Distribution, Contents, and Health Risk Assessment of Cadmium, Lead, and Nickel in Bananas Produced in Ecuador
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Collection and Preparation Process
2.2. Metal Determination
2.3. Quality Control
- Cadmium Certified Reference Material Certipure® (Merck, Darmstadt, Germany), 986 mg/kg ± 4 mg/kg, density 1.0131 g/cm3, Ord. No. 1.19777.0100, Lot No. HC60709577.
- Lead Certified Reference Material (Inorganic Ventures, Christiansburg, VA, USA), 999 μg/cm3 ± 3 μg/cm3, density 1.010 g/cm3, Cat. No. CGNI1, Lot No. J2-NI02103.
- Nickel Certified Reference Material (Inorganic Ventures), 1003 μg/cm3 ± 5 μg/cm3, density 1.002 g/cm3, Cat. No. CGPB1, Lot No. M2-PB656988.
2.4. Human Health Risk Assessment
2.4.1. EDI
2.4.2. Non-Carcinogenic Risk
2.4.3. Carcinogenic Risk
3. Results and Discussion
3.1. Sample Collection and Preparation Process
3.2. Metal Determination
3.3. Quality Control
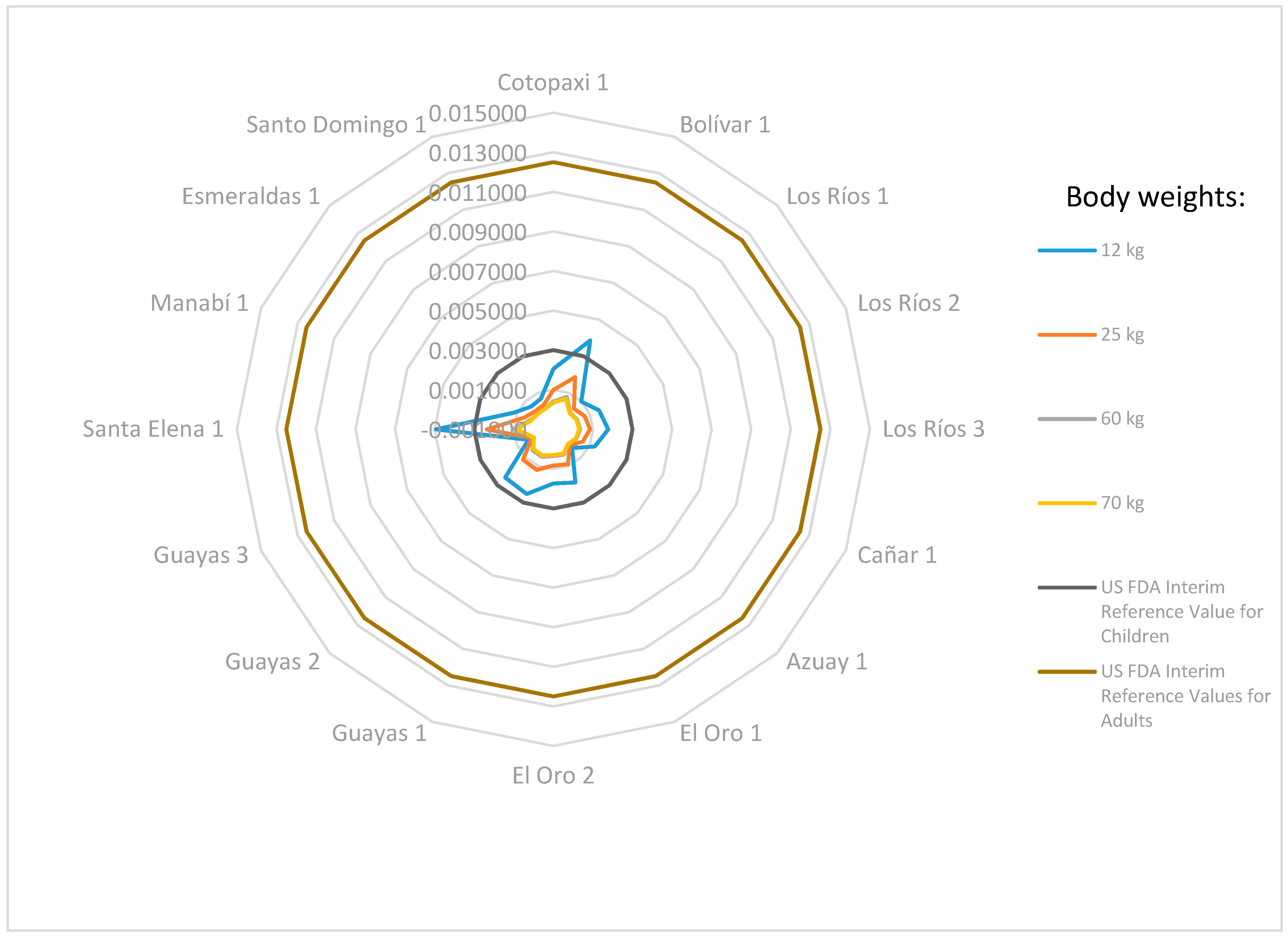
3.4. Health Risk Assessment Results
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Shaheen, N.; Irfan, N.M.; Khan, I.N.; Islam, S.; Islam, M.S.; Ahmed, M.K. Presence of heavy metals in fruits and vegetables: Health risk implications in Bangladesh. Chemosphere 2016, 152, 431–438. [Google Scholar] [CrossRef] [PubMed]
- Vallejo Toro, P.P.; Vásquez Bedoya, L.F.; Correa, I.D.; Bernal Franco, G.R.; Alcántara-Carrió, J.; Palacio Baena, J.A. Impact of terrestrial mining and intensive agriculture in pollution of estuarine surface sediments: Spatial distribution of trace metals in the Gulf of Urabá, Colombia. Mar. Pollut. Bull. 2016, 111, 311–320. [Google Scholar] [CrossRef] [PubMed]
