Punica granatum and Citrus spp. Extract Mix Affects Spoilage Microorganisms Growth Rate in Vacuum-Packaged Cooked Sausages Made from Pork Meat, Emmer Wheat (Triticum dicoccum Schübler), Almond (Prunus dulcis Mill.) and Hazelnut (Corylus avellana L.)
Abstract
1. Introduction
2. Materials and Methods
2.1. Sausage Production and Experimental Design
2.2. Chemical and Physical—Chemical Determinations
2.3. Microbiological Analysis of Sausages
2.4. Sensory Analysis
2.5. Statistical Analysis
3. Results and Discussion
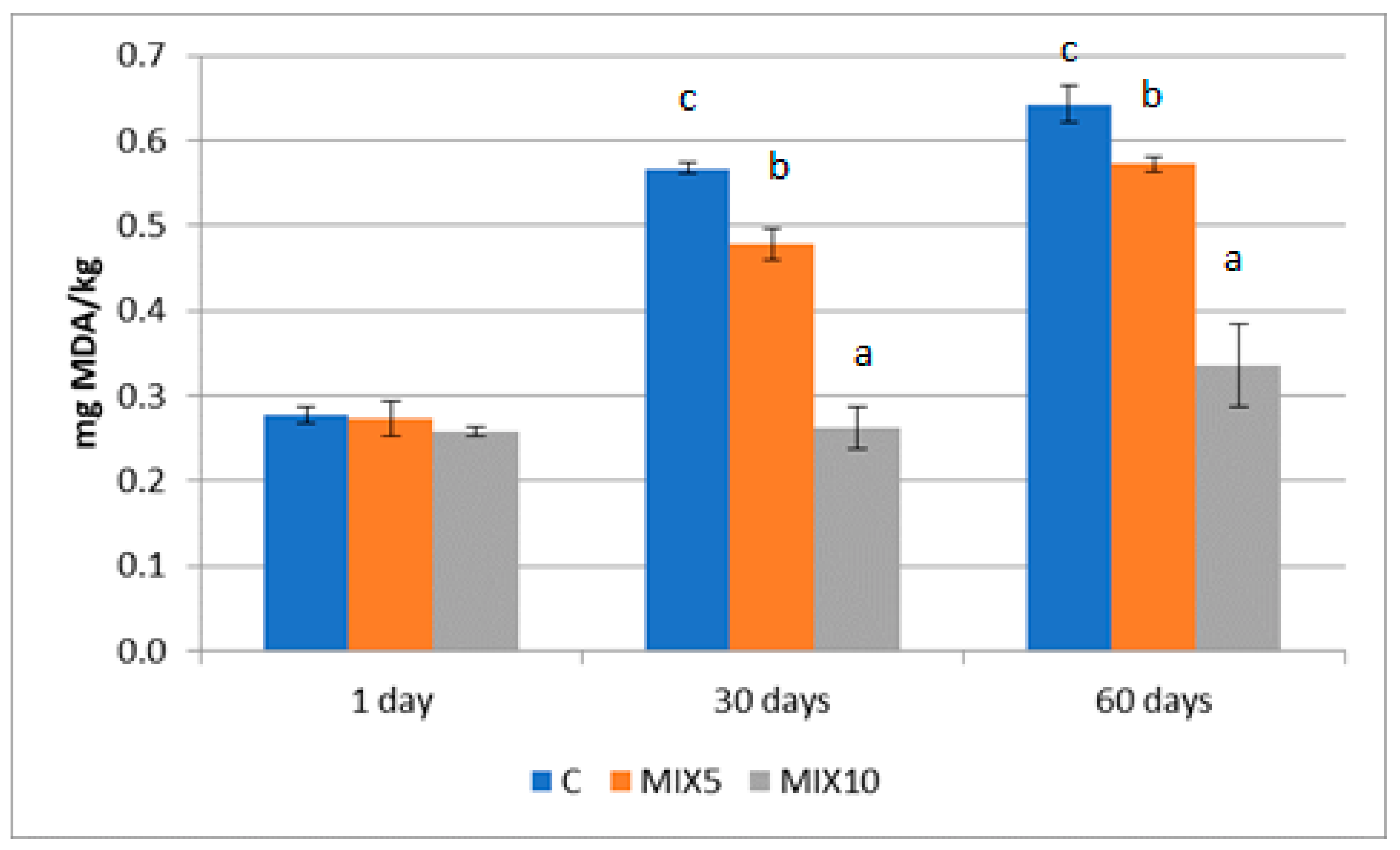
3.1. Chemical Composition and Physical—Chemical Determinations
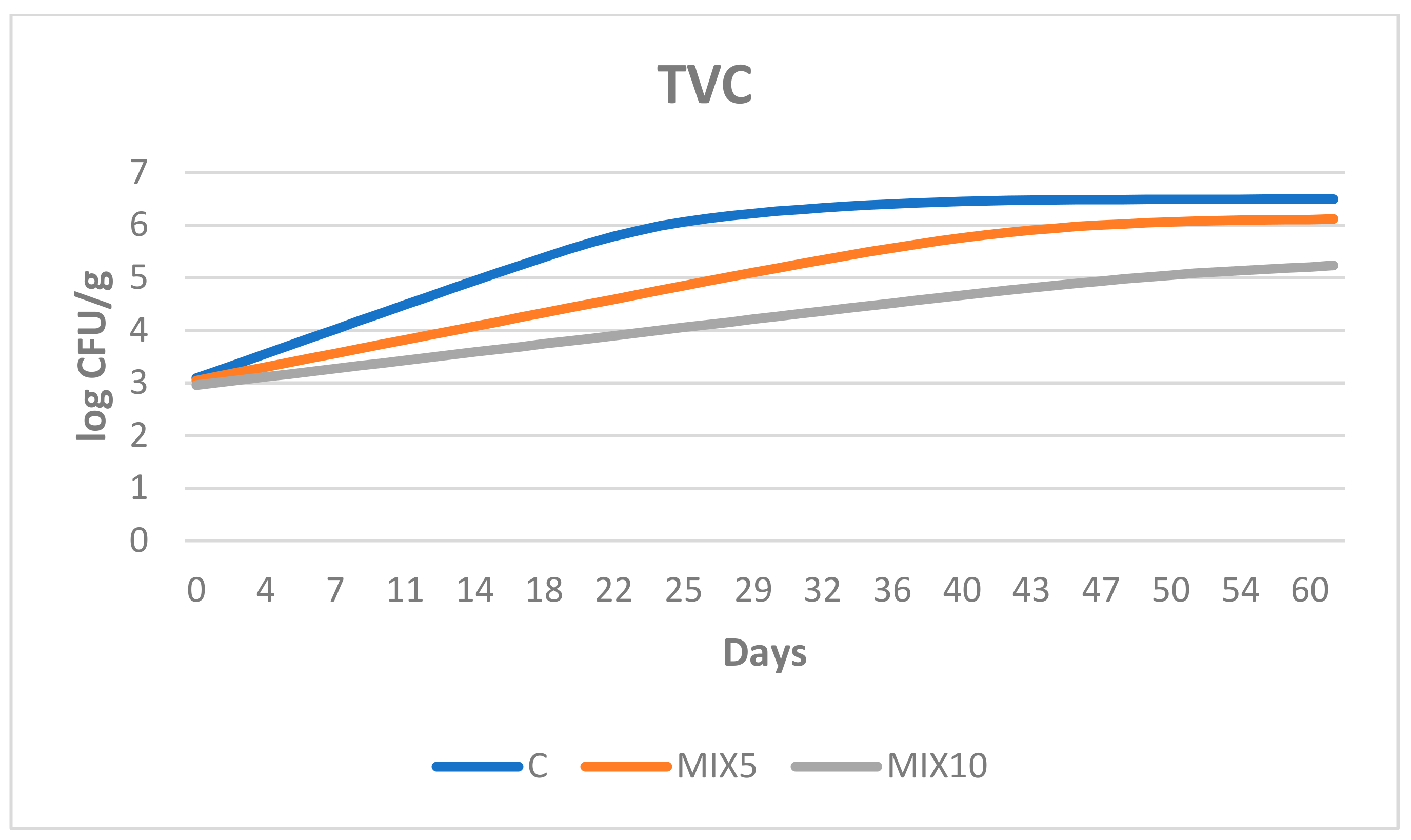
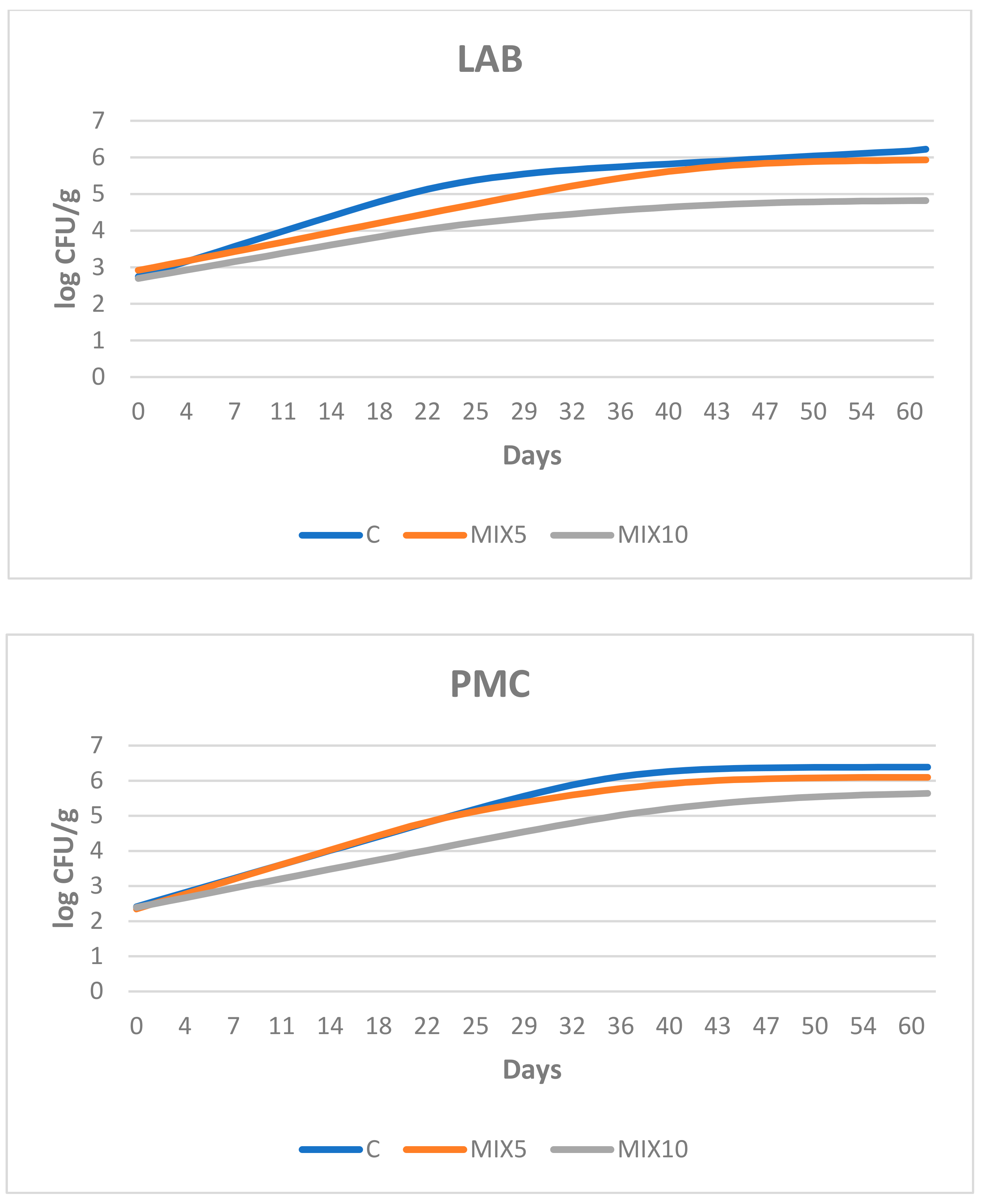
3.2. Microbiological Growth in the Sausages
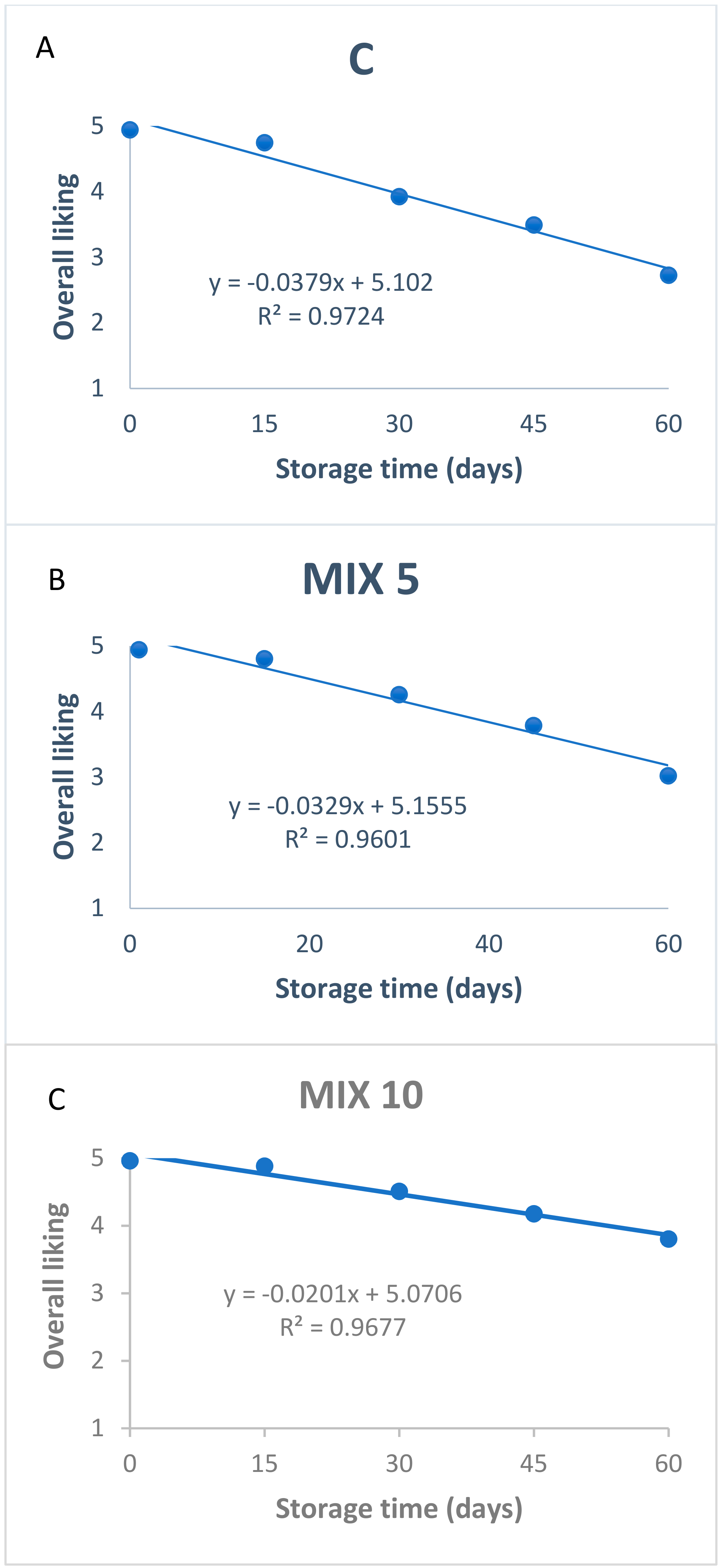
3.3. Sensory Analyses for Shelf-Life
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ayo, J.; Carballo, J.; Solas, M.T.; Jiménez-Colmenero, F. Physicochemical and sensory properties of healthier frankfurters as affected by walnut and fat content. Food Chem. 2008, 107, 1547–1552. [Google Scholar] [CrossRef]
- Jiménez-Colmenero, F.; Sánchez-Muniz, F.J.; Olmedilla-Alonso, B. Design and development of meat-based functional foods with walnut: Technological, nutritional and health impact. Food Chem. 2010, 123, 959–967. [Google Scholar] [CrossRef]
- Mehta, N.; Ahlawat, S.S.; Sharma, D.P.; Dabur, R.S. Novel trends in development of dietary fiber rich meat products—a critical review. J. Food Sci. Technol. 2015, 52, 633–647. [Google Scholar] [CrossRef] [PubMed]
- Weiss, J.; Gibis, M.; Schuh, V.; Salminen, H. Advances in ingredient and processing systems for meat and meat products. Meat Sci. 2010, 86, 196–213. [Google Scholar] [CrossRef]
- Conroy, P.M.; O’Sullivan, M.G.; Hamill, R.M.; Kerry, J.P. Impact on the physical and sensory properties of salt-and fat-reduced traditional Irish breakfast sausages on various age cohorts acceptance. Meat Sci. 2018, 143, 190–198. [Google Scholar] [CrossRef]
- Marcos, C.; Viegas, C.; de Almeida, A.M.; Guerra, M.M. Portuguese traditional sausages: Different types, nutritional composition, and novel trends. J. Ethnic Foods 2016, 3, 51–60. [Google Scholar] [CrossRef]
- Tobin, B.D.; O’Sullivan, M.G.; Hamill, R.M.; Kerry, J.P. The impact of salt and fat level variation on the physiochemical properties and sensory quality of pork breakfast sausages. Meat Sci. 2013, 93, 145–152. [Google Scholar] [CrossRef]
