Seasonal Variations in the Chemical Composition of Liangshan Olive Leaves and Their Antioxidant and Anticancer Activities
Abstract
1. Introduction
2. Material and Methods
2.1. Materials and Chemicals
2.2. Sample Preparation
2.3. Determination of Chemical Composition
2.3.1. Total Phenolic Content (TPC)
2.3.2. Total Flavonoid Content (TFC)
2.3.3. Free Amino Acid Content (FAAC)
2.3.4. Soluble Sugar Content (SSC)
2.3.5. Soluble Protein Content (SPC)
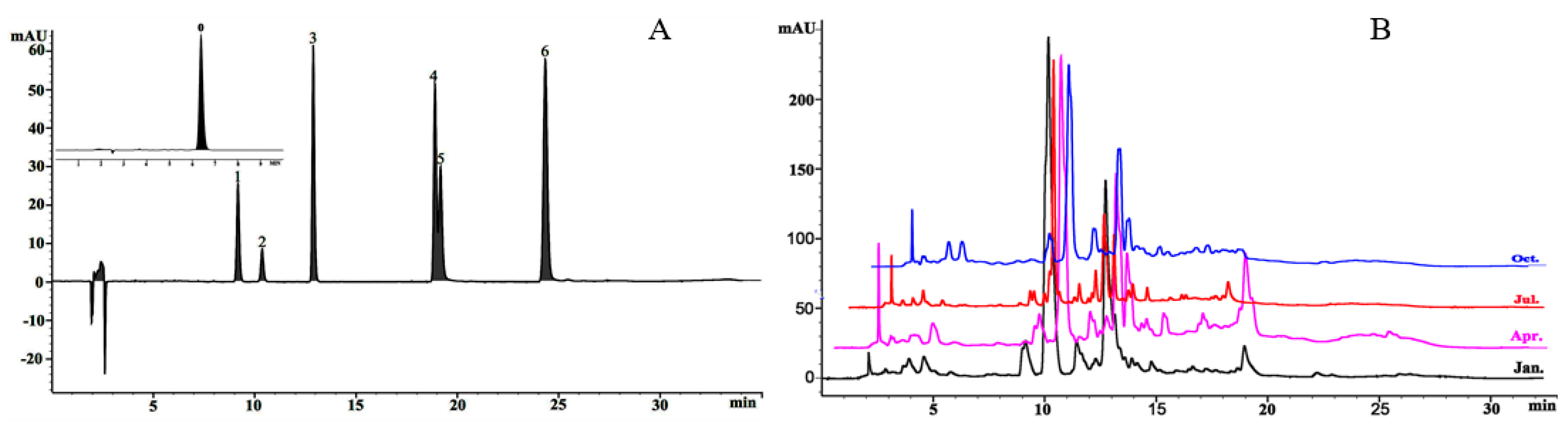
2.4. HPLC Analysis of Phenolic Compounds
2.5. Antioxidant Activity Assays
2.5.1. Superoxide Radical-Scavenging Activity
2.5.2. DPPH Radical-Scavenging Activity (DPPH)
2.5.3. Reducing Power
2.6. Anticancer Activity Assays
2.6.1. Cell Culture
2.6.2. Cell Viability
2.6.3. Mitochondrial Membrane Potential (∆Ψm)
2.6.4. Caspase-3 and Caspase-9 Activity
2.7. Statistical Analysis
3. Results and Discussion
3.1. Seasonal Variations in Chemical Composition
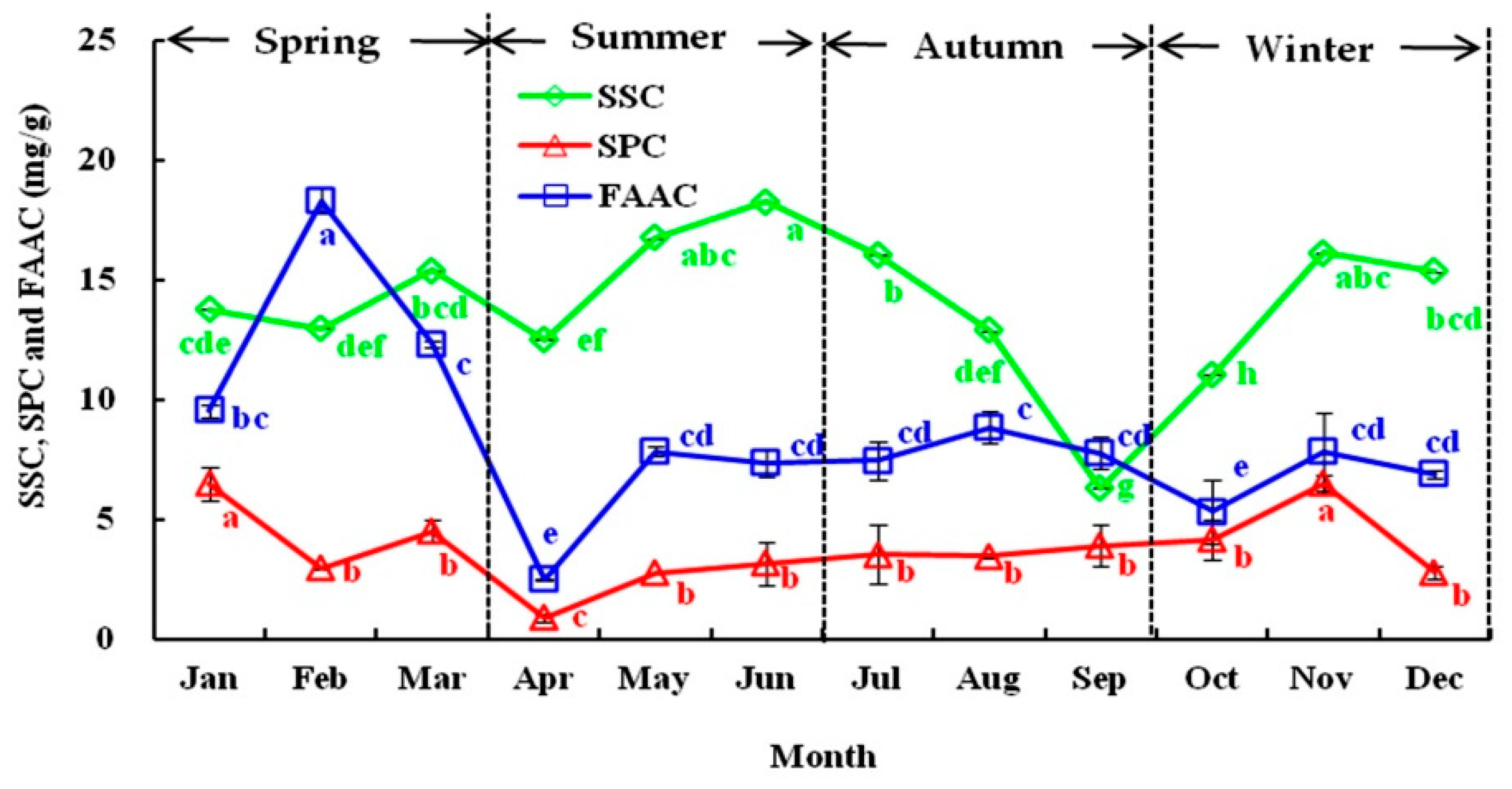
3.1.1. Seasonal Variations in SSC, SPC, and FAAC
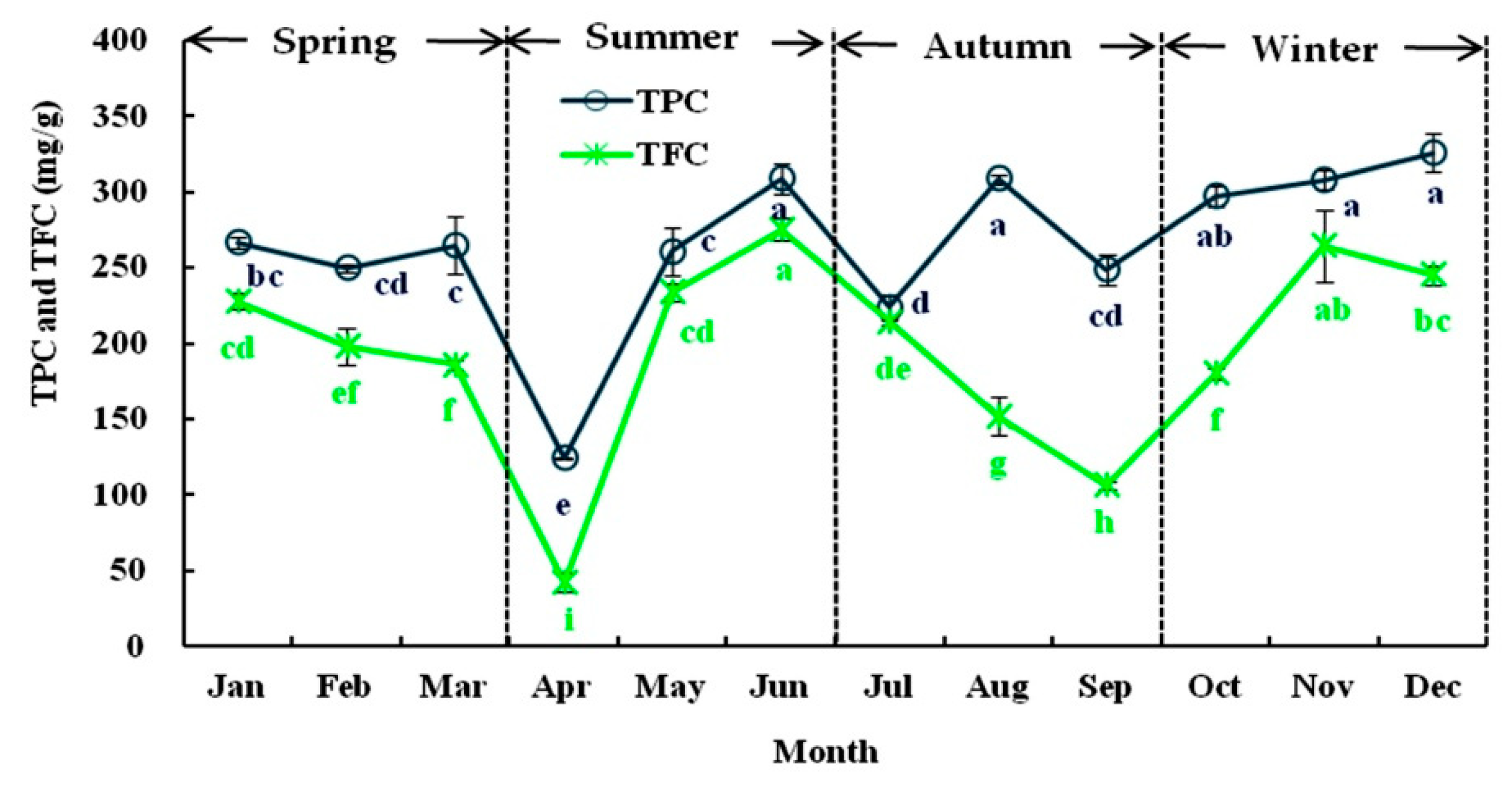
3.1.2. Seasonal Variations in TPC and TFC
3.1.3. Seasonal Variations in Oleuropein (OE) and Flavonoid Composition
3.2. Antioxidant Activity
3.2.1. DPPH Radical-Scavenging Activity
3.2.2. Superoxide Radical-Scavenging Activity (O2−)
3.2.3. Reducing Power
3.3. Anticancer Activity
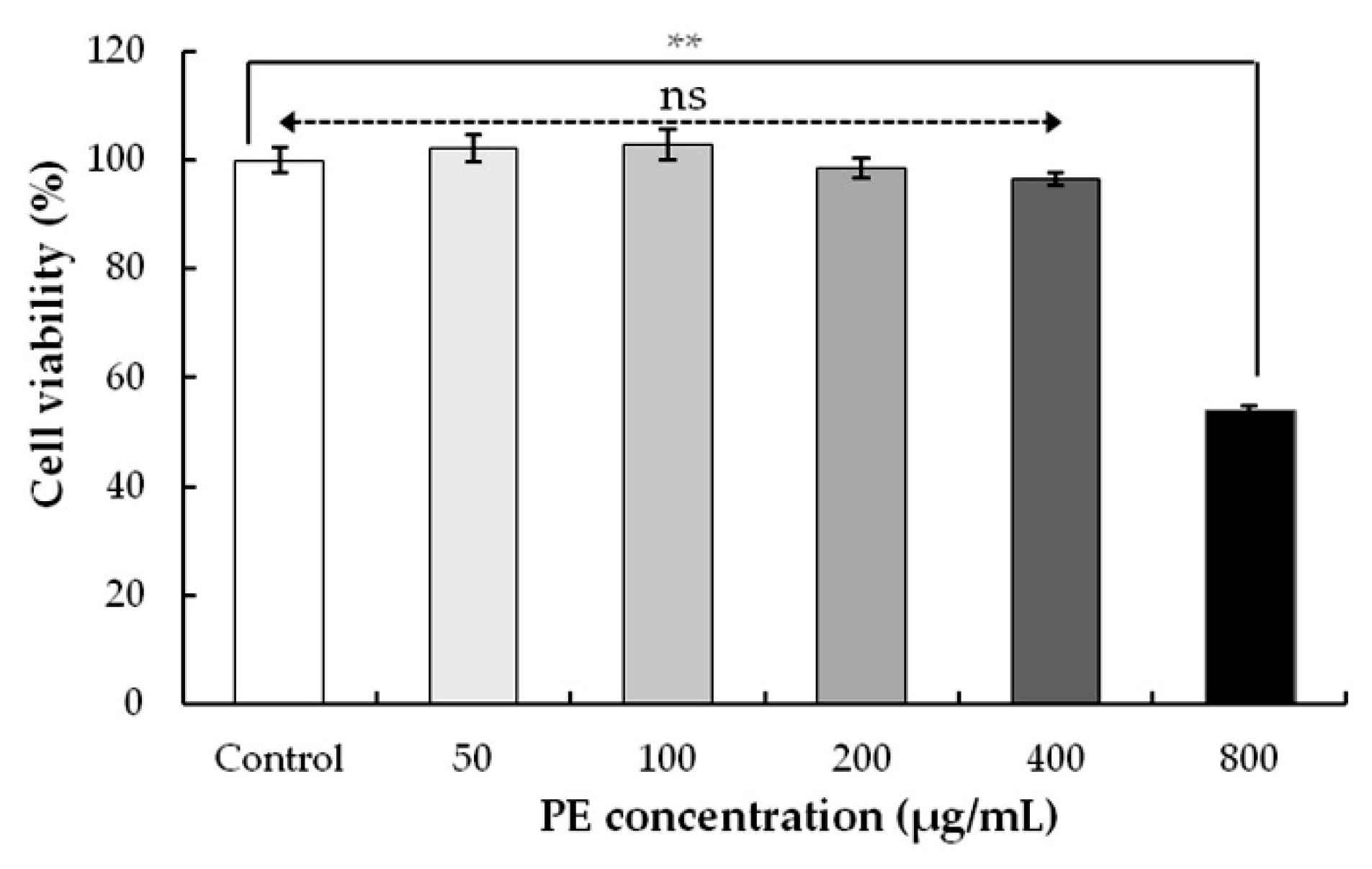
3.3.1. Effect of PE on Cell Viability in HEK293 Cells
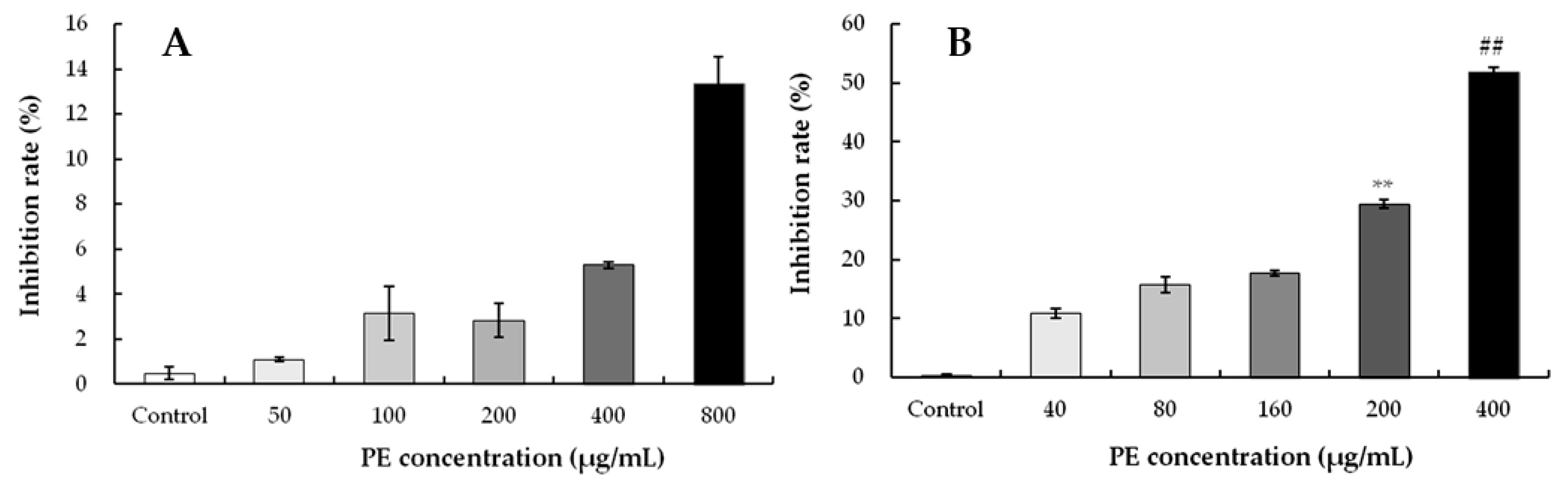
3.3.2. Effect of PE on the Inhibition Rate in HeLa and S180 Cells
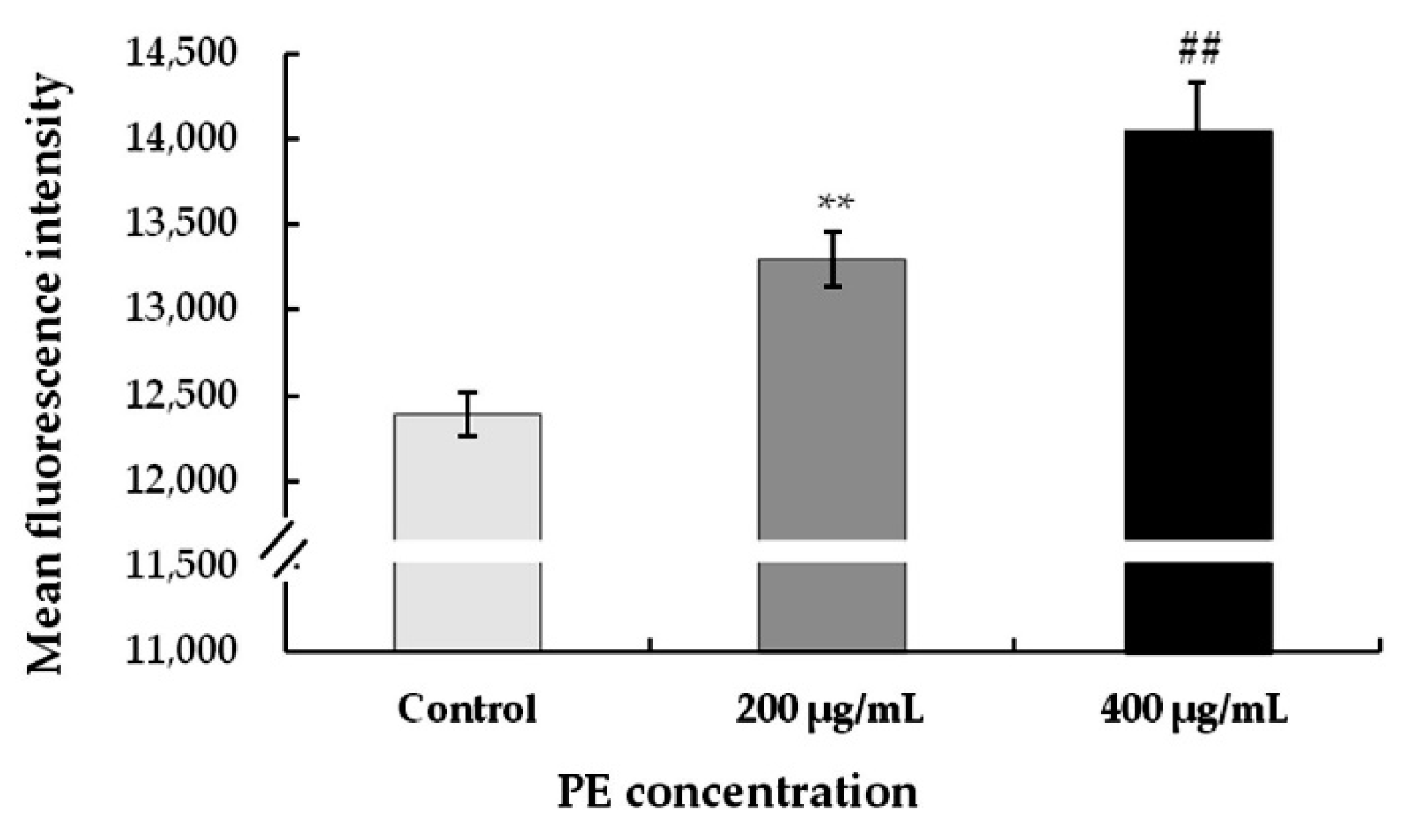
3.3.3. Treatment with PE Induces Mitochondrial Membrane Potential (∆Ψm) Increase in S180 Cells
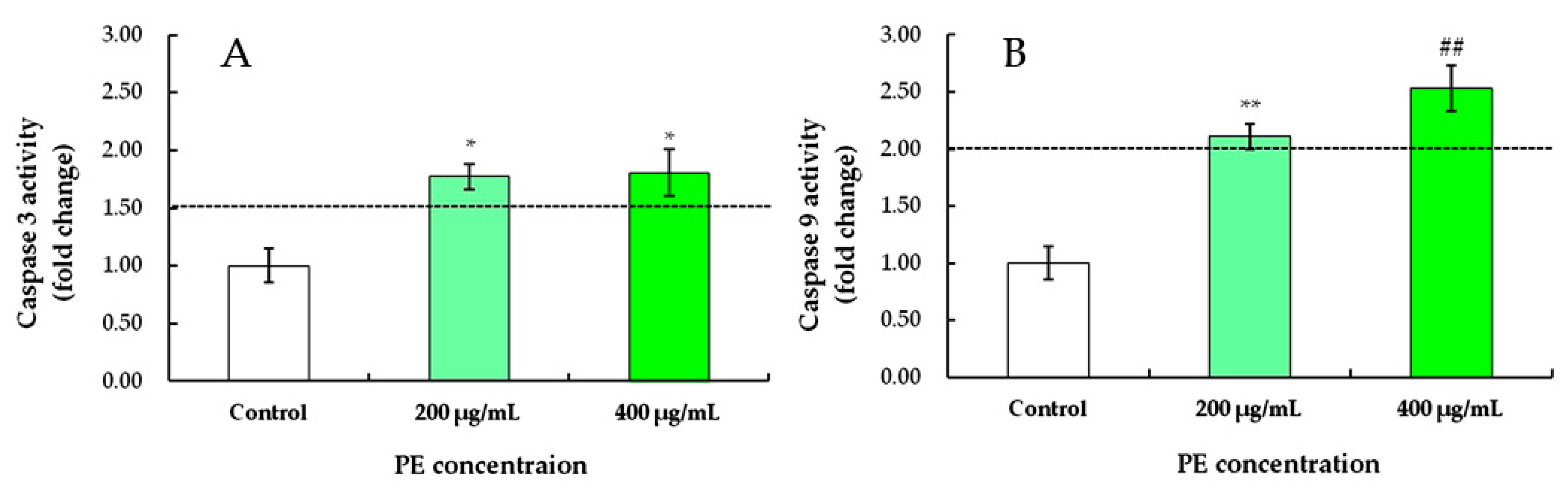
3.3.4. PE Induces Caspase-3 and Caspase-9 Activation in S180 Cells
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Souilem, S.; Fki, I.; Kobayashi, I.; Khalid, N.; Neves, M.A.; Isoda, H.; Sayadi, S.; Nakajima, M. Emerging technologies for recovery of value-added components from olive leaves and their applications in food/feed industries. Food Bioprocess Tech. 2017, 10, 229–248. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, D.; Farooqi, T.J.A.; Ma, L.; Deng, Y.; Jia, Z. The olive ( Olea europaea L.) industry in China: Its status, opportunities and challenges. Agrofor. Syst. 2017, 4, 1–23. [Google Scholar] [CrossRef]
