Differential Effects of Monounsaturated and Polyunsaturated Fats on Satiety and Gut Hormone Responses in Healthy Subjects
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Study Design
2.3. Satiety
2.4. Test Meals
2.5. Hormone Assays
2.6. Statistical Analysis
3. Results
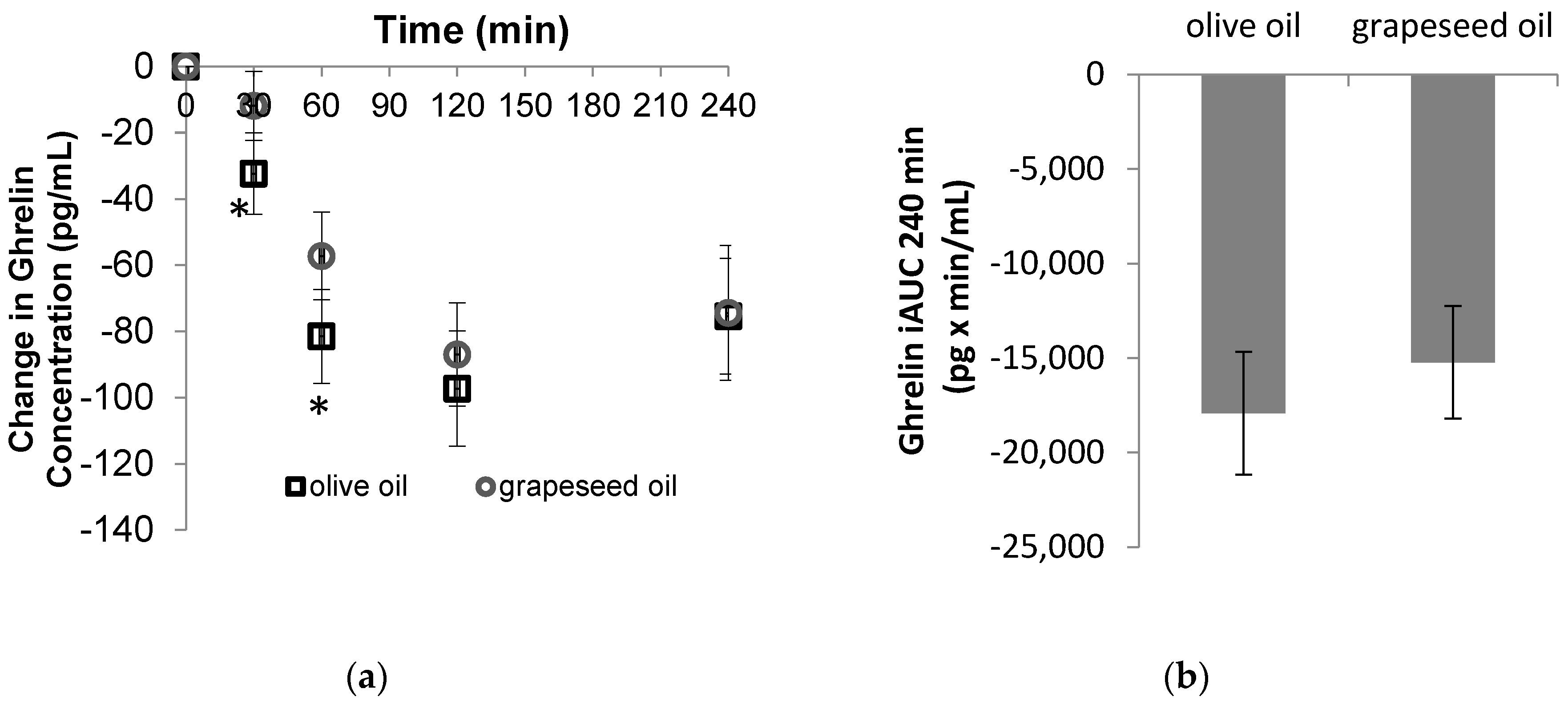
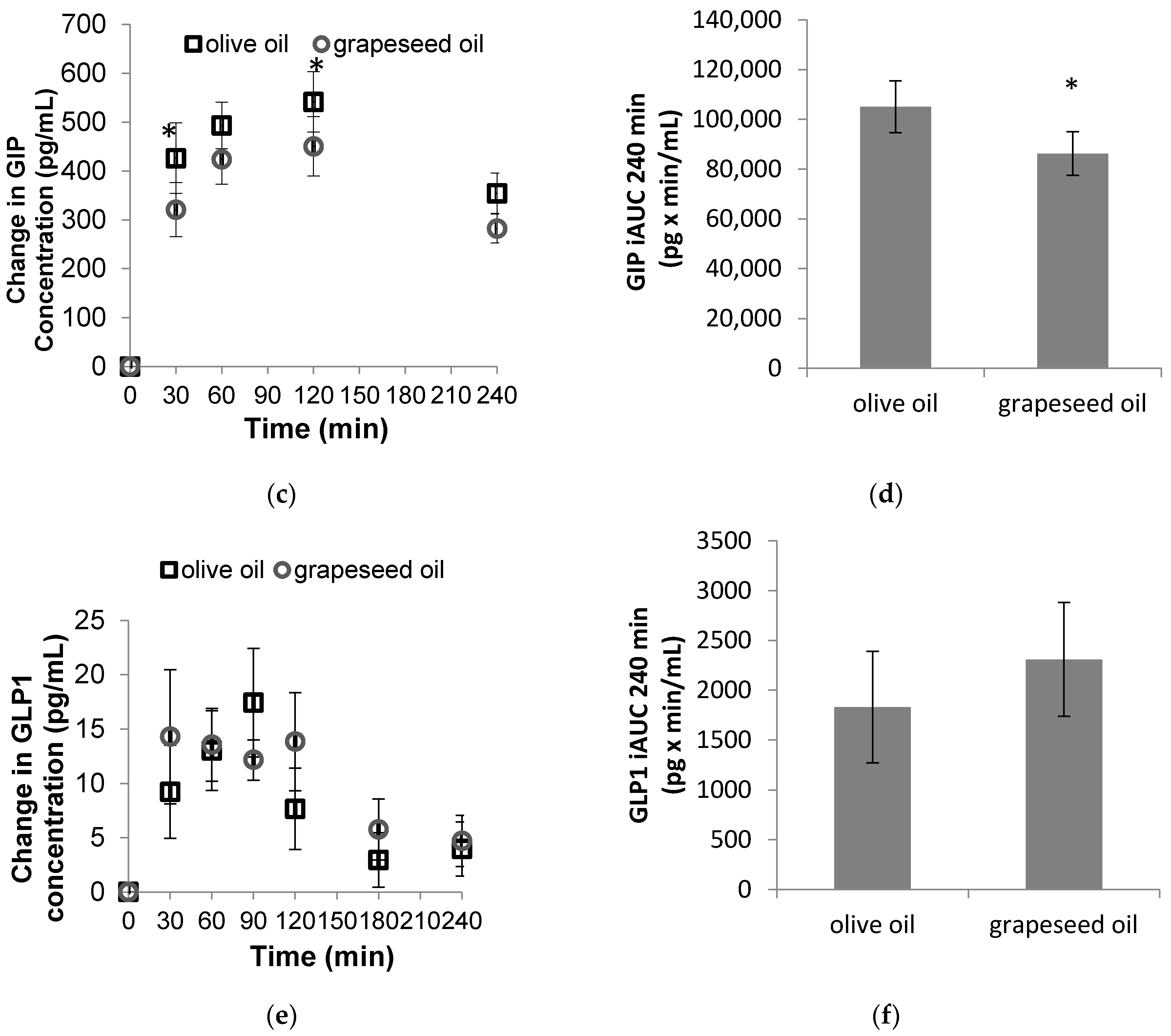
3.1. Hormone Responses
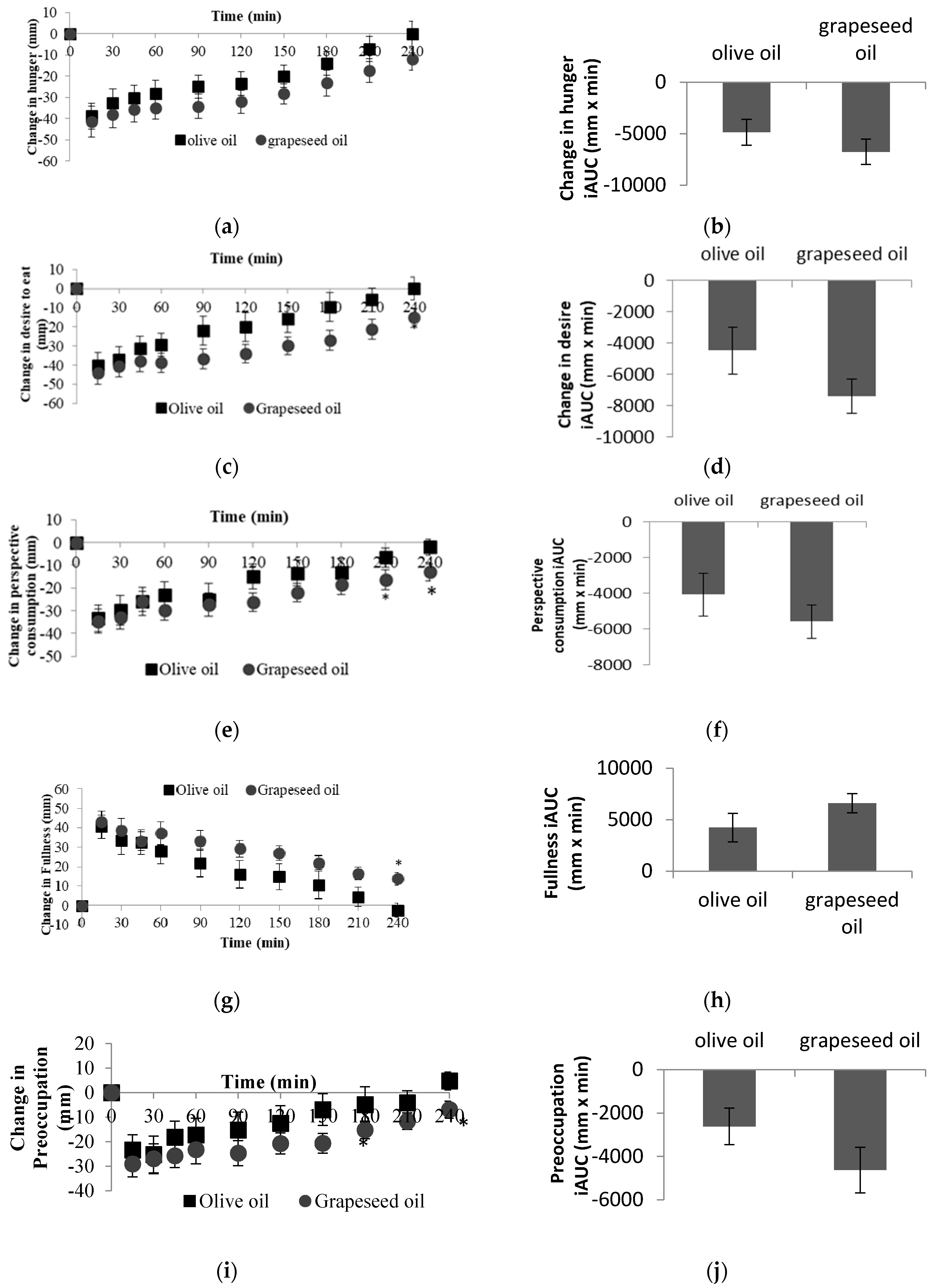
3.2. Subjective Satiety Responses
3.3. Correlations
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Sowers, J.R. Obesity as a cardiovascular risk factor. Am. J. Med. 2003, 115 (Suppl. 8A), 37S–41S. [Google Scholar] [CrossRef] [PubMed]
- Bray, G.A.; Paeratakul, S.; Popkin, B.M. Dietary fat and obesity: A review of animal, clinical and epidemiological studies. Physiol. Behav. 2004, 83, 549–555. [Google Scholar] [CrossRef]
- Lawton, C.L.; Burley, V.J.; Wales, J.K.; Blundell, J.E. Dietary fat and appetite control in obese subjects: Weak effects on satiation and satiety. Int. J. Obes. Relat. Metab. Disord. 1993, 17, 409–416. [Google Scholar]
- Kozimor, A.; Chang, H.; Cooper, J.A. Effects of dietary fatty acid composition from a high fat meal on satiety. Appetite 2013, 69, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Strik, C.M.; Lithander, F.E.; McGill, A.T.; MacGibbon, A.K.; McArdle, B.H.; Poppitt, S.D. No evidence of differential effects of SFA, MUFA or PUFA on post-ingestive satiety and energy intake: A randomised trial of fatty acid saturation. Nutr. J. 2010, 9, 24. [Google Scholar] [CrossRef]
- Stevenson, J.L.; Clevenger, H.C.; Cooper, J.A. Hunger and satiety responses to high-fat meals of varying fatty acid composition in women with obesity. Obesity (Silver Spring) 2015, 23, 1980–1986. [Google Scholar] [CrossRef]