- Antoine, J.M.R.; Hoo Fung, L.A.; Grant, C.N. Assessment of the potential health risks associated with the aluminium, arsenic, cadmium and lead content in selected fruits and vegetables grown in Jamaica. Toxicol. Rep. 2017, 4, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Tóth, G.; Hermann, T.; Da Silva, M.R.; Montanarella, L. Heavy metals in agricultural soils of the European Union with implications for food safety. Environ. Int. 2016, 88, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Peralta-Videa, J.R.; López, M.L.; Narayan, M.; Saupe, G.; Gardea-Torresdey, J. The biochemistry of environmental heavy metal uptake by plants: Implications for the food chain. Int. J. Biochem. Cell Biol. 2009, 41, 1665–1677. [Google Scholar] [CrossRef] [PubMed]
- Järup, L. Hazards of heavy metal contamination. Br. Med. Bull. 2003, 68, 167–182. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gall, J.E.; Boyd, R.S.; Rajakaruna, N. Transfer of heavy metals through terrestrial food webs: A review. Environ. Monit. Assess. 2015. [Google Scholar] [CrossRef] [PubMed]
- Clemens, S.; Feng MA, J. Toxic Heavy Metal and Metalloid Accumulation in Crop Plants and Foods. Annu. Rev. Plant. Biol. 2016, 67, 489–512. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Škrbić, B.; Živančev, J.; Mrmoš, N. Concentrations of arsenic, cadmium and lead in selected foodstuffs from Serbian market basket: Estimated intake by the population from the Serbia. Food Chem. Toxicol. 2013, 58, 440–448. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Olivas, R.; Cámara, C. Speciation Related to Human Health. In Trace Element Speciation for Environment, Food and Health; Royal Society of Chemistry: Manchester, UK, 2001; pp. 331–353. ISBN 0-85404-459-0. [Google Scholar]
- Celik, U.; Oehlenschlager, J. High contents of cadmium, lead, zinc and copper in popular fishery products sold in Turkish supermarkets. Food Control. 2007, 18, 258–261. [Google Scholar] [CrossRef]
- IARC. Agents Classified by the IARC Monographs, Volumes 1–123. Available online: https://monographs.iarc.fr/wp-content/uploads/2019/02/List_of_Classifications.pdf (accessed on 31 May 2019).
