Effect of Slaughter Age on Muscle Fiber Composition, Intramuscular Connective Tissue, and Tenderness of Goat Meat during Post-Mortem Time
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Samples
2.2. Histochemical Analyses
2.3. Meat Quality Measurement
2.4. Tenderness-Related Measurements
2.5. Histology Features of Perimysium
2.6. Statistical Analysis
3. Results and Discussion
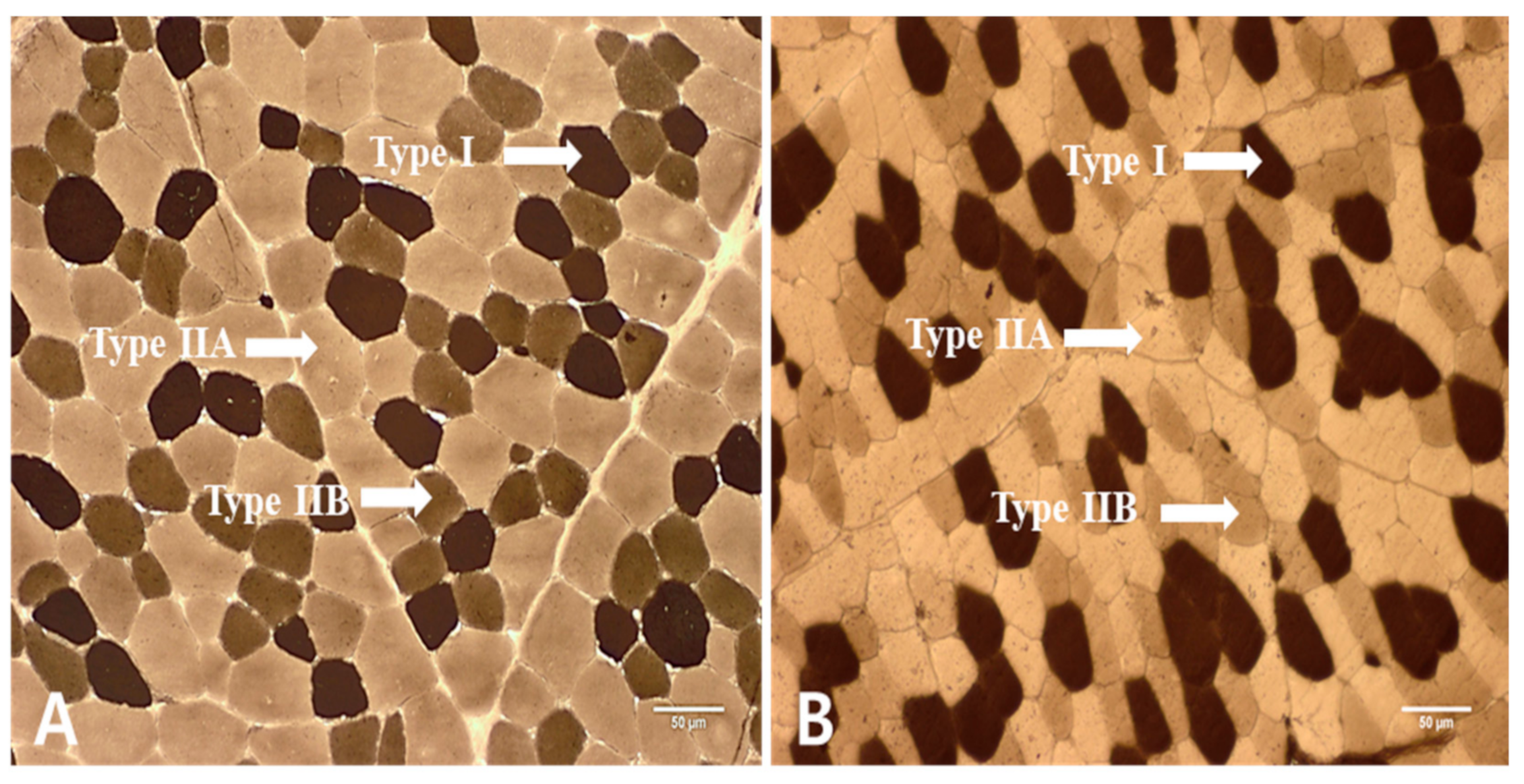
