Determination of Phenolic Compounds, Procyanidins, and Antioxidant Activity in Processed Coffea arabica L. Leaves
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Processing and Extraction of Coffee Leaves
2.3. HPLC Analysis of Bioactive Compounds of Coffee Leaves
2.4. Liquid Chromatography Mass Spectrometer (LC-MS)
2.5. Measurement of DPPH Radical Scavenging Activity
2.6. Measurement of Total Phenolic Content
2.7. Measurement of Total Procyanidins
2.8. Statistical Analysis
3. Results
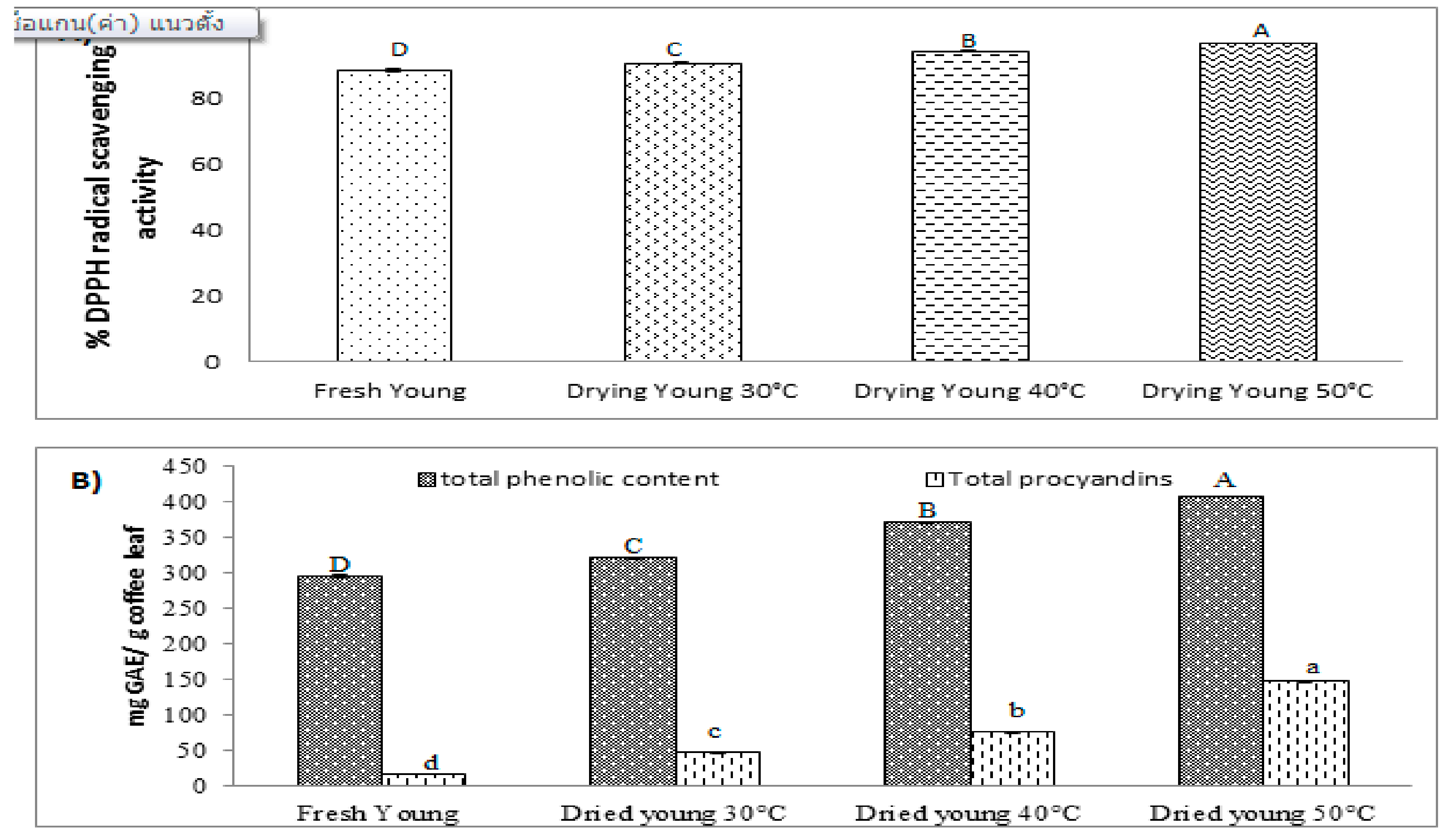
3.1. Effect of Drying Process on DPPH Radical Scavenging Activity
3.2. Effect of Drying Process on Total Phenolic Content
3.3. Effect of Drying Process on Total Procyanidins
3.4. Effect of Different Drying Temperatures on DPPH Radical Scavenging Activity, Total Phenolic Content, and Total Procyanidins
