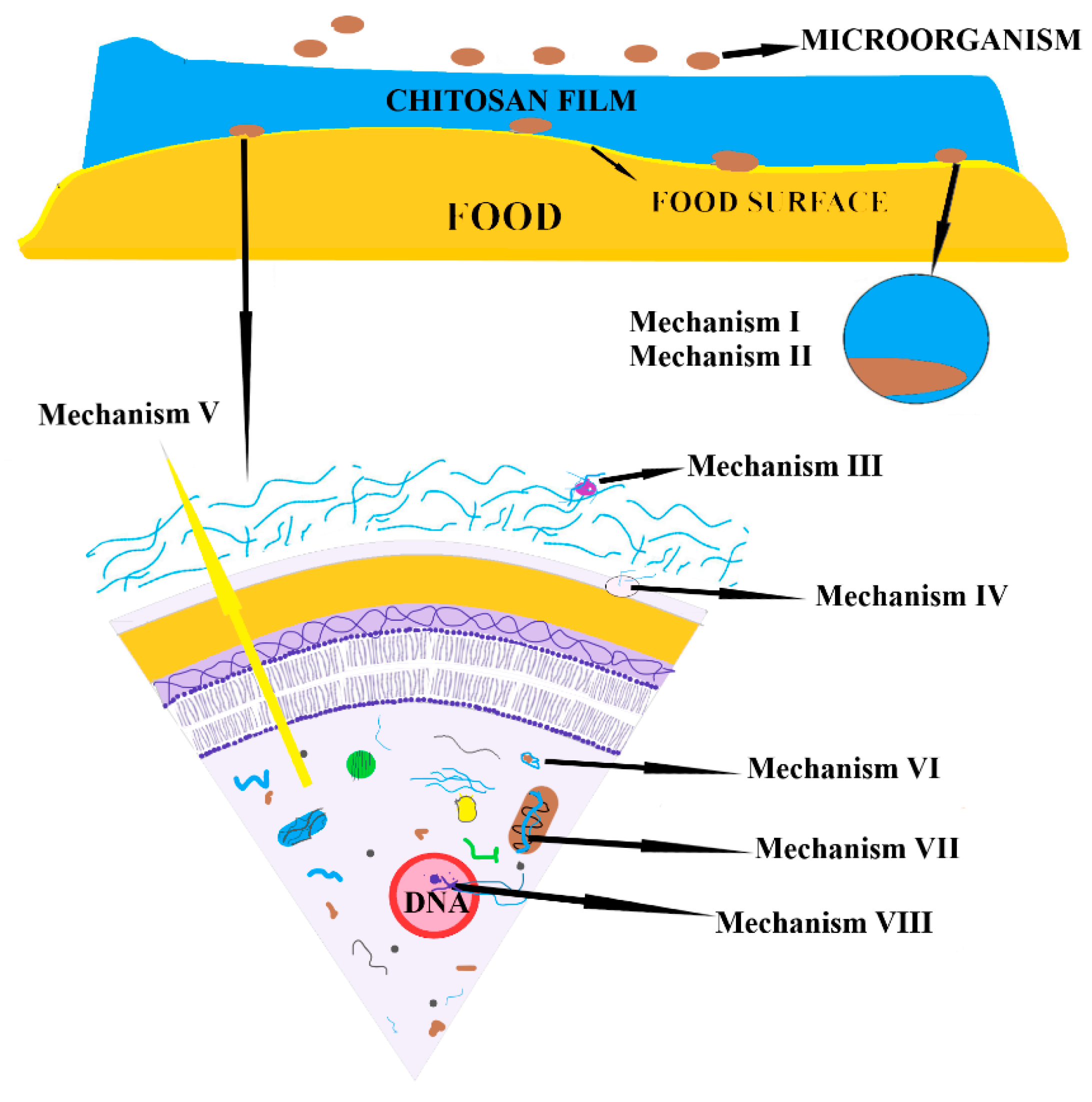
3.2. Polysaccharides-Based Films
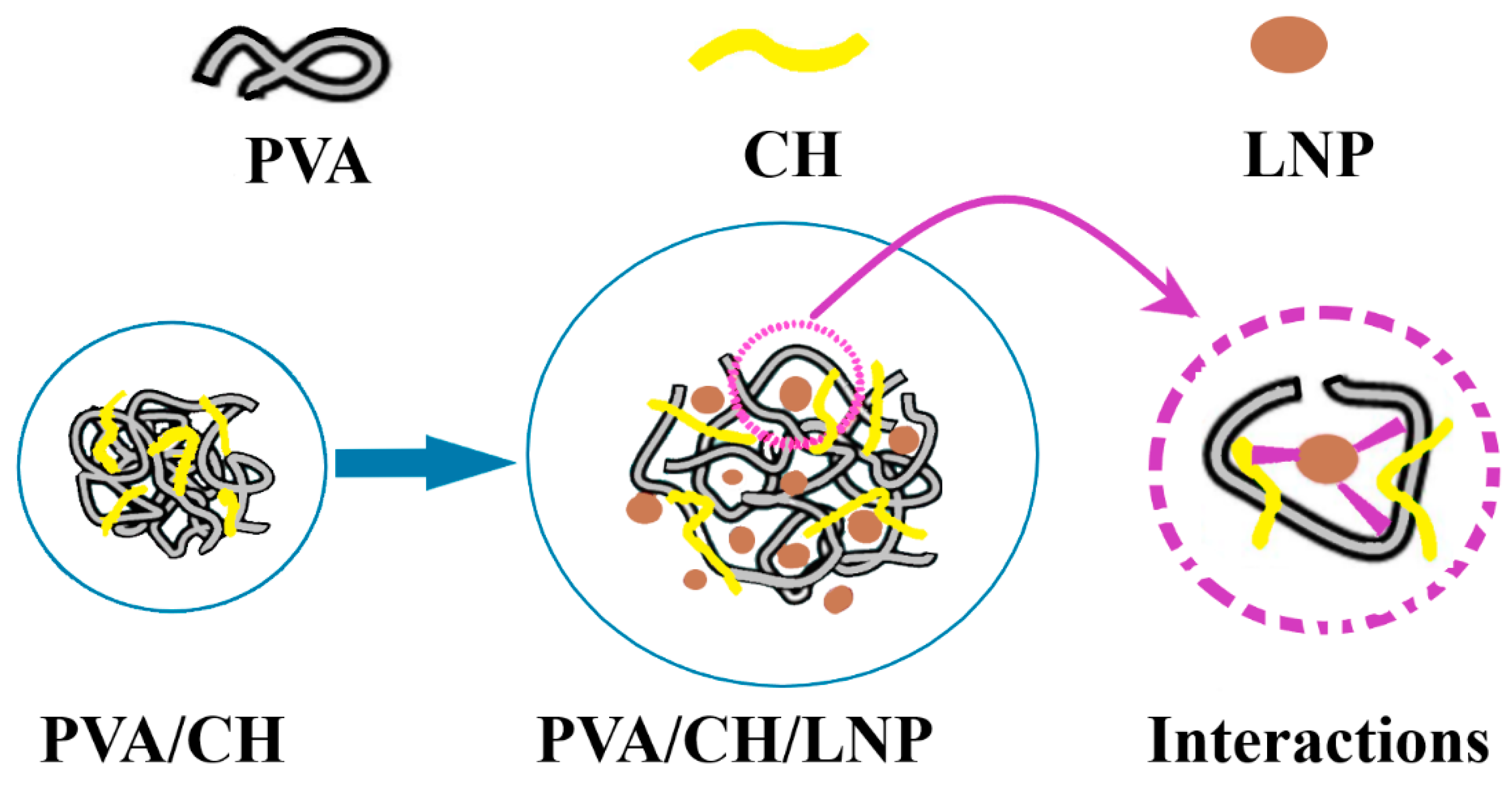
3.3. Other Biopolymers-Based Films
4. Toxicity Studies
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Technical Platform on the Measurement and Reduction of Food Loss and Waste. Available online: http://www.fao.org/platform-food-loss-waste/en/ (accessed on 25 August 2020).
- Ishangulyyev, S.; Kim, S.; Lee, S.H. Understanding Food Loss and Waste-Why Are We Losing and Wasting Food? Foods 2019, 8, 297. [Google Scholar] [CrossRef] [PubMed]
- Jiang, B.; Na, J.X.; Wang, L.L.; Li, D.M.; Liu, C.H.; Feng, Z.B. Reutilization of Food Waste: One-Step Extration, Purification and Characterization of Ovalbumin from Salted Egg White by Aqueous Two-Phase Flotation. Foods 2019, 8, 286. [Google Scholar] [CrossRef] [PubMed]
- Food Packaging Market Size, Share&Trends Analysis Report By Type (Rigid, Flexible), By Material (Paper, Plastic), By Application (Bakery and Confectionery, Dairy Products), By Region, And Segment Forecasts, 2020–2027. Available online: https://www.grandviewresearch.com/industry-analysis/food-packaging-market (accessed on 26 September 2020).
- Wohner, B.; Gabriel, V.H.; Krenn, B.; Krauter, V.; Tacker, M. Environmental and economic assessment of food-packaging systems with a focus on food waste. Case study on tomato ketchup. Sci. Total Environ. 2020, 738. [Google Scholar] [CrossRef]
- EU Circular Economy Action Plan. A New Circular Economy Action Plan for a Cleaner and More Competitive Europe. Available online: https://ec.europa.eu/environment/circular-economy/index_en.htm (accessed on 26 September 2020).
- Kuster-Boluda, I.; Vila, N. Can Health Perceptions, Credibility, and Physical Appearance of Low-Fat Foods Stimulate Buying Intentions? Foods 2020, 9, 866. [Google Scholar] [CrossRef] [PubMed]
- Kuster, I.; Vila, N.; Sarabia, F. Food packaging cues as vehicles of healthy information: Visions of millennials (early adults and adolescents). Food Res. Int. 2019, 119, 170–176. [Google Scholar] [CrossRef]
- Hernandez, A.; Vila, N.; Kuster, I.; Rodriguez, C. Clustering Spanish alcoholic beverage shoppers to focus marketing strategies. Int. J. Wine Bus. Res. 2019, 31, 362–384. [Google Scholar] [CrossRef]
- Vila-Lopez, N.; Kuster-Boluda, I. Consumers’ physiological and verbal responses towards product packages: Could these responses anticipate product choices? Physiol. Behav. 2019, 200, 166–173. [Google Scholar] [CrossRef]
- Vila-Lopez, N.; Küster-Boluda, I. A bibliometric analysis on packaging research: Towards sustainable and healthy packages. Br. Food J. 2020. ahead of print. [Google Scholar] [CrossRef]
- Bou-Mitri, C.; Abdessater, M.; Zgheib, H.; Akiki, Z. Food packaging design and consumer perception of the product quality, safety, healthiness and preference. Nutr. Food Sci. 2020. ahead-of-print. [Google Scholar] [CrossRef]
- Makaremi, M.; Yousefi, H.; Cavallaro, G.; Lazzara, G.; Goh, C.B.S.; Lee, S.M.; Solouk, A.; Pasbakhsh, P. Safely Dissolvable and Healable Active Packaging Films Based on Alginate and Pectin. Polymers 2019, 11, 1594. [Google Scholar] [CrossRef]
- Kaladharan, P.; Singh, V.V.; Asha, P.S.; Edward, L.; Sukhadane, K.S. Marine plastic litter in certain trawl grounds along the peninsular coasts of India. Mar. Pollut. Bull. 2020, 157, 111299. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.Y.; Chen, H.; Yang, J.H.; Long, R.Y.; Li, W.B. Impact of regulatory focus on express packaging waste recycling behavior: Moderating role of psychological empowerment perception. Environ. Sci. Pollut. Res. 2019, 26, 8862–8874. [Google Scholar] [CrossRef] [PubMed]
- White, A.; Lockyer, S. Removing plastic packaging from fresh produce—What’s the impact? Nutr. Bull. 2020, 45, 35–50. [Google Scholar] [CrossRef]
- Guo, J.J.; Huang, X.P.; Xiang, L.; Wang, Y.Z.; Li, Y.W.; Li, H.; Cai, Q.Y.; Mo, C.H.; Wong, M.H. Source, migration and toxicology of microplastics in soil. Environ. Int. 2020, 137, 105263. [Google Scholar] [CrossRef] [PubMed]
- Boz, Z.; Korhonen, V.; Sand, C.K. Consumer Considerations for the Implementation of Sustainable Packaging: A Review. Sustainability 2020, 12, 2192. [Google Scholar] [CrossRef]
- Clark, N.; Trimingham, R.; Wilson, G.T. Incorporating Consumer Insights into the UK Food Packaging Supply Chain in the Transition to a Circular Economy. Sustainability 2020, 12, 6106. [Google Scholar] [CrossRef]
- Zemljic, L.F.; Plohl, O.; Vesel, A.; Luxbacher, T.; Potrc, S. Physicochemical Characterization of Packaging Foils Coated by Chitosan and Polyphenols Colloidal Formulations. Int. J. Mol. Sci. 2020, 21, 495. [Google Scholar] [CrossRef] [PubMed]
- Esmailzadeh, H.; Sangpour, P.; Shahraz, F.; Eskandari, A.; Hejazi, J.; Khaksar, R. CuO/LDPE nanocomposite for active food packaging application: A comparative study of its antibacterial activities with ZnO/LDPE nanocomposite. Polym. Bull. 2020. published online. [Google Scholar] [CrossRef]
- Zia, J.; Paul, U.C.; Heredia-Guerrero, J.A.; Athanassiou, A.; Fragouli, D. Low-density polyethylene/curcumin melt extruded composites with enhanced water vapor barrier and antioxidant properties for active food packaging. Polymer 2019, 175, 137–145. [Google Scholar] [CrossRef]
- Habib, S.; Lehocky, M.; Vesela, D.; Humpolicek, P.; Krupa, I.; Popelka, A. Preparation of Progressive Antibacterial LDPE Surface via Active Biomolecule Deposition Approach. Polymers 2019, 11, 1704. [Google Scholar] [CrossRef]
- Giannakas, A.; Salmas, C.; Leontiou, A.; Tsimogiannis, D.; Oreopoulou, A.; Braouhli, J. Novel LDPE/Chitosan Rosemary and Melissa Extract Nanostructured Active Packaging Films. Nanomaterials 2019, 9, 1105. [Google Scholar] [CrossRef] [PubMed]
- Kuster-Boluda, A.; Vila, N.V.; Kuster, I. Managing international distributors’ complaints: An exploratory study. J. Bus. Ind. Mark. 2020. [Google Scholar] [CrossRef]
- Kerry, J.P.; O’Grady, M.N.; Hogan, S.A. Past, current and potential utilisation of active and intelligent packaging systems for meat and muscle-based products: A review. Meat Sci. 2006, 74, 113–130. [Google Scholar] [CrossRef] [PubMed]
- O’Callaghan, K.A.M.; Kerry, J.P. Assessment of the antimicrobial activity of potentially active substances (nanoparticled and non-nanoparticled) against cheese-derived micro-organisms. Int. J. Dairy Technol. 2014, 67, 483–489. [Google Scholar] [CrossRef]
- Rai, M.; Ingle, A.P.; Gupta, I.; Pandit, R.; Paralikar, P.; Gade, A.; Chaud, M.V.; dos Santos, C.A. Smart nanopackaging for theenhancement of foodshelf life. Environ. Chem. Lett. 2019, 17, 277–290. [Google Scholar] [CrossRef]
- Vilas, C.; Mauricio-Iglesias, M.; Garcia, M.R. Model-based design of smart active packaging systems with antimicrobial activity. Food Packag. Shelf 2020, 24, 100446. [Google Scholar] [CrossRef]
- Jayasena, D.D.; Jo, C. Essential oils as potential antimicrobial agents in meat and meat products: A review. Trends Food Sci. Technol. 2013, 34, 96–108. [Google Scholar] [CrossRef]
- Szabo, K.; Teleky, B.E.; Mitrea, L.; Calinoiu, L.F.; Martau, G.A.; Simon, E.; Varvara, R.A.; Vodnar, D.C. Active Packaging-Poly(Vinyl Alcohol) Films Enriched with Tomato By-Products Extract. Coatings 2020, 10, 141. [Google Scholar] [CrossRef]
- Motelica, L.; Ficai, D.; Oprea, O.C.; Ficai, A.; Andronescu, E. Smart Food Packaging Designed by Nanotechnological and Drug Delivery Approaches. Coatings 2020, 10, 806. [Google Scholar] [CrossRef]
- Shruthy, R.; Jancy, S.; Preetha, R. Cellulose nanoparticles synthesised from potato peel for the development of active packaging film for enhancement of shelf life of raw prawns (Penaeus monodon) during frozen storage. Int. J. Food Sci. Technol. 2020. [Google Scholar] [CrossRef]
- Han, J.H. Antimicrobial food packaging. Food Technol. Chic. 2000, 54, 56–65. [Google Scholar]
- Surendhiran, D.; Li, C.Z.; Cui, H.Y.; Lin, L. Fabrication of high stability active nanofibers encapsulated with pomegranate peel extract using chitosan/PEO for meat preservation. Food Packag. Shelf Life 2020, 23, 100439. [Google Scholar] [CrossRef]
- Settier-Ramirez, L.; Lopez-Carballo, G.; Gavara, R.; Hernandez-Munoz, P. PVOH/protein blend films embedded with lactic acid bacteria and their antilisterial activity in pasteurized milk. Int. J. Food Microbiol. 2020, 322, 108545. [Google Scholar] [CrossRef]
- Ramos, M.; Beltran, A.; Fortunati, E.; Peltzer, M.; Cristofaro, F.; Visai, L.; Valente, A.J.M.; Jimenez, A.; Kenny, J.M.; Garrigos, M.C. Controlled Release of Thymol from Poly(Lactic Acid)-Based Silver Nanocomposite Films with Antibacterial and Antioxidant Activity. Antioxidants 2020, 9, 395. [Google Scholar] [CrossRef] [PubMed]
- Chiabrando, V.; Garavaglia, L.; Giacalone, G. The Postharvest Quality of Fresh Sweet Cherries and Strawberries with an Active Packaging System. Foods 2019, 8, 335. [Google Scholar] [CrossRef] [PubMed]
- Ojogbo, E.; Ward, V.; Mekonnen, T.H. Functionalized starch microparticles for contact-active antimicrobial polymer surfaces. Carbohyd. Polym. 2020, 229, 115442. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.F.; Xia, Q.Y.; Xiao, H.N. Cationic Polymers with Tailored Structures for Rendering Polysaccharide-Based Materials Antimicrobial: An Overview. Polymers 2019, 11, 1283. [Google Scholar] [CrossRef]
- Brezoiu, A.M.; Prundeanu, M.; Berger, D.; Deaconu, M.; Matei, C.; Oprea, O.; Vasile, E.; Negreanu-Pirjol, T.; Muntean, D.; Danciu, C. Properties of Salvia officinalis L. and Thymus serpyllum L. Extracts Free and Embedded into Mesopores of Silica and Titania Nanomaterials. Nanomaterials 2020, 10, 820. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.J.; Wang, X.J.; Gao, M.; Zhao, Y.L.; Chen, Y.Z. Sustained release of an essential oil by a hybrid cellulose nanofiber foam system. Cellulose 2020, 27, 2709–2721. [Google Scholar] [CrossRef]
