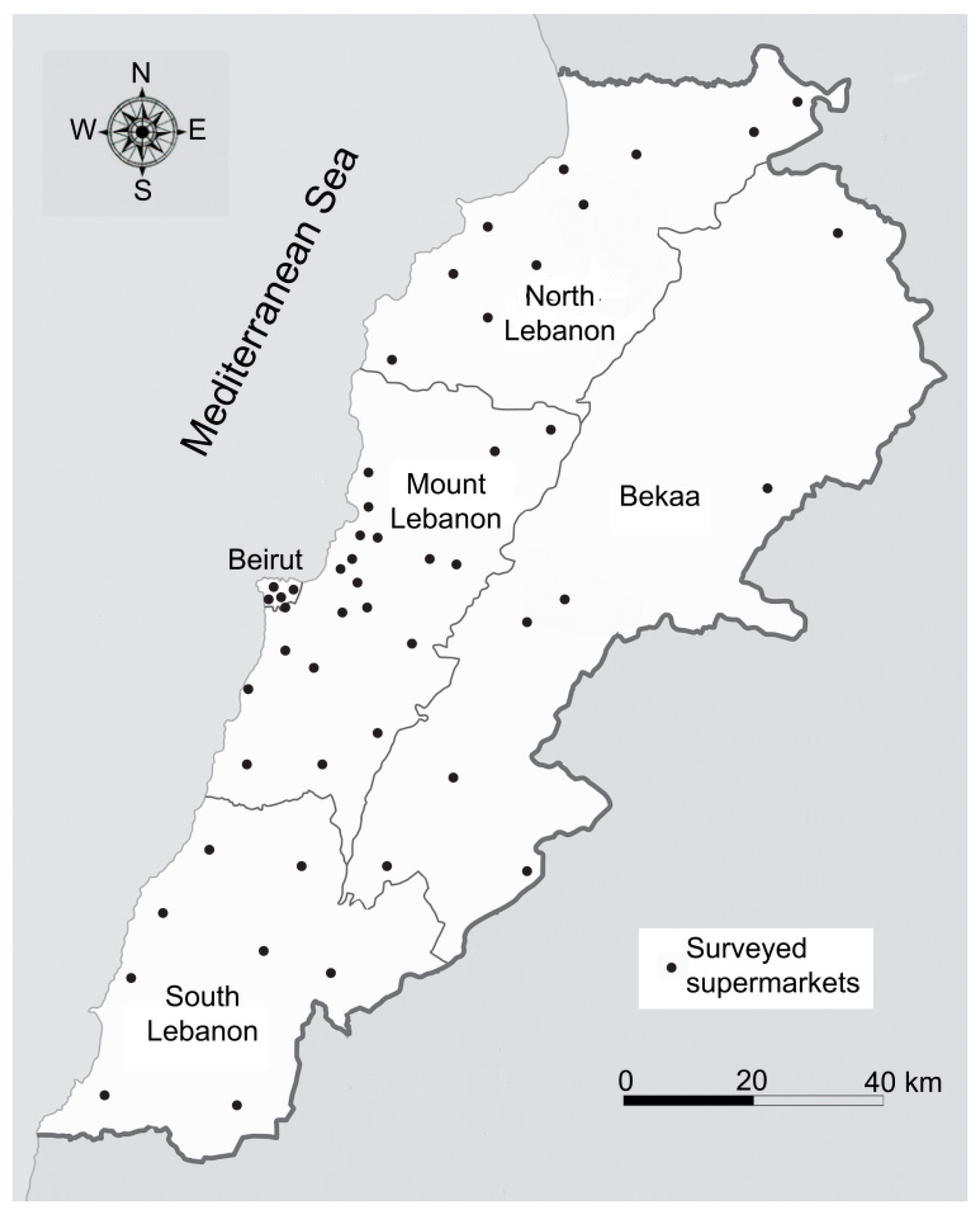
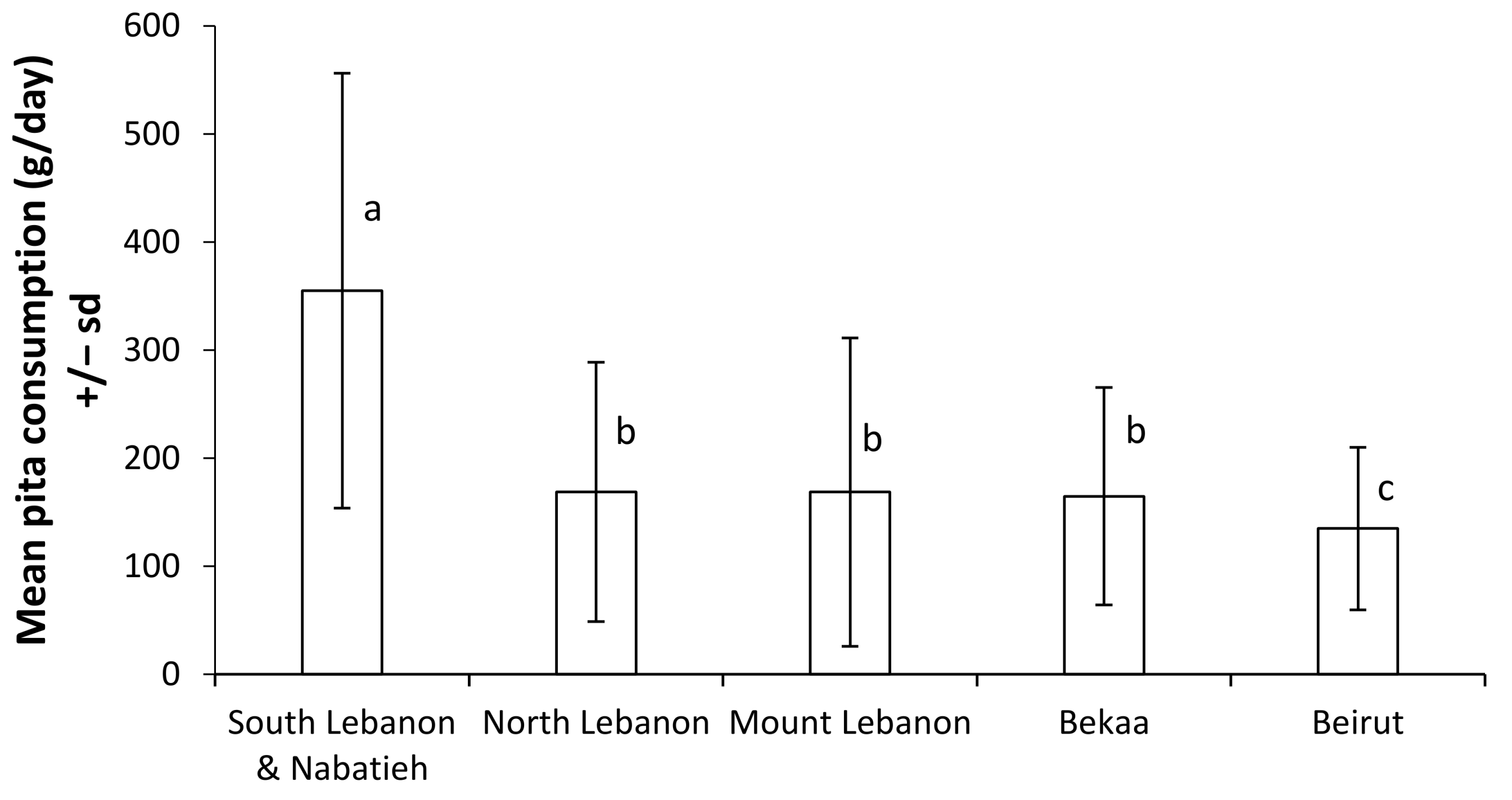
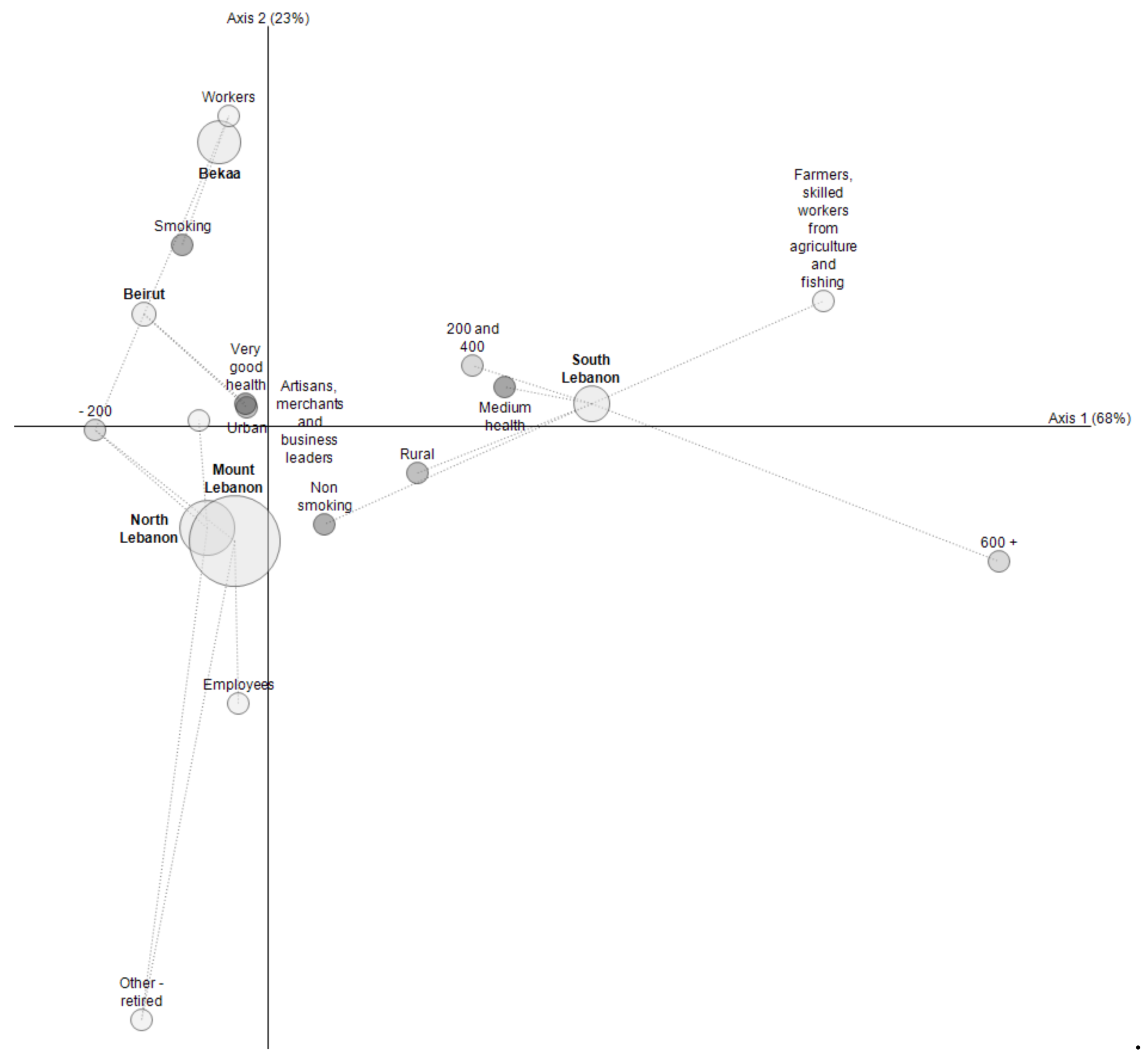
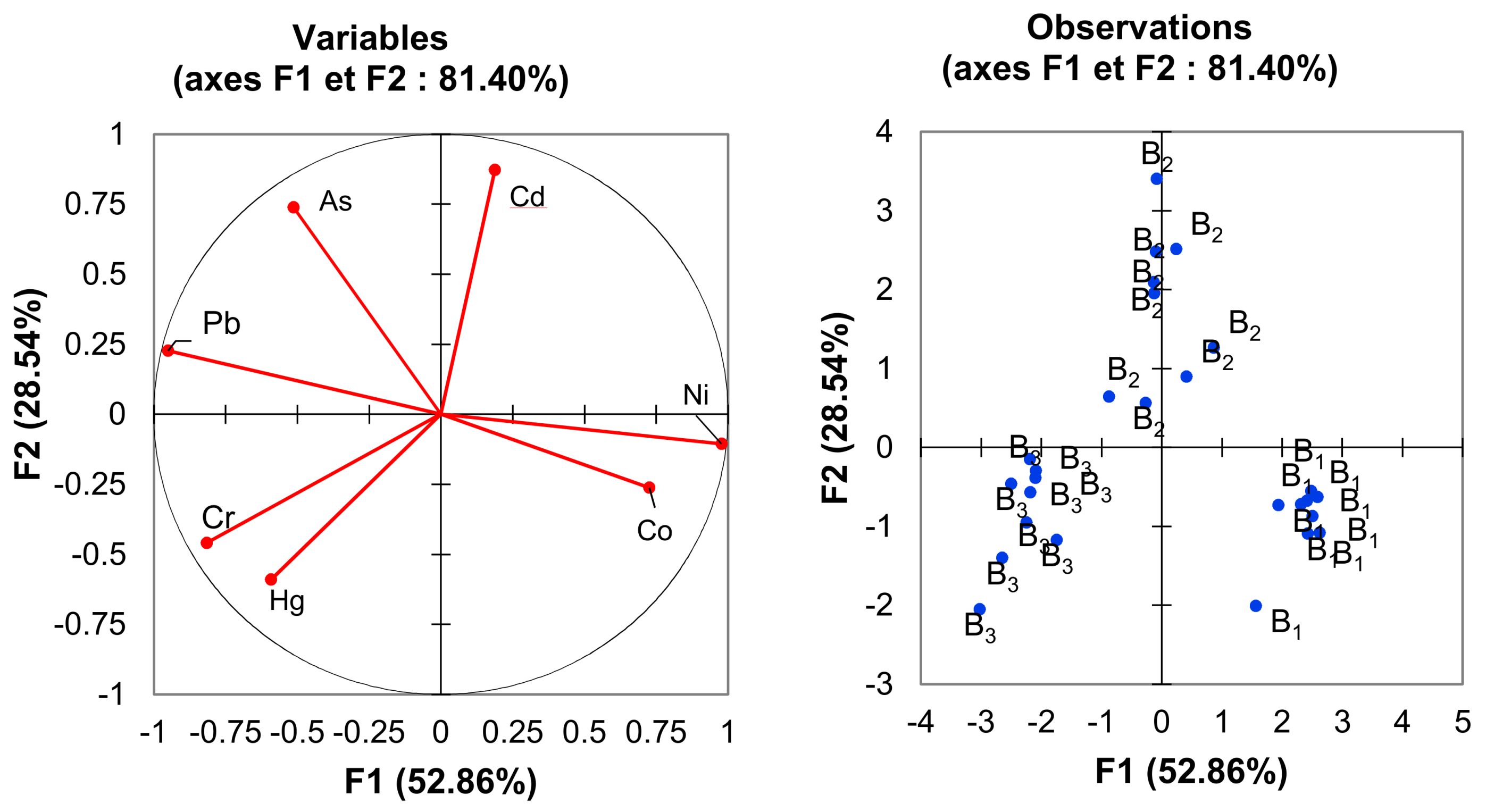
4.2. Risk Characterization
In terms of risk characterization linked to the seven trace elements analyzed in the pita bread, a highly consumed foodstuff, two of the trace elements, Cd and Hg, appeared to not be a safety concern. The results for Cd are in accordance with the Nasreddine [
13] study conducted on the total diet in an adult urban population. Bread may not be the main food involved in the highest exposure to Cd, as these authors showed that the main contributors to the dietary intake of Cd in a Beirut TDS were vegetables (46.8%) followed by breads and cereal-based products (30.9%). Same as for Cd, bread is not the main source of Hg, for which it is fish and seafood, which is a concern for high fish consumers [
17] and, particularly, pregnant women [
15]. We found between 0.70 and 0.89 µg/kg of Hg in Lebanese pita which is by far less than in the study of Mestek and co-workers [
44] who found 13 µg/kg of Hg in bread in the Czech Republic.
In our study, total chromium was analyzed. The B3 pita contained the highest level of Cr (0.36 mg/kg of dry weight). It is known that the major form of Cr in food is Cr (III), and the major contributor of Cr is bread (8%) followed by bakery products. For children, it is milk (9%) followed by pasta (6%) [
22]. In this study, the analytical data from the two brands (B1 and B2) were in accordance with Soares’ [
22] team who quantified the total chromium contents (47.3 ± 20.0 μg/kg) of dry weight white bread samples.