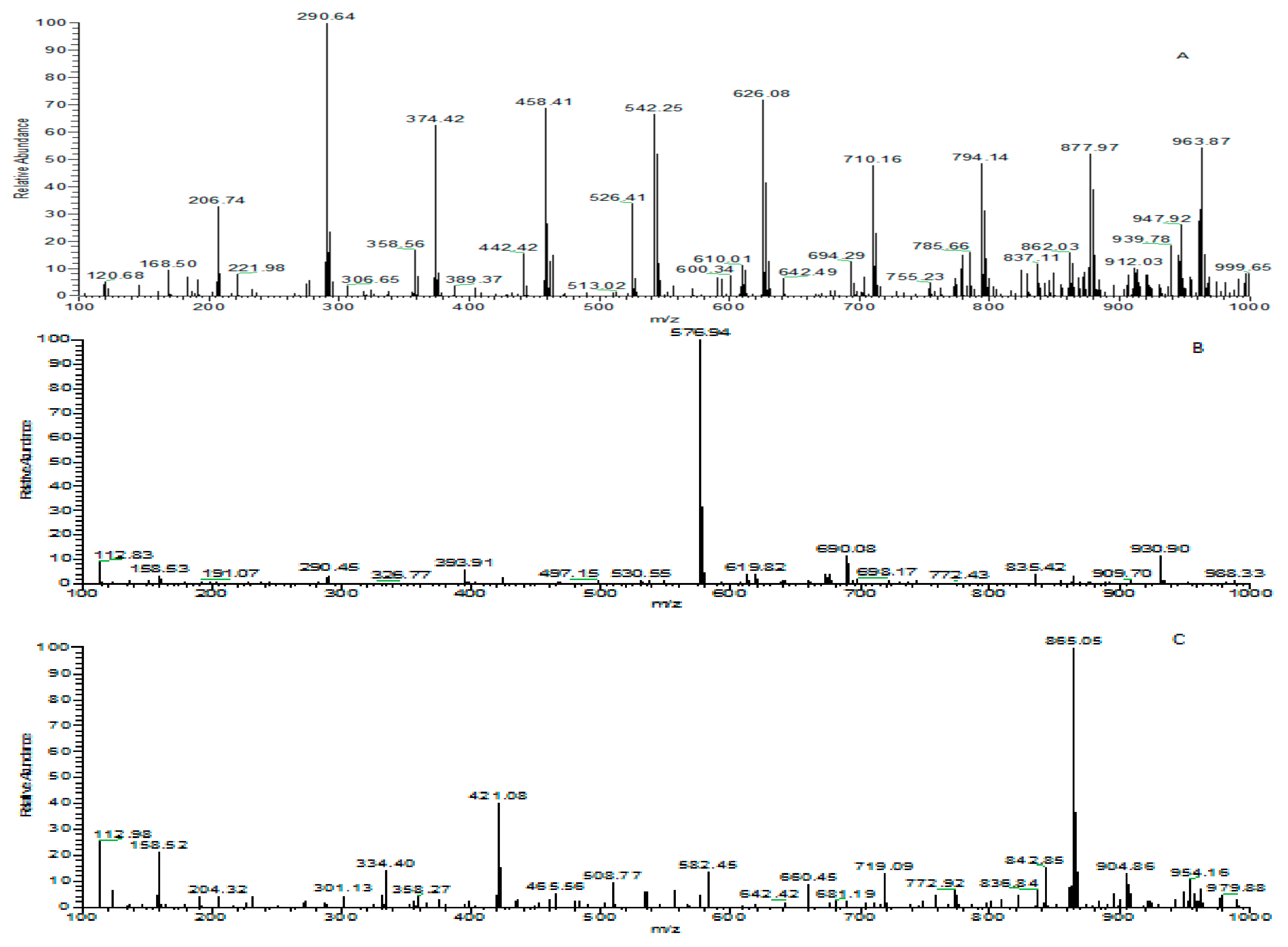
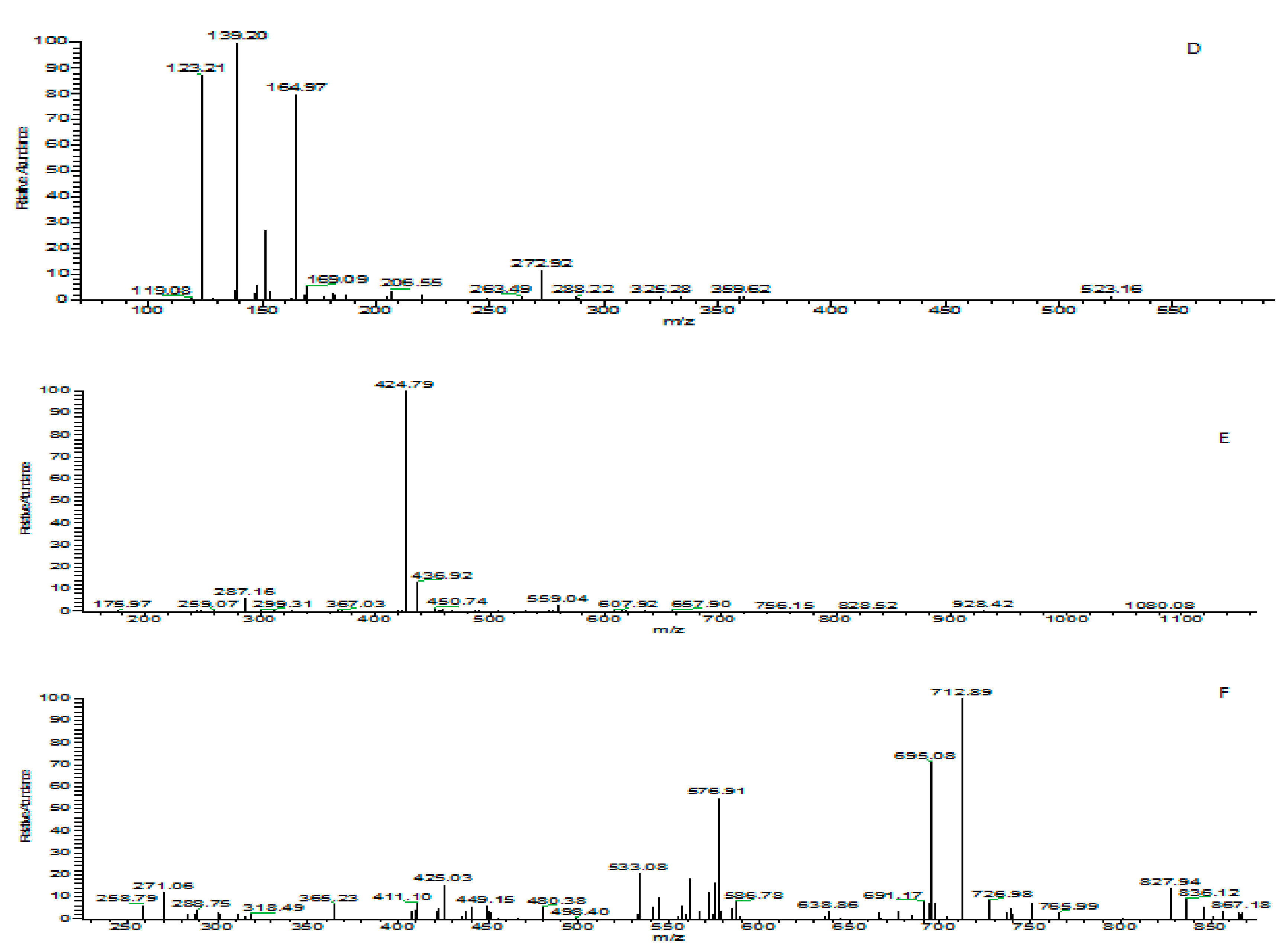
3.5. Identification of Phenolic Compounds in Leaf Extracts of Coffea arabica L.
3.6. Effect of Drying Process on Phenolic Compounds in Coffea arabica L.
3.7. Effect of Different Drying Temperatures on Phenolic Compounds in Coffea arabica L.
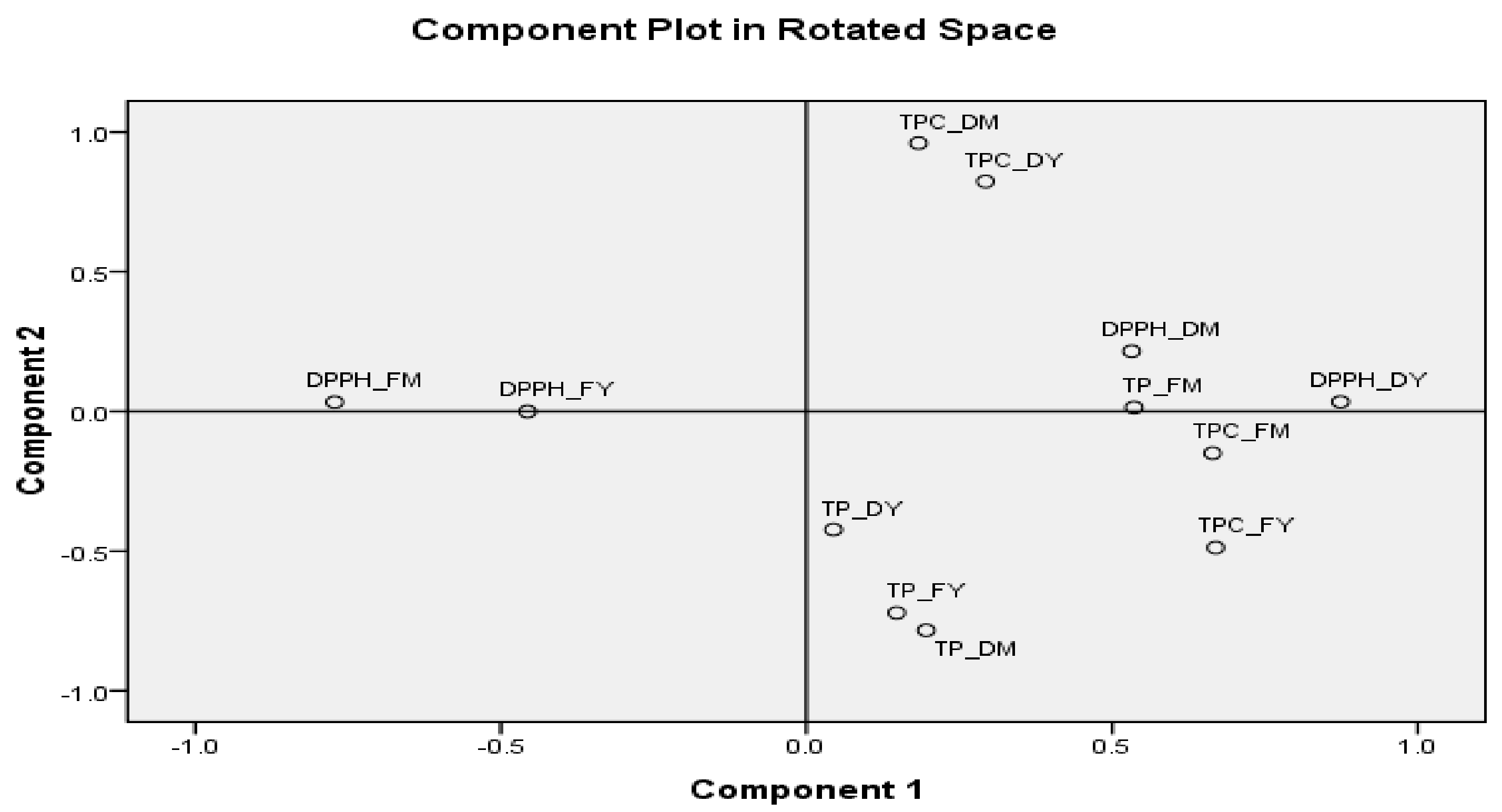
3.8. Correlation of Drying Process on Antioxidant Activity, Total Phenolic Content, and Total Procyanidins
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Campa, C.; Mondolot, L.; Rakotondravao, A.; Bidel, L.P.; Gargadennec, A.; Couturon, E.; La Fisca, P.; Rakotomalala, J.J.; Jay-Allemand, C.; Davis, A.P. A survey if mangiferin and hydroxycinnamic acid ester accumulation in coffee (coffea) leaves biological implications and uses. Ann. Bot. 2012, 110, 595–613. [Google Scholar] [CrossRef] [PubMed]
- Campa, C.; Petivallet, A. Achieving Sustainable Cultivation of Coffee. In Beneficial Compounds from Coffee Leaves; Lashermes, P., Ed.; Burleigh Science Publishing: Sawston, UK, 2017. [Google Scholar]
- Almeida, R.F.; Ulrich, C.M.; Owen, R.W.; Trevisan, M.T.S. Content and solvent extraction in coffee leaves from Brazil. Assoc. Sci. Inf. Coffee Conf. 2014, 42–45. [Google Scholar]
- Júnior, A.P.D.; Shimizu, M.M.; Moura, J.C.M.S.; Catharino, R.R.; Ramos, R.A.; Ribeiro, R.V.; Mazzafera, P. Looking for the physiological role of anthocyanins in the leaves of Coffea arabica. Photochem. Photobiol. 2012, 88, 928–937. [Google Scholar] [CrossRef] [PubMed]
- Patay, E.B.; Nemeth, T.; Nemeth, T.S.; Filep, R.; Vlase, L.; Papp, N. Histological and phytochemical studies of Coffea benghalensis Roxb ex. Schult compared with Coffea arabica L. Farmacia 2016, 64, 125–130. [Google Scholar]
- Ross, I.A. Medicinal Plant of The World; Humana Press: Totowa, NJ, USA, 2005. [Google Scholar]
- Rodríguez-Gómez, R.; Vanheuverzwjin, J.; Souard, F.; Delporte, C.; Stevigny, C.; Stoffelen, P.; De Braekeleer, K.; Kauffmann, J.-M. Determination of three main chlorogenic acids in water extracts of coffee leaves by liquid chromatography coupled to an electrochemical detector. Antioxidants 2018, 7, 143. [Google Scholar] [CrossRef] [PubMed]