- Fang, Z.X.; Zhao, Y.Y.; Warner, R.D.; Johnson, S.K. Active and intelligent packaging in meat industry. Trends Food Sci. Technol. 2017, 61, 60–71. [Google Scholar] [CrossRef]
- Realini, C.E.; Marcos, B. Active and intelligent packaging systems for a modern society. Meat Sci. 2014, 98, 404–419. [Google Scholar] [CrossRef] [PubMed]
- Yildirim, S.; Rocker, B.; Pettersen, M.K.; Nilsen-Nygaard, J.; Ayhan, Z.; Rutkaite, R.; Radusin, T.; Suminska, P.; Marcos, B.; Coma, V. Active Packaging Applications for Food. Compr. Rev. Food Sci. Food Saf. 2018, 17, 165–199. [Google Scholar] [CrossRef]
- Malhotra, B.; Keshwani, A.; Kharkwal, H. Antimicrobial food packaging: Potential and pitfalls. Front. Microbiol. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Suppakul, P.; Miltz, J.; Sonneveld, K.; Bigger, S.W. Antimicrobial properties of basil and its possible application in food packaging. J. Agric. Food Chem. 2003, 51, 3197–3207. [Google Scholar] [CrossRef] [PubMed]
- Saadat, S.; Pandey, G.; Tharmavaram, M.; Braganza, V.; Rawtani, D. Nano-interfacial decoration of Halloysite Nanotubes for the development of antimicrobial nanocomposites. Adv. Colloid Interface Sci. 2020, 275, 102063. [Google Scholar] [CrossRef]
- Arsenie, L.V.; Lacatusu, I.; Oprea, O.; Bordei, N.; Bacalum, M.; Badea, N. Azelaic acid-willow bark extract-panthenol—Loaded lipid nanocarriers improve the hydration effect and antioxidant action of cosmetic formulations. Ind. Crops Prod. 2020, 154, 112658. [Google Scholar] [CrossRef]
- Arfat, Y.A.; Ejaz, M.; Jacob, H.; Ahmed, J. Deciphering the potential of guar gum/Ag-Cu nanocomposite films as an active food packaging material. Carbohyd. Polym. 2017, 157, 65–71. [Google Scholar] [CrossRef]
- Nedelcu, I.A.; Ficai, A.; Sonmez, M.; Ficai, D.; Oprea, O.; Andronescu, E. Silver Based Materials for Biomedical Applications. Curr. Org. Chem. 2014, 18, 173–184. [Google Scholar] [CrossRef]
- Pica, A.; Guran, C.; Andronescu, E.; Oprea, O.; Ficai, D.; Ficai, A. Antimicrobial performances of some film forming materials based on silver nanoparticles. J. Optoelectron. Adv. Mater. 2012, 14, 863–868. [Google Scholar]
- Oprea, O.; Andronescu, E.; Ficai, D.; Ficai, A.; Oktar, F.N.; Yetmez, M. ZnO Applications and Challenges. Curr. Org. Chem. 2014, 18, 192–203. [Google Scholar] [CrossRef]
- Vasile, B.S.; Oprea, O.; Voicu, G.; Ficai, A.; Andronescu, E.; Teodorescu, A.; Holban, A. Synthesis and characterization of a novel controlled release zinc oxide/gentamicin-chitosan composite with potential applications in wounds care. Int. J. Pharm. 2014, 463, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Siripatrawan, U.; Kaewklin, P. Fabrication and characterization of chitosan-titanium dioxide nanocomposite film as ethylene scavenging and antimicrobial active food packaging. Food Hydrocoll. 2018, 84, 125–134. [Google Scholar] [CrossRef]
- de Araujo, M.J.G.; Barbosa, R.C.; Fook, M.V.L.; Canedo, E.L.; Silva, S.M.L.; Medeiros, E.S.; Leite, I.F. HDPE/Chitosan Blends Modified with Organobentonite Synthesized with Quaternary Ammonium Salt Impregnated Chitosan. Materials 2018, 11, 291. [Google Scholar] [CrossRef] [PubMed]
- Nouri, A.; Yaraki, M.T.; Ghorbanpour, M.; Agarwal, S.; Gupta, V.K. Enhanced Antibacterial effect of chitosan film using Montmorillonite/CuO nanocomposite. Int. J. Biol. Macromol. 2018, 109, 1219–1231. [Google Scholar] [CrossRef]
- Salmas, C.; Giannakas, A.; Katapodis, P.; Leontiou, A.; Moschovas, D.; Karydis-Messinis, A. Development of ZnO/Na-Montmorillonite Hybrid Nanostructures Used for PVOH/ZnO/Na-Montmorillonite Active Packaging Films Preparation via a Melt-Extrusion Process. Nanomaterials 2020, 10, 1079. [Google Scholar] [CrossRef]
- Lacatusu, I.; Badea, N.; Badea, G.; Brasoveanu, L.; Stan, R.; Ott, C.; Oprea, O.; Meghea, A. Ivy leaves extract based—Lipid nanocarriers and their bioefficacy on antioxidant and antitumor activities. RSC Adv. 2016, 6, 77243–77255. [Google Scholar] [CrossRef]
- Lacatusu, I.; Badea, N.; Badea, G.; Oprea, O.; Mihaila, M.A.; Kaya, D.A.; Stan, R.; Meghea, A. Lipid nanocarriers based on natural oils with high activity against oxygen free radicals and tumor cell proliferation. Mater. Sci. Eng. C 2015, 56, 88–94. [Google Scholar] [CrossRef]
- Yahaya, W.A.W.; Abu Yazid, N.; Azman, N.A.M.; Almajano, M.P. Antioxidant Activities and Total Phenolic Content of Malaysian Herbs as Components of Active Packaging Film in Beef Patties. Antioxidants 2019, 8, 204. [Google Scholar] [CrossRef]
- Yu, H.H.; Kim, Y.J.; Park, Y.J.; Shin, D.M.; Choi, Y.S.; Lee, N.K.; Paik, H.D. Application of mixed natural preservatives to improve the quality of vacuum skin packaged beef during refrigerated storage. Meat Sci. 2020, 169, 108219. [Google Scholar] [CrossRef]
- Huang, T.Q.; Qian, Y.S.; Wei, J.; Zhou, C.C. Polymeric Antimicrobial Food Packaging and Its Applications. Polymers 2019, 11, 560. [Google Scholar] [CrossRef]
- Liang, S.M.; Wang, L.J. A Natural Antibacterial-Antioxidant Film from Soy Protein Isolate Incorporated with Cortex Phellodendron Extract. Polymers 2018, 10, 71. [Google Scholar] [CrossRef] [PubMed]
- Gingasu, D.; Mindru, I.; Patron, L.; Ianculescu, A.; Vasile, E.; Marinescu, G.; Preda, S.; Diamandescu, L.; Oprea, O.; Popa, M.; et al. Synthesis and Characterization of Chitosan-Coated Cobalt Ferrite Nanoparticles and Their Antimicrobial Activity. J. Inorg. Organomet. Polym. 2018, 28, 1932–1941. [Google Scholar] [CrossRef]
- Radulescu, M.; Ficai, D.; Oprea, O.; Ficai, A.; Andronescu, E.; Holban, A.M. Antimicrobial Chitosan based Formulations with Impact on Different Biomedical Applications. Curr. Pharm. Biotechnol. 2015, 16, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.T.; Dao, U.T.T.; Bui, Q.P.T.; Bach, G.L.; Thuc, C.N.H.; Thuc, H.H. Enhanced antimicrobial activities and physiochemical properties of edible film based on chitosan incorporated with Sonneratia caseolaris (L.) Engl. leaf extract. Prog. Org. Coat. 2020, 140, 105487. [Google Scholar] [CrossRef]
- Sofi, S.A.; Singh, J.; Rafiq, S.; Ashraf, U.; Dar, B.N.; Nayik, G.A. A Comprehensive Review on Antimicrobial Packaging and its Use in Food Packaging. Curr. Nutr. Food Sci. 2018, 14, 305–312. [Google Scholar] [CrossRef]
- Mirabelli, V.; Salehi, S.M.; Angiolillo, L.; Belviso, B.D.; Conte, A.; Del Nobile, M.A.; Di Profio, G.; Caliandro, R. Enzyme Crystals and Hydrogel Composite Membranes as New Active Food Packaging Material. Glob. Chall. 2018, 2. [Google Scholar] [CrossRef]
- Galante, Y.M.; Merlini, L.; Silvetti, T.; Campia, P.; Rossi, B.; Viani, F.; Brasca, M. Enzyme oxidation of plant galactomannans yielding biomaterials with novel properties and applications, including as delivery systems. Appl. Microbiol. Biotechnol. 2018, 102, 4687–4702. [Google Scholar] [CrossRef]
- Avramescu, S.M.; Butean, C.; Popa, C.V.; Ortan, A.; Moraru, I.; Temocico, G. Edible and Functionalized Films/Coatings—Performances and Perspectives. Coatings 2020, 10, 687. [Google Scholar] [CrossRef]
- Feng, K.; Wen, P.; Yang, H.; Li, N.; Lou, W.Y.; Zong, M.H.; Wu, H. Enhancement of the antimicrobial activity of cinnamon essential oil-loaded electrospun nanofilm by the incorporation of lysozyme. RSC Adv. 2017, 7, 1572–1580. [Google Scholar] [CrossRef]
- He, S.K.; Yang, Q.M.; Ren, X.Y.; Zi, J.H.; Lu, S.S.; Wang, S.Q.; Zhang, Y.B.; Wang, Y.F. Antimicrobial Efficiency of Chitosan Solutions and Coatings Incorporated with Clove Oil and/or Ethylenediaminetetraacetate. J. Food Saf. 2014, 34, 345–352. [Google Scholar] [CrossRef]
- Arvanitoyannis, I.S.; Stratakos, A.C. Application of Modified Atmosphere Packaging and Active/Smart Technologies to Red Meat and Poultry: A Review. Food Bioprocess Technol. 2012, 5, 1423–1446. [Google Scholar] [CrossRef]
- Fratianni, F.; De Martino, L.; Melone, A.; De Feo, V.; Coppola, R.; Nazzaro, F. Preservation of Chicken Breast Meat Treated with Thyme and Balm Essential Oils. J. Food Sci. 2010, 75, M528–M535. [Google Scholar] [CrossRef] [PubMed]
- Mulla, M.; Ahmed, J.; Al-Attar, H.; Castro-Aguirre, E.; Arfat, Y.A.; Auras, R. Antimicrobial efficacy of clove essential oil infused into chemically modified LLDPE film for chicken meat packaging. Food Control 2017, 73, 663–671. [Google Scholar] [CrossRef]
- Radulescu, M.; Popescu, S.; Ficai, D.; Sonmez, M.; Oprea, O.; Spoiala, A.; Ficai, A.; Andronescu, E. Advances in Drug Delivery Systems, from 0 to 3D Superstructures. Curr. Drug Targets 2018, 19, 393–405. [Google Scholar] [CrossRef] [PubMed]
- Lopes, F.A.; Soares, N.D.F.; Lopes, C.D.P.; da Silva, W.A.; Baffa, J.C.; Medeiros, E.A.A. Conservation of Bakery Products Through Cinnamaldehyde Antimicrobial Films. Packag. Technol. Sci. 2014, 27, 293–302. [Google Scholar] [CrossRef]
- Rivero, S.; Giannuzzi, L.; Garcia, M.A.; Pinotti, A. Controlled delivery of propionic acid from chitosan films for pastry dough conservation. J. Food Eng. 2013, 116, 524–531. [Google Scholar] [CrossRef]
- Mihaly-Cozmuta, A.; Peter, A.; Craciun, G.; Falup, A.; Mihaly-Cozmuta, L.; Nicula, C.; Vulpoi, A.; Baia, M. Preparation and characterization of active cellulose-based papers modified with TiO2, Ag and zeolite nanocomposites for bread packaging application. Cellulose 2017, 24, 3911–3928. [Google Scholar] [CrossRef]
- Al-Naamani, L.; Dutta, J.; Dobretsov, S. Nanocomposite Zinc Oxide-Chitosan Coatings on Polyethylene Films for Extending Storage Life of Okra (Abelmoschus esculentus). Nanomaterials 2018, 8, 479. [Google Scholar] [CrossRef]