- EPA-IRIS. Cadmium; CASRN 7440-43-9. Integrated Risk Information System (IRIS), Chemical Assessment Summary File, First On-Line 03/31/1987. Available online: https://cfpub.epa.gov/ncea/iris/iris_documents/documents/subst/0141_summary.pdf (accessed on 31 May 2019).
- International Programme on Chemical Safety. Environmental Health Criteria: Cadmium (EHC 134). The International Programme on Chemical Safety (IPCS). Available online: http://www.inchem.org/documents/ehc/ehc/ehc134.htm (accessed on 31 May 2019).
- Zhou, Z.Y.; Fan, Y.P.; Wang, M.J. Heavy metal contamination in vegetables and their control in China. Food Rev. Int. 2000, 16, 239–255. [Google Scholar] [CrossRef]
- EPA-IRIS. Lead and compounds (inorganic); CASRN 7439-92-1. Integrated Risk Information System (IRIS), Chemical Assessment Summary, File First On-Line 03/01/1988. Available online: https://cfpub.epa.gov/ncea/iris/iris_documents/documents/subst/0277_summary.pdf (accessed on 31 May 2019).
- International Programme on Chemical Safety. Environmental Health Criteria: Inorganic lead (EHC 165). Available online: http://www.inchem.org/documents/ehc/ehc/ehc165.htm (accessed on 31 May 2019).
- EPA-IRIS. Nickel, soluble salts; CASRN Various. Integrated Risk Information System (IRIS) Chemical Assessment Summary, File First On-Line 09/30/1987. Available online: https://cfpub.epa.gov/ncea/iris/iris_documents/documents/subst/0271_summary.pdf (accessed on 31 May 2019).
- International Programme on Chemical Safety. Nickel, Nickel Carbonyl, and Some Nickel Compounds Health and Safety Guide (Health Safety Guide 62). Available online: http://www.inchem.org/documents/hsg/hsg/hsg062.htm (accessed on 31 May 2019).
- EPA. Nickel Compounds Hazard Summary. Nickel. Available online: https://www.epa.gov/sites/production/files/2016-09/documents/nickle-compounds.pdf (accessed on 31 May 2019).
- European Commission. Commission Regulation No 1881/2006 of 19 December 2006: Setting maximum levels for certain contaminants in foodstuffs (Text with EEA relevance). Official Journal of the European Union 2006. Available online: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2006:364:0005:0024:EN:PDF (accessed on 31 May 2019).
- FAO/WHO. General Standard for Contaminants and Toxins in Food and Feed CXS 193-1995 (Revision 2018). Available online: http://www.fao.org/fao-who-codexalimentarius/sh-proxy/es/?lnk=1&url=https%253A%252F%252Fworkspace.fao.org%252Fsites%252Fcodex%252FStandards%252FCODEX%2BSTAN%2B193-1995%252FCXS_193e.pdf (accessed on 31 May 2019).
- FAO/WHO. Standard for Bananas (Codex STAN 205-1997) (Revision 2005). Available online: http://www.fao.org/fao-who-codexalimentarius/sh-proxy/es/?lnk=1&url=https%253A%252F%252Fworkspace.fao.org%252Fsites%252Fcodex%252FStandards%252FCODEX%2BSTAN%2B205-1997%252FCXS_205e.pdf (accessed on 31 May 2019).
- EPA. Regional Screening (RBL) Summary Table (November 2018). Available online: https://semspub.epa.gov/work/HQ/197414.pdf (accessed on 31 May 2019).