- Alfenas, R.C.; Mattes, R.D. Effect of fat sources on satiety. Obes. Res. 2003, 11, 183–187. [Google Scholar] [CrossRef]
- Cummings, D.E. Ghrelin and the short- and long-term regulation of appetite and body weight. Physiol. Behav. 2006, 89, 71–84. [Google Scholar] [CrossRef]
- Falko, J.M.; Crockett, S.E.; Cataland, S.; Mazzaferri, E.L. Gastric inhibitory polypeptide (GIP) stimulated by ingestion in man. J. Clin. Endocrinol. Metab. 1975, 41, 260–265. [Google Scholar] [CrossRef]
- Miyawaki, K.; Yamada, Y.; Ban, N.; Ihara, Y.; Tsukiyama, K.; Zhou, H.; Fujimoto, S.; Oku, A.; Tsuda, K.; Toyokuni, S.; et al. Inhibition of gastric inhibitory polypeptide signaling prevents obesity. Nat. Med. 2002, 8, 738–742. [Google Scholar] [CrossRef]
- De Silva, A.; Salem, V.; Long, C.J.; Makwana, A.; Newbould, R.D.; Rabiner, E.A.; Ghatei, M.A.; Bloom, S.R.; Matthews, P.M.; Beaver, J.D.; et al. The gut hormones PYY 3-36 and GLP-1 7-36 amide reduce food intake and modulate brain activity in appetite centers in humans. Cell Metab. 2011, 14, 700–706. [Google Scholar] [CrossRef] [PubMed]
- Verdich, C.; Flint, A.; Gutzwiller, J.P.; Näslund, E.; Beglinger, C.; Hellström, P.M.; Long, S.J.; Morgan, L.M.; Holst, J.J.; Astrup, A. A meta-analysis of the effect of glucagon-like peptide-1 (7-36) amide on ad libitum energy intake in humans. J. Clin. Endocrinol. Metab. 2001, 86, 4382–4389. [Google Scholar]
- Kaviani, S.; Cooper, J.A. Appetite responses to high-fat meals or diets of varying fatty acid composition: A comprehensive review. Eur. J. Clin. Nutr. 2017, 71, 1154–1165. [Google Scholar] [CrossRef]
- MacIntosh, C.G.; Holt, S.H.; Brand-Miller, J.C. The degree of fat saturation does not alter glycemic, insulinemic or satiety responses to a starchy staple in healthy men. J. Nutr. 2003, 133, 2577–2580. [Google Scholar] [CrossRef]