- Xing, Y.G.; Li, X.L.; Guo, X.L.; Li, W.X.; Chen, J.W.; Liu, Q.; Xu, Q.L.; Wang, Q.; Yang, H.; Shui, Y.R.; et al. Effects of Different TiO2 Nanoparticles Concentrations on the Physical and Antibacterial Activities of Chitosan-Based Coating Film. Nanomaterials 2020, 10, 1365. [Google Scholar] [CrossRef]
- Xing, Y.G.; Li, W.X.; Wang, Q.; Li, X.L.; Xu, Q.L.; Guo, X.L.; Bi, X.F.; Liu, X.C.; Shui, Y.R.; Lin, H.B.; et al. Antimicrobial Nanoparticles Incorporated in Edible Coatings and Films for the Preservation of Fruits and Vegetables. Molecules 2019, 24, 1695. [Google Scholar] [CrossRef]
- Sun, L.J.; Yang, S.S.; Qian, X.R.; An, X.H. High-efficacy and long term antibacterial cellulose material: Anchored guanidine polymer via double “click chemistry“. Cellulose 2020, 27, 8799–8812. [Google Scholar] [CrossRef]
- Jouneghani, R.S.; Castro, A.H.F.; Panda, S.K.; Swennen, R.; Luyten, W. Antimicrobial Activity of Selected Banana Cultivars Against Important Human Pathogens, Including Candida Biofilm. Foods 2020, 9, 435. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.B.; Yang, X.H.; Tang, B.; Yuan, L.J.; Wang, K.; Liu, X.Y.; Zhu, X.L.; Li, J.N.; Ge, Z.C.; Chen, S.G. New insights into synergistic antimicrobial and antifouling cotton fabrics via dually finished with quaternary ammonium salt and zwitterionic sulfobetaine. Chem. Eng. J. 2018, 336, 123–132. [Google Scholar] [CrossRef]
- Braga, L.R.; Rangel, E.T.; Suarez, P.A.Z.; Machado, F. Simple synthesis of active films based on PVC incorporated with silver nanoparticles: Evaluation of the thermal, structural and antimicrobial properties. Food Packag. Shelf Life 2018, 15, 122–129. [Google Scholar] [CrossRef]
- Bastante, C.C.; Cardoso, L.C.; Ponce, M.T.F.; Serrano, C.M.; de la Ossa-Fernandez, E.J.M. Characterization of olive leaf extract polyphenols loaded by supercritical solvent impregnation into PET/PP food packaging films. J. Supercrit. Fluid 2018, 140, 196–206. [Google Scholar] [CrossRef]
- Ahmed, J.; Mulla, M.; Arfat, Y.A.; Bher, A.; Jacob, H.; Auras, R. Compression molded LLDPE films loaded with bimetallic (Ag-Cu) nanoparticles and cinnamon essential oil for chicken meat packaging applications. LWT Food Sci. Technol. 2018, 93, 329–338. [Google Scholar] [CrossRef]
- Egodage, D.P.; Jayalath, H.T.S.; Samarasekara, A.M.P.B.; Amarasinghe, D.A.S.; Madushani, S.P.A.; Senerath, S.M.N.S. Novel Antimicrobial Nano Coated Polypropylene Based Materials for Food Packaging Systems. In Proceedings of the 2017 3rd International Moratuwa Engineering Research Conference (Mercon), Moratuwa, Sri Lanka, 29–31 May 2017; pp. 88–92. [Google Scholar]
- Wieczynska, J.; Cavoski, I. Antimicrobial, antioxidant and sensory features of eugenol, carvacrol and trans-anethole in active packaging for organic ready-to-eat iceberg lettuce. Food Chem. 2018, 259, 251–260. [Google Scholar] [CrossRef] [PubMed]
- Llana-Ruiz-Cabello, M.; Pichardo, S.; Bermudez, J.M.; Banos, A.; Ariza, J.J.; Guillamon, E.; Aucejo, S.; Camean, A.M. Characterisation and antimicrobial activity of active polypropylene films containing oregano essential oil and Allium extract to be used in packaging for meat products. Food Addit. Contam. A 2018, 35, 782–791. [Google Scholar] [CrossRef] [PubMed]
- Dong, Z.; Xu, F.J.; Ahmed, I.; Li, Z.X.; Lin, H. Characterization and preservation performance of active polyethylene films containing rosemary and cinnamon essential oils for Pacific white shrimp packaging. Food Control 2018, 92, 37–46. [Google Scholar] [CrossRef]
- da Silva, C.F.; de Oliveira, F.S.M.; Gaetano, V.F.; Vinhas, G.M.; Cardoso, S.A. Orange essential oil as antimicrobial additives in poly(vinyl chloride) films. Polimeros 2018, 28, 332–338. [Google Scholar] [CrossRef]
- Krehula, L.K.; Papic, A.; Krehula, S.; Gilja, V.; Foglar, L.; Hrnjak-Murgic, Z. Properties of UV protective films of poly(vinyl-chloride)/TiO2 nanocomposites for food packaging. Polym. Bull. 2017, 74, 1387–1404. [Google Scholar] [CrossRef]
- Li, X.H.; Xing, Y.; Jiang, Y.H.; Ding, Y.L.; Li, W.L. Antimicrobial activities of ZnO powder-coated PVC film to inactivate food pathogens. Int. J. Food Sci. Technol. 2009, 44, 2161–2168. [Google Scholar] [CrossRef]
- Singh, S.; Lee, M.; Gaikwad, K.K.; Lee, Y.S. Antibacterial and amine scavenging properties of silver-silica composite for post-harvest storage of fresh fish. Food Bioprod. Process. 2018, 107, 61–69. [Google Scholar] [CrossRef]
- Gogliettino, M.; Balestrieri, M.; Ambrosio, R.L.; Anastasio, A.; Smaldone, G.; Proroga, Y.T.R.; Moretta, R.; Rea, I.; De Stefano, L.; Agrillo, B.; et al. Extending the Shelf-Life of Meat and Dairy Products via PET-Modified Packaging Activated With the Antimicrobial Peptide MTP1. Front. Microbiol. 2020, 10. [Google Scholar] [CrossRef] [PubMed]
- Silva, C.C.G.; Silva, S.P.M.; Ribeiro, S.C. Application of Bacteriocins and Protective Cultures in Dairy Food Preservation. Front. Microbiol. 2018, 9. [Google Scholar] [CrossRef]
- Santos, J.C.P.; Sousa, R.C.S.; Otoni, C.G.; Moraes, A.R.F.; Souza, V.G.L.; Medeiros, E.A.A.; Espitia, P.J.P.; Pires, A.C.S.; Coimbra, J.S.R.; Soares, N.F.F. Nisin and other antimicrobial peptides: Production, mechanisms of action, and application in active food packaging. Innov. Food Sci. Emerg. 2018, 48, 179–194. [Google Scholar] [CrossRef]
- Agrillo, B.; Balestrieri, M.; Gogliettino, M.; Palmieri, G.; Moretta, R.; Proroga, Y.T.R.; Rea, I.; Cornacchia, A.; Capuano, F.; Smaldone, G.; et al. Functionalized Polymeric Materials with Bio-Derived Antimicrobial Peptides for “Active” Packaging. Int. J. Mol. Sci. 2019, 20, 601. [Google Scholar] [CrossRef]
- Corrales-Urena, Y.R.; Souza-Schiaber, Z.; Lisboa, P.N.; Marquenet, F.; Michael Noeske, P.L.; Gatjen, L.; Rischka, K. Functionalization of hydrophobic surfaces with antimicrobial peptides immobilized on a bio-interfactant layer. RSC Adv. 2020, 10, 376–386. [Google Scholar] [CrossRef]
- Torres, L.M.F.C.; Braga, N.A.; Gomes, I.P.; Almeida, M.T.; Santos, T.L.; de Mesquita, J.P.; da Silva, L.M.; Martins, H.R.; Kato, K.C.; dos Santos, W.T.P.; et al. Nanobiostructure of fibrous-like alumina functionalized with an analog of the BP100 peptide: Synthesis, characterization and biological applications. Colloid Surf. B 2018, 163, 275–283. [Google Scholar] [CrossRef]
- Lu, P.; Guo, M.Y.; Xu, Z.J.; Wu, M. Application of Nanofibrillated Cellulose on BOPP/LDPE Film as Oxygen Barrier and Antimicrobial Coating Based on Cold Plasma Treatment. Coatings 2018, 8, 207. [Google Scholar] [CrossRef]
- Zhu, R.N.; Liu, X.; Song, P.P.; Wang, M.; Xu, F.; Jiang, Y.J.; Zhang, X.M. An approach for reinforcement of paper with high strength and barrier properties via coating regenerated cellulose. Carbohyd. Polym. 2018, 200, 100–105. [Google Scholar] [CrossRef] [PubMed]
- Shankar, S.; Rhim, J.W. Antimicrobial wrapping paper coated with a ternary blend of carbohydrates (alginate, carboxymethyl cellulose, carrageenan) and grapefruit seed extract. Carbohyd. Polym. 2018, 196, 92–101. [Google Scholar] [CrossRef] [PubMed]
- Divsalar, E.; Tajik, H.; Moradi, M.; Forough, M.; Lotfi, M.; Kuswandi, B. Characterization of cellulosic paper coated with chitosan-zinc oxide nanocomposite containing nisin and its application in packaging of UF cheese. Int. J. Biol. Macromol. 2018, 109, 1311–1318. [Google Scholar] [CrossRef] [PubMed]
- Brodnjak, U.V.; Tihole, K. Chitosan Solution Containing Zein and Essential Oil as Bio Based Coating on Packaging Paper. Coatings 2020, 10, 497. [Google Scholar] [CrossRef]
- Mandolini, M.; Campi, F.; Favi, C.; Germanil, M. Manufacturing processes re-engineering for cost reduction: The investment casting case study. In Proceedings of the Asme International Design Engineering Technical Conferences and Computers and Information in Engineering Conference, 2019, Vol 4; American Society of Mechanical Engineers: New York, NY, USA, 2020; Volume 4. [Google Scholar]
- Khezrian, A.; Shahbazi, Y. Application of nanocompostie chitosan and carboxymethyl cellulose films containing natural preservative compounds in minced camel’s meat. Int. J. Biol. Macromol. 2018, 106, 1146–1158. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.W.; Guo, C.R.; Hu, Y.Z.; Guo, Y.R.; Pan, Q.J. The preparation and antibacterial activity of cellulose/ZnO composite: A review. Open Chem. 2018, 16, 9–20. [Google Scholar] [CrossRef]
- Kaya, M.; Khadem, S.; Cakmak, Y.S.; Mujtaba, M.; Ilk, S.; Akyuz, L.; Salaberria, A.M.; Labidi, J.; Abdulqadir, A.H.; Deligoz, E. Antioxidative and antimicrobial edible chitosan films blended with stem, leaf and seed extracts of Pistacia terebinthus for active food packaging. RSC Adv. 2018, 8, 3941–3950. [Google Scholar] [CrossRef]
- Sun, L.J.; Sun, J.J.; Liu, D.J.; Fu, M.H.; Yang, X.; Guo, Y.R. The preservative effects of chitosan film incorporated with thinned young apple polyphenols on the quality of grass carp (Ctenopharyngodon idellus) fillets during cold storage: Correlation between the preservative effects and the active properties of the film. Food Packag. Shelf Life 2018, 17, 1–10. [Google Scholar] [CrossRef]
- Beltran, C.A.U.; Florez, I.F. Preparation of bio-packages from native starches and essential oils prolonging shelf life in strawberries. Rev. Colomb. Investig. 2018, 5, 71–86. [Google Scholar] [CrossRef]
- Issa, A.T.; Schimmel, K.A.; Worku, M.; Shahbazi, A.; Ibrahim, S.A.; Tahergorabi, R. Sweet Potato Starch-Based Nanocomposites: Development, Characterization, and Biodegradability. Starch-Starke 2018, 70. [Google Scholar] [CrossRef]