- Wang, B.; Qu, J.; Luo, S.; Feng, S.; Li, T.; Yuan, M.; Huang, Y.; Liao, J.; Yang, R.; Ding, C. Optimization of ultrasound-assisted extraction of flavonoids from olive (Olea europaea) leaves and evaluation of their antioxidant and anticancer activities. Molecules 2018, 23, 2513. [Google Scholar] [CrossRef] [PubMed]
- Su, C.; Sun, J.; Zhu, W.; Peng, L. History, distribution, and potential of the olive industry in China: A Review. Sustainability 2018, 10, 1426. [Google Scholar] [CrossRef]
- Xie, P.J.; Huang, L.X.; Zhang, C.H.; You, F.; Zhang, Y.L. Reduced pressure extraction of oleuropein from olive leaves (Olea europaea L.) with ultrasound assistance. Food Bioprod. Process. 2015, 93, 29–38. [Google Scholar] [CrossRef]
- Cavaca, L.A.; Afonso, C.A. Oleuropein: A valuable bio-renewable synthetic building block. Eur. J. Org. Chem. 2018, 2018, 581–589. [Google Scholar] [CrossRef]
- Şahin, S.; Bilgin, M. Olive tree (Olea europaea L.) leaf as a waste by-product of table olive and olive oil industry: A review. J. Sci. Food Agric. 2018, 98, 1271–1279. [Google Scholar]
- Kiritsakis, K.; Goula, A.M.; Adamopoulos, K.G.; Gerasopoulos, D. Valorization of olive leaves: Spray drying of olive leaf extract. Waste Biomass Valori. 2018, 9, 619–633. [Google Scholar] [CrossRef]
- Rodrigues, F.; Pimentel, F.B.; Oliveira, M.B.P. Olive by-products: Challenge application in cosmetic industry. Ind. Crop. Prod. 2015, 70, 116–124. [Google Scholar] [CrossRef]
- Ranalli, A.; Contento, S.; Lucera, L.; Di Febo, M.; Marchegiani, D.; Di Fonzo, V. Factors affecting the contents of iridoid oleuropein in olive leaves (Olea europaea L.). J. Agric. Food Chem. 2006, 54, 434–440. [Google Scholar] [CrossRef]
- El, S.N.; Karakaya, S. Olive tree (Olea europaea) leaves: Potential beneficial effects on human health. Nutr. Rev. 2010, 67, 632–638. [Google Scholar] [CrossRef] [PubMed]
- Malheiro, R.; Casal, S.; Teixeira, H.; Bento, A.; Pereira, J.A. Effect of olive leaves addition during the extraction process of overmature fruits on olive oil quality. Food Bioprocess Technol. 2013, 6, 509–521. [Google Scholar] [CrossRef]
- Shahdadi, F.; Mirzaei, H.; Garmakhany, A.D. Study of phenolic compound and antioxidant activity of date fruit as a function of ripening stages and drying process. J. Food Sci. Technol. 2015, 52, 1814–1819. [Google Scholar] [CrossRef] [PubMed]