- Flint, A.; Helt, B.; Raben, A.; Toubro, S.; Astrup, A. Effects of different dietary fat types on postprandial appetite and energy expenditure. Obes. Res. 2003, 11, 1449–1455. [Google Scholar] [CrossRef]
- Poppitt, S.D.; Strik, C.M.; MacGibbon, A.K.; McArdle, B.H.; Budgett, S.C.; McGill, A.T. Fatty acid chain length, postprandial satiety and food intake in lean men. Physiol. Behav. 2010, 101, 161–167. [Google Scholar] [CrossRef]
- Sun, L.; Tan, K.W.J.; Lim, J.Z.; Magkos, F.; Henry, C.J. Dietary fat and carbohydrate quality have independent effects on postprandial glucose and lipid responses. Eur. J. Nutr. 2018, 57, 243–250. [Google Scholar] [CrossRef]
- Yanagimachi, T.; Fujita, Y.; Takeda, Y.; Honjo, J.; Sakagami, H.; Kitsunai, H.; Takiyama, Y.; Abiko, A.; Makino, Y.; Kieffer, T.J.; et al. Kieffer TJDipeptidyl peptidase-4 inhibitor treatment induces a greater increase in plasma levels of bioactive GIP than GLP-1 in non-diabetic subjects. Mol. Metab. 2017, 6, 226–231. [Google Scholar] [CrossRef]
- Bak, M.J.; Wewer Albrechtsen, N.J.; Pedersen, J.; Knop, F.K.; Vilsbøll, T.; Jørgensen, N.B.; Hartmann, B.; Deacon, C.F.; Dragsted, L.O.; Holst, J.J. Specificity and sensitivity of commercially available assays for glucagon-like peptide-1 (GLP-1): Implications for GLP-1 measurements in clinical studies. Diabetes Obes. Metab. 2014, 16, 1155–1164. [Google Scholar] [CrossRef]
- Rolls, B.J.; Kim-Harris, S.; Fischman, M.W.; Foltin, R.W.; Moran, T.H.; Stoner, S.A. Satiety after preloads with different amounts of fat and carbohydrate: Implications for obesity. Am. J. Clin. Nutr. 1994, 60, 476–487. [Google Scholar] [CrossRef]
- Halton, T.L.; Hu, F.B. The effects of high protein diets on thermogenesis, satiety and weight loss: A critical review. J. Am. Coll. Nutr. 2004, 23, 373–385. [Google Scholar] [CrossRef]