- Ranucci, D.; Miraglia, D.; Branciari, R.; Morganti, G.; Roila, R.; Zhou, K.; Jang, H.; Braconi, P. Frankfurters made with pork meat, emmer wheat (Triticum dicoccum Schübler) and almonds nut (Prunus dulcis Mill.): Evaluation during storage of a novel food from an ancient recipe. Meat Sci. 2018, 145, 440–446. [Google Scholar] [CrossRef]
- Fernández-López, J.; Lucas-González, R.; Viuda-Martos, M.; Sayas-Barberá, E.; Navarro, C.; Haros, C.M.; Pérez-Álvarez, J.A. Chia (Salvia hispanica L.) products as ingredients for reformulating frankfurters: Effects on quality properties and shelf-life. Meat Sci. 2019, 156, 139–145. [Google Scholar] [CrossRef]
- Nayeem, M.; Chauhan, K.; Khan, M.; Siddiqui, M.; Sidduqui, H. Development and shelf life studies of buffalo meat sausages incorporated with foxtail millet (Setaria italica). Int. J. Chem. Stud. 2017, 5, 648–654. [Google Scholar]
- Agregán, R.; Barba, F.J.; Gavahian, M.; Franco, D.; Khaneghah, A.M.; Carballo, J.; Ferreira, I.C.F.R.; da Silva Barretto, A.C.; Lorenzo, J.M. Fucus vesiculosus extracts as natural antioxidants for improvement of physicochemical properties and shelf life of pork patties formulated with oleogels. J. Sci. Food Agric. 2019, 99, 4561–4570. [Google Scholar] [CrossRef] [PubMed]
- Roila, R.; Branciari, R.; Staccini, B.; Ranucci, D.; Miraglia, D.; Altissimi, M.S.; Mercuri, M.L.; Haouet, N.M. Contribution of vegetables and cured meat to dietary nitrate and nitrite intake in Italian population: Safe level for cured meat and controversial role of vegetables. Ital. J. Food Saf. 2018, 7, 7692. [Google Scholar] [CrossRef] [PubMed]
- Bakels, C.; Jacomet, S. Access to luxury foods in Central Europe during the Roman period: The archaeobotanical evidence. World Archaeol. 2003, 34, 542–557. [Google Scholar] [CrossRef]
- Pagnoux, C.; Celant, A.; Coubray, S.; Fiorentino, G.; Zech-Matterne, V. The introduction of Citrus to Italy, with reference to the identification problems of seed remains. Veg. Hist. Archaeobot. 2013, 22, 421–438. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis of AOAC International, 17th ed.; AOAC International: Gathersberg, MD, USA, 2000. [Google Scholar]
- Corradini, C.; Canali, G.; Cogliandro, E.; Nicoletti, I. Separation of alditols of interest in food products by high-performance anion-exchange chromatography with pulsed amperometric detection. J. Chromatogr. A 1997, 791, 343–349. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis of AOAC International, 16th ed.; AOAC International: Gathersberg, MD, USA, 1995. [Google Scholar]
