The Introduction of Thousands of Tonnes of Glyphosate in the food Chain—An Evaluation of Glyphosate Tolerant Soybeans
Abstract
1. A Quantitative Success
2. A Qualitative Evaluation
3. Research Question
3.1. Are GT Soybeans Compositionally Equivalent to Their Non-GM Counterparts?
3.2. How does the Quality of Feed Made from Roundup Ready Soybeans Compare to Feed from Conventional or Organic Soybeans?
3.3. Can Adverse Dose-related Effects from Glyphosate Residues be Detected in Consumers?
3.4. Are Glyphosate and Roundup Practically Non-toxic?
4. The Effective Residue Levels of Glyphosate Are Rising Contrary to Published Recommendations
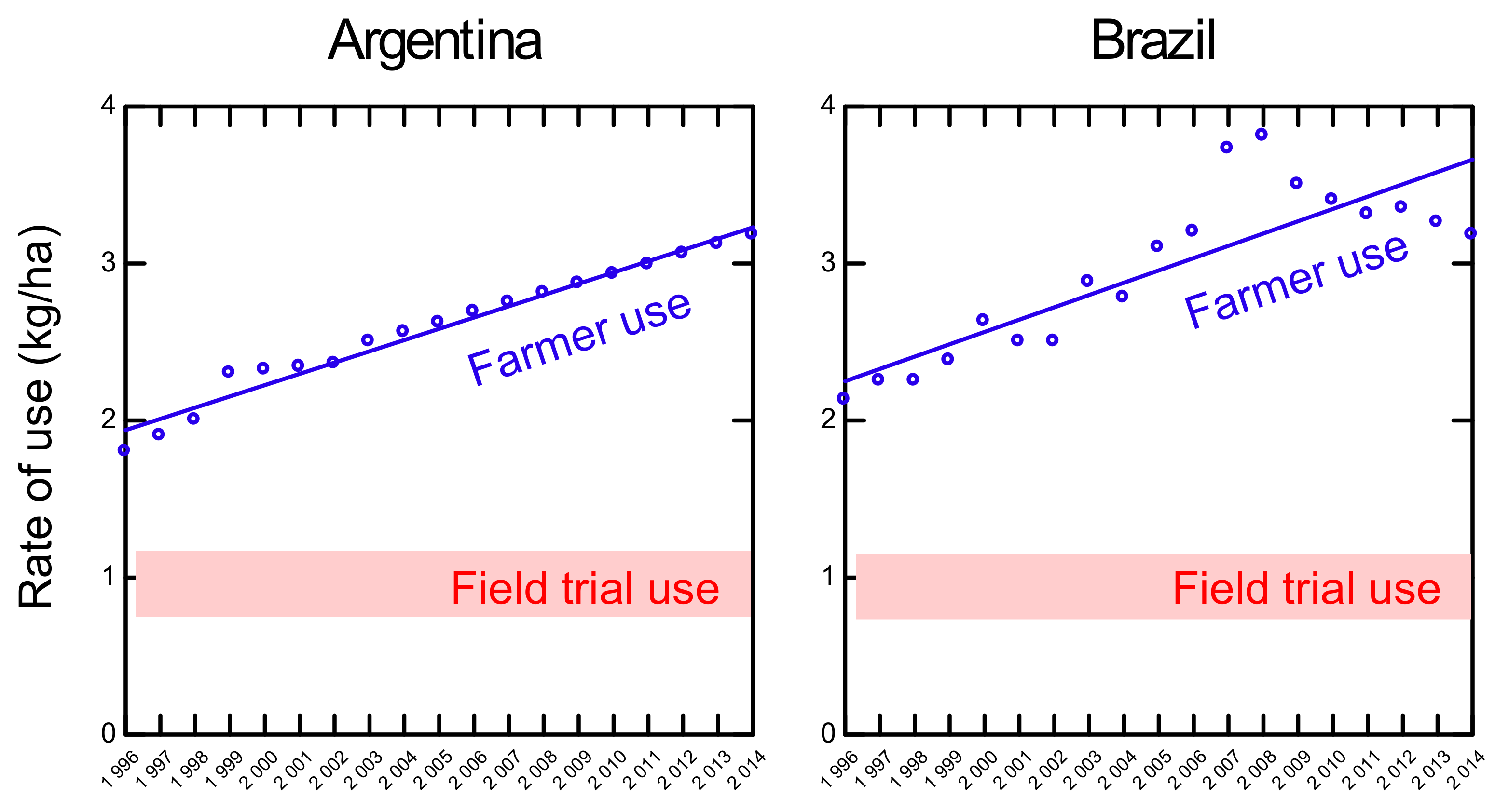
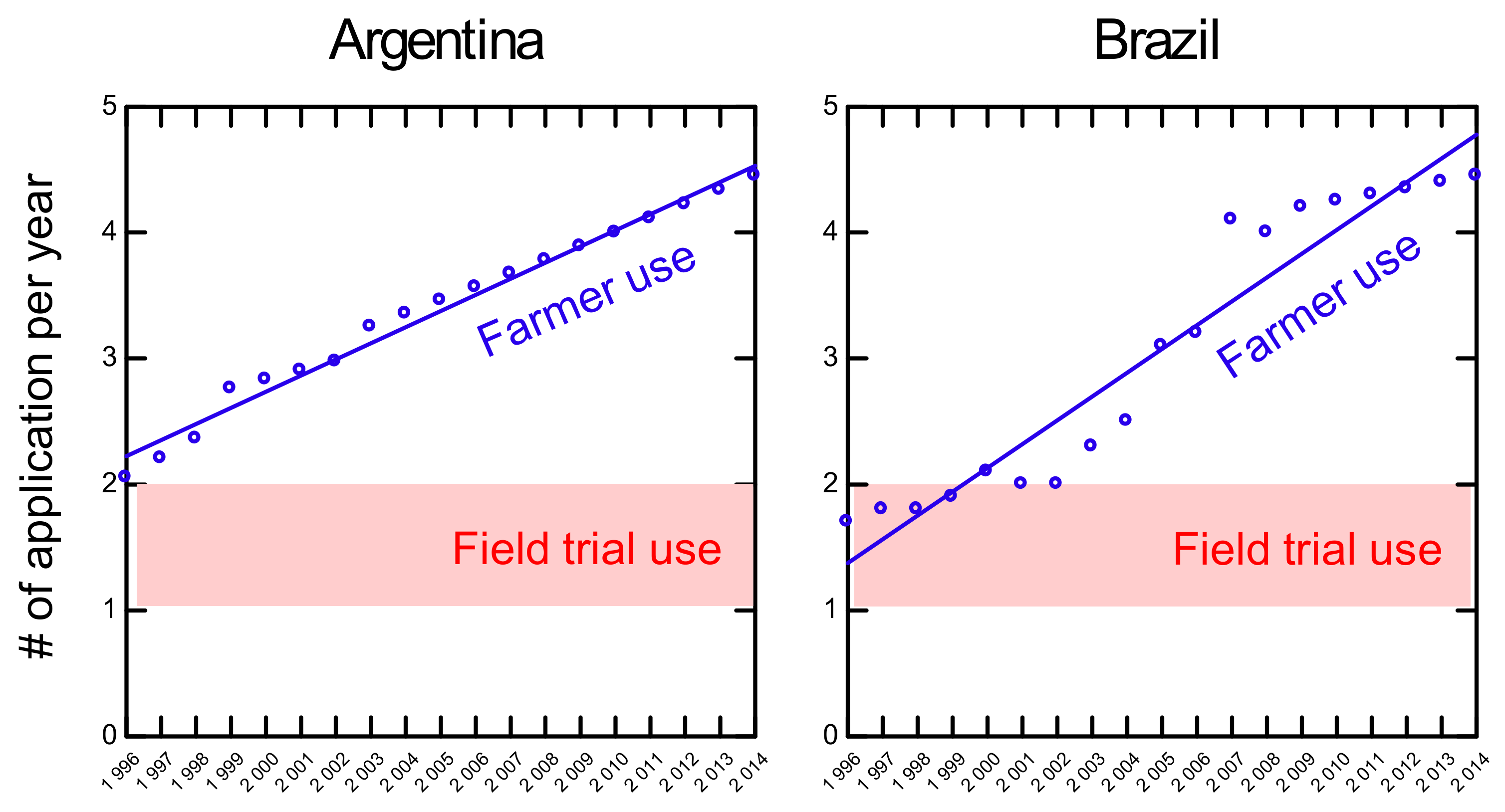