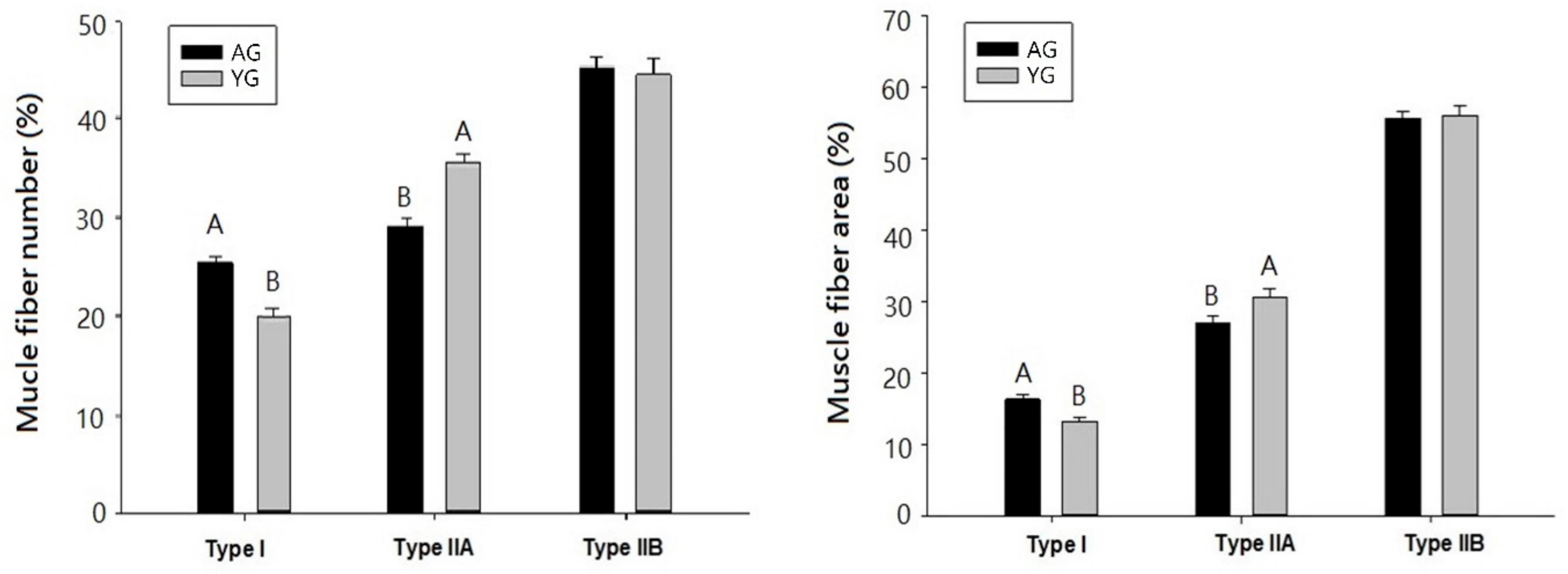
3.1. Histochemical Characteristics
3.2. Muscle pH, Meat Color, and WHC
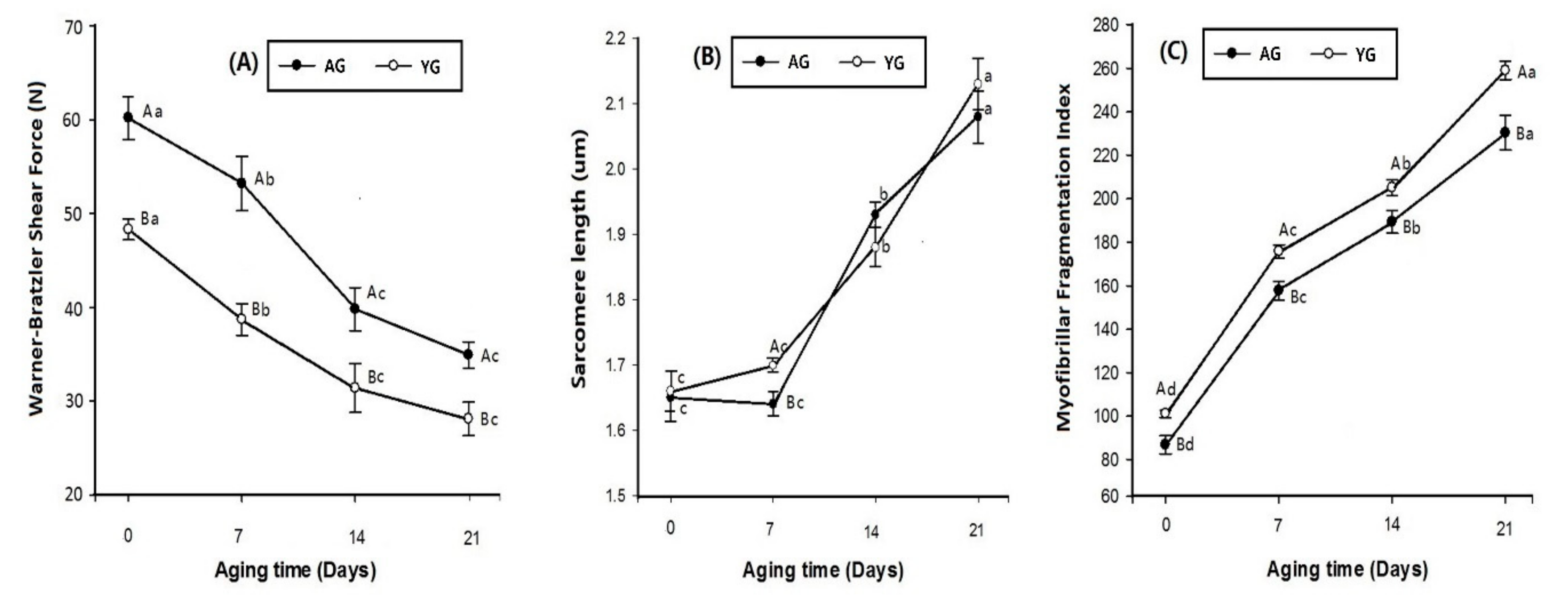
3.3. Tenderness
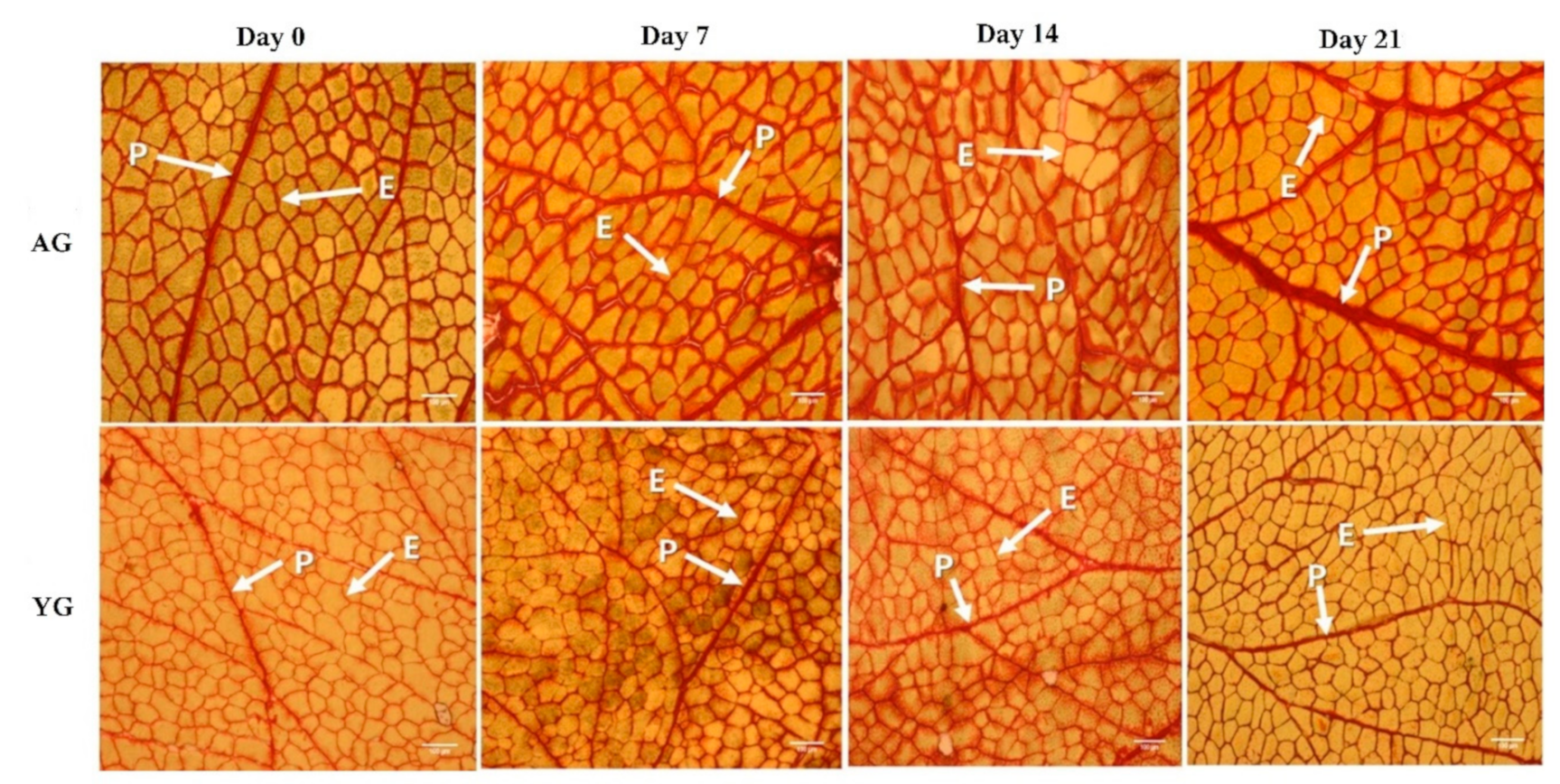
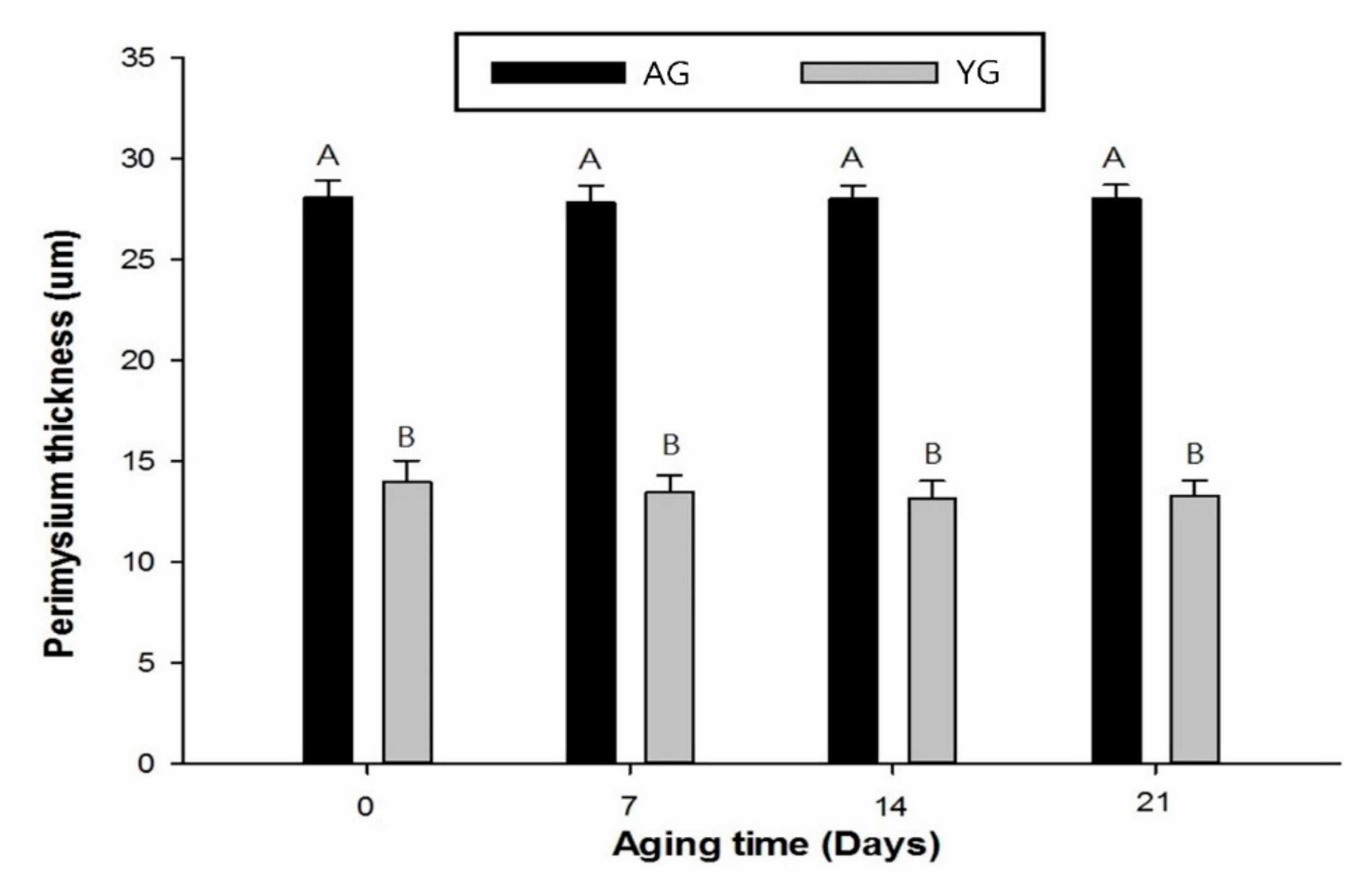
3.4. Perimysium Thickness
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Statistical Yearbook of Agriculture, Food and Rural Affairs; Ministry of Agriculture, Food and Rural Affairs: Sejong-si, Korea, 2018.