- Souard, F.; Delporte, C.; Stoffelen, P.; Thévenot, E.A.; Noret, N.; Dauvergne, B.; Kauffmann, J.-M.; Van Antwerpen, P.; Stévigny, C. Metabolomics fingerprint of coffee species determined by untargeted-profiling study using LC-HRMS. Food Chem. 2018, 245, 603–612. [Google Scholar] [CrossRef] [PubMed]
- Ariga, T.; Hamano, M. Radical scavenging action and its mode in procyanidins B-1 and B-3 from azuki beans to peroxyl radicals. Agric. Boil. Chem. 1990, 54, 2499–2504. [Google Scholar] [CrossRef]
- Arpentine, G.; Femandez, Y.; Bourzeix, M.; Mitjavila, S. Relation between the structure of a series of procyanidins and their radical superoxide scavenger capacity. Proc. Group Polyphen. 1992, 16, 237–240. [Google Scholar]
- Virgili, F.; Kobuchi, H.; Packe, L. Procyanidins extracted from pins maritime (pycnogenol) scavenger of free radical species and modulators of nitrogen monoxide metabolism in activated murine RAW 264.7 macrophages. Free Radic. Biol. Med. 1998, 24, 1120–1129. [Google Scholar] [CrossRef]
- Zhou, H.-C.; Tam, N.F.-Y.; Lin, Y.-M.; Ding, Z.-H.; Chai, W.-M.; Wei, S.-D. Relationships between degree of polymerization and antioxidant activities: A study on proanthocyanidins from the leaves of a medicinal mangrove plant ceriops tagal. PLoS ONE 2014, 9, e107606. [Google Scholar] [CrossRef]
- Chen, X.-X.; Liang, G.; Chai, W.-M.; Feng, H.-L.; Zhou, H.-T.; Shi, Y.; Chen, Q.-X. Antioxidant and antityrosinase proanthocyanidins from Polyalthia longifolia leaves. J. Biosci. Bioeng. 2014, 118, 583–587. [Google Scholar] [CrossRef] [PubMed]
- Rabeta, M.S.; Lai, S.Y. Effect of Drying, Fermented and unfermented tea of Ocimum tenuiflorum Linn. on the antioxidant capacity. Int. Food Res. J. 2013, 20, 1601–1608. [Google Scholar]
- Aziah, A.N.; Komathi, C. Physicochemical and functional properties of peeled and unpeeled pumpkin flour. J. Food Sci. 2009, 74, S328–S333. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.-M.; Ma, Z.; Kitts, D.D. Effects of processing method and age of leaves on phytochemical profiles and bioactivity of coffee leaves. Food Chem. 2018, 249, 143–153. [Google Scholar] [CrossRef]