- Tarladgis, B.G.; Watts, B.M.; Yonathan, M. Distillation method for the determination of malonaldehyde ın rancid foods. J. Am. Oil Chem. Soc. 1960, 37, 44–48. [Google Scholar] [CrossRef]
- ISO 4833-1. Microbiology of the Food Chain—Horizontal Method for the Enumeration of Microorganisms—Part 1: Colony Count at 30 Degrees C by the Pour Plate Technique; International Organization for Standardization: Geneva, Switzerland, 2013. [Google Scholar]
- ISO 21528-2. Microbiology of the food chain—Horizontal method for the detection and enumeration of Enterobacteriaceae—Part 2: Colony-count technique; International Organization for Standardization: Geneva, Switzerland, 2017. [Google Scholar]
- ISO 17410. Microbiology of food and animal feeding stuffs—Horizontal method for the enumeration of psychrotrophic microorganisms; International Organization for Standardization: Geneva, Switzerland, 2001. [Google Scholar]
- ISO 6579-1. Microbiology of the Food Chain—Horizontal Method for the Detection, Enumeration and Serotyping of Salmonella—Part 1: Detection of Salmonella Spp; International Organization for Standardization: Geneva, Switzerland, 2017. [Google Scholar]
- ISO 11290-1. Microbiology of the food chain—Horizontal method for the detection and enumeration of Listeria monocytogenes and of Listeria spp.—Part 1: Detection method; International Organization for Standardization: Geneva, Switzerland, 2017. [Google Scholar]
- Branciari, R.; Ranucci, D.; Urbani, E.; Valiani, A.; Trabalza-Marinucci, M.; Dal Bosco, A.; Franceschini, R. Freshwater fish burgers made from four different fish species as a valuable strategy appreciated by consumers for introducing EPA and DHA into a human diet. J. Aquat. Food Prod. Technol. 2017, 26, 686–694. [Google Scholar] [CrossRef]
- Aaslyng, M.D.; Vestergaard, C.; Koch, A.G. The effect of salt reduction on sensory quality and microbial growth in hotdog sausages, bacon, ham and salami. Meat Sci. 2014, 96, 47–55. [Google Scholar] [CrossRef]
- Roila, R.; Valiani, A.; Ranucci, D.; Ortenzi, R.; Servili, M.; Veneziani, G.; Branciari, R. Antimicrobial efficacy of a polyphenolic extract from olive oil by-product against “Fior di latte” cheese spoilage bacteria. Int. J. Food Microbiol. 2019, 295, 49–53. [Google Scholar] [CrossRef]