- United States Food and Drugs Administration. Lead in Food, Foodwares, and Dietary Supplements. Available online: https://www.fda.gov/food/metals/lead-food-foodwares-and-dietary-supplements (accessed on 31 May 2019).
- European Food Safety Authority. Chemical Hazards Data—OpenFoodTox. Available online: https://www.efsa.europa.eu/en/data/chemical-hazards-data (accessed on 31 May 2019).
- Roibás, L.; Elbehri, A.; Hospido, A. Carbon footprint along the Ecuadorian banana supply chain: Methodological improvements and calculation tool. J. Cleaner Prod. 2016, 112, 2441–2451. [Google Scholar] [CrossRef]
- Banco Central del Ecuador. Exportaciones FOB por Producto Principal. Available online: https://contenido.bce.fin.ec/home1/estadisticas/bolmensual/IEMensual.jsp (accessed on 31 May 2019).
- Ministerio de Comercio Exterior. Informe Sobre el Sector Bananero Ecuatoriano. Available online: http://panama.embajada.gob.ec/wp-content/uploads/2017/06/informe_sobre_el_sector_bananero_ecuatoriano_29.05.2017_def..pdf (accessed on 31 May 2019).
- Sial, T.A.; Khan, M.N.; Lan, Z.; Kumbhar, F.; Zhao, Y.; Zhang, J.; Kumbhar, F.; Ying, Z.; Zhang, J.; Sun, D.; et al. Contrasting effects of banana peels waste and its biochar on greenhouse gas emissions and soil biochemical properties. Process. Saf. Environ. Prot. 2018, 122, 366–377. [Google Scholar] [CrossRef]
- Mahindrakar, K.V.; Rathod, V.K. Utilization of banana peels for removal of strontium (II) from water. Environ. Technol. Innov. 2018, 11, 371–383. [Google Scholar] [CrossRef]
- Feitosa, A.A.; Ritter, E.; Teixeira, W.G.; Abreu de Rezende, F.; Jürgen, K. Sorption of Ammonium in Banana Peel and Orange Bagasse Biochars. In Proceedings of the 8th International Congress on Environmental Geotechnics, Hangzhou, China, 28 October–1 November 2018; Springer: Basingstoke, UK, 2019; pp. 577–584. [Google Scholar]
- Brisbois, B. Bananas, pesticides and health in southwestern Ecuador: A scalar narrative approach to targeting public health responses. Soc. Sci. Med. 2016, 150, 184–191. [Google Scholar] [CrossRef] [PubMed]
- CIIEMAD-IPN. Método General Por Microondas de Digestión Ácida en Matrices Ambientales. Available online: http://www.ciiemad.ipn.mx/SGC/Documents/Procedimientos/IPN_AC-06-00.pdf (accessed on 31 May 2019).
- AOAC. Guidelines for Single Laboratory Validation of Chemical Methods for Dietary Supplements and Botanicals. Available online: https://www.aoac.org/aoac_prod_imis/AOAC_Docs/StandardsDevelopment/SLV_Guidelines_Dietary_Supplements.pdf (accessed on 31 May 2019).
- Antunović, B.; Barlow, S.; Chesson, A.; Flynn, A.; Hardy, A.; Jeger, M.; Vannier, P. Guidance on selected default values to be used by the EFSA Scientific Committee, Scientific Panels and Units in the absence of actual measured data. EFSA J. 2012, 10, 1–32. [Google Scholar]
- WHO. Weight for age BOYS. Available online: https://www.who.int/growthref/wfa_boys_5_10years_z.pdf?ua=1 (accessed on 31 May 2019).
- WHO. Weight for age GIRLS. Available online: https://www.who.int/growthref/wfa_girls_5_10years_z.pdf?ua=1 (accessed on 31 May 2019).