5. Application Rates of Glyphosate on Herbicide Tolerant Soybeans: Farmers’ Use vs Field Trial Use
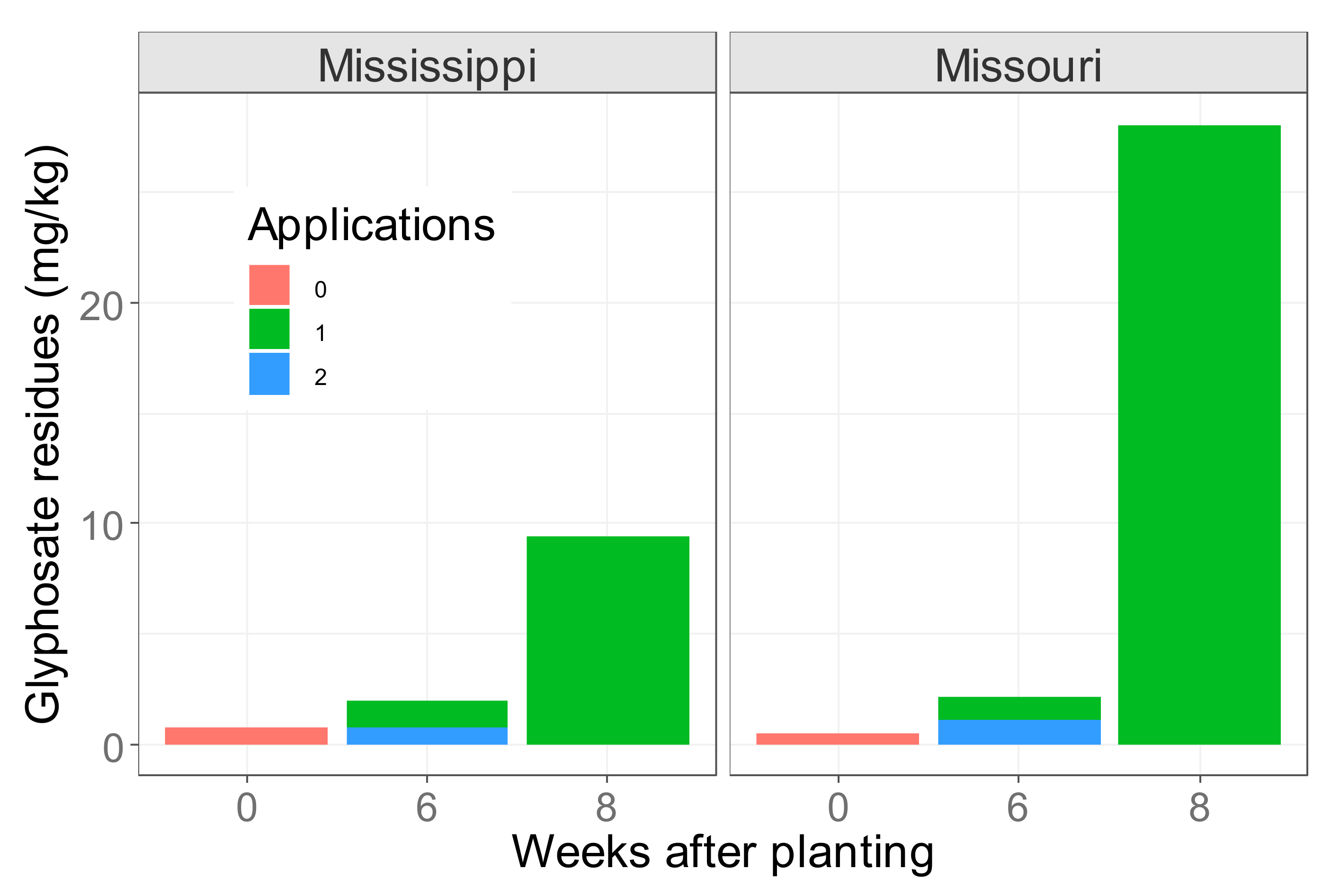
6. Glyphosate Residue Levels and Timing of Spraying
7. How Much Glyphosate Enters the Food Chain?
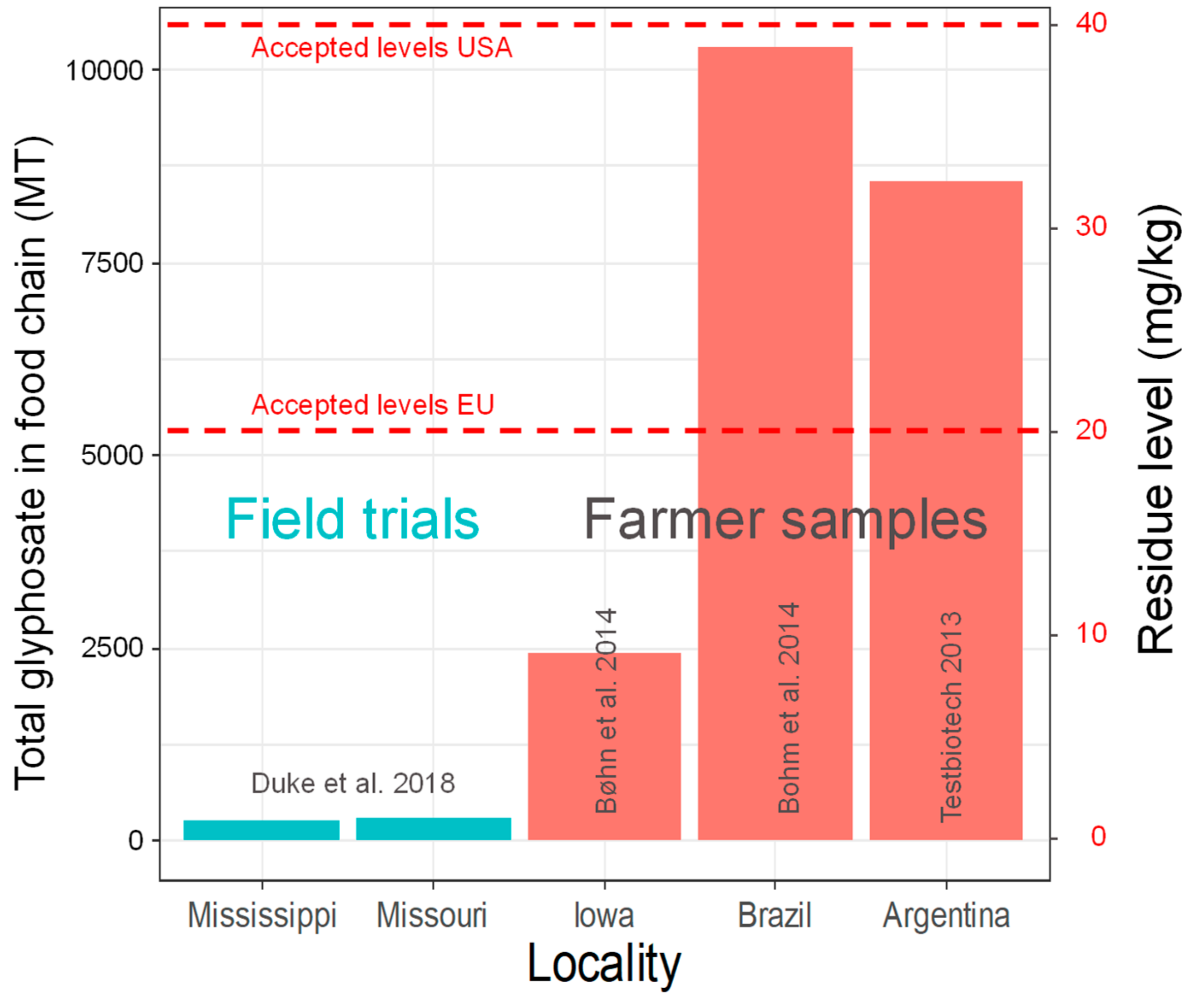
8. Sprayed Adjuvants to Herbicides Can Be More Toxic But Are Not Monitored
9. A Need for International Coordination of Pesticide Residues in Food and Feed
- Article 29 of Regulation 1107/2009: active substances and synergists have to be approved, the maximum residue levels for the specific agricultural products have to be specified;
- Article 4 of Regulation 1107/2009: pesticides must not have any harmful effects on human or animal health, taking into account known cumulative and synergistic effects;