- Bovill, R.; Bew, J.; Cook, N.; D’agostino, M.; Wilkinson, N.; Baranyi, J. Predictions of growth for Listeria monocytogenes and Salmonella during fluctuating temperature. Int. J. Food Microbiol. 2000, 59, 157–165. [Google Scholar] [CrossRef]
- García, M.R.; Vilas, C.; Herrera, J.R.; Bernárdez, M.; Balsa-Canto, E.; Alonso, A.A. Quality and shelf-life prediction for retail fresh hake (Merluccius merluccius). Int. J. Food Microbiol. 2015, 208, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Ranucci, D.; Miraglia, D.; Trabalza-Marinucci, M.; Acuti, G.; Codini, M.; Ceccarini, M.R.; Forte, C.; Branciari, R. Dietary effects of oregano (Origanum vulgaris L.) plant or sweet chestnut (Castanea sativa Mill.) wood extracts on microbiological, chemico-physical characteristics and lipid oxidation of cooked ham during storage. Ital. J. Food Saf. 2015, 4, 5497. [Google Scholar]
- Simion, A.M.C.; Vizireanu, C.; Alexe, P.; Franco, I.; Carballo, J. Effect of the use of selected starter cultures on some quality, safety and sensorial properties of Dacia sausage, a traditional Romanian dry-sausage variety. Food Control 2014, 35, 123–131. [Google Scholar] [CrossRef]
- Vermeiren, L.; Devlieghere, F.; Debevere, J. Evaluation of meat born lactic acid bacteria as protective cultures for the biopreservation of cooked meat products. Int. J. Food Microbiol. 2004, 96, 149–164. [Google Scholar] [CrossRef]
- Fonseca, S.; Cachaldora, A.; Gómez, M.; Franco, I.; Carballo, J. Effect of different autochthonous starter cultures on the volatile compounds profile and sensory properties of Galician chorizo, a traditional Spanish dry fermented sausage. Food Control 2013, 33, 6–14. [Google Scholar] [CrossRef]
- Ranucci, D.; Loschi, A.R.; Miraglia, D.; Stocchi, R.; Branciari, R.; Rea, S. Effect of selected starter cultures on physical, chemical and microbiological characteristics and biogenic amine content in Protected Geographical Indication Ciauscolo salami. Ital. J. Food Saf. 2016, 5, 5568. [Google Scholar]
- Gil, M.I.; Tomás-Barberán, F.A.; Hess-Pierce, B.; Holcroft, D.M.; Kader, A.A. Antioxidant activity of pomegranate juice and its relationship with phenolic composition and processing. J. Agric. Food Chem. 2000, 48, 4581–4589. [Google Scholar] [CrossRef]
- Madrigal-Carballo, S.; Rodriguez, G.; Krueger, C.G.; Dreher, M.; Reed, J.D. Pomegranate (Punica granatum) supplements: Authenticity, antioxidant and polyphenol composition. J. Funct. Foods 2009, 1, 324–329. [Google Scholar] [CrossRef]