- Kim, J.O.; Kim, M.N.; Ha, Y.L. Processing of Korean black goat’s meat to remove goaty flavor. Food Sci. Biotechnol. 1993, 2, 26–29. [Google Scholar]
- Young, H.-T.; Kim, M.-W.; Choi, H.-J. Studies on the characterization of black goat meat and bone beverage containing honey with red ginseng. Korean J. Food Nutr. 2005, 18, 135–139. [Google Scholar]
- Mushi, D.; Safari, J.; Mtenga, L.; Kifaro, G.; Eik, L. Effects of concentrate levels on fattening performance, carcass and meat quality attributes of Small East African× Norwegian crossbred goats fed low quality grass hay. Livest. Sci. 2009, 124, 148–155. [Google Scholar] [CrossRef]
- Lee, J.-G.; Jeong, W.-C.; Joo, S.-T. Meat Quality Characteristics of the Four Major Muscles from Korean Native Black Goat. J. Agric. Life Sci. 2018, 52, 37–44. [Google Scholar] [CrossRef]
- Hwang, Y.-H.; Bakhsh, A.; Ismail, I.; Lee, J.-G.; Joo, S.-T. Effects of Intensive Alfalfa Feeding on Meat Quality and Fatty Acid Profile of Korean Native Black Goats. Korean J. Food Sci. Anim. Resour. 2018, 38, 1092. [Google Scholar] [CrossRef] [PubMed]
- Bakhsh, A.; Ismail, I.; Hwang, Y.-H.; Lee, J.-G.; Joo, S.-T. Comparison of Blood Loss and Meat Quality Characteristics in Korean Black Goat Subjected to Head-Only Electrical Stunning or without Stunning. Korean J. Food Sci. Anim. Resour. 2018, 38, 1286. [Google Scholar] [CrossRef] [PubMed]
- Bekhit, A.E.-D.A.; van de Ven, R.; Fahri, F.; Hopkins, D.L. Effect of pulsed electric field treatment on cold-boned muscles of different potential tenderness. Food Bioprocess Technol. 2014, 7, 3136–3146. [Google Scholar] [CrossRef]
- Sifre, L.; Berge, P.; Engel, E.; Martin, J.-F.; Bonny, J.-M.; Listrat, A.; Taylor, R.; Culioli, J. Influence of the spatial organization of the perimysium on beef tenderness. J. Agric. Food Chem. 2005, 53, 8390–8399. [Google Scholar] [CrossRef]
- Monsón, F.; Sañudo, C.; Sierra, I. Influence of breed and ageing time on the sensory meat quality and consumer acceptability in intensively reared beef. Meat Sci. 2005, 71, 471–479. [Google Scholar] [CrossRef]
- Farouk, M.; Mustafa, N.M.; Wu, G.; Krsinic, G. The “sponge effect” hypothesis: An alternative explanation of the improvement in the waterholding capacity of meat with ageing. Meat Sci. 2012, 90, 670–677. [Google Scholar] [CrossRef]
- Vergara, H.; Molina, A.; Gallego, L. Influence of sex and slaughter weight on carcass and meat quality in light and medium weight lambs produced in intensive systems. Meat Sci. 1999, 52, 221–226. [Google Scholar] [CrossRef]
- Purchas, R. Opportunities and challenges in meat production from sheep. Aust. J. Exp. 2007, 47, 1239–1243. [Google Scholar] [CrossRef]
- Astruc, T. Connective tissue: Structure, function, and influence on meat quality. Encyclopedia Meat Sci. 2014, 1, 321–328. [Google Scholar]
- Veiseth, E.; Shackelford, S.; Wheeler, T.; Koohmaraie, M. Comparison of myofibril fragmentation index from fresh and frozen pork and lamb longissimus. J. Anim. Sci. 2001, 79, 904–906. [Google Scholar] [CrossRef]
- De Huidobro, F.R.; Miguel, E.; Onega, E.; Blázquez, B. Changes in meat quality characteristics of bovine meat during the first 6 days post mortem. Meat Sci. 2003, 65, 1439–1446. [Google Scholar] [CrossRef]
- Hopkins, D.; Thompson, J. The relationship between post-mortem calcium concentration or pH and indicators of proteolysis in ovine muscle. Meat Sci. 2002, 61, 411–414. [Google Scholar] [CrossRef]
- Brooke, M.H.; Kaiser, K.K. Muscle fiber types: How many and what kind? Arch. Neurol. 1970, 23, 369–379. [Google Scholar] [CrossRef]