- Yamassaki, F.T.; Campestrini, L.H.; Zawadzki-Baggio, S.F.; Maurer, J.B.B. Avocado leaves: Influence of drying process, thermal incubation, and storage conditions on preservation of polyphenolic compounds and antioxidant activity. Int. J. Food Prop. 2017, 20, 1–14. [Google Scholar] [CrossRef]
- Thiesen, L.C.; Block, L.C.; Zonta, S.L.; Bittencourt, C.M.D.S.; Ferreira, R.A.; Filho, V.C.; Couto, A.G.; Bersolin, T.M.B. Simultaneous determination of epicatechin and procyanidin A2 marker in litch chinensis by high-performance liquid chromatography. Rev. Bras. Farmacogn. 2016, 26, 168–173. [Google Scholar] [CrossRef][Green Version]
- Chao, J.J.; Lo, D.; Pamboon, N.; Wu, M.C. In vitro antioxidant capacity and antiproliferative effects of different parts and extractions from Ajuga bracteosa on HepG2 cell line. Int. Res. J. Pharm. Pharmacol. 2012, 2, 110–117. [Google Scholar]
- Miliauskas, G.; Venskutonis, P.; Van Beek, T. Screening of radical scavenging activity of some medicinal and aromatic plant extracts. Food Chem. 2004, 85, 231–237. [Google Scholar] [CrossRef]
- Sun, B.; Ricardo-Da-Silva, J.M.; Spranger, I. Critical factors of vanillin assay for catechins and proanthocyanidins. J. Agric. Food Chem. 1998, 46, 4267–4274. [Google Scholar] [CrossRef]
- Taufik, Y.; Widiantara, T.; Garnida, Y. The effect of drying temperature on the antioxidant activity of black mulberry leaf tea (Morus nigra). Rasayan J. Chem. 2016, 9, 889–895. [Google Scholar]
- Qa’Dan, F.; Mansoor, K.; Al-Adham, I.; Schmidt, M.; Nahrstedt, A. Proanthocyanidins from ginkgo biloba leaf extract and their radical scavenging activity. Pharm. Boil. 2011, 49, 471–476. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Genovese, M.; Pinto, M.D.S.; Gonçalves, A.D.S.S.; Lajolo, F. Bioactive compounds and antioxidant capacity of exotic fruits and commercial frozen pulps from Brazil. Food Sci. Technol. Int. 2008, 14, 207–214. [Google Scholar] [CrossRef]
- Hlanla, L.N. Effect of Fermentation Temperature and Duration on Chemical Composition of Bush Tea (Athrixia phylicoides DC.). Master’s Thesis, University of Limpopo, Turfloop Campus, Mankweng, South Africa, 2010. [Google Scholar]