- Naveena, B.M.; Sen, A.R.; Vaithiyanathan, S.; Babji, Y.; Kondaiah, N. Comparative efficacy of pomegranate juice, pomegranate rind powder extract and BHT as antioxidants in cooked chicken patties. Meat Sci. 2008, 80, 1304–1308. [Google Scholar] [CrossRef]
- Devatkal, S.K.; Narsaiah, K.; Borah, A. Anti-oxidant effect of extracts of kinnow rind, pomegranate rind and seed powders in cooked goat meat patties. Meat Sci. 2010, 85, 155–159. [Google Scholar] [CrossRef]
- Kanatt, S.R.; Chander, R.; Sharma, A. Antioxidant and antimicrobial activity of pomegranate peel extract improves the shelf life of chicken products. Int. J. Food Sci. Technol. 2010, 45, 216–222. [Google Scholar] [CrossRef]
- Bocco, A.; Cuvelier, M.E.; Richard, H.; Berset, C. Antioxidant activity and phenolic composition of citrus peel and seed extracts. J. Agric. Food Chem. 1998, 46, 2123–2129. [Google Scholar] [CrossRef]
- Fernández-López, J.; Fernández-Ginés, J.M.; Aleson-Carbonell, L.; Sendra, E.; Sayas-Barberá, E.; Pérez-Alvarez, J.A. Application of functional citrus by-products to meat products. Trends Food Sci. Technol. 2004, 15, 176–185. [Google Scholar] [CrossRef]
- Yang, J.; Liu, R.H.; Halim, L. Antioxidant and antiproliferative activities of common edible nut seeds. LWT-Food Sci. Technol. 2009, 42, 1–8. [Google Scholar] [CrossRef]
- Gokoglu, N.; Yerlikaya, P.; Uran, H.; Topuz, O.K. The effect of modified atmosphere packaging on the quality and shelf life of frankfurther type-sausages. J. Food Qual. 2010, 33, 367–380. [Google Scholar] [CrossRef]
- Firuzi, M.R.; Niakousari, M.; Eskandari, M.H.; Keramat, M.; Gahruie, H.H.; Khaneghah, A.M. Incorporation of pomegranate juice concentrate and pomegranate rind powder extract to improve the oxidative stability of frankfurter during refrigerated storage. LWT-Food Sci. Technol. 2019, 102, 237–245. [Google Scholar] [CrossRef]
- Devatkal, S.K.; Thorat, P.; Manjunatha, M. Effect of vacuum packaging and pomegranate peel extract on quality aspects of ground goat meat and nuggets. J. Food Sci. Technol. 2014, 51, 2685–2691. [Google Scholar] [CrossRef]
- Qin, Y.Y.; Zhang, Z.H.; Li, L.; Xiong, W.; Shi, J.Y.; Zhao, T.R.; Fan, J. Antioxidant effect of pomegranate rind powder extract, pomegranate juice, and pomegranate seed powder extract as antioxidants in raw ground pork meat. Food Sci. Biotechnol. 2013, 22, 1063–1069. [Google Scholar] [CrossRef]