- De Onis, M.; Onyango, A.W.; Borghi, E.; Siyam, A.; Siekmann, J. Development of a WHO growth reference for school-aged children and adolescents. Bull. World Health Organ. 2007, 85, 660–667. [Google Scholar] [CrossRef] [PubMed]
- EPA. Risk Assessment Guidance for Superfund Volume I. Human Health Evaluation Manual (Part A). (Interim Final). Available online: https://www.epa.gov/sites/production/files/2015-09/documents/rags_a.pdf (accessed on 31 May 2019).
- EPA. Guidelines for the Health Risk Assessment of Chemical Mixtures. Available online: https://www.epa.gov/sites/production/files/2014-11/documents/chem_mix_1986.pdf (accessed on 31 May 2019).
- EPA. Risk Assessment Guidance for Superfund: Part A, Process for Conducting Probabilistic Risk Assessment, 3. Available online: https://www.epa.gov/risk/risk-assessment-guidance-superfund-rags-volume-iii-part (accessed on 31 May 2019).
- Del Cioppo Morstadt, J.; Salazar Veloz, R. Ecuador: Exportación de Banano (Musa sp.) Estudio Sectorial del Banano Ecuatoriano de Exportación. Available online: https://www.researchgate.net/publication/309395087_ECUADOR_EXPORTACION_DE_BANANO_Musa_sp_ESTUDIO_SECTORIAL_DEL_BANANO_ECUATORIANO_DE_EXPORTACION (accessed on 31 May 2019).
- Felix, B.I.; Mite, F.; Carrillo, M.; Pino, M. Avances De Investigacion Del Proyecto Determinación de Metales Contaminantes en Cultivos de Exportación y su Repercusion sobre la Calidad de los Mismos. Available online: http://www.secsuelo.org/wp-content/uploads/2015/06/5-Determinacion-de-metales-contaminantes-Felix-I.pdf (accessed on 31 May 2019).
- Orisakwe, O.E.; Nduka, J.K.; Amadi, C.N.; Dike, D.O.; Bede, O. Heavy metals health risk assessment for population via consumption of food crops and fruits in Owerri, South Eastern, Nigeria. Chem. Cent. J. 2012, 6, 77. [Google Scholar] [CrossRef] [PubMed]
| Provinces | Samples | Cd | Ni | Pb | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Cc | RSD | Accuracy | Cc | RSD | Accuracy | Cc | RSD | Accuracy | ||
| Cotopaxi | Cotopaxi 1 | 9.3 | 5.54% | 95.22% | 105.6 | 1.54% | 107.31% | 224.4 | 0.33% | 93.50% |
| Bolívar | Bolívar 1 | 32.5 | 5.38% | 110.34% | 27.7 | 5.93% | 101.29% | 420.9 | 1.88% | 114.62% |
| Los Ríos | Los Ríos 1 | 20.1 | 7.73% | 93.50% | 23.2 | 3.61% | 105.90% | 107.7 | 3.27% | 93.72% |
| Los Ríos 2 | 19.4 | 5.26% | 95.73% | 29.1 | 6.16% | 83.96% | 163.0 | 2.26% | 115.31% | |
| Los Ríos 3 | 30.2 | 5.93% | 96.52% | 21.5 | 6.87% | 112.57% | 193.5 | 2.63% | 106.29% | |
| Cañar | Cañar 1 | 33.5 | 6.68% | 119.78% | 21.8 | 6.32% | 80.63% | 138.1 | 0.78% | 108.48% |
| Azuay | Azuay 1 | 26.0 | 5.55% | 88.49% | 29.8 | 0.83% | 107.39% | 36.9 | 0.81% | 109.31% |