- Del Baño, M.J.; Lorente, J.; Castillo, J.; Benavente-García, O.; Marín, M.P.; Del Río, J.A.; Ibarra, L. Flavonoid distribution during the development of leaves, flowers stems and roots of rosmarinus officinalis postulation of a biosythetic pathway. J. Agric. Food Chem. 2004, 52, 4987–4992. [Google Scholar] [CrossRef] [PubMed]
- Chew, Y.L.; Goh, J.K.; Lim, Y.Y. Assessment of in vitro antioxidant capacity and polyphenolic composition of selected medicinal herb from leguminaosae family in peninsular Malaysia. Food Chem. 2009, 116, 13–18. [Google Scholar] [CrossRef]
- Bubueanu, C.; Pavaloiu, R.; Pirvu, L. HPLC profiles and antioxidant activities from leaves to green and roasted beans of Coffea arabica. Malays. J. Med Biol. 2016, 3, 31–36. [Google Scholar]
- Agrawal, P.K. Carbon-13-NMR of Flavonoids; Elsevier: New York, NY, USA, 1989; pp. 135–155. [Google Scholar]
- Chang, C.H.; Lin, H.Y.; Chang, C.Y.; Liu, Y.C. Comparisons on the antioxidant properties of fresh, freeze-dried and hot-air-dried tomatoes. Food Eng. 2005, 77, 478–485. [Google Scholar] [CrossRef]
- Bhakta, D.; Ganjewala, D. Effect of Leaf Positions on total phenolics, flavonoids and proanthocyanidins content and antioxidant activities in Lantana camara (L). J. Sci. Res. 2009, 1, 363–369. [Google Scholar] [CrossRef]
- Hadadi, S.A.; Li, H.; Rafie, R.; Kaseloo, P.; Witiak, S.M.; Siddiqui, R.A. Anti-oxidation properties of leaves, skin, pulp, and seeds extracts from green papaya and their anti-cancer activities in breast cancer cells. J. Cancer Metastasis Treat. 2018, 4, 25. [Google Scholar] [CrossRef][Green Version]
- Rebaya, A.; Belghith, S.I.; Cherif, J.K.; Trabelsi-Ayadi, M. Total phenolic compounds and antioxidant potential of rokrose (Cistus salviifolius) leaves and flowers grown in Tunisia. Int. J. Pharmacogn. Phytochem. Res. 2016, 8, 327–331. [Google Scholar]
- Tahirovic, A.; Basic, N. Phenolic content and antioxidant capacity of Viscum alaum leaves and twigs from various host tress. Work. Fac. For. Univ. Sarajevo 2017, 2, 93–104. [Google Scholar]
- Kerkhofs, N.; Lister, C.; Savage, G. Change in colour and anti-oxidant content of tomato cultivars following forced-air drying. Plant Foods Hum. Nutr. 2005, 60, 117–121. [Google Scholar] [CrossRef] [PubMed]