- Tedeschi, G.; Guzman-Puyol, S.; Ceseracciu, L.; Paul, U.C.; Picone, P.; Di Carlo, M.; Athanassiou, A.; Heredia-Guerrero, J.A. Multifunctional Bioplastics Inspired by Wood Composition: Effect of Hydrolyzed Lignin Addition to Xylan-Cellulose Matrices. Biomacromolecules 2020, 21, 910–920. [Google Scholar] [CrossRef] [PubMed]
- Nechita, P.; Roman, M. Review on Polysaccharides Used in Coatings for Food Packaging Papers. Coatings 2020, 10, 566. [Google Scholar] [CrossRef]
- Fabra, M.J.; Falco, I.; Randazzo, W.; Sanchez, G.; Lopez-Rubio, A. Antiviral and antioxidant properties of active alginate edible films containing phenolic extracts. Food Hydrocoll. 2018, 81, 96–103. [Google Scholar] [CrossRef]
- Aziz, M.S.A.; Salama, H.E.; Sabaa, M.W. Biobased alginate/castor oil edible films for active food packaging. LWT Food Sci. Technol. 2018, 96, 455–460. [Google Scholar] [CrossRef]
- Hu, Z.; Hong, P.Z.; Liao, M.N.; Kong, S.Z.; Huang, N.; Ou, C.Y.; Li, S.D. Preparation and Characterization of Chitosan-Agarose Composite Films. Materials 2016, 9, 816. [Google Scholar] [CrossRef]
- Kusznierewicz, B.; Staroszczyk, H.; Malinowska-Panczyk, E.; Parchem, K.; Bartoszek, A. Novel ABTS-dot-blot method for the assessment of antioxidant properties of food packaging. Food Packag. Shelf Life 2020, 24, 100478. [Google Scholar] [CrossRef]
- Lei, J.; Zhou, L.; Tang, Y.J.; Luo, Y.; Duan, T.; Zhu, W.K. High-Strength Konjac Glucomannan/Silver Nanowires Composite Films with Antibacterial Properties. Materials 2017, 10, 524. [Google Scholar] [CrossRef]
- Wu, C.H.; Li, Y.L.; Sun, J.S.; Lu, Y.Z.; Tong, C.L.; Wang, L.; Yan, Z.M.; Pang, J. Novel konjac glucomannan films with oxidized chitin nanocrystals immobilized red cabbage anthocyanins for intelligent food packaging. Food Hydrocoll. 2020, 98, 105245. [Google Scholar] [CrossRef]
- Li, S.B.; Yi, J.J.; Yu, X.M.; Wang, Z.Y.; Wang, L. Preparation and characterization of pullulan derivative/chitosan composite film for potential antimicrobial applications. Int. J. Biol. Macromol. 2020, 148, 258–264. [Google Scholar] [CrossRef]
- Hassan, A.H.A.; Cutter, C.N. Development and evaluation of pullulan-based composite antimicrobial films (CAF) incorporated with nisin, thymol and lauric arginate to reduce foodborne pathogens associated with muscle foods. Int. J. Food Microbiol. 2020, 320, 108519. [Google Scholar] [CrossRef]
- da Silva, F.T.; da Cunha, K.F.; Fonseca, L.M.; Antunes, M.D.; El Halal, S.L.M.; Fiorentini, A.M.; Zavareze, E.D.; Dias, A.R.G. Action of ginger essential oil (Zingiber officinale) encapsulated in proteins ultrafine fibers on the antimicrobial control in situ. Int. J. Biol. Macromol. 2018, 118, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Echeverria, I.; Lopez-Caballero, M.E.; Gomez-Guillen, M.C.; Mauri, A.N.; Montero, M.P. Active nanocomposite films based on soy proteins-montmorillonite- clove essential oil for the preservation of refrigerated bluefin tuna (Thunnus thynnus) fillets. Int. J. Food Microbiol. 2018, 266, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Kouravand, F.; Jooyandeh, H.; Barzegar, H.; Hojjati, M. Characterization of cross-linked whey protein isolate-based films containing Satureja Khuzistanica Jamzad essential oil. J. Food Process. Perservation 2018, 42. [Google Scholar] [CrossRef]
- Aziz, S.G.G.; Almasi, H. Physical Characteristics, Release Properties, and Antioxidant and Antimicrobial Activities of Whey Protein Isolate Films Incorporated with Thyme (Thymus vulgaris L.) Extract-Loaded Nanoliposomes. Food Bioprocess. Technol. 2018, 11, 1552–1565. [Google Scholar] [CrossRef]
- Altan, A.; Aytac, Z.; Uyar, T. Carvacrol loaded electrospun fibrous films from zein and poly(lactic acid) for active food packaging. Food Hydrocoll. 2018, 81, 48–59. [Google Scholar] [CrossRef]
- Ahmad, M.; Nirmal, N.P.; Danish, M.; Chuprom, J.; Jafarzedeh, S. Characterisation of composite films fabricated from collagen/chitosan and collagen/soy protein isolate for food packaging applications. RSC Adv. 2016, 6, 82191–82204. [Google Scholar] [CrossRef]
- Ocak, B. Film-forming ability of collagen hydrolysate extracted from leather solid wastes with chitosan. Environ. Sci. Pollut. Res. 2018, 25, 4643–4655. [Google Scholar] [CrossRef]
- Figueroa-Lopez, K.J.; Castro-Mayorga, J.L.; Andrade-Mahecha, M.M.; Cabedo, L.; Lagaron, J.M. Antibacterial and Barrier Properties of Gelatin Coated by Electrospun Polycaprolactone Ultrathin Fibers Containing Black Pepper Oleoresin of Interest in Active Food Biopackaging Applications. Nanomaterials 2018, 8, 199. [Google Scholar] [CrossRef]
- Kim, H.; Beak, S.E.; Song, K.B. Development of a hagfish skin gelatin film containing cinnamon bark essential oil. LWT Food Sci. Technol. 2018, 96, 583–588. [Google Scholar] [CrossRef]
- Abdollahzadeh, E.; Ojagh, S.M.; Fooladi, A.A.I.; Shabanpour, B.; Gharahei, M. Effects of Probiotic Cells on the Mechanical and Antibacterial Properties of Sodium-Caseinate Films. Appl. Food Biotechnol. 2018, 5, 155–162. [Google Scholar] [CrossRef]
- Chevalier, E.; Assezat, G.; Prochazka, F.; Oulahal, N. Development and characterization of a novel edible extruded sheet based on different casein sources and influence of the glycerol concentration. Food Hydrocoll. 2018, 75, 182–191. [Google Scholar] [CrossRef]
- Akyuz, L.; Kaya, M.; Ilk, S.; Cakmak, Y.S.; Salaberria, A.M.; Labidi, J.; Yilmaz, B.A.; Sargin, I. Effect of different animal fat and plant oil additives on physicochemical, mechanical, antimicrobial and antioxidant properties of chitosan films. Int. J. Biol. Macromol. 2018, 111, 475–484. [Google Scholar] [CrossRef] [PubMed]
- Tonyali, B.; McDaniel, A.; Amamcharla, J.; Trinetta, V.; Yucel, U. Release kinetics of cinnamaldehyde, eugenol, and thymol from sustainable and biodegradable active packaging films. Food Packag. Shelf Life 2020, 24, 100484. [Google Scholar] [CrossRef]
- Saurabh, C.K.; Gupta, S.; Variyar, P.S. Development of guar gum based active packaging films using grape pomace. J. Food Sci. Technol. 2018, 55, 1982–1992. [Google Scholar] [CrossRef]
- Zhang, D.; Yu, G.H.; Long, Z.; Xiao, H.N.; Qian, L.Y. Bio-Wax Latex-Modified Paper as Antimicrobial and Water-Vapor-Resistant Packaging Material. J. Wood Chem. Technol. 2016, 36, 182–191. [Google Scholar] [CrossRef]
- Swaroop, C.; Shukla, M. Nano-magnesium oxide reinforced polylactic acid biofilms for food packaging applications. Int. J. Biol. Macromol. 2018, 113, 729–736. [Google Scholar] [CrossRef] [PubMed]
- Lopusiewicz, L.; Jedra, F.; Mizielinska, M. New Poly(lactic acid) Active Packaging Composite Films Incorporated with Fungal Melanin. Polymers 2018, 10, 386. [Google Scholar] [CrossRef]
- Shao, P.; Yan, Z.P.; Chen, H.J.; Xiao, J. Electrospun poly(vinyl alcohol)/permutite fibrous film loaded with cinnamaldehyde for active food packaging. J. Appl. Polym. Sci. 2018, 135. [Google Scholar] [CrossRef]
- Arrieta, M.P.; de Dicastillo, C.L.; Garrido, L.; Roa, K.; Galotto, M.J. Electrospun PVA fibers loaded with antioxidant fillers extracted from Durvillaea antarctica algae and their effect on plasticized PLA bionanocomposites. Eur. Polym. J. 2018, 103, 145–157. [Google Scholar] [CrossRef]
- Chen, C.W.; Xu, Z.W.; Ma, Y.R.; Liu, J.L.; Zhang, Q.J.; Tang, Z.P.; Fu, K.J.; Yang, F.X.; Xie, J. Properties, vapour-phase antimicrobial and antioxidant activities of active poly(vinyl alcohol) packaging films incorporated with clove oil. Food Control 2018, 88, 105–112. [Google Scholar] [CrossRef]
- Baek, N.; Kim, Y.T.; Marcy, J.E.; Duncan, S.E.; O‘Keefe, S.F. Physical properties of nanocomposite polylactic acid films prepared with oleic acid modified titanium dioxide. Food Packag. Shelf Life 2018, 17, 30–38. [Google Scholar] [CrossRef]
- Alboofetileh, M.; Rezaei, M.; Hosseini, H.; Abdollahi, M. Morphological, physico-mechanical, and antimicrobial properties of sodium alginate-montmorillonite nanocomposite films incorporated with marjoram essential oil. J. Food Process. Perservation 2018, 42, e13596. [Google Scholar] [CrossRef]
- Youssef, A.M.; Abdel-Aziz, M.E.; El-Sayed, E.S.A.; Abdel-Aziz, M.S.; El-Hakim, A.A.A.; Kamel, S.; Turky, G. Morphological, electrical & antibacterial properties of trilayered Cs/PAA/PPy bionanocomposites hydrogel based on Fe3O4-NPs. Carbohyd. Polym. 2018, 196, 483–493. [Google Scholar] [CrossRef]
- Yousuf, B.; Qadri, O.S.; Srivastava, A.K. Recent developments in shelf-life extension of fresh-cut fruits and vegetables by application of different edible coatings: A review. LWT Food Sci. Technol. 2018, 89, 198–209. [Google Scholar] [CrossRef]
- Zhang, C.; Li, W.H.; Zhu, B.F.; Chen, H.Y.; Chi, H.; Li, L.; Qin, Y.Y.; Xue, J. The Quality Evaluation of Postharvest Strawberries Stored in Nano-Ag Packages at Refrigeration Temperature. Polymers 2018, 10, 894. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Gaikwad, K.K.; Lee, Y.S. Antimicrobial and antioxidant properties of polyvinyl alcohol bio composite films containing seaweed extracted cellulose nano-crystal and basil leaves extract. Int. J. Biol. Macromol. 2018, 107, 1879–1887. [Google Scholar] [CrossRef]
- Youssef, A.M.; El-Sayed, S.M. Bionanocomposites materials for food packaging applications: Concepts and future outlook. Carbohyd. Polym. 2018, 193, 19–27. [Google Scholar] [CrossRef]
- Maleki, G.; Sedaghat, N.; Woltering, E.J.; Farhoodi, M.; Mohebbi, M. Chitosan-limonene coating in combination with modified atmosphere packaging preserve postharvest quality of cucumber during storage. J. Food Meas. Charact. 2018, 12, 1610–1621. [Google Scholar] [CrossRef]
