Garlic (Allium sativum L.): A Brief Review of Its Antigenotoxic Effects
Abstract
:1. Introduction
2. Garlic
2.1. Overview
2.2. Antigenotoxic Studies of Garlic
3. Perspectives and Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Mendoza-Pérez, J.A.; Fregoso-Aguilar, T.A. Chemistry of natural antioxidants and studies performed with different plants collected in Mexico. In Oxidative Stress and Chronic Degenerative Diseases—A Role for Antioxidants; Morales-González, J.A., Ed.; InTech: Rijeka, Croatia, 2013; pp. 59–85. [Google Scholar]
- Barranco-Pedraza, L.M.; Batista-Hernández, I.L. Social contribution of traditional and Natural Medicine in the Cuban public health. Rev. Humanid. Médicas 2013, 13, 713–727. [Google Scholar]
- López-Romero, D.; Izquierdo-Vega, J.A.; Morales-González, J.A.; Madrigal-Bujaidar, E.; Chamorro-Cevallos, G.; Sánchez-Gutiérrez, M.; Betanzos-Cabrera, G.; Álvarez-González, I.; Morales-González, Á.; Madrigal-Santillán, E. Evidence of Some Natural Products with Antigenotoxic Effects. Part 2: Plants, Vegetables, and Natural Resin. Nutrients 2018, 10, 1954. [Google Scholar] [CrossRef] [PubMed]
- Madrigal-Santillán, E.; Madrigal-Bujaidar, E.; Cruz-Jaime, S.; Valadez-Vega, M.C.; Sumaya-Martínez, M.T.; Pérez-Ávila, K.G.; Morales-González, J.A. The Chemoprevention of Chronic Degenerative Disease Through Dietary Antioxidants: Progress, Promise and Evidences. In Oxidative Stress and Chronic Degenerative Diseases—A Role for Antioxidants; Morales-González, J.A., Ed.; InTech: Rijeka, Croatia, 2013; pp. 155–185. [Google Scholar]
- Ferguson, L.R.; Philpott, M.; Karunasinghe, N. Dietary cancer and prevention using antimutagens. Toxicology 2004, 198, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Nagarathna, P.; Johnson Wesley, M.; Sriram Reddy, P.; Reena, K. Review on genotoxicity, its molecular mechanisms and prevention. Int. J. Pharm. Sci. Rev. Res. 2013, 22, 236–243. [Google Scholar]
- Słoczynska, K.; Powroznik, B.; Pekala, E.; Waszkielewicz, A.M. Antimutagenic compounds and their possible mechanisms of action. J. Appl. Genet. 2014, 55, 273–285. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Allium sativum. Available online: http://www.kew.org/science-conservation/plants-fungi/allium-sativum-garlic (accessed on 30 May 2019).
- Acosta-Rodríguez, G.F.; Lujan-Favela, M.; Parra-Quezada, R.A. Growth and yield of garlic cultivars in Delicias, Chihuahua, México. Agric. Téc. Méx. 2008, 34, 177–188. [Google Scholar]
- What Is the Situation of Garlic in México? Available online: http://hidroponia.mx/cual-es-la-situacion-del-ajo-en-mexico/ (accessed on 30 May 2019).
- Bayan, L.; Koulivand, P.H.; Gorji, A. Garlic: A review of potential therapeutic effects. Avicenna J. Phytomed. 2014, 4, 1–14. [Google Scholar]
- Adaki, S.; Adaki, R.; Shah, K.; Karagir, A. Garlic: Review of literature. Indian J. Cancer 2014, 51, 577–581. [Google Scholar] [CrossRef]
- Thomson, M.; Ali, M. Garlic [Allium sativum]: A review of its potential use as an anti-cancer agent. Curr. Cancer Drug Targets 2003, 3, 67–81. [Google Scholar] [CrossRef]
- Rana, S.V.; Pal, R.; Vaiphei, K.; Sharma, S.K.; Ola, R.P. Garlic in health and disease. Nutr. Res. Rev. 2011, 24, 60–71. [Google Scholar] [CrossRef] [Green Version]
- Ramírez-Concepción, H.R.; Castro-Velasco, L.N.; Martínez-Santiago, E. Therapeutic Effects of Garlic (Allium sativum). Health Adm. 2016, 3, 39–47. [Google Scholar]
- Aubri, J. Garlic & Digestion. Available online: https://www.livestrong.com/article/476618-garlic-digestion/ (accessed on 22 July 2019).