- Green, S.M.; Burley, V.J.; Blundell, J.E. Effect of fat- and sucrose-containing foods on the size of eating episodes and energy intake in lean males: Potential for causing overconsumption. Eur. J. Clin. Nutr. 1994, 48, 547–555. [Google Scholar]
- Bjermo, H.; Iggman, D.; Kullberg, J.; Dahlman, I.; Johansson, L.; Persson, L.; Berglund, J.; Pulkki, K.; Basu, S.; Uusitupa, M.; et al. Effects of n-6 PUFAs compared with SFAs on liver fat, lipoproteins, and inflammation in abdominal obesity: A randomized controlled trial. Am. J. Clin. Nutr. 2012, 95, 1003–1012. [Google Scholar] [CrossRef] [PubMed]
- Jones, P.J.; Ridgen, J.E.; Phang, P.T.; Birmingham, C.L. Influence of dietary fat polyunsaturated to saturated ratio on energy substrate utilization in obesity. Metabolism 1992, 41, 396–401. [Google Scholar] [CrossRef]
- Thomsen, C.; Rasmussen, O.; Lousen, T.; Holst, J.J.; Fenselau, S.; Schrezenmeir, J.; Hermansen, K. Differential effects of saturated and monounsaturated fatty acids on postprandial lipemia and incretin responses in healthy subjects. Am. J. Clin. Nutr. 1999, 69, 1135–1143. [Google Scholar] [CrossRef]
- Stubbs, R.J.; Hughes, D.A.; Johnstone, A.M.; Rowley, E.; Reid, C.; Elia, M.; Stratton, R.; Delargy, H.; King, N.; Blundell, J.E. The use of visual analogue scales to assess motivation to eat in human subjects: A review of their reliability and validity with an evaluation of new hand-held computerized systems for temporal tracking of appetite ratings. Br. J. Nutr. 2000, 84, 405–415. [Google Scholar] [CrossRef]
- Lawton, C.L.; Delargy, H.J.; Brockman, J.; Smith, F.C.; Blundell, J.E. The degree of saturation of fatty acids influences post-ingestive satiety. Br. J. Nutr. 2000, 83, 473–482. [Google Scholar] [CrossRef]