- Kaewklin, P.; Siripatrawan, U.; Suwanagul, A.; Lee, Y.S. Active packaging from chitosan-titanium dioxide nanocomposite film for prolonging storage life of tomato fruit. Int. J. Biol. Macromol. 2018, 112, 523–529. [Google Scholar] [CrossRef]
- Yan, J.; Luo, Z.; Ban, Z.; Lu, H.; Li, D.; Yang, D.; Aghdam, M.S.; Li, L. The effect of the layer-by-layer (LBL) edible coating on strawberry qualityand metabolites during storage. Postharvest Biol. Technol. 2019, 147, 29–38. [Google Scholar] [CrossRef]
- Shahbazi, Y. Application of carboxymethyl cellulose and chitosan coatings containing Mentha spicata essential oil in fresh strawberries. Int. J. Biol. Macromol. 2018, 112, 264–272. [Google Scholar] [CrossRef] [PubMed]
- Li, L.L.; Wang, H.L.; Chen, M.M.; Jiang, S.W.; Jiang, S.T.; Li, X.J.; Wang, Q.Y. Butylated hydroxyanisole encapsulated in gelatin fiber mats: Volatile release kinetics, functional effectiveness and application to strawberry preservation. Food Chem. 2018, 269, 142–149. [Google Scholar] [CrossRef] [PubMed]
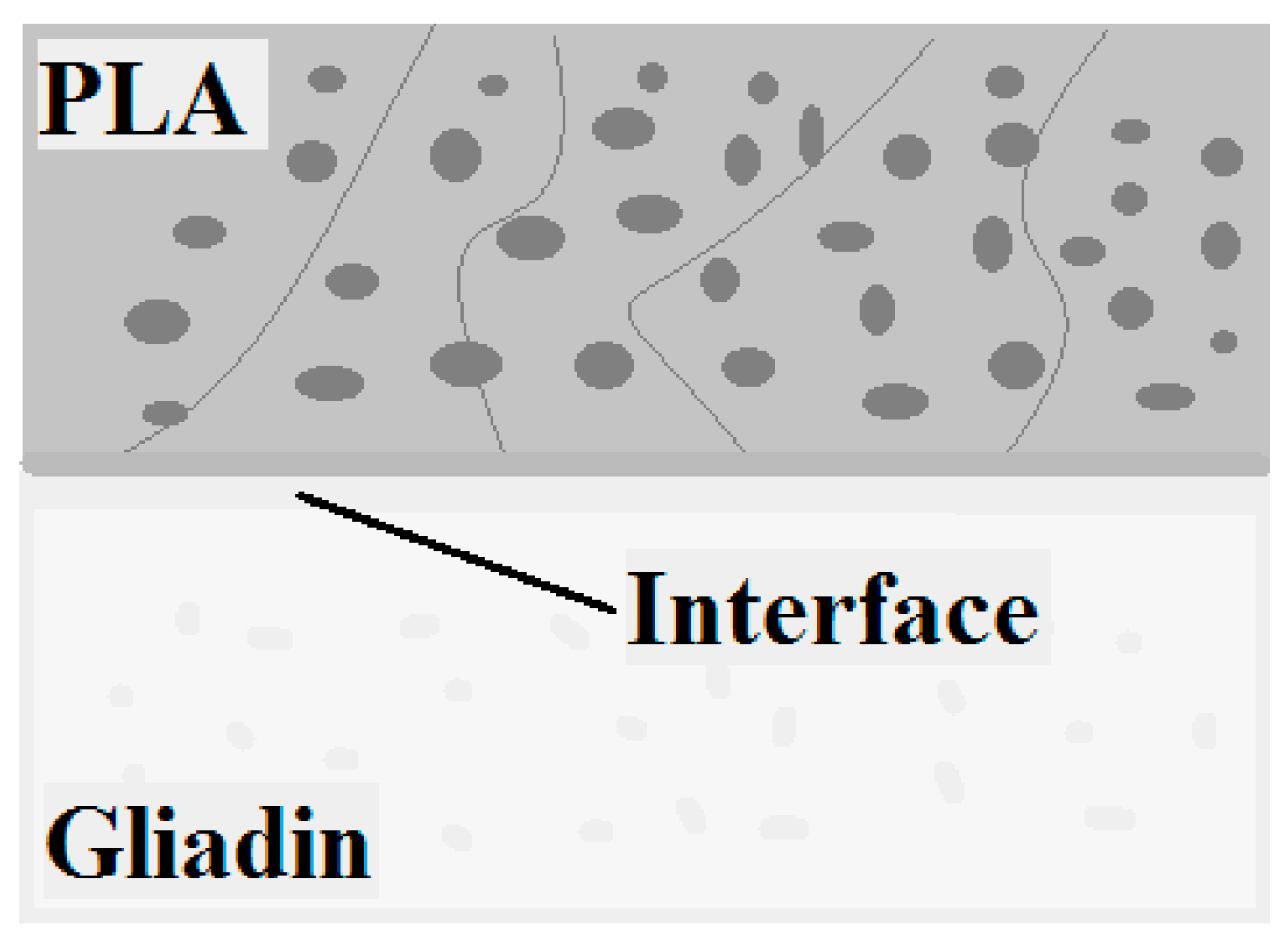
- Zhu, J.Y.; Tang, C.H.; Yin, S.W.; Yang, X.Q. Development and characterisation of polylactic acid-gliadin bilayer/trilayer films as carriers of thymol. Int. J. Food Sci. Technol. 2018, 53, 608–618. [Google Scholar] [CrossRef]
- Rezaei, F.; Shahbazi, Y. Shelf-life extension and quality attributes of sauced silver carp fillet: A comparison among direct addition, edible coating and biodegradable film. LWT Food Sci. Technol. 2018, 87, 122–133. [Google Scholar] [CrossRef]
- Hassanzadeh, P.; Moradi, M.; Vaezi, N.; Moosavy, M.H.; Mahmoudi, R. Effects of chitosan edible coating containing grape seed extract on the shelf-life of refrigerated rainbow trout fillet. Vet. Res. Forum. 2018, 9, 73–79. [Google Scholar]
- Ma, Y.C.; Li, L.; Wang, Y.F. Development of PLA-PHB-based biodegradable active packaging and its application to salmon. Packag. Technol. Sci. 2018, 31, 739–746. [Google Scholar] [CrossRef]
- Wang, Q.Y.; Lei, J.; Ma, J.J.; Yuan, G.F.; Sun, H.Y. Effect of chitosan-carvacrol coating on the quality of Pacific white shrimp during iced storage as affected by caprylic acid. Int. J. Biol. Macromol. 2018, 106, 123–129. [Google Scholar] [CrossRef]
- Ejaz, M.; Arfat, Y.A.; Mulla, M.; Ahmed, J. Zinc oxide nanorods/clove essential oil incorporated Type B gelatin composite films and its applicability for shrimp packaging. Food Packag. Shelf Life 2018, 15, 113–121. [Google Scholar] [CrossRef]
- Zegarra, M.D.C.P.; Santos, A.M.P.; Silva, A.M.A.D.; Melo, E.D. Chitosan films incorporated with antioxidant extract of acerola agroindustrial residue applied in chicken thigh. J. Food Process. Perservation 2018, 42. [Google Scholar] [CrossRef]
- Souza, V.G.L.; Pires, J.R.A.; Vieira, E.T.; Coelhoso, I.M.; Duarte, M.P.; Fernando, A.L. Shelf Life Assessment of Fresh Poultry Meat Packaged in Novel Bionanocomposite of Chitosan/Montmorillonite Incorporated with Ginger Essential Oil. Coatings 2018, 8, 177. [Google Scholar] [CrossRef]
- Pires, J.R.A.; de Souza, V.G.L.; Fernando, A.L. Chitosan/montmorillonite bionanocomposites incorporated with rosemary and ginger essential oil as packaging for fresh poultry meat. Food Packag. Shelf Life 2018, 17, 142–149. [Google Scholar] [CrossRef]
- Lin, L.; Zhu, Y.L.; Cui, H.Y. Electrospun thyme essential oil/gelatin nanofibers for active packaging against Campylobacter jejuni in chicken. LWT Food Sci. Technol. 2018, 97, 711–718. [Google Scholar] [CrossRef]
- Yang, W.E.; Xie, Y.H.; Jin, J.H.; Liu, H.; Zhang, H.X. Development and Application of an Active Plastic Multilayer Film by Coating a Plantaricin BM-1 for Chilled Meat Preservation. J. Food Sci. 2019, 84, 1864–1870. [Google Scholar] [CrossRef] [PubMed]
- Rad, F.H.; Sharifan, A.; Asadi, G. Physicochemical and antimicrobial properties of kefiran /waterborne polyurethane film incorporated with essential oils on refrigerated ostrich meat. LWT Food Sci. Technol. 2018, 97, 794–801. [Google Scholar] [CrossRef]
- Pabast, M.; Shariatifar, N.; Beikzadeh, S.; Jahed, G. Effects of chitosan coatings incorporating with free or nano-encapsulated Satureja plant essential oil on quality characteristics of lamb meat. Food Control 2018, 91, 185–192. [Google Scholar] [CrossRef]
- Talebi, F.; Misaghi, A.; Khanjari, A.; Kamkar, A.; Gandomi, H.; Rezaeigolestani, M. Incorporation of spice essential oils into poly-lactic acid film matrix with the aim of extending microbiological and sensorial shelf life of ground beef. LWT Food Sci. Technol. 2018, 96, 482–490. [Google Scholar] [CrossRef]
- Zhao, Y.J.; Teixeira, J.S.; Ganzle, M.M.; Saldana, M.D.A. Development of antimicrobial films based on cassava starch, chitosan and gallic acid using subcritical water technology. J. Supercrit. Fluid 2018, 137, 101–110. [Google Scholar] [CrossRef]
- Ribeiro-Santos, R.; de Melo, N.R.; Andrade, M.; Azevedo, G.; Machado, A.V.; Carvalho-Costa, D.; Sanches-Silva, A. Whey protein active films incorporated with a blend of essential oils: Characterization and effectiveness. Packag. Technol. Sci. 2018, 31, 27–40. [Google Scholar] [CrossRef]
- Youssef, A.M.; El-Sayed, S.M.; El-Sayed, H.S.; Salama, H.H.; Assem, F.M.; Abd El-Salam, M.H. Novel bionanocomposite materials used for packaging skimmed milk acid coagulated cheese (Karish). Int. J. Biol. Macromol. 2018, 115, 1002–1011. [Google Scholar] [CrossRef]
- Lotfi, M.; Tajik, H.; Moradi, M.; Forough, M.; Divsalar, E.; Kuswandi, B. Nanostructured chitosan/monolaurin film: Preparation, characterization and antimicrobial activity against Listeria monocytogenes on ultrafiltered white cheese. LWT Food Sci. Technol. 2018, 92, 576–583. [Google Scholar] [CrossRef]
- Yang, S.Y.; Cao, L.; Kim, H.; Beak, S.E.; Bin Song, K. Utilization of Foxtail Millet Starch Film Incorporated with Clove Leaf Oil for the Packaging of Queso Blanco Cheese as a Model Food. Starch-Starke 2018, 70. [Google Scholar] [CrossRef]
- Guitian, M.V.; Ibarguren, C.; Soria, M.C.; Hovanyecz, P.; Banchio, C.; Audisio, M.C. Anti-Listeria monocytogenes effect of bacteriocin-incorporated agar edible coatings applied on cheese. Int. Dairy J. 2019, 97, 92–98. [Google Scholar] [CrossRef]
- Mushtaq, M.; Gani, A.; Gani, A.; Punoo, H.A.; Masoodi, F.A. Use of pomegranate peel extract incorporated zein film with improved properties for prolonged shelf life of fresh Himalayan cheese (Kalari/kradi). Innov. Food Sci. Emerg. 2018, 48, 25–32. [Google Scholar] [CrossRef]
- Orsuwan, A.; Sothornvit, R. Active Banana Flour Nanocomposite Films Incorporated with Garlic Essential Oil as Multifunctional Packaging Material for Food Application. Food Bioprocess. Technol. 2018, 11, 1199–1210. [Google Scholar] [CrossRef]
- Pavli, F.; Tassou, C.; Nychas, G.J.E.; Chorianopoulos, N. Probiotic Incorporation in Edible Films and Coatings: Bioactive Solution for Functional Foods. Int. J. Mol. Sci. 2018, 19, 150. [Google Scholar] [CrossRef]
- Yousefi, M.; Azizi, M.; Ehsani, A. Antimicrobial coatings and films on meats: A perspective on the application of antimicrobial edible films or coatings on meats from the past to future. Bali Med. J. 2018, 7, 87–96. [Google Scholar] [CrossRef]
- Wang, H.X.; Qan, J.; Ding, F.Y. Emerging Chitosan-Based Films for Food Packaging Applications. J. Agric. Food Chem. 2018, 66, 395–413. [Google Scholar] [CrossRef]
- Dutta, A.S.P.K. Extraction of Chitin-Glucan Complex from Agaricus bisporus: Characterization and Antibacterial Activity. J. Polym. Mater. 2017, 34, 1–9. [Google Scholar]
- Priyadarshi, R.; Sauraj; Kumar, B.; Negi, Y.S. Chitosan film incorporated with citric acid and glycerol as an active packaging material for extension of green chilli shelf life. Carbohyd. Polym. 2018, 195, 329–338. [Google Scholar] [CrossRef]
- Sogut, E.; Seydim, A.C. Development of Chitosan and Polycaprolactone based active bilayer films enhanced with nanocellulose and grape seed extract. Carbohyd. Polym. 2018, 195, 180–188. [Google Scholar] [CrossRef]
- Xu, Y.X.; Willis, S.; Jordan, K.; Sismour, E. Chitosan nanocomposite films incorporating cellulose nanocrystals and grape pomace extracts. Packag. Technol. Sci. 2018, 31, 631–638. [Google Scholar] [CrossRef]
- Uranga, J.; Puertas, A.I.; Etxabide, A.; Duenas, M.T.; Guerrero, P.; de la Caba, K. Citric acid-incorporated fish gelatin/chitosan composite films. Food Hydrocoll. 2019, 86, 95–103. [Google Scholar] [CrossRef]
- Uranga, J.; Etxabide, A.; Guerrero, P.; de la Caba, K. Development of active fish gelatin films with anthocyanins by compression molding. Food Hydrocoll. 2018, 84, 313–320. [Google Scholar] [CrossRef]
- Ramziia, S.; Ma, H.; Yao, Y.Z.; Wei, K.R.; Huang, Y.Q. Enhanced antioxidant activity of fish gelatin-chitosan edible films incorporated with procyanidin. J. Appl. Polym. Sci. 2018, 135. [Google Scholar] [CrossRef]
- Perez-Cordoba, L.J.; Norton, I.T.; Batchelor, H.K.; Gkatzionis, K.; Spyropoulos, F.; Sobral, P.J.A. Physico-chemical, antimicrobial and antioxidant properties of gelatin-chitosan based films loaded with nanoemulsions encapsulating active compounds. Food Hydrocoll. 2018, 79, 544–559. [Google Scholar] [CrossRef]
- Soni, B.; Mahmoud, B.; Chang, S.; El-Giar, E.M.; Hassan, E.B. Physicochemical, antimicrobial and antioxidant properties of chitosan/TEMPO biocomposite packaging films. Food Packag. Shelf Life 2018, 17, 73–79. [Google Scholar] [CrossRef]
- Robledo, N.; Lopez, L.; Bunger, A.; Tapia, C.; Abugoch, L. Effects of Antimicrobial Edible Coating of Thymol Nanoemulsion/Quinoa Protein/Chitosan on the Safety, Sensorial Properties, and Quality of Refrigerated Strawberries (Fragaria × ananassa) Under Commercial Storage Environment. Food Bioprocess. Technol. 2018, 11, 1566–1574. [Google Scholar] [CrossRef]