- Rosen, R.T.; Hiserodt, R.D.; Fukuda, E.K.; Ruiz, R.J.; Zhou, Z.; Lech, J.; Rosen, S.L.; Hartman, T.G. Determination of allicin, S-allylcysteine and volatile metabolites of garlic in breath, plasma or simulated gastric fluids. J. Nutr. 2001, 131, 968S–971S. [Google Scholar] [CrossRef]
- Amagase, H. Clarifying the real bioactive constituents of garlic. J. Nutr. 2006, 136, 716S–725S. [Google Scholar] [CrossRef]
- Colín-González, A.L.; Santana, R.A.; Silva-Islas, C.A.; Chánez-Cárdenas, M.E.; Santamaría, A.; Maldonado, P.D. The antioxidant mechanisms underlying the aged garlic extract- and S-allylcysteine-induced protection. Oxid. Med. Cell. Longev. 2012, 2012, 907162. [Google Scholar] [CrossRef]
- Park, T.; Oh, J.H.; Lee, J.H.; Park, S.C.; Jang, Y.P.; Lee, Y.J. Oral Administration of (S)-Allyl-l-Cysteine and Aged Garlic Extract to Rats: Determination of Metabolites and Their Pharmacokinetics. Planta Med. 2017, 83, 1351–1360. [Google Scholar] [CrossRef]
- Lawson, L.D.; Hunsaker, S.M. Allicin Bioavailability and Bioequivalence from Garlic Supplements and Garlic Foods. Nutrients 2018, 10, 812. [Google Scholar] [CrossRef]
- Das, T.; Choudhury, A.R.; Sharma, A.; Talukder, G. Modification of clastogenicity of three known clastogens by garlic extract in mice in vivo. Environ. Mol. Mutagen. 1993, 21, 383–388. [Google Scholar] [CrossRef]
- Choudhury, A.R.; Das, T.; Sharma, A.; Talukder, G. Inhibition of clastogenic effects of arsenic through continued oral administration of garlic extract in mice in vivo. Mutat. Res. 1997, 392, 237–242. [Google Scholar] [CrossRef]
- Shukla, Y.; Taneja, P. Antimutagenic effects of garlic extract on chromosomal aberrations. Cancer Lett. 2002, 176, 31–36. [Google Scholar] [CrossRef]
- Anuradha, S.; Rudrama Devi, K. Anti-genotoxic effects of crude garlic extract on cisplatin induced toxicity on germ cells and morphology of sperms in in vivo mouse. J. Cell Anim. Biol. 2011, 5, 279–282. [Google Scholar]
- Bhattacharya, K.; Yadava, S.; Papp, T.; Schiffmann, D.; Rahman, Q. Reduction of chrysotile asbestos-induced genotoxicity in human peripheral blood lymphocytes by garlic extract. Toxicol. Lett. 2004, 153, 327–332. [Google Scholar] [CrossRef]
- Salmon, C.P.; Knize, M.G.; Felton, J.S. Effects of marinating on heterocyclic amine carcinogen formation in grilled chicken. Food Chem. Toxicol. 1997, 35, 433–441. [Google Scholar] [CrossRef]
- Nerurkar, P.V.; LeMarchand, L.; Cooney, R.V. Effects of marinating with Asian marinades or western barbecue sauce on PhIP and MeIQx formation in barbecued beef. Nutr. Cancer 1999, 34, 147–152. [Google Scholar] [CrossRef]
- Ikken, Y.; Morales, P.; Martínez, A.; Marín, M.L.; Haza, A.I.; Cambero, M.I. Antimutagenic effect of fruit and vegetable ethanolic extrac ts against N-nitrosamines evaluated by the Ames test. J. Agric. Food Chem. 1999, 47, 3257–3264. [Google Scholar] [CrossRef]
- Bárta, I.; Smerák, P.; Polívková, Z.; Sestáková, H.; Langová, M.; Turek, B.; Bártová, J. Current trends and perspectives in nutrition and cancer prevention. Neoplasma 2006, 53, 19–25. [Google Scholar]
- Singh, S.P.; Abraham, S.K.; Kesavan, P.C. In vivo radioprotection with garlic extract. Mutat. Res. 1995, 345, 147–153. [Google Scholar] [CrossRef]
- Singh, S.P.; Abraham, S.K.; Kesavan, P.C. Radioprotection of mice following garlic pretreatment. Br. J. Cancer Suppl. 1996, 27, S102–S104. [Google Scholar]
- Arivazhagan, S.; Nagini, S.; Santhiya, S.T.; Ramesh, A. Protection of N-methyl-N′-nitro-N-nitrosoguanidine-induced in vivo clastogenicity by aqueous garlic extract. Asia Pac. J. Clin. Nutr. 2001, 10, 238–241. [Google Scholar] [CrossRef]
- Odunola, O.A. Comparative effects of some local food condiments on sodium arsenite-induced clastogenicity. Afr. J. Med. Med. Sci. 2003, 32, 75–80. [Google Scholar]
- Yadav, S.; Rahman, Q.; Geh, S.; Pant, A.B. Modulatory effects of fresh garlic extract on chrysotile asbestos induced genotoxicity: An in vitro study. Bull. Environ. Contam. Toxicol. 2006, 77, 477–483. [Google Scholar] [CrossRef]