- Lockyer, S.; Rowland, I.; Spencer, J.P.E.; Yaqoob, P.; Stonehouse, W. Impact of phenolic-rich olive leaf extract on blood pressure, plasma lipids and inflammatory markers: A randomised controlled trial. Eur. J. Nutr. 2017, 56, 1421–1432. [Google Scholar] [CrossRef]
- Boss, A.; Bishop, K.S.; Marlow, G.; Barnett, M.P.; Ferguson, L.R. Evidence to support the anti-cancer effect of olive leaf extract and future directions. Nutrients 2016, 8, 513. [Google Scholar] [CrossRef]
- Li, X.; Liu, Y.; Jia, Q.; LaMacchia, V.; O’Donoghue, K.; Huang, Z. A systems biology approach to investigate the antimicrobial activity of oleuropein. J. Ind. Microbiol. Biotechnol. 2016, 43, 1705–1717. [Google Scholar] [CrossRef]
- Benavente-GarcíA, O.; Castillo, J.; Lorente, J.; Ortuño, A.; Rio, J.A.D. Antioxidant activity of phenolics extracted from Olea europaea L. leaves. Food Chem. 2000, 68, 457–462. [Google Scholar] [CrossRef]
- Carrera-González, M.; Ramírez-Expósito, M.; Mayas, M.; Martínez-Martos, J. Protective role of oleuropein and its metabolite hydroxytyrosol on cancer. Trends Food Sci. Technol. 2013, 31, 92–99. [Google Scholar] [CrossRef]
- Talhaoui, N.; Gómez-Caravaca, A.M.; Roldan, C.; Leon, L.; De la Rosa, R.l.; Fernandez-Gutierrez, A.; Segura-Carretero, A. Chemometric analysis for the evaluation of phenolic patterns in olive leaves from six cultivars at different growth stages. J. Agric. Food Chem. 2015, 63, 1722–1729. [Google Scholar] [CrossRef]
- Fleuriet, A.; Macheix, J.-J. Quinyl esters and glucose derivatives of hydroxycinnamic acids during growth and ripening of tomato fruit. Phytochemistry 1981, 20, 667–671. [Google Scholar] [CrossRef]
- Talhaoui, N.; Taamalli, A.; Gómez-Caravaca, A.M.; Fernández-Gutiérrez, A.; Segura-Carretero, A. Phenolic compounds in olive leaves: Analytical determination, biotic and abiotic influence, and health benefits. Food Res. Int. 2015, 77, 92–108. [Google Scholar] [CrossRef]
- Talhaoui, N.; Gómez-Caravaca, A.M.; León, L.; Rosa, R.D.l.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Determination of phenolic compounds of ‘Sikitita’ olive leaves by HPLC-DAD-TOF-MS. Comparison with its parents ‘Arbequina’ and ‘Picual’ olive leaves. Lwt-Food Sci. Technol. 2014, 58, 28–34. [Google Scholar] [CrossRef]
- Xiao, Q.; Zhang, L.; Zhou, L.; Wu, K. Study on olive development in China. Am. Eurasian J. Agric. Environ. Sci. 2009, 5, 414–419. [Google Scholar]