- Yun, Y.H.; Lee, C.M.; Kim, Y.S.; Yoon, S.D. Preparation of chitosan/polyvinyl alcohol blended films containing sulfosuccinic acid as the crosslinking agent using UV curing process. Food Res. Int. 2017, 100, 377–386. [Google Scholar] [CrossRef]
- Yang, W.; Fortunati, E.; Bertoglio, F.; Owczarek, J.S.; Bruni, G.; Kozanecki, M.; Kenny, J.M.; Torre, L.; Visai, L.; Puglia, D. Polyvinyl alcohol/chitosan hydrogels with enhanced antioxidant and antibacterial properties induced by lignin nanoparticles. Carbohyd. Polym. 2018, 181, 275–284. [Google Scholar] [CrossRef]
- Warsiki, E. Application of chitosan as biomaterial for active packaging of ethylene absorber. In Proceedings of the 2nd International Conference on Biomass: Toward Sustainable Biomass Utilization for Industrial and Energy Applications, Bogor, Indonesia, 24–25 July 2017; Volume 141. [Google Scholar] [CrossRef]
- Souza, V.G.L.; Rodrigues, C.; Valente, S.; Pimenta, C.; Pires, J.R.A.; Alves, M.M.; Santos, C.F.; Coelhoso, I.M.; Fernando, A.L. Eco-Friendly ZnO/Chitosan Bionanocomposites Films for Packaging of Fresh Poultry Meat. Coatings 2020, 10, 110. [Google Scholar] [CrossRef]
- Suo, B.; Li, H.R.; Wang, Y.X.; Li, Z.; Pan, Z.L.; Ai, Z.L. Effects of ZnO nanoparticle-coated packaging film on pork meat quality during cold storage. J. Sci. Food Agric. 2017, 97, 2023–2029. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Mudai, A.; Roy, B.; Basumatary, I.B.; Mukherjee, A.; Dutta, J. Biodegradable Hybrid Nanocomposite of Chitosan/Gelatin and Green Synthesized Zinc Oxide Nanoparticles for Food Packaging. Foods 2020, 9, 1143. [Google Scholar] [CrossRef] [PubMed]
- Zheng, K.W.; Li, W.; Fu, B.Q.; Fu, M.F.; Ren, Q.L.; Yang, F.; Qin, C.Q. Physical, antibacterial and antioxidant properties of chitosan films containing hardleaf oatchestnut starch and Litsea cubeba oil. Int. J. Biol. Macromol. 2018, 118, 707–715. [Google Scholar] [CrossRef] [PubMed]
- Sharma, H.; Mendiratta, S.K.; Agrawal, R.K.; Talukder, S.; Kumar, S. Studies on the potential application of various blends of essential oils as antioxidant and antimicrobial preservatives in emulsion based chicken sausages. Br. Food J. 2018, 120, 1398–1411. [Google Scholar] [CrossRef]
- Shahbazi, Y.; Karami, N.; Shavisi, N. Effect of Mentha spicata essential oil on chemical, microbial, and sensory properties of minced camel meat during refrigerated storage. J. Food Saf. 2018, 38. [Google Scholar] [CrossRef]
- Azadbakht, E.; Maghsoudlou, Y.; Khomiri, M.; Kashiri, M. Development and structural characterization of chitosan films containing Eucalyptus globulus essential oil: Potential as an antimicrobial carrier for packaging of sliced sausage. Food Packag. Shelf Life 2018, 17, 65–72. [Google Scholar] [CrossRef]
- Niu, B.; Yan, Z.P.; Shao, P.; Kang, J.; Chen, H.J. Encapsulation of Cinnamon Essential Oil for Active Food Packaging Film with Synergistic Antimicrobial Activity. Nanomaterials 2018, 8, 598. [Google Scholar] [CrossRef]
- Dolea, D.; Rizo, A.; Fuentes, A.; Barat, J.M.; Fernandez-Segovia, I. Effect of thyme and oregano essential oils on the shelf life of salmon and seaweed burgers. Food Sci. Technol. Int. 2018, 24, 394–403. [Google Scholar] [CrossRef]
- Ju, J.; Xu, X.M.; Xie, Y.F.; Guo, Y.H.; Cheng, Y.L.; Qian, H.; Yao, W.R. Inhibitory effects of cinnamon and clove essential oils on mold growth on baked foods. Food Chem. 2018, 240, 850–855. [Google Scholar] [CrossRef]
- Priyadarshi, R.; Sauraj; Kumar, B.; Deeba, F.; Kulshreshtha, A.; Negi, Y.S. Chitosan films incorporated with Apricot (Prunus armeniaca) kernel essential oil as active food packaging material. Food Hydrocoll. 2018, 85, 158–166. [Google Scholar] [CrossRef]
- Zhang, Z.J.; Li, N.; Li, H.Z.; Li, X.J.; Cao, J.M.; Zhang, G.P.; He, D.L. Preparation and characterization of biocomposite chitosan film containing Perilla frutescens (L.) Britt. essential oil. Ind. Crops Prod. 2018, 112, 660–667. [Google Scholar] [CrossRef]
- Ojagh, S.M.; Rezaei, M.; Razavi, S.H.; Hosseini, S.M.H. Effect of chitosan coatings enriched with cinnamon oil on the quality of refrigerated rainbow trout. Food Chem. 2010, 120, 193–198. [Google Scholar] [CrossRef]
- Xing, Y.G.; Lin, H.B.; Cao, D.; Xu, Q.L.; Han, W.F.; Wang, R.R.; Che, Z.M.; Li, X.H. Effect of Chitosan Coating with Cinnamon Oil on the Quality and Physiological Attributes of China Jujube Fruits. Biomed. Res. Int. 2015, 835151. [Google Scholar] [CrossRef] [PubMed]
- Srikandace, Y.; Indrarti, L.; Indriyati; Sancoyorini, M.K. Antibacterial activity of bacterial cellulose-based edible film incorporated with Citrus spp essential oil. IOP Conf. Ser. Earth Environ. 2018, 160. [Google Scholar] [CrossRef]
- Ortiz, C.M.; Salgado, P.R.; Dufresne, A.; Mauri, A.N. Microfibrillated cellulose addition improved the physicochemical and bioactive properties of biodegradable films based on soy protein and clove essential oil. Food Hydrocoll. 2018, 79, 416–427. [Google Scholar] [CrossRef]
- Yu, Z.L.; Sun, L.; Wang, W.; Zeng, W.C.; Mustapha, A.; Lin, M.S. Soy protein-based films incorporated with cellulose nanocrystals and pine needle extract for active packaging. Ind. Crops Prod. 2018, 112, 412–419. [Google Scholar] [CrossRef]
- Fardioui, M.; Kadmiri, I.M.; Qaiss, A.E.; Bouhfid, R. Bio-active nanocomposite films based on nanocrystalline cellulose reinforced styrylquinoxalin-grafted-chitosan: Antibacterial and mechanical properties. Int. J. Biol. Macromol. 2018, 114, 733–740. [Google Scholar] [CrossRef]
- Freire, C.S.R.; Fernandes, S.C.M.; Silvestre, A.J.D.; Neto, C.P. Novel cellulose-based composites based on nanofibrillated plant and bacterial cellulose: Recent advances at the University of Aveiro—A review. Holzforschung 2013, 67, 603–612. [Google Scholar] [CrossRef]
- Shabanpour, B.; Kazemi, M.; Ojagh, S.M.; Pourashouri, P. Bacterial cellulose nanofibers as reinforce in edible fish myofibrillar protein nanocomposite films. Int. J. Biol. Macromol. 2018, 117, 742–751. [Google Scholar] [CrossRef]
- Gilbert, J.; Cheng, C.J.; Jones, O.G. Vapor Barrier Properties and Mechanical Behaviors of Composite Hydroxypropyl Methylcelluose/Zein Nanoparticle Films. Food Biophys. 2018, 13, 25–36. [Google Scholar] [CrossRef]
- Shen, Z.; Kamdem, D.P. Antimicrobial activity of sugar beet lignocellulose films containing tung oil and cedarwood essential oil. Cellulose 2015, 22, 2703–2715. [Google Scholar] [CrossRef]
- Coma, V.; Sebti, I.; Pardon, P.; Deschamps, A.; Pichavant, F.H. Antimicrobial edible packaging based on cellulosic ethers, fatty acids, and nisin incorporation to inhibit Listeria innocua and Staphylococcus aureus. J. Food Protect. 2001, 64, 470–475. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.M.; Shu, Y.; Su, S.P.; Zhu, J. One-step coagulation to construct durable anti-fouling and antibacterial cellulose film exploiting Ag@AgCl nanoparticle- triggered photo-catalytic degradation. Carbohyd. Polym. 2018, 181, 499–505. [Google Scholar] [CrossRef] [PubMed]
- Zahedi, Y.; Fathi-Achachlouei, B.; Yousefi, A.R. Physical and mechanical properties of hybrid montmorillonite/zinc oxide reinforced carboxymethyl cellulose nanocomposites. Int. J. Biol. Macromol. 2018, 108, 863–873. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, P.; Hubbe, M.A.; Lucia, L.; Pal, L. High performance nanocellulose-based composite coatings for oil and grease resistance. Cellulose 2018, 25, 3377–3391. [Google Scholar] [CrossRef]
- Shahbazi, Y. Characterization of nanocomposite films based on chitosan and carboxymethylcellulose containing Ziziphora clinopodioides essential oil and methanolic Ficus carica extract. J. Food Process. Perservation 2018, 42. [Google Scholar] [CrossRef]
- Wu, Z.G.; Huang, X.J.; Li, Y.C.; Xiao, H.Z.; Wang, X.Y. Novel chitosan films with laponite immobilized Ag nanoparticles for active food packaging. Carbohyd. Polym. 2018, 199, 210–218. [Google Scholar] [CrossRef]
- Hu, D.Y.; Wang, H.X.; Wang, L.J. Physical properties and antibacterial activity of quaternized chitosan/carboxymethyl cellulose blend films. LWT Food Sci. Technol. 2016, 65, 398–405. [Google Scholar] [CrossRef]
- Salari, M.; Khiabani, M.S.; Mokarram, R.R.; Ghanbarzadeh, B.; Kafil, H.S. Development and evaluation of chitosan based active nanocomposite films containing bacterial cellulose nanocrystals and silver nanoparticles. Food Hydrocoll. 2018, 84, 414–423. [Google Scholar] [CrossRef]
- Nunes, M.R.; Castilho, M.D.M.; Veeck, A.P.D.; da Rosa, C.G.; Noronha, C.M.; Maciel, M.V.O.B.; Barreto, P.M. Antioxidant and antimicrobial methylcellulose films containing Lippia alba extract and silver nanoparticles. Carbohyd. Polym. 2018, 192, 37–43. [Google Scholar] [CrossRef]
- Shankar, S.; Oun, A.A.; Rhim, J.W. Preparation of antimicrobial hybrid nano-materials using regenerated cellulose and metallic nanoparticles. Int. J. Biol. Macromol. 2018, 107, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Caetano, K.D.; Lopes, N.A.; Costa, T.M.H.; Brandelli, A.; Rodrigues, E.; Flores, S.H.; Cladera-Olivera, F. Characterization of active biodegradable films based on cassava starch and natural compounds. Food Packag. Shelf Life 2018, 16, 138–147. [Google Scholar] [CrossRef]
- Dinika, I.; Utama, G.L. Cheese whey as potential resource for antimicrobial edible film and active packaging production. Food Raw Mater. 2019, 7, 229–239. [Google Scholar] [CrossRef]