- Casas-Agustench, P.; López-Uriarte, P.; Bulló, M.; Ros, E.; Gómez-Flores, A.; Salas-Salvadó, J. Acute effects of three high-fat meals with different fat saturations on energy expenditure, substrate oxidation and satiety. Clin. Nutr. 2009, 28, 39–45. [Google Scholar] [CrossRef]
- Feltrin, K.L.; Patterson, M.; Ghatei, M.A.; Bloom, S.R.; Meyer, J.H.; Horowitz, M.; Feinle-Bisset, C. Effect of fatty acid chain length on suppression of ghrelin and stimulation of PYY, GLP-2 and PP secretion in healthy men. Peptides 2006, 27, 1638–1643. [Google Scholar] [CrossRef]
- Little, T.J.; Horowitz, M.; Feinle-Bisset, C. Modulation by high-fat diets of gastrointestinal function and hormones associated with the regulation of energy intake: Implications for the pathophysiology of obesity. Am. J. Clin. Nutr. 2007, 86, 531–541. [Google Scholar] [CrossRef]
- Riediger, T.; Traebert, M.; Schmid, H.A.; Scheel, C.; Lutz, T.A.; Scharrer, E. Site-specific effects of ghrelin on the neuronal activity in the hypothalamic arcuate nucleus. Neurosci. Lett. 2003, 341, 151–155. [Google Scholar] [CrossRef]
- Nakazato, M.; Murakami, N.; Date, Y.; Kojima, M.; Matsuo, H.; Kangawa, K.; Matsukura, S. A role for ghrelin in the central regulation of feeding. Nature 2001, 409, 194–198. [Google Scholar] [CrossRef]
- Naslund, E.; Barkeling, B.; King, N.; Gutniak, M.; Blundell, J.E.; Holst, J.J.; Rössner, S.; Hellström, P.M. Energy intake and appetite are suppressed by glucagon-like peptide-1 (GLP-1) in obese men. Int. J. Obes. Relat. Metab. Disord. 1999, 23, 304–311. [Google Scholar] [CrossRef] [PubMed]
- Turton, M.D.; O’Shea, D.; Gunn, I.; Beak, S.A.; Edwards, C.M.; Meeran, K.; Choi, S.J.; Taylor, G.M.; Heath, M.M.; Lambert, P.D.; et al. A role for glucagon-like peptide-1 in the central regulation of feeding. Nature 1996, 379, 69–72. [Google Scholar] [CrossRef]