- AMSA. Meat Color Measurement Guidelines; American Meat Science Association: Champaign-Urbana, IL, USA, 2012. [Google Scholar]
- Hwang, Y.-H.; Kim, G.-D.; Jeong, J.-Y.; Hur, S.-J.; Joo, S.-T. The relationship between muscle fiber characteristics and meat quality traits of highly marbled Hanwoo (Korean native cattle) steers. Meat Sci. 2010, 86, 456–461. [Google Scholar] [CrossRef]
- Joo, S.-T. Determination of water-holding capacity of porcine musculature based on released water method using optimal load. Korean J. Food Sci. Anim. Resour. 2018, 38, 823. [Google Scholar]
- AMSA. Research Guidelines for Cookery; American Meat Science Association in Cooperation with National Live Stock and Meat Board: Chicago, IL, USA, 1995; p. 8. [Google Scholar]
- Cross, H.; West, R.; Dutson, T. Comparison of methods for measuring sarcomere length in beef semitendinosus muscle. Meat Sci. 1981, 5, 261–266. [Google Scholar] [CrossRef]
- Hopkins, D.; Littlefield, P.; Thompson, J. A research note on factors affecting the determination of myofibrillar fragmentation. Meat Sci. 2000, 56, 19–22. [Google Scholar] [CrossRef]
- Nakyinsige, K.; Sazili, A.; Zulkifli, I.; Goh, Y.; Bakar, F.A.; Sabow, A. Influence of gas stunning and halal slaughter (no stunning) on rabbits welfare indicators and meat quality. Meat Sci. 2014, 98, 701–708. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Flint, F.O.; Pickering, K. Demonstration of collagen in meat products by an improved picro-sirius red polarisation method. Analyst 1984, 109, 1505–1506. [Google Scholar] [CrossRef] [PubMed]
- Li, C.B.; Zhou, G.H.; Xu, X.L. Dynamical Changes of Beef Intramuscular Connective Tissue and Muscle Fiber during Heating and their Effects on Beef Shear Force. Food Bioprocess Technol. 2010, 3, 521–527. [Google Scholar] [CrossRef]
- Ozawa, S.; Mitsuhashi, T.; Mitsumoto, M.; Matsumoto, S.; Itoh, N.; Itagaki, K.; Kohno, Y.; Dohgo, T. The characteristics of muscle fiber types of longissimus thoracis muscle and their influences on the quantity and quality of meat from Japanese Black steers. Meat Sci. 2000, 54, 65–70. [Google Scholar] [CrossRef]
- Čandek-Potokar, M.; Žlender, B.; Lefaucheur, L.; Bonneau, M. Effects of age and/or weight at slaughter on longissimus dorsi muscle: Biochemical traits and sensory quality in pigs. Meat Sci. 1998, 48, 287–300. [Google Scholar] [CrossRef]
- Ryu, Y.; Choi, Y.; Lee, S.; Shin, H.; Choe, J.; Kim, J.; Hong, K.C.; Kim, B.-C. Comparing the histochemical characteristics and meat quality traits of different pig breeds. Meat Sci. 2008, 80, 363–369. [Google Scholar] [CrossRef]
- Picard, B.; Gagniere, H.; Geay, Y.; Hocquette, J.; Robelin, J. Study of the influence of age and weaning on the contractile and metabolic characteristics of bovine muscle. Reprod. Nutr. Dev. 1995, 35, 71–84. [Google Scholar] [CrossRef]
- Swatland, H.J. Structure and Development of Meat Animals; Prentice Hall, Inc.: Upper Saddle River, NJ, USA, 1984. [Google Scholar]
- Wegner, J.; Albrecht, E.; Fiedler, I.; Teuscher, F.; Papstein, H.-J.; Ender, K. Growth-and breed-related changes of muscle fiber characteristics in cattle. J. Anim. Sci. 2000, 78, 1485–1496. [Google Scholar] [CrossRef]
- Mach, N.; Bach, A.; Velarde, A.; Devant, M. Association between animal, transportation, slaughterhouse practices, and meat pH in beef. Meat Sci. 2008, 78, 232–238. [Google Scholar] [CrossRef] [PubMed]
- Marichal, A.; Castro, N.; Capote, J.; Zamorano, M.; Argüello, A. Effects of live weight at slaughter (6, 10 and 25 kg) on kid carcass and meat quality. Livest. Prod. Sci. 2003, 83, 247–256. [Google Scholar] [CrossRef]