- Recital 5 of Regulation 396/2005: residues should not be present at levels presenting an unacceptable risk to humans and, where relevant, to animals;
- Recital 10 of Regulation 396/2005: specific MRLs for each pesticide in food and feed products have to be established;
- Recital 26 of Regulation 396/2005: MRLs have to be set for food and feed produced outside the Community if produced by different agricultural practices as regards the use of plant protection products;
- Article 14 of Regulation 396/2005: the presence of pesticide residues arising from sources other than current plant protection uses and their known cumulative and synergistic effects have to be determined; any potential risks to consumers with a high intake and high vulnerability have to be taken into account.
10. Research Material for Independent Testing is not Openly Available
11. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- James, C. Global Status of Commercialized Biotech/GM Crops: 2017; ISAAA: Ithaca, NY, USA, 2017. [Google Scholar]
- Brookes, G.; Barfoot, P. Farm income and production impacts of using GM crop technology 1996–2015. Gm Crop. Food 2017, 8, 156–193. [Google Scholar] [CrossRef] [PubMed]
- Benbrook, C.M. Trends in glyphosate herbicide use in the United States and globally. Environ. Sci. Eur. 2016, 28, 1. [Google Scholar] [CrossRef] [PubMed]
- USDA. World Agricultural Supply and Demand Estimates; United States Department of Agriculture: Washington, DC, USA, 2018; pp. 1–40.