- Premkumar, K.; Kavitha, S.; Santhiya, S.T.; Ramesh, A.R.; Suwanteerangkul, J. Interactive effects of saffron with garlic and curcumin against cyclophosphamide induced genotoxicity in mice. Asia Pac. J. Clin. Nutr. 2004, 13, 292–294. [Google Scholar]
- Bhuvaneswari, V.; Velmurugan, B.; Abraham, S.K.; Nagini, S. Tomato and garlic by gavage modulate 7,12-dimethylbenz[a]anthracene-induced genotoxicity and oxidative stress in mice. Braz. J. Med. Biol. Res. 2004, 37, 1029–1034. [Google Scholar] [CrossRef] [Green Version]
- Odunola, O.A.; Adetutu, A.; Olorunnisola, O.S.; Ola-Davis, O. Protection against 2-acetyl aminofluorene-induced toxicity in mice by garlic (Allium sativum), bitter kola (Garcina kola seed) and honey. Afr. J. Med. Med. Sci. 2005, 34, 167–172. [Google Scholar]
- Kumaraguruparan, R.; Chandra Mohan, K.V.; Abraham, S.K.; Nagini, S. Attenuation of N-methyl-N’-nitro-N-nitrosoguanidine induced genotoxicity and oxidative stress by tomato and garlic combination. Life Sci. 2005, 76, 2247–2255. [Google Scholar] [CrossRef]
- Khalil, W.K.; Ahmed, K.A.; Park, M.H.; Kim, Y.T.; Park, H.H.; Abdel-Wahhab, M.A. The inhibitory effects of garlic and Panax ginseng extract standardized with ginsenoside Rg3 on the genotoxicity, biochemical, and histological changes induced by ethylenediaminetetraacetic acid in male rats. Arch. Toxicol. 2008, 82, 183–195. [Google Scholar] [CrossRef]
- Assayed, M.E.; Khalaf, A.A.; Salem, H.A. Protective effects of garlic extract and vitamin C against in vivo cypermethrin-induced cytogenetic damage in rat bone-marrow. Mutat. Res. 2010, 702, 1–7. [Google Scholar] [CrossRef]
- Zeng, T.; Li, Y.; Zhang, C.L.; Yu, L.H.; Zhu, Z.P.; Zhao, X.L.; Xie, K.Q. Garlic oil suppressed the hematological disorders induced by chemotherapy and radiotherapy in tumor-bearing mice. J. Food Sci. 2013, 78, H936–H942. [Google Scholar] [CrossRef]
- German, J.; Alhadeff, B. Analysis of sister-chromatid exchanges. Curr. Protoc. Hum. Genet. 2001, 2, 1–10. [Google Scholar] [CrossRef]
- Edelman, J.R.; Lin, Y.J. Translocation of unstable heterochromatin as the mechanism of sister chromatid exchange formation: A proposed hypothesis. Cytobios 2001, 106, 171–191. [Google Scholar]
- Sowjanya, B.L.; Devi, K.R.; Madhavi, D. Modulatory effects of garlic extract against the cyclophosphamide induced genotoxicity in human lymphocytes in vitro. J. Environ. Biol. 2009, 30, 663–666. [Google Scholar]
- Östling, O.; Johanson, K.J. Microelectrophoretic study of radiation-induced DNA damages in individual mammalian cells. Biochem. Biophys. Res. Commun. 1984, 123, 291–298. [Google Scholar] [CrossRef]
- Singh, N.P.; McCoy, M.T.; Tice, R.R.; Schneider, E.L. A simple technique for quantitation of low levels of DNA damage in individual cells. Exp. Cell Res. 1988, 175, 184–191. [Google Scholar] [CrossRef] [Green Version]
- Araldi, R.P.; de Melo, T.C.; Mendes, T.B.; de Sá Júnior, P.L.; Nozima, B.H.; Ito, E.T.; de Carvalho, R.F.; de Souza, E.B.; de Cassia Stocco, R. Using the comet and micronucleus assays for genotoxicity studies: A review. Biomed. Pharmacother. 2015, 72, 74–82. [Google Scholar] [CrossRef]
- Singh, V.; Belloir, C.; Siess, M.H.; Le Bon, A.M. Inhibition of carcinogen-induced DNA damage in rat liver and colon by garlic powders with varying alliin content. Nutr. Cancer 2006, 55, 178–184. [Google Scholar] [CrossRef]
- Kaur, P.; Singh, R. In vivo interactive effect of garlic oil and vitamin E against stavudine induced genotoxicity in Mus musculus. Indian J. Exp. Biol. 2007, 45, 807–811. [Google Scholar]
- Liu, H.G.; Xu, L.H. Garlic oil prevents tributyltin-induced oxidative damage in vivo and in vitro. J. Food Prot. 2007, 70, 716–721. [Google Scholar] [CrossRef]
- Park, J.H.; Park, Y.K.; Park, E. Antioxidative and antigenotoxic effects of garlic (Allium sativum L.) prepared by different processing methods. Plant Foods Hum. Nutr. 2009, 64, 244–249. [Google Scholar] [CrossRef]
- Abid-Essefi, S.; Zaied, C.; Bouaziz, C.; Salem, I.B.; Kaderi, R.; Bacha, H. Protective effect of aqueous extract of Allium sativum against zearalenone toxicity mediated by oxidative stress. Exp. Toxicol. Pathol. 2012, 64, 689–695. [Google Scholar] [CrossRef]