- Bouarab-Chibane, L.; Ouled-Bouhedda, B.; Leonard, L.; Gemelas, L.; Bouajila, J.; Ferhout, H.; Cottaz, A.; Joly, C.; Degraeve, P.; Oulahal, N. Preservation of fresh ground beef patties using plant extracts combined with a modified atmosphere packaging. Eur. Food Res. Technol. 2017, 243, 1997–2009. [Google Scholar] [CrossRef]
- Basiri, S.; Shekarforoush, S.S.; Aminlari, M.; Akbari, S. The effect of pomegranate peel extract (PPE) on the polyphenol oxidase (PPO) and quality of Pacific white shrimp (Litopenaeus vannamei) during refrigerated storage. LWT-Food Sci. Technol. 2015, 60, 1025–1033. [Google Scholar] [CrossRef]
- Mexis, S.F.; Chouliara, E.; Kontominas, M.G. Shelf life extension of ground chicken meat using an oxygen absorber and a citrus extract. LWT-Food Sci. Technol. 2012, 49, 21–27. [Google Scholar] [CrossRef]
- Viuda-Martos, M.; Ruiz-Navajas, Y.; Fernández-López, J.; Pérez-Álvarez, J.A. Effect of added citrus fibre and spice essential oils on quality characteristics and shelf-life of mortadella. Meat Sci. 2010, 85, 568–576. [Google Scholar] [CrossRef] [PubMed]
- Alsaggaf, M.S.; Moussa, S.H.; Tayel, A.A. Application of fungal chitosan incorporated with pomegranate peel extract as edible coating for microbiological, chemical and sensorial quality enhancement of Nile tilapia fillets. Int. J. Biol. Macromol. 2017, 99, 499–505. [Google Scholar] [CrossRef] [PubMed]
- Tsiraki, M.I.; Yehia, H.M.; Elobeid, T.; Osaili, T.; Sakkas, H.; Savvaidis, I.N. Viability of and Escherichia coli O157: H7 and Listeria monocytogenes in a delicatessen appetizer (yogurt-based) salad as affected by citrus extract (Citrox©) and storage temperature. Food Microbiol. 2018, 69, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Saleh, A.; Morshdy, A.E.M.; Abd-El-Salam, E.H.; Hussein, M.A.; Elewa, E.S.; Mahmoud, A.F.A. Effect of pomegranate peel powder on the hygienic quality of beef sausage. J. Microbiol. Biotech. Food Sci. 2017, 6, 1300. [Google Scholar] [CrossRef]
- Houben, J.H.; Eckenhausen, F. Surface pasteurization of vacuum-sealed precooked ready-to-eat meat products. J. Food Prot. 2006, 69, 459–468. [Google Scholar] [CrossRef]
- Commission Regulation (EC). N. 2073/2005 of 15 November 2005 on Microbiological Criteria for Foodstuffs. Official Journal of the European Union L 338/1. Available online: https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=CELEX%3A32005R2073 (accessed on 10 November 2019).