- Cheng, Z.; Zhan, M.; Yang, Z.; Zumstein, K.; Chen, H.; Huang, Q. The major qualitative characteristics of olive (Olea europaea L.) cultivated in southwest China. Front. Plant Sci. 2017, 8, 559. [Google Scholar] [CrossRef]
- Zhan, M.M.; Cheng, Z.Z.; Su, G.C.; Wang, A.Y.; Chen, H.P.; Shan, Z.; Yang, Z.S.; Huang, Q.M. Genetic relationships analysis of olive cultivars grown in China. Genet. Mol. Res. GMR 2015, 14, 5958–5969. [Google Scholar] [CrossRef]
- Xiang, C.; Xu, Z.; Liu, J.; Li, T.; Yang, Z.; Ding, C. Quality, composition, and antioxidant activity of virgin olive oil from introduced varieties at Liangshan. Lwt-Food Sci. Technol. 2017, 78, 226–234. [Google Scholar] [CrossRef]
- Popova, M.; Bankova, V.; Butovska, D.; Petkov, V.; Nikolova-Damyanova, B.; Sabatini, A.G.; Marcazzan, G.L.; Bogdanov, S. Validated methods for the quantification of biologically active constituents of poplar-type propolis. Phytochem. Anal. Int. J. Plant Chem. Biochem. Tech. 2004, 15, 235–240. [Google Scholar] [CrossRef]
- Arvouet-Grand, A.; Vennat, B.; Pourrat, A.; Legret, P. Standardization of a propolis extract and identification of the main constituents. J. Pharm. Belg. 1994, 49, 462–468. [Google Scholar]
- Yemm, E.; Cocking, E.; Ricketts, R. The determination of amino-acids with ninhydrin. Analyst 1955, 80, 209–214. [Google Scholar] [CrossRef]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.t.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Meda, A.; Lamien, C.E.; Romito, M.; Millogo, J.; Nacoulma, O.G. Determination of the total phenolic, flavonoid and proline contents in Burkina Fasan honey, as well as their radical scavenging activity. Food Chem. 2005, 91, 571–577. [Google Scholar] [CrossRef]
- Zhishen, J.; Mengcheng, T.; Jianming, W. The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem. 1999, 64, 555–559. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.-E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. Lwt-Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Moein, M.R.; Moein, S.; Ahmadizadeh, S. Radical scavenging and reducing power of Salvia mirzayanii subfractions. Molecules 2008, 13, 2804–2813. [Google Scholar] [CrossRef]
- Eris, A.; Gulen, H.; Barut, E.; Cansev, A. Annual patterns of total soluble sugars and proteins related to coldhardiness in olive (Olea europaea L.‘Gemlik’). J. Hortic. Sci. Biotechnol. 2007, 82, 597–604. [Google Scholar] [CrossRef]