- Ali, H.M. Mitigative role of garlic and vitamin E against cytotoxic, genotoxic, and apoptotic effects of lead acetate and mercury chloride on WI-38 cells. Pharmacol. Rep. 2018, 70, 804–811. [Google Scholar] [CrossRef]
- Heuser, V.; Andrade, V.; Peres, A.; Braga, L.; Chies, J. Influence of age and sex on the spontaneous DNA damage detected by micronucleus test and comet assay in mice peripheral blood cells. Cell Biol. Int. 2008, 32, 1223–1229. [Google Scholar] [CrossRef]
- Izzo, A.A.; Capasso, R.; Capasso, F. Eating garlic and onion: A matter of life or death. Br. J. Cancer 2004, 91, 194. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.H.; Kwon, J.Y.; Lee, J.K.; Seo, Y.R. Recent advances in in vivo genotoxicity testing: Prediction of carcinogenic potential using comet and micronucleus assay in animal models. J. Cancer Prev. 2013, 18, 277–288. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, Y.F.; Sekihashi, K.; Izumiyama, F.; Nishidate, E.; Saga, A.; Ishida, K.; Tsuda, S. The comet assay with multiple mouse organs: Comparison of comet assay results and carcinogenicity with 208 chemicals selected from the IARC monographs and U.S. NTP Carcinogenicity Database. Crit. Rev. Toxicol. 2000, 30, 629–799. [Google Scholar] [CrossRef] [PubMed]
- Bhagat, J. Combinations of genotoxic tests for the evaluation of group 1 IARC carcinogens. J. Appl. Toxicol. 2018, 38, 81–99. [Google Scholar] [CrossRef] [PubMed]
- Kirkland, D.; Aardema, M.; Henderson, L.; Müller, L. Evaluation of the ability of a battery of three in vitro genotoxicity tests to discriminate rodent carcinogens and non-carcinogens I. Sensitivity, specificity and relative predictivity. Mutat. Res. 2005, 584, 1–256. [Google Scholar] [CrossRef] [PubMed]
- Amin, A.R.M.R.; Kucuk, O.; Khuri, F.R.; Shin, D.M. Perspectives for cancer prevention with natural compounds. J. Clin. Oncol. 2009, 27, 2712–2725. [Google Scholar] [CrossRef] [PubMed]
- Mohsenzadegan, M.; Seif, F.; Farajollahi, M.M.; Khoshmirsafa, M. Anti-oxidants as chemopreventive agents in prostate cancer: A gap between preclinical and clinical studies. Recent Pat. Anticancer Drug Discov. 2018, 13, 224–239. [Google Scholar] [CrossRef]
- Paller, C.J.; Denmeade, S.R.; Carducci, M.A. Challenges of conducting clinical trials of natural products to combat cancer. Clin. Adv. Hematol. Oncol. 2016, 14, 447–455. [Google Scholar]
- Sahu, S.C. Dual role of organosulfur compounds in foods: A review. J. Environ. Sci. Health C Environ. Carcinog. Ecotoxicol. Rev. 2002, 20, 61–76. [Google Scholar] [CrossRef]
| Action Site | Types of Mechanisms |
|---|---|
| Extracellular | Inhibition of mutagen uptake |
Inhibition of endogenous formation
| |
| Complexation and/or deactivation | |
| Favouring absorption of protective agents | |
| Intracellular | Blocking or competition
|
| Stimulation of trapping and detoxification in non-target cells | |
| Modification of transmembrane transport | |
Modulation of xenobiotic metabolising enzymes
| |
| Modulation of DNA metabolism and repair | |
| Regulation of signaling pathways | |
| Enhancement of apoptosis | |
| Maintenance of genomic stability |
| Year | Aim of the Study | Conclusion | Type of Study | References |
|---|---|---|---|---|
| 1995 1996 | Singh et al. evaluated the genoprotector effect of a garlic extract (GE) against the chromosomal damage induced by three doses (0.5, 1.0, and 2.0 Gy) of gamma radiation. Previously, three doses of GE were administered orally (125, 250, and 500 mg/kg) for 5 days and, later, the mice were irradiated. | The results demonstrated that the previous treatment significantly reduced the frequency of micronucleated polychromatic erythrocytes (MNPE) induced by radiation and that the protector effect was dose dependent. | In vivo | [31,32] |