- Al-Zoreki, N.S. Antimicrobial activity of pomegranate (Punica granatum L.) fruit peels. Int. J. Food Microbiol. 2009, 134, 244–248. [Google Scholar] [CrossRef]
- Shan, B.; Cai, Y.Z.; Brooks, J.D.; Corke, H. Antibacterial and antioxidant effects of five spice and herb extracts as natural preservatives of raw pork. J. Sci. Food Agric. 2009, 89, 1879–1885. [Google Scholar] [CrossRef]
- Hayrapetyan, H.; Hazeleger, W.C.; Beumer, R.R. Inhibition of Listeria monocytogenes by pomegranate (Punica granatum) peel extract in meat paté at different temperatures. Food Control 2012, 23, 66–72. [Google Scholar] [CrossRef]
- Li, G.; Xu, Y.; Wang, X.; Zhang, B.; Shi, C.; Zhang, W.; Xia, X. Tannin-rich fraction from pomegranate rind damages membrane of Listeria monocytogenes. Foodborne Path. Dis. 2014, 11, 313–319. [Google Scholar] [CrossRef]
- Kang, J.H.; Song, K.B. Effect of pomegranate (Punica granatum) pomace extract as a washing agent on the inactivation of Listeria monocytogenes inoculated on fresh produce. Int. J. Food Sci. Technol. 2017, 52, 2295–2302. [Google Scholar] [CrossRef]
- Casquete, R.; Castro, S.M.; Martín, A.; Ruiz-Moyano, S.; Saraiva, J.A.; Córdoba, M.G.; Teixeira, P. Evaluation of the effect of high pressure on total phenolic content, antioxidant and antimicrobial activity of citrus peels. Innov. Food Sci. Emerg. Technol. 2015, 31, 37–44. [Google Scholar] [CrossRef]
- Fernandez-Lopez, J.; Zhi, N.; Aleson-Carbonell, L.; Pérez-Alvarez, J.A.; Kuri, V. Antioxidant and antibacterial activities of natural extracts: Application in beef meatballs. Meat Sci. 2005, 69, 371–380. [Google Scholar] [CrossRef] [PubMed]
- Giménez, A.; Ares, F.; Ares, G. Sensory shelf-life estimation: A review of current methodological approaches. Food Res. Int. 2012, 49, 311–325. [Google Scholar] [CrossRef]
- Heo, C.; Kim, H.W.; Choi, Y.S.; Kim, C.J.; Paik, H.D. Shelf-life estimation of frankfurter sausage containing dietary fiber from rice bran using predictive modeling. Korean J. Food Sci. Anim. Resour. 2009, 29, 47–54. [Google Scholar] [CrossRef]
- Tovunac, I.; Galić, K.; Prpić, T.; Jurić, S. Effect of packaging conditions on the shelf-life of chicken frankfurters with and without lactate addition. Food Sci. Technol. Int. 2011, 17, 167–175. [Google Scholar] [CrossRef]
- Cachaldora, A.; García, G.; Lorenzo, J.M.; García-Fontán, M.C. Effect of modified atmosphere and vacuum packaging on some quality characteristics and the shelf-life of “morcilla”, a typical cooked blood sausage. Meat Sci. 2013, 93, 220–225. [Google Scholar] [CrossRef]
- Balamurugan, S.; Inmanee, P.; Souza, J.D.; Strange, P.; Pirak, T.; Barbut, S. Effects of High-Pressure Processing and Hot Water Pasteurization of Cooked Sausages on Inactivation of Inoculated Listeria monocytogenes, Natural Populations of Lactic Acid Bacteria, Pseudomonas spp., and Coliforms and Their Recovery during Storage at 4 and 10 °C. J. Food Prot. 2018, 81, 1245–1251. [Google Scholar]
| % | C | MIX5 | MIX10 | SEM |
|---|---|---|---|---|
| Moisture | 51.08 | 51.21 | 51.15 | 0.022 |
| Protein | 14.55 | 14.96 | 14.56 | 0.080 |
| Lipid | 17.25 | 16.96 | 17.04 | 0.049 |
| Carbohydrate | 15.02 | 14.79 | 15.11 | 0.055 |
| Fibre | 7.62 | 7.56 | 7.64 | 0.014 |
| Ash | 2.10 | 2.07 | 2.14 | 0.012 |
| NaCl | 1.51 | 1.48 | 1.52 | 0.008 |
| n | Storage Time (Days) | SEM | p-Value | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 15 | 30 | 45 | 60 | T | ST | T × ST | ||||