- Dorta, E.; Loba, M.G.; González, G. Using drying treatment to stabilize mango peel and seed effect on antioxidant activity. LWT Foods Sci. Technol. 2012, 45, 261–268. [Google Scholar] [CrossRef]
- Navarro-Hoyos, M.; Lebrón-Aguilar, R.; Quintanilla-López, J.E.; Cueva, C.; Hevia, D.; Quesada, S.; Azofeifa, G.; Moreno-Arribas, M.V.; Monagas, M.; Bartolomé, B. Proanthocyanidin characterization and bioactivity of extracts from different parts of Uncaria tomentosa L. (cat’s claw). Antioxidant (Basel) 2017, 6, 12. [Google Scholar] [CrossRef] [PubMed]
- Ramsay, A.; Mueller-Harvey, I. Proanthocyanidins from Averrha bilimbi fruits and leaves. J. Food Compos. Anal. 2016, 47, 16–20. [Google Scholar] [CrossRef]
- Hilton, P.J.; Palmer-Jones, R.; Palmer-Jones, R. Relationship between the flavanol composition of fresh tea shoots and the theaflavin content of manufactured tea. J. Sci. Food Agric. 1973, 24, 813–818. [Google Scholar] [CrossRef]
- Katsube, T.; Tsurunaga, Y.; Sugiyama, M.; Furuno, T.; Yamasaki, Y. Effect of air-drying temperature on antioxidant capacity and stability of polyphenolic compounds in mulberry (Morus alba L.) leaves. Food Chem. 2009, 113, 964–969. [Google Scholar] [CrossRef]
- Tiho, T.; Yao, N.J.C.; Brou, Y.C.; Adima, A.A. Drying temperature effect in total phenols and flavonoids content and antioxidant activity of Borassus aethiopum mart ripe fruits. J. Food Res. 2017, 6, 50–64. [Google Scholar] [CrossRef]
- Ciulu, M.; Quirantes-Piné, R.; Spano, N.; Sanna, G.; Borrás-Linares, I.; Segura-Carretero, A. Evaluation of new extraction approaches to obtain phenolic compound-rich extracts from Stevia rebaudiana Bertoni leaves. Ind. Crop. Prod. 2017, 108, 106–112. [Google Scholar] [CrossRef]
- Figueroa, J.G.; Borrás-Linares, I.; Lozano-Sánchez, J.; Quirantes-Piné, R.; Segura-Carretero, A.; Borrás-Linare, I.; Lozano-Sánchez, J.; Quirantes-Piné, R.; Segura-Carretero, A. Optimization of drying process and pressurized liquid extraction for recovery of bioactive compounds from avocado peel by-product. ELECTROPHORESIS 2018, 39, 1908–1916. [Google Scholar] [CrossRef]
- Murakami, I.; Nakamura, T.; Ishibashi, Y.; Shibuya, R.; Ayano, E.; Morita-Murase, Y.; Nagata, Y.; Kanazawa, H. Simultaneous Determination of catechins and procyanidins in bottles tea drink by LC-MS. Chromatography 2006, 83, 56–63. [Google Scholar]