Compared to Cr (VI), Cr (III) is considered an essential element for humans and authorized in food supplements in Europe without any upper limit [
45]. But, in 2010, the EFSA [
46] recommended that total Cr does not exceed 250 µg/day, a value established by the WHO [
26]. Furthermore, Cr (III) can complex with lactate and picolinate, causing concern as it has been demonstrated to be genotoxic [
27]. In contrast, Cr (VI) is known to be highly toxic and carcinogenic (Group 1, [
47]). It also induces skin dermatitis, then the interconversion will be of relevance in terms of risk assessment.
Even if data on the presence of Cr (VI) in food are missing, it is considered that food is a large reducing medium and that oxidation of Cr (III) to Cr (VI) would not occur. Additionally, most of the Cr (VI) is considered to be reduced in the stomach to Cr (III) [
27].
Humans are exposed to Cr mainly via food, but it can be present also in water. Concerning bottled water in Lebanese markets [
48], a study of the physicochemical and heavy metal parameters in 32 samples of bottled waters, 25 of these samples showed values below 1 µg/L for Cr, while the other samples showed values ranging between 1 and 4 μg/L in the drinking water. Although naturally occurring Cr (VI) is rarely found in the environment, its presence in ground and surface waters, as a consequence of oxidation of Cr (III)-bearing minerals by Mn (IV) oxides, has been reported recently [
49]. Furthermore, in terms of the total quantity found in bread, we do not know its respective part coming from the wheat flour or water used during the bread-making process. Wheat are among species that demonstrate the ability to bio-accumulate chromium the most, just after tomato [
5]. An average value of Cr below 0.05 mg/kg of dry weight was measured in wheat seeds in Swedish long-term soil-fertility experiments between 1967 and 2003 [
50].
The recipient might also be a source of contamination if Cr is released. Indeed, migration of chromium from steel material occurred in the past mostly from Asia (China) and was above the upper legal limits for food contact material [
51].
The EFSA panel recommended the generation of data using sensitive analytical methodologies which specifically measure the content of Cr (III) and Cr (VI) in food and drinking water in different EU Member States [
27]; the issue is the same for Lebanon. However, Cr speciation is difficult to conduct, especially in complex matrices. Chromium speciation depends on several factors such as pH, concentration, oxidation state, and complexation with natural components present in the matrix such as peptide [
49]. Soares and co-workers [
22] mentioned the presence of Cr (VI) in bread after extraction of Cr by alkaline solutions. However, Novotnik et al. [
38] demonstrated that the only Cr species in bread is Cr (III) by spiking with enriched isotopes of
53Cr (III) and
50Cr (VI), a more accurate quantification, and using HPLC-ICP-MS. Thus, we made a characterization in regards to Cr (III) and, in this way, there were no safety concerns in this study for Cr (III) in Lebanese pita bread.
For cobalt, the exposure of young people under 15 years old at the 95th percentile exceeded the TDI by a percentage of 131%. Bread and cereals are significant source of Co exposure (18.1%), with a daily intake of cobalt estimated to be around 11.4 µg/day according to the Nasreddine study [
13]. Indeed, in their study they found 6.36 µg/kg of cobalt in bread and cereals. In this study, the level of Co was 91 µg/kg for the B1 pita brand, which is 7 fold more than in Australian bread and 13.5 fold higher than the data from the Nasreddine study [
13] but 1.8 fold less than in Ethiopian bread [
52].
In Lebanon, imported flour samples used in Lebanese white pita presented values for cobalt between 2 ± 2 and 33 ± 7 µg/kg which is far less than in the pita. Cobalt contamination can also occur during bread preparation via the water used. The mean level of Co in tap water is approximately 9 µg/L (IRAL, personal communication). In 2013, the Environmental Working Group’s (EWG) mission detected Co in only one sample (6 analyzed) and found 1.01 µg/L [
53]. Furthermore, cobalt can also be released from food contact materials (FCMs) as it is used in ceramics [
54]; thus, we can wonder if cobalt migration can occur from recipients used for making bread in Lebanon.