| El Oro | El Oro 1 | 29.8 | 5.39% | 92.73% | 24.9 | 6.40% | 109.60% | 208.2 | 3.47% | 86.55% |
| El Oro 2 | 29.6 | 1.17% | 86.47% | 24.5 | 5.34% | 87.06% | 189.3 | 0.56% | 82.68% | |
| Guayas | Guayas 1 | 17.4 | 7.43% | 105.97% | 30.5 | 6.87% | 108.01% | 276.5 | 3.74% | 111.64% |
| Guayas 2 | 24.3 | 7.77% | 87.96% | 22.3 | 5.58% | 116.60% | 266.5 | 1.18% | 83.36% | |
| Guayas 3 | 17.9 | 5.95% | 101.74% | 21.6 | 5.74% | 116.36% | 40.1 | 3.16% | 88.98% | |
| Santa Elena | Santa Elena 1 | 21.3 | 7.19% | 82.90% | 17.7 | 4.31% | 85.99% | 538.0 | 1.81% | 84.27% |
| Manabí | Manabí 1 | 47.3 | 4.07% | 97.19% | 30.8 | 4.84% | 84.89% | 128.8 | 2.93% | 81.29% |
| Esmeraldas | Esmeraldas 1 | 13.8 | 6.48% | 86.64% | 16.1 | 5.77% | 96.12% | 67.0 | 3.74% | 81.96% |
| Santo Domingo | Santo Domingo 1 | 14.9 | 4.62% | 107.59% | 19.4 | 4.38% | 95.89% | 72.9 | 0.51% | 109.04% |
| Threshold values | 50.0 a | - | - | 300.0 b | - | - | 100.0 a,c | - | - | |
| Country | Metal | Mean concentrations | EDI | THQ | CR | References |
|---|---|---|---|---|---|---|
| Bangladesh | Cd | ND | NA | 0 | NA | [1] |
| Ni | 37.0 | 2.80 × 10−5 | 1.00 × 10−3 | NA | ||
| Pb | 3.0 | 2.20 × 10−6 | 6.00 × 10−4 | 1.90 × 10−8 | ||
| Serbia | Cd | <0.3 | 0.002 | NA | NA | [9] |
| Pb | 60.0 | 1.254 | NA | NA | ||
| Jamaica | Cd | 57.0 | 0.028 | 0.028 | NA | [3] |
| Pb | 10.0 | 0.005 | 0.002 | NA | ||
| Nigeria | Cd | ND | NA | NA | NA | [45] |
| Pb | 460.0 | 0.0028 | NA | NA | ||
| Ecuador * | Cd | 24.0 | 4.44 × 10−5 | 0.044 | NA | The present study |
| Ni | 29.0 | 5.35 × 10−5 | 0.005 | NA | ||
| Pb | 192.0 | 3.52 × 10−4 | 0.099 | 3.77 × 10−7 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Romero-Estévez, D.; Yánez-Jácome, G.S.; Simbaña-Farinango, K.; Navarrete, H. Distribution, Contents, and Health Risk Assessment of Cadmium, Lead, and Nickel in Bananas Produced in Ecuador. Foods 2019, 8, 330. https://doi.org/10.3390/foods8080330
Romero-Estévez D, Yánez-Jácome GS, Simbaña-Farinango K, Navarrete H. Distribution, Contents, and Health Risk Assessment of Cadmium, Lead, and Nickel in Bananas Produced in Ecuador. Foods. 2019; 8(8):330. https://doi.org/10.3390/foods8080330
Chicago/Turabian StyleRomero-Estévez, David, Gabriela S. Yánez-Jácome, Karina Simbaña-Farinango, and Hugo Navarrete. 2019. "Distribution, Contents, and Health Risk Assessment of Cadmium, Lead, and Nickel in Bananas Produced in Ecuador" Foods 8, no. 8: 330. https://doi.org/10.3390/foods8080330
APA StyleRomero-Estévez, D., Yánez-Jácome, G. S., Simbaña-Farinango, K., & Navarrete, H. (2019). Distribution, Contents, and Health Risk Assessment of Cadmium, Lead, and Nickel in Bananas Produced in Ecuador. Foods, 8(8), 330. https://doi.org/10.3390/foods8080330