- FDA. FDA Statement of Policy: Foods Derived from New Plant Varieties. 1992 [Cited US FDA. Policy Brief in the Federal Register (57 FR 22984) 28 May 2015]. Available online: http://www.fda.gov/food/biotechnology/default.html (accessed on 18 October 2018).
- Bawa, A.; Anilakumar, K. Genetically modified foods: Safety, risks and public concerns—A review. J. Food Sci. Technol. 2013, 50, 1035–1046. [Google Scholar] [CrossRef] [PubMed]
- Traavik, T.; Heinemann, J.A. Genetic Engineering and Omitted Health Research: Still no Answers to Ageing Questions; Chapter 9 in Biosafety, First; Traavik, T., Ching, L.L., Eds.; Third World Network: Penang, Malaysia, 2009. [Google Scholar]
- Domingo, J.L.; Bordonaba, J.G. A literature review on the safety assessment of genetically modified plants. Environ. Int. 2011, 37, 734–742. [Google Scholar] [CrossRef] [PubMed]
- Domingo, J.L. Safety assessment of GM plants: An updated review of the scientific literature. Food Chem. Toxicol. 2016, 95, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Millstone, E.; Brunner, E.; Mayer, S. Beyond ‘substantial equivalence’. Nature 1999, 401, 525–526. [Google Scholar] [CrossRef] [PubMed]
- Diels, J.; Cunha, M.; Manaia, C.; Sabugosa-Madeira, B.; Silva, M. Association of financial or professional conflict of interest to research outcomes on health risks or nutritional assessment studies of genetically modified products. Food Policy 2011, 36, 197–203. [Google Scholar] [CrossRef]
- Waltz, E. Under wraps. Nat. Biotechnol. 2009, 27, 880–882. [Google Scholar] [CrossRef]
- Waltz, E. Battlefield. Nature 2009, 461, 27–32. [Google Scholar] [CrossRef]
- Rang, A.; Linke, B.; Jansen, B. Detection of RNA variants transcibed from the transgene in Roundup Ready soybean. Eur. Food Res. Technol. 2005, 220, 438–443. [Google Scholar] [CrossRef]
- Myers, J.P.; Antoniou, M.N.; Blumberg, B.; Carroll, L.; Colborn, T.; Everett, L.G.; Hansen, M.; Landrigan, P.J.; Lanphear, B.P.; Mesnage, R. Concerns over use of glyphosate-based herbicides and risks associated with exposures: A consensus statement. Environ. Health 2016, 15, 1. [Google Scholar] [CrossRef] [PubMed]
- Van Straalen, N.M.; Legler, J. Decision-making in a storm of discontent. Science 2018, 360, 958–960. [Google Scholar] [CrossRef] [PubMed]
- Bøhn, T.; Cuhra, M.; Traavik, T.; Sanden, M.; Fagan, J.; Primicerio, R. Compositional differences in soybeans on the market: Glyphosate accumulates in Roundup Ready GM soybeans. Food Chem. 2014, 153, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Duke, S.O.; Rimando, A.M.; Reddy, K.N.; Cizdziel, J.V.; Bellaloui, N.; Shaw, D.R.; Williams, M.M.; Maul, J.E. Lack of transgene and glyphosate effects on yield, and mineral and amino acid content of glyphosate-resistant soybean. Pest Manag. Sci. 2018, 74, 1166–1173. [Google Scholar] [CrossRef]
- Cuhra, M.; Traavik, T.; Bøhn, T. Life cycle fitness differences in Daphnia magna fed Roundup-Ready soybean or conventional soybean or organic soybean. Aquac. Nutr. 2014, 21, 702–713. [Google Scholar] [CrossRef]
- Cuhra, M.; Traavik, T.; Dando, M.; Primicerio, R.; Holderbaum, D.; Bøhn, T. Glyphosate-residues in Roundup-ready soybean impair D. magna life-cycle. J. Agric. Chem. Environ. 2015, 4, 24–36. [Google Scholar] [CrossRef][Green Version]
- Williams, G.M.; Kroes, R.; Munro, I.C. Safety evaluation and risk assessment of the herbicide Roundup and its active ingredient, glyphosate, for humans. Regul. Toxicol. Pharmacol. 2000, 31, 117–165. [Google Scholar] [CrossRef]
- Giesy, J.P.; Dobson, S.; Solomon, K.R. Ecotoxicological risk assessment for Roundup herbicide. Rev. Environ. Contam. Toxicol 2000, 167, 35–120. [Google Scholar]
- McKee, M.M.; McAllister, W.; Schofield, M. Chronic Toxicity of Glyphosate (AB-82-036) to Daphnia magna under Flowthrough Test Conditions, Project No. 28742; Analytical biochemistry Laboratories, Submitted by Monsanto Agricultural Project No. on December 27, 1982, for Registration No.; 1982; pp. 524–308, 1982. Accession No. 249160; US EPA Library: Boston, MA, USA, Unpublished study document.