- Lazovic, B.; Miranovic, K.; Gasic, O.; Popovic, M. Olive Protein Content and Amino Acid Composition. In Proceedings of the III International Symposium on Olive Growing 474, Chania, Greece, 22 September 1997; pp. 465–468. [Google Scholar]
- Papoti, V.T.; Tsimidou, M.Z. Impact of sampling parameters on the radical scavenging potential of olive (Olea europaea L.) leaves. J. Agric. Food Chem. 2009, 57, 3470–3477. [Google Scholar] [CrossRef]
- Heimler, D.; Pieroni, A.; Cimato, A.; Sani, G.; Tattini, M. Seasonal trend of flavonoids, flavonoid glycosides and biflavonoids in ten olive cultivars. Acta Hortic. 1994, 356, 372–374. [Google Scholar] [CrossRef]
- Orak, H.H.; Karamać, M.; Amarowicz, R.; Orak, A.; Penkacik, K. Genotype-related differences in the phenolic compound profile and antioxidant activity of extracts from olive (Olea europaea L.) leaves. Molecules 2019, 24, 1130. [Google Scholar] [CrossRef]
- Malik, N.S.; Bradford, J.M. Changes in oleuropein levels during differentiation and development of floral buds in ‘Arbequina’olives. Sci. Hortic. 2006, 110, 274–278. [Google Scholar] [CrossRef]
- Hashemi, P.; Delfan, B.; Ghiasvand, A.; Alborzi, M.; Raeisi, F. A study of the effects of cultivation variety, collection time, and climate on the amount of oleuropein in olive leaves. Acta Chromatogr. 2010, 22, 133–140. [Google Scholar] [CrossRef]
- Hura, K.; Rapacz, M.; Hura, T.; Żur, I.; Filek, M. The effect of cold on the response of Brassica napus callus tissue to the secondary metabolites of Leptosphaeria maculans. Acta Physiol. Plant. 2015, 37, 13. [Google Scholar] [CrossRef]
- Quirantes-Piné, R.; Lozano-Sánchez, J.; Herrero, M.; Ibáñez, E.; Segura-Carretero, A.; Fernández-Gutiérrez, A. HPLC-ESI-QTOF-MS as a powerful analytical tool for characterising phenolic compounds in olive-leaf extracts. Phytochem. Anal. 2013, 24, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Chimi, H.; Morel, I.; Lescoat, G.; Pasdeloup, N.; Cillard, P.; Cillard, J. Inhibition of iron toxicity in rat hepatocyte culture by natural phenolic compounds. Toxicol. Vitr. 1995, 9, 695–702. [Google Scholar] [CrossRef]
- Shahidi, F.; Janitha, P.; Wanasundara, P. Phenolic antioxidants. Crit. Rev. Food Sci. Nutr. 1992, 32, 67–103. [Google Scholar] [CrossRef]
- Cotelle, N. Role of flavonoids in oxidative stress. Curr. Top. Med. Chem. 2001, 1, 569–590. [Google Scholar] [CrossRef]
- Dai, J.; Mumper, R.J. Plant phenolics: Extraction, analysis and their antioxidant and anticancer properties. Molecules 2010, 15, 7313–7352. [Google Scholar] [CrossRef]