- Trongchuen, K.; Ounkaew, A.; Kasemsiri, P.; Hiziroglu, S.; Mongkolthanaruk, W.; Wannasutta, R.; Pongsa, U.; Chindaprasirt, P. Bioactive Starch Foam Composite Enriched With Natural Antioxidants from Spent Coffee Ground and Essential Oil. Starch-Starke 2018, 70. [Google Scholar] [CrossRef]
- Ounkaew, A.; Kasemsiri, P.; Kamwilaisak, K.; Saengprachatanarug, K.; Mongkolthanaruk, W.; Souvanh, M.; Pongsa, U.; Chindaprasirt, P. Polyvinyl Alcohol (PVA)/Starch Bioactive Packaging Film Enriched with Antioxidants from Spent Coffee Ground and Citric Acid. J. Polym. Environ. 2018, 26, 3762–3772. [Google Scholar] [CrossRef]
- Iamareerat, B.; Singh, M.; Sadiq, M.B.; Anal, A.K. Reinforced cassava starch based edible film incorporated with essential oil and sodium bentonite nanoclay as food packaging material. J. Food Sci. Technol. 2018, 55, 1953–1959. [Google Scholar] [CrossRef]
- Jash, A.; Paliyath, G.; Lim, L.T. Activated release of bioactive aldehydes from their precursors embedded in electrospun poly(lactic acid) nonwovens. RSC Adv. 2018, 8, 19930–19938. [Google Scholar] [CrossRef]
- Beak, S.; Kim, H.; Song, K.B. Sea Squirt Shell Protein and Polylactic Acid Laminated Films Containing Cinnamon Bark Essential Oil. J. Food Sci. 2018, 83, 1896–1903. [Google Scholar] [CrossRef]
- Yin, X.; Sun, C.; Tian, M.Y.; Wang, Y. Preparation and Characterization of Salicylic Acid/Polylactic Acid Composite Packaging Materials. Lect. Notes Electr. Eng. 2018, 477, 811–818. [Google Scholar] [CrossRef]
- Ye, J.S.; Wang, S.Y.; Lan, W.J.; Qin, W.; Liu, Y.W. Preparation and properties of polylactic acid-tea polyphenol-chitosan composite membranes. Int. J. Biol. Macromol. 2018, 117, 632–639. [Google Scholar] [CrossRef]
- Ahmed, J.; Arfat, Y.A.; Bher, A.; Mulla, M.; Jacob, H.; Auras, R. Active Chicken Meat Packaging Based on Polylactide Films and Bimetallic Ag-Cu Nanoparticles and Essential Oil. J. Food Sci. 2018, 83, 1299–1310. [Google Scholar] [CrossRef] [PubMed]
- Li, W.H.; Zhang, C.; Chi, H.; Li, L.; Lan, T.Q.; Han, P.; Chen, H.Y.; Qin, Y.Y. Development of Antimicrobial Packaging Film Made from Poly(Lactic Acid) Incorporating Titanium Dioxide and Silver Nanoparticles. Molecules 2017, 22, 1170. [Google Scholar] [CrossRef] [PubMed]
- Valerini, D.; Tammaro, L.; Di Benedetto, F.; Vigliotta, G.; Capodieci, L.; Terzi, R.; Rizzo, A. Aluminum-doped zinc oxide coatings on polylactic acid films for antimicrobial food packaging. Thin Solid Films 2018, 645, 187–192. [Google Scholar] [CrossRef]
- Li, W.H.; Li, L.; Cao, Y.; Lan, T.Q.; Chen, H.Y.; Qin, Y.Y. Effects of PLA Film Incorporated with ZnO Nanoparticle on the Quality Attributes of Fresh-Cut Apple. Nanomaterials 2017, 7, 207. [Google Scholar] [CrossRef] [PubMed]
- Li, M.X.; Zhang, F.; Liu, Z.D.; Guo, X.F.; Wu, Q.; Qiao, L.R. Controlled Release System by Active Gelatin Film Incorporated with beta-Cyclodextrin-Thymol Inclusion Complexes. Food Bioprocess. Technol. 2018, 11, 1695–1702. [Google Scholar] [CrossRef]
- Raquel, B.; Cristina, N.; Filomena, S. Encapsulation Systems for Antimicrobial Food Packaging Components: An Update. Molecules 2020, 25, 1134. [Google Scholar]
- Dou, L.X.; Li, B.F.; Zhang, K.; Chu, X.; Hou, H. Physical properties and antioxidant activity of gelatin-sodium alginate edible films with tea polyphenols. Int. J. Biol. Macromol. 2018, 118, 1377–1383. [Google Scholar] [CrossRef]
- Bonilla, J.; Poloni, T.; Lourenco, R.V.; Sobral, P.J.A. Antioxidant potential of eugenol and ginger essential oils with gelatin/chitosan films. Food Biosci. 2018, 23, 107–114. [Google Scholar] [CrossRef]
- Moreno, O.; Atares, L.; Chiralt, A.; Cruz-Romero, M.C.; Kerry, J. Starch-gelatin antimicrobial packaging materials to extend the shelf life of chicken breast fillets. LWT Food Sci. Technol. 2018, 97, 483–490. [Google Scholar] [CrossRef]
- Liu, Y.Y.; Li, Y.; Deng, L.L.; Zou, L.; Feng, F.Q.; Zhang, H. Hydrophobic Ethylcellulose/Gelatin Nanofibers Containing Zinc Oxide Nanoparticles for Antimicrobial Packaging. J. Agric. Food Chem. 2018, 66, 9498–9506. [Google Scholar] [CrossRef]
- He, Q.Y.; Zhang, Y.C.; Cai, X.X.; Wang, S.Y. Fabrication of gelatin-TiO2 nanocomposite film and its structural, antibacterial and physical properties. Int. J. Biol. Macromol. 2016, 84, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Shukla, A.; Baul, P.P.; Mitra, A.; Halder, D. Biodegradable hybrid nanocomposites of chitosan/gelatin and silver nanoparticles for active food packaging applications. Food Packag. Shelf Life 2018, 16, 178–184. [Google Scholar] [CrossRef]
- Venkatesan, R.; Rajeswari, N.; Tamilselvi, A. Antimicrobial, mechanical, barrier, and thermal properties of bio-based poly (butylene adipate-co-terephthalate) (PBAT)/Ag2O nanocomposite films for packaging application. Polym. Adv. Technol 2018, 29, 61–68. [Google Scholar] [CrossRef]
- Zehetmeyer, G.; Meira, S.M.M.; Scheibel, J.M.; da Silva, C.D.; Rodembusch, F.S.; Brandelli, A.; Soares, R.M.D. Biodegradable and antimicrobial films based on poly(butylene adipate-co-terephthalate) electrospun fibers. Polym. Bull. 2017, 74, 3243–3268. [Google Scholar] [CrossRef]
- Khare, S.; DeLoid, G.M.; Molina, R.M.; Gokulan, K.; Couvillion, S.P.; Bloodsworth, K.J.; Eder, E.K.; Wong, A.R.; Hoyt, D.W.; Bramer, L.M.; et al. Effects of ingested nanocellulose on intestinal microbiota and homeostasis in Wistar Han rats. Nanoimpact 2020, 18. [Google Scholar] [CrossRef] [PubMed]
- DeLoid, G.M.; Cao, X.Q.; Molina, R.M.; Silva, D.I.; Bhattacharya, K.; Ng, K.W.; Loo, S.C.J.; Brain, J.D.; Demokritou, P. Toxicological effects of ingested nanocellulose in in vitro intestinal epithelium and in vivo rat models. Environ. Sci. Nano 2019, 6, 2105–2115. [Google Scholar] [CrossRef]
- Chen, Y.J.; Lin, Y.J.; Nagy, T.; Kong, F.B.; Guo, T.L. Subchronic exposure to cellulose nanofibrils induces nutritional risk by non-specifically reducing the intestinal absorption. Carbohyd. Polym. 2020, 229. [Google Scholar] [CrossRef]
- Dimitrijevic, M.; Karabasil, N.; Boskovic, M.; Teodorovic, V.; Vasilev, D.; Djordjevic, V.; Kilibarda, N.; Cobanovic, N. Safety aspects of nanotechnology applications in food packaging. In Proceedings of the 58th International Meat Industry Conference (Meatcon2015), Mt. Zlatibor, Serbia, 4–7 October 2015; Volume 5, pp. 57–60. [Google Scholar] [CrossRef]
- dos Santos, C.A.; Ingle, A.P.; Rai, M. The emerging role of metallic nanoparticles in food. Appl. Microbiol. Biotechnol. 2020, 104, 2373–2383. [Google Scholar] [CrossRef]
- Doskocz, N.; Zaleska-Radziwill, M.; Affek, K.; Lebkowska, M. Ecotoxicity of selected nanoparticles in relation to micro-organisms in the water ecosystem. Desalin Water Treat. 2020, 186, 50–55. [Google Scholar] [CrossRef]
- Noori, A.; Ngo, A.; Gutierrez, P.; Theberge, S.; White, J.C. Silver nanoparticle detection and accumulation in tomato (Lycopersicon esculentum). J. Nanopart Res. 2020, 22. [Google Scholar] [CrossRef]
- Dash, S.R.; Kundu, C.N. Promising opportunities and potential risk of nanoparticle on the society. IET Nanobiotechnol. 2020, 14, 253–260. [Google Scholar] [CrossRef]
- Garcia, C.V.; Shin, G.H.; Kim, J.T. Metal oxide-based nanocomposites in food packaging: Applications, migration, and regulations. Trends Food Sci. Technol. 2018, 82, 21–31. [Google Scholar] [CrossRef]
- Souza, V.G.L.; Fernando, A.L. Nanoparticles in food packaging: Biodegradability and potential migration to food-A review. Food Packag. Shelf Life 2016, 8, 63–70. [Google Scholar] [CrossRef]
- Istiqola, A.; Syafiuddin, A. A review of silver nanoparticles in food packaging technologies: Regulation, methods, properties, migration, and future challenges. J. Chin. Chem. Soc. 2020. [Google Scholar] [CrossRef]
- Sahoo, R.K. Packaging: Polymer-Metal-Based Micro- and Nanocomposites. Encycl. Polym. Appl. 2019, I–III, 2021–2040. [Google Scholar] [CrossRef]
- Morais, L.D.; Macedo, E.V.; Granjeiro, J.M.; Delgado, I.F. Critical evaluation of migration studies of silver nanoparticles present in food packaging: A systematic review. Crit. Rev. Food Sci. 2019. [Google Scholar] [CrossRef] [PubMed]
- Ramachandran, R.; Krishnaraj, C.; Kumar, V.K.A.; Harper, S.L.; Kalaichelvan, T.P.; Yun, S.I. In vivo toxicity evaluation of biologically synthesized silver nanoparticles and gold nanoparticles on adult zebrafish: A comparative study. 3 Biotech 2018, 8, 441. [Google Scholar] [CrossRef]
- Zhou, Q.F.; Liu, L.H.; Liu, N.A.; He, B.; Hu, L.G.; Wang, L.N. Determination and characterization of metal nanoparticles in clams and oysters. Ecotoxicol. Environ. Saf. 2020, 198. [Google Scholar] [CrossRef]
- Gong, Y.; Chai, M.W.; Ding, H.; Shi, C.; Wang, Y.; Li, R.L. Bioaccumulation and human health risk of shellfish contamination to heavy metals and As in most rapid urbanized Shenzhen, China. Environ. Sci. Pollut. Res. 2020, 27, 2096–2106. [Google Scholar] [CrossRef]
- Shah, N.; Khan, A.; Ali, R.; Marimuthu, K.; Uddin, M.N.; Rizwan, M.; Rahman, K.U.; Alam, M.; Adnan, M.; Jawad, S.M.; et al. Monitoring Bioaccumulation (in Gills and Muscle Tissues), Hematology, and Genotoxic Alteration in Ctenopharyngodon idella Exposed to Selected Heavy Metals. Biomed. Res. Int. 2020, 2020. [Google Scholar] [CrossRef]
- Wu, J.; Wang, G.Y.; Vijver, M.G.; Bosker, T.; Peijnenburg, W.J.G.M. Foliar versus root exposure of AgNPs to lettuce: Phytotoxicity, antioxidant responses and internal translocation. Environ. Pollut. 2020, 261. [Google Scholar] [CrossRef] [PubMed]