- McAllister, W.; Forbis, A. Acute Toxicity of Technical Glyphosate (AB–78–201) to Daphnia magna. Study Reviewed and Approved 8–30–85 by EEB/HED US EPA, US EPA Library: Boston, MA, USA, 1978; Unpublished study document.
- Cuhra, M.; Traavik, T.; Bøhn, T. Clone- and age-dependent toxicity of a glyphosate commercial formulation and its active ingredient in Daphnia magna. Ecotoxicology 2013, 22, 251–262. [Google Scholar] [CrossRef]
- MSDS N-(PHOSPHONOMETHYL) GLYSINE, MONOISOPROPY L-AMINE SALT 40 WT. SOLUTION IN WATER. Available online: https://www.sigmaaldrich.com/catalog/product/aldrich/338109?lang=zh®ion=CN (accessed on 11 December 2019).
- Cuhra, M. Glyphosate Non-Toxicity: The Genesis of a Scientific Fact. J. Biol. Phys. Chem. 2015, 15, 89–96. [Google Scholar] [CrossRef]
- Cuhra, M.; Bøhn, T. Silent Spring for Academic Dialogue: Biotech Industry Leaves Critique Unopposed. J. Biol. Phys. Chem. 2017, 17, 137–138. [Google Scholar] [CrossRef]
- Felline, S.; Del Coco, L.; Kaleb, S.; Guarnieri, G.; Fraschetti, S.; Terlizzi, A.; Fanizzi, F.; Falace, A. The response of the algae Fucus virsoides (Fucales, Ochrophyta) to Roundup® solution exposure: A metabolomics approach. Environ. Pollut. 2019, 254, 112977. [Google Scholar] [CrossRef]
- Sikorski, Ł.; Baciak, M.; Bęś, A.; Adomas, B. The effects of glyphosate-based herbicide formulations on Lemna minor, a non-target species. Aquat. Toxicol. 2019, 209, 70–80. [Google Scholar] [CrossRef]
- Rzymski, P.; Klimaszyk, P.; Kubacki, T.; Poniedziałek, B. The effect of glyphosate-based herbicide on aquatic organisms–a case study. Limnol. Rev. 2013, 13, 215–220. [Google Scholar] [CrossRef]
- Zhang, L.; Rana, I.; Taioli, E.; Shaffer, R.M.; Sheppard, L. Exposure to glyphosate-based herbicides and Risk for non-hodgkin lymphoma: A meta-analysis and supporting evidence. Mutat. Res. Rev. Mutat. Res. 2019, 781, 186–206. [Google Scholar] [CrossRef]
- Aristilde, L.; Reed, M.L.; Wilkes, R.A.; Youngster, T.; Kukurugya, M.A.; Katz, V.; Sasaki, C.R. Glyphosate-induced specific and widespread perturbations in the metabolome of soil Pseudomonas species. Front. Environ. Sci. 2017, 5, 34. [Google Scholar] [CrossRef]
- Cai, W.; Ji, Y.; Song, X.; Guo, H.; Han, L.; Zhang, F.; Liu, X.; Zhang, H.; Zhu, B.; Xu, M. Effects of glyphosate exposure on sperm concentration in rodents: A systematic review and meta-analysis. Environ. Toxicol. Pharmacol. 2017, 55, 148–155. [Google Scholar] [CrossRef]
- Kubsad, D.; Nilsson, E.E.; King, S.E.; Sadler-Riggleman, I.; Beck, D.; Skinner, M.K. Assessment of glyphosate induced epigenetic transgenerational inheritance of pathologies and sperm epimutations: Generational toxicology. Sci. Rep. 2019, 9, 6372. [Google Scholar] [CrossRef]
- Portier, C.J.; Armstrong, B.K.; Baguley, B.C.; Baur, X.; Belyaev, I.; Bellé, R.; Belpoggi, F.; Biggeri, A.; Bosland, M.C.; Bruzzi, P. Differences in the carcinogenic evaluation of glyphosate between the International Agency for Research on Cancer (IARC) and the European Food Safety Authority (EFSA). J. Epidemiol. Commun. Health 2016, 70, 741–745. [Google Scholar] [CrossRef]
- Efsa. Conclusion on the peer review of the pesticide risk assessment of the active substance glyphosate. EFSA J. 2015, 13, 4302. [Google Scholar] [CrossRef]
- Fritschi, L.; McLaughlin, J.; Sergi, C.M.; Calaf, G.M.; Le Curieux, F.; Forastiere, F.; Kromhout, H.; Egeghy, P. Carcinogenicity of tetrachlorvinphos, parathion, malathion, diazinon, and glyphosate. Red (Lancet) 2015, 114, 1–2. [Google Scholar]
- Major Pesticides Are More Toxic to Human Cells than Their Declared Active Principles. Available online: http://dx.doi.org/10.1155/2014/179691 (accessed on 26 February 2014).