- Beysen, C.; Karpe, F.; Fielding, B.A.; Clark, A.; Levy, J.C.; Frayn, K.N. Interaction between specific fatty acids, GLP-1 and insulin secretion in humans. Diabetologia 2002, 45, 1533–1541. [Google Scholar]
- Verdich, C.; Toubro, S.; Buemann, B.; Lysgård Madsen, J.; Juul Holst, J.; Astrup, A. The role of postprandial releases of insulin and incretin hormones in meal-induced satiety--effect of obesity and weight reduction. Int. J. Obes. Relat. Metab. Disord. 2001, 25, 1206–1214. [Google Scholar] [CrossRef]
- Raben, A.; Tagliabue, A.; Christensen, N.J.; Madsen, J.; Holst, J.J.; Astrup, A. Resistant starch: The effect on postprandial glycemia, hormonal response, and satiety. Am. J. Clin. Nutr. 1994, 60, 544–551. [Google Scholar] [CrossRef]
- Raben, A.; Andersen, K.; Karberg, M.A.; Holst, J.J.; Astrup, A. Acetylation of or beta-cyclodextrin addition to potato beneficial effect on glucose metabolism and appetite sensations. Am. J. Clin. Nutr. 1997, 66, 304–314. [Google Scholar] [CrossRef]
- Raben, A.; Andersen, H.B.; Christensen, N.J.; Madsen, J.; Holst, J.J.; Astrup, A. Evidence for an abnormal postprandial response to a high-fat meal in women predisposed to obesity. Am. J. Physiol. 1994, 267, E549–E559. [Google Scholar] [CrossRef]
- Cooper, J.A.; Watras, A.C.; Paton, C.M.; Wegner, F.H.; Adams, A.K.; Schoeller, D.A. Impact of exercise and dietary fatty acid composition from a high-fat diet on markers of hunger and satiety. Appetite 2011, 56, 171–178. [Google Scholar] [CrossRef]
| Olive Oil | Grapeseed Oil | |
|---|---|---|
| Amounts per Serving (g) | 44.4 | 40.0 |
| Calories (kcal) | 364.0 | 360.0 |
| Total fat (g) | 40.4 | 40.0 |
| Saturated Fat (g) | 6.1 | 4.0 |
| Monounsaturated fat (g) | 31.3 | 7.6 |
| Polyunsaturated fat (g) | 3.0 | 28.4 |
| Measurements | Average | SD |
|---|---|---|
| Age (year) | 27.1 | 6.4 |
| Weight (kg) | 70.8 | 10.3 |
| Height (cm) | 174.4 | 4.5 |