- Karami, M.; Alimon, A.; Sazili, A.; Goh, Y.; Ivan, M. Effects of dietary antioxidants on the quality, fatty acid profile, and lipid oxidation of longissimus muscle in Kacang goat with aging time. Meat Sci. 2011, 88, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Xu, Q.; Dai, R.; Ni, Y. Effects of natural antioxidants on colour stability, lipid oxidation and metmyoglobin reducing activity in raw beef patties. Acta Sci. Pol. Technol. Aliment. 2015, 14, 37–44. [Google Scholar] [CrossRef]
- Ilavarasan, R.; Abraham, R.J.; Rao, V.A.; Pandiyan, V.; Babu, R.N.; Ruban, S.W. Effect of Age on Physico-Chemical and Nutritional Composition of Indigenous Kodiadu Goat Meat. J. Anim. Res. 2015, 5, 585. [Google Scholar] [CrossRef]
- De Palo, P.; Maggiolino, A.; Centoducati, P.; Tateo, A. Slaughtering age effect on carcass traits and meat quality of Italian heavy draught horse foals. Asian Aust. J. Anim. Sci. 2013, 26, 1637. [Google Scholar] [CrossRef]
- Kim, G.-D.; Jeong, J.-Y.; Jung, E.-Y.; Yang, H.-S.; Lim, H.-T.; Joo, S.-T. The influence of fiber size distribution of type IIB on carcass traits and meat quality in pigs. Meat Sci. 2013, 94, 267–273. [Google Scholar] [CrossRef]
- Prates, J.A.M.; e Costa, F.J.G.; Ribeiro, A.M.R.; Correia, A.A.D. Contribution of major structural changes in myofibrils to rabbit meat tenderisation during ageing. Meat Sci. 2002, 61, 103–113. [Google Scholar] [CrossRef]
- Ismail, I.; Hwang, Y.-H.; Joo, S.-T. Effect of Different Temperature and Time Combinations on Quality Characteristics of Sous-vide Cooked Goat Gluteus Medius and Biceps Femoris. Food Bioprocess Technol. 2019, 12, 1000–1009. [Google Scholar] [CrossRef]
- American Meat Science Association. Research Guidelines for Cookery, Sensory Evaluation, and Instrumental Tenderness Measurements of Meat; American Meat Science Association: Champaign, IL, USA, 2015. [Google Scholar]
- Shackelford, S.; Wheeler, T.; Koohmaraie, M. Tenderness classification of beef: II. Design and analysis of a system to measure beef longissimus shear force under commercial processing conditions. J. Anim. Sci. 1999, 77, 1474–1481. [Google Scholar] [CrossRef]
- Calkins, C.; Dutson, T.; Smith, G.; Carpenter, Z.; Davis, G. Relationship of fiber type composition to marbling and tenderness of bovine muscle. J. Food Sci. 1981, 46, 708–710. [Google Scholar] [CrossRef]
- Silva, L.H.; Assis, D.E.; Estrada, M.M.; Assis, G.J.; Zamudio, G.D.; Carneiro, G.B.; Valadares Filho, S.C.; Paulino, M.F.; Chizzotti, M.L. Carcass and meat quality traits of Nellore young bulls and steers throughout fattening. Livest. Sci. 2019, 229, 28–36. [Google Scholar] [CrossRef]
- Smulders, F.; Marsh, B.; Swartz, D.; Russell, R.; Hoenecke, M. Beef tenderness and sarcomere length. Meat Sci. 1990, 28, 349–363. [Google Scholar] [CrossRef]
- Pen, S. Effect of Automated Pre Rigor Stretching on Beef Tenderness Development; Auckland University of Technology: Auckland, New Zealand, 2012. [Google Scholar]
- Weaver, A.; Bowker, B.; Gerrard, D. Sarcomere length influences postmortem proteolysis of excised bovine semitendinosus muscle. J. Anim. Sci. 2008, 86, 1925–1932. [Google Scholar] [CrossRef] [PubMed]
- Wojtysiak, D. Effect of age on structural properties of intramuscular connective tissue, muscle fibre, collagen content and meat tenderness in pig longissimus lumborum muscle. Folia Biol. 2013, 61, 221–226. [Google Scholar] [CrossRef]