- Szewczyk, K.; Zidorn, C.; Biernasiuk, A.; Komsta, Ł.; Granica, S. Polyphenols from impatiens (Balsaminaceae) and their antioxidant and antimicrobial activities. Ind. Crop. Prod. 2016, 86, 262–272. [Google Scholar] [CrossRef]
- Taamalli, A.; Arráez-Román, D.; Barrajón-Catalán, E.; Ruiz-Torres, V.; Pérez-Sánchez, A.; Herrero, M.; Ibañez, E.; Micol, V.; Zarrouk, M.; Segura-Carretero, A. Use of advanced techniques for the extraction of phenolic compounds from Tunisian olive leaves: Phenolic composition and cytotoxicity against human breast cancer cells. Food Chem. Toxicol. 2012, 50, 1817–1825. [Google Scholar] [CrossRef]
- Fu, S.; Arráez-Roman, D.; Segura-Carretero, A.; Menéndez, J.A.; Menéndez-Gutiérrez, M.P.; Micol, V.; Fernández-Gutiérrez, A. Qualitative screening of phenolic compounds in olive leaf extracts by hyphenated liquid chromatography and preliminary evaluation of cytotoxic activity against human breast cancer cells. Anal. Bioanal. Chem. 2010, 397, 643–654. [Google Scholar] [CrossRef]
- Hashim, Y.Z.Y.; Rowland, I.R.; McGlynn, H.; Servili, M.; Selvaggini, R.; Taticchi, A.; Esposto, S.; Montedoro, G.; Kaisalo, L.; Wähälä, K. Inhibitory effects of olive oil phenolics on invasion in human colon adenocarcinoma cells in vitro. Int. J. Cancer 2008, 122, 495–500. [Google Scholar] [CrossRef]
- Abaza, L.; Talorete, T.; Yamada, P.; Kurita, Y.; Zarrouk, M.; Isoda, H. Tunisian Gerboui olive leaf extract induces growth inhibition and differentiation of human leukemia HL-60 cells. J. Biosci. Biotechnol. Biochem. 2007, 7, 1306–1312. [Google Scholar] [CrossRef] [PubMed]
- Fares, R.; Bazzi, S.; Baydoun, S.E.; Abdel-Massih, R.M. The antioxidant and anti-proliferative activity of the Lebanese Olea europaea extract. Plant Foods Hum. Nutr. 2011, 66, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Barrajón-Catalán, E.; Taamalli, A.; Quirantes-Piné, R.; Roldan-Segura, C.; Arráez-Román, D.; Segura-Carretero, A.; Micol, V.; Zarrouk, M. Differential metabolomic analysis of the potential antiproliferative mechanism of olive leaf extract on the JIMT-1 breast cancer cell line. J. Pharm. Biomed. Anal. 2015, 105, 156–162. [Google Scholar] [CrossRef] [PubMed]
- Mijatovic, S.A.; Timotijevic, G.S.; Miljkovic, D.M.; Radovic, J.M.; Maksimovic-Ivanic, D.D.; Dekanski, D.P.; Stosic-Grujicic, S.D. Multiple antimelanoma potential of dry olive leaf extract. Int. J. Cancer 2011, 128, 1955–1965. [Google Scholar] [CrossRef] [PubMed]