| Body mass index (kg/m2) | 23.3 | 3.2 |
| Body fat percent (%) | 19.1 | 5.1 |
| Waist circumference (cm) | 79.7 | 9.3 |
| Hip circumference (cm) | 96 | 7.2 |
| Waist-to-hip ratio | 0.89 | 0.05 |
| iAUC240 | Hunger | Desire | Fullness | Quantity | Preoccupation | ||||||
| r | p | r | p | r | p | r | p | r | p | ||
| MUFA | GIP | 0.57 * | 0.04 | 0.58 * | 0.04 | −0.57 * | 0.04 | 0.59 * | 0.03 | 0.22 | 0.47 |
| Ghrelin | −0.13 | 0.66 | −0.18 | 0.57 | −0.07 | 0.82 | 0.03 | 0.92 | −0.58 * | 0.04 | |
| PUFA | GIP | 0.01 | 0.98 | 0.17 | 0.59 | −0.01 | 0.99 | −0.07 | 0.83 | −0.03 | 0.92 |
| Ghrelin | −0.33 | 0.27 | −0.30 | 0.31 | 0.19 | 0.53 | −0.18 | 0.57 | −0.57 * | 0.04 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, L.; Goh, H.J.; Govindharajulu, P.; Khee-Shing Leow, M.; Henry, C.J. Differential Effects of Monounsaturated and Polyunsaturated Fats on Satiety and Gut Hormone Responses in Healthy Subjects. Foods 2019, 8, 634. https://doi.org/10.3390/foods8120634
Sun L, Goh HJ, Govindharajulu P, Khee-Shing Leow M, Henry CJ. Differential Effects of Monounsaturated and Polyunsaturated Fats on Satiety and Gut Hormone Responses in Healthy Subjects. Foods. 2019; 8(12):634. https://doi.org/10.3390/foods8120634
Chicago/Turabian StyleSun, Lijuan, Hui Jen Goh, Priya Govindharajulu, Melvin Khee-Shing Leow, and Christiani Jeyakumar Henry. 2019. "Differential Effects of Monounsaturated and Polyunsaturated Fats on Satiety and Gut Hormone Responses in Healthy Subjects" Foods 8, no. 12: 634. https://doi.org/10.3390/foods8120634
APA StyleSun, L., Goh, H. J., Govindharajulu, P., Khee-Shing Leow, M., & Henry, C. J. (2019). Differential Effects of Monounsaturated and Polyunsaturated Fats on Satiety and Gut Hormone Responses in Healthy Subjects. Foods, 8(12), 634. https://doi.org/10.3390/foods8120634