Several forms of cobalt (II) (sulphate, dichloride) are classified as presumed human carcinogens via the inhalatory route [
55]. The IARC classified the soluble form of cobalt II in class 2B but as metal, Co was classified in 2A [
56]. Although Co (II) salts are able to induce genotoxicity in vitro and in vivo after oral or parenteral exposure; however, there is a lack of carcinogenesis studies in humans and animals following an oral route [
57]. The EFSA concluded that it cannot be excluded that cobalt could have a non-threshold toxicological effect [
58]. In France, ANSES made the recommendation to reduce food exposure to cobalt in infants under 3 years old [
39]. Thus, for cobalt, the daily intake from all dietary sources may be quite pertinent to monitor in order to determine the total exposure of young people under 15 years old in Lebanon.
In regards to nickel, the B1 brand contained the highest value (1292 µg/kg), which was above values found in studies for Nigerian, Ethiopian, and Spanish bread [
52,
59,
60] but under those obtained in Iranian bread [
61]. However, for another brand the value (365 µg/kg) was in the same range.
The average daily intake of nickel was estimated at 126.27 µg/day bread and cereals contributing to a 54.73 µg/day Ni intake in the Lebanese adult urban population [
13].
In a French TDS study regarding the infant population, the value determined in bread was approximately 69.5 µg/kg [
39]. The contribution of drinking water to total exposure to nickel was very small across dietary surveys and age classes [
14]. We do not have an explanation, but this high level could be due to the recipient used by bakers. Using the TDI 2.8 µg/kg bw/day [
14] for oral chronic exposure, the data demonstrate that the percentage of Ni TDI was exceeded 3 to 4 fold for all populations with the 10–14 years old range being the most exposed. These data are in accordance with the TDS studies also showing a safety concern in France for very young people (<3 years old) [
39].
Nickel compounds and metallic nickel are classified as human carcinogens, respectively, group 1 and group 2B after inhalation [
62]. Available studies in animals suggest that exposure to nickel salts lead to renal effects and increases neonatal mortality with the kidney being the principal target. Reproductive and developmental toxicity were the critical effects after chronic oral exposure retained for risk characterization. However, oral exposure to nickel salts could be at the origin of contact dermatitis for sensitive populations. Indeed, oral nickel can also cause an oral and chronic effect in people dermally sensitized [
14].
In humans, absorption of Ni varies according to the method of exposure (in water it is 40 fold more absorbed compared to food; 0.7% and the bioavailability varies with the food type [
63]). There are no maximum levels in food for nickel; in contrast, a value of 20 μg/L is fixed in water intended for human consumption and in mineral waters in Europe [
64,
65].
In this study, arsenic exposure was more worrying, as we found a high level of arsenic for all brand analyzed (235–400 µg/kg) which is around 63 fold higher than those found in a bread study in the UK [
66] and 32 fold higher than those mentioned in a French infant TDS study [
39]. Bou Khouzam et al. [
1] mentioned seasonal variation in the arsenic concentration in bread (33 µg/kg in dry season, which is 4 fold higher than in wet season). Arsenic was not studied by Nasreddine et al. [
13] and calculated MOEs were very low for all bakeries and the population. These data are in accordance with the data and risk characterization in 19 countries in Europe [
16], for example, in France where there is a safety concern with MOEs lower than 36 [
39], suggesting that the situation in Lebanon is worrying.
The inorganic form of As, which is known to be toxic, may cause human malignancies. It was classified as carcinogenic (group 1) in Reference [
67]. The toxicity, due to the inorganic form, occurs by an epigenetic mechanism, and there is increasing evidence that early life exposure affecting fetal and infant growth [
68] may cause chronic disease later in life [
69]. This highlights the urgent need to obtain data on the effect of the toxicity of As on development [
39]. Arsenic passes through the placental barrier in humans [
70], resulting in similar levels in both the fetus and mother. Both the inorganic form and its methylated metabolites are transferred [
71]. The effect of As on the endocrine system has also been investigated for many years showing a disruption on the gonadal, adrenal, and thyroid endocrine systems [
72]. Furthermore, it is has been shown a toxic effect due to an interaction between Cd and As [
73,
74]. Drinking water, crops irrigated with contaminated water, and food prepared with contaminated water are sources of exposure [
75].