- Fraser, K.; Harrison, S.J.; Lane, G.A.; Otter, D.E.; Hemar, Y.; Quek, S.-Y.; Rasmussen, S. HPLC–MS/MS profiling of proanthocyanidins in teas: A comparative study. J. Food Compos. Anal. 2012, 26, 43–51. [Google Scholar] [CrossRef]
- Falleh, H.; Oueslati, S.; Guyot, S.; Dali, A.B.; Abdelly, C.; Riadh, K. LC/ESI-MS/MS characterization of procyanidins and propelargonidins responible for the strong antioxidant activity of the edible halophyte Mesembryanthemun edule L. Food Chem. 2011, 127, 1732–1738. [Google Scholar] [CrossRef]
- Lin, L.-Z.; Sun, J.; Chen, P.; Monagas, M.J.; Harnly, J.M. UHPLC-PDA-ESI/HRMSn profiling method to identify and quantify oligomeric proanthocyanidins in plant products. J. Agric. Food Chem. 2014, 62, 9387–9400. [Google Scholar] [CrossRef]

| Samples | %DPPH Radical Scavenging Activity |
|---|---|
| Butyl hydroxyl anisole (BHA) | 91.04 ± 0.84 E |
| Fresh Young | 92.93 ± 0.51 C |
| Fresh Mature | 92.24 ± 0.95 D |
| Dried Young 40 °C, 8 h | 95.01 ± 0.44 A |
| Dried Mature 40 °C, 8 h | 93.40 ± 0.70 B |
| Compound Number | Compound | Molecular Formula | RT (min) | [M-H]− (m/z) | FY (mg/g) | FM (mg/g) | DY40 (mg/g) | DM40 (mg/g) |
|---|---|---|---|---|---|---|---|---|
| 1 | Catechin or Epicatechin | C15H14O6 | 2.17 | 289 | 11.24 ± 0.02 a | 11.21 ± 0.04 a | 11.23 ± 0.06 a | 11.23 ± 0.03 a |
| 2 | Mangiferin or Isomangiferin | C19H18O11 | 3.33 | 421 | 20.34 ± 0.06 c | 19.03 ± 0.07 d | 49.83 ± 0.01 a | 42.46 ± 0.05 b |
| 3 | Procyanidin B | C30H26O12 | 5.92 | 577 | 17.40 ± 0.05 c | 12.49 ± 0.02 d | 25.40 ± 0.04 a | 18.41 ± 0.07 b |
| 4 | Caffeoylquinic acids (CQA) | C16H18O9 | 8.37 | 353 | 14.00 ± 0.10 c | 12.01 ± 0.11 d | 19.30 ± 0.02 a | 15.17 ± 0.03 b |
| 5 | Caffeine | C8H10N4O2 | 9.43 | 193 | 79.79 ± 0.02 d | 80.65 ± 0.03 c | 320.08 ± 0.07 a | 259.31 ± 0.07 b |
| 6 | Quercetin-3-O-glucoside | C21H19O12 | 12.36 | 463 | 16.34 ± 0.15 c | 11.99 ± 0.18 d | 22.65 ± 0.09 a | 17.27 ± 0.02 b |
| 7 | Procyanidin C | C45H38O18 | 12.94 | 865 | 88.18 ± 0.06 c | 88.18 ± 0.08 c | 121.76 ± 0.02 b | 148.02 ± 0.05 a |