| 2001 | The objective of the study was to determine the inhibitor effect of GE on the clastogenicity of N-methyl-N′-nitro-N-nitrosoguanidine (MNNG) in Wistar rats. Similar to the two studies by Singh et al., the GE was previously administered (250 mg/kg) orally and, later, the mutagen was injected. | At the end of the study, it was observed that the pretreatment of the aqueous extract diminished the number of extracts induced by the nitrosamine carcinogen. | In vivo | [33] |
| 2003 | In this study, in vivo, the modulating potential of four aqueous extracts (Garlic, ginger (Zingiber officinale), sconio (Pimpinella anisumm LINNE), and cloves (Syzygium aromaticum)) was evaluated on the clastogenic effects of sodium arsenite in rat bone marrow cells. | The results of the investigation confirmed the following: (a) All of the extracts showed slight clastogenicity on the order of garlic > ginger, and sconio > cloves; (b) The previous treatment for 7 days with the extracts of the condiments diminished the formation of MNPE induced by the inorganic clastogen, being on the order of ginger > garlic > cloves > sconio and (c) This anticlastogenic effect can be attributed to the antioxidant properties of their chemical components. | In vivo | [34] |
| 2004 2006 | Considering that chrysotile asbestos fibers (CAFs) are environmental carcinogens, researchers from India evaluated the reduction of their genotoxicity in human peripheral blood lymphocytes by means of a garlic extract (GE). Additionally, through the electronic spin resonance (ESR), they determined the generation of hydroxyl radicals. | Their results showed that, when it was administered to the cellular culture, simultaneously the asbestos (1.0 µg/mL) plus the extract at two different concentrations (5.0 and 10 µg/mL), a clear reduction was observed in the induction of MN. On the other hand, the ESR test demonstrated that the asbestos alone induces nearly two times the signals of ESR, which diminished with the presence of the GE. The conjunction of these data suggested that the protector effect of the extract can be related to its antioxidant potential. | In vitro | [26,35] |
| 2004 | This study analyzed the interactive effects of saffron with garlic and curcumin on the genotoxicity induced by cyclophosphamide (CP) in mouse bone marrow. The animals were orally treated previously with saffron (100 mg/kg), garlic (250 mg/kg), and curcumin (10 mg/kg), alone or in combination, for 5 days, 2 h prior to the administration of the CP. | The maximal reduction of the frequency of MNPE induced by the mutagen was observed when the three test compounds were administered together. In addition, the protective effects were more pronounced in the group treated with garlic in comparison with the groups administered with curcumin and/or saffron. | In vivo | [36] |
| 2004 | The objective of the investigation was to determine the combined effect of orally administration tomato and garlic in the face of the genetic damage induced by 7,12-dimethylbenz [a] anthracene (DMBA) in Swiss albino mice. Different experimental groups were included, among these those treated with each extract individually and the combination of both compounds (tomato (500 mg/kg) and garlic (125 mg/kg)) plus the administration of the DMBA. In addition to evaluating the incidence of MN, there was the determination of lipid peroxidation and the concentrations of glutathione, glutathione peroxidase, and glutathione-S-transferase in the liver | On finalizing the experiment, it was confirmed that the animals treated with DMBA presented a greater frequency of MN and an elevated lipid peroxidation. Although the previous treatment with tomato or garlic significantly reduced genotoxicity and the parameters of oxidative stress induced by DMBA, the combination of both extracts showed a more profound effect of this inhibition. These data suggested that the combination of functional foods (especially in doses corresponding to average daily consumption, as in the case of this experiment), can be an effective alternative for inhibiting damage to the DNA produced by carcinogens. | In vivo | [37] |