- Fang, S.-H.; Nishimura, T.; Takahashi, K. Relationship between development of intramuscular connective tissue and toughness of pork during growth of pigs. J. Anim. Sci. 1999, 77, 120–130. [Google Scholar] [CrossRef]
- Nishimura, T.; Hattori, A.; Takahashi, K. Structural changes in intramuscular connective tissue during the fattening of Japanese black cattle: Effect of marbling on beef tenderization. J. Anim. Sci. 1999, 77, 93–104. [Google Scholar] [CrossRef]
- Roy, B.C.; Walker, B.; Rahman, M.M.; Bruce, H.L.; McMullen, L. Role of myofibers, perimysium and adipocytes in horse meat toughness. Meat Sci. 2018, 146, 109–121. [Google Scholar] [CrossRef]
- An, J.; Zheng, J.; Li, J.; Zeng, D.; Qu, L.; Xu, G.; Yang, N. Effect of myofiber characteristics and thickness of perimysium and endomysium on meat tenderness of chickens. Poult. Sci. 2010, 89, 1750–1754. [Google Scholar] [CrossRef]
| Parameter | Age | Storage Days | |||
|---|---|---|---|---|---|
| 0 | 7 | 14 | 21 | ||
| pH | AG | 5.62 ± 0.02 | 5.63 ± 0.02 | 5.64 ± 0.03 | 5.67 ± 0.02 A |
| YG | 5.60 ± 0.01 | 5.59 ± 0.03 | 5.61 ± 0.02 | 5.58 ± 0.01 B | |
| CIE L* | AG | 42.18 ± 0.41 b | 44.56 ± 0.38 a | 44.28 ± 0.70 a | 45.28 ± 0.50 a |
| YG | 42.48 ± 0.50 b | 44.78 ± 0.56 a | 44.88 ± 0.36 a | 45.40 ± 0.27 a | |
| CIE a* | AG | 21.12 ± 0.16 Ab | 22.02 ± 0.12 Aa | 21.08 ± 0.20 b | 20.54 ± 0.71 c |
| YG | 20.70 ± 0.06 Bb | 21.44 ± 0.15 Ba | 20.62 ± 0.06 b | 20.40 ± 0.08 c | |
| CIE b* | AG | 5.22 ± 0.20 a | 4.31 ± 0.12 b | 4.36 ± 0.05 b | 4.47 ± 0.11 b |
| YG | 4.76 ± 0.20 a | 4.36 ± 0.20 b | 4.31 ± 0.21 b | 4.17 ± 0.13 b | |
| Cooking loss (%) | AG | 18.16 ± 0.47 | 17.96 ± 0.21 | 17.88 ± 0.62 | 17.78 ± 0.76 |
| YG | 18.40 ± 0.50 | 18.22 ± 0.16 | 17.70 ± 0.28 | 17.74 ± 0.38 | |
| Released water (%) | AG | 5.72 ± 0.13 Bb | 6.05 ± 0.19 Bab | 6.41 ± 0.20 Ba | 6.01 ± 0.14 Bab |
| YG | 6.50 ± 0.50 Ab | 6.99 ± 0.35 Aab | 7.37 ± 0.24 Aa | 7.20 ± 0.14 Aab | |
| Measurements | Fiber Number Percentage | Fiber Area Percentage | ||||
|---|---|---|---|---|---|---|
| Type I | Type IIA | Type IIB | Type I | Type IIA | Type IIB | |
| PMT | 0.56 | −0.40 | −0.63 | 0.80 ** | −0.82 ** | −0.87 ** |
| WBSF | −0.47 | −0.49 | 0.58 | 0.69* | −0.71 * | 0.76 * |
| SL | −0.15 | 0.09 | 0.15 | −0.04 | 0.15 | −0.02 |
| MFI | −0.73 * | 0.18 | 0.63 | −0.80 ** | 0.48 | 0.74 * |
| RW% | −0.37 | 0.41 | 0.47 | −0.59 | 0.67 * | 0.67 * |
| CL% | 0.19 | 0.32 | 0.29 | −0.06 | 0.01 | 0.03 |
| L* | 0.42 | 0.57 | −058 | 0.34 | −0.01 | 0.23 |
| a* | 0.66 * | −0.38 | −0.67 * | 0.73 * | −0.53 | −0.70 * |
| b* | −0.15 | −023 | 0.22 | 0.33 | −0.46 | 0.40 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bakhsh, A.; Hwang, Y.-H.; Joo, S.-T. Effect of Slaughter Age on Muscle Fiber Composition, Intramuscular Connective Tissue, and Tenderness of Goat Meat during Post-Mortem Time. Foods 2019, 8, 571. https://doi.org/10.3390/foods8110571
Bakhsh A, Hwang Y-H, Joo S-T. Effect of Slaughter Age on Muscle Fiber Composition, Intramuscular Connective Tissue, and Tenderness of Goat Meat during Post-Mortem Time. Foods. 2019; 8(11):571. https://doi.org/10.3390/foods8110571
Chicago/Turabian StyleBakhsh, Allah, Young-Hwa Hwang, and Seon-Tea Joo. 2019. "Effect of Slaughter Age on Muscle Fiber Composition, Intramuscular Connective Tissue, and Tenderness of Goat Meat during Post-Mortem Time" Foods 8, no. 11: 571. https://doi.org/10.3390/foods8110571
APA StyleBakhsh, A., Hwang, Y.-H., & Joo, S.-T. (2019). Effect of Slaughter Age on Muscle Fiber Composition, Intramuscular Connective Tissue, and Tenderness of Goat Meat during Post-Mortem Time. Foods, 8(11), 571. https://doi.org/10.3390/foods8110571