- Kuida, K.; Haydar, T.F.; Kuan, C.Y.; Gu, Y.; Taya, C.; Karasuyama, H.; Su, M.S.-S.; Rakic, P.; Flavell, R.A. Reduced apoptosis and cytochrome c–mediated caspase activation in mice lacking caspase 9. Cell 1998, 94, 325–337. [Google Scholar] [CrossRef]
- Enari, M.; Sakahira, H.; Yokoyama, H.; Okawa, K.; Iwamatsu, A.; Nagata, S. A caspase-activated DNase that degrades DNA during apoptosis, and its inhibitor ICAD. Nature 1998, 391, 43. [Google Scholar] [CrossRef] [PubMed]
| CP | LCR (mg/mL) | CE | RC (R2) | RT (min) |
|---|---|---|---|---|
| Oleuropein | 0.1–1.7 | y = 607.01x + 9.2935 | 0.9990 | 6.388 |
| Rutin | 0.1–2.5 | y = 576.09x + 61.592 | 0.9988 | 9.166 |
| Luteolin-4’-O-glucoside | 1–19 | y = 336.79x + 220.01 | 0.9902 | 10.360 |
| Apigenin-7-O-glucoside | 0.1–1.3 | y = 3304.4x − 8.3598 | 0.9995 | 12.882 |
| Luteolin | 0.01–0.031 | y = 25,711x − 17.17 | 0.9905 | 18.890 |
| Quercetin | 0.005–0.041 | y = 25,590x − 6.9509 | 0.9991 | 19.162 |
| Apigenin | 0.007–0.021 | y = 4241.7x − 3.6054 | 0.9944 | 24.332 |
| Month | Oleuropein | Rutin | Luteolin-4’-O-Glucoside | Apigenin-7-O-Glucoside | Luteolin | Quercetin | Apigenin |
|---|---|---|---|---|---|---|---|
| January | 108.57 ± 2.43c | 0.38 ± 0.043c | 39.72 ± 2.37c | 2.07 ± 0.11ab | 0.057 ± 0.002cd | 0.463 ± 0.028b | nd |
| February | 136.70 ± 1.57b | 0.42 ± 0.038bc | 63.08 ± 6.14b | 2.47 ± 0.22a | 0.061 ± 0.008cd | 0.537 ± 0.092b | nd |
| March | 151.74 ± 2.20a | 0.07 ± 0.012ef | 17.21 ± 0.79d | 0.86 ± 0.13e | 0.093 ± 0.008cd | 0.458 ± 0.058b | nd |
| April | 64.55 ± 3.79f | nq | nq | 0.65 ± 0.42e | 0.786 ± 0.096a | nq | 0.177 ± 0.016 |
| May | 34.45 ± 0.16h | 0.07 ± 0.006ef | 11.81 ± 1.41d | 0.84 ± 0.09e | 0.365 ± 0.147b | nq | nd |
| June | 78.23 ± 0.12e | 0.21 ± 0.003d | 20.14 ± 0.26d | 1.47 ± 0.03cd | 0.149 ± 0.022c | 0.402 ± 0.066b | 0.131 ± 0.004 |
| July | 54.34 ± 1.46g | 0.13 ± 0.012de | 10.65 ± 1.41d | 0.83 ± 0.13e | 0.044 ± 0.004cd | 0.164 ± 0.101c | nd |
| August | 52.16 ± 0.33g | 0.14 ± 0.012de | 16.23 ± 1.25d | 1.02 ± 0.07de | 0.017 ± 0.001cd | 0.803 ± 0.080a | 0.135 ± 0.018 |
| September | 35.44 ± 0.10h | 0.34 ± 0.035c | 16.19 ± 1.34d | 0.95 ± 0.07e | nq | 0.462 ± 0.081b | nd |
| October | 91.42 ± 0.47d | 0.50 ± 0.062b | 18.37 ± 2.46d | 0.61 ± 0.08e | 0.032 ± 0.007cd | 0.020 ± 0.001c | nd |