The EFSA’s recommendation is to reduce inorganic arsenic exposure in food and develop robust validated methods to determine the inorganic form [
16]. We do not distinguish between organic and inorganic form of As in this study. It is known that arsenic intake is higher from solid foods than from liquids.
Furthermore, speciation plays a major role in determining the amount of arsenic absorbed after consumption of As contaminated food as well as bioavailability with significant interspecies differences for organic arsenic. Regards to inorganic form, absorption can be high as demonstrated with contaminated rice [
76,
77].
Representative data on speciation are indeed scarce. The EFSA panel assumed a proportion of the contaminant to inorganic form (the toxic one) to vary between 50 (best case) to 100% (worst case) of the total arsenic reported in food commodities in Europe, other than fish and seafood [
16]. Furthermore, in bread, 92% of the inorganic form has been found [
78]. Cereals and cereal-based products were identified as largely contributing to the daily exposure to inorganic arsenic, as it was quantified as 53 µg up to 72 µg of arsenic per kilo for all cereals and cereal product categories. In wheat samples, it was demonstrated that 91% to 95% of As was in the inorganic form [
77,
78]. In wheat plants cultivated in the Bekaa region, the element As presented values between 288.3 ± 14.7 and 743.7 ± 25.4 mg/kg. In the irrigation water used in the Bekaa sites cultivated for wheat plants, the As content varied between 0.8 ± 0.3 μg/L and 1.2 ± 0.2 μg/L (IRAL data). Indeed, the level of As in drinking water in Lebanon varied between 1.2 and 4.5 µg/L [
79]. Water used for making bread can be the main contributor to the level of arsenic in the pita with the main form recovered in drinking water as inorganic. It is important to note than that change in the speciation of As can occur during food preparation [
80].
Concerning the level of lead in the different brands: B2 and B3 contents were more than 2.5 fold higher than B1 (74, 203, and 260 µg/kg, respectively). The order of magnitude of B1 is in accordance with Turkish, Iranian, Ethiopian, and Spanish studies on bread [
52,
59] and 7 fold more higher than in data on the Czech Republic [
44]. The B3 pita lead content was 32 fold higher than Lebanese data [
13] which focused on Beirut. The authors noticed that the lead exposure in Beirut was even two fold lower compared to a previous study [
43]. Thus, our data were not in accordance, but it took into account all the Lebanese regions including rural ones. The lead origin may be from wheat. Several samples of Lebanese wheat cultivated in the Bekaa region were studied, showing values of lead varying between 4.5 and 23 µg/kg but far less than in the bread, noting that the lead values in Lebanese wheat samples were lower than the values showing in wheat samples in Saudi Arabian markets (2.81 ± 0.08 mg/kg dry weight) [
12]. While, the lead levels, measured in five wheat flour samples in Lebanese markets varied between 0.0045 ± 0.002 and 0.023 ± 0.016 mg/kg dry weight, they were lower than values presented in wheat flour samples in the Canary Islands and used for bread production (varying between 0.037 ± 0.013 and 0.056 ± 0.045 mg/kg fresh weight) [
81].
Furthermore, lead can also be a contaminant issued from recipient food contact materials or water used for bread making.
Human data show that the developing nervous system is a critical target in children. Various water soluble and insoluble lead can induce tumors in rodents, especially renal tumors as a carcinogenic/promoter; brain gliomas were also observed. Classified lead compounds are probably carcinogenic to humans on the basis of limited evidence of carcinogenicity in humans and sufficient evidence in animals [
56].
It is important to note that due to the long lead half-life, chronic exposure is of most concern and children absorb lead after ingestion to a much greater extent (70% of the amount of lead ingested) than adults (20%) [
82].
All MOEs calculated with exposure only via pita consumption in this study were very low, but in the same range than in France and Europe [
17,
39,
41], suggesting an exposure safety concern and that it is necessary to decrease Pb exposure, for example, by varying the diet due to the difficulty in reducing Pb contamination.
For both lead and arsenic, this contamination occurred for all bread brands analyzed in this study and there is a safety concern (
Supplementary Materials Table S3).