| 2005 | The protection against the toxicity of 2- acetyl aminofluorene (2-AAF) exerted by the Garlic (Ga), Honey (Ho) and Bitter kola (Bk) was investigated. Mice organized into different experimental groups were dosed for 7 days with suspensions of Ga, H, and Bk as dietetic supplements; on termination of this period, they were administered a unique intraperitoneal dose of 2-AAF. Finally, the degree of clastogenicity/anticlastogenicity and liver damage was measured. | The results revealed the carcinogen induced the rate of MNPE of the bone marrow by approximately five times in comparison the control group; contrariwise, on being combined with the different suspensions of the dietetic supplements, the capacity of 2-AAF diminished to induce MN on the order of Ho > Ga > Bi. Likewise, it was observed that the activity of the gamma glutamyltransferase induced by the carcinogen was reduced on the order of Ho > Bi > Ga (serum) and Bi > Ga = Ho (liver). | In vivo | [38] |
| 2005 | The purpose of the study was to evaluate the attenuation of the genotoxicity and of the oxidative stress produced by the MNNG through pretreatment with garlic and tomato in Swiss mice using the MN assay and estimating the extension of lipid peroxidation and the state of the antioxidants of the redox cycle of the glutathione. | At the conclusion of the study, the following were confirmed: (a) the increase in MN and of lipid peroxidation is associated with the disequilibrium of the antioxidant defenses generated by the carcinogen; (b) the previous treatment with tomato and garlic (especially when administered in a combined manner) attenuated the genotoxicity induced by MNNG, and (c) the genoprotective effect of these natural compounds was associated with the reduction of glutathione (GSH) and of the enzymes dependent on GSH, glutathione peroxidase (GPx), and glutathione-S-transferase (GST). | In vivo | [39] |
| 2006 | The purpose of the research was to examine the anticlastogenic potential of the different vegetable homogenates (including garlic, red cabbage, cauliflower, broccoli, carrot, and onion), and some chemically identified substances in pure form (resveratrol, diallylsulfide, phenethylisothiocyanate, ellagic acid, epigallocatechin gallate, genistein and curcumin) against AFB1, IQ and MNU. | In general, the results evidenced that all of the homogenates and the substances of plant origin showed an important anticlastogenic effect against the three mutagens. However, garlic was found among the most significant homogenates. | In vivo | [30] |
| 2008 | The study objectives included the following two: (1) to evaluate the potency of ethylendiaminotetraacetic acid (EDTA) to induce biochemical changes, bone marrow micronuclei, and damage to the DNA, and (2) to determine the inhibitory capacity of the extract of Panax ginseng and garlic on the toxic effects of the EDTA in adult male albino rats. | The group administered only with EDTA alone showed a diminution in the biochemical parameters of the serum and the activity of the antioxidant enzymes. Also increased were lipid peroxidation and the incidence of MNPE, while in animals with the combined treatment with Panax ginseng and garlic plus EDTA significantly improved all of the parameters tested. | In vivo | [40] |
| 2010 | There is clear evidence that the insecticide cypermethrin pyretroid (CYP) is a clastogenic agent. Due to this, the protector effect of an extract of garlic (500 mg/kg) and vitamin C (VTC) against the cytogenic damage induced by CYP in the bone marrow of male white rats was analyzed. Similar to other studies, GE as well as VTC were administered individually and in combination orally for 5 days prior to the application of CYP. | As expected, the administration of CYP significantly clastogenic effects, that is, it raised the frequency of MNPE and of structural chromosomal aberrations in cells in metaphase of the bone marrow. On the other hand, the results revealed the genoprotective effect of GE and VTC, especially when they are administered in combined fashion. | In vivo | [41] |