| November | 142.56 ± 6.44b | 0.91 ± 0.095a | 74.32 ± 8.52a | 1.91 ± 0.19bc | 0.068 ± 0.002cd | 0.189 ± 0.094c | nd |
| December | 106.38 ± 1.47c | 0.53 ± 0.015b | 53.81 ± 1.75b | 1.86 ± 0.05bc | 0.067 ± 0.001cd | 0.581 ± 0.009b | nd |
| Concentration (mg/mL) | 0.20 | 0.40 | 0.60 | 0.80 | 1.00 | 1.20 | IC50 | |
|---|---|---|---|---|---|---|---|---|
| DPPH radical scavenging (%) | VC | 92.61 + 0.18 b | 93.84 + 0.11 a | 93.91 + 0.54 a | 93.82 + 0.12 a | 94.20 + 0.07 a | 94.19 + 0.14 a | |
| PE | 56.88 + 0.57 d | 89.62 + 0.77 c | 91.53 + 0.34 b | 93.68 + 0.15 a | 91.17 + 0.31 b | 91.29 + 0.29 b | 0.14 | |
| Superoxide radical scavenging (%) | VC | 95.55 + 0.43 a | 95.21 + 0.30 a | 95.84 + 0.19 a | 95.28 + 0.28 a | 95.55 + 0.43 a | 95.21 + 0.29 a | |
| PE | 23.12 + 1.83 e | 24.65 + 0.65 e | 34.26 + 0.53 d | 43.34 + 0.51 c | 53.85 + 0.29 b | 62.26 + 1.76 a | 0.93 | |
| Reducing power | VC | 1.16 + 0.19 b | 1.52 + 0.03 a | 1.50 + 0.01 a | 1.47 + 0.02 a | 1.51 + 0.02 a | 1.69 + 0.02a | |
| PE | 0.30 + 0.012 f | 0.55 + 0.006 e | 0.69 + 0.012 d | 0.90 + 0.030 c | 1.07 + 0.058 b | 1.34 + 0.003 a | ||
| Cells | IC50 (μg/mL) |
|---|---|
| HEK293 | 841.48 |
| Hela | 7139.23 |
| S180 | 457.69 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, B.; Qu, J.; Feng, S.; Chen, T.; Yuan, M.; Huang, Y.; Liao, J.; Yang, R.; Ding, C. Seasonal Variations in the Chemical Composition of Liangshan Olive Leaves and Their Antioxidant and Anticancer Activities. Foods 2019, 8, 657. https://doi.org/10.3390/foods8120657
Wang B, Qu J, Feng S, Chen T, Yuan M, Huang Y, Liao J, Yang R, Ding C. Seasonal Variations in the Chemical Composition of Liangshan Olive Leaves and Their Antioxidant and Anticancer Activities. Foods. 2019; 8(12):657. https://doi.org/10.3390/foods8120657
Chicago/Turabian StyleWang, Bixia, Jipeng Qu, Shiling Feng, Tao Chen, Ming Yuan, Yan Huang, Jinqiu Liao, Ruiwu Yang, and Chunbang Ding. 2019. "Seasonal Variations in the Chemical Composition of Liangshan Olive Leaves and Their Antioxidant and Anticancer Activities" Foods 8, no. 12: 657. https://doi.org/10.3390/foods8120657
APA StyleWang, B., Qu, J., Feng, S., Chen, T., Yuan, M., Huang, Y., Liao, J., Yang, R., & Ding, C. (2019). Seasonal Variations in the Chemical Composition of Liangshan Olive Leaves and Their Antioxidant and Anticancer Activities. Foods, 8(12), 657. https://doi.org/10.3390/foods8120657