| 8 | Rutin (isomer1 or isomer2) | C27H30O16 | 18.11 | 609 | 23.76 ± 0.01 a | 12.34 ± 0.03 c | 24.24 ± 0.05 a | 14.84 ± 0.01 b |
| 9 | 3,4-Dicaffeoylquinic acid (3,4-diCQA) | C25H24O12 | 20.01 | 515 | 11.37 ± 0.09 b | 11.39 ± 0.05 b | 12.00 ± 0.06 a | 11.39 ± 0.07 b |
| Compound Number | Compound | FY (mg/g) | DY30 (mg/g) | DY40 (mg/g) | DY50 (mg/g) |
|---|---|---|---|---|---|
| 1 | Catechin or Epicatechin | 11.24 ± 0.02 a | 11.21 ± 0.08 a | 11.23 ± 0.03 a | 11.25 ± 0.04 a |
| 2 | Mangiferin or Isomangiferin | 20.34 ± 0.06 d | 39.26 ± 0.05 c | 86.09 ± 0.02 b | 108.78 ± 0.04 a |
| 3 | Procyanidin B | 17.40 ± 0.05 d | 29.86 ± 0.01 c | 41.16 ± 0.07 b | 57.70 ± 0.05 a |
| 4 | Caffeoylquinic acids (CQA) | 14.00 ± 0.10 d | 15.31 ± 0.10 c | 18.17 ± 0.08 b | 20.23 ± 0.02 a |
| 5 | Caffeine | 79.79 ± 0.02 d | 160.83 ± 0.03 c | 301.66 ± 0.07 b | 361.93 ± 0.06 a |
| 6 | Quercetin-3-O-glucoside | 16.34 ± 0.15 c | 16.56 ± 0.04 c | 20.13 ± 0.02 b | 25.94 ± 0.05 a |
| 7 | Procyanidin C | 88.18 ± 0.06 d | 115.26 ± 0.06 c | 178.16 ± 0.01 b | 351.07 ± 0.08 a |
| 8 | Rutin (isomer1 or isomer2) | 23.76 ± 0.01 d | 25.15 ± 0.03 c | 28.69 ± 0.07 b | 39.75 ± 0.04 a |
| 9 | 3,4-Dicaffeoylquinic acid (3,4-diCQA) | 11.37 ± 0.09 c | 16.01 ± 0.09 b | 16.35 ± 0.03 b | 18.82 ± 0.05 a |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ngamsuk, S.; Huang, T.-C.; Hsu, J.-L. Determination of Phenolic Compounds, Procyanidins, and Antioxidant Activity in Processed Coffea arabica L. Leaves. Foods 2019, 8, 389. https://doi.org/10.3390/foods8090389
Ngamsuk S, Huang T-C, Hsu J-L. Determination of Phenolic Compounds, Procyanidins, and Antioxidant Activity in Processed Coffea arabica L. Leaves. Foods. 2019; 8(9):389. https://doi.org/10.3390/foods8090389
Chicago/Turabian StyleNgamsuk, Samuchaya, Tzou-Chi Huang, and Jue-Liang Hsu. 2019. "Determination of Phenolic Compounds, Procyanidins, and Antioxidant Activity in Processed Coffea arabica L. Leaves" Foods 8, no. 9: 389. https://doi.org/10.3390/foods8090389
APA StyleNgamsuk, S., Huang, T.-C., & Hsu, J.-L. (2019). Determination of Phenolic Compounds, Procyanidins, and Antioxidant Activity in Processed Coffea arabica L. Leaves. Foods, 8(9), 389. https://doi.org/10.3390/foods8090389