| 2013 | Although the anticancerigenous property of the garlic has been demonstrated in different studies, there is little evidence of its protector ability on the adverse effects of chemo- and radiotherapy. In order to clarify this, a mouse model was established with a tumor xenograft through a subcutaneous injection of H22 tumor cells, which were employed to investigate the genoprotective capacity of garlic oil (GO) on chemo- and radiotherapy. For the first evaluation, the animals who were tumor carriers were treated for 14 days with cyclophosphamide (CP) individually and with the combination of CP plus GO (25 and 50 mg/kg), while for the radiotherapy test, the animals were radiated only once (5 Gy) and were administered with GO. | The results showed that GO did not increase the rate of inhibition of CP/radiation of the tumor, which indicated that GO could not increase the chemo/radiosensitivity of the cancerous cells. In addition, the treatment with GO significantly inhibited the diminution of the contents of DNA and the proportion of bone marrow micronuclei. These findings support the idea that the consumption of GO can benefit patients with cancer who receive chemotherapy or radiotherapy. | In vivo | [42] |
| Year | Aim of the Study | Conclusion | Type of Study | References |
|---|---|---|---|---|
| 2006 | Singh et al. evaluated the protective effect of three garlic powders (GP) obtained from bulbs cultivated in soils with different levels of sulfur against DNA damage induced by N-nitrosodimethylamine (NDMA) and AFB1 | After 2 weeks of pretreatment with the GP, a reduction was observed of between 35 and 60% of the damage generated by both mutagens. This supports the idea that fertilization with sulfur can exert an impact on the genoprotection of the garlic bulbs, which is probably related to an increase in the content of the alliin, a sulfured phytochemical of garlic. | In vivo | [49] |
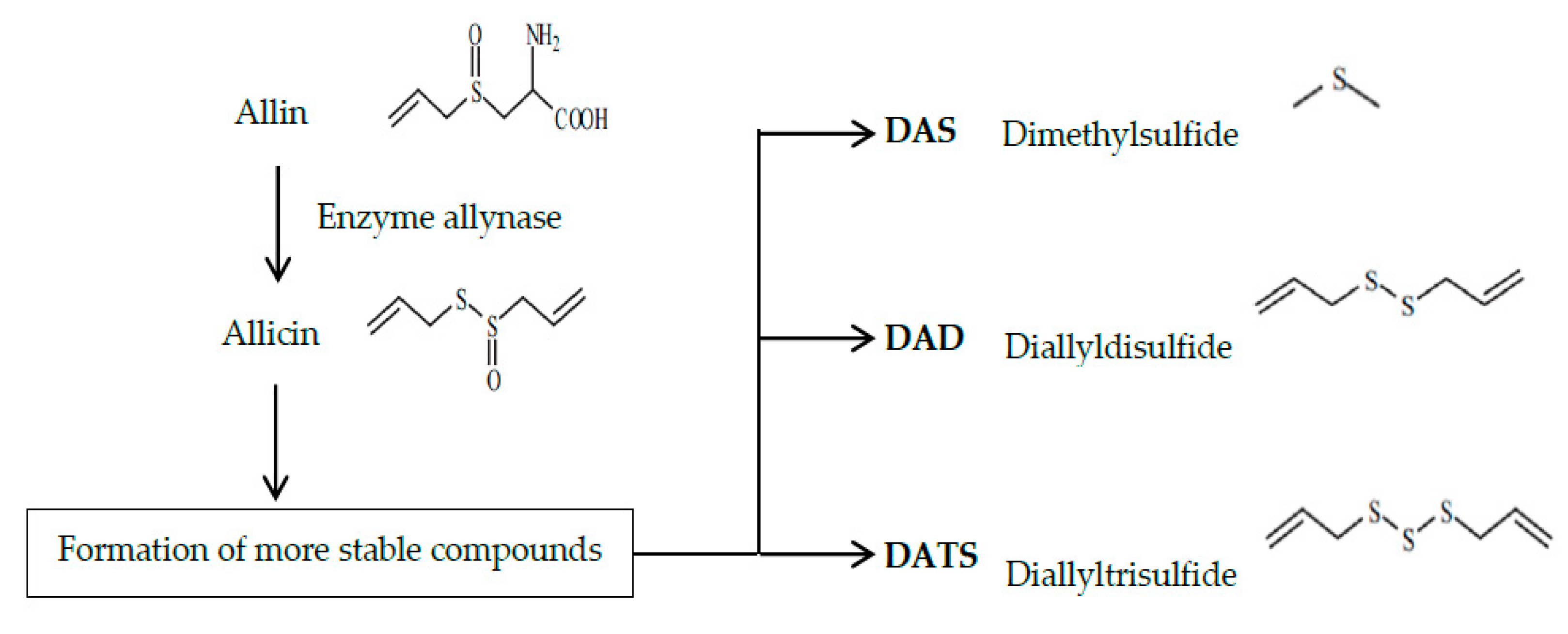
| 2006 | The objective was to determine the capacity of a garlic homogenate (GH) and the diallylsulfur (DAS) to reduce DNA fragmentation produced by AFB1, IQ, and MNU. | The results showed that GH as well as DAS fulfilled the expected objective, notably protecting the genetic material from the damage induced by the three mutagens. The latter supports the evidence that phytochemicals in the diet can perform important functions as chemopreventive agents. | In vivo | [30] |
| 2007 | In this investigation, the genomic damage was analyzed that was caused by stavudine (anti-HIV infection drug (Zerit d4T)) and the possible effect of its improvement on employing garlic oil (GO) and vitamin E (VTE). Two doses (low and high) of GO and VTE were administered, separately and in combination for 6 days, to the animals and, later, a sole dose of Zerit d4T. | VTE as well as GO, separately and in combination, reduced the clastogenicity of Zerit d4T, observing that the genoprotective effect was more pronounced with the high dose of GO plus the vitamin. These results suggest that both agents work interactively by means of an antioxidant mechanism. | In vivo | [50] |
| 2007 | Tributyltin (TBT) is a biocide employed as an additive in anti-fouling paint on the hulls of ships, wharfs, and buoys, to avoid the growth of marine organisms. Unfortunately, it can be transmitted to humans by contaminated shellfish and, to date, no effective strategy is known to eliminate the toxic effects of these foods. Therefore, in this investigation, the capacity of garlic oil (GO) to prevent the damage produced by TBT in vivo and in vitro was explored. | It was observed that, in mice as well as in amniotic cell cultures (human FL), when treated previously with GO, this significantly diminished the production of reactive oxygen species and the number of DNA cells damaged by TBT. This suggested that GO can reduce the oxidative damage induced by TBT in vivo as well as in vitro through an antioxidant mechanism. | In vivo In vitro | [51] |