- Becaro, A.A.; Siqueira, M.C.; Puti, F.C.; de Moura, M.R.; Correa, D.S.; Marconcini, J.M.; Mattoso, L.H.C.; Ferreira, M.D. Cytotoxic and genotoxic effects of silver nanoparticle/carboxymethyl cellulose on Allium cepa. Environ. Monit. Assess. 2017, 189. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.X.; Liu, H.; Chen, G.C.; Zhao, Q.; Guo, H.Y.; Minocha, R.; Long, S.; Tang, Y.Z.; Saad, E.M.; DeLaTorreRoche, R.; et al. Dual roles of glutathione in silver nanoparticle detoxification and enhancement of nitrogen assimilation in soybean (Glycine max (L.) Merrill). Environ. Sci. Nano 2020, 7, 1954–1966. [Google Scholar] [CrossRef]
- Yang, Z.Z.; Xiao, Y.F.; Jiao, T.T.; Zhang, Y.; Chen, J.; Gao, Y. Effects of Copper Oxide Nanoparticles on the Growth of Rice (Oryza sativa L.) Seedlings and the Relevant Physiological Responses. Int. J. Environ. Res. Public Health 2020, 17, 1260. [Google Scholar] [CrossRef]
- Yusefi-Tanha, E.; Fallah, S.; Rostamnejadi, A.; Pokhrel, L.R. Particle size and concentration dependent toxicity of copper oxide nanoparticles (CuONPs) on seed yield and antioxidant defense system in soil grown soybean (Glycine max cv. Kowsar). Sci. Total Environ. 2020, 715. [Google Scholar] [CrossRef]
- Rajput, V.; Minkina, T.; Sushkova, S.; Behal, A.; Maksimov, A.; Blicharska, E.; Ghazaryan, K.; Movsesyan, H.; Barsova, N. ZnO and CuO nanoparticles: A threat to soil organisms, plants, and human health. Environ. Geochem. Health 2020, 42, 147–158. [Google Scholar] [CrossRef]
- Rajput, V.; Minkina, T.; Fedorenko, A.; Sushkova, S.; Mandzhieva, S.; Lysenko, V.; Duplii, N.; Fedorenko, G.; Dvadnenko, K.; Ghazaryan, K. Toxicity of copper oxide nanoparticles on spring barley (Hordeum sativum distichum). Sci Total Environ. 2018, 645, 1103–1113. [Google Scholar] [CrossRef]
- Baskar, V.; Safia, N.; Preethy, K.S.; Dhivya, S.; Thiruvengadam, M.; Sathishkumar, R. A comparative study of phytotoxic effects of metal oxide (CuO, ZnO and NiO) nanoparticles on in-vitro grown Abelmoschus esculentus. Plant. Biosyst. 2020. [Google Scholar] [CrossRef]
- Jeon, Y.R.; Yu, J.; Choi, S.J. Fate Determination of ZnO in Commercial Foods and Human Intestinal Cells. Int. J. Mol. Sci. 2020, 21, 433. [Google Scholar] [CrossRef]
- Pereira, F.F.; Paris, E.C.; Bresolin, J.D.; Mitsuyuki, M.C.; Ferreira, M.D.; Correa, D.S. The Effect of ZnO Nanoparticles Morphology on the Toxicity Towards Microalgae Pseudokirchneriella subcapitata. J. Nanosci. Nanotechnol. 2020, 20, 48–63. [Google Scholar] [CrossRef]
- Vasile, O.R.; Serdaru, I.; Andronescu, E.; Trusca, R.; Surdu, V.A.; Oprea, O.; Ilie, A.; Vasile, B.S. Influence of the size and the morphology of ZnO nanoparticles on cell viability. CR Chim. 2015, 18, 1335–1343. [Google Scholar] [CrossRef]
- Voss, L.; Saloga, P.E.J.; Stock, V.; Bohmert, L.; Braeuning, A.; Thunemann, A.F.; Lampen, A.; Sieg, H. Environmental Impact of ZnO Nanoparticles Evaluated by in Vitro Simulated Digestion. ACS Appl. Nano Mater. 2020, 3, 724–733. [Google Scholar] [CrossRef]
- Meng, J.; Zhou, X.L.; Yang, J.; Qu, X.J.; Cui, S.X. Exposure to low dose ZnO nanoparticles induces hyperproliferation and malignant transformation through activating the CXCR2/NF-kappa B/STAT3/ERK and AKT pathways in colonic mucosal cells. Environ. Pollut. 2020, 263. [Google Scholar] [CrossRef] [PubMed]
- Musial, J.; Krakowiak, R.; Mlynarczyk, D.T.; Goslinski, T.; Stanisz, B.J. Titanium Dioxide Nanoparticles in Food and Personal Care Products-What Do We Know about Their Safety? Nanomaterials 2020, 10, 1110. [Google Scholar] [CrossRef] [PubMed]
- Kurtz, C.C.; Mitchell, S.; Nielsen, K.; Crawford, K.D.; Mueller-Spitz, S.R. Acute high-dose titanium dioxide nanoparticle exposure alters gastrointestinal homeostasis in mice. J. Appl. Toxicol. 2020, 40, 1384–1395. [Google Scholar] [CrossRef] [PubMed]
- Hashem, M.M.; Abo-El-Sooud, K.; Abd-Elhakim, Y.M.; Badr, Y.A.H.; El-Metwally, A.E.; Bahy-El-Diena, A. The long-term oral exposure to titanium dioxide impaired immune functions and triggered cytotoxic and genotoxic impacts in rats. J. Trace Elem. Med. Biol. 2020, 60. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.Q.; Han, Y.H.; Gu, M.; Du, H.J.; Song, M.Y.; Zhu, X.A.; Ma, G.X.; Pan, C.; Wang, W.C.; Zhao, E.M.; et al. Foodborne Titanium Dioxide Nanoparticles Induce Stronger Adverse Effects in Obese Mice than Non-Obese Mice: Gut Microbiota Dysbiosis, Colonic Inflammation, and Proteome Alterations. Small 2020. [Google Scholar] [CrossRef]
- Bettencourt, A.; Goncalves, L.M.; Gramacho, A.C.; Vieira, A.; Rolo, D.; Martins, C.; Assuncao, R.; Alvito, P.; Silva, M.J.; Louro, H. Analysis of the Characteristics and Cytotoxicity of Titanium Dioxide Nanomaterials Following Simulated In Vitro Digestion. Nanomaterials 2020, 10, 1516. [Google Scholar] [CrossRef]
- Najnin, H.; Alam, N.; Mujeeb, M.; Ahsan, H.; Siddiqui, W.A. Biochemical and toxicological analysis of Cinnamomum tamala essential oil in Wistar rats. J. Food Process. Perservation 2020, 44. [Google Scholar] [CrossRef]
- Costa, W.K.; de Oliveira, J.R.S.; de Oliveira, A.M.; Santos, I.B.D.; da Cunha, R.X.; de Freitas, A.F.S.; da Silva, J.W.L.M.; Silva, V.B.G.; de Aguiar, J.C.R.D.F.; da Silva, A.G.; et al. Essential oil from Eugenia stipitata McVaugh leaves has antinociceptive, anti-inflammatory and antipyretic activities without showing toxicity in mice. Ind. Crops Prod. 2020, 144. [Google Scholar] [CrossRef]
- Bonin, E.; Carvalho, V.M.; Avila, V.D.; dos Santos, N.C.A.; Benassi-Zanqueta, E.; Lancheros, C.A.C.; Previdelli, I.T.S.; Ueda-Nakamura, T.; de Abreu, B.A.; do Prado, I.N. Baccharis dracunculifolia: Chemical constituents, cytotoxicity and antimicrobial activity. LWT Food Sci. Technol. 2020, 120. [Google Scholar] [CrossRef]
- Abou Baker, D.H.; Al-Moghazy, M.; ElSayed, A.A.A. The in vitro cytotoxicity, antioxidant and antibacterial potential of Satureja hortensis L. essential oil cultivated in Egypt. Bioorg. Chem. 2020, 95. [Google Scholar] [CrossRef] [PubMed]
| Film Forming Class | Substance | Reference |
|---|---|---|
| Polysaccharides | Cellulose | [110,111] |
| Chitosan | [112,113] | |
| Starch | [114,115] | |
| Hemicellulose | [116,117] | |
| Alginate | [118,119] | |
| Agarose | [120,121] | |
| Glucomannan | [122,123] | |
| Pullulan | [124,125] | |
| Proteins | Soy-protein | [126,127] |
| Whey-protein | [128,129] | |
| Zein | [126,130] | |
| Collagen | [131,132] | |
| Gelatine | [133,134] | |
| Casein | [135,136] | |
| Lipids and waxes | Plant oil and animal fat | [137,138] |
| Waxes | [139,140] | |
| Others | PLA | [141,142] |
| PVA | [143,144] |
| Product Preserved | Packaging Material | Antimicrobial Agent | Reference |
|---|---|---|---|
| Iceberg lettuce | Cellulose | Clove and oregano oils | [91] |
| Cucumber | Chitosan | Limonene | [153] |
| Tomato | Chitosan | TiO2 nanoparticles | [154] |
| Strawberries | Chitosan/CMC | Chitosan/citric acid | [155] |
| Strawberries | PLA | AgNPs | [150] |
| Strawberries | Chitosan/CMC | Mentha spicata oil | [156] |
| Strawberries | Gelatin | Butylated hydroxyanisole | [157] |
| Fish | PLA | Thymol | [158] |
| Crap fillets | Alginate/CMC | Ziziphora clinopodioides oil / ZnO | [159] |
| Rainbow trout fillet | Chitosan | Grape seed extract | [160] |
| Salmon | PLA | Glycerol monolaurate | [161] |
| Shrimps | Chitosan | Carvacrol | [162] |
| Shrimps | Gelatin | ZnO /clove oil | [163] |
| Chicken | Chitosan | Acerola residue extract | [164] |
| Poultry | Chitosan | Ginger oil | [165,166] |
| Chicken | Gelatin | Thyme oil | [167] |
| Chicken | Pullulan | Nisin | [125] |
| Chicken | Chitosan/PET | Plantaricin | [168] |
| Ostrich meat | Kefiran/polyurethane | Zataria multiflora oil | [169] |
| Lamb meat | Chitosan | Satureja plant oil | [170] |
| Ground beef | PLA/NC | Mentha piperita, Bunium percicum | [171] |
| Ham | Chitosan/starch | Gallic acid | [172] |
| Salami | Whey protein | Cinnamomum cassia, Rosmarinus officinalis oils | [173] |
| Cheese | Chitosan/PVA | TiO2 | [174] |
| Cheese | Cellulose /Chitosan | Monolaurin | [175] |
| Cheese | Starch | Clove leaf oil | [176] |
| Cheese | Agar | Enterocin | [177] |
| Cheese | Zein | Pomegranate peel extract | [178] |
| Peanuts (roasted) | Banana flour (starch) | Garlic essential oil | [179] |
| Food Type | Essential Oil / Extract | References |
|---|---|---|
| Meat | Acerola extract, oregano oil, ginger oil, betel oil, mentha spicata oil, rosemarin oil, ziziphora clinopodioides oil, zataria multiflora oil, satureja plant oil, thyme oil, grape seed extract, eucalyptus oil | [151,159,160,162,164,165,166,167,169,170,171,173,200,201,202] |
| Vegetables and fruits | Litsea cubeba oil, clove and oregano oils, mentha spicata, cinnamon oil, limonene, thyme oil | [91,114,153,156,178,199,203,204] |
| Pastry and bread | Garlic essential oil, pomegranate peel extract, cinnamon oil, clove oil | [178,179,205] |
| Dairy and cheese | Clove leaf oil, ginger oil | [107,126,176] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Motelica, L.; Ficai, D.; Ficai, A.; Oprea, O.C.; Kaya, D.A.; Andronescu, E. Biodegradable Antimicrobial Food Packaging: Trends and Perspectives. Foods 2020, 9, 1438. https://doi.org/10.3390/foods9101438
Motelica L, Ficai D, Ficai A, Oprea OC, Kaya DA, Andronescu E. Biodegradable Antimicrobial Food Packaging: Trends and Perspectives. Foods. 2020; 9(10):1438. https://doi.org/10.3390/foods9101438
Chicago/Turabian StyleMotelica, Ludmila, Denisa Ficai, Anton Ficai, Ovidiu Cristian Oprea, Durmuş Alpaslan Kaya, and Ecaterina Andronescu. 2020. "Biodegradable Antimicrobial Food Packaging: Trends and Perspectives" Foods 9, no. 10: 1438. https://doi.org/10.3390/foods9101438
APA StyleMotelica, L., Ficai, D., Ficai, A., Oprea, O. C., Kaya, D. A., & Andronescu, E. (2020). Biodegradable Antimicrobial Food Packaging: Trends and Perspectives. Foods, 9(10), 1438. https://doi.org/10.3390/foods9101438