- Defarge, N.; Takacs, E.; Lozano, V.L.; Mesnage, R.; Vendômois, J.S.; Seralini, G.E.; Szekacs, A. Co-formulants in glyphosate-based herbicides disrupt aromatase activity in human cells below toxic levels. Int. J. Environ. Res. Public Health 2016, 13, 264. [Google Scholar] [CrossRef] [PubMed]
- Gunatilake, S.; Seneff, S.; Orlando, L. Glyphosate’s synergistic toxicity in combination with other factors as a cause of chronic kidney disease of unknown origin. Int. J. Environ. Res. Public Health 2019, 16, 2734. [Google Scholar] [CrossRef] [PubMed]
- FAO. The JMPR Practice in Estimating Maximim Residues Levels and Proposing Maximum Residue Limits. Available online: http://www.fao.org/3/X5848E/X5848e07.htm#5.1.3%20information%20received%20on%20good%20agricultural%20practices (accessed on 31 October 2019).
- Cuhra, M.; Bøhn, T.; Cuhra, P. Glyphosate: Too Much of a Good Thing? Front. Environ. Sci. 2016, 4, 1–14. [Google Scholar] [CrossRef]
- Xu, J.; Smith, S.; Smith, G.; Wang, W.; Li, Y. Glyphosate contamination in grains and foods: An overview. Food Control 2019, 106, 106710. [Google Scholar] [CrossRef]
- Heap, I. The international survey of herbicide resistant weeds. Pest Manag. Sci. 2014, 70, 1306–1315. [Google Scholar] [CrossRef]
- Powles, S.B. Evolution in action: Glyphosate-resistant weeds threaten world crops. Outlooks Pest Manag. 2008, 19, 256–259. [Google Scholar] [CrossRef]
- Hawkins, N.J.; Bass, C.; Dixon, A.; Neve, P. The evolutionary origins of pesticide resistance. Biol. Rev. 2019, 94, 135–155. [Google Scholar] [CrossRef]
- Cuhra, M. Evolution of glyphosate resistance: Is the rhizosphere microbiome a key factor? J. Biol. Phys. Chem. 2018, 18, 78–93. [Google Scholar] [CrossRef]
- Duke, S.O.; Rimando, A.M.; Pace, P.F.; Reddy, K.N.; Smeda, R.J. Isoflavone, glyphosate, and aminomethylphosphonic acid levels in seeds of glyphosate-treated, glyphosate-resistant soybean. J. Agric. Food Chem. 2003, 51, 340–344. [Google Scholar] [CrossRef]
- Avila-Vazquez, M.; Difilippo, F.S.; Mac Lean, B.; Maturano, E.; Etchegoyen, A. Environmental exposure to glyphosate and reproductive health impacts in agricultural population of Argentina. J. Environ. Prot. 2018, 9, 241. [Google Scholar] [CrossRef]
- Almeida, V.E.S.d.; Friedrich, K.; Tygel, A.F.; Melgarejo, L.; Carneiro, F.F. Use of genetically modified crops and pesticides in Brazil: Growing hazards. Cienc. Saude Coletiva 2017, 22, 3333–3339. [Google Scholar] [CrossRef] [PubMed]
- Miyazaki, J.; Bauer-Panskus, A.; Bøhn, T.; Reichenbecher, W.; Then, C. Insufficient risk assessment of herbicide tolerant genetically engineered soybeans intended for import into the EU. Environ. Sci. Eur. 2019, (in press). [CrossRef]
- Battaglin, W.; Meyer, M.; Kuivila, K.; Dietze, J. Glyphosate and its degradation product AMPA occur frequently and widely in US soils, surface water, groundwater, and precipitation. JAWRA J. Am. Water Resour. Assoc. 2014, 50, 275–290. [Google Scholar] [CrossRef]
- Bohm, B.; Mariza, G.; Rombaldi, C.V.; Genovese, M.I.; Castilhos, D.; Rodrigues Alves, B.J.; Rumjanek, N.G. Glyphosate effects on yield, nitrogen fixation, and seed quality in glyphosate-resistant soybean. Crop Sci. 2014, 54, 1737–1743. [Google Scholar] [CrossRef]
- Guidelines for the Conduct of Food Safety Assessment of Foods Derived from Recombinant-DNA Plants. Available online: http://bch.cbd.int/database/record.shtml?documentid=42122 (accessed on 13 May 2009).