| pH | C | 10 | 6.61 w | 6.23 x | 6.18 x | 6.08 ya | 5.60 za | 0.030 | <0.001 | <0.001 | <0.001 |
| MIX5 | 10 | 6.64 w | 6.20 x | 6.17 x | 6.12 yab | 6.02 yb | |||||
| MIX10 | 10 | 6.65 w | 6.27 x | 6.21 x | 6.20 xb | 6.14 yc | |||||
| aw | C | 10 | 0.966 | 0.964 | 0.965 | 0.965 | 0.965 | 0.001 | 0.063 | 0.072 | 0.068 |
| MIX5 | 10 | 0.966 | 0.966 | 0.966 | 0.967 | 0.966 | |||||
| MIX10 | 10 | 0.970 | 0.966 | 0.967 | 0.966 | 0.965 | |||||
| Storage Time (Days) | p-Value | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 15 | 30 | 45 | 60 | SEM | T | ST | T × ST | ||
| TVC | C | 3.05 v | 5.19 wc | 5.80 xc | 6.15 yb | 7.18 zc | 0.068 | <0.001 | <0.001 | <0.001 |
| MIX5 | 2.99 v | 4.24 wb | 5.11 xb | 5.98 yb | 6.11 yb | |||||
| MIX10 | 2.85 v | 3.83 wa | 4.17 xa | 4.87 ya | 5.24 za | |||||
| Lactobacillus spp. count | C | 2.54 v | 4.88 wb | 5.12 wb | 5.58 xb | 6.85 yc | 0.089 | <0.001 | <0.001 | <0.001 |
| MIX5 | 2.86 v | 4.11 wa | 4.97 xb | 5.82 yb | 5.93 yb | |||||
| MIX10 | 2.58 v | 3.90 wa | 4.15 wa | 4.60 xa | 5.04 ya | |||||
| PMC | C | 2.26 v | 4.47 wb | 5.45 xc | 5.97 yb | 6.75 zc | 0.101 | <0.001 | <0.001 | <0.001 |
| MIX5 | 2.26 v | 4.31 wb | 5.16 xb | 6.04 yb | 6.27 yb | |||||
| MIX10 | 2.20 v | 3.89 wa | 4.04 wa | 4.99 xa | 5.33 ya | |||||
| Percentage | C (Days) | MIX5 (Days) | MIX10 (Days) |
|---|---|---|---|
| 1% | 7.595 | 8.951 | 11.236 |
| 5% | 15.000 | 17.390 | 21.464 |
| 10% | 20.259 | 23.318 | 28.566 |
| 1st Quartile 25% | 30.815 | 35.110 | 42.567 |
| Median 50% | 44.487 | 50.241 | 60.357 |
| 3rd Quartile 75% | 59.419 | 66.637 | 79.480 |
| 90% | 73.441 | 81.942 | 97.221 |
| 95% | 81.971 | 91.217 | 107.930 |
| 99% | 98.092 | 108.684 | 128.026 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ranucci, D.; Roila, R.; Andoni, E.; Braconi, P.; Branciari, R. Punica granatum and Citrus spp. Extract Mix Affects Spoilage Microorganisms Growth Rate in Vacuum-Packaged Cooked Sausages Made from Pork Meat, Emmer Wheat (Triticum dicoccum Schübler), Almond (Prunus dulcis Mill.) and Hazelnut (Corylus avellana L.). Foods 2019, 8, 664. https://doi.org/10.3390/foods8120664
Ranucci D, Roila R, Andoni E, Braconi P, Branciari R. Punica granatum and Citrus spp. Extract Mix Affects Spoilage Microorganisms Growth Rate in Vacuum-Packaged Cooked Sausages Made from Pork Meat, Emmer Wheat (Triticum dicoccum Schübler), Almond (Prunus dulcis Mill.) and Hazelnut (Corylus avellana L.). Foods. 2019; 8(12):664. https://doi.org/10.3390/foods8120664
Chicago/Turabian StyleRanucci, David, Rossana Roila, Egon Andoni, Paolo Braconi, and Raffaella Branciari. 2019. "Punica granatum and Citrus spp. Extract Mix Affects Spoilage Microorganisms Growth Rate in Vacuum-Packaged Cooked Sausages Made from Pork Meat, Emmer Wheat (Triticum dicoccum Schübler), Almond (Prunus dulcis Mill.) and Hazelnut (Corylus avellana L.)" Foods 8, no. 12: 664. https://doi.org/10.3390/foods8120664
APA StyleRanucci, D., Roila, R., Andoni, E., Braconi, P., & Branciari, R. (2019). Punica granatum and Citrus spp. Extract Mix Affects Spoilage Microorganisms Growth Rate in Vacuum-Packaged Cooked Sausages Made from Pork Meat, Emmer Wheat (Triticum dicoccum Schübler), Almond (Prunus dulcis Mill.) and Hazelnut (Corylus avellana L.). Foods, 8(12), 664. https://doi.org/10.3390/foods8120664