| 2009 | Considering that there is evidence that high temperatures and/or cooking processes can affect the bioactivities of fruits and vegetables, the antioxidant and antigenotoxic effects of an Aged garlic extract (AGE) were analyzed in comparison with a raw garlic extract (RGE) and a heated garlic extract (HGE) obtained by different processing methods | At the end of the experiment, the results showed the following: (a) the greatest total phenol content corresponded to AGE; (b) the antioxidant activity evaluated through DPPH indicated that the HGE was significantly higher, while the activity of the SOD was in decreasing order RGE > AGE > HE, and (c) AGE was the extract type that notably reduced the damage to the DNA induced by H2O2 in the culture of leukocytes, showing an inhibition rate of approximately 70%. The data suggested that the thermal process can diminish the antioxidant and genoprotector activity of the garlic. | In vitro | [52] |
| 2012 | The objective of the present study was to evaluate the protective capacity of an aqueous extract of Allium sativum (AEA) against the cytotoxicity induced by zearalenone (ZEN), the generation of reactive oxygen species (ROS), and the fragmentation of the DNA in a culture of Vero cells. In general, the cytotoxicity was analyzed utilizing the MTT viability assay, while the antioxidant activity was carried out measuring the activity of the catalase. Finally, to determine whether the induction of oxidative stress was associated with the DNA lesions, the DNA fragmentation was sought by means of the comet assay (CA). | The results indicated that ZEN induced several toxic effects and significant alterations measured by its action in the oxidative stress, while in the combined treatment of ZEN plus AEA (250 μg/mL), an important reduction was observed of all of these damages in all of the markers evaluated, especially a significant reduction of DNA fragmentation generated by the mycotoxin | In vitro | [53] |
| 2018 | Evaluation of the interactive effect of the garlic and the vitamin E (VTE) against the cytotoxic and genotoxic damage of lead acetate (Led) and the mercury chloride (Mer) in a culture of human pulmonary cells (WI-38). Initially, the WI-38 cells were treated with garlic and VTE for 24 h and, later, with Led and/or Mer alone or combined for 24 h. | In general, it was observed that Led or Mer or the combination of both induced serious damage to the DNA, a phenomenon that was reverted when the cells were pretreated with VTE plus garlic. The latter suggests that the garlic can interact with the vitamin, generating a very promising protector effect against the toxic effect of heavy metals. | In vitro | [54] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morales-González, J.A.; Madrigal-Bujaidar, E.; Sánchez-Gutiérrez, M.; Izquierdo-Vega, J.A.; Valadez-Vega, M.d.C.; Álvarez-González, I.; Morales-González, Á.; Madrigal-Santillán, E. Garlic (Allium sativum L.): A Brief Review of Its Antigenotoxic Effects. Foods 2019, 8, 343. https://doi.org/10.3390/foods8080343
Morales-González JA, Madrigal-Bujaidar E, Sánchez-Gutiérrez M, Izquierdo-Vega JA, Valadez-Vega MdC, Álvarez-González I, Morales-González Á, Madrigal-Santillán E. Garlic (Allium sativum L.): A Brief Review of Its Antigenotoxic Effects. Foods. 2019; 8(8):343. https://doi.org/10.3390/foods8080343
Chicago/Turabian StyleMorales-González, José Antonio, Eduardo Madrigal-Bujaidar, Manuel Sánchez-Gutiérrez, Jeannett A. Izquierdo-Vega, María del Carmen Valadez-Vega, Isela Álvarez-González, Ángel Morales-González, and Eduardo Madrigal-Santillán. 2019. "Garlic (Allium sativum L.): A Brief Review of Its Antigenotoxic Effects" Foods 8, no. 8: 343. https://doi.org/10.3390/foods8080343
APA StyleMorales-González, J. A., Madrigal-Bujaidar, E., Sánchez-Gutiérrez, M., Izquierdo-Vega, J. A., Valadez-Vega, M. d. C., Álvarez-González, I., Morales-González, Á., & Madrigal-Santillán, E. (2019). Garlic (Allium sativum L.): A Brief Review of Its Antigenotoxic Effects. Foods, 8(8), 343. https://doi.org/10.3390/foods8080343