- Testbiotech. High Levels of Residues from Spraying with Glyphosate Found in Soybeans in Argentina; Testbiotech: Munich, Germany, 2013; pp. 1–14. [Google Scholar]
- Effects of Herbicide Glyphosate and Glyphosate-Based Formulations on Aquatic Ecosystems. Available online: https://www.intechopen.com/books/herbicides-and-environment/effects-of-herbicide-glyphosate-and-glyphosate-based-formulations-on-aquatic-ecosystems (accessed on 7 April 2010).
- Mesnage, R.; Benbrook, C.; Antoniou, M.N. Insight into the confusion over surfactant co-formulants in glyphosate-based herbicides. Food Chem. Toxicol. 2019, 128, 137–145. [Google Scholar] [CrossRef]
- EU Commission. Request to Consider the Impact of Glyphosate Residues in Feed on ANIMAL health; Health and Food Safety Directorate-General: Brussels, Belgium, 2016. [Google Scholar]
- EFSA. Review of the existing maximum residue levels for glyphosate according to Article 12 of Regulation (EC) No 396/2005. EFSA J. 2018, 16, e05263. [Google Scholar]
- Bøhn, T. Criticism of EFSA’s scientific opinion on combinatorial effects of ‘stacked’ GM plants. Food Chem. Toxicol. 2018, 111, 268–274. [Google Scholar] [CrossRef]
- Cuhra, M. Review of GMO safety assessment studies: Glyphosate residues in Roundup Ready crops is an ignored issue. Environ. Sci. Eur. 2015, 27, 1–14. [Google Scholar] [CrossRef]
- Viljoen, C. Letter to the editor. Food Chem. Toxicol. 2013, 59, 809–810. [Google Scholar] [CrossRef]
- Calas, A.-G.; Richard, O.; Même, S.; Beloeil, J.-C.; Doan, B.-T.; Gefflaut, T.; Même, W.; Crusio, W.E.; Pichon, J.; Montécot, C. Chronic exposure to glufosinate-ammonium induces spatial memory impairments, hippocampal MRI modifications and glutamine synthetase activation in mice. NeuroToxicology 2008, 29, 740–747. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Conclusion regarding the peer review of the pesticide risk assessment of the active substance glufosinate: Conclusion regarding the peer review of the pesticide risk assessment of the active substance glufosinate. EFSA J. 2005, 3, 27r. [Google Scholar] [CrossRef]
- Nielsen, K.M. Biosafety data as confidential business information. PLoS Biol. 2013, 11, 1–6. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bøhn, T.; Millstone, E. The Introduction of Thousands of Tonnes of Glyphosate in the food Chain—An Evaluation of Glyphosate Tolerant Soybeans. Foods 2019, 8, 669. https://doi.org/10.3390/foods8120669
Bøhn T, Millstone E. The Introduction of Thousands of Tonnes of Glyphosate in the food Chain—An Evaluation of Glyphosate Tolerant Soybeans. Foods. 2019; 8(12):669. https://doi.org/10.3390/foods8120669
Chicago/Turabian StyleBøhn, Thomas, and Erik Millstone. 2019. "The Introduction of Thousands of Tonnes of Glyphosate in the food Chain—An Evaluation of Glyphosate Tolerant Soybeans" Foods 8, no. 12: 669. https://doi.org/10.3390/foods8120669
APA StyleBøhn, T., & Millstone, E. (2019). The Introduction of Thousands of Tonnes of Glyphosate in the food Chain—An Evaluation of Glyphosate Tolerant Soybeans. Foods, 8(12), 669. https://doi.org/10.3390/foods8120669





